Preparation and Antioxidant Activity of Chitosan Dimers with Different Sequences
Abstract
:1. Introduction
2. Results
2.1. Preparation and Characterization of the Chitin Oligomers
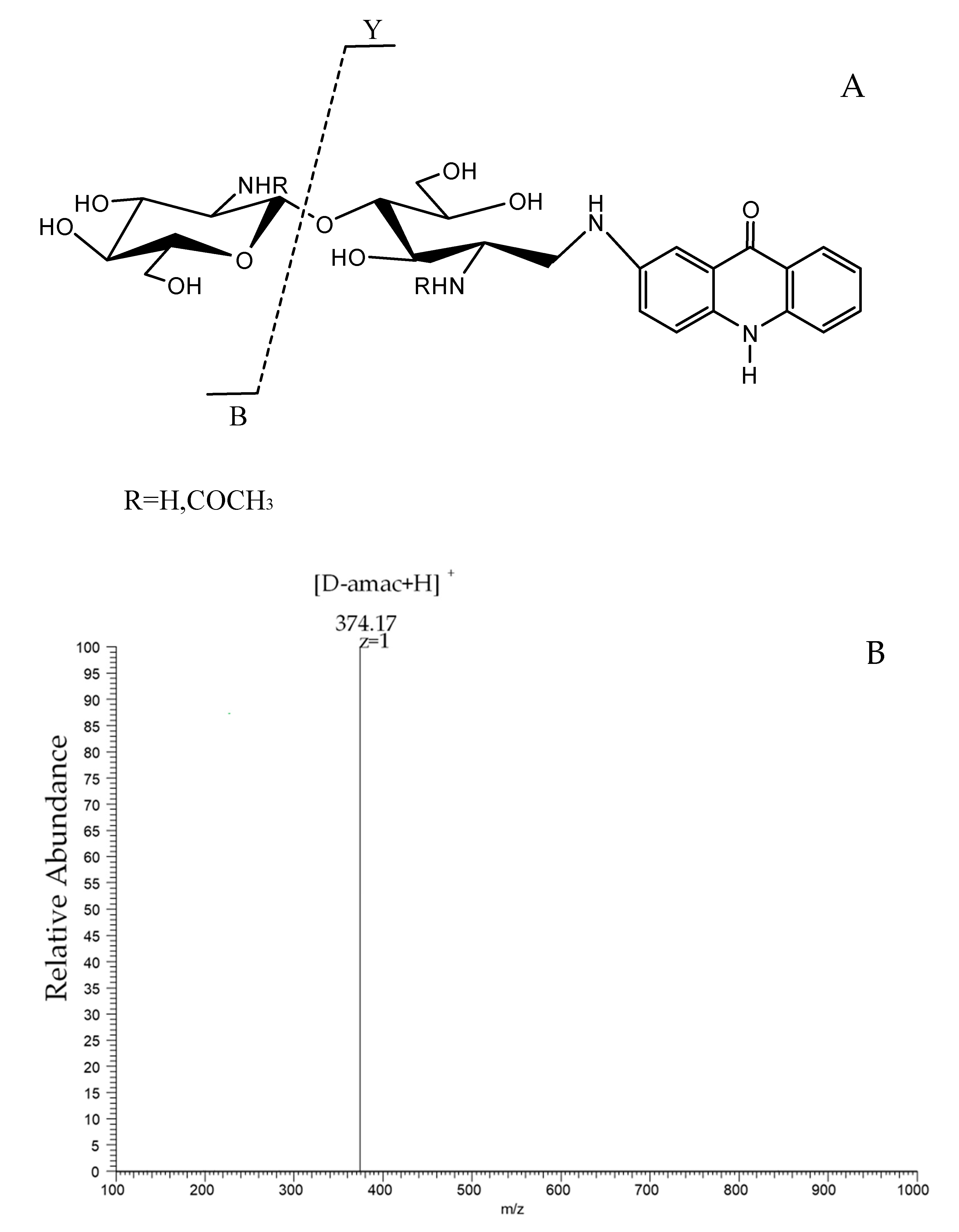
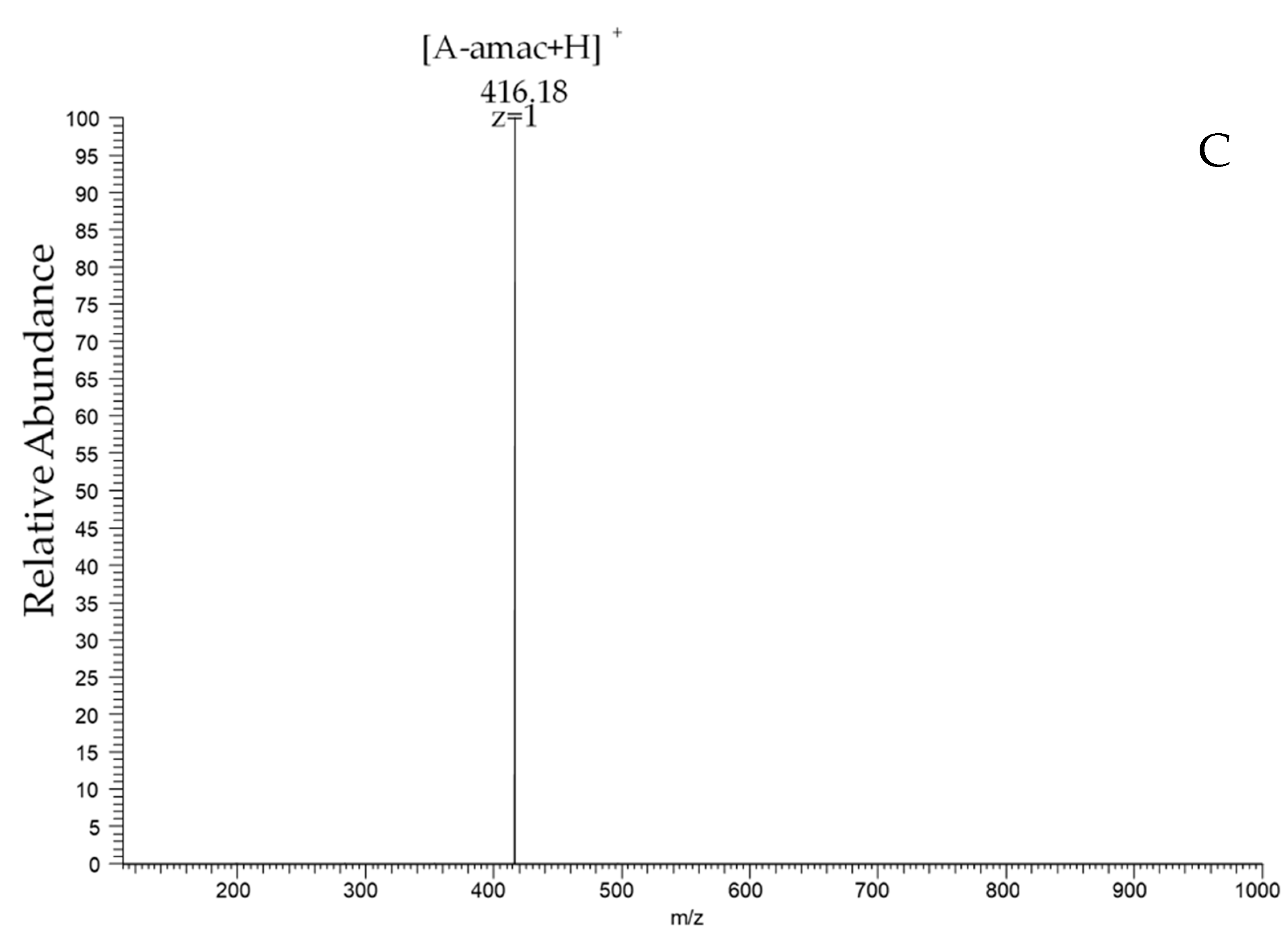
2.2. Preparation and Characterization of Two Heterogenous Sequences of the Chitosan Dimer
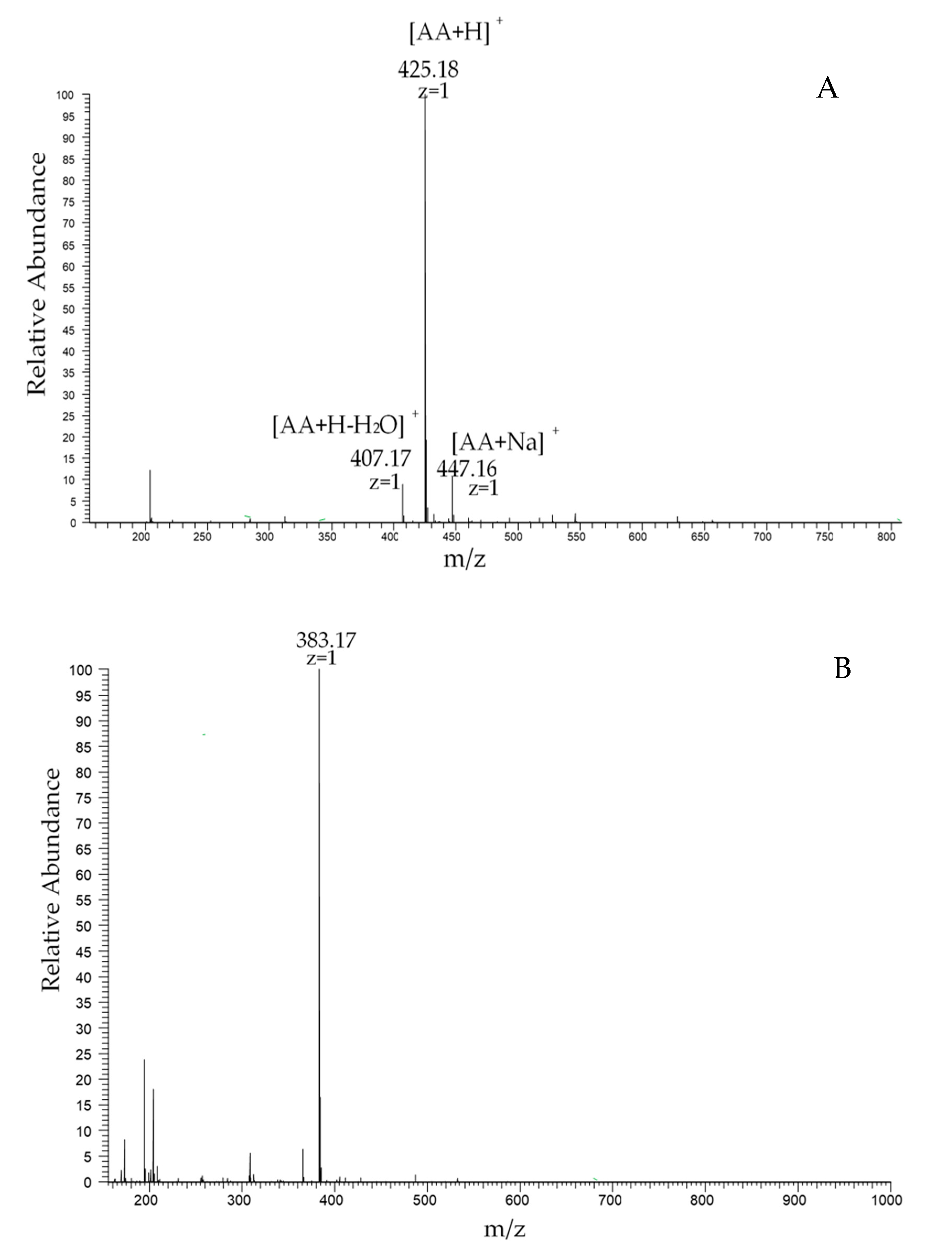
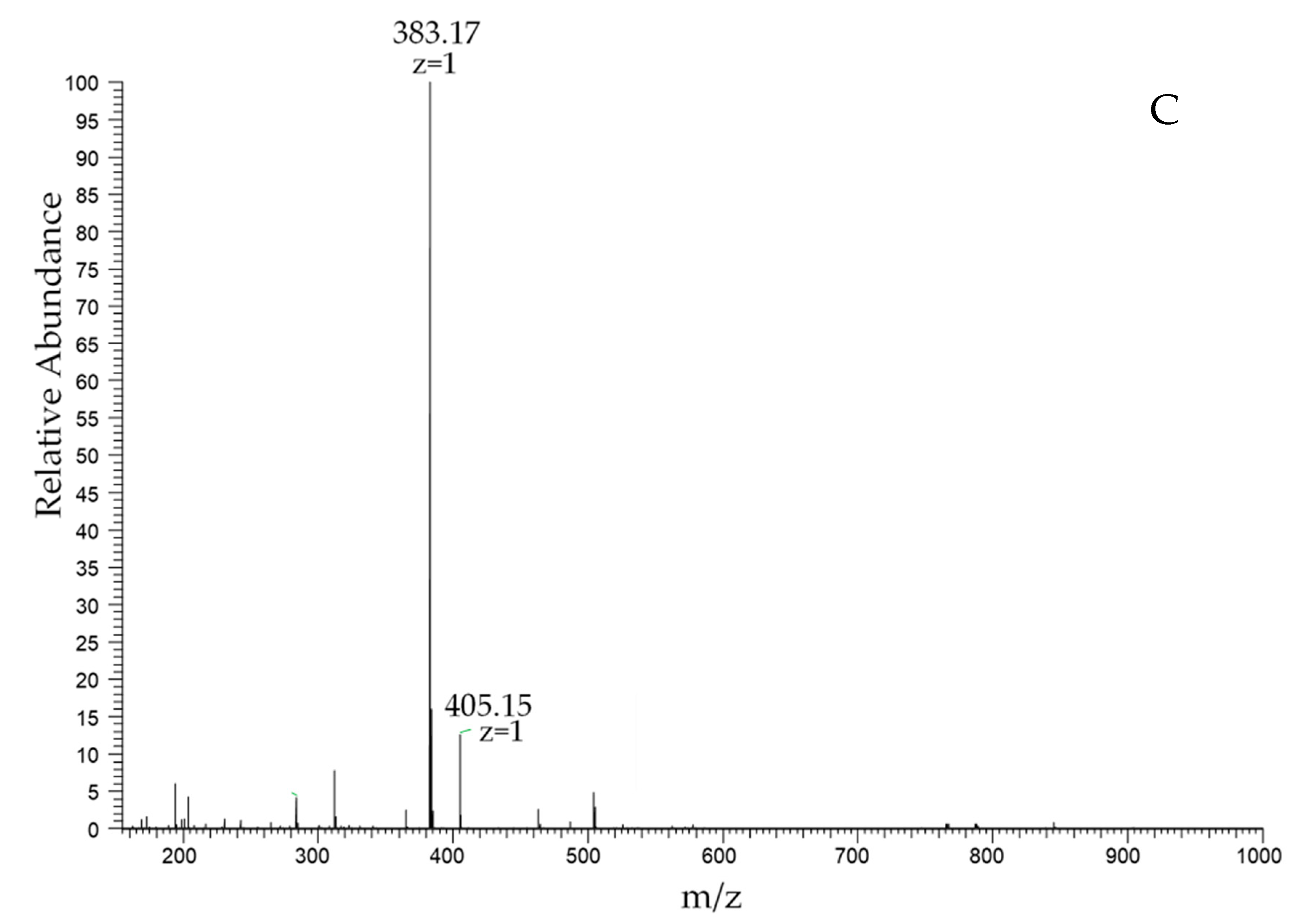
2.3. High-Performance Liquid Chromatography (HPLC) Analysis of Chitosan Dimers
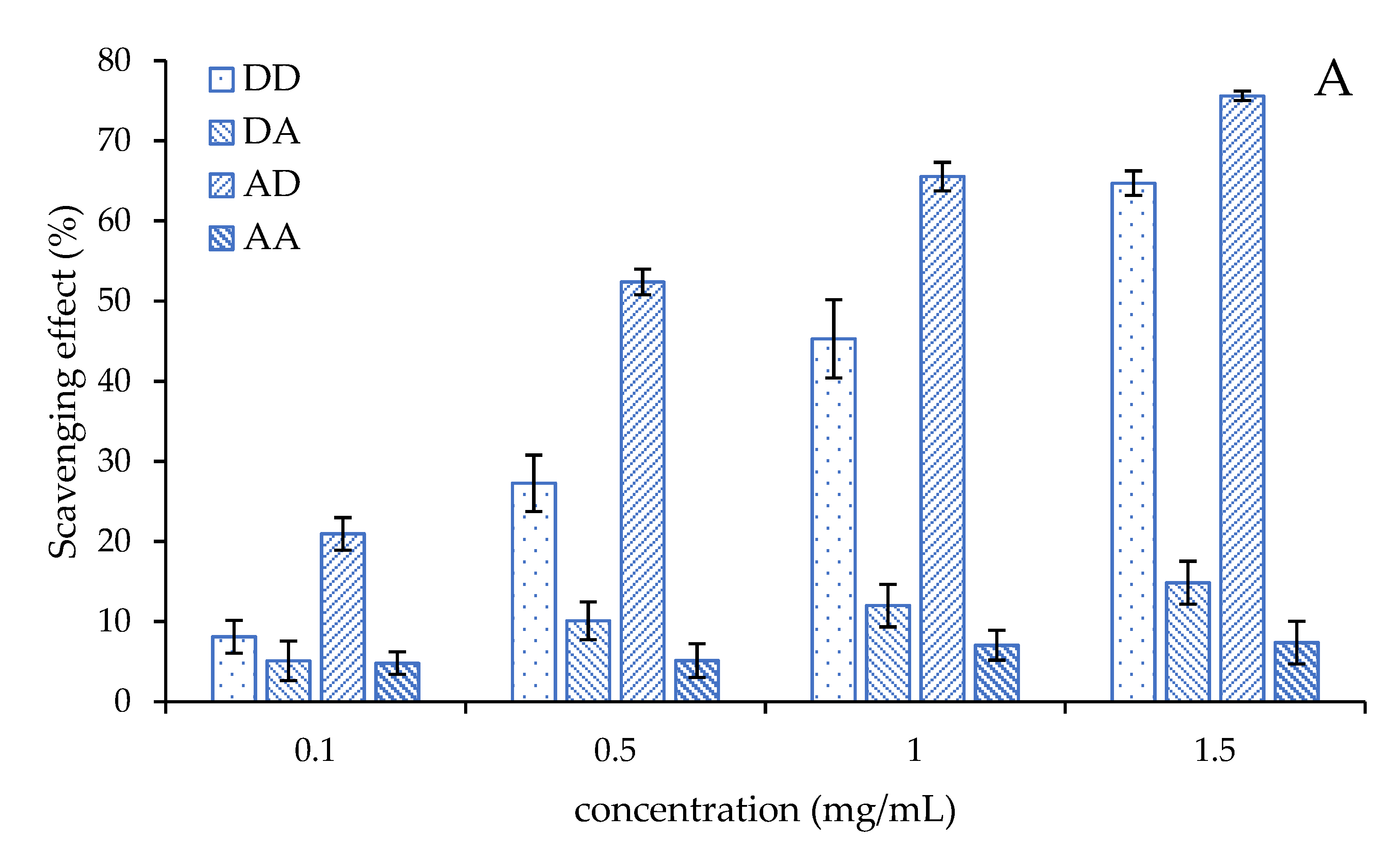
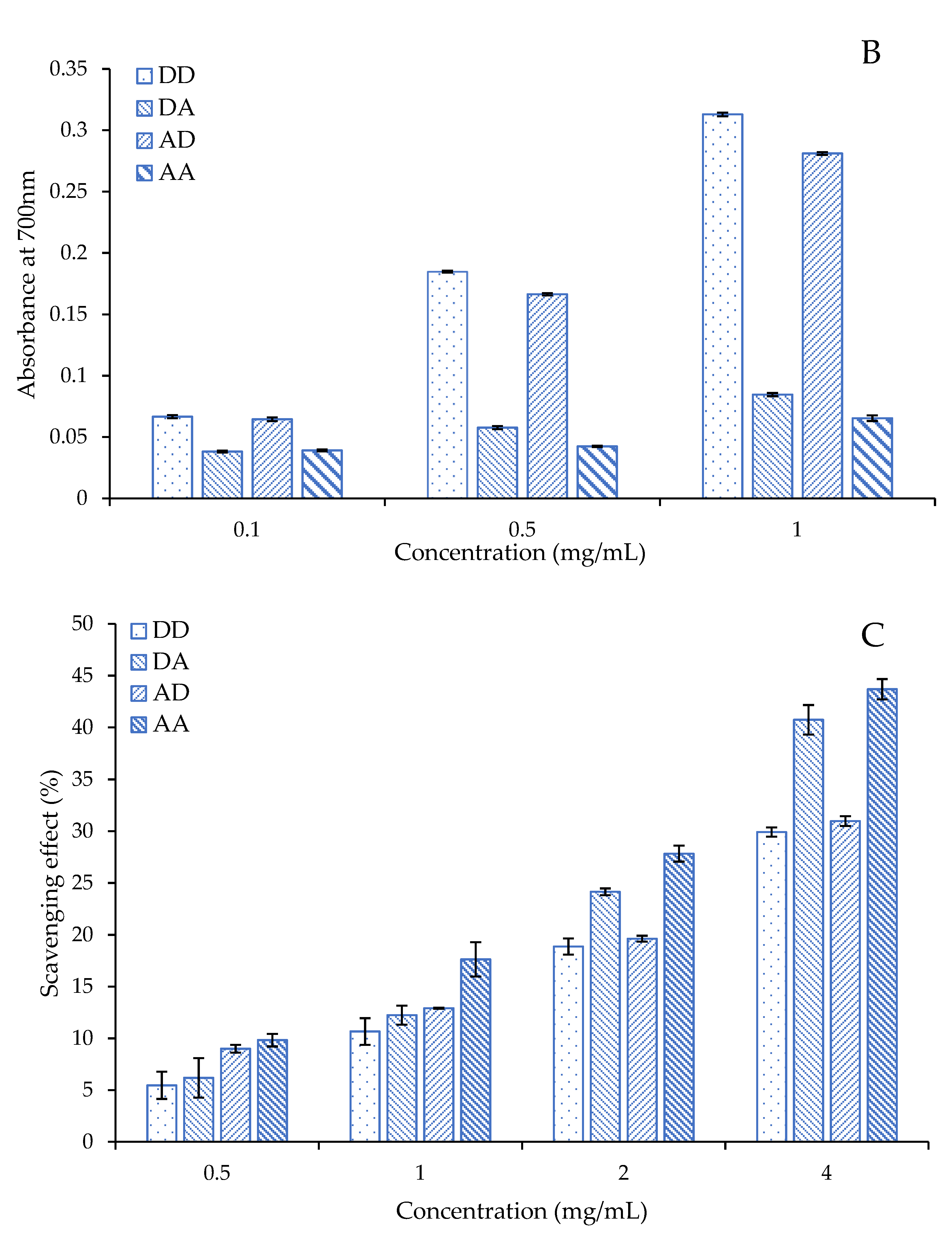
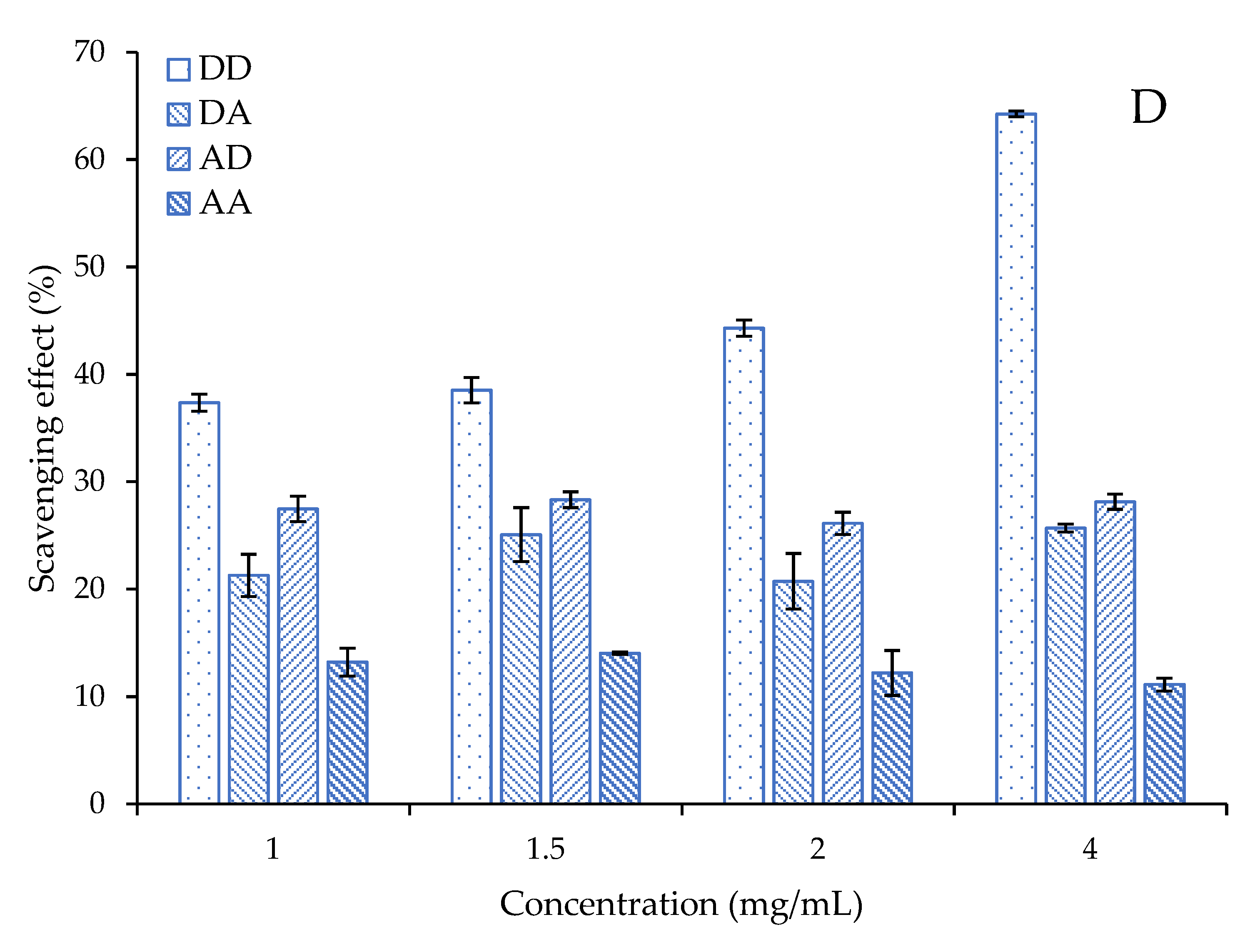
2.4. Antioxidant Activity of the Chitosan Dimers
3. Discussion
4. Materials and Methods
4.1. Material
4.2. Separation of N-Acetyl Chitooligomers by Bio Rad P10
4.3. Biological Preparation of the Four Chitosan Dimers
4.4. Reductive Amination of Chitooligosaccharides with 2-Aminoacridone (Amac)
4.5. HPLC and ESI-MS Analysis
4.6. Superoxide Radical Scavenging Assay
4.7. Measurement of Reducing Power
4.8. Hydroxyl Radical Scavenging Assay
4.9. DPPH Scavenging Assay
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Galley, J.C.; Straub, A.C. Redox Control of Vascular Function. Arterioscler. Thromb. Vasc. Biol. 2017, 37, E178–E184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Talha, M.; Mir, A.R.; Habib, S.; Abidi, M.; Warsi, M.S.; Islam, S. Moinuddin, Hydroxyl radical induced structural perturbations make insulin highly immunogenic and generate an auto-immune response in type 2 diabetes mellitus. