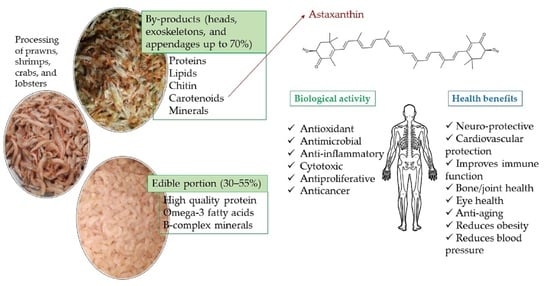
Astaxanthin from Crustaceans and Their Byproducts: A Bioactive Metabolite Candidate for Therapeutic Application
Abstract
1. Introduction
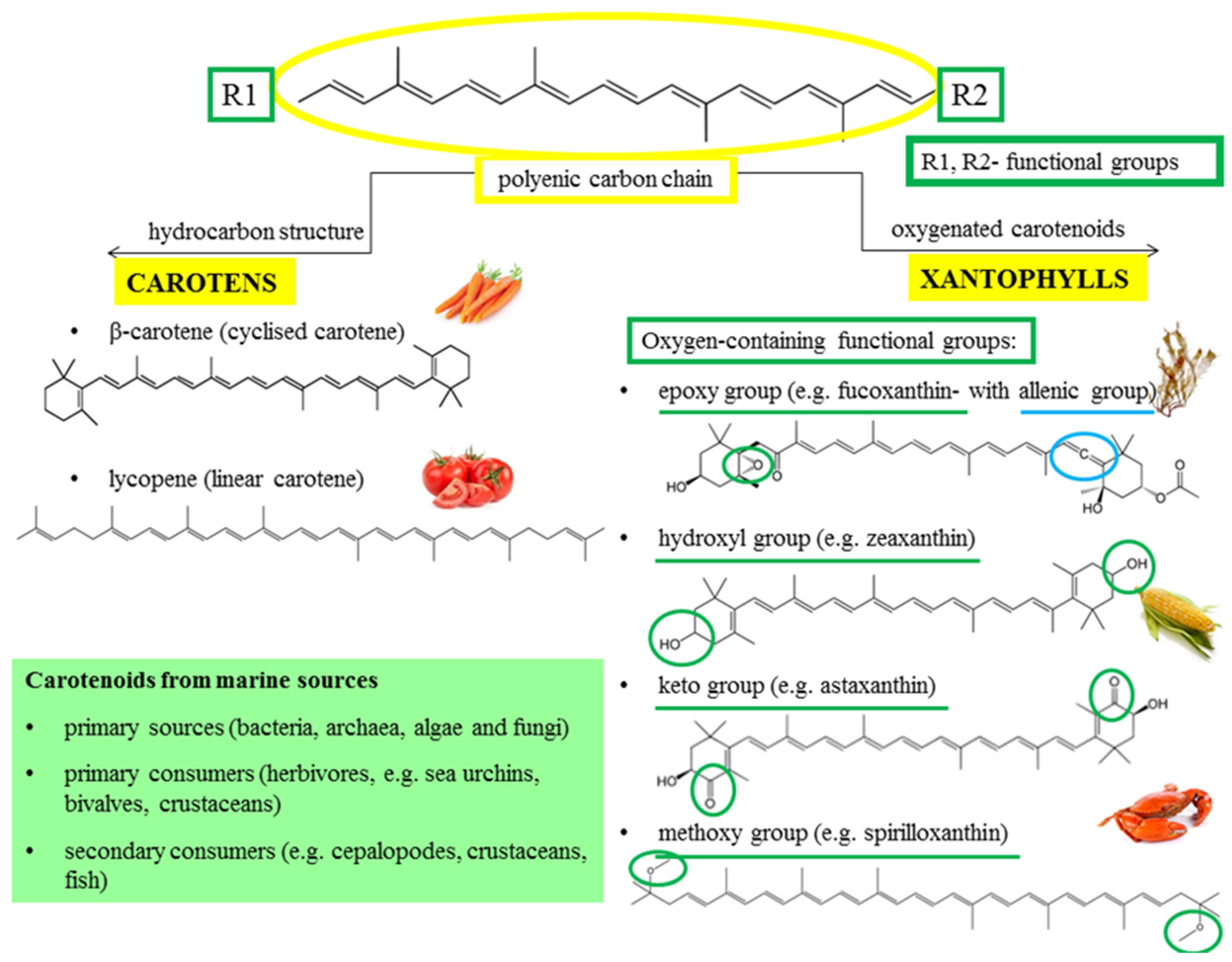
2. The Structure and Division of the Carotenoids from Marine Origin
2.1. Chemical Structure and Classes
- (a)
- carotenes—linear hydrocarbons that can be cyclized at the ends (e.g., β-carotene and lycopene), and
- (b)
2.2. Carotenoids from Marine Organisms
3. Carotenoids from Crustaceans
3.1. Astaxanthin from Crustaceans
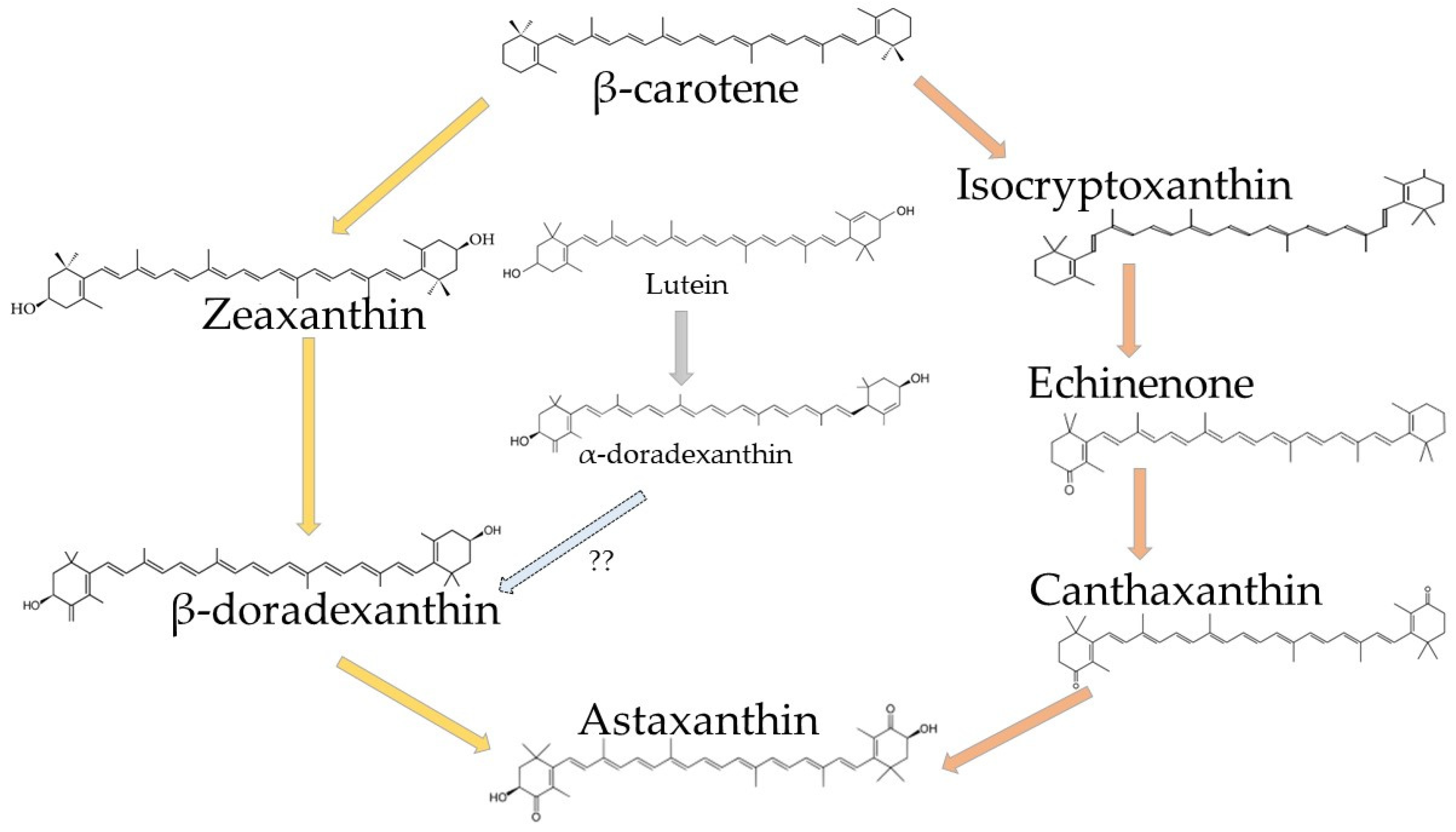
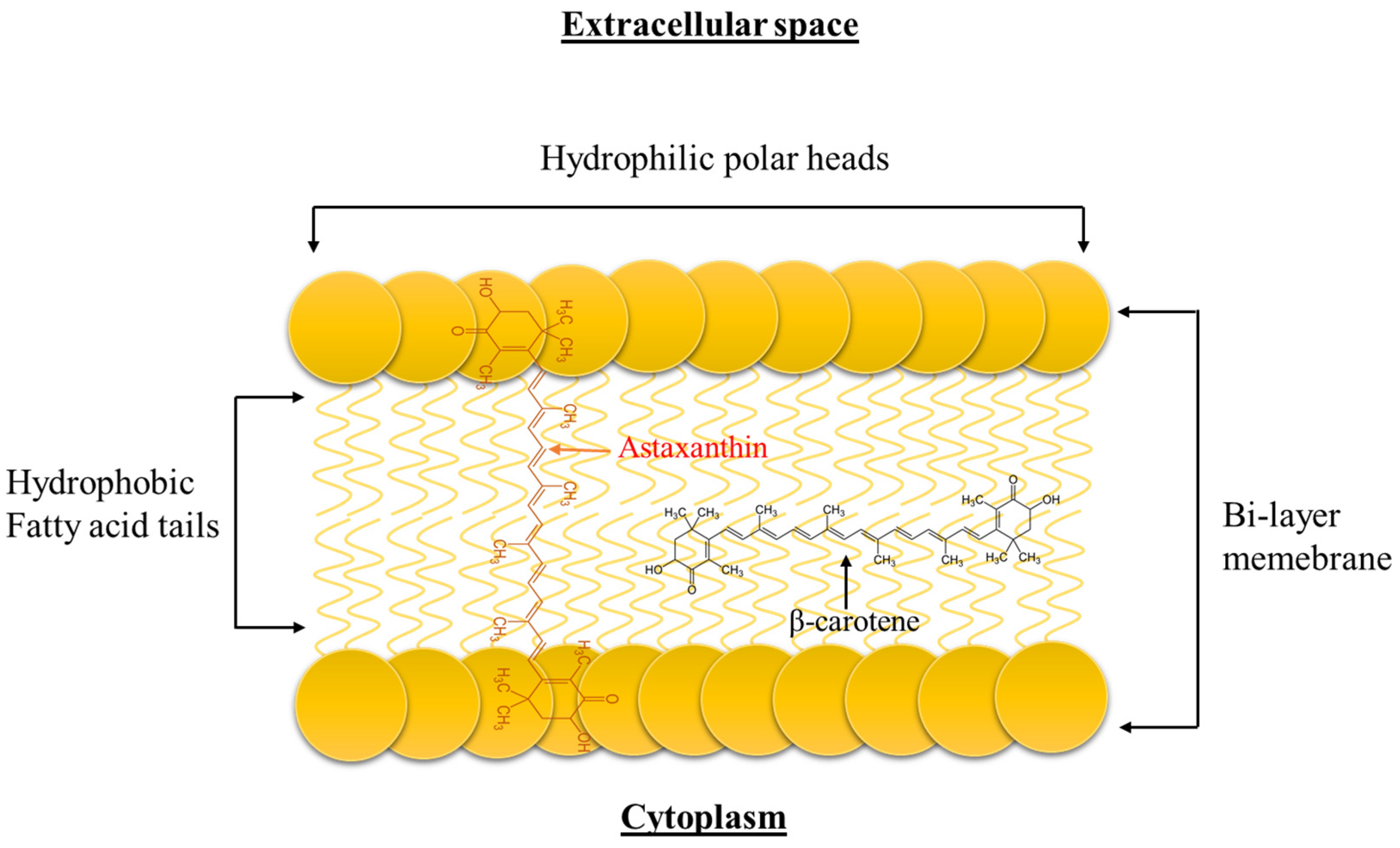
3.1.1. Structure and Biochemistry
3.1.2. Sources
3.1.3. Extraction
3.2. Biological Activity of Astaxanthin
3.3. Health Benefits, Therapeutic Application, and Safety of Astaxanthin Application in Humans
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture 2020; Sustainability in Action: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Özogul, F.; Hamed, I.; Özogul, Y.; Regenstein, J.M. Crustacean By-Products. In Encyclopedia of Food Chemistry; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 33–38. [Google Scholar] [CrossRef]
- Šimat, V. Valorization of Seafood Processing By-Products. In Valorization of Agri-Food Wastes and By-Products: Recent Trends, Innovations and Sustainability Challenges; Bhat, R., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2021; pp. 515–536. [Google Scholar]
- Nirmal, N.P.; Santivarangkna, C.; Rajput, M.S.; Benjakul, S. Trends in Shrimp Processing Waste Utilization: An Industrial Prospective. Trends Food Sci. Technol. 2020, 103, 20–35. [Google Scholar] [CrossRef]
- Visioli, F.; Artaria, C. Astaxanthin in Cardiovascular Health and Disease: Mechanisms of Action, Therapeutic Merits, and Knowledge Gaps. Food Funct. 2017, 8, 39–63. [Google Scholar] [CrossRef] [PubMed]
- Davinelli, S.; Nielsen, M.E.; Scapagnini, G. Astaxanthin in Skin Health, Repair, and Disease: A Comprehensive Review. Nutrients 2018, 10, 522. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, S.; Abbaszadeh, F.; Dargahi, L.; Jorjani, M. Astaxanthin: A Mechanistic Review on Its Biological Activities and Health Benefits. Pharmacol. Res. 2018, 136, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.P.M.; Souza, A.C.R.; Vasconcelos, A.R.; Prado, P.S.; Name, J.J. Antioxidant and Anti-Inflammatory Mechanisms of Action of Astaxanthin in Cardiovascular Diseases (Review). Int. J. Mol. Med. 2021, 47, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Raza, S.H.A.; Naqvi, S.R.Z.; Abdelnour, S.A.; Schreurs, N.; Mohammedsaleh, Z.M.; Khan, I.; Shater, A.F.; Abd El-Hack, M.E.; Khafaga, A.F.; Quan, G.; et al. Beneficial Effects and Health Benefits of Astaxanthin Molecules on Animal Production: A Review. Res. Vet. Sci. 2021, 138, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Donoso, A.; González-Durán, J.; Muñoz, A.A.; González, P.A.; Agurto-Muñoz, C. Therapeutic Uses of Natural Astaxanthin: An Evidence-Based Review Focused on Human Clinical Trials. Pharmacol. Res. 2021, 166, 105479. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumar, R.; Kumari, A.; Panwar, A.; Diksha. Astaxanthin: A Super Antioxidant from Microalgae and Its Therapeutic Potential. J. Basic Microbiol. 2021, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Mohd Hatta, F.A.; Othman, R. Carotenoids as Potential Biocolorants: A Case Study of Astaxanthin Recovered from Shrimp Waste. In Carotenoids: Properties, Processing and Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 289–325. [Google Scholar] [CrossRef]
