Chemical Characterization of Atlantic Cod (Gadus morhua) Collagen Hydrolyzed Using Enzyme Preparation Derived from Red King Crab (Paralithodes camtschaticus) and Its Potential as a Core Component of Bacterial Culture Medium
Abstract
:1. Introduction
2. Results and Discussion
2.1. Proteolytic Activity of the EPs Using a Standard Substrate
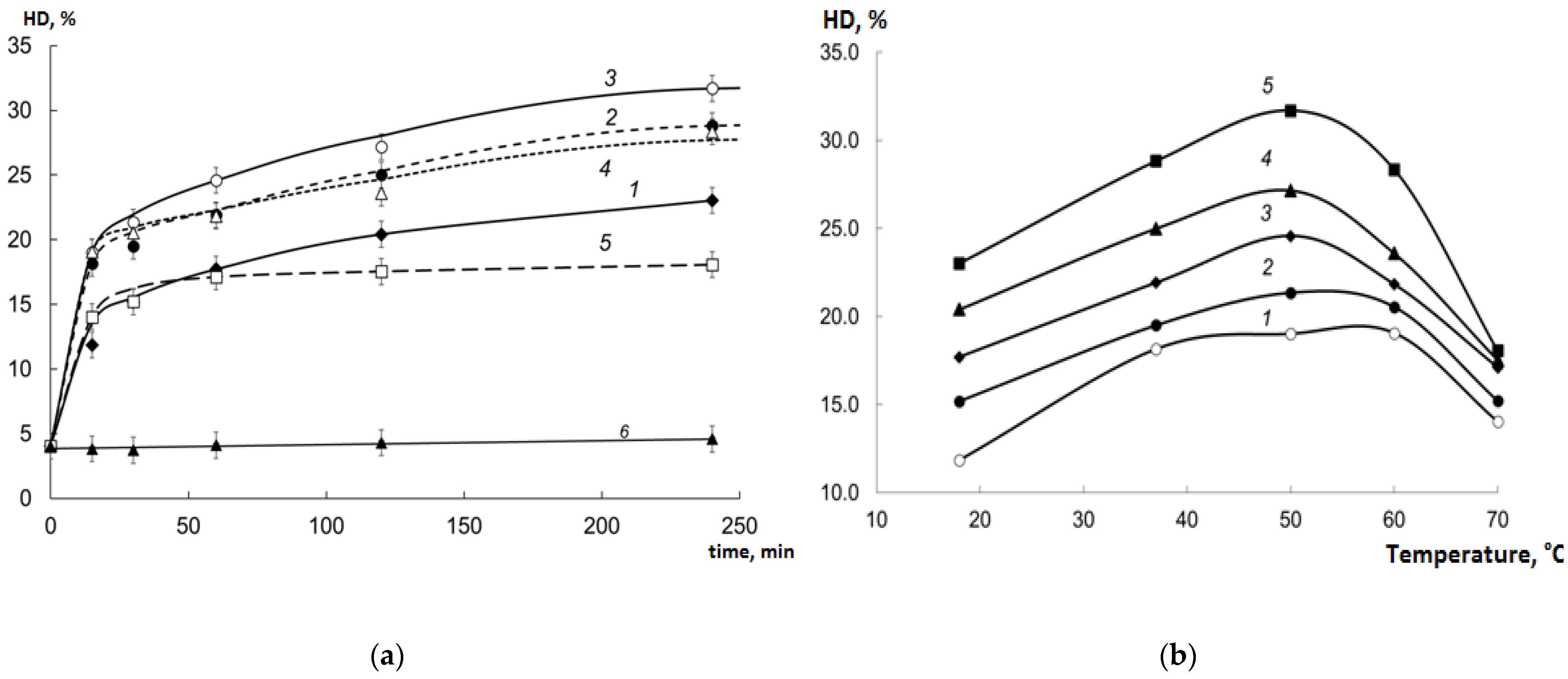
2.2. HD of Collagen Hydrolysates (CHs) Prepared Using Various EPs
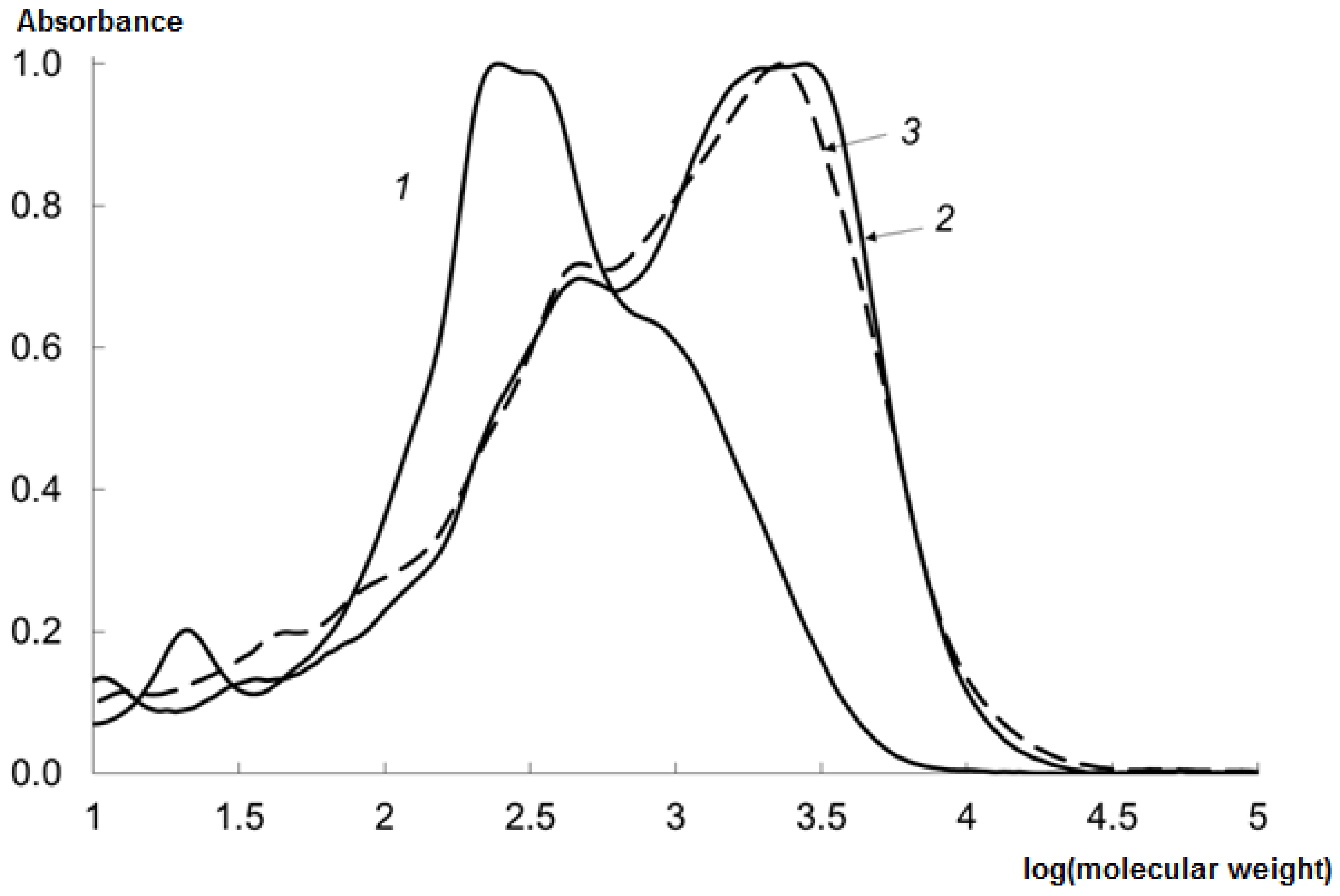
2.3. Molecular Weight Distribution (MWD) of CHs Prepared Using Various EPs
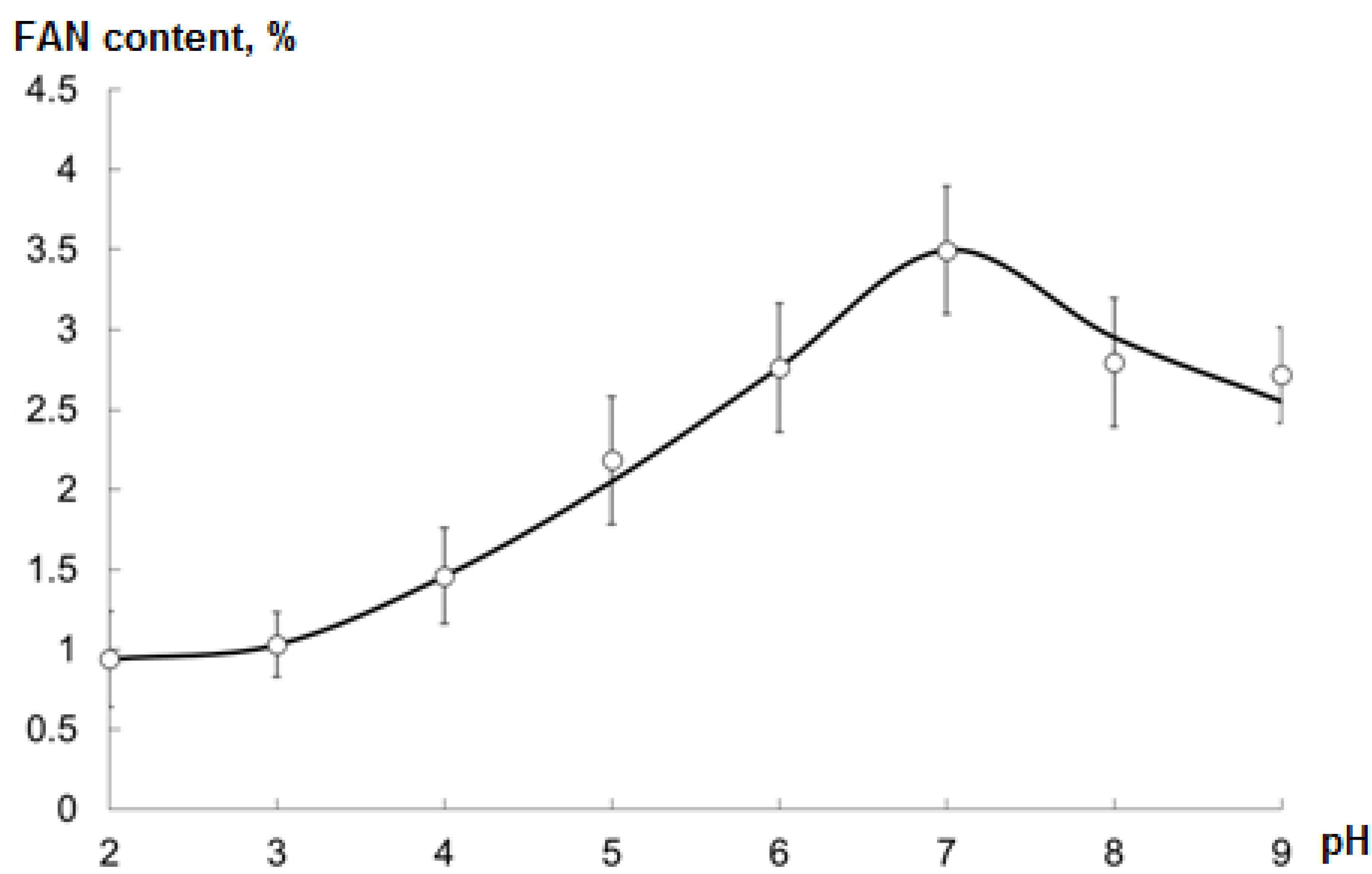
2.4. Optimal pH during Hydrolysis
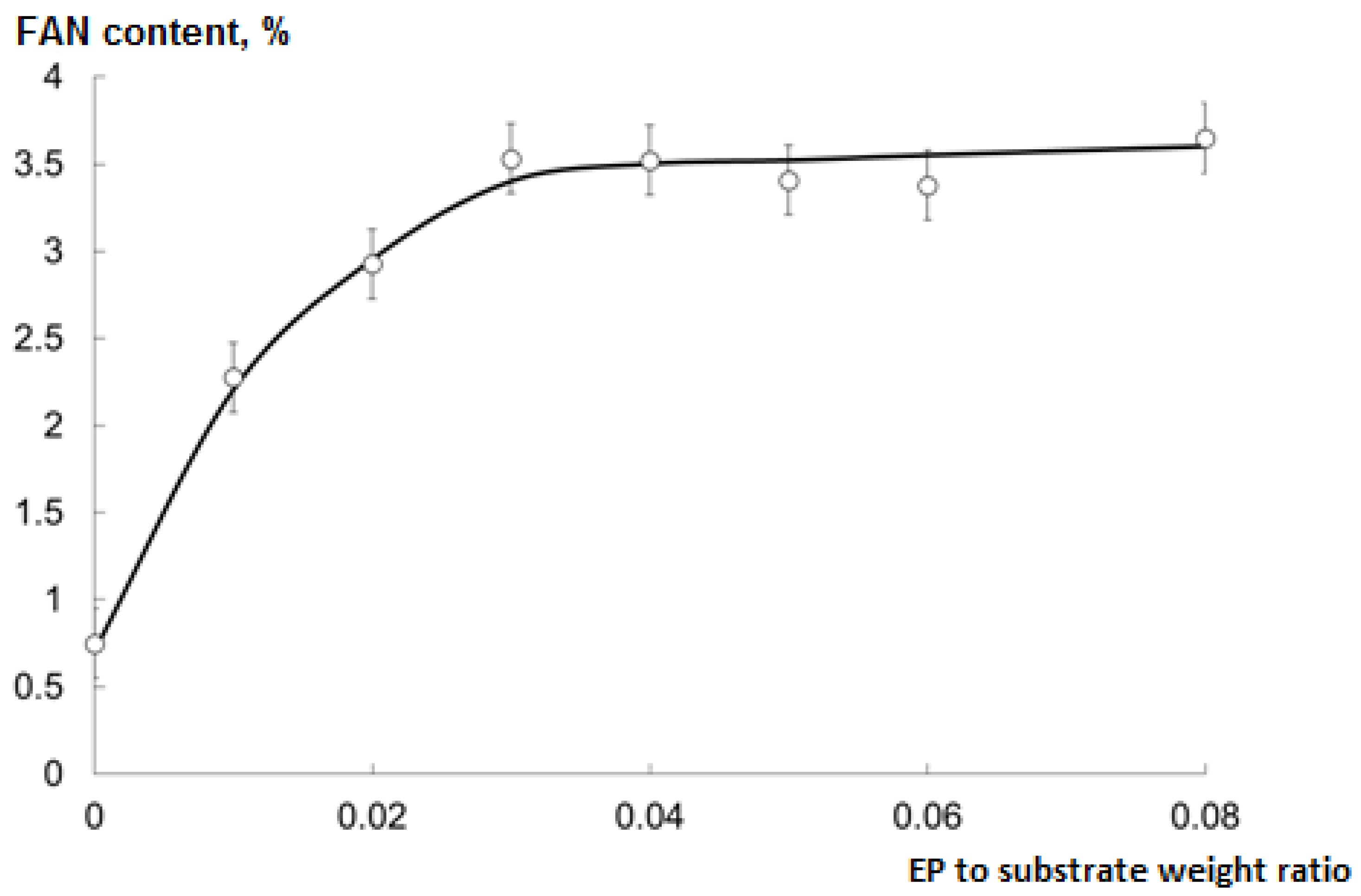
2.5. Optimal EP to Substrate Ratio for Hydrolysis
2.6. Temperature and Temporal Regime of Hydrolysis
2.7. Analysis of MWD of the CHs
2.8. Amino Acid Composition of the Untreated Collagen and CHs
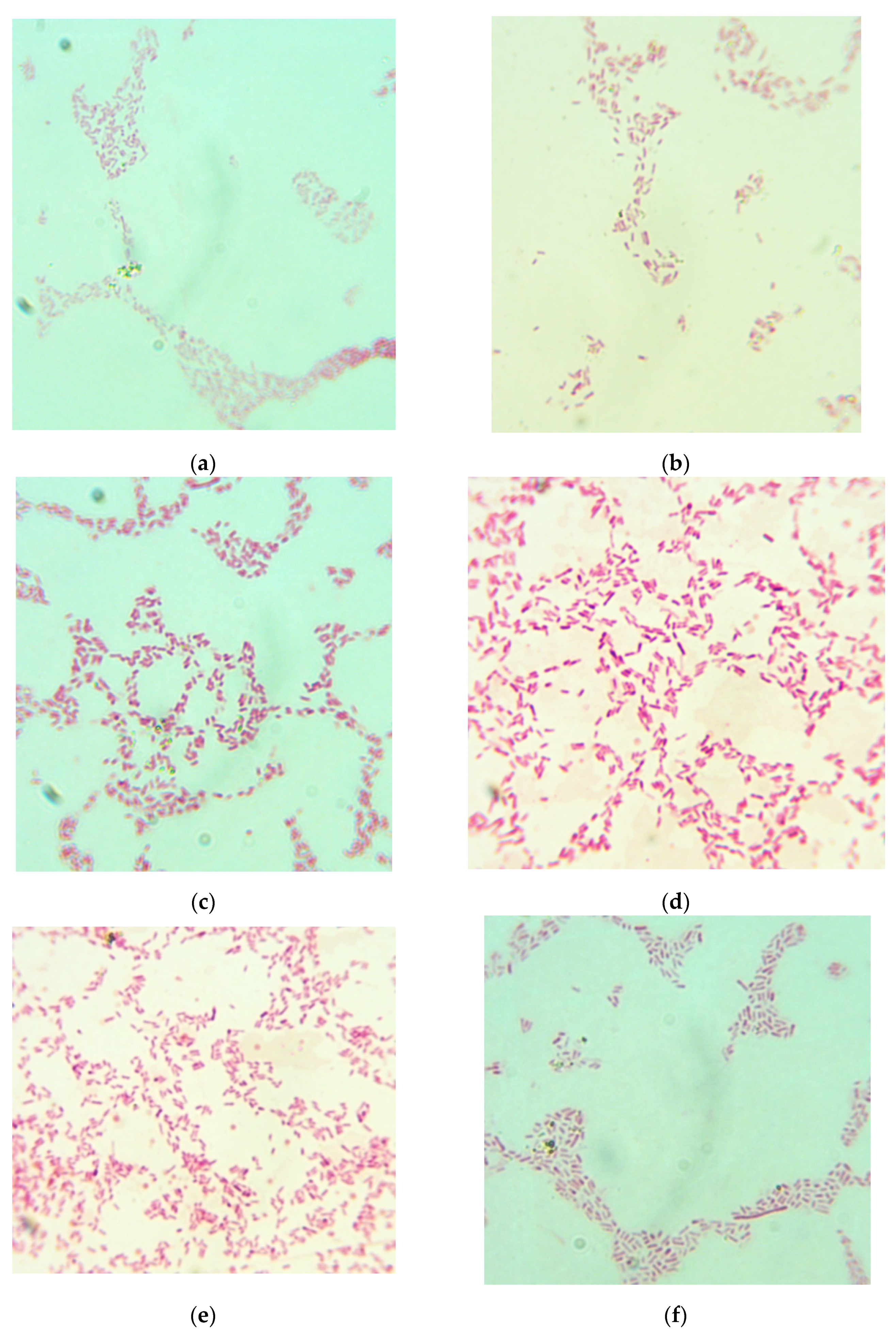
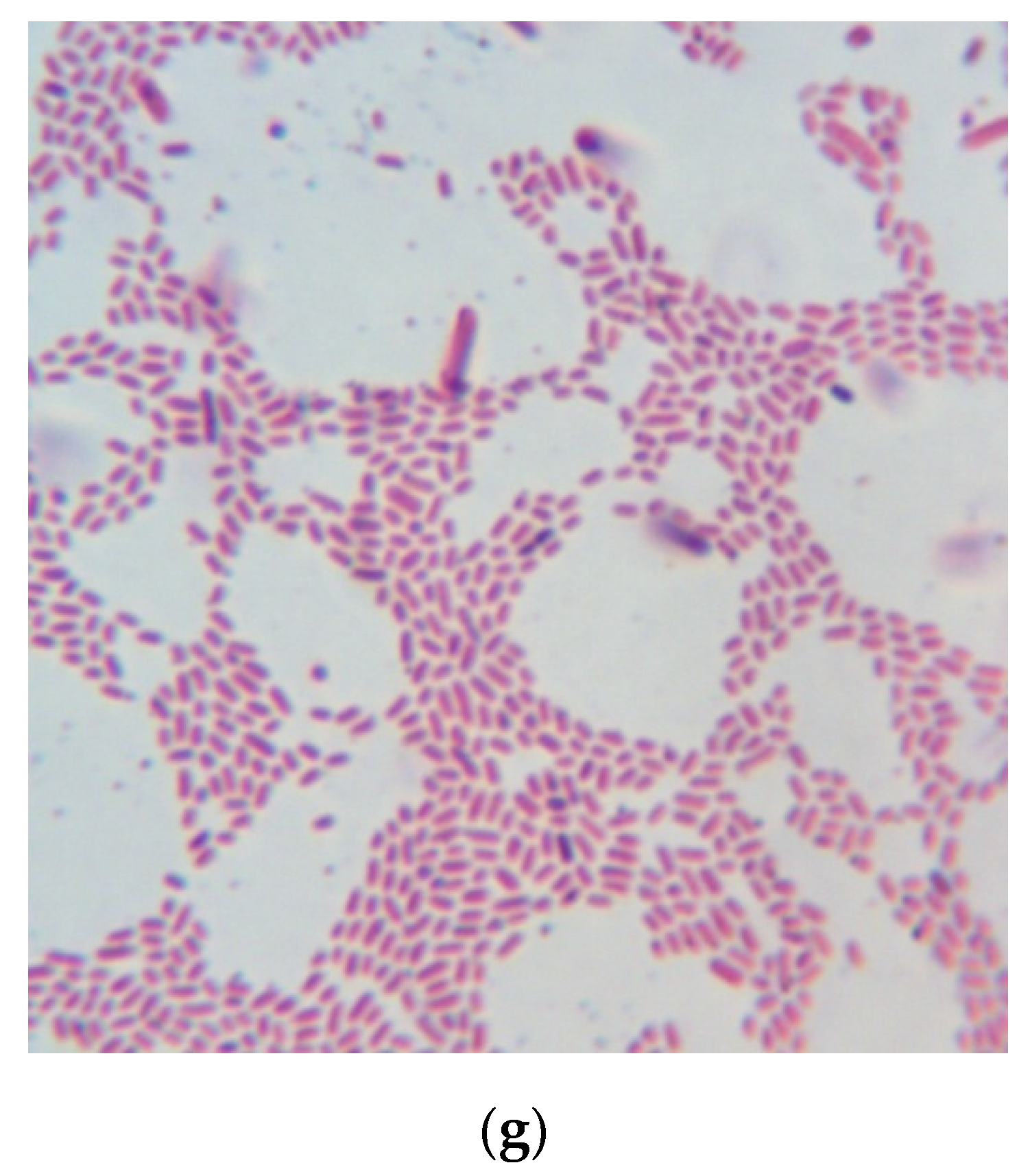
2.9. Bacterial Culture Medium Testing
2.10. Cost Analysis of Bacterial Culture Media
3. Materials and Methods
3.1. EPsfor Hydrolysis
3.2. Estimation of Proteolytic Activity of the EPs Using a Standard Substrate
3.3. Collagen Isolation and Hydrolysis
3.4. FAN Content and HD Determination
3.5. Determination of pH
3.6. MWD Analysis
3.7. Amino Acid Analysis
3.8. Bacterial Culture Medium Testing
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Fratzl, P. Collagen. Structure and Mechanics; Springer Science + Business Media: New York, NY, USA, 2008. [Google Scholar]
