Diversity and Antimicrobial Activity of Vietnamese Sponge-Associated Bacteria
Abstract
1. Introduction
2. Results
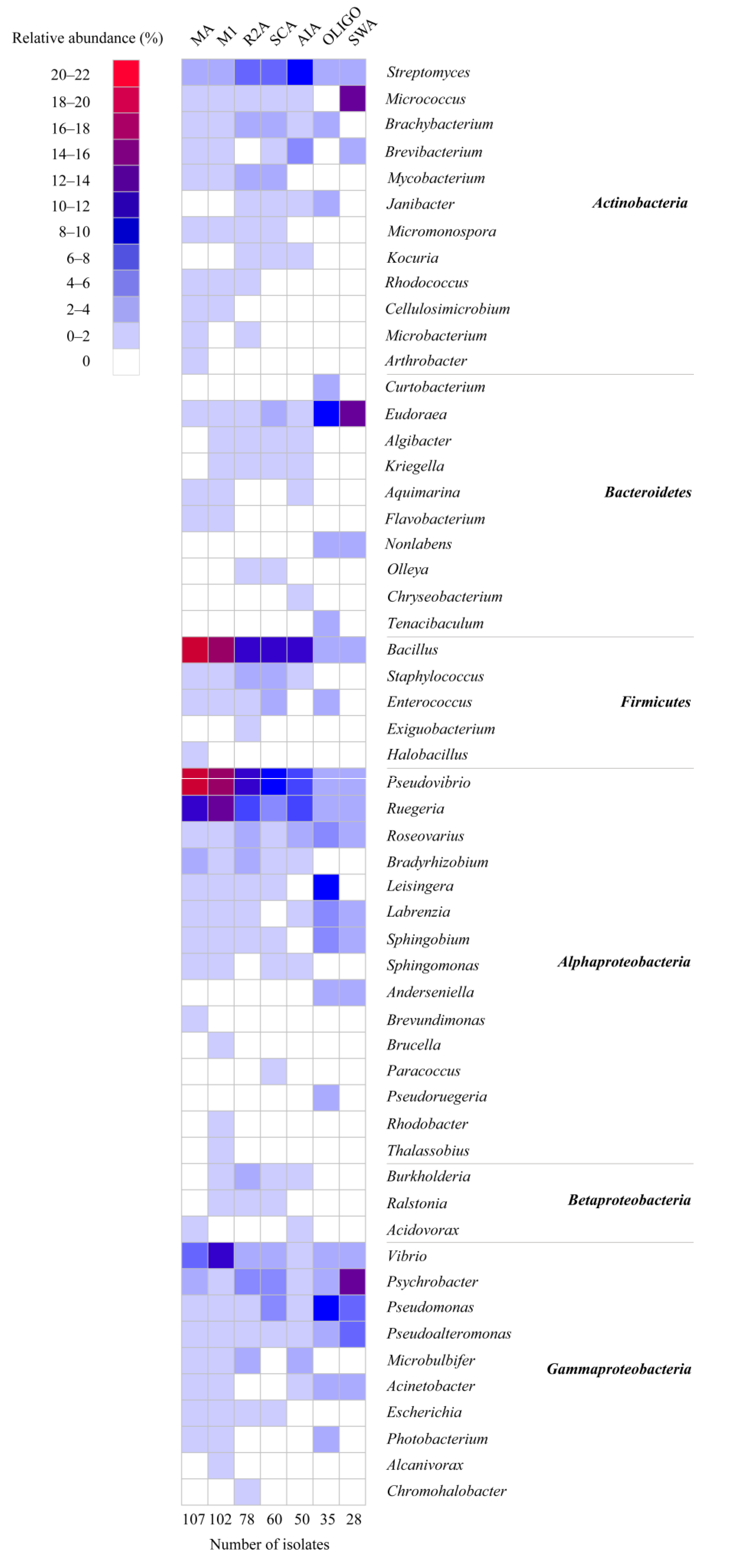
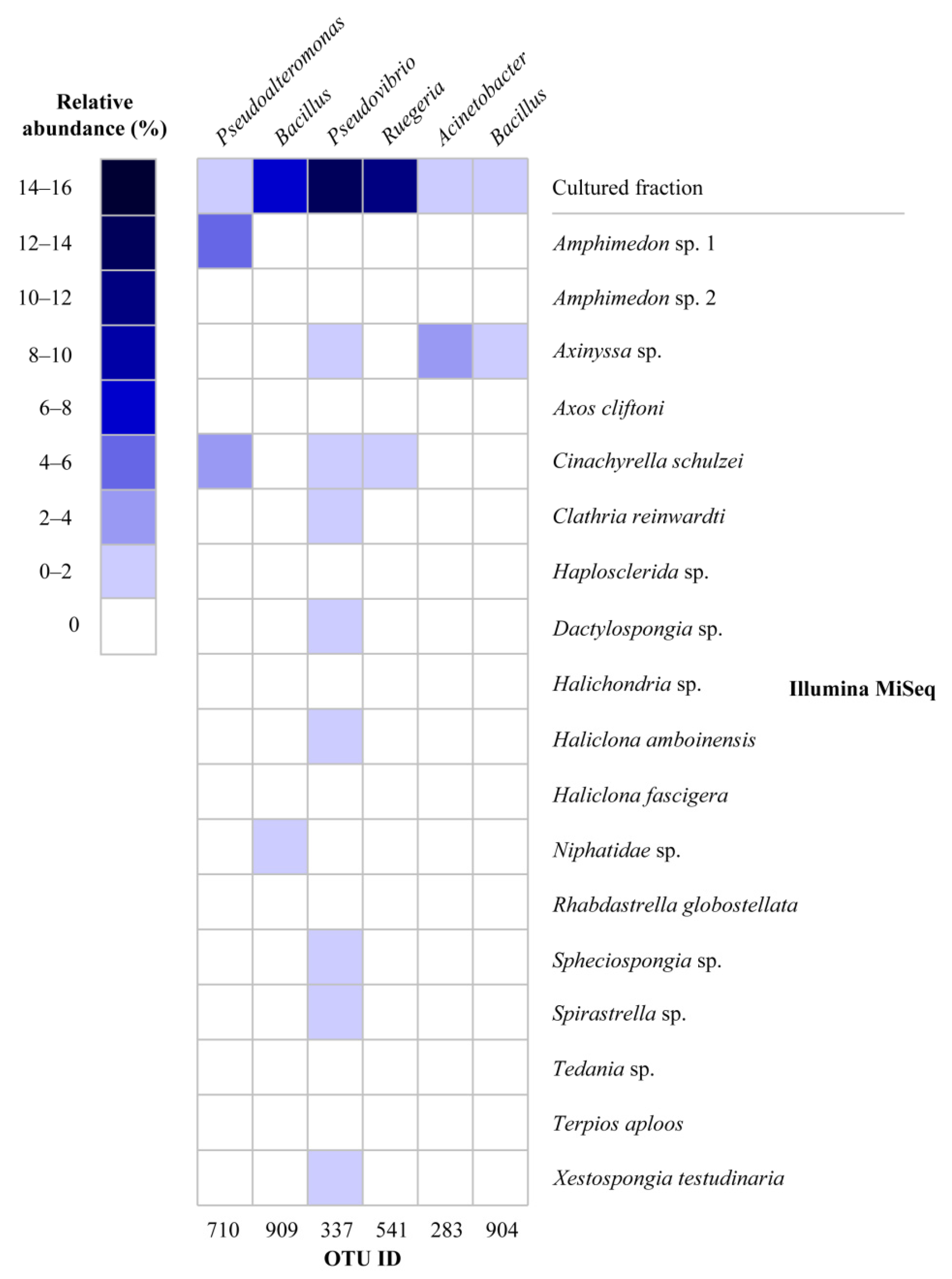
2.1. Diversity of Sponge-Associated Cultivable Bacteria
2.2. Antimicrobial Activity of Isolated Strains
2.3. Isolation and Identification of the Compounds Produced by Bacillus sp. M1_CRV_171
2.4. Antimicrobial Activity of the Isolated Secondary Metabolites
3. Discussion
3.1. Cultivable Bacteria from Vietnamese Sponges
3.2. Antimicrobial Activity of Sponge-Associated Bacteria
4. Materials and Methods
4.1. Collection and Identification of Sponges
4.2. Isolation of Bacteria from Sponges
4.3. Screening for Antimicrobial Activity of the Isolates
4.4. Identification of the Isolates by 16S rRNA Gene Analysis
4.5. Cultivation, Extraction, and Isolation of Secondary Metabolites
4.6. Antimicrobial Activity of the Secondary Metabolites
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Collignon, P.; Beggs, J.J. Socioeconomic Enablers for Contagion: Factors Impelling the Antimicrobial Resistance Epidemic. Antibiotics 2019, 8, 86. [Google Scholar] [CrossRef]
- Malik, B.; Bhattacharyya, S. Antibiotic drug-resistance as a complex system driven by socio-economic growth and antibiotic misuse. Sci. Rep. 2019, 9, 9788. [Google Scholar] [CrossRef] [PubMed]
- Saleem, Z.; Saeed, H.; Hassali, M.A.; Godman, B.; Asif, U.; Yousaf, M.; Ahmed, Z.; Riaz, H.; Raza, S.A. Pattern of inappropriate antibiotic use among hospitalized patients in Pakistan: A longitudinal surveillance and implications. Antimicrob. Resist. Infect. Control. 2019, 8, 188. [Google Scholar] [CrossRef] [PubMed]
- Manyi-Loh, C.; Mamphweli, S.; Meyer, E.; Okoh, A. Antibiotic Use in Agriculture and Its Consequential Resistance in Environmental Sources: Potential Public Health Implications. Molecules 2018, 23, 795. [Google Scholar] [CrossRef] [PubMed]
- Cantón, R.; Horcajada, J.P.; Oliver, A.; Garbajosa, P.R.; Vila, J. Inappropriate use of antibiotics in hospitals: The complex relationship between antibiotic use and antimicrobial resistance. Enferm. Infecc. Microbiol. Clínica 2013, 31, 3–11. [Google Scholar] [CrossRef]
- O’Neill, J. Antimicrobial Resistance: Tackling a crisis for the health and wealth of nations. In Review on Antimicrobial Resistance: Chaired by Jim O’Neill, 2014; Review on AMR: London, UK, 2014. [Google Scholar]
- Taylor, P.L.; Wright, G.D. Novel approaches to discovery of antibacterial agents. Anim. Health Res. Rev. 2008, 9, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Projan, S.J. Why is big Pharma getting out of antibacterial drug discovery? Curr. Opin. Microbiol. 2003, 6, 427–430. [Google Scholar] [CrossRef]
- Kunakom, S.; Eustáquio, A.S. Natural Products and Synthetic Biology: Where We Are and Where We Need to Go. mSystems 2019, 4, e00113-19. [Google Scholar] [CrossRef]
- Breitling, R.; Takano, E. Synthetic Biology of Natural Products. Cold Spring Harb. Perspect Biol. 2016, 8, a023994. [Google Scholar] [CrossRef]
- Hughes, C.C.; Fenical, W. Antibacterials from the sea. Chemistry 2010, 16, 12512–12525. [Google Scholar] [CrossRef]
- Montaser, R.; Luesch, H. Marine natural products: A new wave of drugs? Future Med. Chem. 2011, 3, 1475–1489. [Google Scholar] [CrossRef]
- Choudhary, A.; Naughton, L.M.; Montánchez, I.; Dobson, A.D.W.; Rai, D.K. Current Status and Future Prospects of Marine Natural Products (MNPs) as Antimicrobials. Mar. Drugs 2017, 15, 272. [Google Scholar] [CrossRef]
- Mehbub, F.M.; Lei, J.; Franco, C.; Zhang, W. Marine Sponge Derived Natural Products between 2001 and 2010: Trends and Opportunities for Discovery of Bioactives. Mar. Drugs 2014, 12, 4539–4577. [Google Scholar] [CrossRef]
- Carroll, A.R.; Copp, B.R.; Davis, R.A.; Keyzers, R.A.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2019, 36, 122–173. [Google Scholar] [CrossRef]
- Jaspars, M.; De Pascale, D.; Andersen, J.H.; Reyes, F.; Crawford, A.D.; Ianora, A. The marine biodiscovery pipeline and ocean medicines of tomorrow. J. Mar. Biol. Assoc. UK 2016, 96, 151–158. [Google Scholar] [CrossRef]
- Sipkema, D.; Franssen, M.C.R.; Osinga, R.; Tramper, J.; Wijffels, R.H. Marine Sponges as Pharmacy. Mar. Biotechnol. 2005, 7, 142–162. [Google Scholar] [CrossRef]
- Proksch, P.; Edrada-Ebel, R.; Ebel, R. Drugs from the Sea—Opportunities and Obstacles. Mar. Drugs 2003, 1, 5–17. [Google Scholar] [CrossRef]
- Wagner-Döbler, I.; Beil, W.; Lang, S.; Meiners, M.; Laatsch, H. Integrated Approach to Explore the Potential of Marine Microorganisms for the Production of Bioactive Metabolites. In Tools and Applications of Biochemical Engineering Science; Schügerl, K., Zeng, A.P., Aunins, J.G., Bader, A., Bell, W., Biebl, H., Biselli, M., Carrondo, M.J.T., Castilho, L.R., Chang, H.N., et al., Eds.; Springer: Berlin/Heidelberg, Germany, 2002; pp. 207–238. [Google Scholar]
- Thoms, C.; Schupp, P. Biotechnological Potential of Marine Sponges and their Associated Bacteria as Producers of New Pharmaceuticals (Part II). J. Int. Biotechnol. Law 2005, 2, 257. [Google Scholar] [CrossRef]
