Impact of Phlorotannin Extracts from Fucus vesiculosus on Human Gut Microbiota
Abstract
:1. Introduction
2. Results and Discussion
2.1. Stability, Bioaccessibility and Antioxidant Activity of F. vesiculosus Extracts throughout the Simulated GIT
2.2. Prebiotic Effect
2.3. Evolution of the Gut Microbiota Profile Groups
2.4. Organic Acids Profile and pH Variation
3. Materials and Methods
3.1. Chemicals
3.2. Extraction Procedure
3.3. Gastrointestinal Digestion Simulation
3.4. Determination of the Phlorotannin Content and Antioxidant Activities
3.5. Determination of the Phlorotannin Content and Antioxidant Activities
3.6. In Vitro Fermentation Assays
3.7. Gut Microbiota Evaluation
3.7.1. DNA Extraction
3.7.2. Real-Time PCR for Microbial Analysis of Stool
3.7.3. Determination of Organic Acids
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Markowiak, P.; Ślizewska, K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Wen, L.; Duffy, A. Factors Influencing the Gut Microbiota, Inflammation, and Type 2 Diabetes. J. Nutr. 2017, 147, 1468S–1475S. [Google Scholar] [CrossRef] [Green Version]
- Nishida, A.; Inoue, R.; Inatomi, O.; Bamba, S.; Naito, Y.; Andoh, A. Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin. J. Gastroenterol. 2018, 11, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Demirci, M.; Tokman, H.B.; Uysal, H.K.; Demiryas, S.; Karakullukcu, A.; Saribas, S.; Cokugras, H.; Kocazeybek, B.S. Reduced Akkermansia muciniphila and Faecalibacterium prausnitzii levels in the gut microbiota of children with allergic asthma. Allergol. Immunopathol. 2019, 47, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Hevia, A.; Milani, C.; López, P.; Cuervo, A.; Arboleya, S.; Duranti, S.; Turroni, F.; González, S.; Suárez, A.; Gueimonde, M.; et al. Intestinal dysbiosis associated with systemic lupus erythematosus. MBio 2014, 5, e01548-14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, M.; Qian, Z.; Yin, J.; Xu, W.; Zhou, X. The role of intestinal microbiota in cardiovascular disease. J. Cell. Mol. Med. 2019, 23, 2343–2350. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Hua, D.; Wang, Q.; Yang, L.; Wang, X.; Luo, A.; Yang, C. The Role of Bacteria and Its Derived Metabolites in Chronic Pain and Depression: Recent Findings and Research Progress. Int. J. Neuropsychopharmacol. 2020, 23, 26–41. [Google Scholar] [CrossRef]
