Assessing the Toxicity of Lagocephalus sceleratus Pufferfish from the Southeastern Aegean Sea and the Relationship of Tetrodotoxin with Gonadal Hormones
Abstract
1. Introduction
2. Results and Discussion
2.1. Concentration Levels of TTX and Related Analogues
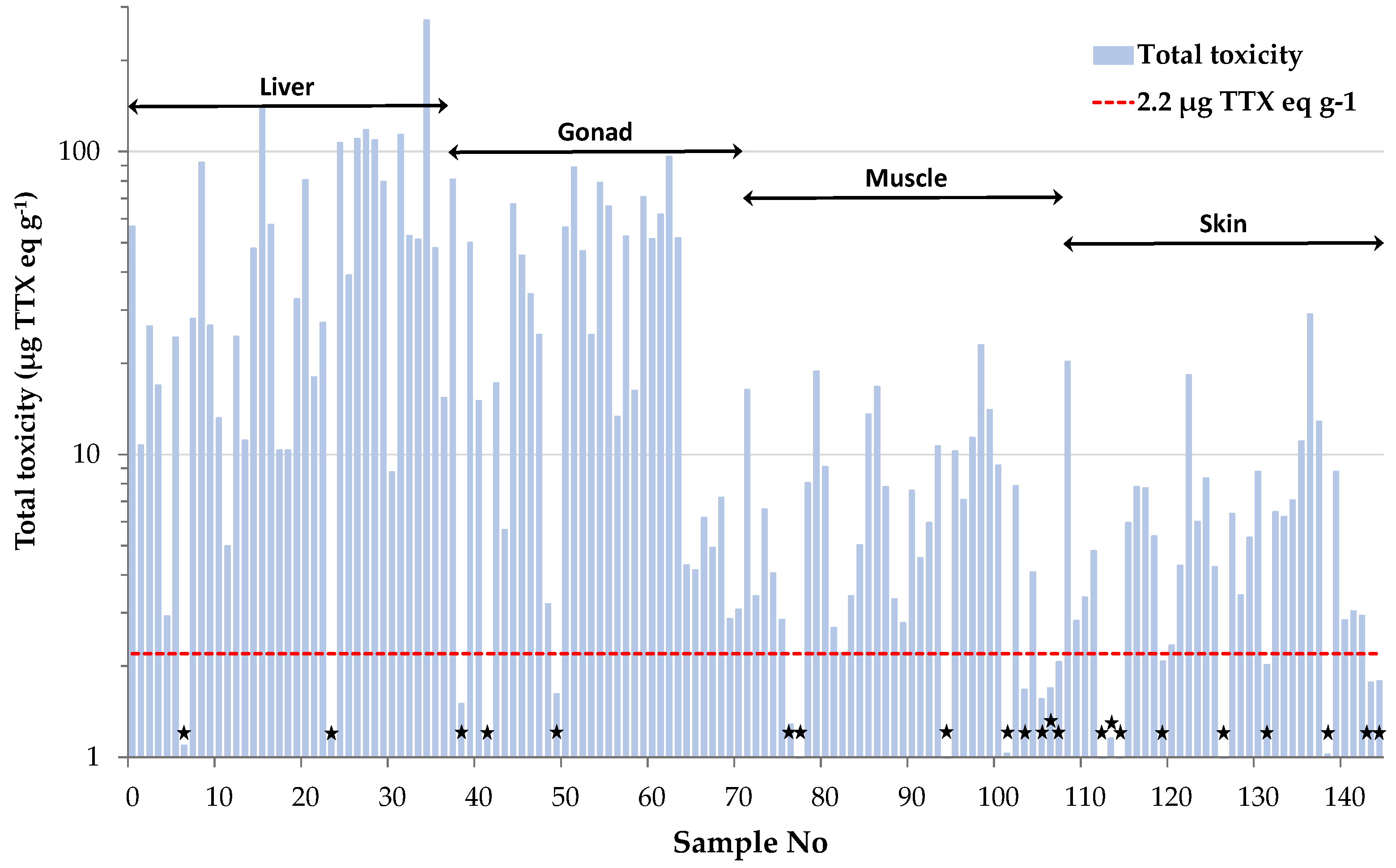
2.2. TTX-based Toxicity of L. sceleratus
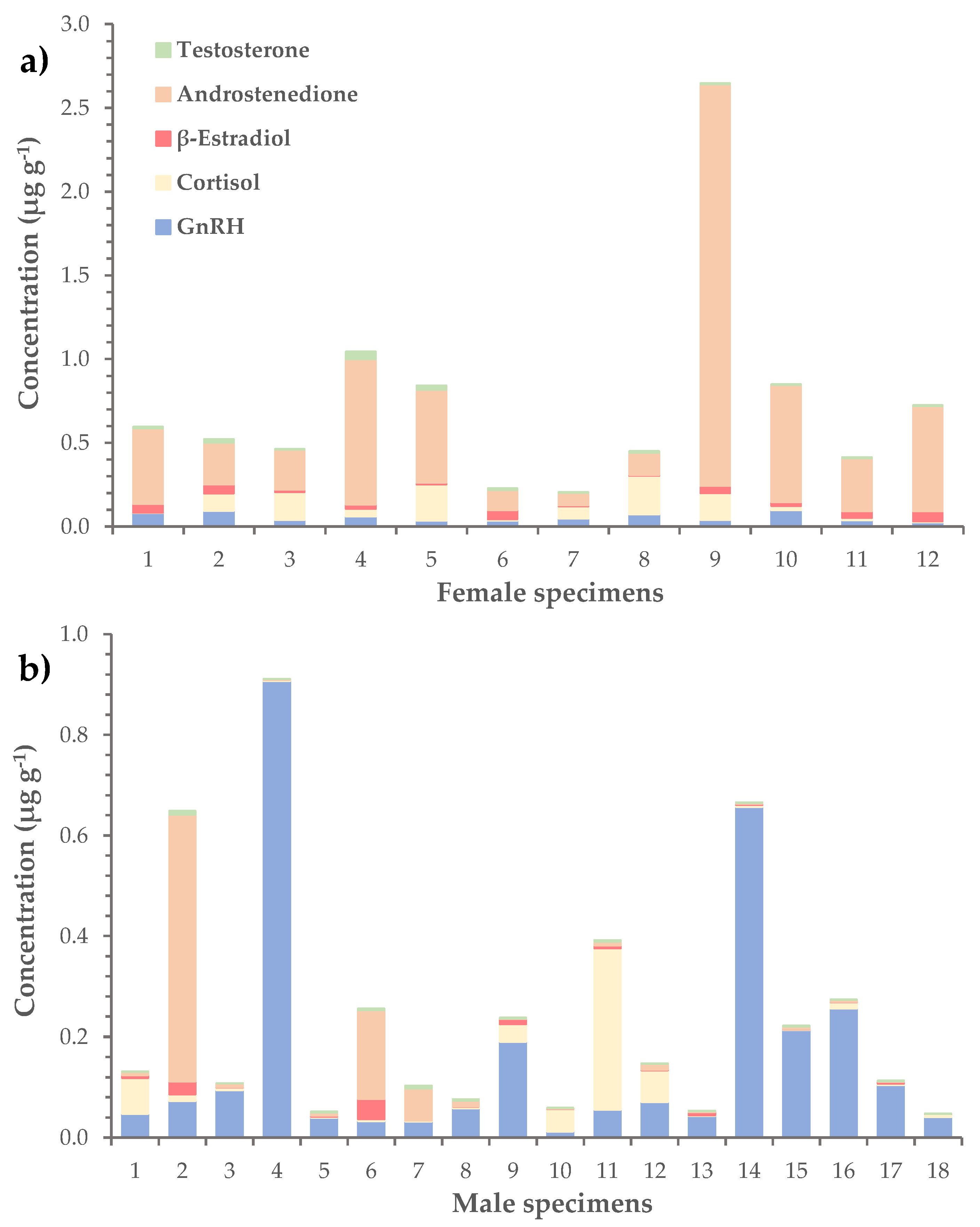
2.3. Hormones in L. sceleratus Gonads
3. Materials and Methods
3.1. TTX Analysis
3.2. Hormone Analysis
3.3. Quality Control and Assurance
3.4. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ulman, A.; Yildiz, T.; Demirel, N.; Canak, O.; Yemişken, E.; Pauly, D. The biology and ecology of the invasive silver-cheeked toadfish (Lagocephalus sceleratus), with emphasis on the Eastern Mediterranean. NeoBiota 2021, 68, 145–175. [Google Scholar] [CrossRef]
- Kasapidis, P.; Peristeraki, P.; Tserpes, G.; Magoulas, A. First record of the Lessepsian migrant Lagocephalus sceleratus (Gmelin 1789) (Osteichthyes: Tetraodontidae) in the Cretan Sea (Aegean, Greece). Aquat. Invasions 2007, 2, 71–73. [Google Scholar] [CrossRef]
- Kalogirou, S. Ecological characteristics of the invasive pufferfish Lagocephalus sceleratus (Gmelin, 1789) in Rhodes, Eastern Mediterranean Sea. A case study. Mediterr. Mar. Sci. 2013, 14, 251–260. [Google Scholar] [CrossRef]
- Ulman, A.; Kalogirou, S.; Pauly, D. The Dynamics of Maximum Lengths for the Invasive Silver-Cheeked Toadfish (Lagocephalus sceleratus) in the Eastern Mediterranean Sea. J. Mar. Sci. Eng. 2022, 10, 387. [Google Scholar] [CrossRef]
- Christidis, G.; Batziakas, E.; Peristeraki, P.; Kosoglou, I.; Tampakakis, K.; Somarakis, S.; Tserpes, G. The Impact of Lagocephalus sceleratus (Gmelin, 1789) on Small-Scale Fisheries in Crete: Preliminary Results. In Proceedings of the Marine and Inland Waters Research Symposium, Porto Heli, Argolida, Greece, 16–19 September 2022; pp. 357–361. [Google Scholar]
- Christidis, G.; Mandalakis, M.; Anastasiou, T.I.; Tserpes, G.; Peristeraki, P.; Somarakis, S. Keeping Lagocephalus sceleratus off the table: Sources of variation in the quantity of TTX, TTX analogues, and risk of tetrodotoxication. Toxins 2021, 13, 896. [Google Scholar] [CrossRef] [PubMed]
- Akbora, H.D.; Kunter, İ.; Erçetïn, T.; Elagöz, A.M.; Çïçek, B.A. Determination of tetrodotoxin (TTX) levels in various tissues of the silver cheeked puffer fish (Lagocephalus sceleratus (Gmelin, 1789)) in Northern Cyprus Sea (Eastern Mediterranean). Toxicon 2020, 175, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kosker, A.R.; Özogul, F.; Durmus, M.; Ucar, Y.; Ayas, D.; Regenstein, J.M.; Özogul, Y. Tetrodotoxin levels in pufferfish (Lagocephalus sceleratus) caught in the Northeastern Mediterranean Sea. Food Chem. 2016, 210, 332–337. [Google Scholar] [CrossRef]