- Taylor, M.W.; Radax, R.; Steger, D.; Wagner, M. Sponge-Associated Microorganisms: Evolution, Ecology, and Biotechnological Potential. Microbiol. Mol. Biol. Rev. 2007, 71, 295–347. [Google Scholar] [CrossRef]
- Fuerst, J.A. Diversity and biotechnological potential of microorganisms associated with marine sponges. Appl. Microbiol. Biotechnol. 2014, 98, 7331–7347. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Hill, R.T. New drugs from marine microbes: The tide is turning. J. Ind. Microbiol. Biotechnol. 2006, 33, 539–544. [Google Scholar] [CrossRef]
- Schmidt, E.W.; Bewley, C.A.; Faulkner, D.J. Theopalauamide, a Bicyclic Glycopeptide from Filamentous Bacterial Symbionts of the Lithistid Sponge Theonella swinhoei from Palau and Mozambique. J. Org. Chem. 1998, 63, 1254–1258. [Google Scholar] [CrossRef]
- Santos-Gandelman, J.; Giambiagi-deMarval, M.; Oelemann, W.; Laport, M. Biotechnological Potential of Sponge-Associated Bacteria. Curr. Pharm. Biotechnol. 2014, 15, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Laport, M.S.; Santos, O.C.S.; Muricy, G. Marine Sponges: Potential Sources of New Antimicrobial Drugs. Curr. Pharm. Biotechnol. 2009, 10, 86–105. [Google Scholar] [CrossRef]
- Indraningrat, A.A.; Smidt, H.; Sipkema, D. Bioprospecting Sponge-Associated Microbes for Antimicrobial Compounds. Mar. Drugs 2016, 14, 87. [Google Scholar] [CrossRef]
- Dat, T.T.H.; Steinert, G.; Thi Kim Cuc, N.; Smidt, H.; Sipkema, D. Archaeal and bacterial diversity and community composition from 18 phylogenetically divergent sponge species in Vietnam. PeerJ 2018, 6, e4970. [Google Scholar] [CrossRef] [PubMed]
- Wattana-Amorn, P.; Charoenwongsa, W.; Williams, C.; Crump, M.P.; Apichaisataienchote, B. Antibacterial activity of cyclo(L-Pro-L-Tyr) and cyclo(D-Pro-L-Tyr) from Streptomyces sp. strain 22-4 against phytopathogenic bacteria. Nat. Prod. Res. 2016, 30, 1980–1983. [Google Scholar] [CrossRef]
- Lee, S.-J.; Cho, J.-Y.; Cho, J.-I.; Moon, J.-H.; Park, K.D.; Lee, Y.J.; Park, K.-H. Isolation and Characterization of Antimicrobial Substance Macrolactin A Produced from Bacillus amyloliquefaciens CHO104 Isolated from Soil. J. Microbiol. Biotechnol. 2004, 14, 525–531. [Google Scholar]
- Romero-Tabarez, M.; Jansen, R.; Sylla, M.; Lünsdorf, H.; Häussler, S.; Santosa, D.A.; Timmis, K.N.; Molinari, G. 7-O-malonyl macrolactin A, a new macrolactin antibiotic from Bacillus subtilis active against methicillin-resistant Staphylococcus aureus, vancomycin-resistant enterococci, and a small-colony variant of Burkholderia cepacia. Antimicrob. Agents Chemother. 2006, 50, 1701–1709. [Google Scholar] [CrossRef]
- Nagao, T.; Adachi, K.; Sakai, M.; Nishijima, M.; Sano, H. Novel Macrolactins as antibiotic lactones from a marine bacterium. J. Antibiot. 2001, 54, 333–339. [Google Scholar] [CrossRef]
- Mondol, M.A.M.; Tareq, F.S.; Kim, J.H.; Lee, M.A.; Lee, H.-S.; Lee, Y.-J.; Lee, J.S.; Shin, H.J. Cyclic Ether-Containing Macrolactins, Antimicrobial 24-Membered Isomeric Macrolactones from a Marine Bacillus sp. J. Nat. Prod. 2011, 74, 2582–2587. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Q.; Liu, W.C.; Zhu, P.; Yang, J.L.; Cheng, K.D. Phylogenetic diversity of bacteria associated with the marine sponge Gelliodes carnosa collected from the Hainan Island coastal waters of the South China Sea. Microb. Ecol. 2011, 62, 800–812. [Google Scholar] [CrossRef] [PubMed]
- Sipkema, D.; Schippers, K.; Maalcke, W.J.; Yang, Y.; Salim, S.; Blanch, H.W. Multiple Approaches to Enhance the Cultivability of Bacteria Associated with the Marine Sponge Haliclona (gellius) sp. Appl. Environ. Microbiol. 2011, 77, 2130–2140. [Google Scholar] [CrossRef]
- Montalvo, N.F.; Davis, J.; Vicente, J.; Pittiglio, R.; Ravel, J.; Hill, R.T. Integration of culture-based and molecular analysis of a complex sponge-associated bacterial community. PLoS ONE 2014, 9, e90517. [Google Scholar] [CrossRef]
- Hardoim, C.C.P.; Costa, R. Microbial Communities and Bioactive Compounds in Marine Sponges of the Family Irciniidae—A Review. Mar. Drugs 2014, 12, 5089–5122. [Google Scholar] [CrossRef] [PubMed]
- Versluis, D.; McPherson, K.; van Passel, M.W.J.; Smidt, H.; Sipkema, D. Recovery of Previously Uncultured Bacterial Genera from Three Mediterranean Sponges. Mar. Biotechnol. 2017, 19, 454–468. [Google Scholar] [CrossRef]
- Slaby, B.M.; Hackl, T.; Horn, H.; Bayer, K.; Hentschel, U. Metagenomic binning of a marine sponge microbiome reveals unity in defense but metabolic specialization. ISME J. 2017, 11, 2465–2478. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.A.; Palladino, G.; Wemheuer, B.; Steinert, G.; Sipkema, D.; Williams, T.J.; Thomas, T. Phylogeny resolved, metabolism revealed: Functional radiation within a widespread and divergent clade of sponge symbionts. ISME J. 2021, 15, 503–519. [Google Scholar] [CrossRef]
- Esteves, A.I.; Amer, N.; Nguyen, M.; Thomas, T. Sample Processing Impacts the Viability and Cultivability of the Sponge Microbiome. Front. Microbiol. 2016, 7, 499. [Google Scholar] [CrossRef]
