Marine-Inspired Drugs and Biomaterials in the Perspective of Pancreatic Cancer Therapies
Abstract
1. Introduction
2. Pancreatic Cancer: A Silent Killer
2.1. Current Treatments and Challenges
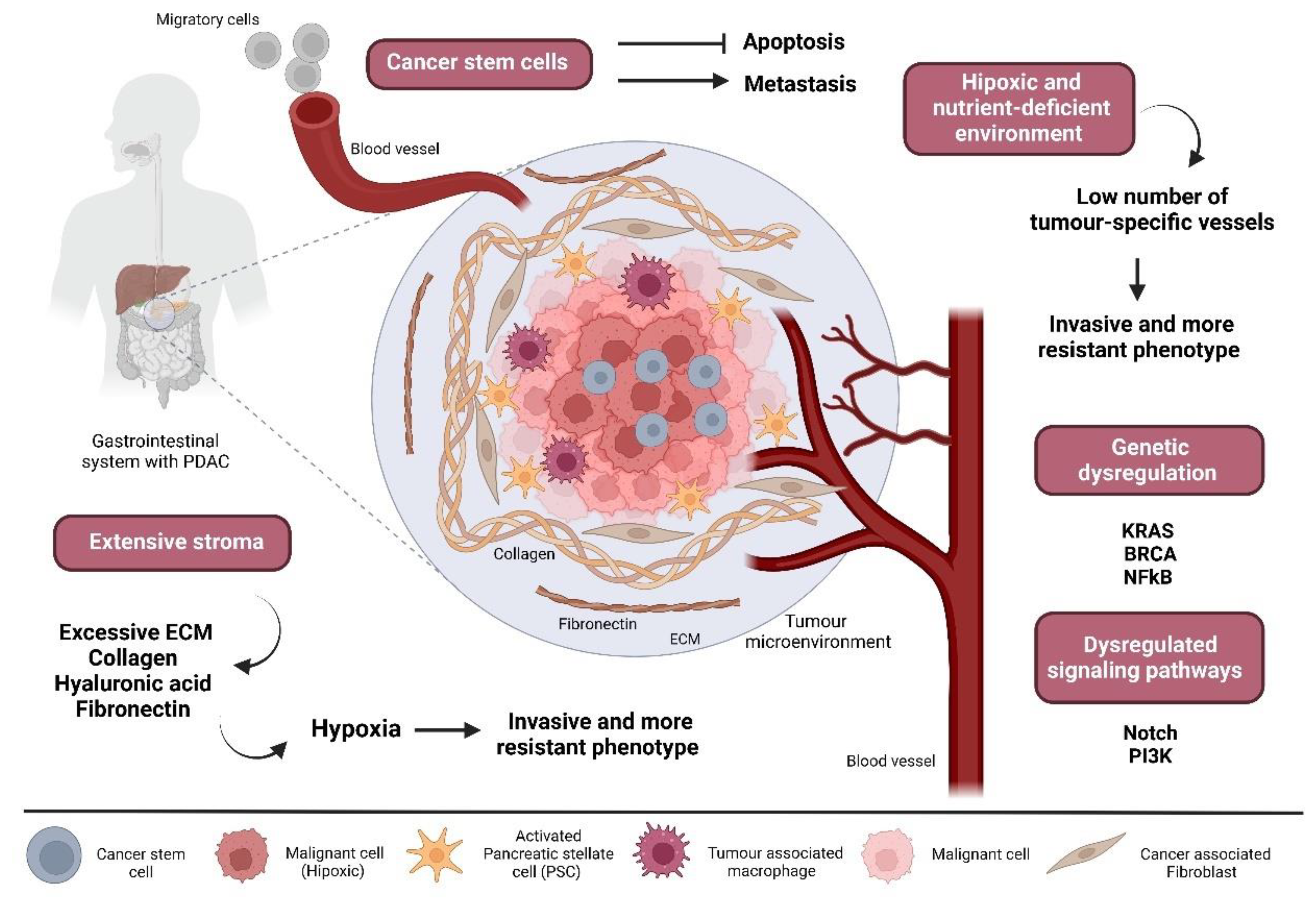
2.2. The Pancreatic Cancer Ecosystem as a Potential Therapeutic Target against Chemoresistance
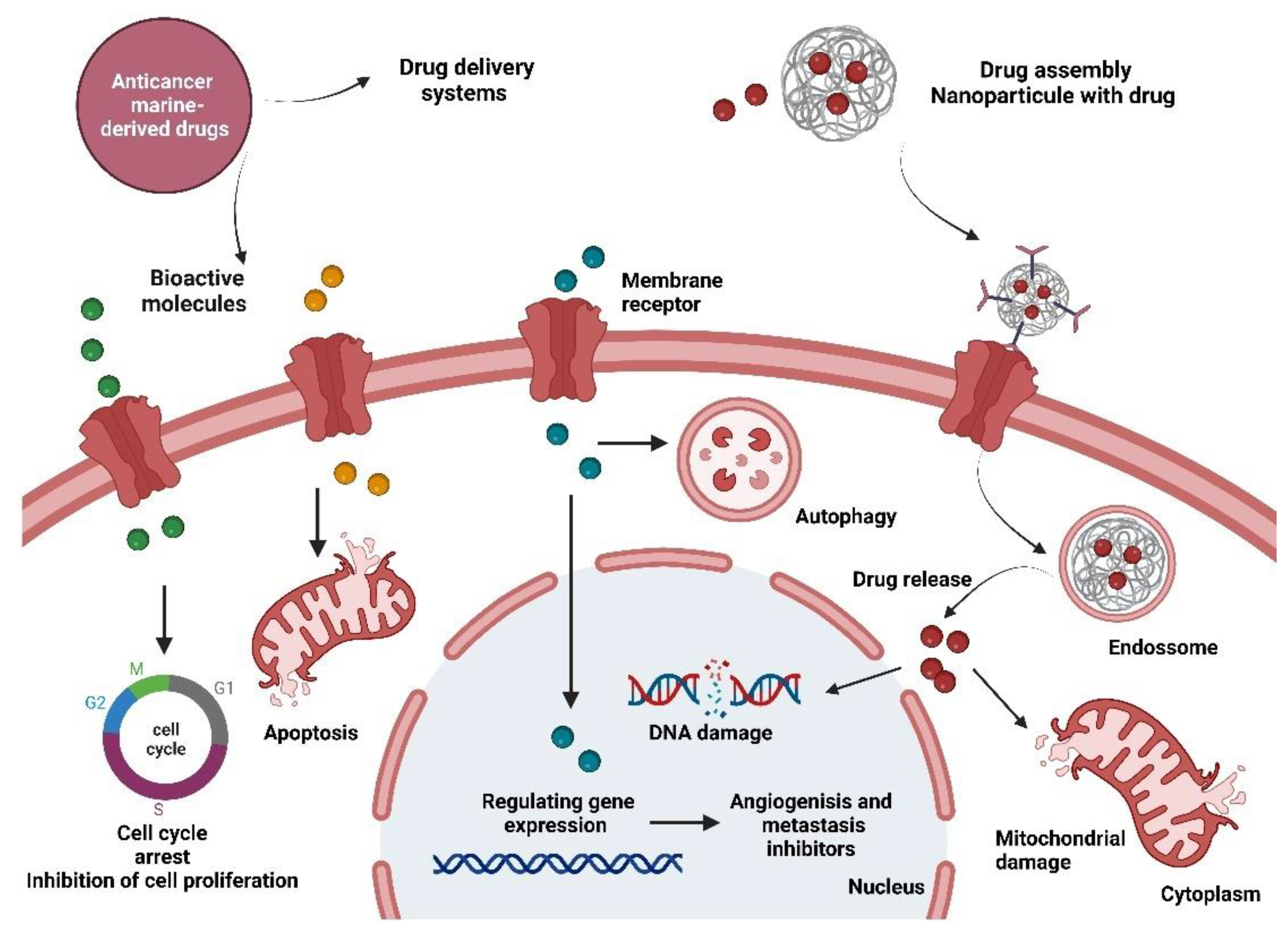
3. Marine Organisms and Anticancer Drugs for Pancreatic Cancer
3.1. Marine Sponges
3.2. Marine Molluscs
3.3. Bryozoans
3.4. Macroalgae
3.4.1. Fucoidan
3.4.2. Polyphenols
3.5. Marine Fungi
3.6. Marine Bacteria
3.7. Clinical Trials with Marine Natural Products Addressing Pancreatic Cancer
4. Marine-Inspired Materials for Drug Delivery in Pancreatic Cancer
4.1. Chitosan-Based Drug Delivery Systems
4.2. Fucoidan-based Drug Delivery Systems
4.3. Alginate-based Drug Delivery Systems
5. Looking Forward: The Future of Marine-Inspired Drugs and Biomaterials against Pancreatic Cancer
5.1. Biomarkers and Precise Medicine
5.2. Three-dimensional Cancer Models
5.3. Immunotherapy
6. Conclusions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Singh, R.D.; Mody, S.K.; Patel, H.B.; Devi, S.; Sarvaiya, V.N.; Patel, H.A.; Patel, B.R. Antimicrobial drug discovery: Evident shifting from terrestrial to marine micro-organisms. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 2322–2327. [Google Scholar] [CrossRef]
- Amin, A.R.; Kucuk, O.; Khuri, F.R.; Shin, D.M. Perspectives for cancer prevention with natural compounds. J. Clin. Oncol. 2009, 27, 2712. [Google Scholar] [CrossRef] [PubMed]
- Calcabrini, C.; Catanzaro, E.; Bishayee, A.; Turrini, E.; Fimognari, C. Marine sponge natural products with anticancer potential: An updated review. Mar. Drugs 2017, 15, 310. [Google Scholar] [CrossRef] [PubMed]
- Dagogo-Jack, I.; Shaw, A.T. Tumour heterogeneity and resistance to cancer therapies. Nat. Rev. Clin. Oncol. 2018, 15, 8194. [Google Scholar] [CrossRef] [PubMed]
- Joyce, J.A. Therapeutic targeting of the tumor microenvironment. Cancer Cell 2005, 7, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Chan, K.; Qi, Y.; Lu, L.; Ning, F.; Wu, M.; Du, J. Participation of CCL1 in snail-positive fibroblasts in colorectal cancer contribute to 5-fluorouracil/paclitaxel chemoresistance. Cancer Res. Treat. Off. J. Korean Cancer Assoc. 2018, 50, 894. [Google Scholar] [CrossRef]
- Surveillance, Epidemiology, and End Results Program (based on 2008–2012 Cases and Deaths). Available online: http://seer.cancer.gov (accessed on 24 March 2020).
