Effects of Daily Kelp (Laminaria japonica) Intake on Body Composition, Serum Lipid Levels, and Thyroid Hormone Levels in Healthy Japanese Adults: A Randomized, Double-Blind Study
Abstract
:1. Introduction
2. Results
2.1. Subject Characteristics
2.2. Daily Energy and Nutrient Intakes
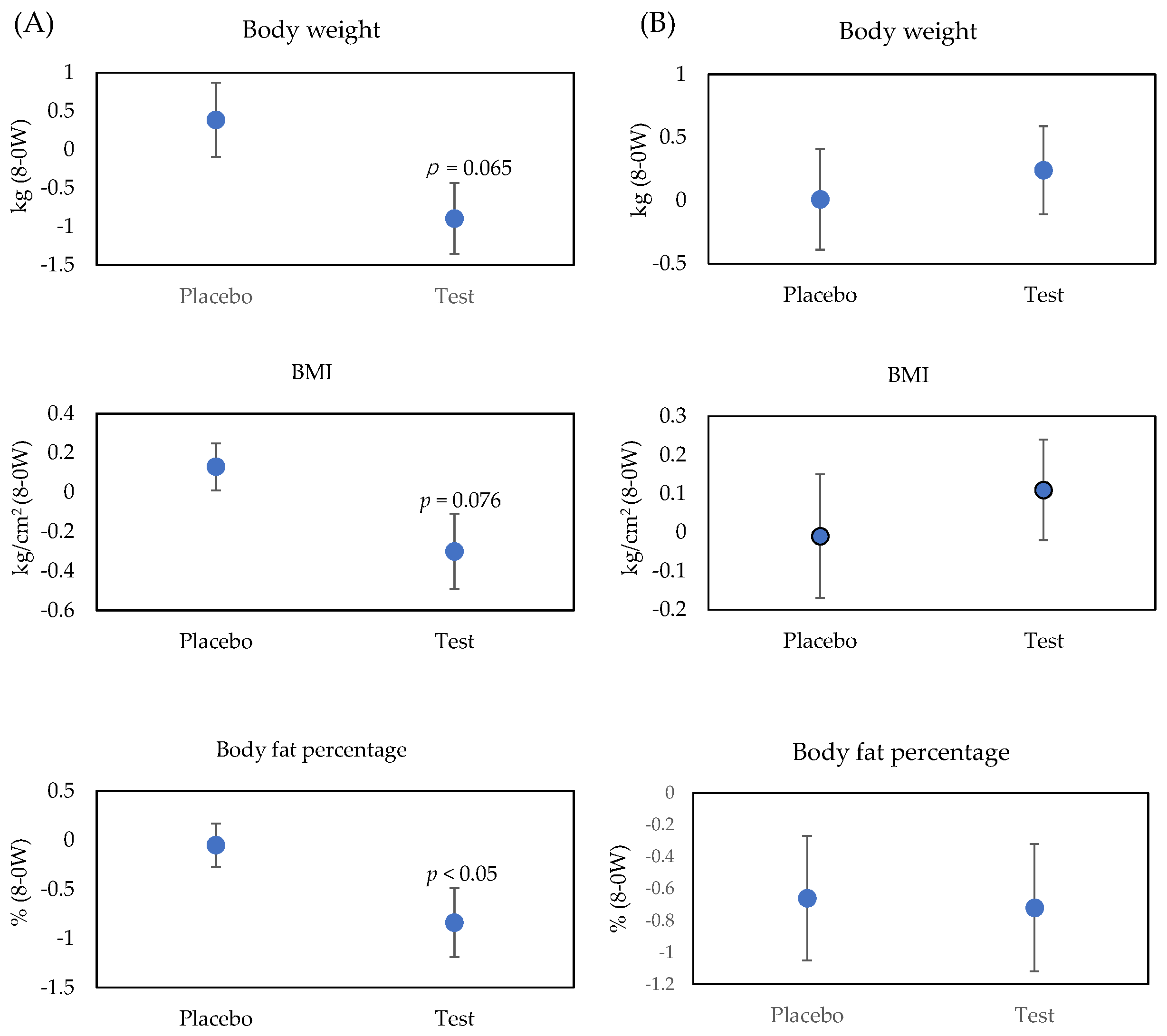
2.3. Anthropometric Measurements and Visceral Fat Area
2.4. Serum Lipid Values
2.5. Serum Thyroid Hormones
3. Discussion
4. Materials and Methods
4.1. Subjects
- Aged 20–59 years;
- BMI 25–30 kg/m2.