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 255, 119640. [Google Scholar] [CrossRef]
- Dhalla, N.S.; Temsah, R.M.; Netticadan, T. Role of oxidative stress in cardiovascular diseases. J. Hypertens. 2000, 18, 655–673. [Google Scholar] [CrossRef] [PubMed]
- Jenner, P. Oxidative stress in Parkinson’s disease. Ann. Neurol. 2003, 53, S26–S36. [Google Scholar] [CrossRef] [PubMed]
- Surdu, A.M.; Pinzariu, O.; Ciobanu, D.M.; Negru, A.G.; Cainap, S.S.; Lazea, C.; Iacob, D.; Saraci, G.; Tirinescu, D.; Borda, I.M.; et al. Vitamin D and Its Role in the Lipid Metabolism and the Development of Atherosclerosis. Biomedicines 2021, 9, 172. [Google Scholar] [CrossRef] [PubMed]
- Dorszewska, J.; Kowalska, M.; Prendecki, M.; Piekut, T.; Kozlowska, J.; Kozubski, W. Oxidative stress factors in Parkinson’s disease. Neural Regen. Res. 2021, 16, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Cerda, C.; Sanchez, C.; Climent, B.; Vazquez, A.; Iradi, A.; El Amrani, F.; Bediaga, A.; Saez, G.T. Oxidative Stress and DNA Damage in Obesity-Related Tumorigenesis. In Oxidative Stress and Inflammation in Non-Communicable Diseases–Molecular Mechanisms and Perspectives in Therapeutics; Camps, J., Ed.; Springer: Cham, Switzerland, 2014; Volume 824, pp. 5–17. [Google Scholar]