- Clapp, M.; Aurora, N.; Herrera, L.; Bhatia, M.; Wilen, E.; Wakefield, S. Gut microbiota’s effect on mental health: the gut-brain axis. Clin. Pract. 2017, 7, 987. [Google Scholar] [CrossRef]
- Hirschberg, S.; Gisevius, B.; Duscha, A.; Haghikia, A. Implications of diet and the gut microbiome in neuroinflammatory and neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 3109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [Green Version]
- Mohanty, D.; Misra, S.; Mohapatra, S.; Sahu, P.S. Prebiotics and synbiotics: Recent concepts in nutrition. Food Biosci. 2018, 26, 152–160. [Google Scholar] [CrossRef]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef] [Green Version]
- Costantini, L.; Molinari, R.; Farinon, B.; Merendino, N. Impact of omega-3 fatty acids on the gut microbiota. Int. J. Mol. Sci. 2017, 18, 2645. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alves-Santos, A.M.; Sugizaki, C.S.A.; Lima, G.C.; Naves, M.M.V. Prebiotic effect of dietary polyphenols: A systematic review. J. Funct. Foods 2020, 74, 104169. [Google Scholar] [CrossRef]
- González-Sarrías, A.; Romo-Vaquero, M.; García-Villalba, R.; Cortés-Martín, A.; Selma, M.V.; Espín, J.C. The Endotoxemia Marker Lipopolysaccharide-Binding Protein is Reduced in Overweight-Obese Subjects Consuming Pomegranate Extract by Modulating the Gut Microbiota: A Randomized Clinical Trial. Mol. Nutr. Food Res. 2018, 62, 1800160. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, S.; Guglielmetti, S.; Riso, P.; Arioli, S.; Klimis-Zacas, D.; Porrini, M. Six-week consumption of a wild blueberry powder drink increases Bifidobacteria in the human gut. J. Agric. Food Chem. 2011, 59, 12815–12820. [Google Scholar] [CrossRef]
- Catarino, D.M.; Silva, M.A.; Cardoso, M.S. Fucaceae: A Source of Bioactive Phlorotannins. Int. J. Mol. Sci. 2017, 18, 1327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Charoensiddhi, S.; Conlon, M.A.; Vuaran, M.S.; Franco, C.M.M.; Zhang, W. Polysaccharide and phlorotannin-enriched extracts of the brown seaweed Ecklonia radiata influence human gut microbiota and fermentation in vitro. J. Appl. Phycol. 2017, 29, 2407–2416. [Google Scholar] [CrossRef]