- Bucciarelli, G.M.; Lechner, M.; Fontes, A.; Kats, L.B.; Eisthen, H.L.; Shaffer, H.B. From poison to promise: The evolution of tetrodotoxin and its potential as a therapeutic. Toxins 2021, 13, 517. [Google Scholar] [CrossRef]
- Hagen, N.A.; Cantin, L.; Constant, J.; Haller, T.; Blaise, G.; Ong-Lam, M.; Du Souich, P.; Korz, W.; Lapointe, B. Tetrodotoxin for Moderate to Severe Cancer-Related Pain: A Multicentre, Randomized, Double-Blind, Placebo-Controlled, Parallel-Design Trial. Pain Res. Manag. 2017, 7212713. [Google Scholar] [CrossRef]
- Zhan, C.; Wang, W.; Santamaria, C.; Wang, B.; Rwei, A.; Timko, B.P.; Kohane, D.S. Ultrasensitive phototriggered local anesthesia. Physiol. Behav. 2017, 176, 139–148. [Google Scholar] [CrossRef]
- Song, H.; Li, J.; Lu, C.L.; Kang, L.; Xie, L.; Zhang, Y.Y.; Zhou, X.B.; Zhong, S. Tetrodotoxin alleviates acute heroin withdrawal syndrome: A multicentre, randomized, double-blind, placebo-controlled study. Clin. Exp. Pharmacol. Physiol. 2011, 38, 510–514. [Google Scholar] [CrossRef] [PubMed]
- EXPLIAS. Design and Piloting Methods of Commercial Exploitation of Marine Alien Species Towards the Contribution to the Control of Their Population. Available online: https://explias.gr/ (accessed on 31 August 2023).
- Doğdu, S.A.; Turan, C.; Depci, T.; Ayas, D. Natural hydroxyapatite obtained from pufferfish teeth for potential dental application. J. Ceram. Process. Res. 2021, 22, 356–361. [Google Scholar] [CrossRef]
- Doğdu, S.A.; Turan, C.; Ayas, D. Isolation and Characterization of Collagen and Gelatin From Skin of Silver Cheeked Pufferfish Lagocephalus sceleratus for Pharmaceutical and Biomedical Applications. Nat. Eng. Sci. 2019, 4, 308–314. [Google Scholar] [CrossRef]
- Acar, C.; Ishizaki, S.; Nagashima, Y. Toxicity of the Lessepsian pufferfish Lagocephalus sceleratus from eastern Mediterranean coasts of Turkey and species identification by rapid PCR amplification. Eur. Food Res. Technol. 2017, 243, 49–57. [Google Scholar] [CrossRef]
- Tani, I. Toxicological Studies in Japanese Puffers. J. Toxicol. Sci. 1984, 9, 1–11. [Google Scholar]
- Kawabata, T. Pu er toxin. In The Manual for the Methods of Food Sanitation Tests; Bureau, E.H., Ed.; Japan Food Hygiene Association: Tokyo, Japan, 1978; p. 232. [Google Scholar]