- Gulcin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Liu, A.; Hu, S.; Ares, I.; Martinez-Larranaga, M.-R.; Wang, X.; Martinez, M.; Anadon, A.; Martinez, M.-A. Synthetic phenolic antioxidants: Metabolism, hazards and mechanism of action. Food Chem. 2021, 353, 129488. [Google Scholar] [CrossRef]
- Li, G.Y.; Luo, Z.C.; Yuan, F.; Yu, X.B. Combined process of high-pressure homogenization and hydrothermal extraction for the extraction of fucoidan with good antioxidant properties from Nemacystus decipients. Food Bioprocess Technol. 2017, 106, 35–42. [Google Scholar] [CrossRef]
- Chale-Dzul, J.; Freile-Pelegrin, Y.; Robledo, D.; Moo-Puc, R. Protective effect of fucoidans from tropical seaweeds against oxidative stress in HepG2 cells. J. Appl. Psychol. 2017, 29, 2229–2238. [Google Scholar] [CrossRef]
- Fernando, I.P.S.; Sanjeewa, K.K.A.; Samarakoon, K.W.; Lee, W.W.; Kim, H.S.; Kim, E.A.; Gunasekara, U.; Abeytunga, D.T.U.; Nanayakkara, C.; de Silva, E.D.; et al. FTIR characterization and antioxidant activity of water soluble crude polysaccharides of Sri Lankan marine algae. Algae 2017, 32, 75–86. [Google Scholar] [CrossRef] [Green Version]
- Mou, J.J.; Li, Q.; Qi, X.H.; Yang, J. Structural comparison, antioxidant and anti-inflammatory properties of fucosylated chondroitin sulfate of three edible sea cucumbers. Carbohydr. Polym. 2018, 185, 41–47. [Google Scholar] [CrossRef]