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics, 2012. CA A Cancer J. Clin. 2012, 62, 10–29. [Google Scholar] [CrossRef]
- Rawla, P.; Sunkara, T.; Gaduputi, V. Epidemiology of pancreatic cancer: Global trends, etiology and risk factors. World J. Oncol. 2019, 10, 10. [Google Scholar] [CrossRef]
- Raimondi, S.; Maisonneuve, P.; Lowenfels, A.B. Epidemiology of pancreatic cancer: An overview. Nat. Rev. Gastroenterol. Hepatol. 2009, 6, 699. [Google Scholar] [CrossRef]
- Beger, H.G.; Rau, B.; Gansauge, F.; Poch, B.; Link, K.H. Treatment of pancreatic cancer: Challenge of the facts. World J. Surg. 2003, 27, 1075–1084. [Google Scholar] [CrossRef]
- Wong, H.H.; Lemoine, N.R. Pancreatic cancer: Molecular pathogenesis and new therapeutic targets. Nat. Rev. Gastroenterol. Hepatol. 2009, 6, 412. [Google Scholar] [CrossRef] [PubMed]
- Fink, D.M.; Steele, M.M.; Hollingsworth, M.A. The lymphatic system and pancreatic cancer. Cancer Lett. 2016, 381, 217–236. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Bennouna, J. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef]
- Garrido-Laguna, I.; Hidalgo, M. Pancreatic cancer: From state-of-the-art treatments to promising novel therapies. Nat. Rev. Clin. Oncol. 2015, 12, 319. [Google Scholar] [CrossRef]
- Tas, F.; Sen, F.; Keskin, S.; Kilic, L.; Yildiz, I. Prognostic factors in metastatic pancreatic cancer: Older patients are associated with reduced overall survival. Mol. Clin. Oncol. 2013, 1, 788–792. [Google Scholar] [CrossRef]
- Burris, H.A.; Moore, M.J.; Andersen, J.; Green, M.R.; Rothenberg, M.L.; Modiano, M.R.; Tarassoff, P. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997, 15, 2403–2413. [Google Scholar] [CrossRef] [PubMed]
- Fiorini, C.; Cordani, M.; Padroni, C.; Blandino, G.; Di Agostino, S.; Donadelli, M. Mutant p53 stimulates chemoresistance of pancreatic adenocarcinoma cells to gemcitabine. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2015, 1853, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Weizman, N.; Krelin, Y.; Shabtay-Orbach, A.; Amit, M.; Binenbaum, Y.; Wong, R.J.; Gil, Z. Macrophages mediate gemcitabine resistance of pancreatic adenocarcinoma by upregulating cytidine deaminase. Oncogene 2014, 33, 3812–3819. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.P.; Gallick, G.E. Gemcitabine resistance in pancreatic cancer: Picking the key players. Clin. Cancer Res. 2008, 2008, 1284–1285. [Google Scholar] [CrossRef]
- Glassman, D.C.; Palmaira, R.L.; Covington, C.M.; Desai, A.M.; Ku, G.Y.; Li, J.; Kenneth, H.Y. Nanoliposomal irinotecan with fluorouracil for the treatment of advanced pancreatic cancer, a single institution experience. BMC Cancer 2018, 18, 693. [Google Scholar] [CrossRef]
- Haddock, M.G.; Swaminathan, R.; Foster, N.R.; Hauge, M.D.; Martenson, J.A.; Camoriano, J.K.; Alberts, S.R. Gemcitabine, cisplatin, and radiotherapy for patients with locally advanced pancreatic adenocarcinoma: Results of the North Central Cancer Treatment Group Phase II Study N9942. J. Clin. Oncol. 2007, 25, 2567–2572. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Merl, M.Y.; Chabot, J.; Saif, M.W. Updates of adjuvant therapy in pancreatic cancer: Where are we and where are we going? JOP J. Pancreas 2010, 11, 310–312. [Google Scholar]
- Amrutkar, M.; Gladhaug, I.P. Pancreatic cancer chemoresistance to gemcitabine. Cancers 2017, 9, 157. [Google Scholar] [CrossRef]
- Swayden, M.; Iovanna, J.; Soubeyran, P. Pancreatic cancer chemo-resistance is driven by tumor phenotype rather than tumor genotype. Heliyon 2018, 4, e01055. [Google Scholar] [CrossRef]
- Rucki, A.A.; Zheng, L. Pancreatic cancer stroma: Understanding biology leads to new therapeutic strategies. World J. Gastroenterol. WJG 2014, 20, 2237. [Google Scholar] [CrossRef] [PubMed]
- Provenzano, P.P.; Cuevas, C.; Chang, A.E.; Goel, V.K.; Von Hoff, D.D.; Hingorani, S.R. Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell 2012, 21, 418–429. [Google Scholar] [CrossRef] [PubMed]
- Michl, P.; Gress, T.M. Improving drug delivery to pancreatic cancer: Breaching the stromal fortress by targeting hyaluronic acid. Gut 2012, 61, 1377–1379. [Google Scholar] [CrossRef]
- Chauhan, V.P.; Stylianopoulos, T.; Martin, J.D.; Popović, Z.; Chen, O.; Kamoun, W.S.; Jain, R.K. Normalization of tumour blood vessels improves the delivery of nanomedicines in a size-dependent manner. Nat. Nanotechnol. 2012, 7, 383–388. [Google Scholar] [CrossRef]
- Nagathihalli, N.S.; Castellanos, J.A.; Shi, C.; Beesetty, Y.; Reyzer, M.L.; Caprioli, R.; Merchant, N.B. Signal transducer and activator of transcription 3, mediated remodeling of the tumor microenvironment results in enhanced tumor drug delivery in a mouse model of pancreatic cancer. Gastroenterology 2015, 149, 1932–1943. [Google Scholar] [CrossRef]
- Wörmann, S.M.; Song, L.; Ai, J.; Diakopoulos, K.N.; Kurkowski, M.U.; Görgülü, K.; Neesse, A. Loss of P53 function activates JAK2–STAT3 signaling to promote pancreatic tumor growth, stroma modification, and gemcitabine resistance in mice and is associated with patient survival. Gastroenterology 2016, 151, 180–193. [Google Scholar] [CrossRef]
- Chauhan, V.P.; Martin, J.D.; Liu, H.; Lacorre, D.A.; Jain, S.R.; Kozin, S.V.; Popović, Z. Angiotensin inhibition enhances drug delivery and potentiates chemotherapy by decompressing tumour blood vessels. Nat. Commun. 2013, 4, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Vennin, C.; Chin, V.T.; Warren, S.C.; Lucas, M.C.; Herrmann, D.; Magenau, A.; Nobis, M. Transient tissue priming via ROCK inhibition uncouples pancreatic cancer progression, sensitivity to chemotherapy, and metastasis. Sci. Transl. Med. 2017, 9, eaai8504. [Google Scholar] [CrossRef] [PubMed]
- Nanda, S. High stromal expression of α-smooth-muscle actin correlates with aggressive pancreatic cancer biology. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 652. [Google Scholar] [CrossRef]
- Liu, S.L.; Cao, S.G.; Li, Y.; Sun, B.; Chen, D.; Wang, D.S.; Zhou, Y.B. Pancreatic stellate cells facilitate pancreatic cancer cell viability and invasion. Oncol. Lett. 2019, 17, 2057–2062. [Google Scholar] [CrossRef]
- Di Carlo, C.; Brandi, J.; Cecconi, D. Pancreatic cancer stem cells: Perspectives on potential therapeutic approaches of pancreatic ductal adenocarcinoma. World J. Stem Cells 2018, 10, 172. [Google Scholar] [CrossRef]
- Plaks, V.; Kong, N.; Werb, Z. The cancer stem cell niche: How essential is the niche in regulating stemness of tumor cells? Cell Stem Cell 2015, 16, 225–238. [Google Scholar] [CrossRef]
- Valle, S.; Martin-Hijano, L.; Alcalá, S.; Alonso-Nocelo, M.; Sainz, B., Jr. The ever-evolving concept of the cancer stem cell in pancreatic cancer. Cancers 2018, 10, 33. [Google Scholar] [CrossRef]