- Antonelli, P.; Salerno, B.; Bordin, P.; Peruzzo, A.; Orsini, M.; Arcangeli, G.; Barco, L.; Losasso, C. Tetrodotoxin in live bivalve mollusks from Europe: Is it to be considered an emerging concern for food safety? Compr. Rev. Food Sci. Food Saf. 2021, 21, 719–737. [Google Scholar] [CrossRef]
- Vlamis, A.; Katikou, P.; Rodriguez, I.; Rey, V.; Alfonso, A.; Papazachariou, A.; Zacharaki, T.; Botana, A.M.; Botana, L.M. First detection of tetrodotoxin in greek shellfish by UPLC-MS/MS potentially linked to the presence of the dinoflagellate Prorocentrum minimum. Toxins 2015, 7, 1779–1807. [Google Scholar] [CrossRef]
- Knutsen, H.K.; Alexander, J.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Edler, L.; Grasl-Kraupp, B.; et al. Risks for public health related to the presence of tetrodotoxin (TTX) and TTX analogues in marine bivalves and gastropods. EFSA J. 2017, 15, 4752. [Google Scholar] [CrossRef]
- Itoi, S.; Yoshikawa, S.; Tatsuno, R.; Suzuki, M.; Asahina, K.; Yamamoto, S.; Takanashi, S.; Takatani, T.; Arakawa, O.; Sakakura, Y.; et al. Difference in the localization of tetrodotoxin between the female and male pufferfish Takifugu niphobles, during spawning. Toxicon 2012, 60, 1000–1004. [Google Scholar] [CrossRef]
- Ikeda, K.; Emoto, Y.; Tatsuno, R.; Wang, J.J.; Ngy, L.; Taniyama, S.; Takatani, T.; Arakawa, O. Maturation-associated changes in toxicity of the pufferfish Takifugu poecilonotus. Toxicon 2010, 55, 289–297. [Google Scholar] [CrossRef]
- Gao, W.; Kanahara, Y.; Tatsuno, R.; Soyano, K.; Nishihara, G.N.; Urata, C.; Takatani, T.; Arakawa, O. Maturation-associated changes in internal distribution and intra-ovarian microdistribution of tetrodotoxin in the pufferfish Takifugu pardalis. Fish. Sci. 2018, 84, 723–732. [Google Scholar] [CrossRef]
- Itoi, S.; Yoshikawa, S.; Asahina, K.; Suzuki, M.; Ishizuka, K.; Takimoto, N.; Mitsuoka, R.; Yokoyama, N.; Detake, A.; Takayanagi, C.; et al. Larval pufferfish protected by maternal tetrodotoxin. Toxicon 2014, 78, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Okita, K.; Yamazaki, H.; Sakiyama, K.; Yamane, H.; Niina, S.; Takatani, T.; Arakawa, O.; Sakakura, Y. Puffer smells tetrodotoxin. Ichthyol. Res. 2013, 60, 386–389. [Google Scholar] [CrossRef]
- Stacey, N. Hormones, pheromones and reproductive behavior. Fish Physiol. Biochem. 2003, 28, 229–235. [Google Scholar] [CrossRef]
- Zhou, L.; Li, M.; Wang, D. Role of sex steroids in fish sex determination and differentiation as revealed by gene editing. Gen. Comp. Endocrinol. 2021, 313, 113893. [Google Scholar] [CrossRef]