- Lim, K.C.; Yusoff, F.M.; Shariff, M.; Kamarudin, M.S. Astaxanthin as Feed Supplement in Aquatic Animals. Rev. Aquac. 2018, 10, 738–773. [Google Scholar] [CrossRef]
- Mezquita, P.C.; Huerta, B.E.B.; Ramírez, J.C.P.; Hinojosa, C.P.O. Milks Pigmentation with Astaxanthin and Determination of Colour Stability during Short Period Cold Storage. J. Food Sci. Technol. 2015, 52, 1634–1641. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Grand View Research, Inc. Astaxanthin Market Size, Industry Report, 2021–2028. Available online: https://www.grandviewresearch.com/industry-analysis/global-astaxanthin-market (accessed on 13 February 2022).
- Villaró, S.; Ciardi, M.; Morillas-España, A.; Sánchez-Zurano, A.; Acién-Fernández, G.; Lafarga, T. Microalgae Derived Astaxanthin: Research and Consumer Trends and Industrial Use as Food. Foods 2021, 10, 2303. [Google Scholar] [CrossRef] [PubMed]
- Athanasios, B.; Zoumboulakis, P. Valuable Products from Algae Using New Magnetic Cultivation and Extraction Techniques; VALUEMAG project (Contract No. H2020-745695). Report on market penetration. 2020. [Google Scholar]
- Nguyen, K.D. Astaxanthin: A Comparative Case of Synthetic vs. Natural Production. Chem. Biomol. Eng. Publ. Other Work 2013, 1–9. [Google Scholar]
- Stachowiak, B.; Szulc, P. Astaxanthin for the Food Industry. Molecules 2021, 26, 2666. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Huang, B.; Liu, J. The Carotenoids of Shrimps (Decapoda: Caridea and Dendrobranchiata) Cultured in China. J. Crustac. Biol. 2018, 38, 523–530. [Google Scholar] [CrossRef]
- Mezzomo, N.; Ferreira, S.R.S. Carotenoids Functionality, Sources, and Processing by Supercritical Technology: A Review. J. Chem. 2016, 2016, 3164312. [Google Scholar] [CrossRef]
- Fernandes, A.S.; do Nascimento, T.C.; Jacob-Lopes, E.; De Rosso, V.V.; Zepka, L.Q. Introductory Chapter: Carotenoids—A Brief Overview on Its Structure, Biosynthesis, Synthesis, and Applications. Prog. Carotenoid Res. 2018, 1, 1–16. [Google Scholar] [CrossRef][Green Version]
- Nagarajan, J.; Ramanan, R.N.; Raghunandan, M.E.; Galanakis, C.M.; Krishnamurthy, N.P. Carotenoids; Elsevier Inc.: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Schieber, A.; Weber, F. Carotenoids. In Handbook on Natural Pigments in Food and Beverages: Industrial Applications for Improving Food Color; Woodhead Publishing: Cambridge, UK, 2016; pp. 101–123. [Google Scholar] [CrossRef]
- Rivera-Madrid, R.; Carballo-Uicab, V.M.; Cárdenas-Conejo, Y.; Aguilar-Espinosa, M.; Siva, R. Overview of Carotenoids and Beneficial Effects on Human Health. In Carotenoids: Properties, Processing and Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–40. [Google Scholar] [CrossRef]
- Yabuzaki, J. Carotenoids Database. Available online: http://carotenoiddb.jp/ (accessed on 13 February 2022).