- Karsdal, M. Biochemistry of Collagens, Laminins and Elastin. Structure, Function and Biomarkers; Academic Press: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Moreira-Silva, J.; Diogo, G.; Marques, A.; Silva, T.; Reis, R. Marine collagen isolation and processing envisaging biomedical applications. In Biomaterials from Nature for Advanced Devices and Therapies; Neves, N., Reis, R., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2016; pp. 16–36. [Google Scholar]
- Suresh, P.; Kudre, T.; Johny, L. Sustainable valorization of seafood processing by-product/discard. In Waste to Wealth; Singhania, R., Agarwal, R., Kumar, R., Sukumaran, R., Eds.; Springer: Singapore, 2018; pp. 111–139. [Google Scholar]
- Pal, G.K.; Suresh, P.V. Sustainable valorisation of seafood by-products: Recovery of collagen and development of collagen-based novel functional food ingredients. Innov. Food Sci. Emerg. Technol. 2016, 37, 201–215. [Google Scholar] [CrossRef]
- Schmidt, M.M.; Dornelles, R.C.P.; Mello, R.O.; Kubota, E.H.; Mazutti, M.A.; Kempka, A.P.; Demiate, I.M. Collagen extraction process. Int. Food Res. J. 2016, 23, 913–922. [Google Scholar]
- Subhan, F.; Hussain, Z.; Tauseef, I.; Shehzad, A.; Wahid, F. A review on recent advances and applications of fish collagen. Crit. Rev. Food Sci. Nutr. 2021, 61, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Nuñez, S.M.; Guzmán, F.; Valencia, P.; Almonacid, S.; Cárdenas, C. Collagen as a source of bioactive peptides: A bioinformatics approach. Electron. J. Biotechnol. 2020, 48, 101–108. [Google Scholar] [CrossRef]