- Lei, Y.; Zhang, D.; Yu, J.; Dong, H.; Zhang, J.; Yang, S. Targeting autophagy in cancer stem cells as an anticancer therapy. Cancer Lett. 2017, 393, 33–39. [Google Scholar] [CrossRef]
- Rao, C.V.; Mohammed, A. New insights into pancreatic cancer stem cells. World J. Stem Cells 2015, 7, 547. [Google Scholar] [CrossRef]
- Minet, E.; Arnould, T.; Michel, G.; Roland, I.; Mottet, D.; Raes, M.; Michiels, C. ERK activation upon hypoxia: Involvement in HIF-1 activation. FEBS Lett. 2000, 468, 53–58. [Google Scholar] [CrossRef]
- Courtnay, R.; Ngo, D.C.; Malik, N.; Ververis, K.; Tortorella, S.M.; Karagiannis, T.C. Cancer metabolism and the Warburg effect: The role of HIF-1 and PI3K. Mol. Biol. Rep. 2015, 42, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Dhani, N.C.; Serra, S.; Pintilie, M.; Schwock, J.; Xu, J.; Gallinger, S.; Hedley, D.W. Analysis of the intra-and intertumoral heterogeneity of hypoxia in pancreatic cancer patients receiving the nitroimidazole tracer pimonidazole. Br. J. Cancer 2015, 113, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Weng, L.; Jia, Y.; Liu, B.; Wu, S.; Xue, L.; Shang, M. PTBP3 promotes malignancy and hypoxia-induced chemoresistance in pancreatic cancer cells by ATG12 up-regulation. J. Cell. Mol. Med. 2020, 24, 2917–2930. [Google Scholar] [CrossRef] [PubMed]
- Ranniger, K.; Saldino, R.M. Arteriographic diagnosis of pancreatic lesions. Radiology 1966, 86, 470–474. [Google Scholar] [CrossRef]
- Kisker, O.; Onizuka, S.; Banyard, J.; Komiyama, T.; Becker, C.M.; Achilles, E.G.; Pirie-Shepherd, S.R. Generation of multiple angiogenesis inhibitors by human pancreatic cancer. Cancer Res. 2001, 61, 7298–7304. [Google Scholar]
- Tang, D.; Wang, D.; Yuan, Z.; Xue, X.; Zhang, Y.; An, Y.; Jiang, K. Persistent activation of pancreatic stellate cells creates a microenvironment favorable for the malignant behavior of pancreatic ductal adenocarcinoma. Int. J. Cancer 2013, 132, 993–1003. [Google Scholar] [CrossRef]
- Yilmaz, M.; Christofori, G. Mechanisms of motility in metastasizing cells. Mol. Cancer Res. 2010, 8, 629–642. [Google Scholar] [CrossRef]
- Zhao, X.; Gao, S.; Ren, H.; Sun, W.; Zhang, H.; Sun, J.; Hao, J. Hypoxia-inducible factor-1 promotes pancreatic ductal adenocarcinoma invasion and metastasis by activating transcription of the actin-bundling protein fascin. Cancer Res. 2014, 74, 2455–2464. [Google Scholar] [CrossRef]
- Erkan, M.; Kurtoglu, M.; Kleeff, J. The role of hypoxia in pancreatic cancer: A potential therapeutic target? Expert Rev. Gastroenterol. Hepatol. 2016, 10, 301–316. [Google Scholar] [CrossRef]
- Muz, B.; de la Puente, P.; Azab, F.; Azab, A.K. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia 2015, 3, 83. [Google Scholar] [CrossRef]
- Khalifa, S.A.; Elias, N.; Farag, M.A.; Chen, L.; Saeed, A.; Hegazy, M.E.F.; Chang, F.R. Marine natural products: A source of novel anticancer drugs. Mar. Drugs 2019, 17, 491. [Google Scholar] [CrossRef] [PubMed]
- Han, B.N.; Hong, L.L.; Gu, B.B.; Sun, Y.T.; Wang, J.; Liu, J.T.; Lin, H.W. Natural Products from Sponges. In Symbiotic Microbiomes of Coral Reefs Sponges and Corals; Springer: Berlin/Heidelberg, Germany, 2019; pp. 329–463. [Google Scholar]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef]
- Fedoreev, S.A.; Prokof’eva, N.G.; Denisenko, V.A.; Rebachuk, N.M. Cytotoxic activity of aaptamines from suberitid marine sponges. Pharm. Chem. J. 1988, 22, 615–618. [Google Scholar] [CrossRef]
- Zapolska-Downar, D.; Zapolski-Downar, A.; Markiewski, M.; Ciechanowicz, A.; Kaczmarczyk, M.; Naruszewicz, M. Selective inhibition by probucol of vascular cell adhesion molecule-1 (VCAM-1) expression in human vascular endothelial cells. Atherosclerosis 2001, 155, 123–130. [Google Scholar] [CrossRef]
- Blackburn, C.L.; Hopmann, C.; Sakowicz, R.; Berdelis, M.S.; Goldstein, L.S.; Faulkner, D.J. Adociasulfates 1− 6, Inhibitors of Kinesin Motor Proteins from the Sponge Haliclona (aka Adocia) sp. J. Org. Chem. 1999, 64, 5565–5570. [Google Scholar] [CrossRef] [PubMed]
- Anjum, K.; Abbas, S.Q.; Shah, S.A.A.; Akhter, N.; Batool, S.; ul Hassan, S.S. Marine sponges as a drug treasure. Biomol. Ther. 2016, 24, 347. [Google Scholar] [CrossRef] [PubMed]
- Perry, N.B.; Blunt, J.W.; Munro, M.H.; Thompson, A.M. Antiviral and antitumor agents from a New Zealand sponge, Mycale sp. 2. Structures and solution conformations of mycalamides A and B. J. Org. Chem. 1990, 55, 223–227. [Google Scholar] [CrossRef]
- Cortes, J.; O’Shaughnessy, J.; Loesch, D.; Blum, J.L.; Vahdat, L.T.; Petrakova, K.; Vladimirov, V. Eribulin monotherapy versus treatment of physician’s choice in patients with metastatic breast cancer (EMBRACE): A phase 3 open-label randomised study. Lancet 2011, 377, 914–923. [Google Scholar] [CrossRef]
- Guzmán, E.A.; Harmody, D.; Pitts, T.P.; Vera-Diaz, B.; Winder, P.L.; Yu, Y.; Wright, A.E. Inhibition of IL-8 secretion on BxPC-3 and MIA PaCa-2 cells and induction of cytotoxicity in pancreatic cancer cells with marine natural products. Anti-Cancer Drugs 2017, 28, 153–160. [Google Scholar] [CrossRef]
- Waugh, D.J.; Wilson, C. The interleukin-8 pathway in cancer. Clin. Cancer Res. 2008, 14, 6735–6741. [Google Scholar] [CrossRef]
- Wright, A.E.; Roth, G.P.; Hoffman, J.K.; Divlianska, D.B.; Pechter, D.; Sennett, S.H.; Pomponi, S.A. Isolation, synthesis, and biological activity of aphrocallistin, an adenine-substituted bromotyramine metabolite from the hexactinellida sponge Aphrocallistes beatrix. J. Nat. Prod. 2009, 72, 1178–1183. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.H.; Sakemi, S.; Burres, N.; McCarthy, P. Isobatzellines A, B, C, and D. Cytotoxic and antifungal pyrroloquinoline alkaloids from the marine sponge Batzella sp. J. Org. Chem. 1990, 55, 4964–4966. [Google Scholar] [CrossRef]
- Guzmán, E.A.; Johnson, J.D.; Carrier, M.K.; Meyer, C.I.; Pitts, T.P.; Gunasekera, S.P.; Wright, A.E. Selective cytotoxic activity of the marine derived batzelline compounds against pancreatic cancer cell lines. Anti-Cancer Drugs 2009, 20, 149. [Google Scholar] [CrossRef] [PubMed]
- Bewley, C.A.; Debitus, C.; Faulkner, D.J. Microsclerodermins A and B. Antifungal cyclic peptides from the lithistid sponge Microscleroderma sp. J. Am. Chem. Soc. 1994, 116, 7631–7636. [Google Scholar] [CrossRef]
- Guzmán, E.A.; Maers, K.; Roberts, J.; Kemami-Wangun, H.V.; Harmody, D.; Wright, A.E. The marine natural product microsclerodermin A is a novel inhibitor of the nuclear factor kappa B and induces apoptosis in pancreatic cancer cells. Investig. New Drugs 2015, 33, 86–94. [Google Scholar] [CrossRef]
- Wright, A.E.; Pomponi, S.A.; Longley, R.E.; Isbrucker, R.A. Antiproliferative Activity of Microsclerodermins. U.S. Patent No. 6,384,187, 7 May 2002. [Google Scholar]