- Oliveira, R.F.; Gonçalves, D.M. Hormones and social behaviour of Teleost fish. In Fish Behaviour, 1st ed.; Magnhagen, C., Braithwaite, V.A., Forsgren, E., Kapoor, B.G., Eds.; Science Publishers: Enfield, NH, USA, 2008; pp. 61–150. [Google Scholar] [CrossRef]
- Amano, M.; Amiya, N.; Takaoka, M.; Sato, H.; Takatani, T.; Arakawa, O.; Sakakura, Y. Tetrodotoxin functions as a stress relieving substance in Juvenile tiger puffer Takifugu rubripes. Toxicon 2019, 171, 54–61. [Google Scholar] [CrossRef]
- Rodríguez, P.; Alfonso, A.; Otero, P.; Katikou, P.; Georgantelis, D.; Botana, L.M. Liquid chromatography-mass spectrometry method to detect Tetrodotoxin and Its analogues in the puffer fish Lagocephalus sceleratus (Gmelin, 1789) from European waters. Food Chem. 2012, 132, 1103–1111. [Google Scholar] [CrossRef]
- Bane, V.; Brosnan, B.; Barnes, P.; Lehane, M.; Furey, A. High-resolution mass spectrometry analysis of tetrodotoxin (TTX) and its analogues in puffer fish and shellfish. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2016, 33, 1468–1489. [Google Scholar] [CrossRef]
- Katikou, P.; Georgantelis, D.; Sinouris, N.; Petsi, A.; Fotaras, T. First report on toxicity assessment of the Lessepsian migrant pufferfish Lagocephalus sceleratus (Gmelin, 1789) from European waters (Aegean Sea, Greece). Toxicon 2009, 54, 50–55. [Google Scholar] [CrossRef]
- Kosker, A.R.; Özogul, F.; Ayas, D.; Durmus, M.; Ucar, Y.; Regenstein, J.M.; Özogul, Y. Tetrodotoxin levels of three pufferfish species (Lagocephalus sp.) caught in the North-Eastern Mediterranean sea. Chemosphere 2019, 219, 95–99. [Google Scholar] [CrossRef]
- Matsumura, K. Tetrodotoxin as a pheromone. Nature 1995, 378, 563–564. [Google Scholar] [CrossRef] [PubMed]
- Sabrah, M.M.; El-Ganainy, A.A.; Zaky, M.A. Biology and toxicity of the pufferfish Lagocephalus sceleratus (Gmelin, 1789) from the gulf of Suez. Egypt. J. Aquat. Res. 2006, 32, 283–297. [Google Scholar]
- Wang, J.; Araki, T.; Tatsuno, R.; Nina, S.; Ikeda, K.; Takatani, T.; Arakawa, O. Transfer profile of orally and intramuscularly administered tetrodotoxin to artificial hybrid specimens of the pufferfish Takifugu rubripes and Takifugu porphyreus. J. Food Hyg. Soc. Jpn. 2012, 53, 33–38. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ikeda, K.; Murakami, Y.; Emoto, Y.; Ngy, L.; Taniyama, S.; Yagi, M.; Takatani, T.; Arakawa, O. Transfer profile of intramuscularly administered tetrodotoxin to non-toxic cultured specimens of the pufferfish Takifugu rubripes. Toxicon 2009, 53, 99–103. [Google Scholar] [CrossRef]
- Matsumoto, T.; Nagashima, Y.; Kusuhara, H.; Ishizaki, S.; Shimakura, K.; Shiomi, K. Evaluation of hepatic uptake clearance of tetrodotoxin in the puffer fish Takifugu rubripes. Toxicon 2008, 52, 369–374. [Google Scholar] [CrossRef]