- Hakuta, A.; Yamaguchi, Y.; Okawa, T.; Yamamoto, S.; Sakai, Y.; Aihara, M. Anti-inflammatory effect of collagen tripeptide in atopic dermatitis. J. Dermatol. Sci. 2017, 88, 357–364. [Google Scholar] [CrossRef] [Green Version]
- Zamorano-Apodaca, J.C.; García-Sifuentes, C.O.; Carvajal-Millán, E.; Vallejo-Galland, B.; Scheuren-Acevedo, S.M.; Lugo-Sánchez, M.E. Biological and functional properties of peptide fractions obtained from collagen hydrolysate derived from mixed by-products of different fish species. Food Chem. 2020, 331, 127350. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Wu, C.; Liu, D.; Yang, X.; Huang, J.; Zhang, J.; Liao, B.; He, H. Antioxidant and anti-freezing peptides from salmon collagen hydrolysate prepared by bacterial extracellular protease. Food Chem. 2018, 248, 346–352. [Google Scholar] [CrossRef]
- Song, H.; Li, B. Beneficial Effects of Collagen Hydrolysate: A Review on Recent Developments. Biomed. J. Sci. Tech. Res. 2017, 1, 458–461. [Google Scholar] [CrossRef]
- Shu, Y.; Ren, H.; Ao, R.; Qi, W.C.; Zhang, Z.S. Comparison of physical and chemical characteristics of collagen from the skin of cod (Gadusmacrocephaius). Genet. Mol. Res. 2017, 16, gmr16027940. [Google Scholar] [CrossRef]
- Sun, L.; Hou, H.; Li, B.; Zhang, Y. Characterization of acid- and pepsin-soluble collagen extracted from the skin of Nile tilapia (Oreochromis niloticus). Int. J. Biol. Macromol. 2017, 99, 8–14. [Google Scholar] [CrossRef]
- Klimova, O.A.; Chebotarev, V.Y. Collagenolytic protease complex from hepatopancreas of kamchatka crab: Enzyme activity of individual components. Bull Exp. Biol. Med. 1999, 128, 994–998. [Google Scholar] [CrossRef]
