An Efficient and Quick Analytical Method for the Quantification of an Algal Alkaloid Caulerpin Showed In-Vitro Anticancer Activity against Colorectal Cancer
Abstract
1. Introduction
2. Results
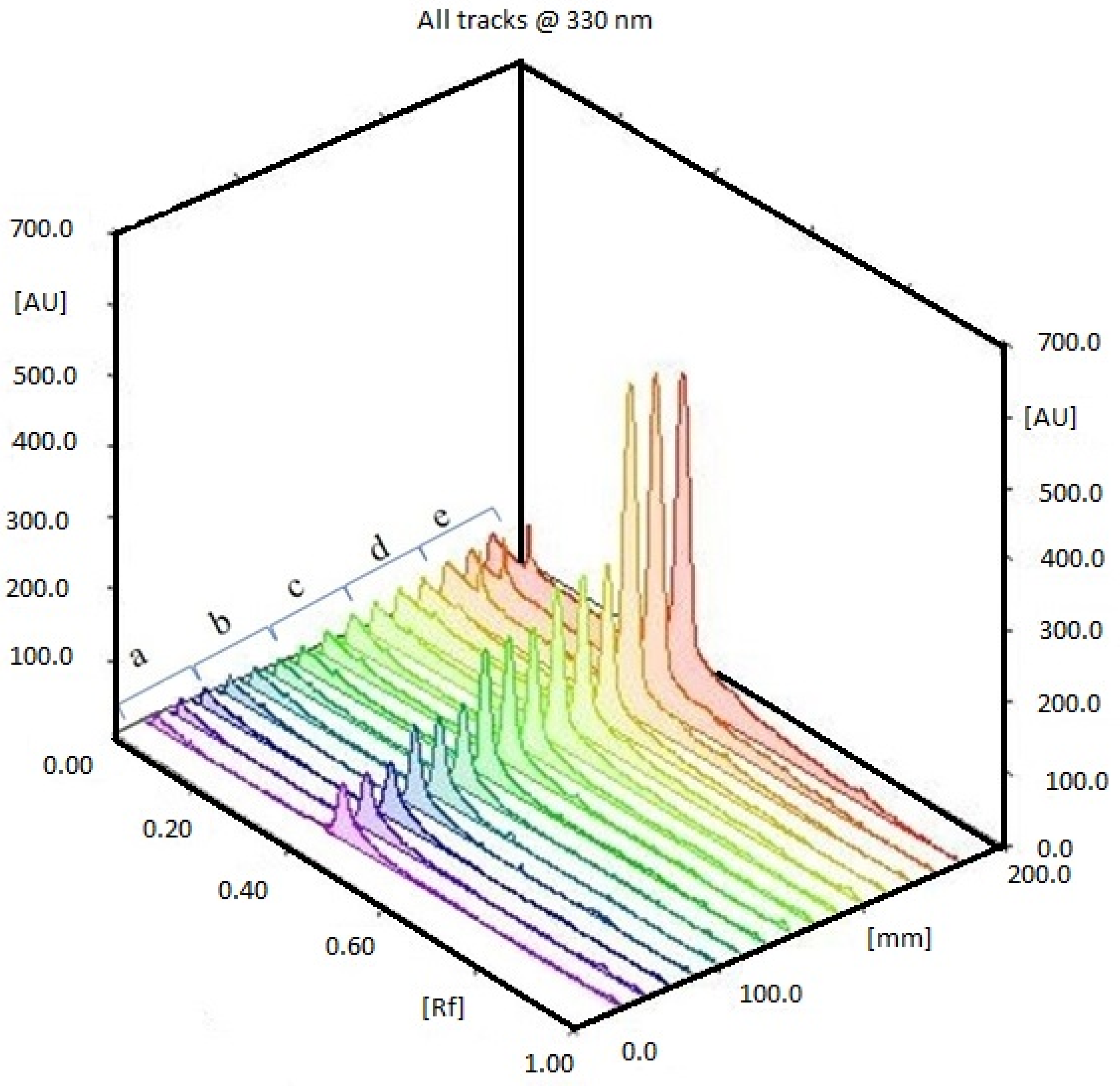
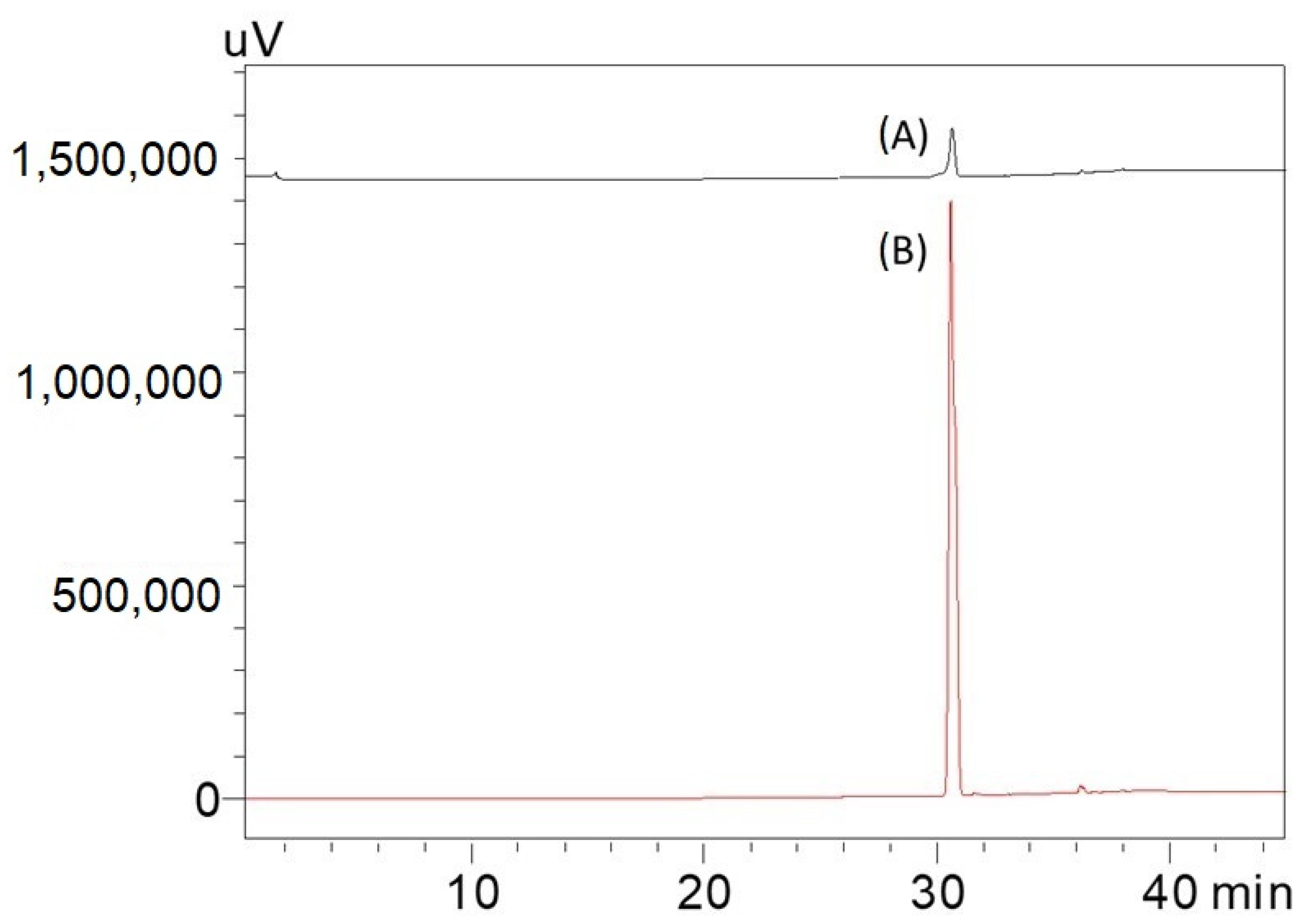
2.1. HPTLC Validation of the Method for Quantification of Caulerpin
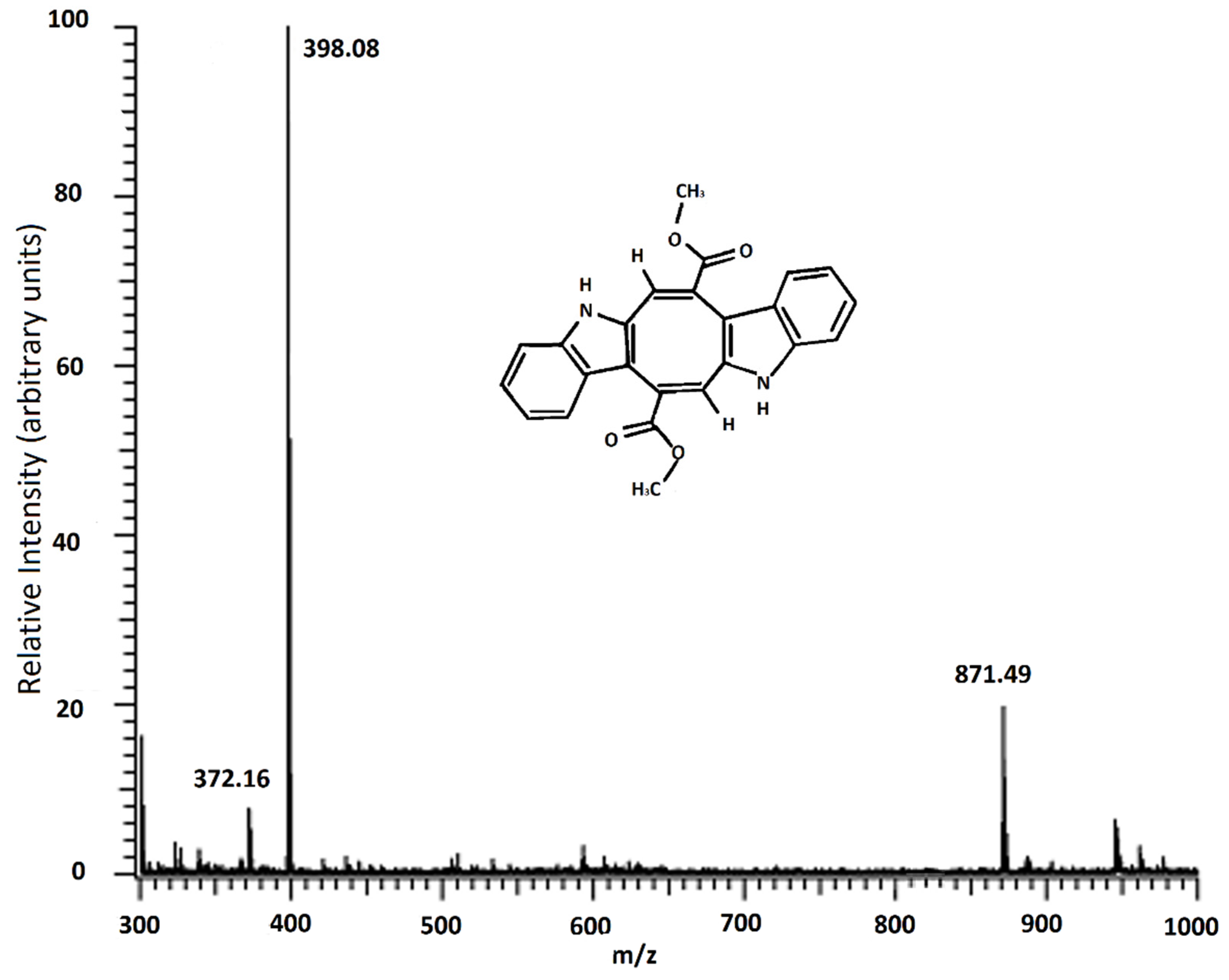
2.2. MALDI-TOF/MS Result of Caulerpin
2.3. The Effect of Caulerpin on Colorectal Cancer Cell Line Viability
2.4. Inhibition of Migration and Invasion and Inducing Apoptosis of HCT-116 and HT-29 Cell Lines by Caulerpin
3. Discussion
4. Materials and Methods
4.1. Instrumentation and Chemicals
4.2. Caulerpa Sampling
4.3. Extraction of Caulerpa spp. and Isolation of Caulerpin
4.4. Optimization of HPTLC Analysis
4.5. Method Validation of Caulerpin
4.6. Cell Lines and Reagents
4.7. Cell Culture
4.8. Cytotoxicity Assay
4.9. Colony Formation Assay
4.10. Migration Assay
4.11. Invasion Assay
4.12. Apoptosis Analysis
4.13. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Diagne, C.; Leroy, B.; Gozlan, R.E.; Vaissière, A.C.; Assailly, C.; Nuninger, L.; Roiz, D.; Jourdain, F.; Jarić, I.; Courchamp, F. InvaCost, a Public Database of the Economic Costs of Biological Invasions Worldwide. Sci. Data 2020, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Diagne, C.; Leroy, B.; Vaissière, A.C.; Gozlan, R.E.; Roiz, D.; Jarić, I.; Salles, J.M.; Bradshaw, C.J.A.; Courchamp, F. High and Rising Economic Costs of Biological Invasions Worldwide. Nature 2021, 592, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Schaffelke, B.; Hewitt, C.L. Impacts of Introduced Seaweeds. Bot. Mar. 2008, 50, 397–417. [Google Scholar] [CrossRef]
- Pierucci, A.; De La Fuente, G.; Cannas, R.; Chiantore, M. A New Record of the Invasive Seaweed Caulerpa cylindracea Sonder in the South Adriatic Sea. Heliyon 2019, 5, e02449. [Google Scholar] [CrossRef]
- Mehra, R.; Bhushan, S.; Bast, F.; Singh, S. Marine Macroalga Caulerpa: Role of Its Metabolites in Modulating Cancer Signaling. Mol. Biol. Rep. 2019, 46, 3545–3555. [Google Scholar] [CrossRef] [PubMed]