- Viera, I.; Pérez-Gálvez, A.; Roca, M. Bioaccessibility of Marine Carotenoids. Mar. Drugs 2018, 16, 397. [Google Scholar] [CrossRef] [PubMed]
- Ngamwonglumlert, L.; Devahastin, S.; Food, A. Carotenoids. In Encyclopedia of Food Chemistry, 1st ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 40–52. [Google Scholar] [CrossRef]
- Kiokias, S.; Proestos, C.; Varzakas, T. A Review of the Structure, Biosynthesis, Absorption of Carotenoids-Analysis and Properties of Their Common Natural Extracts. Curr. Res. Nutr. Food Sci. J. 2016, 4, 25–37. [Google Scholar] [CrossRef]
- de Carvalho, C.C.C.R.; Caramujo, M.J. Carotenoids in Aquatic Ecosystems and Aquaculture: A Colorful Business with Implications for Human Health. Front. Mar. Sci. 2017, 4, 93. [Google Scholar] [CrossRef]
- Maoka, T. Carotenoids in Marine Animals. Mar. Drugs 2011, 9, 278–293. [Google Scholar] [CrossRef] [PubMed]
- Galasso, C.; Corinaldesi, C.; Sansone, C. Carotenoids from Marine Organisms: Biological Functions and Industrial Applications. Antioxidants 2017, 6, 96. [Google Scholar] [CrossRef] [PubMed]
- Miyashita, K.; Hosokawa, M. Health Impact of Marine Carotenoids. J. Food Bioact. 2018, 1, 31–40. [Google Scholar] [CrossRef]
- de Queiroz Antonino, R.; Lia Fook, B.; de Oliveira Lima, V.; de Farias Rached, R.; Lima, E.; da Silva Lima, R.; Peniche Covas, C.; Lia Fook, M. Preparation and Characterization of Chitosan Obtained from Shells of Shrimp (Litopenaeus Vannamei Boone). Mar. Drugs 2017, 15, 141. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.N.; Lee, P.P.; Chen, W.N. Microbial Extraction of Chitin from Seafood Waste Using Sugars Derived from Fruit Waste-Stream. AMB Express 2020, 10, 17. [Google Scholar] [CrossRef] [PubMed]
- Hülsey, M.J. Shell Biorefinery: A Comprehensive Introduction. Green Energy Environ. 2018, 3, 318–327. [Google Scholar] [CrossRef]
- Nekvapil, F.; Ganea, I.V.; Ciorîță, A.; Hirian, R.; Ogresta, L.; Glamuzina, B.; Roba, C.; Pinzaru, S.C. Wasted Biomaterials from Crustaceans as a Compliant Natural Product Regarding Microbiological, Antibacterial Properties and Heavy Metal Content for Reuse in Blue Bioeconomy: A Preliminary Study. Materials 2021, 14, 4558. [Google Scholar] [CrossRef] [PubMed]
- Arbia, W.; Arbia, L.; Adour, L.; Amrane, A. Chitin Extraction from Crustacean Shells Using Biological Methods—A Review. Food Technol. Biotechnol. 2013, 51, 12–25. [Google Scholar]
- Casadidio, C.; Peregrina, D.V.; Gigliobianco, M.R.; Deng, S.; Censi, R.; Di Martino, P. Chitin and Chitosans: Characteristics, Eco-Friendly Processes, and Applications in Cosmetic Science. Mar. Drugs 2019, 17, 369. [Google Scholar] [CrossRef] [PubMed]
- Morin-Crini, N.; Lichtfouse, E.; Torri, G.; Crini, G. Applications of Chitosan in Food, Pharmaceuticals, Medicine, Cosmetics, Agriculture, Textiles, Pulp and Paper, Biotechnology, and Environmental Chemistry. Environ. Chem. Lett. 2019, 17, 1667–1692. [Google Scholar] [CrossRef]
- Ahmadkelayeh, S.; Hawboldt, K. Extraction of Lipids and Astaxanthin from Crustacean By-Products: A Review on Supercritical CO2 Extraction. Trends Food Sci. Technol. 2020, 103, 94–108. [Google Scholar] [CrossRef]
- Hamdi, M.; Nasri, R.; Dridi, N.; Li, S.; Nasri, M. Development of Novel High-Selective Extraction Approach of Carotenoproteins from Blue Crab (Portunus Segnis) Shells, Contribution to the Qualitative Analysis of Bioactive Compounds by HR-ESI-MS. Food Chem. 2020, 302, 125334. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Hua, S.; Huang, Z.; Gu, Z.; Cheng, L.; Hong, Y. Comparison of Bioaccessibility of Astaxanthin Encapsulated in Starch-Based Double Emulsion with Different Structures. Carbohydr. Polym. 2021, 272, 118475. [Google Scholar] [CrossRef] [PubMed]
- Seabra, L.M.J.; Pedrosa, L.F.C. Astaxanthin: Structural and Functional Aspects. Rev. Nutr. 2010, 23, 1041–1050. [Google Scholar] [CrossRef]
- Brotosudarmo, T.H.P.; Limantara, L.; Setiyono, E.; Heriyanto. Structures of Astaxanthin and Their Consequences for Therapeutic Application. Int. J. Food Sci. 2020, 2020, 2156582. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Qiao, X.; Gu, J.; Li, X.; Cao, Y.; Xu, J.; Xue, C. Influence of Molecular Structure of Astaxanthin Esters on Their Stability and Bioavailability. Food Chem. 2021, 343, 128497. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Kim, B.; Lee, J.-Y. Astaxanthin Structure, Metabolism, and Health Benefits. J. Hum. Nutr. Food Sci. 2013, 1, 1003. [Google Scholar]
- Ambati, R.R.; Moi, P.S.; Ravi, S.; Aswathanarayana, R.G. Astaxanthin: Sources, Extraction, Stability, Biological Activities and Its Commercial Applications—A Review. Mar. Drugs 2014, 12, 128–152. [Google Scholar] [CrossRef] [PubMed]
- Qiu, D.; Wu, Y.-C.; Zhu, W.-L.; Yin, H.; Yi, L.-T. Identification of Geometrical Isomers and Comparison of Different Isomeric Samples of Astaxanthin. J. Food Sci. 2012, 77, C934–C940. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Nile, S.H.; Park, S.W. Carotenoids from Fruits and Vegetables: Chemistry, Analysis, Occurrence, Bioavailability and Biological Activities. Food Res. Int. 2015, 76, 735–750. [Google Scholar] [CrossRef] [PubMed]
- Higuera-Ciapara, I.; Félix-Valenzuela, L.; Goycoolea, F.M. Astaxanthin: A Review of Its Chemistry and Applications. Crit. Rev. Food Sci. Nutr. 2006, 46, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Gulzar, S.; Benjakul, S. Impact of Pulsed Electric Field Pretreatment on Yield and Quality of Lipid Extracted from Cephalothorax of Pacific White Shrimp (Litopenaeus Vannamei) by Ultrasound-assisted Process. Int. J. Food Sci. Technol. 2020, 55, 619–630. [Google Scholar] [CrossRef]
- Gómez-Guillén, M.C.; Montero, P.; López-Caballero, M.E.; Baccan, G.C.; Gómez-Estaca, J. Bioactive and Technological Functionality of a Lipid Extract from Shrimp (L. Vannamei) Cephalothorax. LWT 2018, 89, 704–711. [Google Scholar] [CrossRef]
- Rhodes, A.C.E. Dietary Effects on Carotenoid Composition in the Marine Harpacticoid Copepod Nitokra Lacustris. J. Plankton Res. 2007, 29, i73–i83. [Google Scholar] [CrossRef]
- Montoya, J.M.; Velazco Mata, S.; Acosta, J.L.; Herrera Cabrera, B.E.; López Valdez, L.G.; Reyes, C.; Barrales Cureño, J.H. Obtaining of Astaxanthin from Crab Exosqueletons and Shrimp Head Shells. Biointerface Res. Appl. Chem. 2021, 11, 13516–13523. [Google Scholar] [CrossRef]
- Routray, W.; Dave, D.; Cheema, S.K.; Ramakrishnan, V.V.; Pohling, J. Biorefinery Approach and Environment-Friendly Extraction for Sustainable Production of Astaxanthin from Marine Wastes. Crit. Rev. Biotechnol. 2019, 39, 469–488. [Google Scholar] [CrossRef] [PubMed]
- Babin, A.; Motreuil, S.; Teixeira, M.; Bauer, A.; Rigaud, T.; Moreau, J.; Moret, Y. Origin of the Natural Variation in the Storage of Dietary Carotenoids in Freshwater Amphipod Crustaceans. PLoS ONE 2020, 15, e0231247. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, Y.; Yoshida, H.; Kondo, K. Potential Anti-Atherosclerotic Properties of Astaxanthin. Mar. Drugs 2016, 14, 35. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.M.R.; Liang, Y.; Cheng, J.J.; Daroch, M. Astaxanthin-Producing Green Microalga Haematococcus Pluvialis: From Single Cell to High Value Commercial Products. Front. Plant Sci. 2016, 7, 531. [Google Scholar] [CrossRef] [PubMed]
- Nunes, A.N.; Roda, A.; Gouveia, L.F.; Fernández, N.; Bronze, M.R.; Matias, A.A. Astaxanthin Extraction from Marine Crustacean Waste Streams: An Integrate Approach between Microwaves and Supercritical Fluids. ACS Sustain. Chem. Eng. 2021, 9, 3050–3059. [Google Scholar] [CrossRef]
- Dalei, J.; Sahoo, D. Extraction and Characterisation of Astaxanthin from the Crustacean Shell Waste from Shrimp Processing Industries. Int. J. Pharm. Sci. Res. 2015, 6, 2532–2537. [Google Scholar] [CrossRef]
- Hu, J.; Lu, W.; Lv, M.; Wang, Y.; Ding, R.; Wang, L. Extraction and Purification of Astaxanthin from Shrimp Shells and the Effects of Different Treatments on Its Content. Rev. Bras. Farmacogn. 2019, 29, 24–29. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, Q.; Zhang, D.; Wei, S.; Sun, Q.; Xia, Q.; Shi, W.; Ji, H.; Liu, S. Comparison of the Proximate Composition and Nutritional Profile of Byproducts and Edible Parts of Five Species of Shrimp. Foods 2021, 10, 2603. [Google Scholar] [CrossRef] [PubMed]
- Messina, C.M.; Manuguerra, S.; Arena, R.; Renda, G.; Ficano, G.; Randazzo, M.; Fricano, S.; Sadok, S.; Santulli, A. In Vitro Bioactivity of Astaxanthin and Peptides from Hydrolisates of Shrimp (Parapenaeus Longirostris) By-Products: From the Extraction Process to Biological Effect Evaluation, as Pilot Actions for the Strategy “From Waste to Profit”. Mar. Drugs 2021, 19, 216. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas-Bojórquez, L.A.; Gutiérrez-Grijalva, E.P.; González-Aguilar, G.A.; López-Martinez, L.X.; Castillo-López, R.I.; Bastidas-Bastidas, P.D.J.; Heredia, J.B. Valorization of Fermented Shrimp Waste with Supercritical CO2 Conditions: Extraction of Astaxanthin and Effect of Simulated Gastrointestinal Digestion on Its Antioxidant Capacity. Molecules 2021, 26, 4465. [Google Scholar] [CrossRef] [PubMed]