- Steinert, G.; Whitfield, S.; Taylor, M.W.; Thoms, C.; Schupp, P.J. Application of Diffusion Growth Chambers for the Cultivation of Marine Sponge-Associated Bacteria. Mar. Biotechnol. 2014, 16, 594–603. [Google Scholar] [CrossRef]
- Webster, N.S.; Hill, R.T. The culturable microbial community of the Great Barrier Reef sponge Rhopaloeides odorabile is dominated by an alpha-protebacterium. Mar. Biol. 2001, 138, 843–851. [Google Scholar] [CrossRef]
- O’Halloran, J.A.; Barbosa, T.M.; Morrissey, J.P.; Kennedy, J.; O’Gara, F.; Dobson, A.D.W. Diversity and antimicrobial activity of Pseudovibrio spp. from Irish marine sponges. J. Appl. Microbiol. 2011, 110, 1495–1508. [Google Scholar] [CrossRef]
- Enticknap, J.J.; Kelly, M.; Peraud, O.; Hill, R.T. Characterization of a culturable alphaproteobacterial symbiont common to many marine sponges and evidence for vertical transmission via sponge larvae. Appl. Environ. Microbiol. 2006, 72, 3724–3732. [Google Scholar] [CrossRef] [PubMed]
- Hentschel, U.; Schmid, M.; Wagner, M.; Fieseler, L.; Gernert, C.; Hacker, J. Isolation and phylogenetic analysis of bacteria with antimicrobial activities from the Mediterranean sponges Aplysina aerophoba and Aplysina cavernicola. FEMS Microbiol. Ecol. 2001, 35, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Muscholl-Silberhorn, A.; Thiel, V.; Imhoff, J.F. Abundance and bioactivity of cultured sponge-associated bacteria from the Mediterranean sea. Microb. Ecol. 2008, 55, 94–106. [Google Scholar] [CrossRef]
- Garland, J.L.; Cook, K.L.; Adams, J.L.; Kerkhof, L. Culturability as an Indicator of Succession in Microbial Communities. Microb. Ecol. 2001, 42, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Hernández-González, I.L.; Moreno-Hagelsieb, G.; Olmedo-Álvarez, G. Environmentally-driven gene content convergence and the Bacillus phylogeny. BMC Evol. Biol. 2018, 18, 148. [Google Scholar] [CrossRef]
- Ian, E.; Malko, D.B.; Sekurova, O.N.; Bredholt, H.; Rückert, C.; Borisova, M.E.; Albersmeier, A.; Kalinowski, J.; Gelfand, M.S.; Zotchev, S.B. Genomics of Sponge-Associated Streptomyces spp. Closely Related to Streptomyces albus J1074: Insights into Marine Adaptation and Secondary Metabolite Biosynthesis Potential. PLoS ONE 2014, 9, e96719. [Google Scholar] [CrossRef]
- Alex, A.; Antunes, A. Whole Genome Sequencing of the Symbiont Pseudovibrio sp. from the Intertidal Marine Sponge Polymastia penicillus Revealed a Gene Repertoire for Host-Switching Permissive Lifestyle. Genome Biol. Evol. 2015, 7, 3022–3032. [Google Scholar] [CrossRef]
- Alex, A.; Antunes, A. Genus-wide comparison of Pseudovibrio bacterial genomes reveal diverse adaptations to different marine invertebrate hosts. PLoS ONE 2018, 13, e0194368. [Google Scholar] [CrossRef]
- Belbahri, L.; Bouket, A.C.; Rekik, I.; Alenezi, F.N.; Vallat, A.; Luptakova, L.; Petrovova, E.; Oszako, T.; Cherrad, S.; Vacher, S.; et al. Comparative Genomics of Bacillus amyloliquefaciens Strains Reveals a Core Genome with Traits for Habitat Adaptation and a Secondary Metabolites Rich Accessory Genome. Front. Microbiol. 2017, 8, 1438. [Google Scholar] [CrossRef]
- Versluis, D.; Nijsse, B.; Naim, M.A.; Koehorst, J.J.; Wiese, J.; Imhoff, J.F.; Schaap, P.J.; van Passel, M.W.J.; Smidt, H.; Sipkema, D. Comparative Genomics Highlights Symbiotic Capacities and High Metabolic Flexibility of the Marine Genus Pseudovibrio. Genome Biol. Evol. 2018, 10, 125–142. [Google Scholar] [CrossRef] [PubMed]
- Romano, S. Ecology and Biotechnological Potential of Bacteria Belonging to the Genus Pseudovibrio. Appl. Environ. Microbiol. 2018, 84, e02516-17. [Google Scholar] [CrossRef] [PubMed]
- Steil, L.; Hoffmann, T.; Budde, I.; Völker, U.; Bremer, E. Genome-Wide Transcriptional Profiling Analysis of Adaptation of Bacillus subtilis to High Salinity. J. Bacteriol. 2003, 185, 6358. [Google Scholar] [CrossRef]
- Tian, X.; Zhang, Z.; Yang, T.; Chen, M.; Li, J.; Chen, F.; Yang, J.; Li, W.; Zhang, B.; Zhang, Z.; et al. Comparative Genomics Analysis of Streptomyces Species Reveals Their Adaptation to the Marine Environment and Their Diversity at the Genomic Level. Front. Microbiol. 2016, 7, 998. [Google Scholar] [CrossRef]
- Brito, P.H.; Chevreux, B.; Serra, C.R.; Schyns, G.; Henriques, A.O.; Pereira-Leal, J.B. Genetic Competence Drives Genome Diversity in Bacillus subtilis. Genome Biol. Evol. 2018, 10, 108–124. [Google Scholar] [CrossRef]
- Lin, H.; Yu, M.; Wang, X.; Zhang, X.-H. Comparative genomic analysis reveals the evolution and environmental adaptation strategies of vibrios. BMC Genom. 2018, 19, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bondarev, V.; Richter, M.; Romano, S.; Piel, J.; Schwedt, A.; Schulz-Vogt, H.N. The genus Pseudovibrio contains metabolically versatile bacteria adapted for symbiosis. Environ. Microbiol. 2013, 15, 2095–2113. [Google Scholar] [CrossRef] [PubMed]
- Díez-Vives, C.; Esteves, A.I.S.; Costa, R.; Nielsen, S.; Thomas, T. Detecting signatures of a sponge-associated lifestyle in bacterial genomes. Environ. Microbiol. Rep. 2018, 10, 433–443. [Google Scholar] [CrossRef]