- De Souza, É.T.; De Lira, D.P.; De Queiroz, A.C.; Da Silva, D.J.C.; De Aquino, A.B.; Campessato Mella, E.A.; Lorenzo, V.P.; De Miranda, G.E.C.; De Araújo-Júnior, J.X.; De Oliveira Chaves, M.C.; et al. The Antinociceptive and Anti-Inflammatory Activities of Caulerpin, a Bisindole Alkaloid Isolated from Seaweeds of the Genus Caulerpa. Mar. Drugs 2009, 7, 689–704. [Google Scholar] [CrossRef]
- Nagappan, T.; Vairappan, C.S. Nutritional and Bioactive Properties of Three Edible Species of Green Algae, Genus Caulerpa (Caulerpaceae). J. Appl. Phycol. 2014, 26, 1019–1027. [Google Scholar] [CrossRef]
- Lorenzo, V.P.; Filho, J.M.B.; Scotti, L.; Scotti, M.T. Combined Structure- and Ligand-Based Virtual Screening to Evaluate Caulerpin Analogs with Potential Inhibitory Activity against Monoamine Oxidase B. Rev. Bras. Farm. 2015, 25, 690–697. [Google Scholar] [CrossRef][Green Version]
- Canché Chay, C.I.; Cansino, R.G.; Espitia Pinzón, C.I.; Torres-Ochoa, R.O.; Martínez, R. Synthesis and Anti-Tuberculosis Activity of the Marine Natural Product Caulerpin and Its Analogues. Mar. Drugs 2014, 12, 1757–1772. [Google Scholar] [CrossRef]
- Cavalcante-Silva, L.H.A.; Falcão, M.A.P.; Vieira, A.C.S.; Viana, M.D.M.; De Araújo-Júnior, J.X.; Sousa, J.C.F.; Da Silva, T.M.S.; Barbosa-Filho, J.M.; Noël, F.; De Miranda, G.E.C.; et al. Assessment of Mechanisms Involved in Antinociception Produced by the Alkaloid Caulerpine. Molecules 2014, 19, 14699–14709. [Google Scholar] [CrossRef]
- Pinto, A.M.V.; Leite, J.P.G.; Ferreira, W.J.; Cavalcanti, D.N.; Villaça, R.C.; Giongo, V.; Teixeira, V.L.; Paixão, I.C.N.d.P. Marine Natural Seaweed Products as Potential Antiviral Drugs against Bovine Viral Diarrhea Virus. Rev. Bras. Farm. 2012, 22, 813–817. [Google Scholar] [CrossRef]
- Macedo, N.R.P.V.; Ribeiro, M.S.; Villaça, R.C.; Ferreira, W.; Pinto, A.M.; Teixeira, V.L.; Cirne-Santos, C.; Paixão, I.C.N.P.; Giongo, V. Caulerpin as a Potential Antiviral Drug against Herpes Simplex Virus Type 1. Rev. Bras. Farm. 2012, 22, 861–867. [Google Scholar] [CrossRef]