- Yuan, Y.; Zheng, Y.; Zhou, J.; Geng, Y.; Zou, P.; Li, Y.; Zhang, C. Polyphenol-Rich Extracts from Brown Macroalgae Lessonia trabeculate Attenuate Hyperglycemia and Modulate Gut Microbiota in High-Fat Diet and Streptozotocin-Induced Diabetic Rats. J. Agric. Food Chem. 2019, 67, 12472–12480. [Google Scholar] [CrossRef]
- McRae, J.M.; Kennedy, J.A. Wine and grape tannin interactions with salivary proteins and their impact on astringency: A review of current research. Molecules 2011, 16, 2348–2364. [Google Scholar] [CrossRef] [Green Version]
- Quan, W.; Tao, Y.; Lu, M.; Yuan, B.; Chen, J.; Zeng, M.; Qin, F.; Guo, F.; He, Z. Stability of the phenolic compounds and antioxidant capacity of five fruit (apple, orange, grape, pomelo and kiwi) juices during in vitro-simulated gastrointestinal digestion. Int. J. Food Sci. Technol. 2018, 53, 1131–1139. [Google Scholar] [CrossRef]
- Rodríguez-Roque, M.J.; Rojas-Graü, M.A.; Elez-Martínez, P.; Martín-Belloso, O. Changes in vitamin C, phenolic, and carotenoid profiles throughout in vitro gastrointestinal digestion of a blended fruit juice. J. Agric. Food Chem. 2013, 61, 1859–1867. [Google Scholar] [CrossRef]
- Grgić, J.; Šelo, G.; Planinić, M.; Tišma, M.; Bucić-Kojić, A. Role of the encapsulation in bioavailability of phenolic compounds. Antioxidants 2020, 9, 923. [Google Scholar] [CrossRef]
- Dima, C.; Assadpour, E.; Dima, S.; Jafari, S.M. Bioavailability and bioaccessibility of food bioactive compounds; overview and assessment by in vitro methods. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2862–2884. [Google Scholar] [CrossRef]
- Martelli, F.; Favari, C.; Mena, P.; Guazzetti, S.; Ricci, A.; Del Rio, D.; Lazzi, C.; Neviani, E.; Bernini, V. Antimicrobial and fermentation potential of himanthalia elongata in food applications. Microorganisms 2020, 8, 248. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.J.; Lee, D.G.; Park, S.H.; Kim, M.; Kong, C.S.; Kim, Y.Y.; Lee, S.H. Comparison of biological activities in Sargassum siliquanstrum fermented by isolated lactic acid bacteria. Biotechnol. Bioprocess Eng. 2015, 20, 341–348. [Google Scholar] [CrossRef]
- Gupta, S.; Abu-Ghannam, N.; Rajauria, G. Effect of heating and probiotic fermentation on the phytochemical content and antioxidant potential of edible Irish brown seaweeds. Bot. Mar. 2012, 55, 527–537. [Google Scholar] [CrossRef]