- Having a history of a severe circulatory, respiratory, digestive, urinary system, endocrine, or blood disease;
- Receiving therapeutic medication at the time of the agreement of informed consent;
- Having a food or drug allergy;
- Will not eat kelp;
- Regular consumption of foods for specified health use, foods with functional claims, supplements, and health foods rich in dietary fiber;
- Refraining from or restricted from iodine-rich foods;
- Having an excessive alcohol intake (more than 30 g/d of alcohol);
- Having extremely irregular eating habits or irregular rhythms of life such as shift workers and late-night workers;
- Participating in another study involving the intake of drugs or food within one month before screening tests or planning to participate in another study involving the intake of drugs or food after the agreement of informed consent;
- Any male who donated 400 mL of whole blood 3 months before the start of the trial;
- Any female who donated 400 mL of whole blood 4 months before the start of the trial.
4.2. Test Supplements
4.3. Study Design and Intervention
4.4. Clinical Analyses
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| HDL | high-density lipoprotein |
| ITT | intention to treat |
| LDL | low-density lipoprotein |
| MS | metabolic syndrome |
| PP | per-protocol |
| SE | standard error of the mean |
References
- Klop, B.; Elte, J.W.F.; Cabezas, M.C. Dyslipidemia in obesity: Mechanisms and potential targets. Nutrients 2013, 5, 1218–1240. [Google Scholar] [CrossRef] [Green Version]
- Daskalopoulou, S.S.; Athyros, V.G.; Kolovou, G.D.; Anagnostopoulou, K.K.; Mikhailidis, D.P. Definitions of metabolic syndrome: Where are we now? Curr. Vasc. Pharm. 2006, 4, 185–197. [Google Scholar] [CrossRef] [PubMed]
- InterAct Consortium. Dietary fibre and incidence of type 2 diabetes in eight European countries: The EPIC-InterAct Study and a meta-analysis of prospective studies. Diabetologia 2015, 58, 1394–1408. [Google Scholar] [CrossRef] [Green Version]
- Yao, B.; Fang, H.; Xu, W.; Yan, Y.; Xu, H.; Liu, Y.; Mo, M.; Zhang, H.; Zhao, Y. Dietary fiber intake and risk of type 2 diabetes: A dose response analysis of prospective studies. Eur. J. Epidemiol. 2014, 29, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, A.; Mann, J.; Cummings, J.; Winter, N.; Mete, E.; Morenga, L.T. Carbohydrate quality and human health: A series of systematic reviews and meta-analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Qian, Y.; Pan, Y.; Li, P.; Yang, J.; Ye, X.; Xu, G. Association between dietary fiber intake and risk of coronary heart disease: A meta-analysis. Clin. Nutr. 2015, 34, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health, Labour and Welfare, Japan. The National Health and Nutrition Survey in Japan. 2019. Available online: https://www.mhlw.go.jp/content/000710991.pdf (accessed on 6 May 2021).