- Ponomareva, T.; Timchenko, M.; Filippov, M.; Lapaev, S.; Sogorin, E. Prospects of Red King Crab Hepatopancreas Processing: Fundamental and Applied Biochemistry. Recycling 2021, 6, 3. [Google Scholar] [CrossRef]
- Mukhin, V.A.; Novikov, V.Y. Enzymatic protein-hydrolysate in poultry feed. Zootechniya 2001, 10, 21–23. [Google Scholar]
- Mukhin, V.A.; Novikov, V.Y.; Ryzhikova, L.S. A Protein Hydrolysate Enzymatically Produced from the Industrial Waste of Processing Icelandic Scallop Chlamysislandica. Appl. Biochem. Microbiol. 2001, 37, 292–296. [Google Scholar] [CrossRef]
- Afinogenov, G.E.; Domorad, A.A. Nutrient medium for clostridia isolation. USSR Patent SU 1,751,987 A1, 1 October 1996. [Google Scholar]
- Amerkhanova, A.M.; Gins, V.K.; Aleshkin, V.A.; Bandojan, A.K.; Khachatrjan, G.V.; Zubkova, E.S.; Gins, M.S.; Kononkov, P.F.; Bojarkina, L.A. Nutrient medium for culturing bifidobacteria. Russian Federation Patent RU 2,214,454 C2, 20 October 2003. [Google Scholar]
- Liu, Z.; Oliveira, A.C.; Su, Y.C. Purification and characterization of pepsin-solubilized collagen from skin and connective tissue of giant red sea cucumber (Parastichopus californicus). J. Agric. Food Chem. 2010, 58, 1270–1274. [Google Scholar] [CrossRef] [PubMed]
- Duan, R.; Zhang, J.; Du, X.; Yao, X.; Konno, K. Properties of collagen from skin, scale and bone of carp (Cyprinus carpio). Food Chem. 2009, 112, 702–706. [Google Scholar] [CrossRef]