- de Silva, M.P.K.S.K.; Senaarachchi, W.A.R.K. Efficiency of Biotransformation of Shellfish Waste to Carotenoprotein by Autolysis and Crab-Shrimp Endo-Enzymes. J. Aquat. Food Prod. Technol. 2021, 30, 526–534. [Google Scholar] [CrossRef]
- Dave, D.; Liu, Y.; Pohling, J.; Trenholm, S.; Murphy, W. Astaxanthin Recovery from Atlantic Shrimp (Pandalus Borealis) Processing Materials. Bioresour. Technol. Rep. 2020, 11, 100535. [Google Scholar] [CrossRef]
- Cheong, J.Y.; Muskhazli, M.; Nor Azwady, A.A.; Ahmad, S.A.; Adli, A.A. Three Dimensional Optimisation for the Enhancement of Astaxanthin Recovery from Shrimp Shell Wastes by Aeromonas Hydrophila. Biocatal. Agric. Biotechnol. 2020, 27, 101649. [Google Scholar] [CrossRef]
- Nunes, A.N.; Rodaa, A.; Matias, A.A. Recovery of Astaxanthin Pigments from Marine Crustacean Waste Streams Using Supercritical Fluid Technology. In Book of Abstracts The European Summer School in High Pressure Technology; Verlag der Technischen Universität: Graz, Austria, 2019; pp. 24–28. [Google Scholar]
- da Silva, A.K.N.; Rodrigues, B.D.; da Silva, L.H.M.; da Rodrigues, A.M.C. Drying and Extraction of Astaxanthin from Pink Shrimp Waste (Farfantepenaeus Subtilis): The Applicability of Spouted Beds. Food Sci. Technol. 2018, 38, 454–461. [Google Scholar] [CrossRef]
- Chintong, S.; Phatvej, W.; Rerk-Am, U.; Waiprib, Y.; Klaypradit, W. In Vitro Antioxidant, Antityrosinase, and Cytotoxic Activities of Astaxanthin from Shrimp Waste. Antioxidants 2019, 8, 128. [Google Scholar] [CrossRef]
- Irna, C.; Jaswir, I.; Othman, R.; Jimat, D.N. Comparison between High-Pressure Processing and Chemical Extraction: Astaxanthin Yield from Six Species of Shrimp Carapace. J. Diet. Suppl. 2018, 15, 805–813. [Google Scholar] [CrossRef]
- Darachai, P.; Limpawattana, M.; Hawangjoo, M.; Klaypradit, W. Effects of Shrimp Waste Types and Their Cooking on Properties of Extracted Astaxanthin and Its Characteristics in Liposomes. J. Food Nutr. Res. 2019, 7, 530–536. [Google Scholar] [CrossRef]
- Li, J.; Sun, W.; Ramaswamy, H.S.; Yu, Y.; Zhu, S.; Wang, J.; Li, H. High Pressure Extraction of Astaxanthin from Shrimp Waste (Penaeus Vannamei Boone): Effect on Yield and Antioxidant Activity. J. Food Process Eng. 2017, 40, e12353. [Google Scholar] [CrossRef]
- Gómez-Estaca, J.; Calvo, M.M.; Álvarez-Acero, I.; Montero, P.; Gómez Guillén, M.C. Characterization and Storage Stability of Astaxanthin Esters, Fatty Acid Profile and a -Tocopherol of Lipid Extract from Shrimp (L. Vannamei) Waste with Potential Applications as Food Ingredient. Food Chem. 2017, 216, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Antunes-Valcareggi, S.A.; Ferreira, S.R.S.; Hense, H. Enzymatic Hydrolysis of Blue Crab (Callinectes Sapidus) Waste Processing to Obtain Chitin, Protein, and Astaxanthin-Enriched Extract. Int. J. Environ. Agric. Res. 2017, 3, 81–92. [Google Scholar]
- Shazana, A.R.; Masturah, M.; Badlishah, S.B.; Rashidi, O.; Russly, A.R. Optimisation of Supercritical Fluid Extraction of Astaxanthin From Penaeus Monodon Waste Using Ethanol-Modified Carbon Dioxide. J. Eng. Sci. Technol. 2016, 11, 722–736. [Google Scholar]
- Soultani, G.; Strati, I.F. Assessment of Functional Lipid Constituents of Red (Aristaeomorpha Foliacea) and Pink (Parapenaeus Longirostris) Shrimps. J. Aquac. Res. Dev. 2016, 7, 452. [Google Scholar] [CrossRef]
- Caramujo, M.-J.; de Carvalho, C.C.C.R.; Silva, S.J.; Carman, K.R. Dietary Carotenoids Regulate Astaxanthin Content of Copepods and Modulate Their Susceptibility to UV Light and Copper Toxicity. Mar. Drugs 2012, 10, 998–1018. [Google Scholar] [CrossRef] [PubMed]
- Sharayei, P.; Azarpazhooh, E.; Zomorodi, S.; Einafshar, S.; Ramaswamy, H.S. Optimization of Ultrasonic-Assisted Extraction of Astaxanthin from Green Tiger (Penaeus semisulcatus) Shrimp Shell. Ultrason. Sonochem. 2021, 76, 105666. [Google Scholar] [CrossRef]
- Gulzar, S.; Benjakul, S. Ultrasound Waves Increase the Yield and Carotenoid Content of Lipid Extracted from Cephalothorax of Pacific White Shrimp (Litopenaeus vannamei). Eur. J. Lipid Sci. Technol. 2018, 120, 1700495. [Google Scholar] [CrossRef]
- Sánchez-Camargo, A.P.; Almeida Meireles, M.Â.; Lopes, B.L.F.; Cabral, F.A. Proximate Composition and Extraction of Carotenoids and Lipids from Brazilian Redspotted Shrimp Waste (Farfantepenaeus paulensis). J. Food Eng. 2011, 102, 87–93. [Google Scholar] [CrossRef]
- Sánchez-Camargo, A.P.; Martinez-Correa, H.A.; Paviani, L.C.; Cabral, F.A. Supercritical CO2 Extraction of Lipids and Astaxanthin from Brazilian Redspotted Shrimp Waste (Farfantepenaeus paulensis). J. Supercrit. Fluids 2011, 56, 164–173. [Google Scholar] [CrossRef]
- Sánchez-Camargo, A.P.; Meireles, M.Â.A.; Ferreira, A.L.K.; Saito, E.; Cabral, F.A. Extraction of ω-3 Fatty Acids and Astaxanthin from Brazilian Redspotted Shrimp Waste Using Supercritical CO2+ethanol Mixtures. J. Supercrit. Fluids 2012, 61, 71–77. [Google Scholar] [CrossRef]
- Wang, W.; Liu, M.; Fawzy, S.; Xue, Y.; Wu, M.; Huang, X.; Yi, G.; Lin, Q. Effects of Dietary Phaffia Rhodozyma Astaxanthin on Growth Performance, Carotenoid Analysis, Biochemical and Immune-Physiological Parameters, Intestinal Microbiota, and Disease Resistance in Penaeus Monodon. Front. Microbiol. 2021, 12, 762689. [Google Scholar] [CrossRef] [PubMed]
- Kidd, P. Astaxanthin, Cell Membrane Nutrient with Diverse Clinical Benefits and Anti-Aging Potential. Altern. Med. Rev. 2011, 16, 355–364. [Google Scholar] [PubMed]
- Naguib, Y.M.A. Antioxidant Activities of Astaxanthin and Related Carotenoids. J. Agric. Food Chem. 2000, 48, 1150–1154. [Google Scholar] [CrossRef] [PubMed]
- Martin, H.D.; Ruck, C.; Schmidt, M.; Sell, S.; Beutner, S.; Mayer, B.; Walsh, R. Chemistry of Carotenoid Oxidation and Free Radical Reactions. Pure Appl. Chem. 1999, 71, 2253–2262. [Google Scholar] [CrossRef]
- Nishida, Y.; Yamashita, E.; Miki, W. Quenching Activities of Common Hydrophilic and Lipophilic Antioxidants against Singlet Oxygen Using Chemiluminescence Detection System. Carotenoid Sci. 2007, 11, 16–20. [Google Scholar]
- Memar, M.Y.; Ghotaslou, R.; Samiei, M.; Adibkia, K. Antimicrobial Use of Reactive Oxygen Therapy: Current Insights. Infect. Drug Resist. 2018, 11, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhou, X.; Huang, Y.; Liao, B.; Cheng, L.; Ren, B. Reactive Oxygen Species in Pathogen Clearance: The Killing Mechanisms, the Adaption Response, and the Side Effects. Front. Microbiol. 2021, 11, 622534. [Google Scholar] [CrossRef] [PubMed]
- Aribisala, J.O.; Nkosi, S.; Idowu, K.; Nurain, I.O.; Makolomakwa, G.M.; Shode, F.O.; Sabiu, S. Astaxanthin-Mediated Bacterial Lethality: Evidence from Oxidative Stress Contribution and Molecular Dynamics Simulation. Oxid. Med. Cell. Longev. 2021, 2021, 7159652. [Google Scholar] [CrossRef] [PubMed]
- Karpiński, T.M.; Ożarowski, M.; Alam, R.; Łochyńska, M.; Stasiewicz, M. What Do We Know about Antimicrobial Activity of Astaxanthin and Fucoxanthin? Mar. Drugs 2022, 20, 36. [Google Scholar] [CrossRef] [PubMed]
- Suganya, V.; Asheeba, S. Antioxidant and Antimicrobial Activity of Astaxanthin Isolated from Three Varieties of Crabs. Int. J. Recent Sci. Res. 2015, 6, 6753–6758. [Google Scholar]
- Ushakumari, U.N.; Ramanujan, R. Isolation of Astaxanthin from Marine Yeast and Study of Its Pharmacological Activity. Int. Curr. Pharm. J. 2013, 2, 67–69. [Google Scholar] [CrossRef]
- Mageswari, A.; Subramanian, P.; Srinivasan, R.; Karthikeyan, S.; Gothandam, K.M. Astaxanthin from Psychrotrophic Sphingomonas Faeni Exhibits Antagonism against Food-Spoilage Bacteria at Low Temperatures. Microbiol. Res. 2015, 179, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Irna, C.; Jaswir, I.; Othman, R.; Jimat, D.N. Antioxidant and Antimicrobial Activities of Astaxanthin from Penaeus Monodon in Comparison between Chemical Extraction and High Pressure Processing (HPP). Int. Food Res. J. 2017, 24, 508–513. [Google Scholar]
- Rather, A.H.; Singh, S.; Choudhary, S. Antibacterial Activity of Haematococcus Pluvialis Crude Astaxanthin Extract. J. Drug Deliv. Ther. 2021, 11, 28–30. [Google Scholar] [CrossRef]