- Nagashima, Y.; Toyoda, M.; Hasobe, M.; Shimakura, K.; Shiomi, K. In vitro accumulation of tetrodotoxin in pufferfish liver tissue slices. Toxicon 2003, 41, 569–574. [Google Scholar] [CrossRef]
- Noguchi, T.; Arakawa, O. Tetrodotoxin—Distribution and accumulation in aquatic organisms, and cases of human intoxication. Mar. Drugs 2008, 6, 220–242. [Google Scholar] [CrossRef]
- Zhang, X.; Zong, J.; Chen, S.; Li, M.; Lu, Y.; Wang, R.; Xu, H. Accumulation and elimination of tetrodotoxin in the pufferfish Takifugu obscurus by dietary administration of the wild toxic gastropod Nassarius semiplicata. Toxins 2020, 12, 278. [Google Scholar] [CrossRef]
- Itoi, S.; Ueda, H.; Yamada, R.; Takei, M.; Sato, T.; Oshikiri, S.; Wajima, Y.; Ogata, R.; Oyama, H.; Shitto, T.; et al. Including planocerid flatworms in the diet effectively toxifies the pufferfish, Takifugu niphobles. Sci. Rep. 2018, 8, 12302. [Google Scholar] [CrossRef]
- Noguchi, T.; Arakawa, O.; Takatani, T. Toxicity of pufferfish Takifugu rubripes cultured in netcages at sea or aquaria on land. Comp. Biochem. Physiol. Part D Genom. Proteom. 2006, 1, 153–157. [Google Scholar] [CrossRef]
- Honda, S.; Arakawa, O.; Takatani, T.; Tachibana, K.; Yagi, M.; Tanigawa, A.; Noguchi, T. Toxification of cultured pufferfish Takifugu rubripes by feeding on tetrodotoxin-containing diet. Nippon Suisan Gakkaishi 2005, 71, 815–820. [Google Scholar] [CrossRef]
- Turner, A.D.; Dhanji-Rapkova, M.; Coates, L.; Bickerstaff, L.; Milligan, S.; O’Neill, A.; Faulkner, D.; McEneny, H.; Baker-Austin, C.; Lees, D.N.; et al. Detection of Tetrodotoxin Shellfish Poisoning (TSP) toxins and causative factors in bivalve molluscs from the UK. Mar. Drugs 2017, 15, 277. [Google Scholar] [CrossRef] [PubMed]
- Pratheepa, V.; Vasconcelos, V. Microbial diversity associated with tetrodotoxin production in marine organisms. Environ. Toxicol. Pharmacol. 2013, 36, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Auawithoothij, W.; Noomhorm, A. Shewanella putrefaciens, a major microbial species related to tetrodotoxin (TTX)-accumulation of puffer fish Lagocephalus lunaris. J. Appl. Microbiol. 2012, 113, 459–465. [Google Scholar] [CrossRef]
- Natural Toxin Risk Profile: Fish: Pufferfish Toxin. Available online: https://www.mhlw.go.jp/topics/syokuchu/poison/animal_det_01.html (accessed on 31 August 2023).
- Natural Toxin Risk Profile: Fish: Pufferfish Toxin (Summary Version). Available online: https://www.mhlw.go.jp/topics/syokuchu/poison/animal_01.html#betsuhyo (accessed on 31 August 2023).