- Zhang, M.-Z.; Chen, Q.; Yang, G.-F. A Review on Recent Developments of Indole-Containing Antiviral Agents. Eur. J. Med. Chem. 2015, 89, 421–441. [Google Scholar] [CrossRef] [PubMed]
- Esteves, P.O.; de Oliveira, M.C.; de Souza Barros, C.; Cirne-Santos, C.C.; Laneuvlille, V.T.; Palmer Paixão, I.C. Antiviral Effect of Caulerpin Against Chikungunya. Nat. Prod. Commun. 2019, 14, 1–6. [Google Scholar] [CrossRef]
- Ahmed, S.A.; Abdelrheem, D.A.; El-Mageed, H.R.A.; Mohamed, H.S.; Rahman, A.A.; Elsayed, K.N.M.; Ahmed, S.A. Destabilizing the Structural Integrity of COVID-19 by Caulerpin and Its Derivatives along with Some Antiviral Drugs: An in Silico Approaches for a Combination Therapy. Struct. Chem. 2020, 31, 2391–2412. [Google Scholar] [CrossRef]
- Abdelrheem, D.A.; Ahmed, S.A.; Abd El-Mageed, H.R.; Mohamed, H.S.; Rahman, A.A.; Elsayed, K.N.M.; Ahmed, S.A. The Inhibitory Effect of Some Natural Bioactive Compounds against SARS-CoV-2 Main Protease: Insights from Molecular Docking Analysis and Molecular Dynamic Simulation. J. Environ. Sci. Health A Toxic Hazard. Subst. Environ. Eng. 2020, 55, 1373–1386. [Google Scholar] [CrossRef] [PubMed]
- Çavaş, L.; Dag, C.; Carmena-Barreño, M.; Martínez-cortés, C.; Pedro, J.; Pérez-Sánchez, H. Secondary Metabolites from Caulerpa cylindracea (Sonder) Could Be Alternative Natural Antiviral Compounds for COVID-19: A Further in Silico Proof. Biol. Med. Chem. 2020. [Google Scholar] [CrossRef]
- Vitale, R.M.; D’aniello, E.; Gorbi, S.; Martella, A.; Silvestri, C.; Giuliani, M.E.; Fellous, T.; Gentile, A.; Carbone, M.; Cutignano, A.; et al. Fishing for Targets of Alien Metabolites: A Novel Peroxisome Proliferator-Activated Receptor (PPAR) Agonist from a Marine Pest. Mar. Drugs 2018, 16, 431. [Google Scholar] [CrossRef]
- Felline, S.; Mollo, E.; Cutignano, A.; Grauso, L.; Andaloro, F.; Castriota, L.; Consoli, P.; Falautano, M.; Sinopoli, M.; Terlizzi, A. Preliminary Observations of Caulerpin Accumulation from the Invasive Caulerpa cylindracea in Native Mediterranean Fish Species. Aquat. Biol. 2017, 26, 27–31. [Google Scholar] [CrossRef]