- Guan, L.; Liu, J.; Yu, H.; Tian, H.; Wu, G.; Liu, B.; Dong, P.; Li, J.; Liang, X. Water-Dispersible Astaxanthin-Rich Nanopowder: Preparation, Oral Safety and Antioxidant Activity in Vivo. Food Funct. 2019, 10, 1386–1397. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Hu, S.; Fleming, E.; Lee, J.Y.; Luo, Y. Chitosan-Caseinate-Dextran Ternary Complex Nanoparticles for Potential Oral Delivery of Astaxanthin with Significantly Improved Bioactivity. Int. J. Biol. Macromol. 2020, 151, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.Z.; Li, M.Y.; Wang, Y.T.; Zhu, M.J. Astaxanthin from Phaffia Rhodozyma: Microencapsulation with Carboxymethyl Cellulose Sodium and Microcrystalline Cellulose and Effects of Microencapsulated Astaxanthin on Yogurt Properties. LWT 2018, 96, 152–160. [Google Scholar] [CrossRef]
- Tirado, D.F.; Palazzo, I.; Scognamiglio, M.; Calvo, L.; Della Porta, G.; Reverchon, E. Astaxanthin Encapsulation in Ethyl Cellulose Carriers by Continuous Supercritical Emulsions Extraction: A Study on Particle Size, Encapsulation Efficiency, Release Profile and Antioxidant Activity. J. Supercrit. Fluids 2019, 150, 128–136. [Google Scholar] [CrossRef]
- Montanari, E.; Di Meo, C.; Coviello, T.; Gueguen, V.; Pavon-Djavid, G.; Matricardi, P. Intracellular Delivery of Natural Antioxidants via Hyaluronan Nanohydrogels. Pharmaceutics 2019, 11, 532. [Google Scholar] [CrossRef] [PubMed]
- Weintraub, S.; Shpigel, T.; Harris, L.G.; Schuster, R.; Lewis, E.C.; Lewitus, D.Y. Astaxanthin-Based Polymers as New Antimicrobial Compounds. Polym. Chem. 2017, 8, 4182–4189. [Google Scholar] [CrossRef]
- Karuppusamy, S.; Kim, H.; Saravana, P.S.; Chun, B.S.; Kang, H.W. Astaxanthin-Alpha Tocopherol Nanoemulsion Formulation by Emulsification Methods: Investigation on Anticancer, Wound Healing, and Antibacterial Effects. Colloids Surf. B Biointerfaces 2018, 172, 170–179. [Google Scholar] [CrossRef]
- Sukmawati; Fawwaz, M.; Pratama, M.; Hasrawati, A. Potential of Astaxanthin from Asian Tiger Shrimp (Penaeus Monodon) Shell Extract as an Antibacterial and Anti-Inflammatory. J. Glob. Pharma Technol. 2019, 11, 2017–2022. [Google Scholar]
- Kohandel, Z.; Farkhondeh, T.; Aschner, M.; Pourbagher-Shahri, A.M.; Samarghandian, S. Anti-Inflammatory Action of Astaxanthin and Its Use in the Treatment of Various Diseases. Biomed. Pharmacother. 2022, 145, 112179. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.J.; Lu, J.W.; Liu, F.C.; Lee, C.H.; Lee, H.S.; Ho, Y.J.; Hsieh, T.H.; Wu, C.C.; Wang, C.C. Astaxanthin Attenuates Joint Inflammation Induced by Monosodium Urate Crystals. FASEB J. 2020, 34, 11215–11226. [Google Scholar] [CrossRef]
- Hwang, Y.H.; Hong, S.G.; Mun, S.K.; Kim, S.J.; Lee, S.J.; Kim, J.J.; Kang, K.Y.; Yee, S.T. The Protective Effects of Astaxanthin on the OVA-Induced Asthma Mice Model. Molecules 2017, 22, 2019. [Google Scholar] [CrossRef]
- Bharathiraja, S.; Manivasagan, P.; Bui, N.Q.; Oh, Y.O.; Lim, I.G.; Park, S.; Oh, J. Cytotoxic Induction and Photoacoustic Imaging of Breast Cancer Cells Using Astaxanthin-Reduced Gold Nanoparticles. Nanomaterials 2016, 6, 78. [Google Scholar] [CrossRef]
- Zhang, X.; Yin, W.; Qi, Y.; Li, X.; Zhang, W.; He, G. Microencapsulation of Astaxanthin in Alginate Using Modified Emulsion Technology: Preparation, Characterization, and Cytostatic Activity. Can. J. Chem. Eng. 2017, 95, 412–419. [Google Scholar] [CrossRef]
- Haung, H.Y.; Wang, Y.C.; Cheng, Y.C.; Kang, W.; Hu, S.H.; Liu, D.; Xiao, C.; Wang, H.M.D.; Ali, D. A Novel Oral Astaxanthin Nanoemulsion from Haematococcus Pluvialis Induces Apoptosis in Lung Metastatic Melanoma. Oxid. Med. Cell. Longev. 2020, 2020, 2647670. [Google Scholar] [CrossRef]
- Ni, X.; Yu, H.; Wang, S.; Zhang, C.; Shen, S. Astaxanthin Inhibits PC-3 Xenograft Prostate Tumor Growth in Nude Mice. Mar. Drugs 2017, 15, 66. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Xu, F.; Wang, M.; Li, L.; Qiao, T.; Cui, H.; Li, Z.; Sun, C. Dietary Natural Astaxanthin at an Early Stage Inhibits N-Nitrosomethylbenzylamine-Induced Esophageal Cancer Oxidative Stress and Inflammation via Downregulation of NFκB and COX2 in F344 Rats. Onco. Targets. Ther. 2019, 12, 5087–5096. [Google Scholar] [CrossRef] [PubMed]
- Ohno, T.; Shimizu, M.; Shirakami, Y.; Miyazaki, T.; Ideta, T.; Kochi, T.; Kubota, M.; Sakai, H.; Tanaka, T.; Moriwaki, H. Preventive Effects of Astaxanthin on Diethylnitrosamine-Induced Liver Tumorigenesis in C57/BL/KsJ-Db/Db Obese Mice. Hepatol. Res. 2016, 46, E201–E209. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Liang, P.; Qu, Y.; An, T.; Wang, J.; Deng, X.; Bai, L.; Shen, P.; Bai, D. Protective Effect of Astaxanthin against SnS2 Nanoflowers Induced Testes Toxicity by Suppressing RIPK1-RIPK3-MLKL Signaling in Mice. Food Chem. Toxicol. 2020, 145, 111736. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.C.; Huang, H.H.; Wu, Y.J.; Manousakas, I.; Yang, C.C.; Kuo, S.M. Therapeutic and Protective Effects of Liposomal Encapsulation of Astaxanthin in Mice with Alcoholic Liver Fibrosis. Int. J. Mol. Sci. 2019, 20, 4057. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.H.; Chang, C.C.; Lin, S.T.; Chyau, C.C.; Peng, R.Y. Improved Hepatoprotective Effect of Liposome-Encapsulated Astaxanthin in Lipopolysaccharide-Induced Acute Hepatotoxicity. Int. J. Mol. Sci. 2016, 17, 1128. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, S.; Bi, J.; Gu, J.; Deng, Y.; Liu, C. Astaxanthin Pretreatment Attenuates Acetaminophen-Induced Liver Injury in Mice. Int. Immunopharmacol. 2017, 45, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Özbeyli, D.; Gürler, E.B.; Buzcu, H.; Çilingir-Kaya, Ö.T.; Çam, M.E.; Yüksel, M. Astaxanthin Alleviates Oxidative Damage in Acute Pancreatitis via Direct Antioxidant Mechanisms. Turkish J. Gastroenterol. 2020, 31, 706–712. [Google Scholar] [CrossRef]
- Zhuge, F.; Ni, Y.; Wan, C.; Liu, F.; Fu, Z. Anti-Diabetic Effects of Astaxanthin on an Stz-Induced Diabetic Model in Rats. Endocr. J. 2021, 68, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Akduman, H.; Tayman, C.; Çakir, U.; Çakir, E.; Dilli, D.; Türkmenoğlu, T.T.; Gönel, A. Astaxanthin Prevents Lung Injury Due to Hyperoxia and Inflammation. Comb. Chem. High Throughput Screen. 2021, 24, 1243–1250. [Google Scholar] [CrossRef]
- Li, H.; Li, J.; Hou, C.; Li, J.; Peng, H.; Wang, Q. The Effect of Astaxanthin on Inflammation in Hyperosmolarity of Experimental Dry Eye Model in Vitro and in vivo. Exp. Eye Res. 2020, 197, 108113. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, K.; Hongo, N.; Fujishita, M.; Takahashi, Y.; Adachi, Y. Protective Effect of Astaxanthin on Skin Deterioration. J. Clin. Biochem. Nutr. 2017, 61, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Jeon, S.H.; Ham, H.J.; Lee, H.P.; Song, M.J.; Hong, J.T. Improved Anti-Inflammatory Effects of Liposomal Astaxanthin on a Phthalic Anhydride-Induced Atopic Dermatitis Model. Front. Immunol. 2020, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yaqoob, Z.; Arshad, M.S.; Imran, M.; Munir, H.; Qaisrani, T.B.; Khalid, W.; Asghar, Z.; Suleria, H.A.R. Mechanistic Role of Astaxanthin Derived from Shrimp against Certain Metabolic Disorders. Food Sci. Nutr. 2022, 10, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Faraone, I.; Sinisgalli, C.; Ostuni, A.; Armentano, M.F.; Carmosino, M.; Milella, L.; Russo, D.; Labanca, F.; Khan, H. Astaxanthin Anticancer Effects Are Mediated through Multiple Molecular Mechanisms: A Systematic Review. Pharmacol. Res. 2020, 155, 104689. [Google Scholar] [CrossRef] [PubMed]
- Grimmig, B.; Kim, S.-H.; Nash, K.; Bickford, P.C.; Douglas Shytle, R. Neuroprotective Mechanisms of Astaxanthin: A Potential Therapeutic Role in Preserving Cognitive Function in Age and Neurodegeneration. GeroScience 2017, 39, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lim, J.; Kim, H. Astaxanthin Inhibits Mitochondrial Dysfunction and Interleukin-8 Expression in Helicobacter Pylori-Infected Gastric Epithelial Cells. Nutrients 2018, 10, 1320. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Cui, Z.; Cui, H.; Wang, Y.; Zhong, C. Astaxanthin Protects Astrocytes against Trauma-Induced Apoptosis through Inhibition of NKCC1 Expression via the NF-ΚB Signaling Pathway. BMC Neurosci. 2017, 18, 42. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Song, M.; Gao, Z.; Cai, X.; Dixon, W.; Chen, X.; Cao, Y.; Xiao, H. Stereoisomers of Astaxanthin Inhibit Human Colon Cancer Cell Growth by Inducing G2/M Cell Cycle Arrest and Apoptosis. J. Agric. Food Chem. 2016, 64, 7750–7759. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Niu, H.; Shao, A.; Wu, C.; Dixon, B.; Zhang, J.; Yang, S.; Wang, Y. Astaxanthin as a Potential Neuroprotective Agent for Neurological Diseases. Mar. Drugs 2015, 13, 5750–5766. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-K.; Park, Y.-S.; Choi, D.-K.; Chang, H.-I. Effects of Astaxanthin on the Production of NO and the Expression of COX-2 and INOS in LPS-Stimulated BV2 Microglial Cells. J. Microbiol. Biotechnol. 2008, 18, 1990–1996. [Google Scholar] [PubMed]