- Dybdal-Hargreaves, N.F.; Risinger, A.L.; Mooberry, S.L. Eribulin mesylate: Mechanism of action of a unique microtubule-targeting agent. Clin. Cancer Res. 2015, 21, 2445–2452. [Google Scholar] [CrossRef]
- Ciavatta, M.L.; Lefranc, F.; Carbone, M.; Mollo, E.; Gavagnin, M.; Betancourt, T.; Kiss, R. Marine Mollusk-Derived Agents with Antiproliferative Activity as Promising Anticancer Agents to Overcome Chemotherapy Resistance. Med. Res. Rev. 2017, 37, 702–801. [Google Scholar] [CrossRef]
- Kindler, H.L.; Tothy, P.K.; Wolff, R.; McCormack, R.A.; Abbruzzese, J.L.; Mani, S.; Vokes, E.E. Phase II trials of dolastatin-10 in advanced pancreaticobiliary cancers. Investig. New Drugs 2005, 23, 489–493. [Google Scholar] [CrossRef]
- Serova, M.; De Gramont, A.; Bieche, I.; Riveiro, M.E.; Galmarini, C.M.; Aracil, M.; Raymond, E. Predictive factors of sensitivity to elisidepsin, a novel Kahalalide F-derived marine compound. Mar. Drugs 2013, 11, 944–959. [Google Scholar] [CrossRef]
- Hong, S.M.; Li, A.; Olino, K.; Wolfgang, C.L.; Herman, J.M.; Schulick, R.D.; Goggins, M. Loss of E-cadherin expression and outcome among patients with resectable pancreatic adenocarcinomas. Mod. Pathol. 2011, 24, 1237–1247. [Google Scholar] [CrossRef]
- Figuerola, B.; Avila, C. The Phylum Bryozoa as a promising source of anticancer drugs. Mar. Drugs 2019, 17, 477. [Google Scholar] [CrossRef] [PubMed]
- Morris, B.D.; Prinsep, M.R. Amathaspiramides A− F, Novel Brominated Alkaloids from the Marine Bryozoan Amathia wilsoni. J. Nat. Prod. 1999, 62, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Shimokawa, J.; Chiyoda, K.; Umihara, H.; Fukuyama, T. Antiproliferative activity of amathaspiramide alkaloids and analogs. Chem. Pharm. Bull. 2016, 64, 1239–1241. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.B.; Wu, G.J.; Wang, X.Y.; Tang, X.J.; Gong, L. Bryostatin I inhibits growth and proliferation of pancreatic cancer cells via suppression of NF-κB activation. Trop. J. Pharm. Res. 2016, 15, 2071–2076. [Google Scholar] [CrossRef][Green Version]
- Lam, A.; Sparano, J.A.; Vinciguerra, V.; Ocean, A.J.; Christos, P.; Hochster, H.; Kaubisch, A. Phase II study of paclitaxel plus the protein kinase C inhibitor bryostatin-1 in advanced pancreatic carcinoma. Am. J. Clin. Oncol. 2010, 33, 121. [Google Scholar] [CrossRef] [PubMed]
- Kollár, P.; Rajchard, J.; Balounová, Z.; Pazourek, J. Marine natural products: Bryostatins in preclinical and clinical studies. Pharm. Biol. 2014, 52, 237–242. [Google Scholar] [CrossRef]
- El-Rayes, B.F.; Gadgeel, S.; Shields, A.F.; Manza, S.; Lorusso, P.; Philip, P.A. Phase I study of bryostatin 1 and gemcitabine. Clin. Cancer Res. 2006, 12, 7059–7062. [Google Scholar] [CrossRef][Green Version]
- Ruan, B.F.; Zhu, H.L. The chemistry and biology of the bryostatins: Potential PKC inhibitors in clinical development. Curr. Med. Chem. 2012, 19, 2652–2664. [Google Scholar] [CrossRef]
- Leandro, A.; Pereira, L.; Gonçalves, A.M. Diverse applications of marine macroalgae. Mar. Drugs 2020, 18, 17. [Google Scholar] [CrossRef]
- Rocha, D.H.; Seca, A.M.; Pinto, D.C. Seaweed secondary metabolites in vitro and in vivo anticancer activity. Mar. Drugs 2018, 16, 410. [Google Scholar] [CrossRef]
- Alves, C.; Silva, J.; Pinteus, S.; Gaspar, H.; Alpoim, M.C.; Botana, L.M.; Pedrosa, R. From marine origin to therapeutics: The antitumor potential of marine algae-derived compounds. Front. Pharmacol. 2018, 9, 777. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Hu, J.; Yang, B.; Lin, X.P.; Zhou, X.F.; Yang, X.W.; Liu, Y. Chemical composition of seaweeds. In Seaweed Sustainability; Academic Press: Cambridge, MA, USA, 2015; pp. 79–124. [Google Scholar]
- Namvar, F.; Mohamad, R.; Baharara, J.; Zafar-Balanejad, S.; Fargahi, F.; Rahman, H.S. Antioxidant, antiproliferative, and antiangiogenesis effects of polyphenol-rich seaweed (Sargassum muticum). BioMed Res. Int. 2013, 2013, 604787. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.; Soares, A.I.; Neves, N.M.; Reis, R.L.; Marques, A.P.; Silva, T.H.; Martins, A. Fucoidan immobilized at the surface of a fibrous mesh presents toxic effects over melanoma cells, but not over non-cancer skin cells. Biomacromolecules 2020, 21, 2745–2754. [Google Scholar] [CrossRef]
- Haq, S.H.; Al-Ruwaished, G.; Al-Mutlaq, M.A.; Naji, S.A.; Al-Mogren, M.; Al-Rashed, S.; Al-Mussallam, A. Antioxidant, Anticancer Activity and Phytochemical Analysis of Green Algae, Chaetomorpha Collected from the Arabian Gulf. Sci. Rep. 2019, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Saadaoui, I.; Rasheed, R.; Abdulrahman, N.; Bounnit, T.; Cherif, M.; Al Jabri, H.; Mraiche, F. Algae-Derived Bioactive Compounds with Anti-Lung Cancer Potential. Mar. Drugs 2020, 18, 197. [Google Scholar] [CrossRef] [PubMed]
- Narayani, S.S.; Saravanan, S.; Ravindran, J.; Ramasamy, M.S.; Chitra, J. In vitro anticancer activity of fucoidan extracted from Sargassum cinereum against Caco-2 cells. Int. J. Biol. Macromol. 2019, 138, 618–628. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Xu, J.; Ge, K.; Tian, Q.; Zhao, P.; Guo, Y. Anti-inflammatory effect of low molecular weight fucoidan from Saccharina japonica on atherosclerosis in apoE-knockout mice. Int. J. Biol. Macromol. 2018, 118, 365–374. [Google Scholar] [CrossRef]
- Kan, J.; Hood, M.; Burns, C.; Scholten, J.; Chuang, J.; Tian, F.; Gui, M. A novel combination of wheat peptides and fucoidan attenuates ethanol-induced gastric mucosal damage through anti-oxidant, anti-inflammatory, and pro-survival mechanisms. Nutrients 2017, 9, 978. [Google Scholar] [CrossRef]
- Oliveira, C.; Granja, S.; Neves, N.M.; Reis, R.L.; Baltazar, F.; Silva, T.H.; Martins, A. Fucoidan from Fucus vesiculosus inhibits new blood vessel formation and breast tumor growth in vivo. Carbohydr. Polym. 2019, 223, 115034. [Google Scholar] [CrossRef]
- Van Weelden, G.; Bobiński, M.; Okła, K.; Van Weelden, W.J.; Romano, A.; Pijnenborg, J. Fucoidan structure and activity in relation to anti-cancer mechanisms. Mar. Drugs 2019, 17, 32. [Google Scholar] [CrossRef]
- Delma, C.R.; Thirugnanasambandan, S.; Srinivasan, G.P.; Raviprakash, N.; Manna, S.K.; Natarajan, M.; Aravindan, N. Fucoidan from marine brown algae attenuates pancreatic cancer progression by regulating p53–NFκB crosstalk. Phytochemistry 2019, 167, 112078. [Google Scholar] [CrossRef] [PubMed]
- Delma, C.R.; Somasundaram, S.T.; Srinivasan, G.P.; Khursheed, M.; Bashyam, M.D.; Aravindan, N. Fucoidan from Turbinaria conoides: A multifaceted ‘deliverable’ to combat pancreatic cancer progression. Int. J. Biol. Macromol. 2015, 74, 447–457. [Google Scholar] [CrossRef]
- Gómez-Guzmán, M.; Rodríguez-Nogales, A.; Algieri, F.; Gálvez, J. Potential role of seaweed polyphenols in cardiovascular-associated disorders. Mar. Drugs 2018, 16, 250. [Google Scholar] [CrossRef] [PubMed]