- Gorbi, S.; Giuliani, M.E.; Pittura, L.; d’Errico, G.; Terlizzi, A.; Felline, S.; Grauso, L.; Mollo, E.; Cutignano, A.; Regoli, F. Could Molecular Effects of Caulerpa racemosa Metabolites Modulate the Impact on Fish Populations of Diplodus Sargus? Mar. Environ. Res. 2014, 96, 2–11. [Google Scholar] [CrossRef]
- Turhan, S.; Cavas, L. The Threat on Your Plate: Do We Just Eat Sarpa Salpa or More? Reg. Stud. Mar. Sci. 2019, 29, 100697. [Google Scholar] [CrossRef]
- Ristivojević, P.; Trifković, J.; Andrić, F.; Milojković-Opsenica, D. Recent Trends in Image Evaluation of HPTLC Chromatograms. J. Liq. Chromatogr. Relat. Technol. 2020, 43, 291–299. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Lenos, K.J.; Bach, S.; Ferreira Moreno, L.; ten Hoorn, S.; Sluiter, N.R.; Bootsma, S.; Vieira Braga, F.A.; Nijman, L.E.; van den Bosch, T.; Miedema, D.M.; et al. Molecular Characterization of Colorectal Cancer Related Peritoneal Metastatic Disease. Nat. Commun. 2022, 13, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sawicki, T.; Ruszkowska, M.; Danielewicz, A. A Review of Colorectal Cancer in Terms of Epidemiology, Risk Factors, Development, Symptoms and Diagnosis. Cancers 2021, 13, 2025. [Google Scholar] [CrossRef] [PubMed]
- Rejhová, A.; Opattová, A.; Čumová, A.; Slíva, D.; Vodička, P. Natural Compounds and Combination Therapy in Colorectal Cancer Treatment. Eur. J. Med. Chem. 2018, 144, 582–594. [Google Scholar] [CrossRef] [PubMed]
- Nowruzi, B.; Jokela, J. Identification of Four Different Chlorophyll Allomers of Nostoc Sp. by Liquid Identification of Four Different Chlorophyll Allomers of Nostoc Sp. by Liquid Chromatography-Mass Spectrometer. Int. J. Plant Stud. 2019, 2, 1–4. [Google Scholar]
- Km, G. Significant Role of Soxhlet Extraction Process in Phytochemical. Mintage J. Pharm. Med. Sci. 2019, 7, 43–47. [Google Scholar]
- Felline, S.; Mollo, E.; Ferramosca, A.; Zara, V.; Regoli, F.; Gorbi, S.; Terlizzi, A. Can a Marine Pest Reduce the Nutritional Value of Mediterranean Fish Flesh? Mar. Biol. 2014, 161, 1275–1283. [Google Scholar] [CrossRef]