- Helber, S.B.; Hoeijmakers, D.J.J.; Muhando, C.A.; Rohde, S.; Schupp, P.J. Sponge chemical defenses are a possible mechanism for increasing sponge abundance on reefs in Zanzibar. PLoS ONE 2018, 13, e0197617. [Google Scholar] [CrossRef]
- Pawlik, J.R. The Chemical Ecology of Sponges on Caribbean Reefs: Natural Products Shape Natural Systems. BioScience 2011, 61, 888–898. [Google Scholar] [CrossRef]
- Rohde, S.; Nietzer, S.; Schupp, P.J. Prevalence and Mechanisms of Dynamic Chemical Defenses in Tropical Sponges. PLoS ONE 2015, 10, e0132236. [Google Scholar] [CrossRef]
- Flowers, A.E.; Garson, M.J.; Webb, R.I.; Dumdei, E.J.; Charan, R.D. Cellular origin of chlorinated diketopiperazines in the dictyoceratid sponge Dysidea herbacea (Keller). Cell Tissue Res. 1998, 292, 597–607. [Google Scholar] [CrossRef]
- Piel, J.; Hui, D.; Wen, G.; Butzke, D.; Platzer, M.; Fusetani, N.; Matsunaga, S. Antitumor polyketide biosynthesis by an uncultivated bacterial symbiont of the marine sponge Theonella swinhoei. Proc. Natl. Acad. Sci. USA 2004, 101, 16222–16227. [Google Scholar] [CrossRef]
- Faulkner, D.J.; Harper, M.K.; Haygood, M.G.; Salomon, C.E.; Schmidt, E.W. Symbiotic bacteria in sponges: Sources of bioactive substances. In Drugs from the Sea; Karger Publishers: Basel, Switzerland, 2000; pp. 107–119. [Google Scholar]
- Haygood, M.G.; Schmidt, E.W.; Davidson, S.K.; Faulkner, D.J. Microbial symbionts of marine invertebrates: Opportunities for microbial biotechnology. J. Mol. Microbiol. Biotechnol. 1999, 1, 33–43. [Google Scholar] [PubMed]
- Hildebrand, M.; Waggoner, L.E.; Lim, G.E.; Sharp, K.H.; Ridley, C.P.; Haygood, M.G. Approaches to identify, clone, and express symbiont bioactive metabolite genes. Nat. Prod. Rep. 2004, 21, 122–142. [Google Scholar] [CrossRef]
- Page, M.G.P. The role of the outer membrane of gram-negative bacteria in antibiotic resistance: Ajax’ shield or Achilles’ heel? In Antibiotic Resistance. Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2012; Volume 211, pp. 67–86. [Google Scholar]
- Naughton, L.M.; Romano, S.; O’Gara, F.; Dobson, A.D.W. Identification of Secondary Metabolite Gene Clusters in the Pseudovibrio Genus Reveals Encouraging Biosynthetic Potential toward the Production of Novel Bioactive Compounds. Front. Microbiol. 2017, 8, 1494. [Google Scholar] [CrossRef]
- Xu, L.; Ye, K.-X.; Dai, W.-H.; Sun, C.; Xu, L.-H.; Han, B.-N. Comparative Genomic Insights into Secondary Metabolism Biosynthetic Gene Cluster Distributions of Marine Streptomyces. Mar. Drugs 2019, 17, 498. [Google Scholar] [CrossRef] [PubMed]
- Harwood, C.R.; Mouillon, J.-M.; Pohl, S.; Arnau, J. Secondary metabolite production and the safety of industrially important members of the Bacillus subtilis group. FEMS Microbiol. Rev. 2018, 42, 721–738. [Google Scholar] [CrossRef]
- Mondol, M.A.M.; Shin, H.J.; Islam, M.T. Diversity of secondary metabolites from marine Bacillus species: Chemistry and biological activity. Mar. Drugs 2013, 11, 2846–2872. [Google Scholar] [CrossRef] [PubMed]
- Caulier, S.; Nannan, C.; Gillis, A.; Licciardi, F.; Bragard, C.; Mahillon, J. Overview of the Antimicrobial Compounds Produced by Members of the Bacillus subtilis Group. Front. Microbiol. 2019, 10, 302. [Google Scholar] [CrossRef] [PubMed]
- Sumi, C.D.; Yang, B.W.; Yeo, I.-C.; Hahm, Y.T. Antimicrobial peptides of the genus Bacillus: A new era for antibiotics. Can. J. Microbiol. 2014, 61, 93–103. [Google Scholar] [CrossRef]
- Grubbs, K.J.; Bleich, R.M.; Santa Maria, K.C.; Allen, S.E.; Farag, S.; AgBiome, T.; Shank, E.A.; Bowers, A.A. Large-Scale Bioinformatics Analysis of Bacillus Genomes Uncovers Conserved Roles of Natural Products in Bacterial Physiology. mSystems 2017, 2, e00040-17. [Google Scholar] [CrossRef]
- Stein, T. Bacillus subtilis antibiotics: Structures, syntheses and specific functions. Mol. Microbiol. 2005, 56, 845–857. [Google Scholar] [CrossRef] [PubMed]
- Kunst, F.; Ogasawara, N.; Moszer, I.; Albertini, A.M.; Alloni, G.; Azevedo, V.; Bertero, M.G.; Bessières, P.; Bolotin, A.; Borchert, S.; et al. The complete genome sequence of the Gram-positive bacterium Bacillus subtilis. Nature 1997, 390, 249–256. [Google Scholar] [CrossRef]
- Chen, X.H.; Koumoutsi, A.; Scholz, R.; Eisenreich, A.; Schneider, K.; Heinemeyer, I.; Morgenstern, B.; Voss, B.; Hess, W.R.; Reva, O.; et al. Comparative analysis of the complete genome sequence of the plant growth–promoting bacterium Bacillus amyloliquefaciens FZB42. Nat. Biotechnol. 2007, 25, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, S.; Mitova, M.; Tommonaro, G. Marine bacteria associated with sponge as source of cyclic peptides. Biomol. Eng. 2003, 20, 311–316. [Google Scholar] [CrossRef]