- Rigby, B.J. Amino-acid composition and thermal stability of the skin collagen of the Antarctic ice-fish. Nature 1968, 219, 166–167. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.L.; Peng, M.; Li, J.; Tang, B.L.; Shao, X.; Zhao, F.; Liu, C.; Zhang, X.Y.; Li, P.Y.; Shi, M.; et al. Preparation and functional evaluation of collagen oligopeptide-rich hydrolysate from fish skin with the serine collagenolytic protease from Pseudoalteromonas sp. SM9913. Sci. Rep. 2017, 7, 15716. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Guillén, M.C.; Turnay, J.; Fernández-Díaz, M.D.; Ulmo, N.; Lizarbe, M.A.; Montero, P. Structural and physical properties of gelatin extracted from different marine species: A comparative study. Food Hydrocoll. 2002, 16, 25–34. [Google Scholar] [CrossRef] [Green Version]
- Top Quality Factory Supply Fish Skin Cod Collagen Peptide. Available online: https://www.alibaba.com/product-detail/Top-Quality-Factory-Supply-Fish-Skin_62437125645.html (accessed on 13 August 2021).
- Industrial Chemicals. Sodium Chloride. Available online: https://trade-him.ru/catalog/promyshlennaya_himiya/natriy_hloristyy.html?ymclid=16287751382323524986000002 (accessed on 13 August 2021).
- Laboratory Supplies. Bacteriological Agar. Available online: http://biom-msk.ru/agar-bakteriologicheskij/ (accessed on 13 August 2021).
- Laboratory Supplies. GRM-Agar. Available online: http://biom-msk.ru/pitatelnyj-agar-dlja-kultivirovanija-mikroorganizmov-suxoj/ (accessed on 13 August 2021).