- Yen, M.T.; Yang, J.H.; Mau, J.L. Antioxidant properties of chitosan from crab shells. Carbohydr. Polym. 2008, 74, 840–844. [Google Scholar] [CrossRef]
- Mourya, V.K.; Inamdar, N.N.; Choudhari, Y.M. Chitooligosaccharides: Synthesis, characterization and applications. Polym. Sci. Ser. A 2011, 53, 583–612. [Google Scholar] [CrossRef]
- Hao, W.; Li, K.; Li, P. Review: Advances in preparation of chitooligosaccharides with heterogeneous sequences and their bioactivity. Carbohydr. Polym. 2021, 252, 117206. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Ma, P.; Yu, W.; Tan, C.; Liu, H.; Xiong, C.; Qiao, Y.; Du, Y. Chitooligosaccharides protect human embryonic hepatocytes against oxidative stress induced by hydrogen peroxide. Mar. Biotechnol. 2010, 12, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.C.; Hong, L.; Chang, P.; Zhang, J.; Lu, S.Y.; Zheng, B.W.; Jiang, Z.F. Chitooligosaccharides attenuate Cu2+-induced cellular oxidative damage and cell apoptosis involving Nrf2 activation. Neurotox. Res. 2015, 27, 411–420. [Google Scholar] [CrossRef]
- Yuan, W.P.; Liu, B.; Liu, C.H.; Wang, X.J.; Zhang, M.S.; Meng, X.M.; Xia, X.K. Antioxidant activity of chito-oligosaccharides on pancreatic islet cells in streptozotocin-induced diabetes in rats. World J. Gastroenterol. 2009, 15, 1339–1345. [Google Scholar] [CrossRef]
- Mengibar, M.; Mateos-Aparicio, I.; Miralles, B.; Heras, A. Influence of the physico-chemical characteristics of chito-oligosaccharides (COS) on antioxidant activity. Carbohydr. Polym. 2013, 97, 776–782. [Google Scholar] [CrossRef]