- Gupta, S.; Abu-Ghannam, N.; Scannell, A.G.M. Growth and kinetics of Lactobacillus plantarum in the fermentation of edible Irish brown seaweeds. Food Bioprod. Process. 2011, 89, 346–355. [Google Scholar] [CrossRef] [Green Version]
- Corona, G.; Ji, Y.; Anegboonlap, P.; Hotchkiss, S.; Gill, C.; Yaqoob, P.; Spencer, J.P.E.; Rowland, I. Gastrointestinal modifications and bioavailability of brown seaweed phlorotannins and effects on inflammatory markers. Br. J. Nutr. 2016, 15, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Guarner, F.; Malagelada, J.R. Gut flora in health and disease. Lancet 2003, 361, 512–519. [Google Scholar] [CrossRef]
- Madureira, A.R.; Campos, D.; Gullon, B.; Marques, C.; Rodríguez-Alcalá, L.M.; Calhau, C.; Alonso, J.L.; Sarmento, B.; Gomes, A.M.; Pintado, M. Fermentation of bioactive solid lipid nanoparticles by human gut microflora. Food Funct. 2016, 7, 516–529. [Google Scholar] [CrossRef] [PubMed]
- Campos, D.A.; Coscueta, E.R.; Vilas-Boas, A.A.; Silva, S.; Teixeira, J.A.; Pastrana, L.M.; Pintado, M.M. Impact of functional flours from pineapple by-products on human intestinal microbiota. J. Funct. Foods 2020, 67, 103830. [Google Scholar] [CrossRef]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [Green Version]
- Gerritsen, J.; Smidt, H.; Rijkers, G.T.; De Vos, W.M. Intestinal microbiota in human health and disease: The impact of probiotics. Genes Nutr. 2011, 6, 209–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dubin, K.; Pamer, E.G. Enterococci and Their Interactions with the Intestinal Microbiome. Bugs Drugs 2017, 5, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Allen, S.J.; Martinez, E.G.; Gregorio, G.V.; Dans, L.F. Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst. Rev. 2010, 11, CD003048. [Google Scholar] [CrossRef]
- Wang, C.; Zhao, J.; Zhang, H.; Lee, Y.K.; Zhai, Q.; Chen, W. Roles of intestinal bacteroides in human health and diseases. Crit. Rev. Food Sci. Nutr. 2020, 1–19. [Google Scholar] [CrossRef]
- Cockburn, D.W.; Koropatkin, N.M. Polysaccharide Degradation by the Intestinal Microbiota and Its Influence on Human Health and Disease. J. Mol. Biol. 2016, 428, 3230–3252. [Google Scholar] [CrossRef]