- Morais, V.; Suárez, N. Economic Evaluation of Streptococcus Pneumoniae Culture Media. Am. J. Biochem. Biotechnol. 2016, 12, 133–138. [Google Scholar] [CrossRef] [Green Version]
- Farid, S.S.; Baron, M.; Stamatis, C.; Nie, W.; Coffman, J. Benchmarking biopharmaceutical process development and manufacturing cost contributions to R&D. MAbs 2020, 12, 1754999. [Google Scholar] [CrossRef] [PubMed]
- Folin, O.; Ciocalteu, V. On tyrosine and tryptophane determinations in proteins. J. Biol. Chem. 1927, 73, 627–650. [Google Scholar] [CrossRef]
- Anson, M.L. The estimation of pepsin, trypsin, papain, and cathepsin with hemoglobin. J. Gen. Physiol. 1938, 22, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Mikhailova, M.V.; Zolotarev, K.V.; Mikhailov, A.N.; Sanzhakov, M.A.; Farafonova, T.E. Differences in Nutritional Value of Various Fish Products Expressed by the Amino Acid Profiles of their Water-soluble Fractions. IJMH 2019, 4, 1–5. [Google Scholar] [CrossRef]


| Amino Acid | Untreated Collagen | CH Prepared at | Collagen | Gelatin [25] | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 18 °C | 37 °C | 50 °C | 60 °C | 70 °C | [21] | [22] | [23] | [24] | |||
| Asp | 59 | 58 | 58 | 58 | 58 | 65 | 51 | 53 | 52 | 50 | 52 |
| Glu | 71 | 66 | 67 | 67 | 67 | 75 | 71 | 80 | 75 | 79 | 78 |
| Ser | 62 | 58 | 60 | 58 | 57 | 61 | 67 | 59 | 69 | 52 | 64 |
| His | 8 | 6 | 7 | 7 | 8 | 9 | 8 | 8 | 8 | 7 | 8 |
| Gly | 358 | 347 | 384 | 428 | 393 | 395 | 332 | 342 | 345 | 325 | 344 |
| Thr | 21 | 20 | 21 | 19 | 20 | 20 | 23 | 23 | 25 | 28 | 25 |
| Arg | 41 | 40 | 42 | 38 | 40 | 39 | 51 | 54 | 51 | 48 | 56 |