- Sun, T.; Zhou, D.; Xie, J.; Mao, F. Preparation of chitosan oligomers and their antioxidant activity. Eur. Food Res. Technol. 2006, 225, 451–456. [Google Scholar] [CrossRef]
- Li, K.; Xing, R.; Liu, S.; Li, R.; Qin, Y.; Meng, X.; Li, P. Separation of chito-oligomers with several degrees of polymerization and study of their antioxidant activity. Carbohydr. Polym. 2012, 88, 896–903. [Google Scholar] [CrossRef]
- Ju, Y.Y.; Huang, Y.Y.; Xiao, M.T.; Ye, J. Hypolipidaemic and antioxidant activities of chito-oligosaccharides in hyperlipidaemic rats induced by high-fat diet. Maejo Int. J. Sci. Technol. 2019, 13, 72–81. [Google Scholar]
- Je, J.Y.; Park, P.J.; Kim, S.-K. Free radical scavenging properties of hetero-chitooligosaccharides using an ESR spectroscopy. Food Chem. Toxicol. 2004, 42, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Park, P.-J.; Je, J.-Y.; Kim, S.-K. Free radical scavenging activities of differently deacetylated chitosans using an ESR spectrometer. Carbohydr. Polym. 2004, 55, 17–22. [Google Scholar] [CrossRef]
- Li, K.; Liu, S.; Xing, R.; Qin, Y.; Li, P. Preparation, characterization and antioxidant activity of two partially N-acetylated chitotrioses. Carbohydr. Polym. 2013, 92, 1730–1736. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, M.A.; Bezos, A.; Susanti; Marmuse, L.; Driguez, H.; Samain, E.; Vauzeilles, B.; Beau, J.M.; Kordbacheh, F.; Rolfe, B.G.; et al. Lipo-chitin oligosaccharides, plant symbiosis signalling molecules that modulate mammalian angiogenesis in vitro. PLoS ONE 2014, 9, e112635. [Google Scholar] [CrossRef] [PubMed]
- Basa, S.; Nampally, M.; Honorato, T.; Das, S.N.; Podile, A.R.; El Gueddari, N.E.; Moerschbacher, B.M. The Pattern of Acetylation Defines the Priming Activity of Chitosan Tetramers. J. Am. Chem. Soc. 2020, 142, 1975–1986. [Google Scholar] [CrossRef] [PubMed]
- John, M.; Rohrig, H.; Schmidt, J.; Wieneke, U.; Schell, J. Rhizobium NodB protein involved in nodulation signal synthesis is a chitooligosaccharide deacetylase. Proc. Natl. Acad. Sci. 1993, 90, 625–629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Wang, L.-X.; Wang, X.; Roseman, S. The chitin catabolic cascade in the marine bacterium Vibrio cholerae: Characterization of a unique chitin oligosaccharide deacetylase. Glycobiology 2007, 17, 1377–1387. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Liu, S.; Xing, R.; Yu, H.; Qin, Y.; Li, R.; Li, P. High-resolution separation of homogeneous chitooligomers series from 2-mers to 7-mers by ion-exchange chromatography. J. Sep. Sci. 2013, 36, 1275–1282. [Google Scholar] [CrossRef]
- Xie, W.M.; Xu, P.X.; Liu, Q. Antioxidant activity of water-soluble chitosan derivatives. Bioorg. Med. Chem. Lett. 2001, 11, 1699–1701. [Google Scholar] [CrossRef]