- Vauzour, D.; Rodriguez-Mateos, A.; Corona, G.; Oruna-Concha, M.J.; Spencer, J.P. Polyphenols and human health: Prevention of disease and mechanisms of action. Nutrients 2010, 2, 1106–1131. [Google Scholar] [CrossRef]
- Hooper, L.; Kroon, P.A.; Rimm, E.B.; Cohn, J.S.; Harvey, I.; Le Cornu, K.A.; Cassidy, A. Flavonoids, flavonoid-rich foods, and cardiovascular risk: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2008, 88, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Renaud, J.; Martinoli, M.G. Considerations for the use of polyphenols as therapies in neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 1883. [Google Scholar] [CrossRef]
- Abdal Dayem, A.; Choi, H.Y.; Yang, G.M.; Kim, K.; Saha, S.K.; Cho, S.G. The anti-cancer effect of polyphenols against breast cancer and cancer stem cells: Molecular mechanisms. Nutrients 2016, 8, 581. [Google Scholar] [CrossRef]
- Li, W.; He, N.; Tian, L.; Shi, X.; Yang, X. Inhibitory effects of polyphenol-enriched extract from Ziyang tea against human breast cancer MCF-7 cells through reactive oxygen species-dependent mitochondria molecular mechanism. J. Food Drug Anal. 2016, 24, 527–538. [Google Scholar] [CrossRef]
- Yuan, Y.V.; Walsh, N.A. Antioxidant and antiproliferative activities of extracts from a variety of edible seaweeds. Food Chem. Toxicol. 2006, 44, 1144–1150. [Google Scholar] [CrossRef]
- Mansur, A.A.; Brown, M.T.; Billington, R.A. The cytotoxic activity of extracts of the brown alga Cystoseira tamariscifolia (Hudson) Papenfuss, against cancer cell lines changes seasonally. J. Appl. Phycol. 2020, 32, 2419–2429. [Google Scholar] [CrossRef]
- Aravindan, S.; Delma, C.R.; Thirugnanasambandan, S.S.; Herman, T.S.; Aravindan, N. Anti-pancreatic cancer deliverables from sea: First-hand evidence on the efficacy, molecular targets and mode of action for multifarious polyphenols from five different brown-algae. PLoS ONE 2013, 8, e61977. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, S.; Ramraj, S.K.; Somasundaram, S.T.; Herman, T.S.; Aravindan, N. Polyphenols from marine brown algae target radiotherapy-coordinated EMT and stemness-maintenance in residual pancreatic cancer. Stem Cell Res. Ther. 2015, 6, 182. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, S.; Ramraj, S.; Kandasamy, K.; Thirugnanasambandan, S.S.; Somasundaram, D.B.; Herman, T.S.; Aravindan, N. Hormophysa triquerta polyphenol, an elixir that deters CXCR4-and COX2-dependent dissemination destiny of treatment-resistant pancreatic cancer cells. Oncotarget 2017, 8, 5717. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, S.; Ramraj, S.K.; Somasundaram, S.T.; Aravindan, N. Novel adjuvants from seaweed impede autophagy signaling in therapy-resistant residual pancreatic cancer. J. Biomed. Sci. 2015, 22, 28. [Google Scholar] [CrossRef]
- Aravindan, S.; Somasundaram, D.B.; Somasundaram, S.T.; Natarajan, M.; Herman, T.S.; Aravindan, N. Targeting acquired oncogenic burden in resilient pancreatic cancer: A novel benefit from marine polyphenols. Mol. Cell. Biochem. 2019, 460, 175–193. [Google Scholar] [CrossRef]
- Singh, A.K.; Rana, H.K.; Pandey, A.K. Fungal-Derived Natural Product: Synthesis, Function, and Applications. In Recent Advancement in White Biotechnology Through Fungi; Springer: Cham, Switzerland, 2019; pp. 229–248. [Google Scholar]
- Bonugli-Santos, R.C.; dos Santos Vasconcelos, M.R.; Passarini, M.R.; Vieira, G.A.; Lopes, V.C.; Mainardi, P.H.; Sette, L.D. Marine-derived fungi: Diversity of enzymes and biotechnological applications. Front. Microbiol. 2015, 6, 269. [Google Scholar] [CrossRef]
- Richards, T.A.; Jones, M.D.; Leonard, G.; Bass, D. Marine fungi: Their ecology and molecular diversity. Annu. Rev. Mar. Sci. 2012, 4, 495–522. [Google Scholar] [CrossRef]
- Badalyan, S.M.; Barkhudaryan, A.; Rapior, S. Recent progress in research on the pharmacological potential of mushrooms and prospects for their clinical application. In Medicinal Mushrooms; Agrawal, D., Dhanasekaran, M., Eds.; Springer: Singapore, 2019; pp. 1–70. [Google Scholar] [CrossRef]
- Ramos, A.A.; Prata-Sena, M.; Castro-Carvalho, B.; Dethoup, T.; Buttachon, S.; Kijjoa, A.; Rocha, E. Potential of four marine-derived fungi extracts as anti-proliferative and cell death-inducing agents in seven human cancer cell lines. Asian Pac. J. Trop. Med. 2015, 8, 798–806. [Google Scholar] [CrossRef]
- Malhão, F.; Ramos, A.A.; Buttachon, S.; Dethoup, T.; Kijjoa, A.; Rocha, E. Cytotoxic and antiproliferative effects of preussin, a hydroxypyrrolidine derivative from the marine sponge-associated fungus Aspergillus candidus KUFA 0062, in a panel of breast cancer cell lines and using 2D and 3D cultures. Mar. Drugs 2019, 17, 448. [Google Scholar] [CrossRef]
- Yu, Z.; Lang, G.; Kajahn, I.; Schmaljohann, R.; Imhoff, J.F. Scopularides A and B, cyclodepsipeptides from a marine sponge-derived fungus, Scopulariopsis brevicaulis. J. Nat. Prod. 2008, 71, 1052–1054. [Google Scholar] [CrossRef]
- Ye, P.; Shen, L.; Jiang, W.; Ye, Y.; Chen, C.T.A.; Wu, X.; Wu, B. Zn-driven discovery of a hydrothermal vent fungal metabolite clavatustide C, and an experimental study of the anti-cancer mechanism of clavatustide B. Mar. Drugs 2014, 12, 3203–3217. [Google Scholar] [CrossRef] [PubMed]
- Heiferman, M.J.; Salabat, M.R.; Ujiki, M.B.; Strouch, M.J.; Cheon, E.C.; Silverman, R.B.; Bentrem, D.J. Sansalvamide induces pancreatic cancer growth arrest through changes in the cell cycle. Anticancer. Res. 2010, 30, 73–78. [Google Scholar] [PubMed]
- Ujiki, M.B.; Ding, X.Z.; Roginsky, A.B.; Salabat, M.R.; Gu, W.; Silverman, R.; Adrian, T.E. Pancreatic cancer cell cycle arrest and apoptosis are induced by a novel depsipeptide sansalvamide analogue. Pancreas 2005, 31, 475. [Google Scholar] [CrossRef]
- Zhang, W.; Zhu, Y.; Yu, H.; Liu, X.; Jiao, B.; Lu, X. Libertellenone H, a natural pimarane diterpenoid, inhibits thioredoxin system and induces ROS-mediated apoptosis in human pancreatic cancer cells. Molecules 2021, 26, 315. [Google Scholar] [CrossRef] [PubMed]
- Karlenius, T.C.; Tonissen, K.F. Thioredoxin and cancer: A role for thioredoxin in all states of tumor oxygenation. Cancers 2010, 2, 209–232. [Google Scholar] [CrossRef]
- Ghareeb, H.; Metanis, N. The thioredoxin system: A promising target for cancer drug development. Chem.–Eur. J. 2020, 26, 10175–10184. [Google Scholar] [CrossRef]
- Qi, C.; Gao, W.; Guan, D.; Wang, J.; Liu, M.; Chen, C.; Zhang, Y. Butenolides from a marine-derived fungus Aspergillus terreus with antitumor activities against pancreatic ductal adenocarcinoma cells. Bioorganic Med. Chem. 2018, 26, 5903–5910. [Google Scholar] [CrossRef]
- Williams, P.G. Panning for chemical gold: Marine bacteria as a source of new therapeutics. Trends Biotechnol. 2009, 27, 45–52. [Google Scholar] [CrossRef]