- Mitova, M.; Tommonaro, G.; Hentschel, U.; Müller, W.E.G.; De Rosa, S. Exocellular Cyclic Dipeptides from a Ruegeria Strain Associated with Cell Cultures of Suberites domuncula. Mar. Biotechnol. 2004, 6, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Huayue, L.; Byung Cheol, L.; Tae Sung, K.; Kyung Sook, B.; Jongki, H.; Sang Ho, C.; Baoquan, B.; Jee Hyung, J. Bioactive Cyclic Dipeptides from a Marine Sponge-Associated Bacterium, Psychrobacter sp. Biomol. Ther. 2008, 16, 356–363. [Google Scholar]
- Sawangwong, P.; Wattanadilok, R.; Kijjoa, A.; Silva, A.M.S.; Eaton, G.; Herz, W. Secondary metabolites from a marine sponge Cliona patera. Biochem. Syst. Ecol. 2008, 36, 493–496. [Google Scholar] [CrossRef]
- Abbamondi, G.R.; De Rosa, S.; Iodice, C.; Tommonaro, G. Cyclic Dipeptides Produced by Marine Sponge-Associated Bacteria as Quorum Sensing Signals. Nat. Prod. Commun. 2014, 9. [Google Scholar] [CrossRef]
- Jayatilake, G.S.; Thornton, M.P.; Leonard, A.C.; Grimwade, J.E.; Baker, B.J. Metabolites from an Antarctic Sponge-Associated Bacterium, Pseudomonas aeruginosa. J. Nat. Prod. 1996, 59, 293–296. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.N.; Lankalapalli, R.S.; Kumar, B.S.D. In Vitro Antibacterial Screening of Six Proline-Based Cyclic Dipeptides in Combination with β-Lactam Antibiotics Against Medically Important Bacteria. Appl. Biochem. Biotechnol. 2014, 173, 116–128. [Google Scholar] [CrossRef]
- Smaoui, S.; Mathieu, F.; Elleuch, L.; Coppel, Y.; Merlina, G.; Karray-Rebai, I.; Mellouli, L. Taxonomy, purification and chemical characterization of four bioactive compounds from new Streptomyces sp. TN256 strain. World J. Microbiol. Biotechnol. 2012, 28, 793–804. [Google Scholar] [CrossRef] [PubMed]
- Sansinenea, E.; Salazar, F.; Jiménez, J.; Mendoza, Á.; Ortiz, A. Diketopiperazines derivatives isolated from Bacillus thuringiensis and Bacillus endophyticus, establishment of their configuration by X-ray and their synthesis. Tetrahedron Lett. 2016, 57, 2604–2607. [Google Scholar] [CrossRef]
- Yonezawa, K.; Yamada, K.; Kouno, I. New Diketopiperazine Derivatives Isolated from Sea Urchin-Derived Bacillus sp. Chem. Pharm. Bull. 2011, 59, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Cimmino, A.; Puopolo, G.; Perazzolli, M.; Andolfi, A.; Melck, D.; Pertot, I.; Evidente, A. Cyclo(L-pro-L-tyr), The Fungicide Isolated From Lysobacter Capsici AZ78: A Structure–Activity Relationship Study. Chem. Heterocycl. Compd. 2014, 50, 290–295. [Google Scholar] [CrossRef]
- Puopolo, G.; Cimmino, A.; Palmieri, M.C.; Giovannini, O.; Evidente, A.; Pertot, I. Lysobacter capsici AZ78 produces cyclo(l-Pro-l-Tyr), a 2,5-diketopiperazine with toxic activity against sporangia of Phytophthora infestans and Plasmopara viticola. J. Appl. Microbiol. 2014, 117, 1168–1180. [Google Scholar] [CrossRef]
- Degrassi, G.; Aguilar, C.; Bosco, M.; Zahariev, S.; Pongor, S.; Venturi, V. Plant Growth-Promoting Pseudomonas putida WCS358 Produces and Secretes Four Cyclic Dipeptides: Cross-Talk with Quorum Sensing Bacterial Sensors. Curr. Microbiol. 2002, 45, 250–254. [Google Scholar] [CrossRef]
- González, J.E.; Keshavan, N.D. Messing with Bacterial Quorum Sensing. Microbiol. Mol. Biol. Rev. 2006, 70, 859. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Castro, R.; Díaz-Pérez, C.; Martínez-Trujillo, M.; del Río, R.E.; Campos-García, J.; López-Bucio, J. Transkingdom signaling based on bacterial cyclodipeptides with auxin activity in plants. Proc. Natl. Acad. Sci. USA 2011, 108, 7253. [Google Scholar] [CrossRef] [PubMed]
- Holden, M.T.G.; Ram Chhabra, S.; De Nys, R.; Stead, P.; Bainton, N.J.; Hill, P.J.; Manefield, M.; Kumar, N.; Labatte, M.; England, D.; et al. Quorum-sensing cross talk: Isolation and chemical characterization of cyclic dipeptides from Pseudomonas aeruginosa and other Gram-negative bacteria. Mol. Microbiol. 1999, 33, 1254–1266. [Google Scholar] [CrossRef]
- Wang, J.-H.; Quan, C.-S.; Qi, X.-H.; Li, X.; Fan, S.-D. Determination of diketopiperazines of Burkholderia cepacia CF-66 by gas chromatography–mass spectrometry. Anal. Bioanal. Chem. 2010, 396, 1773–1779. [Google Scholar] [CrossRef] [PubMed]
- Ryan, R.P.; Dow, J.M. Diffusible signals and interspecies communication in bacteria. Microbiology 2008, 154, 1845–1858. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, W.; Xu, S.X.; Magarvey, N.A.; McCormick, J.K. Lactobacillus reuteri-produced cyclic dipeptides quench agr-mediated expression of toxic shock syndrome toxin-1 in staphylococci. Proc. Natl. Acad. Sci. USA 2011, 108, 3360. [Google Scholar] [CrossRef]
- Teasdale, M.E.; Donovan, K.A.; Forschner-Dancause, S.R.; Rowley, D.C. Gram-Positive Marine Bacteria as a Potential Resource for the Discovery of Quorum Sensing Inhibitors. Mar. Biotechnol. 2011, 13, 722–732. [Google Scholar] [CrossRef]
- Fuqua, C.; Parsek, M.R.; Greenberg, E.P. Regulation of Gene Expression by Cell-to-Cell Communication: Acyl-Homoserine Lactone Quorum Sensing. Annu. Rev. Genet. 2001, 35, 439–468. [Google Scholar] [CrossRef]