- Chambon, R.; Pradeau, S.; Fort, S.; Cottaz, S.; Armand, S. High yield production of Rhizobium NodB chitin deacetylase and its use for in vitro synthesis of lipo-chitinoligosaccharide precursors. Carbohydr. Res. 2017, 442, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Chambon, R.; Despras, G.; Brossay, A.; Vauzeilles, B.; Urban, D.; Beau, J.M.; Armand, S.; Cottaz, S.; Fort, S. Efficient chemoenzymatic synthesis of lipo-chitin oligosaccharides as plant growth promoters. Green Chem. 2015, 17, 3923–3930. [Google Scholar] [CrossRef]
- Bahrke, S.; Einarsson, J.M.; Gislason, J.; Haebel, S.; Letzel, M.C.; Peter-Katalinic, J.; Peter, M.G. Sequence analysis of chitooligosaccharides by matrix-assisted laser desorption ionization postsource decay mass spectrometry. Biomacromolecules 2002, 3, 696–704. [Google Scholar] [CrossRef]
- Nishikimi, M.; Appaji, N.; Yagi, K. Occurrence of superoxide anion in reaction of reduced phenazine methosulfate and molecular-oxygen. Biochem. Biophys. Res. Commun. 1972, 46, 849–854. [Google Scholar] [CrossRef]
- Yen, G.C.; Chen, H.Y. Antioxidant activity of various tea extracts in relation to their antimutagenicity. J. Agr. Food Chem. 1995, 43, 27–32. [Google Scholar] [CrossRef]
- Guo, Z.Y.; Xing, R.E.; Liu, S.; Yu, H.H.; Wang, P.B.; Li, C.P.; Li, P.C. The synthesis and antioxidant activity of the Schiff bases of chitosan and carboxymethyl chitosan. Bioorg. Med. Chem. Lett. 2005, 15, 4600–4603. [Google Scholar] [CrossRef] [PubMed]