- Santocono, M.; Zurria, M.; Berrettini, M.; Fedeli, D.; Falcioni, G. Influence of Astaxanthin, Zeaxanthin and Lutein on DNA Damage and Repair in UVA-Irradiated Cells. J. Photochem. Photobiol. B Biol. 2006, 85, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.K.; Ima-Nirwana, S.; Chin, K. Effects of Astaxanthin on the Protection of Muscle Health (Review). Exp. Ther. Med. 2020, 20, 2941–2952. [Google Scholar] [CrossRef] [PubMed]
- Kamath, B.S.; Srikanta, B.M.; Dharmesh, S.M.; Sarada, R.; Ravishankar, G.A. Ulcer Preventive and Antioxidative Properties of Astaxanthin from Haematococcus Pluvialis. Eur. J. Pharmacol. 2008, 590, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, T.; Tawara, A. Effects of Astaxanthin on Eyestrain Induced by Accommodative Dysfunction. J. Eye 2006, 23, 829. [Google Scholar]
- Manivasagan, P.; Bharathiraja, S.; Santha Moorthy, M.; Mondal, S.; Seo, H.; Dae Lee, K.; Oh, J. Marine Natural Pigments as Potential Sources for Therapeutic Applications. Crit. Rev. Biotechnol. 2018, 38, 745–761. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Chang, M.J.; Choi, H.D.; Youn, Y.-K.; Kim, J.T.; Oh, J.M.; Shin, W.G. Protective Effects of Haematococcus Astaxanthin on Oxidative Stress in Healthy Smokers. J. Med. Food 2011, 14, 1469–1475. [Google Scholar] [CrossRef] [PubMed]
- Bhuvaneswari, S.; Arunkumar, E.; Viswanathan, P.; Anuradha, C.V. Astaxanthin Restricts Weight Gain, Promotes Insulin Sensitivity and Curtails Fatty Liver Disease in Mice Fed a Obesity-Promoting Diet. Process Biochem. 2010, 45, 1406–1414. [Google Scholar] [CrossRef]
- Hussein, G.; Goto, H.; Oda, S.; Iguchi, T.; Sankawa, U.; Matsumoto, K.; Watanabe, H. Antihypertensive Potential and Mechanism of Action of Astaxanthin: II. Vascular Reactivity and Hemorheology in Spontaneously Hypertensive Rats. Biol. Pharm. Bull. 2005, 28, 967–971. [Google Scholar] [CrossRef] [PubMed]
- Satoh, A.; Tsuji, S.; Okada, Y.; Murakami, N.; Urami, M.; Nakagawa, K.; Ishikura, M.; Katagiri, M.; Koga, Y.; Shirasawa, T. Preliminary Clinical Evaluation of Toxicity and Efficacy of A New Astaxanthin-Rich Haematococcus Pluvialis Extract. J. Clin. Biochem. Nutr. 2009, 44, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Palozza, P.; Barone, E.; Mancuso, C.; Picci, N. The Protective Role of Carotenoids against 7-Keto-Cholesterol Formation in Solution. Mol. Cell. Biochem. 2008, 309, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, Q.; Zhao, S.; Ji, X.; Qiu, J.; Wang, J.; Zhou, Y.; Cai, Q.; Zhang, J.; Gao, H. Astaxanthin Attenuated Pressure Overload-Induced Cardiac Dysfunction and Myocardial Fibrosis: Partially by Activating SIRT1. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 1715–1728. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T. Astaxanthin. In Nutraceuticals; Elsevier: Amsterdam, The Netherlands, 2016; pp. 531–539. [Google Scholar] [CrossRef]
- Preuss, H.G.; Echard, B.; Bagchi, D.; Perricone, N.V.; Yamashita, E. Astaxanthin Lowers Blood Pressure and Lessens the Activity of the Renin-Angiotensin System in Zucker Fatty Rats. J. Funct. Foods 2009, 1, 13–22. [Google Scholar] [CrossRef]
- Capelli, B.; Talbott, S.; Ding, L. Astaxanthin Sources: Suitability for Human Health and Nutrition. Funct. Foods Heal. Dis. 2019, 9, 430. [Google Scholar] [CrossRef]
- Li, J.; Guo, C.; Wu, J. Astaxanthin in Liver Health and Disease: A Potential Therapeutic Agent. Drug Des. Devel. Ther. 2020, 14, 2275–2285. [Google Scholar] [CrossRef] [PubMed]
- Mercke Odeberg, J.; Lignell, Å.; Pettersson, A.; Höglund, P. Oral Bioavailability of the Antioxidant Astaxanthin in Humans Is Enhanced by Incorporation of Lipid Based Formulations. Eur. J. Pharm. Sci. 2003, 19, 299–304. [Google Scholar] [CrossRef]
- Zhou, Q.; Xu, J.; Yang, L.; Gu, C.; Xue, C. Thermal Stability and Oral Absorbability of Astaxanthin Esters from Haematococcus Pluvialis in Balb/c Mice. J. Sci. Food Agric. 2019, 99, 3662–3671. [Google Scholar] [CrossRef] [PubMed]
- Ranga Rao, A.; Raghunath Reddy, R.L.; Baskaran, V.; Sarada, R.; Ravishankar, G.A. Characterization of Microalgal Carotenoids by Mass Spectrometry and Their Bioavailability and Antioxidant Properties Elucidated in Rat Model. J. Agric. Food Chem. 2010, 58, 8553–8559. [Google Scholar] [CrossRef] [PubMed]
- Fukami, H.; Namikawa, K.; Sugiura-Tomimori, N.; Sumida, M.; Katano, K.; Nakao, M. Chemical Synthesis of Astaxanthin N-Octanoic Acid Monoester and Diester and Evaluation of Their Oral Absorbability. J. Oleo Sci. 2006, 55, 653–656. [Google Scholar] [CrossRef]
- Talukdar, J.; Dasgupta, S.; Nagle, V.; Bhadra, B. COVID-19: Potential of Microalgae Derived Natural Astaxanthin As Adjunctive Supplement in Alleviating Cytokine Storm. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Niu, T.; Zhou, J.; Wang, F.; Xuan, R.; Chen, J.; Wu, W.; Chen, H. Safety Assessment of Astaxanthin from Haematococcus Pluvialis: Acute Toxicity, Genotoxicity, Distribution and Repeat-Dose Toxicity Studies in Gestation Mice. Regul. Toxicol. Pharmacol. 2020, 115, 104695. [Google Scholar] [CrossRef]
- Yoon, H.-S.; Cho, H.H.; Cho, S.; Lee, S.-R.; Shin, M.-H.; Chung, J.H. Supplementing with Dietary Astaxanthin Combined with Collagen Hydrolysate Improves Facial Elasticity and Decreases Matrix Metalloproteinase-1 and -12 Expression: A Comparative Study with Placebo. J. Med. Food 2014, 17, 810–816. [Google Scholar] [CrossRef]
- Giordano, F.J. Oxygen, Oxidative Stress, Hypoxia, and Heart Failure. J. Clin. Investig. 2005, 115, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-W.; Xu, X.-C.; Liu, T.; Yuan, S. Mitochondrion-Permeable Antioxidants to Treat ROS-Burst-Mediated Acute Diseases. Oxid. Med. Cell. Longev. 2016, 2016, 6859523. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, E. Let Astaxanthin Be Thy Medicine. PharmaNutrition 2015, 3, 115–122. [Google Scholar] [CrossRef]
- Choi, H.D.; Youn, Y.K.; Shin, W.G. Positive Effects of Astaxanthin on Lipid Profiles and Oxidative Stress in Overweight Subjects. Plant Foods Hum. Nutr. 2011, 66, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Ryu, S.K.; King, T.J.; Fujioka, K.; Pattison, J.; Pashkow, F.J.; Tsimikas, S. Effect of an Oral Astaxanthin Prodrug (CDX-085) on Lipoprotein Levels and Progression of Atherosclerosis in LDLR−/− and ApoE−/− Mice. Atherosclerosis 2012, 222, 99–105. [Google Scholar] [CrossRef]
- Kato, T.; Kasai, T.; Sato, A.; Ishiwata, S.; Yatsu, S.; Matsumoto, H.; Shitara, J.; Murata, A.; Shimizu, M.; Suda, S.; et al. Effects of 3-Month Astaxanthin Supplementation on Cardiac Function in Heart Failure Patients with Left Ventricular Systolic Dysfunction-A Pilot Study. Nutrients 2020, 12, 1896. [Google Scholar] [CrossRef]
- Maria, A.G.; Graziano, R.; Nicolantonio, D. Carotenoids: Potential Allies of Cardiovascular Health? Food Nutr. Res. 2015, 59, 26762. [Google Scholar] [CrossRef]
- Park, J.S.; Chyun, J.H.; Kim, Y.K.; Line, L.L.; Chew, B.P. Astaxanthin Decreased Oxidative Stress and Inflammation and Enhanced Immune Response in Humans. Nutr. Metab. 2010, 7, 18. [Google Scholar] [CrossRef]
- Yoshida, H.; Yanai, H.; Ito, K.; Tomono, Y.; Koikeda, T.; Tsukahara, H.; Tada, N. Administration of Natural Astaxanthin Increases Serum HDL-Cholesterol and Adiponectin in Subjects with Mild Hyperlipidemia. Atherosclerosis 2010, 209, 520–523. [Google Scholar] [CrossRef] [PubMed]
- Miyawaki, H.; Takahashi, J.; Tsukahara, H.; Takehara, I. Effects of Astaxanthin on Human Blood Rheology. J. Clin. Biochem. Nutr. 2008, 43, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, T.; Hosoda, K.; Hirano, R.; Kurata, H.; Matsumoto, A.; Miki, W.; Kamiyama, M.; Itakura, H.; Yamamoto, S.; Kondo, K. Inhibition of Low-Density Lipoprotein Oxidation by Astaxanthin. J. Atheroscler. Thromb. 2000, 7, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Shi, Y.; Peng, X.; Liu, H.; Peng, Y.; He, L. Astaxanthin Attenuates Adriamycin-Induced Focal Segmental Glomerulosclerosis. Pharmacology 2015, 95, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Mohan, V.; Edamakanti, C.R.; Sharma, V. Role of Neuroinflammation in Neurodegenerative Disorders. In The Molecular Immunology of Neurological Diseases; Elsevier: Amsterdam, The Netherlands, 2021; pp. 41–49. [Google Scholar] [CrossRef]
- Fakhri, S.; Aneva Yosifova, I.; Farzaei, M.H.; Sobarzo-Sánchez, E. The Neuroprotective Effects of Astaxanthin: Therapeutic Targets and Clinical Perspective. Molecules 2019, 24, 2640. [Google Scholar] [CrossRef] [PubMed]