- Adrian, J. Les algues laminaires sont-elles acceptables en alimentation humaine? Med. Nutr. 1985, 21, 50–55. [Google Scholar]
- Michel, C.; Lahaye, M.; Bonnet, C.; Mabeau, S.; Barry, J.L. In vitro fermentation by human faecal bacteria of total and purified dietary fibres from brown seaweeds. Br. J. Nutr. 1996, 75, 263–280. [Google Scholar] [CrossRef]
- Yoshinaga, J.; Morita, M.; Yukawa, M.; Shiraishi, K.; Kawamura, H. Certified reference material for analytical quality assurance of minor and trace elements in food and related matrixes based on a typical Japanese diet: Interlaboratory study. J. Aoac. Int. 2001, 84, 1202–1208. [Google Scholar] [CrossRef]
- Iso, H.; Kubota, Y. Nutrition and disease in the Japan Collaborative Cohort Study for Evaluation of Cancer (JACC). Asian Pac. J. Cancer Prev. 2007, 8, 35–80. [Google Scholar]
- Yeh, C.J.; Chang, H.Y.; Pan, W.H. Time trend of obesity, the metabolic syndrome and related dietary pattern in Taiwan: From NAHSIT 1993-1996 to NAHSIT 2005-2008. Asia Pac. J. Clin. Nutr. 2011, 20, 292–300. [Google Scholar] [PubMed]
- Lee, H.Y.; Won, J.C.; Kang, Y.J.; Yoon, S.H.; Choi, E.O.; Bae, J.Y.; Sung, M.H.; Kim, H.-R.; Yang, J.H.; Oh, J.; et al. Type 2 diabetes in urban and rural districts in Korea: Factors associated with prevalence difference. J. Korean Med. Sci. 2010, 25, 1777–1783. [Google Scholar] [CrossRef]
- Vaugelade, P.; Hoebler, C.; Bernard, F.; Guillon, F.; Lahaye, M.; Duee, P.H.; Darcy-Vrillon, B. Non-starch polysaccharides extracted from seaweed can modulate intestinal absorption of glucose and insulin response in the pig. Reprod. Nutr. Dev. 2000, 40, 33–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujita, T.; Fujii, Y.; Goto, B.; Miyauchi, A.; Takagi, Y. Peripheral computed tomography (pQCT) detected short-term effect of AAACa (heated oyster shell with heated algal ingredient HAI): A double-blind comparison with CaCO3 and placebo. J. Bone Min. Metab. 2000, 18, 212–215. [Google Scholar] [CrossRef]
- Wang, H.; Ooi, E.V.; Ang, P.O., Jr. Antiviral activities of extracts from Hong Kong seaweeds. J. Zhejiang Univ. Sci. B 2008, 9, 969–976. [Google Scholar] [CrossRef] [Green Version]
- Kumar, C.S.; Ganesan, P.; Suresh, P.V.; Bhaskar, N. Seaweeds as a source of nutritionally beneficial compounds a review. J. Food Sci. Technol. 2008, 45, 1–13. [Google Scholar]
- Peñalver, R.; Lorenzo, J.M.; Ros, G.; Amarowicz, R.; Pateiro, M.; Nieto, G. Seaweeds as a Functional Ingredient for a Healthy Diet. Mar. Drugs 2020, 18, 301. [Google Scholar] [CrossRef]
- Abidov, M.; Ramazanov, Z.; Seifulla, R.; Grachev, S. The Effects of Xanthigen in the Weight Management of Obese Premenopausal Women with Non-alcoholic Fatty Liver Disease and Normal Liver Fat. Diabetes Obes. Metab. 2010, 12, 72–81. [Google Scholar] [CrossRef]
- Heo, S.J.; Yoon, W.J.; Kim, K.N.; Ahn, G.N.; Kang, S.M.; Kang, D.H.; Affan, A.; Oh, C.; Jung, W.K.; Jeon, Y.J. Evaluation of anti-inflammatory effect of fucoxanthin isolated from brown algae in lipopolysaccharide-stimulated RAW 264.7 macrophages. Food Chem. Toxicol. 2010, 48, 2045–2051. [Google Scholar] [CrossRef]
- Grasa-López, A.; Miliar-García, A.; Quevedo-Corona, L.; Paniagua-Castro, N.; Escalona-Cardoso, G.; Reyes-Maldonado, E.; Jaramillo-Flores, M. Undaria pinnatifida and fucoxanthin ameliorate lipogenesis and markers of both inflammation and cardiovascular dysfunction in an animal model of diet-induced obesity. Mar. Drugs 2016, 14, 148. [Google Scholar] [CrossRef] [Green Version]
- Kang, M.C.; Kang, N.; Kim, S.Y.; Lima, I.S.; Ko, S.C.; Kim, Y.T.; Kim, Y.B.; Jeung, H.D.; Choi, K.S.; Jeon, Y.J. Popular edible seaweed, Gelidium amansii prevents against diet-induced obesity. Food Chem. Toxicol. 2016, 90, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Yamori, Y.; Miura, A.; Taira, K. Implications from and for food cultures for cardiovascular diseases: Japanese food, particularly Okinawan diets. Pac. J. Clin. Nutr. 2001, 10, 144–145. [Google Scholar] [CrossRef]
- Arasaki, S.; Arasaki, T. Vegetables from the Sea; Japan Publications Inc.: Tokyo, Japan, 1983; p. 196. [Google Scholar]
- Teas, J.; Pino, S.; Critchley, A.T.; Braverman, L.E. Variability of iodine content in common commercially available edible seaweeds. Thyroid 2004, 14, 836–841. [Google Scholar] [CrossRef] [PubMed]
- Nagataki, S. The average of dietary iodine intake due to the ingestion of seaweeds is 1.2 mg/d in Japan. Thyroid 2008, 18, 667–668. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health, Labour and Welfare. Overview of Dietary Reference Intakes for Japanese. 2015. Available online: https://www.mhlw.go.jp/file/06-Seisakujouhou-10900000-Kenkoukyoku/Overview.pdf (accessed on 6 May 2021).