- Smith, D.; Wang, J.-H.; Swatton, J.E.; Davenport, P.; Price, B.; Mikkelsen, H.; Stickland, H.; Nishikawa, K.; Gardiol, N.; Spring, D.R.; et al. Variations on a Theme: Diverse N-Acyl Homoserine Lactone-Mediated Quorum Sensing Mechanisms in Gram-Negative Bacteria. Sci. Prog. 2006, 89, 167–211. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.W.; Schupp, P.J.; Baillie, H.J.; Charlton, T.S.; de Nys, R.; Kjelleberg, S.; Steinberg, P.D. Evidence for acyl homoserine lactone signal production in bacteria associated with marine sponges. Appl. Environ. Microbiol. 2004, 70, 4387–4389. [Google Scholar] [CrossRef]
- Bin Saidin, J.; Abd Wahid, M.E.; Le Pennec, G. Characterization of the in vitro production of N-acyl homoserine lactones by cultivable bacteria inhabiting the sponge Suberites domuncula. J. Mar. Biol. Assoc. UK 2016, 97, 119–127. [Google Scholar] [CrossRef]
- Bose, U.; Ortori, C.A.; Sarmad, S.; Barrett, D.A.; Hewavitharana, A.K.; Hodson, M.P.; Fuerst, J.A.; Shaw, P.N. Production of N-acyl homoserine lactones by the sponge-associated marine actinobacteria Salinispora arenicola and Salinispora pacifica. FEMS Microbiol. Lett. 2017, 364, fnx002. [Google Scholar] [CrossRef][Green Version]
- Britstein, M.; Saurav, K.; Teta, R.; Sala, G.D.; Bar-Shalom, R.; Stoppelli, N.; Zoccarato, L.; Costantino, V.; Steindler, L. Identification and chemical characterization of N-acyl-homoserine lactone quorum sensing signals across sponge species and time. FEMS Microbiol. Ecol. 2017, 94, fix182. [Google Scholar] [CrossRef]
- Kanoh, S.; Rubin, B.K. Mechanisms of action and clinical application of macrolides as immunomodulatory medications. Clin. Microbiol. Rev. 2010, 23, 590–615. [Google Scholar] [CrossRef]
- Karpiński, T.M. Marine Macrolides with Antibacterial and/or Antifungal Activity. Mar. Drugs 2019, 17, 241. [Google Scholar] [CrossRef]
- Tareq, F.S.; Kim, J.H.; Lee, M.A.; Lee, H.-S.; Lee, J.-S.; Lee, Y.-J.; Shin, H.J. Antimicrobial Gageomacrolactins Characterized from the Fermentation of the Marine-Derived Bacterium Bacillus subtilis under Optimum Growth Conditions. J. Agric. Food Chem. 2013, 61, 3428–3434. [Google Scholar] [CrossRef]
- Lu, X.L.; Xu, Q.Z.; Shen, Y.H.; Liu, X.Y.; Jiao, B.H.; Zhang, W.D.; Ni, K.Y. Macrolactin S, a novel macrolactin antibiotic from marine Bacillus sp. Nat. Prod. Res. 2008, 22, 342–347. [Google Scholar] [CrossRef]
- Yoo, J.-S.; Zheng, C.-J.; Lee, S.; Kwak, J.-H.; Kim, W.-G. Macrolactin N, a new peptide deformylase inhibitor produced by Bacillus subtilis. Bioorganic Med. Chem. Lett. 2006, 16, 4889–4892. [Google Scholar] [CrossRef]
- Gao, C.; Chen, X.; Yu, L.; Jiang, L.; Pan, D.; Jiang, S.; Gan, Y.; Liu, Y.; Yi, X. New 24-Membered Macrolactins Isolated from Marine Bacteria Bacillus siamensis as Potent Fungal Inhibitors against Sugarcane Smut. J. Agric. Food Chem. 2021, 69, 4392–4401. [Google Scholar] [CrossRef]
- Zotchev, S.B.; Stepanchikova, A.V.; Sergeyko, A.P.; Sobolev, B.N.; Filimonov, D.A.; Poroikov, V.V. Rational Design of Macrolides by Virtual Screening of Combinatorial Libraries Generated through in Silico Manipulation of Polyketide Synthases. J. Med. Chem. 2006, 49, 2077–2087. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, H.K.; Kim, K.M.; Kim, C.K.; Jeong, M.H.; Ko, C.Y.; Moon, K.H.; Kang, J.S. Antibacterial activities of macrolactin a and 7-O-succinyl macrolactin a from Bacillus polyfermenticus KJS-2 against vancomycin-resistant enterococci and methicillin-resistant Staphylococcus aureus. Arch. Pharmacal Res. 2011, 34, 147–152. [Google Scholar] [CrossRef]
- Li, W.; Tang, X.-X.; Yan, X.; Wu, Z.; Yi, Z.-W.; Fang, M.-J.; Su, X.; Qiu, Y.-K. A new macrolactin antibiotic from deep sea-derived bacteria Bacillus subtilis B5. Nat. Prod. Res. 2016, 30, 2777–2782. [Google Scholar] [CrossRef]
- Weiner, R.M.; Segall, A.M.; Colwell, R.R. Characterization of a Marine Bacterium Associated with Crassostrea virginica (the Eastern Oyster). Appl. Environ. Microbiol. 1985, 49, 83–90. [Google Scholar] [CrossRef]
- Mincer, T.J.; Jensen, P.R.; Kauffman, C.A.; Fenical, W. Widespread and persistent populations of a major new marine actinomycete taxon in ocean sediments. Appl. Environ. Microbiol. 2002, 68, 5005–5011. [Google Scholar] [CrossRef]
- Reasoner, D.J.; Geldreich, E.E. A New Medium for the Enumeration and Subculture of Bacteria from Potable Water. Appl. Environ. Microbiol. 1985, 49, 1–7. [Google Scholar] [CrossRef]
- Kuster, E.; William, S.T. Selection of media for isolation of streptomyces. Nature 1964, 202, 928–929. [Google Scholar] [CrossRef]
- Olson, J.B.; Lord, C.C.; McCarthy, P.J. Improved Recoverability of Microbial Colonies from Marine Sponge Samples. Microb. Ecol. 2000, 40, 139–147. [Google Scholar] [CrossRef]
- Lane, D.J.; Pace, B.; Olsen, G.J.; Stahl, D.A.; Sogin, M.L.; Pace, N.R. Rapid determination of 16S ribosomal RNA sequences for phylogenetic analyses. Proc. Natl. Acad. Sci. USA 1985, 82, 6955. [Google Scholar] [CrossRef]
- Lane, D.J. 16S/23S rRNA sequencing. In Nucleic Acid Techniques in Bacterial Systematics; Stackebrandt, E., Goodfellow, M., Eds.; John Wiley & Sons: New York, NY, USA, 1991. [Google Scholar]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016; Available online: http://www.R-project.org/ (accessed on 5 December 2018).