- Regulation (EC) No 853/2004 of the European Parliament and of the Council of 29 April 2004 Laying Down Specific Hygiene Rules for Food of Animal Origin. Available online: https://eur-lex.europa.eu/legal-content/en/ALL/?uri=CELEX%3A32004R0853 (accessed on 20 September 2023).
- Commission Implementing Regulation (EU) 2019/627 of 15 March 2019 Laying Down Uniform Practical Arrangements for the Performance of Official Controls on Products of Animal Origin Intended for Human Consumption in Accordance with Regulation (EU) 2017/625 of the European Parliament and of the Council and Amending Commission Regulation (EC) No 2074/2005 as Regards Official Controls. Available online: https://eur-lex.europa.eu/eli/reg_impl/2019/627/oj#:~:text=Commission (accessed on 20 September 2023).
- Bordin, P.; Dall’Ara, S.; Tartaglione, L.; Antonelli, P.; Calfapietra, A.; Varriale, F.; Guiatti, D.; Milandri, A.; Dell’Aversano, C.; Arcangeli, G.; et al. First occurrence of tetrodotoxins in bivalve mollusks from Northern Adriatic Sea (Italy). Food Control 2021, 120, 107510. [Google Scholar] [CrossRef]
- Dell’Aversano, C.; Tartaglione, L.; Polito, G.; Dean, K.; Giacobbe, M.; Casabianca, S.; Capellacci, S.; Penna, A.; Turner, A.D. First detection of tetrodotoxin and high levels of paralytic shellfish poisoning toxins in shellfish from Sicily (Italy) by three different analytical methods. Chemosphere 2019, 215, 881–892. [Google Scholar] [CrossRef]
- Sorensen, P.W.; Pinillos, M.; Scott, A.P. Sexually mature male goldfish release large quantities of androstenedione into the water where it functions as a pheromone. Gen. Comp. Endocrinol. 2005, 140, 164–175. [Google Scholar] [CrossRef]
- Poling, K.R.; Fraser, E.J.; Sorensen, P.W. The three steroidal components of the goldfish preovulatory pheromone signal evoke different behaviors in males. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2001, 129, 645–651. [Google Scholar] [CrossRef]
- Lee, K.H.; Yamaguchi, A.; Rashid, H.; Kadomura, K.; Yasumoto, S.; Matsuyama, M. Estradiol-17beta treatment induces intersexual gonadal development in the pufferfish, Takifugu rubripes. Zoolog. Sci. 2009, 26, 639–645. [Google Scholar] [CrossRef]
- Shahjahan, M.; Hamabata, T.; Motohashi, E.; Doi, H.; Ando, H. Differential expression of three types of gonadotropin-releasing hormone genes during the spawning season in grass puffer, Takifugu niphobles. Gen. Comp. Endocrinol. 2010, 167, 153–163. [Google Scholar] [CrossRef]