- Yukami, A.; Nakajima, E.; Yoshida, M. Release of Iodine from Kombu to Soup Stock and Infiltration of Iodine from Kombu Soup Stock to Ingredient. Trace Nutr. Res. 2016, 33, 55–58. (In Japanese) [Google Scholar]
- Jensen, M.G.; Kristensen, M.; Astrup, A. Effect of alginate supplementation on weight loss in obese subjects completing a 12-wk energy-restricted diet: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 96, 5–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Odunsi, S.T.; Vazquez-Roque, M.I.; Camilleri, M.; Papathanasopoulos, A.; Clark, M.M.; Wodrich, L.; Lempke, M.; McKinzie, S.; Ryks, M.; Burton, D.; et al. Effect of alginate on satiation, appetite, gastric function, and selected gut satiety hormones in overweight and obesity. Obesity 2010, 18, 1579–1584. [Google Scholar] [CrossRef] [Green Version]
- Link, J.C.; Reue, K. Genetic basis for sex differences in obesity and lipid metabolism. Annu. Rev. Nutr. 2017, 37, 225–245. [Google Scholar] [CrossRef]
- Kokubo, Y.; Okamura, T.; Yoshimasa, Y.; Miyamoto, Y.; Kawanishi, K.; Kotani, Y.; Okayama, A.; Tomoike, H. Impact of metabolic syndrome components on the incidence of cardiovascular disease in a general urban Japanese population: The suita study. Hypertens. Res. 2008, 31, 2027–2035. [Google Scholar] [CrossRef]
- Teas, J.; Baldeón, M.E.; Chiriboga, D.E.; Davis, J.R.; Sarriés, A.J.; Braverman, L.E. Could dietary seaweed reverse the metabolic syndrome? Asia. Pac. J. Clin. Nutr. 2009, 18, 145–154. [Google Scholar]
- Aoe, S.; Ichinose, Y.; Kohyama, N.; Komae, K.; Takahashi, A.; Abe, D.; Yoshioka, T.; Yanagisawa, T. Effects of high β-glucan barley on visceral fat obesity in Japanese individuals: A randomized, double-blind study. Nutrition 2017, 42, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, M.; Jensen, M.G. Dietary fibres in the regulation of appetite and food intake. Importance of viscosity. Appetite 2011, 56, 65–70. [Google Scholar] [CrossRef]
- Jensen, G.M.; Kristensen, M.; Belza, A.; Knudsen, J.C.; Astrup, A. Acute effect of alginate-based preload on satiety feelings, energy intake and gastric emptying rate in healthy subjects. Obesity 2012, 20, 1851–1858. [Google Scholar]
- Chu, W.L.; Phang, S.M. Marine Algae as a Potential Source for Anti-Obesity Agents. Mar. Drugs 2016, 14, 222. [Google Scholar]
- Jenkins, D.J.A.; Kendall, C.W.C.; Vuksan, V.; Vidgen, E.; Parker, T.; Faulkner, D.; Mehling, C.C.; Garsetti, M.; Testolin, G.; Cunnane, S.C.; et al. Soluble fiber intake at a dose approved by the US Food and Drug Administration for a claim of health benefits: Serum lipid risk factors for cardiovascular disease assessed in a randomized controlled crossover trial. Am. J. Clin. Nutr. 2002, 75, 834–839. [Google Scholar] [CrossRef]
- Idota, Y.; Kogure, Y.; Kato, T.; Ogawa, M.; Kobayashi, S.; Kakinuma, C.; Yano, K.; Arakawa, H.; Miyajima, C.; Kasahara, F.; et al. Cholesterol-lowering effect of calcium alginate in rats. Biol. Pharm. Bull. 2016, 39, 62–67. [Google Scholar] [CrossRef] [Green Version]
- Zava, T.T.; Zava, D.T. Assessment of Japanese iodine intake based on seaweed consumption in Japan: A literature-based analysis. Thyroid. Res. 2011, 4, 14. [Google Scholar] [CrossRef] [Green Version]
- Teas, J.; Braverman, L.; Kurzer, M.; Pino, S.; Hurley, T.; Hebert, J. Seaweed and soy: Companion foods in Asian cuisine and their effects on thyroid function in American women. J. Med. Food 2007, 10, 90–100. [Google Scholar] [CrossRef] [Green Version]
- Ministry of Health, Labour and Welfare. Ethical Guidelines for Medical and Health Research Involving Human Subjects. Available online: https://www.mhlw.go.jp/file/06-Seisakujouhou-10600000-Daijinkanboukouseikagakuka/0000080278.pdf (accessed on 6 May 2021).