- Zarneshan, S.N.; Fakhri, S.; Farzaei, M.H.; Khan, H.; Saso, L. Astaxanthin Targets PI3K/Akt Signaling Pathway toward Potential Therapeutic Applications. Food Chem. Toxicol. 2020, 145, 111714. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.O.; Panda, B.P.; Parvez, S.; Kaundal, M.; Hussain, S.; Akhtar, M.; Najmi, A.K. Neuroprotective Role of Astaxanthin in Hippocampal Insulin Resistance Induced by Aβ Peptides in Animal Model of Alzheimer’s Disease. Biomed. Pharmacother. 2019, 110, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhu, J.; Yin, W.; Ding, X. Astaxanthin Improves Cognitive Deficits from Oxidative Stress, Nitric Oxide Synthase and Inflammation through Upregulation of PI3K/Akt in Diabetes Rat. Int. J. Clin. Exp. Pathol. 2015, 8, 6083–6094. [Google Scholar] [PubMed]
- Katagiri, M.; Satoh, A.; Tsuji, S.; Shirasawa, T. Effects of Astaxanthin-Rich Haematococcus Pluvialis Extract on Cognitive Function: A Randomised, Double-Blind, Placebo-Controlled Study. J. Clin. Biochem. Nutr. 2012, 51, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Ito, N.; Seki, S.; Ueda, F. The Protective Role of Astaxanthin for UV-Induced Skin Deterioration in Healthy People—A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2018, 10, 817. [Google Scholar] [CrossRef] [PubMed]
- Imai, A.; Oda, Y.; Ito, N.; Seki, S.; Nakagawa, K.; Miyazawa, T.; Ueda, F. Effects of Dietary Supplementation of Astaxanthin and Sesamin on Daily Fatigue: A Randomized, Double-Blind, Placebo-Controlled, Two-Way Crossover Study. Nutrients 2018, 10, 281. [Google Scholar] [CrossRef] [PubMed]
- Katsumata, T.; Ishibashi, T.; Kyle, D. A Sub-Chronic Toxicity Evaluation of a Natural Astaxanthin-Rich Carotenoid Extract of Paracoccus Carotinifaciens in Rats. Toxicol. Rep. 2014, 1, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Iwabayashi, M.; Fujioka, N.; Nomoto, K.; Miyazaki, R.; Takahashi, H.; Hibino, S.; Takahashi, Y.; Nishikawa, K.; Nishida, M.; Yonei, Y. Efficacy and Safety of Eight-Week Treatment with Astaxanthin in Individuals Screened for Increased Oxidative Stress Burden. ANTI-AGING Med. 2009, 6, 15–21. [Google Scholar] [CrossRef]
- Turck, D.; Castenmiller, J.; de Henauw, S.; Hirsch-Ernst, K.I.; Kearney, J.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; Pelaez, C.; et al. Safety of Astaxanthin for Its Use as a Novel Food in Food Supplements. EFSA J. 2020, 18, e05993. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-J.; Lin, J.-Y.; Wang, D.-S.; Chen, C.-H.; Chiou, M.-H. Safety Assessment of Astaxanthin Derived from Engineered Escherichia Coli K-12 Using a 13-Week Repeated Dose Oral Toxicity Study and a Prenatal Developmental Toxicity Study in Rats. Regul. Toxicol. Pharmacol. 2017, 87, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Buesen, R.; Schulte, S.; Strauss, V.; Treumann, S.; Becker, M.; Gröters, S.; Carvalho, S.; van Ravenzwaay, B. Safety Assessment of [3S, 3′S]-Astaxanthin—Subchronic Toxicity Study in Rats. Food Chem. Toxicol. 2015, 81, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Bampidis, V.; Azimonti, G.; de Bastos, M.L.; Christensen, H.; Dusemund, B.; Kouba, M.; Kos Durjava, M.; López-Alonso, M.; López Puente, S.; Marcon, F.; et al. Assessment of the Application for Renewal of Authorisation of Selenomethionine Produced by Saccharomyces Cerevisiae NCYC R397 for All Animal Species. EFSA J. 2019, 17, e05539. [Google Scholar] [CrossRef] [PubMed]
- Jannel, S.; Caro, Y.; Bermudes, M.; Petit, T. Novel Insights into the Biotechnological Production of Haematococcus Pluvialis-Derived Astaxanthin: Advances and Key Challenges to Allow Its Industrial Use as Novel Food Ingredient. J. Mar. Sci. Eng. 2020, 8, 789. [Google Scholar] [CrossRef]
- Lima, S.G.M.; Freire, M.C.L.C.; da Oliveira, V.S.; Solisio, C.; Converti, A.; de Lima, Á.A.N. Astaxanthin Delivery Systems for Skin Application: A Review. Mar. Drugs 2021, 19, 511. [Google Scholar] [CrossRef] [PubMed]
| Species | Byproduct | Extraction Procedure | Astaxanthin Content | Salient Finding | Reference |
|---|---|---|---|---|---|
| Shrimps and prawns (Litopenaeus vannamei (L.v), Macrobrachium rosenbergii (M.r), Penaeus monodon (P.m), Fenneropenaeus chinensis (F.c), and Penaeus japonicus (P.j)) | Head, shell, and tail | Solvent extraction using dichloromethane: methanol (1:3, v/v) | 19.2 µg/g (L.v.), 15.7 µg/g (M.r.), 2.9 µg/g (P.m.), 7.1 µg/g (F.c.), and 5.8 µg/g (P.j.) | The byproduct yield was 44.06–62.53%. | [63] |
| Brown crab (Cancer pagurus) | Crab shells | Microwave (MW) pretreatment in ethanol (at 140 °C and 300 W, during 90 s) followed by supercritical fluid extraction (SFE) (500 bar, 40 °C, and 13 wt % ethanol content, 30 min) | 1023 µg/g dry extract | In comparison to conventional extraction, the SFE conditions after MW pretreatment gave the best results. | [60] |
| Shrimp (Parapenaeus longirostris) | Exoskeleton, including cephalothorax and abdominal parts | Extraction using fish oil (CVO) and different fatty acid ethyl esters (TFA) and by SFE (350 bar, 40 °C, 30 min of static extraction followed by dynamic extraction with a CO2 flow 2.5 L/min for 2 h) | CVO: 149.1 ± 0.8 µg/g TFA: 160.1 ± 8.9 µg/g | The highest astaxanthin yields were obtained for wet byproducts, extracted with ethyl esters fatty acids at a 2.0 ratio. | [64] |
| Shrimp (Litopenaeus vannamei) | Fermented shrimp exoskeleton | SFE (300 bar, 60 °C, and 6 mL/min) | 12.62%, 0.52 µg/g | Extracts showed antioxidant activity in vitro. | [65] |
| Tiger prawn (Penaeus monodon) and mud crab (Scylla serrata) | Discards | Autolysis at 55 °C for 20 min on a hot plate with continuous stirring | 35.76 ± 6.74 μg/g | The highest astaxanthin amount was found when the 60:20 shrimp:crab ratio was used. | [66] |
| Blue crab (Portunus segnis) | Shells | Conventional extraction, enzymatic extraction, Soxhlet, maceration | 5045 μg/g extract | The highest amount of total carotenoid content was found for combined enzyme-assisted extraction and maceration in hexane/isopropanol (50/50; v/v). | [42] |
| Atlantic shrimp (Pandalus borealis) | Shells | UAE solvent extraction by acetone, hexane/isopropanol 3:2 (v/v), and methanol for 5 min at 25 °C | 270.04, 284.48, and 57.34 mg/g | Hexane/isopropanol extraction resulted in the highest amount of extracted astaxanthin. | [67] |
| Shrimp (species not determined) | Shells | Degradation by Aeromonas hydrophila | 2.14 ± 0.13 μg/ml | The optimized culture media for higher astaxanthin recovery is characterized by the following conditions: pH 7.0, monosodium glutamate 3% (w/v), glucose (1% w/v) and 30 °C. | [68] |
| Brown crab (Cancer pagurus) | Residues | Supercritical fluid extraction (500 bar, 40 °C, 30 min, 50 g/min) | 5.18 µg/g | Optimized conditions yielded a 1.5-fold higher content of astaxanthin. | [69] |
| Pink shrimp (Farfantepenaeus subtilis) | Shrimp waste paste | Extraction using palm olein (90 mL/2.5 g) at 50, 60, and 70 °C | 26.38 µg/g (50 °C), 28.62 µg/g (60 °C), and 29.18 µg/g (70 °C) | Extraction at 70 °C yielded 50.42% astaxanthin. | [70] |
| Shrimp (Litopenaeus vannamei) | Shells | Shrimp shells, dried under vacuum (40 °C and 175 MPa), were extracted by ethanol | 28.9 µg/g | The obtained isolate exhibited high antioxidative activity, no toxic effect up to 160 µg/mL on human fibroblast cells, and anti-tyrosinase (12.2 µg/mL) properties. | [71] |
| Shrimps (Parapenaeopsis sculptili, Metapenaeus lysianassa, Macrobrachium rosenbergii, Metapenaeopsis hardwickii, Penaeus merguiensis, and Penaeus monodon) | Carapace | Extraction using acetone and methanol (7:3 v/v) and high-pressure processing (HPP) (210 MPa, 10 min) | 46.95 µg/mL (conventional) 68.26 µg/mL (HPP) | HPP improved astaxanthin extraction by around 45%. P. monodon yielded the highest astaxanthin with a shorter extraction time. | [72] |
| Shrimp (Procambarus clarkia) | Shells | Extracted using ethanol (1:7) for 20 min at 50 °C using ultrasound (40 kHz) and dried under a vacuum | 43.7 µg/g | Extraction using optimized conditions increased purity by 250 times, exhibiting great application abilities. | [62] |
| Shrimp (species not determined) | Fresh head, cooked head, fresh shell and cooked shell | Extraction by cooking at 90 °C for 15 min | 3.64 mg/g (fresh head), 2.38 mg/g (cooked head), 14.65 mg/g (fresh shell), 11.76 mg/g (cooked shell) | Fresh shells contained the highest amount of astaxanthin, and cooking slightly impacted its content. | [73] |
| Shrimp (Penaeus vannamei Boone) | Shells | HPE using acetone, dichloromethane, and ethanol | Range from 42.3–72.9 μg/g depending on applied pressure and time | HPE resulted in higher extraction yield with improved antioxidant activity. | [74] |
| Shrimp (Litopenaeus vannamei) | Cephalothorax, cuticles, pleopods, and tails | Lipid extraction for 30 min with ethyl acetate (10 g/50 mL) | 7 ± 1 mg/g | Valorization of shrimp byproducts by the production of an extract rich in bioactive compounds, such as astaxanthin, PUFAs, and α-tocopherol. | [75] |