| Ala | 92 | 91 | 95 | 87 | 91 | 89 | 106 | 107 | 107 | 102 | 96 |
| Tyr | 2 | 3 | 3 | 3 | 3 | 3 | 4 | 4 | 5 | 10 | 3 |
| Val | 14 | 16 | 16 | 15 | 15 | 15 | 19 | 19 | 19 | 15 | 18 |
| Met | 5 | 1 | 1 | 4 | 5 | 4 | 17 | 15 | 13 | 5 | 17 |
| Trp | 0 | 0 | 0 | 0 | 0 | 0 | ND1 | ND | ND | 0 | ND |
| Ile | 9 | 10 | 10 | 9 | 9 | 9 | 11 | 12 | 11 | 9 | 11 |
| Phe | 9 | 10 | 10 | 9 | 10 | 10 | 12 | 12 | 13 | 41 | 16 |
| Leu | 18 | 19 | 20 | 17 | 19 | 18 | 21 | 22 | 23 | 18 | 22 |
| Lys | 0 | 12 | 3 | 0 | 5 | 0 | 26 | 29 | 25 | 23 | 27 |
| Pro | 134 | 141 | 115 | 94 | 114 | 109 | 91 | 103 | 102 | 84 | 106 |
| Hyp | 60 | 64 | 52 | 49 | 49 | 42 | 55 | 51 | 53 | 70 | 50 |
| Taurine | 2 | 2 | 2 | 2 | 2 | 2 | ND | ND | ND | 0 | ND |
| Sample Number | Hydrolysis Regime (240 min each) | HD, % | Number of colony-forming unit (CFU) per 1 mL (Mean ± SD) | Diameter of Colony, mm | Efficiency Rate (Mean ± SD) |
|---|---|---|---|---|---|
| 1 | 18 °C | 23.0 | (12.0 ± 1.8) × 109 | 1–2 | 0.375 ± 0.056 |
| 2 | 37 °C | 28.8 | (17.6 ± 2.3) × 109 | 3–4 | 0.550 ± 0.072 |
| 3 | 37 °C without EP | 4.6 | (3.0 ± 0.5) × 109 | 1–3 | 0.094 ± 0.016 |
| 4 | 50 °C | 31.7 | (30.5 ± 4.6) × 109 | 2–3 | 0.953 ± 0.144 |
| 5 | 60 °C | 28.3 | (8.4 ± 0.8) × 109 | 4–5 | 0.262 ± 0.025 |
| 6 | 70 °C | 18.1 | (8.4 ± 1.0) × 109 | 0.5–2 | 0.262 ± 0.031 |
| Control medium | Not specified | Not specified | (32.0 ± 5.4) × 109 | 2–3 | 1.000 ± 0.169 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novikov, V.Y.; Shumskaya, N.V.; Mukhin, V.A.; Zolotarev, K.V.; Mikhailov, A.N.; Nakhod, V.I.; Mikhailova, M.V. Chemical