- Lange, K.W.; Hauser, J.; Nakamura, Y.; Kanaya, S. Dietary seaweeds and obesity. Food Sci. Hum. Wellness 2015, 4, 87–96. [Google Scholar] [CrossRef] [Green Version]
- Prosky, L.; Asp, N.-G.; Schweizer, T.F.; Devries, J.W.; Furuda, I. Determination of insoluble, soluble, and total dietary fiber in foods and food products. J. Assoc. Off. Anal. Chem. 1988, 71, 1017–1023. [Google Scholar]
- Pelkman, C.L.; Navia, J.L.; Miller, A.E.; Pohle, R.J. Novel calcium-gelled, alginate-pectin beverage reduced energy intake in nondieting overweight and obese women: Interactions with dietary restraint status. Am. J. Clin. Nutr. 2007, 86, 1595–1602. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Education, Culture, Sports, Science and Technology. Standard Tables of Food Composition in Japan—2015—(Seventh Revised Version). Available online: https://www.mext.go.jp/en/policy/science_technology/policy/title01/detail01/1374030.htm (accessed on 6 May 2021).
- Asaoka, T.; Iwatsuka, H.; Minowa, H. Effect of “CHOLECUT”, a Drink Containing Depolymerized Sodium Alginate, on Serum Total Cholesterol Levels in Healthy Males. Jpn. J. Nutr. Assess 1996, 13, 460–464. (In Japanese) [Google Scholar]
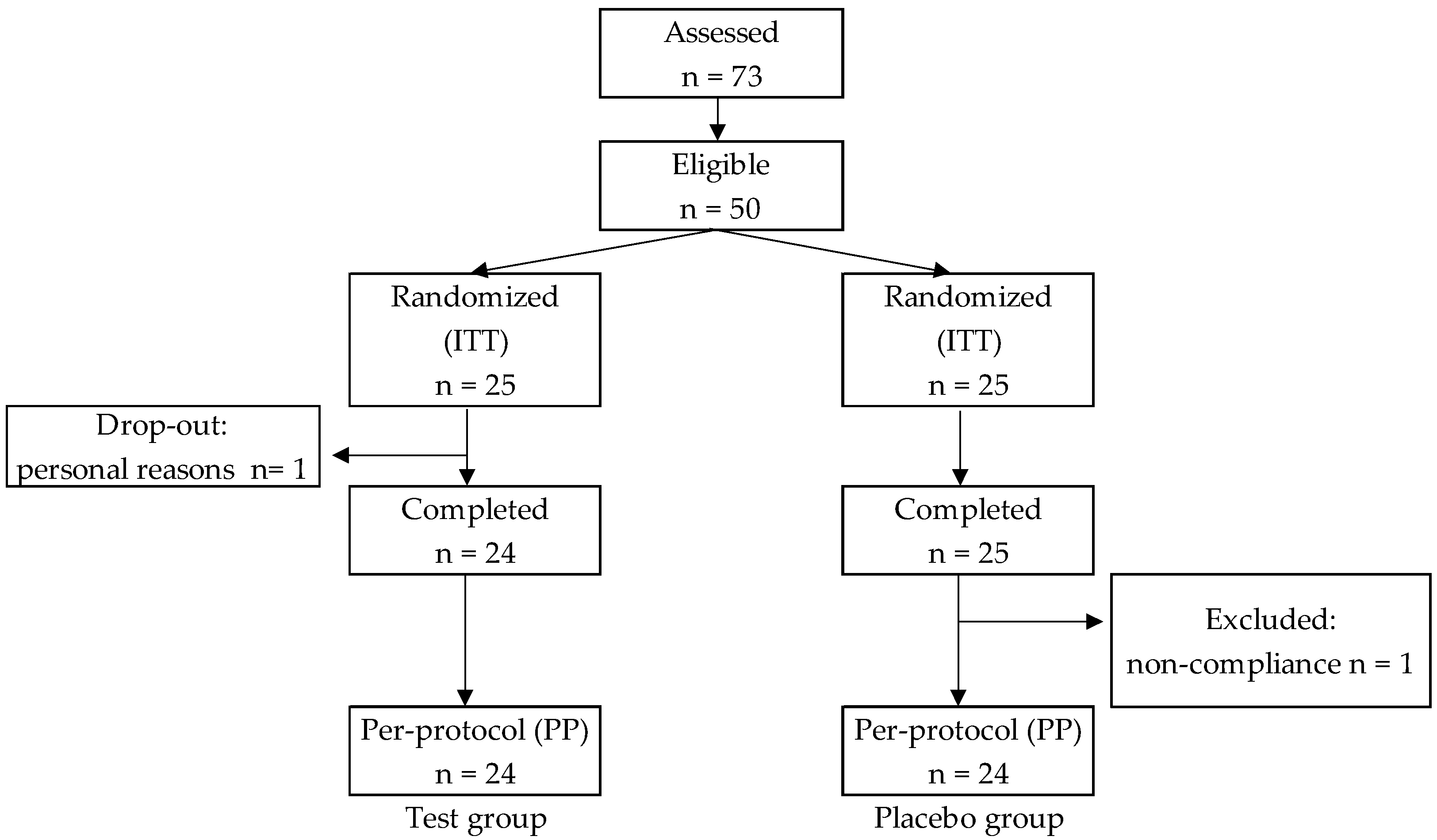
| Placebo | Test | p† | ||
|---|---|---|---|---|
| n | 24 | 24 | ||
| Male | 11 | 12 | ||
| Female | 13 | 12 | ||
| Age (y) | 47.5 ± 1.9 | 47.7 ± 2.1 | 0.96 | |
| Male | 44.9 ± 3.4 | 46.1 ± 3.2 | 0.80 | |
| Female | 49.8 ± 2.0 | 49.3 ± 2.8 | 0.88 | |
| Body weight (kg) | 74.0 ± 1.7 | 74.4 ± 1.4 | 0.86 | |
| Male | 80.9 ± 1.7 | 79.0 ± 1.6 | 0.42 | |
| Female | 68.1 ± 1.5 | 69.8 ± 1.3 | 0.42 | |
| BMI (kg/cm2) | 27.3 ± 0.3 | 27.2 ± 1.2 | 0.79 | |