- Terlizzi, A.; Felline, S.; Lionetto, M.G.; Caricato, R.; Perfetti, V.; Cutignano, A.; Mollo, E. Detrimental Physiological Effects of the Invasive Alga Caulerpa racemosa on the Mediterranean White Seabream Diplodus Sargus. Aquat. Biol. 2011, 12, 109–117. [Google Scholar] [CrossRef]
- Mao, S.C.; Guo, Y.W.; Shen, X. Two Novel Aromatic Valerenane-Type Sesquiterpenes from the Chinese Green Alga Caulerpa Taxifolia. Bioorg. Med. Chem. Lett. 2006, 16, 2947–2950. [Google Scholar] [CrossRef] [PubMed]
- Lucena, A.M.M.; Souza, C.R.M.; Jales, J.T.; Guedes, P.M.M.; De Miranda, G.E.C.; de Moura, A.M.A.; Araújo-Júnior, J.X.; Nascimento, G.J.; Scortecci, K.C.; Santos, B.V.O.; et al. The Bisindole Alkaloid Caulerpin, from Seaweeds of the Genus Caulerpa, Attenuated Colon Damage in Murine Colitis Model. Mar. Drugs 2018, 16, 318. [Google Scholar] [CrossRef] [PubMed]
- Alqarni, M.H.; Foudah, A.I.; Alam, A.; Salkini, M.A.; Alam, P.; Yusufoglu, H.S. Novel HPTLC-Densitometric Method for Concurrent Quantification of Linalool and Thymol in Essential Oils. Arab. J. Chem. 2021, 14, 102916. [Google Scholar] [CrossRef]
- Attala, K.; Eissa, M.S.; El-Henawee, M.M.; Abd El-Hay, S.S. Application of Quality by Design Approach for HPTLC Simultaneous Determination of Amlodipine and Celecoxib in Presence of Process-Related Impurity. Microchem. J. 2021, 162, 105857. [Google Scholar] [CrossRef]
- Chewchinda, S.; Kongkiatpaiboon, S. A Validated HPTLC Method for Quantitative Analysis of Morin in Maclura Cochinchinensis Heartwood. Chin. Herb. Med. 2020, 12, 200–203. [Google Scholar] [CrossRef]
- Golfakhrabadi, F.; Khaledi, M.; Nazemi, M.; Safdarian, M. Isolation, Identification, and HPTLC Quantification of Dehydrodeoxycholic Acid from Persian Gulf Sponges. J. Pharm. Biomed. Anal. 2021, 197, 113962. [Google Scholar] [CrossRef]
- Gupta, S.; Shanker, K.; Srivastava, S.K. HPTLC Method for the Simultaneous Determination of Four Indole Alkaloids in Rauwolfia Tetraphylla: A Study of Organic/Green Solvent and Continuous/Pulse Sonication. J. Pharm. Biomed. Anal. 2012, 66, 33–39. [Google Scholar] [CrossRef]