- Oksanen, J.; Blanchett, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.M.; Wagner, H. Vegan: Community Ecology Package. R Package 2.4.0. 2016. Available online: https://CRAN.R-project.org/package=vegan (accessed on 5 December 2018).
- Joachimiak, M.P.; Weisman, J.L.; May, B.C.H. JColorGrid: Software for the visualization of biological measurements. BMC Bioinform. 2006, 7, 225. [Google Scholar] [CrossRef]
- EUCAST. Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution. Clin. Microbiol. Infect. 2003, 9, ix–xv. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Guinea, J.; Cuenca-Estrell, M.; Meletiadis, J.; Mouton, J.W.; Lagrou, K.; Howard, S.J.; EUCAST-AFST. EUCAST Definitive Document E.DEF 7.3: Method for the Determination of Broth Dilution Minimum Inhibitory Concentrations of Antifungal Agents for Yeasts; EUCAST: Växjö, Sweden, 2015. [Google Scholar]
- Arendrup, M.C.; Guinea, J.; Cuenca-Estrell, M.; Meletiadis, J.; Mouton, J.W.; Lagrou, K.; Howard, S.J.; EUCAST-AFST. EUCAST Definitive Documnet E.DEF 9.3: Method for the Determination of Broth Dilution Minimum Inhibitory Concentrations of Antifungal Agents for Conidia Forming Moulds; EUCAST: Växjö, Sweden, 2015. [Google Scholar]



| Isolated Strains | Genus | Inhibition Zone Diameter (mm) | ||||||
|---|---|---|---|---|---|---|---|---|
| Gram-Negative | Gram-Positive | Yeast | ||||||
| SE | EC | PA | SA | EF | BS | CA | ||
| MA_AMC_32 | Bacillus | - | - | - | 10 ± 2 | - | - | - |
| MA_AMQ_66 | Bacillus | - | 7 ± 2 | - | - | - | 8 ± 3 | - |
| MA_AXT_69 | Bacillus | 7 ± 2 | - | - | - | - | - | - |
| MA_AXT_70 | Bacillus | 5 ± 2 | - | 8 ± 3 | 6 ± 3 | - | 8 ± 3 | - |
| MA_AXC_75 | Bacillus | - | 7 ± 2 | - | - | - | - | - |
| MA_CIS_78 | Bacillus | - | - | - | - | - | - | 6 ± 2 |
| M1_CRV_171 | Bacillus | - | 5 ± 2 | 8 ± 2 | 7 ± 3 | 10 ± 3 | - | 6 ± 2 |
| M1_DAS_199 | Bacillus | - | - | 12 ± 3 | - | - | - | - |
| M1_HAA_234 | Bacillus | - | - | - | - | 5 ± 1 | 11 ± 3 | 7 ± 2 |
| M1_HAA_246 | Bacillus | - | - | - | 6 ± 2 | - | - | - |
| M1_NIS_274 | Bacillus | 5 ± 2 | - | - | - | - | - | - |
| R2A_NIS_276 | Bacillus | - | - | - | - | - | 12 ± 3 | - |
| R2A_RHG_312 | Bacillus | - | - | - | 5 ± 2 | - | - | - |
| R2A_SPV_326 | Bacillus | - | - | 7 ± 2 | - | 5 ± 2 | 7 ± 2 | - |
| R2A_SPV_338 | Bacillus | - | 4 ± 1 | - | - | 3 ± 1 | - | 6 ± 2 |
| SCA_SPS_344 | Bacillus | - | - | - | - | - | 4 ± 1 | - |
| SCA_TES_347 | Bacillus | - | - | - | - | - | - | 4 ± 1 |
| AIA_TEA_438 | Bacillus | - | - | - | - | 4 ± 1 | - | - |
| AIA_XES_454 | Bacillus | - | - | 15± 3 | - | - | - | - |
| AIA_XES_458 | Bacillus | 12 ± 3 | - | - | - | - | - | 10 ± 3 |
| M1_HAF_272 | Bacillus | - | 10 ± 3 | - | - | - | 8 ± 3 | - |
| AIA_SPV_375 | Brevibacterium | - | - | - | - | 12 ± 3 | - | - |
| AIA_HAS_264 | Brevibacterium | - | - | - | - | - | 4 ± 1 | - |
| M1_AXC_175 | Brevibacterium | - | - | - | - | 6 ± 2 | 10 ± 3 | - |
| MA_AMQ_34 | Brevibacterium | - | 8 ± 3 | - | - | - | 9 ± 2 | - |
| R2A_AXC_194 | Kocuria | 10 ± 4 | - | - | 14 ± 4 | - | - | 7 ± 2 |
| MA_AMC_87 | Microbacterium | - | - | - | 6 ± 2 | - | - | - |
| R2A_SPS_90 | Microbacterium | - | - | - | 4 ± 1 | - | - | - |
| M1_AXT_2 | Microbulbifer | - | - | - | 5 ± 1 | - | - | - |
| R2A_DAS_4 | Microbulbifer | - | - | 3 ± 1 | - | - | - | - |
| AIA_TES_7 | Microbulbifer | - | 12 ± 4 | - | - | - | - | - |
| R2A_CIS_91 | Micrococcus | - | - | - | - | - | - | 9 ± 2 |
| SCA_CLR_217 | Micrococcus | 8 ± 3 | - | - | - | - | - | - |
| M1_AXT_88 | Micrococcus | 11 ± 3 | - | - | - | - | - | - |
| R2A_CRV_10 | Micromonospora | - | - | 13 ± 3 | - | - | 8 ± 2 | - |
| SCA_HAS_11 | Micromonospora | - | - | - | 6 ± 2 | 6 ± 2 | - | 4 ± 1 |
| M1_AXC_17 | Pseudoalteromonas | - | - | - | - | 8 ± 3 | - | - |
| MA_AMC_15 | Pseudoalteromonas | 6 ± 2 | - | - | 10 ± 4 | 8 ± 4 | - | 4 ± 2 |
| R2A_CIS_18 | Pseudoalteromonas | - | - | - | - | - | 7 ± 2 | - |
| SCA_CRV_19 | Pseudoalteromonas | - | - | - | - | 6 ± 1 | - | - |
| MA_AMQ_98 | Pseudomonas | - | - | 6 ± 2 | - | - | - | 4 ± 1 |
| M1_AXT_131 | Pseudomonas | - | 12 ± 4 | - | - | - | - | - |
| R2A_AXC_259 | Pseudomonas | - | - | - | - | - | 6 ± 2 | - |
| SCA_CLR_279 | Pseudomonas | - | - | 9 ± 3 | - | - | - | - |
| AIA_HAA_313 | Pseudomonas | - | 11 ± 4 | - | - | 9 ± 2 | 14 ± 4 | - |
| MA_AMC_93 | Pseudovibrio | - | - | - | 3 ± 1 | - | - | - |
| MA_AMC_33 | Pseudovibrio | - | - | 6 ± 3 | - | - | - | - |
| MA_AMQ_100 | Pseudovibrio | - | - | - | - | 12 ± 4 | - | - |
| MA_AXT_177 | Pseudovibrio | 13 ± 4 | - | - | 10 ± 3 | - | - | - |
| MA_AXC_181 | Pseudovibrio | 13 ± 4 | - | - | - | - | - | - |
| MA_CIS_184 | Pseudovibrio | 11 ± 4 | - | - | - | 8 ± 2 | 10 ± 3 | - |
| MA_CIS_186 | Pseudovibrio | - | - | 5 ± 1 | - | - | - | - |
| MA_CIS_195 | Pseudovibrio | - | - | - | - | - | 7 ± 1 | - |