| Male | 27.1 ± 0.3 | 27.0 ± 0.3 | 0.90 | |
| Female | 27.4 ± 0.5 | 27.3 ± 0.4 | 0.85 | |
| Body fat percentage (%) | 34.3 ± 1.4 | 32.9 ± 1.3 | 0.45 | |
| Male | 28.5 ± 1.3 | 27.6 ± 1.1 | 0.59 | |
| Female | 39.2 ± 1.1 | 38.1 ± 1.0 | 0.49 | |
| Visceral fat area (cm2) | 82.3 ± 5.7 | 82.5 ± 5.3 | 0.99 | |
| Male | 92.9 ± 8.2 | 90.5 ± 7.1 | 0.83 | |
| Female | 73.4 ± 7.2 | 74.4 ± 7.3 | 0.92 | |
| Systolic blood pressure (mmHg) | 122.8 ± 1.9 | 125.5 ± 2.4 | 0.38 | |
| Male | 125.5 ± 3.3 | 125.3 ± 3.6 | 0.97 | |
| Female | 120.5 ± 7.8 | 125.6 ± 3.2 | 0.20 | |
| Diastolic blood pressure (mmHg) | 79.1 ± 1.8 | 81.7 ± 2.0 | 0.35 | |
| Male | 81.0 ± 2.7 | 81.7 ± 2.9 | 0.87 | |
| Female | 77.5 ± 2.5 | 81.7 ± 2.9 | 0.29 |
| Week | 0 | 8 | 8-0 W | p (8-0 W) † | |
|---|---|---|---|---|---|
| Energy (kcal) | Placebo | 1922 ± 70 | 1827 ± 87 | −95 ± 52 | 0.22 |
| Test | 1900 ± 64 | 1945 ± 121 | 45 ± 99 | ||
| Protein (g) | Placebo | 73.4 ± 2.6 | 70.1 ± 3.8 | −3.1 ± 2.8 | 0.44 |
| Test | 75.9 ± 3.2 | 76.4 ± 4.4 | 0.5 ± 3.8 | ||
| Fat (g) | Placebo | 65.6 ± 3.4 | 60.4 ± 3.8 | −5.1 ± 2.9 | 0.69 |
| Test | 67.2 ± 3.7 | 64.0 ± 5.5 | −3.1 ± 3.9 | ||
| Carbohydrate (g) | Placebo | 243.8 ± 11.4 | 233.7 ± 12.9 | −10.1 ± 9.1 | 0.15 |
| Test | 236.5 ± 10.8 | 254.2 ± 19.4 | 17.7 ± 16.5 | ||
| Cholesterol (mg) | Placebo | 373.3 ± 23.4 | 345.1 ± 22.0 | −27.1 ± 16.6 | 0.37 |
| Test | 362.4 ± 24.0 | 363.0 ± 28.6 | 0.7 ± 26.1 | ||
| Total dietary fiber (g) | Placebo | 13.6 ± 1.5 | 11.6 ± 0.9 | 1.1 ± 3.3 | 0.84 |
| Test | 13.6 ± 1.2 | 12.6 ± 1.1 | −0.2 ± 5.2 | ||
| Calcium (mg) | Placebo | 484.8 ± 42.9 | 495.5 ± 63.8 | 10.7 ± 42.8 | 0.99 |