- Katakam, S.; Sharma, P.; Anandjiwala, S.; Sharma, S.; Shrivastava, N. Investigation on Apposite Chemical Marker for Quality Control of Tephrosia Purpurea (L.) Pers. by Means of HPTLC-Chemometric Analysis. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2019, 1110–1111, 81–86. [Google Scholar] [CrossRef]
- Nazir, R.; Kumar, V.; Dey, A.; Pandey, D.K. HPTLC Quantification of Diosgenin in Dioscorea Deltoidea: Evaluation of Extraction Efficacy, Organ Selection, Drying Method and Seasonal Variation. S. Afr. J. Bot. 2021, 138, 386–393. [Google Scholar] [CrossRef]
- Romero Rocamora, C.; Ramasamy, K.; Meng Lim, S.; Majeed, A.B.A.; Agatonovic-Kustrin, S. HPTLC Based Approach for Bioassay-Guided Evaluation of Antidiabetic and Neuroprotective Effects of Eight Essential Oils of the Lamiaceae Family Plants. J. Pharm. Biomed. Anal. 2020, 178, 112909. [Google Scholar] [CrossRef]
- Tomar, V.; Beuerle, T.; Sircar, D. A Validated HPTLC Method for the Simultaneous Quantifications of Three Phenolic Acids and Three Withanolides from Withania Somnifera Plants and Its Herbal Products. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2019, 1124, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Yap, W.F.; Tay, V.; Tan, S.H.; Yow, Y.Y.; Chew, J. Decoding Antioxidant and Antibacterial Potentials of Malaysian Green Seaweeds: Caulerpa Racemosa and Caulerpa Lentillifera. Antibiotics 2019, 8, 152. [Google Scholar] [CrossRef] [PubMed]
- Majumder, M.; Debnath, S.; Gajbhiye, R.L.; Saikia, R.; Gogoi, B.; Samanta, S.K.; Das, D.K.; Biswas, K.; Jaisankar, P.; Mukhopadhyay, R. Ricinus Communis L. Fruit Extract Inhibits Migration/Invasion, Induces Apoptosis in Breast Cancer Cells and Arrests Tumor Progression In Vivo. Sci. Rep. 2019, 9, 14493. [Google Scholar] [CrossRef] [PubMed]
- Rocha, F.D.; Soares, A.R.; Houghton, P.J.; Pereira, R.C.; Kaplan, M.A.C.; Teixeira, V.L. Potential Cytotoxic Activity of Some Brazilian Seaweeds on Human Melanoma Cells. Phytother. Res. 2007, 21, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Morgan, J.B.; Coothankandaswamy, V.; Liu, R.; Jekabsons, M.B.; Mahdi, F.; Nagle, D.G.; Zhou, Y.D. The Caulerpa Pigment Caulerpin Inhibits HIF-1 Activation and Mitochondrial Respiration. J. Nat. Prod. 2009, 72, 2104–2109. [Google Scholar] [CrossRef]