| MA_CRV_231 | Pseudovibrio | - | 8 ± 3 | - | - | - | - | - |
| M1_DAS_236 | Pseudovibrio | - | 13 ± 3 | - | - | 7 ± 2 | 10 ± 3 | - |
| M1_HAA_265 | Pseudovibrio | - | - | - | - | - | - | 5 ± 1 |
| R2A_RHG_301 | Pseudovibrio | - | - | 6 ± 2 | - | - | - | - |
| SCA_TES_374 | Pseudovibrio | 8 ± 2 | - | - | 14 ± 4 | - | - | - |
| AIA_TEA_401 | Pseudovibrio | - | 15 ± 4 | - | - | - | - | - |
| MA_AMQ_136 | Ruegeria | - | - | 6 ± 2 | - | - | - | - |
| MA_AXT_139 | Ruegeria | 4 ± 1 | - | - | 7 ± 2 | 7 ± 2 | 13 ± 4 | - |
| MA_CIS_145 | Ruegeria | - | 13 ± 4 | - | - | 7 ± 2 | - | - |
| M1_DAS_153 | Ruegeria | 3 ± 1 | - | - | - | - | - | - |
| R2A_SPV_381 | Ruegeria | - | 6 ± 2 | - | - | - | - | - |
| MA_AMC_38 | Streptomyces | - | - | 3 ± 1 | - | 8 ± 4 | - | - |
| MA_AMQ_39 | Streptomyces | 7 ± 2 | - | - | 5 ± 2 | - | - | - |
| M1_AXT_41 | Streptomyces | - | 13 ± 3 | - | - | - | - | - |
| M1_CIS_51 | Streptomyces | - | - | - | 6 ± 2 | - | 10 ± 3 | - |
| R2A_CLR_53 | Streptomyces | - | 4 ± 1 | - | 4 ± 2 | - | - | - |
| R2A_DAS_58 | Streptomyces | - | - | - | - | - | 8 ± 3 | - |
| R2A_HAS_60 | Streptomyces | - | - | - | - | 7 ± 2 | - | - |
| SCA_HAF_63 | Streptomyces | - | 9 ± 3 | - | - | - | - | - |
| SCA_RHG_65 | Streptomyces | - | - | - | 7 ± 2 | - | - | - |
| AIA_SPV_83 | Streptomyces | - | 6 ± 2 | - | - | - | - | - |
| AIA_SPS_85 | Streptomyces | - | - | - | - | - | 4 ± 2 | - |
| AIA_TEA_127 | Streptomyces | - | - | - | - | 5 ± 2 | - | - |
| AIA_TES_126 | Streptomyces | - | - | - | - | - | 14 ± 4 | - |
| AIA_TES_125 | Streptomyces | - | 4 ±1 | - | 7 ± 2 | - | - | - |
| OLIGO_XES_128 | Streptomyces | 3 ± 1 | - | - | - | - | - | - |
| SWA_XES_129 | Streptomyces | - | 8 ± 3 | - | - | - | 10 ± 3 | - |
| AIA_SPV_84 | Streptomyces | - | - | - | - | - | 10 ± 3 | 7 ± 2 |
| MA_AMC_44 | Vibrio | - | - | - | - | 5 ± 3 | - | 6 ± 2 |
| R2A_SPS_117 | Vibrio | - | - | - | - | - | 8 ± 3 | - |
| MA_AMQ_46 | Vibrio | - | - | - | 10 ± 3 | - | - | - |
| AIA_TEA_122 | Vibrio | - | - | - | - | 4 ± 2 | - | - |
| M1_RHG_114 | Vibrio | - | - | - | - | - | - | 4 ± 1 |
| M1_HAA_109 | Vibrio | - | - | 12 ± 3 | - | - | - | - |
| MA_CIS_55 | Vibrio | - | - | - | - | 5 ± 1 | - | - |
| SCA_TES_120 | Vibrio | 11 ± 3 | - | - | - | 17 ± 3 | - | - |
| M1_CLR_86 | Vibrio | - | - | 13 ± 4 | - | - | - | - |
| Ampicillin | 18 ± 3 | 20 ± 4 | - | 25 ± 4 | 21 ± 3 | 23 ± 3 | na | |
| Kanamycin | 12 ± 2 | 17 ± 3 | - | 19 ± 3 | 18 ± 2 | 19 ± 2 | na | |
| Tetracycline | 10 ± 2 | 19 ± 3 | - | 22 ± 3 | 15 ± 2 | 21 ± 3 | na | |
| Miconazole | na | na | na | na | na | na | 17 ± 2 | |
| Reference Microorganisms | MIC (µg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | Amp | Kan | Tet | Mic | |
| Gram negative bacteria | ||||||||
| E. coli ATCC 25922 | - | 64 | 16 | 32 | 8 | 16 | 4 | na |
| S. enterica ATCC 13076 | - | - | >256 | >256 | 16 | 64 | 32 | na |
| P. aeruginosa ATCC 27853 | - | 8 | 64 | 64 | >256 | >256 | 64 | na |
| P. putida MISR 71218 | - | 128 | 128 | 128 | >256 | 64 | 64 | na |
| V. parahaemolyticus MISR 21116 | >256 | 128 | 64 | 128 | 64 | 32 | 32 | na |
| V. vulnificus MISR 20716 | - | - | 128 | 256 | 32 | 64 | 32 | na |
| V. alginolyticus MISR 30816 | - | - | 256 | 128 | >256 | 32 | 64 | na |
| Gram positive bacteria | ||||||||
| E. faecalis ATCC 29212 | - | - | >256 | >256 | 8 | 128 | 32 | na |
| S. aureus ATCC 25923 | >256 | 16 | 32 | 64 | 16 | 16 | 4 | na |
| B. subtilis ATCC 6633 | - | 128 | 64 | 64 | 8 | 16 | 32 | na |
| B. cereus MISR 12818 | - | 128 | 64 | 64 | 32 | 16 | 64 | na |
| Rhodococcus sp. MISR 16518 | - | 32 | 64 | 128 | 16 | 8 | 32 | na |
| Fungi | ||||||||
| C. albicans ATCC 10231 | - | 128 | 64 | 64 | na | na | na | 32 |
| A. niger MISR 11215 | - | 64 | 64 | 64 | na | na | na | 8 |
| F. oxisporum MISR 20415 | >256 | 64 | 64 | 64 | na | na | na | 32 |
| R. solani MISR 11115 | >256 | 64 | 64 | 64 | na | na | na | 32 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dat, T.T.H.; Cuc, N.T.K.; Cuong, P.V.; Smidt, H.; Sipkema, D. Diversity and Antimicrobial Activity of Vietnamese Sponge-Associated Bacteria. Mar. Drugs 2021, 19, 353. https://doi.org/10.3390/md19070353
Dat TTH, Cuc NTK, Cuong PV, Smidt H, Sipkema D. Diversity and Antimicrobial Activity of Vietnamese Sponge-Associated Bacteria. Marine Drugs. 2021; 19(7):353. https://doi.org/10.3390/md19070353
Chicago/Turabian StyleDat, Ton That Huu, Nguyen Thi Kim Cuc, Pham Viet Cuong, Hauke Smidt, and Detmer Sipkema. 2021. "Diversity and Antimicrobial Activity of Vietnamese Sponge-Associated Bacteria" Marine Drugs 19, no. 7: 353. https://doi.org/10.3390/md19070353
APA StyleDat, T. T. H., Cuc, N. T. K., Cuong, P. V., Smidt, H., & Sipkema, D. (2021). Diversity and Antimicrobial Activity of Vietnamese Sponge-Associated Bacteria. Marine Drugs, 19(7), 353. https://doi.org/10.3390/md19070353