| Test | 523.3 ± 35.8 | 531.9 ± 56.3 | 10.1 ± 66.9 | ||
| Iodine (μg) | Placebo | 548.1 ± 123.4 | 686.9 ± 176.3 | 138.8 ± 107.6 | 0.52 |
| Test | 590.7 ± 150.5 | 836.6 ± 144.3 | 254.9 ± 123.6 |
| Placebo (n = 24) | 8-0 W | Test (n = 24) | 8-0 W | p (8-0 W) † | ||||
|---|---|---|---|---|---|---|---|---|
| Week | 0 | 8 | 0 | 8 | ||||
| Total cholesterol (mmol/L) | Overall | 5.73 ± 0.19 | 5.61 ± 0.20 | −0.12 ± 0.09 | 5.71 ± 0.19 | 5.59 ± 0.22 | −0.12 ± 0.09 | 0.987 |
| HL | 6.29 ± 0.25 | 5.96 ± 0.27 | −0.33 ± 0.11 | 6.33 ± 0.20 | 6.26 ± 0.28 | −0.06 ± 0.13 | 0.125 | |
| Non-HL | 5.07 ± 0.11 | 5.20 ± 0.17 | 0.12 ± 0.11 | 4.98 ± 0.14 | 4.79 ± 0.09 | −0.19 ± 0.14 | 0.111 | |
| LDL cholesterol (mmol/L) | Overall | 3.54 ± 0.16 | 3.45 ± 0.17 | −0.09 ± 0.11 | 3.53 ± 0.18 | 3.38 ± 0.20 | −0.15 ± 0.11 | 0.689 |
| HL | 3.98 ± 0.21 | 3.65 ± 0.28 | −0.33 ± 0.14 | 4.08 ± 0.21 | 3.94 ± 0.28 | −0.14 ± 0.17 | 0.394 | |
| Non-HL | 3.02 ± 0.12 | 3.21 ± 0.15 | 0.19 ± 0.12 | 2.88 ± 0.12 | 2.72 ± 0.10 * | −0.17 ± 0.14 | 0.063 | |
| HDL cholesterol (mmol/L) | Overall | 1.49 ± 0.07 | 1.42 ± 0.07 | −0.07 ± 0.02 | 1.45 ± 0.07 | 1.42 ± 0.07 | −0.02 ± 0.02 | 0.154 |
| HL | 1.45 ± 0.11 | 1.40 ± 0.11 | −0.05 ± 0.03 | 1.31 ± 0.08 | 1.28 ± 0.08 | −0.03 ± 0.03 | 0.675 | |
| Non-HL | 1.53 ± 0.09 | 1.44 ± 0.08 | −0.09 ± 0.03 | 1.60 ± 0.11 | 1.59 ± 0.11 | −0.01 ± 0.04 | 0.125 | |
| Triglyceride (mmol/L) | Overall | 1.58 ± 0.14 | 1.61 ± 0.23 | 0.03 ± 0.21 | 1.48 ± 0.23 | 1.66 ± 0.52 | 0.18 ± 0.45 | 0.754 |
| HL | 2.02 ± 0.17 | 2.15 ± 0.40 | 0.13 ± 0.40 | 1.99 ± 0.36 | 2.34 ± 0.93 | 0.36 ± 0.83 | 0.811 | |
| Non-HL | 1.06 ± 0.09 | 0.96 ± 0.10 | −0.01 ± 0.10 | 0.88 ± 0.09 | 0.86 ± 0.07 | −0.02 ± 0.10 | 0.557 | |