- Movahhedin, N.; Barar, J.; Azad, F.F.; Barzegari, A.; Nazemiyeh, H. Phytochemistry and Biologic Activities of Caulerpa Peltata Native to Oman Sea. Iran. J. Pharm. Res. 2014, 13, 515–521. [Google Scholar]
- Ferramosca, A.; Conte, A.; Guerra, F.; Felline, S.; Rimoli, M.G.; Mollo, E.; Zara, V.; Terlizzi, A. Metabolites from Invasive Pests Inhibit Mitochondrial Complex II: A Potential Strategy for the Treatment of Human Ovarian Carcinoma? Biochem. Biophys. Res. Commun. 2016, 473, 1133–1138. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, H.; Dong, M.; Wu, Z.; Shen, Z.; Xie, Y.; Kong, Z.; Dai, X.; Xu, B. Metabolic Reprogramming and AMPKα1 Pathway Activation by Caulerpin in Colorectal Cancer Cells. Int. J. Oncol. 2017, 50, 161–172. [Google Scholar] [CrossRef]
- Präbst, K.; Engelhardt, H.; Ringgeler, S.; Hübner, H. Basic Colorimetric Proliferation Assays: MTT, WST, and Resazurin. In Cell viability assays; Humana Press: New York, NY, USA, 2017; Volume 1601, pp. 1–17. ISBN 978-1-4939-6959-3. [Google Scholar]
- Cavas, L.; Donut, N.; Mert, N. Artificial Neural Network Modeling of Diuron and Irgarol-Based HPLC Data and Their Levels from the Seawaters in Izmir, Turkey. J. Liq. Chromatogr. Relat. Technol. 2016, 39, 87–95. [Google Scholar] [CrossRef]
- Harron, D.W.G. Technical Requirements for Registration of Pharmaceuticals for Human Use: The ICH Process. Textb. Pharm. Med. 2013, 1994, 447–460. [Google Scholar] [CrossRef]
- Buachan, P.; Chularojmontri, L.; Wattanapitayakul, S.K. Selected Activities of Citrus maxima Merr. Fruits on Human Endothelial Cells: Enhancing Cell Migration and Delaying Cellular Aging. Nutrients 2014, 6, 1618–1634. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Tan, T.; Mao, Z.G.; Lei, N.; Wang, Z.M.; Hu, B.; Chen, Z.Y.; She, Z.G.; Zhu, Y.H.; Wang, H.J. The Marine Metabolite SZ-685C Induces Apoptosis in Primary Human Nonfunctioning Pituitary Adenoma Cells by Inhibition of the Akt Pathway In Vitro. Mar. Drugs 2015, 13, 1569–1580. [Google Scholar] [CrossRef] [PubMed]