- Koliada, A.; Syzenko, G.; Moseiko, V.; Budovska, L.; Puchkov, K.; Perederiy, V.; Gavalko, Y.; Dorofeyev, A.; Romanenko, M.; Tkach, S.; et al. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 2017, 17, 4–9. [Google Scholar] [CrossRef] [Green Version]
- Larsen, N.; Vogensen, F.K.; Van Den Berg, F.W.J.; Nielsen, D.S.; Andreasen, A.S.; Pedersen, B.K.; Al-Soud, W.A.; Sørensen, S.J.; Hansen, L.H.; Jakobsen, M. Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS ONE 2010, 5, e9085. [Google Scholar] [CrossRef]
- Miquel, S.; Martín, R.; Rossi, O.; Bermúdez-Humarán, L.G.; Chatel, J.M.; Sokol, H.; Thomas, M.; Wells, J.M.; Langella, P. Faecalibacterium prausnitzii and human intestinal health. Curr. Opin. Microbiol. 2013, 16, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Dueñas, M.; Muñoz-González, I.; Cueva, C.; Jiménez-Girón, A.; Sánchez-Patán, F.; Santos-Buelga, C.; Moreno-Arribas, M.V.; Bartolomé, B. A survey of modulation of gut microbiota by dietary polyphenols. BioMed Res. Int. 2015, 2015, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Venegas, D.P.; De La Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short chain fatty acids (SCFAs)mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front. Immunol. 2019, 10, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Chambers, E.S.; Preston, T.; Frost, G.; Morrison, D.J. Role of Gut Microbiota-Generated Short-Chain Fatty Acids in Metabolic and Cardiovascular Health. Curr. Nutr. Rep. 2018, 7, 198–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernández-Veledo, S.; Vendrell, J. Gut microbiota-derived succinate: Friend or foe in human metabolic diseases? Rev. Endocr. Metab. Disord. 2019, 20, 439–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hosseini, E.; Grootaert, C.; Verstraete, W.; Van de Wiele, T. Propionate as a health-promoting microbial metabolite in the human gut. Nutr. Rev. 2011, 69, 245–258. [Google Scholar] [CrossRef] [PubMed]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Bäckhed, F. From dietary fiber to host physiology: Short-chain fatty acids as key bacterial metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef] [Green Version]
- Belenguer, A.; Duncan, S.H.; Holtrop, G.; Anderson, S.E.; Lobley, G.E.; Flint, H.J. Impact of pH on lactate formation and utilization by human fecal microbial communities. Appl. Environ. Microbiol. 2007, 73, 6526–6533. [Google Scholar] [CrossRef] [Green Version]