- Sharma, O.P.; Bhat, T.K. DPPH antioxidant assay revisited. Food Chem. 2009, 113, 1202–1205. [Google Scholar] [CrossRef]

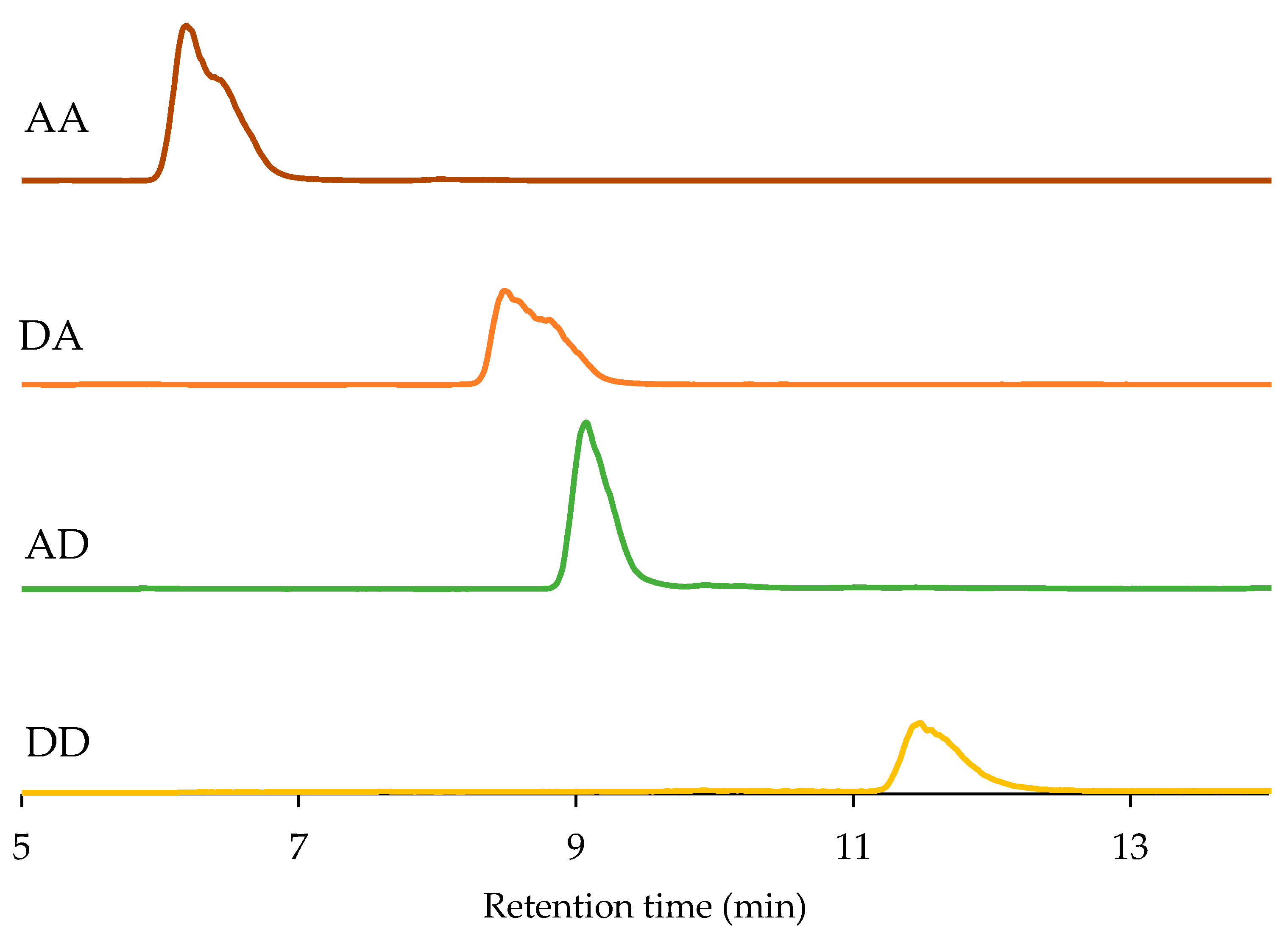
| Sequence | Retention Time (min) | Purity | The [M + H] + Ion |
|---|---|---|---|
| AA | 6.189 | 98.9% | 425.18 |
| DA | 8.489 | 98.0% | 383.17 |
| AD | 9.074 | 98.1% | 383.17 |
| DD | 11.483 | 98.4% | 341.16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hao, W.; Li, K.; Ma, Y.; Li, R.; Xing, R.; Yu, H.; Li, P. Preparation and Antioxidant Activity of Chitosan Dimers with Different Sequences. Mar. Drugs 2021, 19, 366. https://doi.org/10.3390/md19070366
Hao W, Li K, Ma Y, Li R, Xing R, Yu H, Li P. Preparation and Antioxidant Activity of Chitosan Dimers with Different Sequences. Marine Drugs. 2021; 19(7):366. https://doi.org/10.3390/md19070366
Chicago/Turabian StyleHao, Wentong, Kecheng Li, Yuzhen Ma, Rongfeng Li, Ronge Xing, Huahua Yu, and Pengcheng Li. 2021. "Preparation and Antioxidant Activity of Chitosan Dimers with Different Sequences" Marine Drugs 19, no. 7: 366. https://doi.org/10.3390/md19070366
APA StyleHao, W., Li, K., Ma, Y., Li, R., Xing, R., Yu, H., & Li, P. (2021). Preparation and Antioxidant Activity of Chitosan Dimers with Different Sequences. Marine Drugs, 19(7), 366. https://doi.org/10.3390/md19070366