Characterization of Atlantic Cod (Gadus morhua) Collagen Hydrolyzed Using Enzyme Preparation Derived from Red King Crab (Paralithodes camtschaticus) and Its Potential as a Core Component of Bacterial Culture Medium. Mar. Drugs 2021, 19, 472. https://doi.org/10.3390/md19080472
Novikov VY, Shumskaya NV, Mukhin VA, Zolotarev KV, Mikhailov AN, Nakhod VI, Mikhailova MV. Chemical Characterization of Atlantic Cod (Gadus morhua) Collagen Hydrolyzed Using Enzyme Preparation Derived from Red King Crab (Paralithodes camtschaticus) and Its Potential as a Core Component of Bacterial Culture Medium. Marine Drugs. 2021; 19(8):472. https://doi.org/10.3390/md19080472
Chicago/Turabian StyleNovikov, Vitaliy Yu., Nadezhda V. Shumskaya, Vyacheslav A. Mukhin, Konstantin V. Zolotarev, Anton N. Mikhailov, Valeriya I. Nakhod, and Marina V. Mikhailova. 2021. "Chemical Characterization of Atlantic Cod (Gadus morhua) Collagen Hydrolyzed Using Enzyme Preparation Derived from Red King Crab (Paralithodes camtschaticus) and Its Potential as a Core Component of Bacterial Culture Medium" Marine Drugs 19, no. 8: 472. https://doi.org/10.3390/md19080472
APA StyleNovikov, V. Y., Shumskaya, N. V., Mukhin, V. A., Zolotarev, K. V., Mikhailov, A. N., Nakhod, V. I., & Mikhailova, M. V. (2021). Chemical Characterization of Atlantic Cod (Gadus morhua) Collagen Hydrolyzed Using Enzyme Preparation Derived from Red King Crab (Paralithodes camtschaticus) and Its Potential as a Core Component of Bacterial Culture Medium. Marine Drugs, 19(8), 472. https://doi.org/10.3390/md19080472