- Catarino, M.; Silva, A.; Mateus, N.; Cardoso, S. Optimization of Phlorotannins Extraction from Fucus vesiculosus and Evaluation of Their Potential to Prevent Metabolic Disorders. Mar. Drugs 2019, 17, 162. [Google Scholar] [CrossRef] [Green Version]
- Amarante, S.J.; Catarino, M.D.; Marçal, C.; Silva, A.M.S.; Ferreira, R.; Cardoso, S.M. Microwave-Assisted Extraction of Phlorotannins from Fucus vesiculosus. Mar. Drugs 2020, 18, 559. [Google Scholar] [CrossRef]
- Catarino, M.D.; Silva, A.; Cruz, M.T.; Mateus, N.; Silva, A.M.S.; Cardoso, S.M. Phlorotannins from Fucus vesiculosus: Modulation of inflammatory response by blocking NF-κB signaling pathway. Int. J. Mol. Sci. 2020, 21, 6897. [Google Scholar] [CrossRef] [PubMed]
- Pereira, O.; Catarino, M.; Afonso, A.; Silva, A.; Cardoso, S. Salvia elegans, Salvia greggii and Salvia officinalis Decoctions: Antioxidant Activities and Inhibition of Carbohydrate and Lipid Metabolic Enzymes. Molecules 2018, 23, 3169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
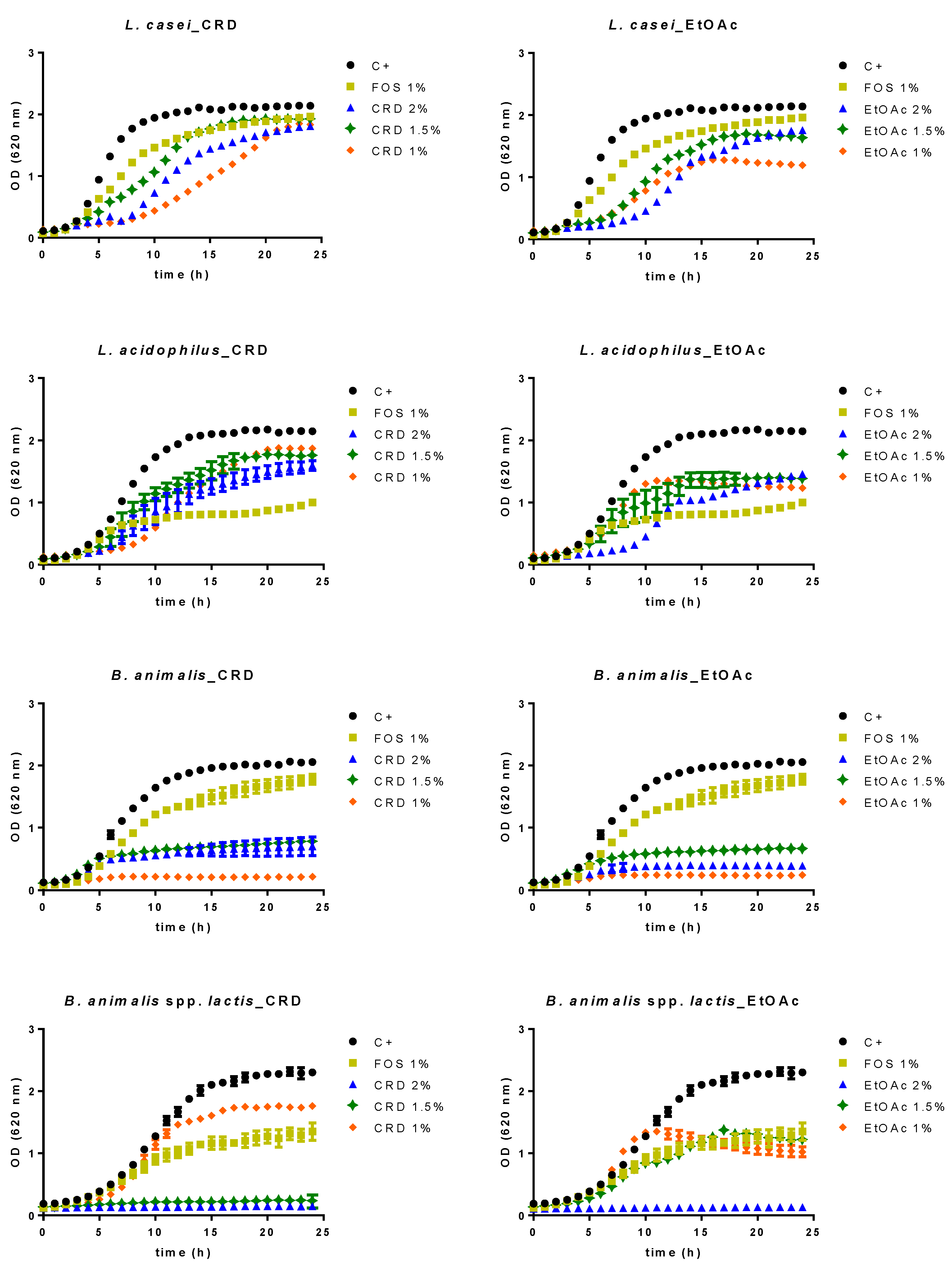
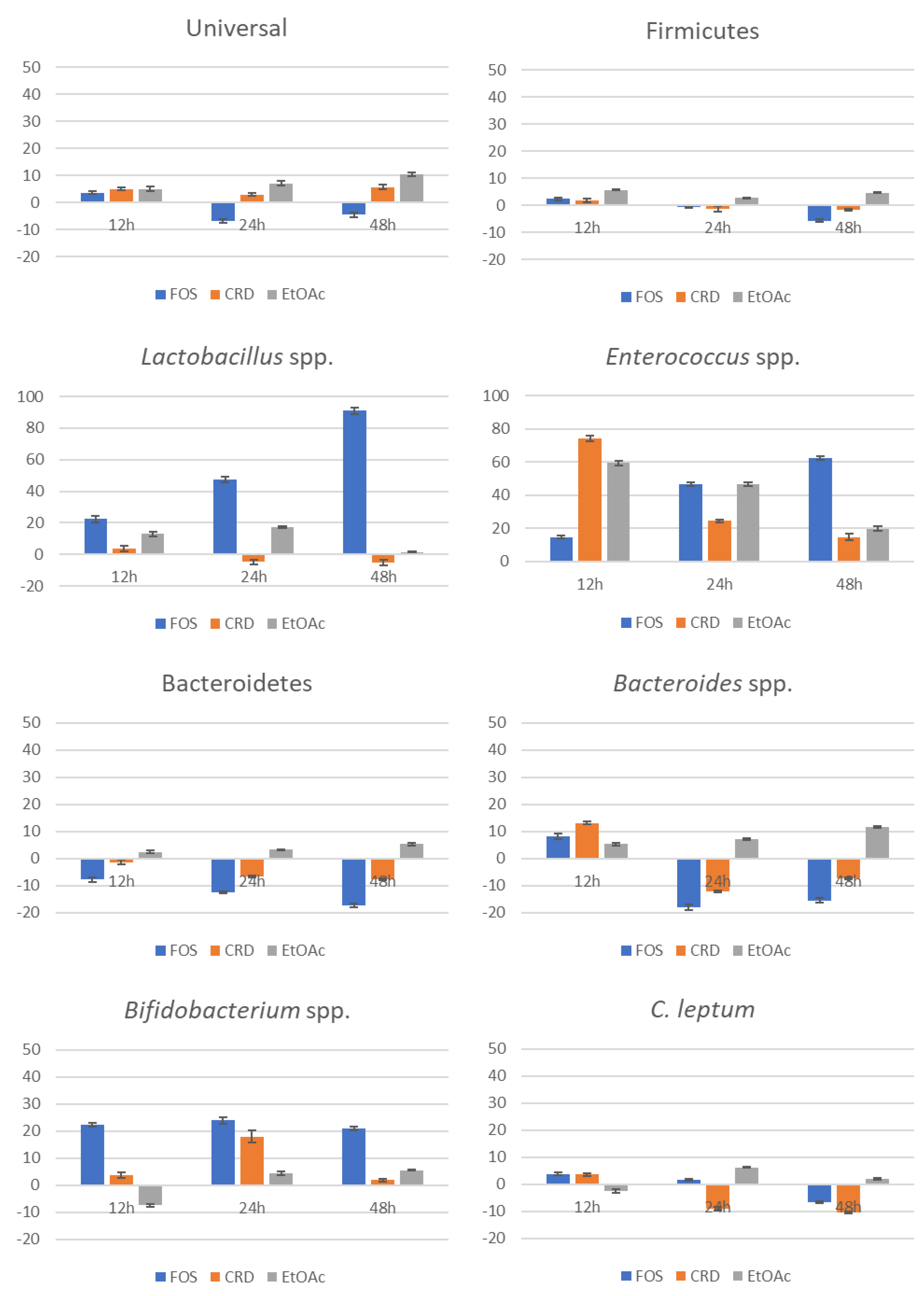
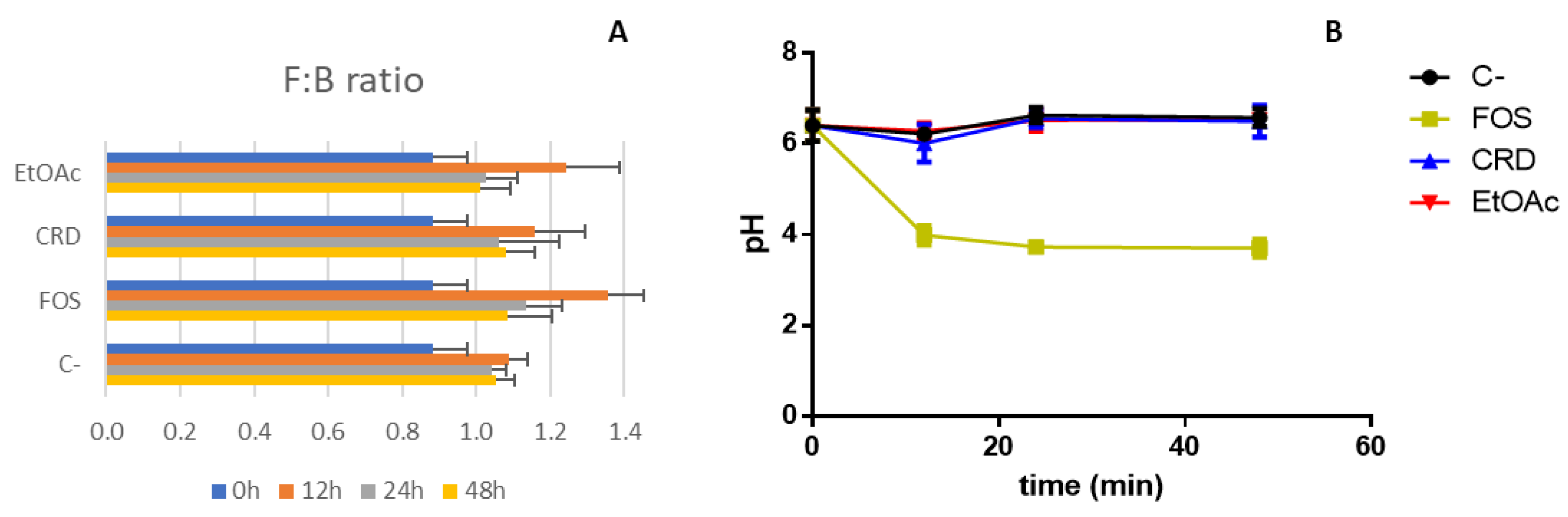
| Sample | GIT Stage | TPhC (mg PGE/g ext) | (1) NO● (IC50 μg/mL) | (2) O2●– (IC50 μg/mL) |
|---|---|---|---|---|
| CRD | Undigested | 9.93 ± 1.48 a | 161 ± 8.8 a | 417 ± 164.5 a |
| Mouth | 6.33 ± 2.96 b | 309 ± 105.2 b | 745 ± 88.2 b | |
| Stomach | 8.52 ± 1.16 a,b | 171 ± 27.1 a | 378 ± 26.6 a | |
| Intestine | 5.17 ± 0.70 b | 287 ± 27.2 a,b | 1105 ± 421.3 b | |
| Retentate * | 4.60 ± 0.26 b | 141 ± 9.1 a | 294 ± 19.3 a | |
| Permeate * | 1.40 ± 0.19 c | 2551 ± 30.7 c | 2580 ± 75.2 c | |
| EtOAc | Undigested | 17.39 ± 1.77 a | 45 ± 2.5 a | 118 ± 17.6 a |
| Mouth | 13.83 ± 0.74 b | 73 ± 11.0 a,b | 221 ± 1.1 a,b | |
| Stomach | 5.67 ± 0.91 c | 109 ± 7.1 a,b | 244 ± 0.4 a,b | |
| Intestine | 3.28 ± 0.55 c | 195 ± 38.5 b,c | 564 ± 19.9 c | |
| Retentate * | 2.97 ± 0.62 c,d | 281 ±16.1 c | 383 ± 18.2 b,c | |
| Permeate * | 0.37 ± 0.10 d | 1531 ± 52.2 d | 3074 ± 32.3 d | |
| Standard compound | - | 36 ± 0.9 | 6 ± 0.5 | |
| Division (Genus) | Number of Copies (n = 5) a |
|---|---|
| Universal | 7.52 ± 0.38 |
| Firmicutes | 4.76 ± 0.20 |