- Reichenbach, H. Myxobacteria, producers of novel bioactive substances. J. Ind. Microbiol. Biotechnol. 2001, 27, 149–156. [Google Scholar] [CrossRef]
- Wang, C.; Fan, Q.; Zhang, X.; Lu, X.; Xu, Y.; Zhu, W.; Hao, L. Isolation, characterization, and pharmaceutical applications of an exopolysaccharide from Aerococcus uriaeequi. Mar. Drugs 2018, 16, 337. [Google Scholar] [CrossRef]
- Shin, D.; Byun, W.S.; Moon, K.; Kwon, Y.; Bae, M.; Um, S.; Oh, D.C. Coculture of marine Streptomyces sp. with Bacillus sp. produces a new piperazic acid-bearing cyclic peptide. Front. Chem. 2018, 6, 498. [Google Scholar] [CrossRef] [PubMed]
- Galaviz-Silva, L.; Iracheta-Villarreal, J.M.; Molina-Garza, Z.J. Bacillus and Virgibacillus strains isolated from three Mexican coasts antagonize Staphylococcus aureus and Vibrio parahaemolyticus. FEMS Microbiol. Lett. 2018, 365, fny202. [Google Scholar] [CrossRef] [PubMed]
- Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J.; Corbett, T.H. Total structure determination of apratoxin A, a potent novel cytotoxin from the marine cyanobacterium Lyngbya majuscula. J. Am. Chem. Soc. 2001, 123, 5418–5423. [Google Scholar] [CrossRef]
- Taori, K.; Paul, V.J.; Luesch, H. Structure and activity of largazole, a potent antiproliferative agent from the Floridian marine cyanobacterium Symploca sp. J. Am. Chem. Soc. 2008, 130, 1806–1807. [Google Scholar] [CrossRef]
- Schneider, K.; Keller, S.; Wolter, F.E.; Röglin, L.; Beil, W.; Seitz, O.; Süssmuth, R.D. Proximicins A, B, and C—Antitumor furan analogues of netropsin from the marine actinomycete Verrucosispora induce upregulation of p53 and the cyclin kinase inhibitor p21. Angew. Chem. Int. Ed. 2008, 47, 3258–3261. [Google Scholar] [CrossRef]
- Trimurtulu, G.; Ohtani, I.; Patterson, G.M.; Moore, R.E.; Corbett, T.H.; Valeriote, F.A.; Demchik, L. Total structures of cryptophycins, potent antitumor depsipeptides from the blue-green alga Nostoc sp. strain GSV 224. J. Am. Chem. Soc. 1994, 116, 4729–4737. [Google Scholar] [CrossRef]
- Gutiérrez, M.; Suyama, T.L.; Engene, N.; Wingerd, J.S.; Matainaho, T.; Gerwick, W.H. Apratoxin D, a potent cytotoxic cyclodepsipeptide from Papua New Guinea collections of the marine cyanobacteria Lyngbya majuscula and Lyngbya sordida. J. Nat. Prod. 2008, 71, 1099–1103. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.C.; Chen, Z.; Jiang, Y.; Akare, S.; Kolber-Simonds, D.; Condon, K.; Nomoto, K. Apratoxin A Shows Novel Pancreas-Targeting Activity through the Binding of Sec 61Apratoxin A, a Novel Pancreas-Targeting Agent. Mol. Cancer Ther. 2016, 15, 1208–1216. [Google Scholar] [CrossRef]
- Luesch, H.; Chanda, S.K.; Raya, R.M.; DeJesus, P.D.; Orth, A.P.; Walker, J.R.; Schultz, P.G. A functional genomics approach to the mode of action of apratoxin A. Nat. Chem. Biol. 2006, 2, 158–167. [Google Scholar] [CrossRef]
- Chen, Q.Y.; Liu, Y.; Luesch, H. Systematic chemical mutagenesis identifies a potent novel apratoxin A/E hybrid with improved in vivo antitumor activity. ACS Med. Chem. Lett. 2011, 2, 861–865. [Google Scholar] [CrossRef]
- Cai, W.; Chen, Q.Y.; Dang, L.H.; Luesch, H. Apratoxin S10, a dual inhibitor of angiogenesis and cancer cell growth to treat highly vascularized tumors. ACS Med. Chem. Lett. 2017, 8, 1007–1012. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Law, B.K.; Luesch, H. Apratoxin a reversibly inhibits the secretory pathway by preventing cotranslational translocation. Mol. Pharmacol. 2009, 76, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Ratnayake, R.; Gerber, M.H.; Chen, Q.Y.; Yu, Y.; Derendorf, H.; Luesch, H. Development of apratoxin S10 (Apra S10) as an anti-pancreatic cancer agent and its preliminary evaluation in an orthotopic patient-derived xenograft (PDX) model. Investig. New Drugs 2019, 37, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Lu, D.; Wu, C.; Li, M.; Ding, Z.; Li, Y.; Li, H. Coibamide A kills cancer cells through inhibiting autophagy. Biochem. Biophys. Res. Commun. 2021, 547, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Abdelnasser, S.M.; Yahya, S.M.; Mohamed, W.F.; Asker, M.M.; Shady, H.M.A.; Mahmoud, M.G.; Gadallah, M.A. Antitumor exopolysaccharides derived from novel marine bacillus: Isolation, characterization aspect and biological activity. Asian Pac. J. Cancer Prev. APJCP 2017, 18, 1847. [Google Scholar]
- Liu, G.; Liu, R.; Shan, Y.; Sun, C. Marine bacterial exopolysaccharide EPS11 inhibits migration and invasion of liver cancer cells by directly targeting collagen I. J. Biol. Chem. 2021, 297, 101133. [Google Scholar] [CrossRef]
- Ryschich, E.; Khamidjanov, A.; Kerkadze, V.; Büchler, M.W.; Zöller, M.; Schmidt, J. Promotion of tumor cell migration by extracellular matrix proteins in human pancreatic cancer. Pancreas 2009, 38, 804–810. [Google Scholar] [CrossRef]
- Grzesiak, J.J.; Cao, H.S.T.; Burton, D.W.; Kaushal, S.; Vargas, F.; Clopton, P.; Bouvet, M. Knockdown of the β1 integrin subunit reduces primary tumor growth and inhibits pancreatic cancer metastasis. Int. J. Cancer 2011, 129, 2905–2915. [Google Scholar] [CrossRef]
- Yu, Y.; Shen, M.; Song, Q.; Xie, J. Biological activities and pharmaceutical applications of polysaccharide from natural resources: A review. Carbohydr. Polym. 2018, 183, 91–101. [Google Scholar] [CrossRef]
- Ahmed, S.; Vaitkevicius, V.K.; Zalupski, M.M.; Du, W.; Arlauskas, P.; Gordon, C.; Kellogg, C.; Shields, A.F. Cisplatin, cytarabine, caffeine, and continuously infused 5-fluorouracil (PACE) in the treatment of advanced pancreatic carcinoma: A phase II study. Am. J. Clin. Oncol. 2000, 23, 420–424. [Google Scholar] [CrossRef]
- Neoptolemos, J.P.; Stocken, D.D.; Bassi, C.; Ghaneh, P.; Cunningham, D.; Goldstein, D.; European Study Group for Pancreatic Cancer. Adjuvant chemotherapy with fluorouracil plus folinic acid vs gemcitabine following pancreatic cancer resection: A randomized controlled trial. JAMA 2010, 304, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Renouf, D.J.; Tang, P.A.; Major, P.; Krzyzanowska, M.K.; Dhesy-Thind, B.; Goffin, J.R.; Moore, M.J. A phase II study of the halichondrin B analog eribulin mesylate in gemcitabine refractory advanced pancreatic cancer. Investig. New Drugs 2012, 30, 1203–1207. [Google Scholar] [CrossRef]
- Belli, C.; Piemonti, L.; D’Incalci, M.; Zucchetti, M.; Porcu, L.; Cappio, S.; Reni, M. Phase II trial of salvage therapy with trabectedin in metastatic pancreatic adenocarcinoma. Cancer Chemother. Pharmacol. 2016, 77, 477–484. [Google Scholar] [CrossRef] [PubMed]
- National Library of Medicine (U.S.). (14 June 2022). Lurbinectedin in Patients with Advanced Pancreatic Cancer with DNA Repair Mutations. Identifier: NCT05229588. Available online: https://clinicaltrials.gov/ct2/show/study/NCT05229588 (accessed on 19 October 2022).