| Blue crab (Callinectes sapidus) | Crab byproducts | Enzymatic hydrolysis with alcalase and bromelain | Range from 12.0–97.7 μg/g residue | Production of chitin and astaxanthin-enriched extract using enzymatic hydrolysis. | [76] |
| Tiger shrimp (Penaeus monodon) | Shrimp waste | Supercritical fluid extraction using carbon dioxide with 15% (v/v) ethanol | 58.50 ± 2.62 µg/g astaxanthin and 12.20 ± 4.16 µg/g free astaxanthin | Use of modeling to determine the best extraction conditions, which were 215.68 bar, 56.88 °C, and 1.89 mL/min for 120 min. | [77] |
| Red (Aristaeomorpha foliacea) and pink shrimp (Parapenaeus longirostris) | Muscle and cephalothorax | Solvent extraction using Bligh and Dyer method | For A. foliacea of total carotenoids: 34.73 ± 0.87 (muscle) and 37.55 ± 0.64 (cephalothorax) % (w/w). For P. longirostris of total carotenoids: 34.32 ± 0.58 (muscle), 49.08 ± 0.82 (cephalothorax) % (w/w). | Analysis showed higher content of PUFAs (mainly omega-3) and high concentrations of carotenoids (astaxanthin followed by lutein). | [78] |
| Activity | Form of Astaxanthin and Its Action | Reference |
|---|---|---|
| Antioxidant | Better activity of isolated astaxanthin from crabs in comparison to the standard compound investigated by scavenging activity against hydrogen peroxide and 2,2-diphenyl-1-picryl hydrazyl (DPPH) radicals, as reducing power and metal-ion-chelating ability. | [94] |
| In vivo antioxidant efficiency on the alcohol-induced oxidative damage in mice of the water-dispersible, astaxanthin-rich nanopowder. | [99] | |
| Improved antioxidant properties of astaxanthin biopolymer nanoparticles in comparison to the free compound tested by in vitro scavenging activity against 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS). | [100] | |
| Higher antioxidant activity of microencapsulated astaxanthin from Phaffia rhodozyma. | [101] | |
| Applied supercritical emulsions extraction technology resulted in encapsulated astaxanthin in ethyl cellulose with good antioxidant activity. | [102] | |
| Effectiveness of astaxanthin in form of nanohydrogels in the neutralization of ROS in vitro. | [103] | |
| Antimicrobial | The extent of ROS involvement in antibacterial activity against S. aureus, B. cereus, P. aeroginosa, and E. coli | [92] |
| High activity of astaxanthin isolate from crabs against E. coli detected using the agar diffusion method. | [94] | |
| Confirmed antagonism of the astaxanthin methanolic isolate from Sphingomonas faeni against common food-borne pathogens. | [96] | |
| Good antimicrobial activity of astaxanthin from crustacean shell byproducts against Escherichia coli, Bacillus, Staphylococcus, and Pseudomonas. | [61] | |
| Good antimicrobial activity of astaxanthin from Penaeus monodon against four bacteria (E. coli, E. aerogenes, S. aureus, and B. subtilis), especially for extracts obtained by high-pressure processing. | [97] | |
| Effectiveness of astaxanthin from H. pluvialis against E. coli, Salmonella typhi, Vibrio cholera, and S. aureus). | [98] | |
| Astaxanthin in bioactive polymers showed significant reduction of bacterial growth and biofilm formation, especially against MRSA. | [104] | |
| The good activity of astaxanthin-alpha tocopherol nanoemulsions through the disruption of the integrity of the bacterial cell membrane detected by MIC, MBC, and disk diffusion methods. | [105] | |
| Astaxanthin from Asian tiger shrimp shell showed good activity in killing and growth inhibition of E. coli, Pseudomonas aeroginosa, Salmonella typhi, S. aureus, and Streptococcus mutans bacteria. | [106] | |
| Anti-inflammatory | Effective in various diseases (diabetes mellitus, Alzheimer’s and Parkinson’s diseases, neuropathic pain, kidney-related diseases, hepatitis, dry eye disease, atopic dermatitis, and inflammatory bowel disease) | [107] |
| The activity is demonstrated by recording suppression of proinflammatory cytokines and inflammatory mediator production in rats with monosodium urate crystal-induced arthritis. | [108] | |
| Astaxanthin alleviated the status of epilepticus-induced hippocampal injury in rats and improved cognitive dysfunction. | [108] | |
| The anti-inflammatory effect of orally administered astaxanthin was confirmed in mice with ovalbumin-induced asthma. | [109] | |
| Cytotoxic, antiproliferative, and anticancer activity | Astaxanthin-alpha tocopherol nanoemulsions showed cytotoxicity as a measure of cell viability of four cell lines (CT26, HeLa, Panc1, and T24) and showed a significant decrease in viability after 1 and 2 days of exposure. | [105] |
| Dose-dependent toxicity and antiproliferative effect of gold nanoparticles synthesized using astaxanthin against human breast cancer cells (MDA-MB-231). | [110] | |
| Microencapsulated astaxanthin showed inhibition of lipid peroxidation and significant cytostatic activity on adipose-derived stem cells. | [111] | |
| Oral treatment of astaxanthin nanoemulsion demonstrated a chemotherapy effect in mice with lung metastatic melanoma by triggering apoptosis. | [112] | |
| Astaxanthin administered intragastrically in mice with PC-3 xenograft prostate tumor significantly inhibited its growth. | [113] | |
| Astaxanthin suppressed the occurrence of N-nitrosomethylbenzylamine-induced esophageal cancer in rats through antioxidant and anti-inflammation capacity increase. | [114] | |
| Significant inhibition of the development of liver cell adenoma and hepatocellular carcinoma in diethylnitrosamine-treated mice by ameliorating serum adiponectin level and improving oxidative stress. | [115] | |
| Effect on subchronic testis injury induced by SnS2 nanoflowers in mice; treatment attenuates testicular ultrastructure alterations and histopathological injury and alleviated testicular inflammation, oxidative stress, apoptosis, and necroptosis. | [116] | |
| Astaxanthin-alpha tocopherol nanoemulsions showed wound healing potential through scratch assay on HeLa, CT26, and T24 cells. | [105] | |
| Hepatoprotective | Astaxanthin-rich nanopowder prepared by nanoencapsulation and freeze-drying showed in vivo antioxidant effect on the alcohol-induced oxidative damage in mice, making the hepatic injury less severe. | [99] |
| Astaxanthin-loaded liposomes provided therapeutic and reparative effects on mice with alcoholic liver fibrosis. | [117] | |
| Astaxanthin encapsulated within liposomes caused a reduction of lipopolysaccharide-induced acute hepatotoxicity in rats. | [118] | |
| Astaxanthin pretreatment reduces the effect of acetaminophen-induced liver injury in mice by reduction of ROS generation, inhibition of oxidative stress, and reduction of apoptosis | [119] | |
| Protection from pancreatic damage and reduces oxidative stress in rats with acute pancreatitis. | [120] | |
| Antidiabetic | Significant decrease of total cholesterol and blood glucose levels and increase of high-density lipoprotein cholesterol levels in rats. | [121] |
| Oral administration of astaxanthin reduced lung damage in rat pups with bronchopulmonary dysplasia (induced by hyperoxia and lipopolysaccharide). | [122] | |
| Eye health | Protective effect against dry eye disease in vitro on human corneal epithelial cells cultures and in vivo in mice. | [123] |
| Skin health | Protective effects on age-related skin deterioration and environmentally induced damage. | [124] |
| Liposomal astaxanthin showed antidermatotic effects in mice with phthalic anhydride-induced atopic dermatitis. | [125] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šimat, V.; Rathod, N.B.; Čagalj, M.; Hamed, I.; Generalić Mekinić, I. Astaxanthin from Crustaceans and Their Byproducts: A Bioactive Metabolite Candidate for Therapeutic Application. Mar. Drugs 2022, 20, 206. https://doi.org/10.3390/md20030206
Šimat V, Rathod NB, Čagalj M, Hamed I, Generalić Mekinić I. Astaxanthin from Crustaceans and Their Byproducts: A Bioactive Metabolite Candidate for Therapeutic Application. Marine Drugs. 2022; 20(3):206. https://doi.org/10.3390/md20030206
Chicago/Turabian StyleŠimat, Vida, Nikheel Bhojraj Rathod, Martina Čagalj, Imen Hamed, and Ivana Generalić Mekinić. 2022. "Astaxanthin from Crustaceans and Their Byproducts: A Bioactive Metabolite Candidate for Therapeutic Application" Marine Drugs 20, no. 3: 206. https://doi.org/10.3390/md20030206
APA StyleŠimat, V., Rathod, N. B., Čagalj, M., Hamed, I., & Generalić Mekinić, I. (2022). Astaxanthin from Crustaceans and Their Byproducts: A Bioactive Metabolite Candidate for Therapeutic Application. Marine Drugs, 20(3), 206. https://doi.org/10.3390/md20030206