- Papadaki, M.; Mandalakis, M.; Anastasiou, T.I.; Pouli, M.; Asderis, M.; Katharios, P.; Papandroulakis, N.; Mylonas, C.C. Histological Evaluation of Sex Differentiation and Early Sex Identification in Hatchery-Produced Greater Amberjack (Seriola dumerili) Reared in Sea Cages. Fish Physiol. Biochem. 2021, 47, 1777–1792. [Google Scholar] [CrossRef] [PubMed]
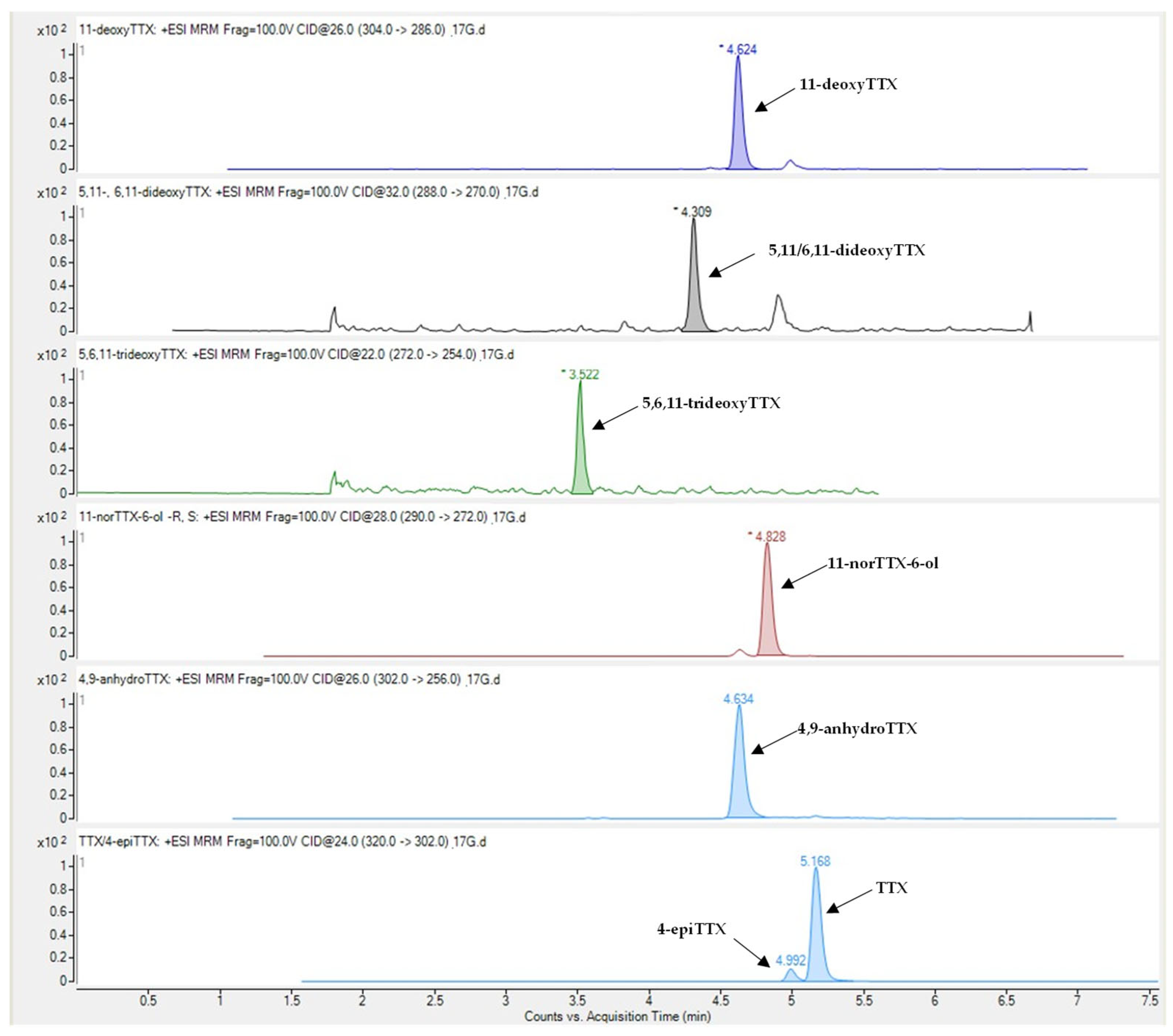
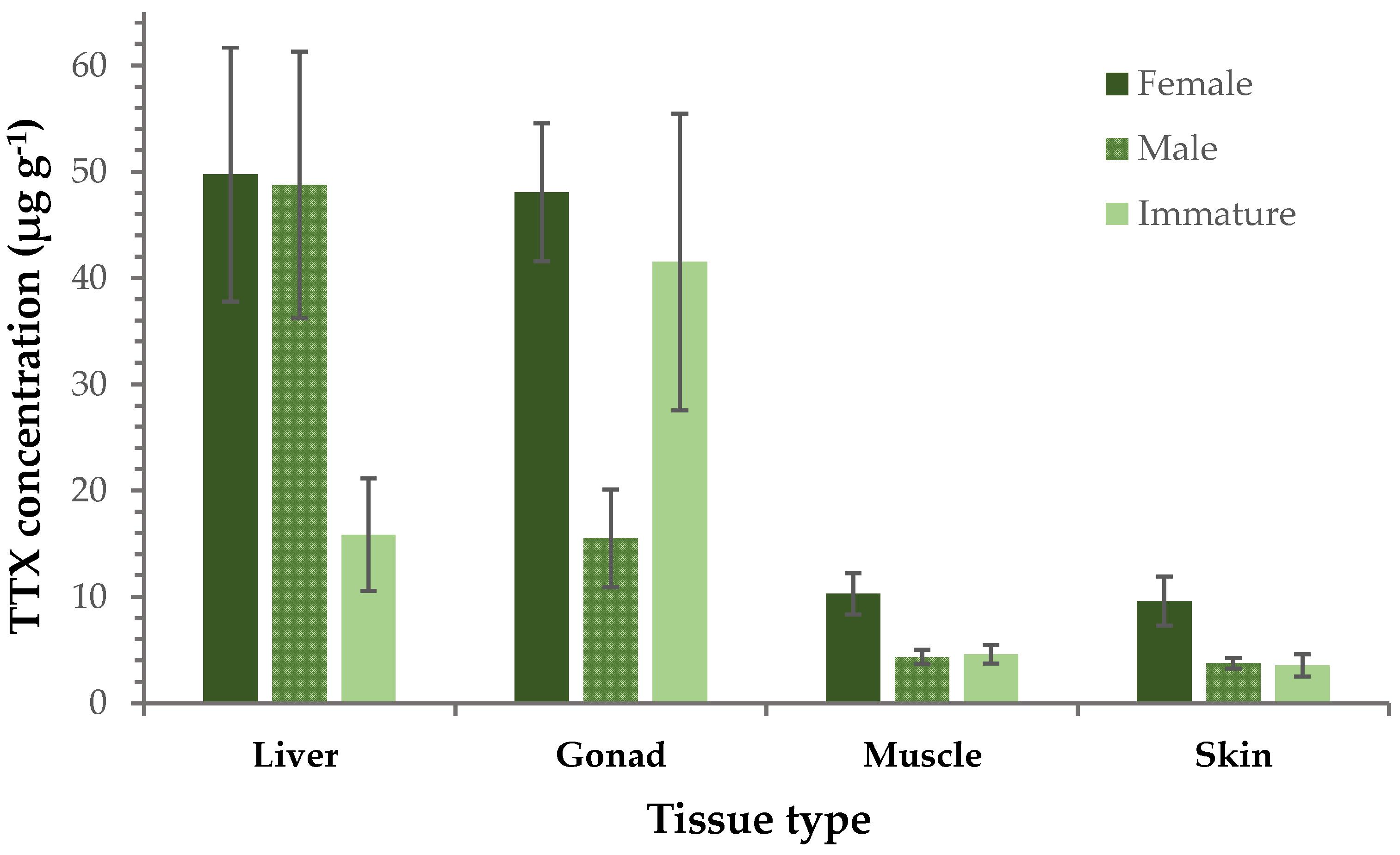
| Liver | Gonad | Muscle | Skin | Detection Rate | |||||
|---|---|---|---|---|---|---|---|---|---|
| Analyte | Average | Range | Average | Range | Average | Range | Average | Range | |
| TTX | 43.7 ± 7.7 | 1.1–239.8 | 30.1 ± 4.5 | 1.2–85.2 | 6.3 ± 0.9 | 0.2–21.7 | 5.6 ± 0.9 | 0.3–27.5 | 100% |
| 4-epiTTX | 5.4 ± 1.6 | 0.08–57.8 | 2.4 ± 0.5 | 0.02–11.1 | 0.3 ± 0.05 | 0.01–1.3 | 0.3 ± 0.07 | 0.04–2.4 | 100% |
| 11-norTTX-6-ol | 31.8 ± 5.6 | 0.07–115.6 | 18.2 ± 3.6 | 0.14–69.6 | 2.3 ± 0.4 | 0.02–9.9 | 2.3 ± 0.4 | 0.1–8.3 | 100% |
| 4,9-anhydroTTX | 3.7 ± 0.7 | n.d.–24.1 | 2.1 ± 0.4 | n.d.–9.1 | 0.2 ± 0.03 | n.d.–0.8 | 0.2 ± 0.04 | n.d.–1.2 | 93% |
| 11-deoxyTTX | 4.5 ± 0.8 | 0.01–20.1 | 1.6 ± 0.3 | 0.1–4.6 | 0.3 ± 0.04 | 0.01–1.0 | 0.3 ± 0.04 | 0.02–1.1 | 100% |
| 5,11/6,11-dideoxyTTX | 0.4 ± 0.1 | 0.002–2.1 | 0.2 ± 0.04 | 0.002–0.9 | 0.02 ± 0.004 | n.d.–0.1 | 0.04 ± 0.01 | 0.01–0.2 | 99% |
| Tissue Type | Total Toxicity |
|---|---|
| Liver | 51.03 ± 8.88 |
| Gonad | 33.99 ± 5.16 |
| Muscle | 6.81 ± 0.92 |
| Skin | 6.14 ± 0.97 |
| Hormone | Average | Range | Detection Rate |
|---|---|---|---|
| GnRH | 0.11 ± 0.03 | n.d.–0.91 | 97% |
| Cortisol | 0.06 ± 0.01 | 0.001–0.32 | 100% |
| β-Estradiol | 0.020 ± 0.004 | n.d.–0.06 | 76% |
| Androstenedione | 0.23 ± 0.08 | n.d.–2.40 | 97% |
| Testosterone | 0.010 ± 0.002 | 0.0003–0.05 | 100% |
| Hormone | Female | Male | Immature |
|---|---|---|---|
| GnRH | 0.05 ± 0.01 | 0.2 ± 0.1 | 0.05 ± 0.01 |
| Cortisol | 0.10 ± 0.02 | 0.03 ± 0.02 | 0.06 ± 0.03 |
| β-Estradiol | 0.03 ± 0.01 | 0.01 ± 0.002 | 0.03 ± 0.02 |
| Androstenedione | 0.6 ± 0.2 | 0.05 ± 0.03 | 0.06 ± 0.04 |
| Testosterone | 0.010 ± 0.004 | 0.002 ± 0.0005 | 0.002 ± 0.0005 |
| Weight | Length | Sex | TTX | 4-epi TTX | 11-nor TTX-6-ol | 4,9-anhydro TTX | 11-deoxy TTX | 5,11/6,11- dideoxyTTX | |
|---|---|---|---|---|---|---|---|---|---|
| Weight | − | 0.949 ** | −0.232 | −0.476 | −0.347 | −0.365 | −0.372 | −0.468 | −0.419 |
| Length | 0.949 ** | − | −0.246 | −0.388 | −0.253 | −0.266 | −0.276 | −0.374 | −0.348 |
| Sex | −0.232 | −0.246 | − | 0.624 ** | 0.658 ** | 0.570 ** | 0.688 ** | 0.631 ** | 0.769 ** |
| GnRH | 0.397 * | 0.374 * | −0.282 | −0.321 | −0.266 | −0.289 | −0.287 | −0.313 | −0.299 |