| Clostridium leptum | 4.97 ± 0.26 |
| Enterococcus spp. | 2.07 ± 0.63 |
| Lactobacillus spp. | 3.27 ± 0.72 |
| Bacteroidetes | 5.46 ± 0.63 |
| Bacteroides spp. | 3.76 ± 0.55 |
| Bifidobacterium spp. | 4.42 ± 0.45 |
| F:B ratio | 0.97 ± 0.23 |
| Organic Acids | Time (h) | Ctrl | FOS | CRD | EtOAc |
|---|---|---|---|---|---|
| Total | 0 | 2.38 ± 0.63 a;A | 2.38 ± 0.63 a;A | 2.38 ± 0.63 a;A | 2.38 ± 0.63 a;A |
| 12 | 5.24 ± 1.98 a;A | 10.89 ± 2.79 b;B | 7.43 ± 2.09 b;A | 7.76 ± 1.92 b;A,B | |
| 24 | 4.90 ± 1.59 a;A | 12.63 ± 2.37 b,c;B | 7.08 ± 2.45 b;A | 7.55 ± 1.75 b;A | |
| 48 | 4.10 ± 2.01 a;A | 14.78 ± 4.00 c;B | 5.04 ± 1.57 a,b;A | 6.38 ± 1.98 b;A | |
| Succinic acid | 0 | 0.45 ± 0.20 a;A | 0.45 ± 0.20 a;A | 0.45 ± 0.20 a;A | 0.45 ± 0.20 a;A |
| 12 | 0.77 ± 0.75 a;A | 1.85 ± 0.92 b;B | 2.29 ± 1.39 c;B | 2.15 ± 1.15 b;B | |
| 24 | 1.12 ± 0.53 a;A | 1.97 ± 0.58 b;B | 2.02 ± 0.93 b,c;A,B | 1.35 ± 0.50 a,b;A,B | |
| 48 | 0.74 ± 0.71 a;A | 2.03 ± 0.85 b;B | 1.12 ± 0.20 a,b;A,B | 1.40 ± 0.89 a,b;A,B | |
| Lactic acid | 0 | ND | ND | ND | ND |
| 12 | 1.21 ± 0.93 a;A | 3.91 ± 1.94 a;B | 0.87 ± 0.22 a;A | 0.87 ± 0.23 a;A | |
| 24 | 0.34 ± 0.14 a;A | 4.81 ± 0.75 a,b;B | 0.76 ± 0.58 a;A | 0.26 ± 0.16 a;A | |
| 48 | ND | 5.49 ± 2.14 b | ND | ND | |
| Acetic acid | 0 | 0.16 ± 0.04 a;A | 0.16 ± 0.04 a;A | 0.16 ± 0.04 a;A | 0.16 ± 0.04 a;A |
| 12 | 0.81 ± 0.10 b;A | 1.36 ± 0.75 b;A | 1.03 ±0.09 b;A | 1.02 ± 0.19 b;A | |
| 24 | 0.82 ± 0.17 b;A | 1.65 ± 0.52 b;B | 0.92 ± 0.22 b;A | 0.96 ± 0.30 b;A | |
| 48 | 0.78 ± 0.20 b;A | 2.77 ± 1.21 c;B | 0.78 ± 0.20 b;A | 0.93 ± 0.28 b;A | |
| Propionic acid | 0 | 0.34 ± 0.09 a;A | 0.34 ± 0.09 a;A | 0.34 ± 0.09 a;A | 0.34 ± 0.09 a;A |
| 12 | 0.53 ± 0.23 a;A | 1.48 ± 0.32 b;B | 1.14 ± 0.49 b;B | 1.43 ± 0.87 b;B | |
| 24 | 0.65 ± 0.35 a;A | 1.89 ± 0.75 b;C | 1.25 ± 0.58 b;B | 0.85 ± 0.27 a,b;A,B | |
| 48 | 0.50 ± 0.24 a;A | 1.64 ± 0.60 b;B | 0.77 ± 0.20 a,b;A | 0.90 ± 0.24 a,b;A | |
| Butyric acid | 0 | 1.41 ± 0.25 a;A | 1.41 ± 0.25 a;A | 1.41 ± 0.25 a;A | 1.41 ± 0.25 a;A |
| 12 | 1.92 ± 0.69 a;A | 2.29 ± 0.99 a;A | 2.10 ± 0.79 a;A | 2.71 ± 0.94 a;A | |
| 24 | 2.24 ± 0.67 a;A | 2.23 ± 0.86 a;A | 2.54 ± 1.05 a;A | 4.12 ± 0.37 b;B | |
| 48 | 2.23 ± 1.35 a;A | 2.70 ± 1.43 a;A | 2.31 ± 0.85 a;A | 4.31 ± 0.62 b;B |