- Millward, M.; Price, T.; Townsend, A.; Sweeney, C.; Spencer, A.; Sukumaran, S.; Spear, M.A. Phase 1 clinical trial of the novel proteasome inhibitor marizomib with the histone deacetylase inhibitor vorinostat in patients with melanoma, pancreatic and lung cancer based on in vitro assessments of the combination. Investig. New Drugs 2012, 30, 2303–2317. [Google Scholar] [CrossRef]
- Normolle, D.P.; Zureikat, A.H.; Hogg, M.E.; Bartlett, D.L.; Lee, K.; Tsung, A.; Lotze, M.T. A Randomized Phase II Preoperative Study of Autophagy Inhibition With High-Dose Hydroxychloroquine and Gemcita-bine/Nab-Paclitaxel in Pancreatic Cancer Patients. Clin. Cancer Res. 2020, 26, 3126–3134. [Google Scholar]
- Philip, P.A.; Lacy, J.; Portales, F.; Sobrero, A.; Pazo-Cid, R.; Mozo, J.L.M.; Hammel, P. Nab-paclitaxel plus gemcitabine in patients with locally advanced pancreatic cancer (LAPACT): A multicentre, open-label phase 2 study. Lancet Gastroenterol. Hepatol. 2020, 5, 285–294. [Google Scholar] [CrossRef]
- Su, T.; Yang, B.; Gao, T.; Liu, T.; Li, J. Polymer nanoparticle-assisted chemotherapy of pancreatic cancer. Ther. Adv. Med. Oncol. 2020, 12, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Xin, Y.; Yin, M.; Zhao, L.; Meng, F.; Luo, L. Recent progress on nanoparticle-based drug delivery systems for cancer therapy. Cancer Biol. Med. 2017, 14, 228–241. [Google Scholar] [CrossRef]
- Lucas, A.T.; Price, L.S.; Schorzman, A.; Zamboni, W.C. Complex effects of tumor microenvironment on the tumor disposition of carrier-mediated agents. Nanomedicine 2017, 12, 2021–2042. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Habtemariam, S. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef]
- Golombek, S.K.; May, J.N.; Theek, B.; Appold, L.; Drude, N.; Kiessling, F.; Lammers, T. Tumor targeting via EPR: Strategies to enhance patient responses. Adv. Drug Deliv. Rev. 2018, 130, 17–38. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.; Park, C.; Yi, G.; Lee, D.; Koo, H. Active targeting strategies using biological ligands for nanoparticle drug delivery systems. Cancers 2019, 11, 640. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Lillard, J.W., Jr. Nanoparticle-based targeted drug delivery. Exp. Mol. Pathol. 2009, 86, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Trickler, W.J.; Khurana, J.; Nagvekar, A.A.; Dash, A.K. Chitosan and glyceryl monooleate nanostructures containing gemcitabine: Potential delivery system for pancreatic cancer treatment. Aaps Pharmscitech 2010, 11, 392–401. [Google Scholar] [CrossRef]
- Arya, G.; Vandana, M.; Acharya, S.; Sahoo, S.K. Enhanced antiproliferative activity of Herceptin (HER2)-conjugated gemcitabine-loaded chitosan nanoparticle in pancreatic cancer therapy. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 859–870. [Google Scholar] [CrossRef] [PubMed]
- Min, Y.J.; Joo, K.R.; Park, N.H.; Yun, T.K.; Nah, Y.W.; Nam, C.W.; Park, J.H. Gemcitabine therapy in patients with advanced pancreatic cancer. Korean J. Intern. Med. 2002, 17, 259–262. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, J.; Xu, Q.; Xu, S.; Wen, J.; Yu, Z.; Yang, D. Folate-chitosan-gemcitabine core-shell nanoparticles targeted to pancreatic cancer. Chin. J. Cancer Res. 2013, 25, 527–535. [Google Scholar]
- Snima, K.S.; Jayakumar, R.; Lakshmanan, V.K. In vitro and in vivo biological evaluation of O-carboxymethyl chitosan encapsulated metformin nanoparticles for pancreatic cancer therapy. Pharm. Res. 2014, 31, 3361–3370. [Google Scholar] [CrossRef]
- Thakkar, A.; Chenreddy, S.; Thio, A.; Khamas, W.; Wang, J.; Prabhu, S. Preclinical systemic toxicity evaluation of chitosan-solid lipid nanoparticle-encapsulated aspirin and curcumin in combination with free sulforaphane in BALB/c mice. Int. J. Nanomed. 2016, 11, 3265–3276. [Google Scholar]
- David, K.I.; Jaidev, L.R.; Sethuraman, S.; Krishnan, U.M. Dual drug loaded chitosan nanoparticles—Sugar-coated arsenal against pancreatic cancer. Colloids Surf. B Biointerfaces 2015, 135, 689–698. [Google Scholar] [CrossRef]
- Arya, G.; Das, M.; Sahoo, S.K. Evaluation of curcumin loaded chitosan/PEG blended PLGA nanoparticles for effective treatment of pancreatic cancer. Biomed. Pharmacother. 2018, 102, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.W.; Jung, W.; Choi, C.; Kim, S.Y.; Son, A.; Kim, H.; Park, H.C. Fucoidan-manganese dioxide nanoparticles potentiate radiation therapy by co-targeting tumor hypoxia and angiogenesis. Mar. Drugs 2018, 16, 510. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Li, H.; Zhang, L.M.; Zhu, Z.; Yang, L. Enhancement of phototoxicity against human pancreatic cancer cells with photosensitizer-encapsulated amphiphilic sodium alginate derivative nanoparticles. Int. J. Pharm. 2014, 473, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Wade, S.J.; Zuzic, A.; Foroughi, J.; Talebian, S.; Aghmesheh, M.; Moulton, S.E.; Vine, K.L. Preparation and in vitro assessment of wet-spun gemcitabine-loaded polymeric fibers: Towards localized drug delivery for the treatment of pancreatic cancer. Pancreatology 2017, 17, 795–804. [Google Scholar] [CrossRef]
- Flynn, J.F.; Wong, C.; Wu, J.M. Anti-EGFR therapy: Mechanism and advances in clinical efficacy in breast cancer. J. Oncol. 2009, 2009, 526963. [Google Scholar] [CrossRef]
- Xiao, J.; Yu, H. Gemcitabine conjugated chitosan and double antibodies (Abc-GC-gemcitabine nanoparticles) enhanced cytoplasmic uptake of gemcitabine and inhibit proliferation and metastasis in human SW1990 pancreatic cancer cells. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2017, 23, 1613–1620. [Google Scholar] [CrossRef]
- Etman, S.M.; Abdallah, O.Y.; Elnaggar, Y.S. Novel fucoidan based bioactive targeted nanoparticles from Undaria pinnatifida for treatment of pancreatic cancer. Int. J. Biol. Macromol. 2020, 145, 390–401. [Google Scholar] [CrossRef]
- Verdaguer, H.; Sauri, T.; Macarulla, T. Predictive and prognostic biomarkers in personalized gastrointestinal cancer treatment. J. Gastrointest. Oncol. 2017, 8, 405–410. [Google Scholar] [CrossRef][Green Version]
- El Bairi, K.; Atanasov, A.G.; Amrani, M.; Afqir, S. The arrival of predictive biomarkers for monitoring therapy response to natural compounds in cancer drug discovery. Biomed. Pharmacother. 2019, 109, 2492–2498. [Google Scholar] [CrossRef]
- Herbst, B.; Zheng, L. Precision medicine in pancreatic cancer: Treating every patient as an exception. Lancet Gastroenterol. Hepatol. 2019, 4, 805–810. [Google Scholar] [CrossRef]
- Moreira, L.; Bakir, B.; Chatterji, P.; Dantes, Z.; Reichert, M.; Rustgi, A.K. Pancreas 3D organoids: Current and future aspects as a research platform for personalized medicine in pancreatic cancer. Cell. Mol. Gastroenterol. Hepatol. 2018, 5, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A.M.; Marques, A.P.; Silva, T.H.; Reis, R.L. Evaluation of the potential of collagen from codfish skin as a biomaterial for biomedical applications. Mar. Drugs 2018, 16, 495. [Google Scholar] [CrossRef] [PubMed]
- Barros, A.A.; Aroso, I.M.; Silva, T.H.; Mano, J.F.; Duarte, A.R.C.; Reis, R.L. In vitro bioactivity studies of ceramic structures isolated from marine sponges. Biomed. Mater. 2016, 11, 045004. [Google Scholar] [CrossRef] [PubMed]
- Moreira-Silva, J.; Diogo, G.S.; Marques, A.L.; Silva, T.H.; Reis, R.L. Marine collagen isolation and processing envisaging biomedical applications. In Biomaterials from Nature for Advanced Devices and Therapies; Neves, N.M., Reis, R.L., Eds.; Wiley: Hoboken, NJ, USA, 2016; pp. 16–36. [Google Scholar] [CrossRef]
- Marques, C.F.; Diogo, G.S.; Pina, S.; Oliveira, J.M.; Silva, T.H.; Reis, R.L. Collagen-based bioinks for hard tissue engineering applications: A comprehensive review. J. Mater. Sci. Mater. Med. 2019, 30, 1–12. [Google Scholar] [CrossRef]
- Paradiso, F.; Fitzgerald, J.; Yao, S.; Barry, F.; Taraballi, F.; Gonzalez, D.; Francis, L. Marine collagen substrates for 2D and 3D ovarian cancer cell systems. Front. Bioeng. Biotechnol. 2019, 7, 343–351. [Google Scholar] [CrossRef]
- Tomás-Bort, E.; Kieler, M.; Sharma, S.; Candido, J.B.; Loessner, D. 3D approaches to model the tumor microenvironment of pancreatic cancer. Theranostics 2020, 10, 5074–5089. [Google Scholar] [CrossRef]
- Schizas, D.; Charalampakis, N.; Kole, C.; Economopoulou, P.; Koustas, E.; Gkotsis, E.; Karamouzis, M.V. Immunotherapy for pancreatic cancer: A 2020 update. Cancer Treat. Rev. 2020, 86, 102016. [Google Scholar] [CrossRef]
- Hewitt, D.B.; Nissen, N.; Hatoum, H.; Musher, B.; Seng, J.; Coveler, A.L.; Lavu, H. A phase 3 randomized clinical trial of chemotherapy with or without algenpantucel-L (hyperacute-pancreas) immunotherapy in subjects with borderline resectable or locally advanced unresectable pancreatic cancer. Ann. Surg. 2022, 275, 45–53. [Google Scholar] [CrossRef]
- National Library of Medicine (U.S.). 18 May 2020–Present. Nivolumab and Ipilimumab and Radiation Therapy in Metastatic, Microsatellite Stable Pancreatic Cancer. Clinical Trial Identifier: NCT04361162. Available online: https://clinicaltrials.gov/ct2/show/NCT04361162 (accessed on 19 October 2022).