| Cortisol | −0.178 | −0.112 | 0.331 | 0.187 | 0.118 | 0.062 | 0.092 | 0.056 | 0.012 |
| β−Estradiol | −0.325 | −0.359 | 0.659 ** | 0.556 ** | 0.520 ** | 0.511 ** | 0.579 ** | 0.603 ** | 0.728 ** |
| Androstenedione | −0.207 | −0.265 | 0.539 ** | 0.132 | 0.11 | 0.163 | 0.145 | 0.193 | 0.212 |
| Testosterone | −0.007 | 0.026 | 0.563 ** | 0.173 | 0.083 | 0.051 | 0.139 | 0.182 | 0.281 |
| Model No | R | R2 | Standard Error of Estimate | F-Stat | p-Value | Variable | B a | β b | t-Test | p-Value | Part-R c |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 0.787 | 0.619 | 16.8 | 21.8 | p < 0.001 | Constant | 42.9 | - | 4.8 | <0.001 | - |
| Sex | 31.1 | 0.584 | 4.7 | <0.001 | 0.572 | ||||||
| Weight | −0.013 | −0.425 | −3.4 | 0.002 | −0.416 | ||||||
| 2 | 0.680 | 0.463 | 20.0 | 16.9 | p < 0.001 | Constant | 38.5 | - | 4.1 | <0.001 | - |
| β-Estradiol | 600.9 | 0.487 | 3.5 | 0.002 | 0.464 | ||||||
| Weight | −0.010 | −0.349 | −2.5 | 0.019 | −0.332 |
| Number of Specimens | Sex | Length | Weight |
|---|---|---|---|
| 12 | Female | 42–65 (53 ± 8) | 1030–3500 (1766 ± 761) |
| 19 | Male | 49–72 (56 ± 7) | 990–4300 (2138 ± 942) |
| 6 | Immature | 23–45 (37 ± 8) | 140–895 (568 ± 294) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anastasiou, T.I.; Kagiampaki, E.; Kondylatos, G.; Tselepides, A.; Peristeraki, P.; Mandalakis, M. Assessing the Toxicity of Lagocephalus sceleratus Pufferfish from the Southeastern Aegean Sea and the Relationship of Tetrodotoxin with Gonadal Hormones. Mar. Drugs 2023, 21, 520. https://doi.org/10.3390/md21100520
Anastasiou TI, Kagiampaki E, Kondylatos G, Tselepides A, Peristeraki P, Mandalakis M. Assessing the Toxicity of Lagocephalus sceleratus Pufferfish from the Southeastern Aegean Sea and the Relationship of Tetrodotoxin with Gonadal Hormones. Marine Drugs. 2023; 21(10):520. https://doi.org/10.3390/md21100520
Chicago/Turabian StyleAnastasiou, Thekla I., Eirini Kagiampaki, Gerasimos Kondylatos, Anastasios Tselepides, Panagiota Peristeraki, and Manolis Mandalakis. 2023. "Assessing the Toxicity of Lagocephalus sceleratus Pufferfish from the Southeastern Aegean Sea and the Relationship of Tetrodotoxin with Gonadal Hormones" Marine Drugs 21, no. 10: 520. https://doi.org/10.3390/md21100520
APA StyleAnastasiou, T. I., Kagiampaki, E., Kondylatos, G., Tselepides, A., Peristeraki, P., & Mandalakis, M. (2023). Assessing the Toxicity of Lagocephalus sceleratus Pufferfish from the Southeastern Aegean Sea and the Relationship of Tetrodotoxin with Gonadal Hormones. Marine Drugs, 21(10), 520. https://doi.org/10.3390/md21100520