| Target Group | Maximum Growth Rate (µmax.h−1) | |||
|---|---|---|---|---|
| Primer Sequence (5′–3′) | Genomic DNA Standard | PCR Product Size (bp) | AT (°C) | |
| Universal | AAA CTC AAA GGA ATT GAC GG ACT TCA CGA GCT GAC | Bacteroides vulgatus ATCC 8482 (DSMZ 1447) | 180 | 45 |
| Firmicutes | ATG TGG TTT AAT TCG AAG CA AGC TGA CGA CAA CCA TGC AC | Lactobacillus gasseri ATCC 33323 (DSMZ 20243) | 126 | 45 |
| Enterococcus spp. | CCC TTA TTG TTA GTT GCC ATC ATT ACT CGT TGT ACT TCC CT TGT | Enterococcus gilvus ATCC BAA-350 (DSMZ 15689) | 144 | 45 |
| Lactobacillus spp. | GAG GCA GCA GTA GGG AAT CTT C GGC CAG TTA CTA CCT CTA TCC TTC TTC | Lactobacillus gasseri ATCC 33323 (DSMZ 20243) | 126 | 55 |
| Bacteroidetes | CAT GTG GTT TAA TTC GAT GAT AGC TGA CGA CAA CCA TGC AG | Bacteroides vulgatus ATCC 8482 (DSMZ 1447) | 126 | 45 |
| Bacteroides spp. | ATA GCC TTT CGA AAG RAA GAT CCA GTA TCA ACT GCA ATT TTA | Bacteroides vulgatus ATCC 8482 (DSMZ 1447) | 495 | 45 |
| Bifidobacterium spp. | CGC GTC TGG TGT GAA AG CCC CAC ATC CAG CAT CCA | Bifidobacterium longum subsp. infantis ATCC 15697 (DSMZ 20088) | 244 | 50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Catarino, M.D.; Marçal, C.; Bonifácio-Lopes, T.; Campos, D.; Mateus, N.; Silva, A.M.S.; Pintado, M.M.; Cardoso, S.M. Impact of Phlorotannin Extracts from Fucus vesiculosus on Human Gut Microbiota. Mar. Drugs 2021, 19, 375. https://doi.org/10.3390/md19070375
Catarino MD, Marçal C, Bonifácio-Lopes T, Campos D, Mateus N, Silva AMS, Pintado MM, Cardoso SM. Impact of Phlorotannin Extracts from Fucus vesiculosus on Human Gut Microbiota. Marine Drugs. 2021; 19(7):375. https://doi.org/10.3390/md19070375
Chicago/Turabian StyleCatarino, Marcelo D., Catarina Marçal, Teresa Bonifácio-Lopes, Débora Campos, Nuno Mateus, Artur M. S. Silva, Maria Manuela Pintado, and Susana M. Cardoso. 2021. "Impact of Phlorotannin Extracts from Fucus vesiculosus on Human Gut Microbiota" Marine Drugs 19, no. 7: 375. https://doi.org/10.3390/md19070375
APA StyleCatarino, M. D., Marçal, C., Bonifácio-Lopes, T., Campos, D., Mateus, N., Silva, A. M. S., Pintado, M. M., & Cardoso, S. M. (2021). Impact of Phlorotannin Extracts from Fucus vesiculosus on Human Gut Microbiota. Marine Drugs, 19(7), 375. https://doi.org/10.3390/md19070375