- Saeed, A.F.; Su, J.; Ouyang, S. Marine-derived drugs: Recent advances in cancer therapy and immune signaling. Biomed. Pharmacother. 2021, 134, 111091. [Google Scholar] [CrossRef]
- Amer, N.F.; Knaan, T.L. Natural Products of Marine Origin for the Treatment of Colorectal and Pancreatic Cancers: Mechanisms and Potential. Int. J. Mol. Sci. 2022, 23, 8048. [Google Scholar] [CrossRef]
| Tested Drugs | Phase Trial | PC Type | Main Outcomes | Ref. |
|---|---|---|---|---|
| Cisplatin, cytarabine, caffeine, and fluorouracil | II | Advanced PDAC | Produced significant responses in PC but the toxicity was significant | [146] |
| Fluorouracil Plus Folinic Acid vs. Gemcitabine | III | Resected PDAC | Compared with the use of fluorouracil plus folinic acid, gemcitabine did not result in improved overall survival in patients with totally resected PC | [147] |
| Eribulin mesylat | II | Gemcitabine refractory PDAC | The drug was well tolerated and did not result in any responses in refractory PC | [148] |
| Paclitaxel plus Bryostatin-1 | II | Locally advanced or metastatic PDAC | The combination of these drugs was not an effective therapy | [78] |
| Trabectedin | II | Gemcitabine refractory and Metastatic PDAC | Some ability to modulate inflammatory process was reported but single-agent trabectedin had no activity as salvage therapy | [149] |
| Lurbinectedin | II | Advanced PC with DNA repair mutations | Ongoing | [150] |
| Marizomib and vorinostat | I | Metastatic PDAC | The combination of the full dose was tolerable in patients, with safety findings consistent with either drug alone | [151] |
| Hydroxychloroquine in combination with gemcitabine and nab-paclitaxel | II | Resettable PDAC | Greater tumour response, improved serum biomarker response, and evidence of autophagy inhibition and immune activity were reported | [152] |
| Nab-Paclitaxel plus gemcitabine | II | Locally advanced PD | Tolerability and activity for locally advanced PC were reported | [153] |
| System | Drug | Cell Type/Animal Model | Main Outcomes | Ref. |
|---|---|---|---|---|
| Chitosan/glyceryl monooleate NPs | Gemcitabine | - BxPC-3 - MIA-PaCa-2 | Drug-loaded NPs increased GEM accumulation and enhanced cytotoxic effects more than the naïve drug. | [161] |
| Herceptin-conjugated gemcitabine-loaded chitosan NPs (HER2-Gem-CS-NPs) | Gemcitabine | - MIA-PaCa-2 - PANC 1 | The targeting capability of the developed system was validated. Increased cytotoxic effects were observed when compared with free drug and unconjugated NPs | [162] |
| Anti-EGFR Glycol-chitosan NPs loaded with gemcitabine ((Abc)-GC-Gem) | Gemcitabine | - SW1990 | (Abc)-GC-Gem NPs targeted and inhibited cancer cell proliferation. | [163] |
| Folate-chitosan-gemcitabine core-shell NPs (FA-Chi-Gem) | Gemcitabine | - COLO357 - Orthotopic xenograft mice model | NPs inhibited pancreatic cancer cell proliferation and impaired tumour growth in vivo | [164] |
| Metformin encapsulated O-Carboxymethyl chitosan NPs (O-CMC-met) | Metformin | - MIA-PaCa-2 - Mice model | NPs downregulated gene expression (p21, vanin 1, and MMP9) in pancreatic cancer cells. There was a normal distribution of the NPs in vivo and no adverse effects over major organs | [165] |
| Chitosan-coated solid-lipid NPs | Aspirin Curcumin Sulforaphane | - BALB/c mice | No changes in blood counts, no abnormalities in different organs, and no toxicities | [166] |
| Chitosan NPs | Quercetin 5-fluorouracil | - MIA-PaCa-2 | The encapsulation of both drugs increased cytotoxicity more than single-loaded NPs | [167] |
| Chitosan and PEG-coated curcumin-loaded Poly d,l-lactide-co-glycolide | Curcumin | - PANC-1 - MIA-PaCa-2 | In vitro results demonstrated that NPs enhanced the cellular uptake, cytotoxicity, pro-apoptotic, anti-migratory, and anti-invasive properties as compared to free drugs | [168] |
| Fucoidan-coated manganese dioxide nanoparticles (Fuco-MnO2-NPs) | - | - AsPC-1 - BxPC-3 - BxPC3 xenograft mouse model | Fuco-MnO2-NPs and RT resulted in a greater tumour growth delay than RT alone | [169] |
| Fucoidan/lactoferrin NPs | - | - PANC-1 | NPs increased the cytotoxicity and prevented the migration and invasion of pancreatic cancer cells | [175] |
| Photosensitizer-encapsulated amphiphilic sodium alginate derivative (Photosan-CSAD) | - | - PANC-1 | NPs increased fluorescence activity and ROS generation, resulting in strong phototoxicity | [170] |
| Fibres-spun from alginate or chitosan loaded with gemcitabine | Gemcitabine | - MIA-PaCa-2 - PANC-1 | Demonstrated a decrease in cells viability when compared with control fibres (no-gemcitabine) | [171] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandes, A.S.; Oliveira, C.; Reis, R.L.; Martins, A.; Silva, T.H. Marine-Inspired Drugs and Biomaterials in the Perspective of Pancreatic Cancer Therapies. Mar. Drugs 2022, 20, 689. https://doi.org/10.3390/md20110689
Fernandes AS, Oliveira C, Reis RL, Martins A, Silva TH. Marine-Inspired Drugs and Biomaterials in the Perspective of Pancreatic Cancer Therapies. Marine Drugs. 2022; 20(11):689. https://doi.org/10.3390/md20110689
Chicago/Turabian StyleFernandes, Andreia S., Catarina Oliveira, Rui L. Reis, Albino Martins, and Tiago H. Silva. 2022. "Marine-Inspired Drugs and Biomaterials in the Perspective of Pancreatic Cancer Therapies" Marine Drugs 20, no. 11: 689. https://doi.org/10.3390/md20110689
APA StyleFernandes, A. S., Oliveira, C., Reis, R. L., Martins, A., & Silva, T. H. (2022). Marine-Inspired Drugs and Biomaterials in the Perspective of Pancreatic Cancer Therapies. Marine Drugs, 20(11), 689. https://doi.org/10.3390/md20110689