| Concentration (ng µL−1) | Concentration (µg g−1) | |
|---|---|---|
| CC48 | 453.46 ± 21.11 | 108.83 ± 5.07 |
| CC72 | 402.04 ± 18.93 | 96.49 ± 4.54 |
| CL48 | n.d. | n.d. |
| CL72 | 35.16 ± 16.08 (below the LOQ) | 8.44 ± 3.86 (below the LOQ) |
| CCM | 562.14 ± 26.43 | 112.43 ± 5.29 |
| CLM | 42.09 ± 8.21 (below the LOQ) | 8.42 ± 1.64 (below the LOQ) |
| Validation Parameters | Caulerpin (330 nm) |
|---|---|
| Calibration equation | y = 28.872x + 3233.4 |
| Linearity (R2) | 0.9635 |
| Slope | 28.872 |
| Shift | 3233.4 |
| Range (ng band−1) | 25–500 |
| LOD (ng band−1) | 20.47 |
| LOQ (ng band−1) | 67.56 |
| Recovery (%) | 89.81 |
| RSD (%) | 19.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mert-Ozupek, N.; Calibasi-Kocal, G.; Olgun, N.; Basbinar, Y.; Cavas, L.; Ellidokuz, H. An Efficient and Quick Analytical Method for the Quantification of an Algal Alkaloid Caulerpin Showed In-Vitro Anticancer Activity against Colorectal Cancer. Mar. Drugs 2022, 20, 757. https://doi.org/10.3390/md20120757
Mert-Ozupek N, Calibasi-Kocal G, Olgun N, Basbinar Y, Cavas L, Ellidokuz H. An Efficient and Quick Analytical Method for the Quantification of an Algal Alkaloid Caulerpin Showed In-Vitro Anticancer Activity against Colorectal Cancer. Marine Drugs. 2022; 20(12):757. https://doi.org/10.3390/md20120757
Chicago/Turabian StyleMert-Ozupek, Nazli, Gizem Calibasi-Kocal, Nur Olgun, Yasemin Basbinar, Levent Cavas, and Hulya Ellidokuz. 2022. "An Efficient and Quick Analytical Method for the Quantification of an Algal Alkaloid Caulerpin Showed In-Vitro Anticancer Activity against Colorectal Cancer" Marine Drugs 20, no. 12: 757. https://doi.org/10.3390/md20120757
APA StyleMert-Ozupek, N., Calibasi-Kocal, G., Olgun, N., Basbinar, Y., Cavas, L., & Ellidokuz, H. (2022). An Efficient and Quick Analytical Method for the Quantification of an Algal Alkaloid Caulerpin Showed In-Vitro Anticancer Activity against Colorectal Cancer. Marine Drugs, 20(12), 757. https://doi.org/10.3390/md20120757