| Placebo (n = 24) | 8-0 W | Test (n = 24) | 8-0 W | p (8-0 W) † | |||
|---|---|---|---|---|---|---|---|
| Week | 0 | 8 | 0 | 8 | |||
| TSH (mIU/L) | 1.43 ± 0.15 | 1.43 ± 0.58 | −0.01 ± 0.08 | 1.65 ± 0.39 | 1.65 ± 0.99 | 0.01 ± 0.24 | 0.653 |
| FT3 (pmol/L) | 4.24 ± 0.08 | 4.06 ± 0.10 | −0.18 ± 0.07 | 4.11 ± 0.10 | 4.05 ± 0.11 | −0.06 ± 0.09 | 0.294 |
| FT4 (pmol/L) | 0.12 ± 0.00 | 0.13 ± 0.00 | 0.01 ± 0.00 | 0.12 ± 0.00 | 0.13 ± 0.00 | 0.01 ± 0.00 | 0.826 |
| Placebo | Test | |
|---|---|---|
| Boiled kelp powder | 0 | 39.3 |
| Maltitol | 47.5 | 24.2 |
| Microcrystalline cellulose | 47.5 | 31.5 |
| Silicon dioxide | 1.5 | 2 |
| Tricalcium phosphate | 0 | 2 |
| Calcium stearate | 1.1 | 1 |
| Coloring | 2.1 | 0 |
| Sodium glutamate | 0.2 | 0 |
| Kelp flavor powder | 0.1 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aoe, S.; Yamanaka, C.; Ohtoshi, H.; Nakamura, F.; Fujiwara, S. Effects of Daily Kelp (Laminaria japonica) Intake on Body Composition, Serum Lipid Levels, and Thyroid Hormone Levels in Healthy Japanese Adults: A Randomized, Double-Blind Study. Mar. Drugs 2021, 19, 352. https://doi.org/10.3390/md19070352
Aoe S, Yamanaka C, Ohtoshi H, Nakamura F, Fujiwara S. Effects of Daily Kelp (Laminaria japonica) Intake on Body Composition, Serum Lipid Levels, and Thyroid Hormone Levels in Healthy Japanese Adults: A Randomized, Double-Blind Study. Marine Drugs. 2021; 19(7):352. https://doi.org/10.3390/md19070352
Chicago/Turabian StyleAoe, Seiichiro, Chiemi Yamanaka, Hirofumi Ohtoshi, Fumiko Nakamura, and Suguru Fujiwara. 2021. "Effects of Daily Kelp (Laminaria japonica) Intake on Body Composition, Serum Lipid Levels, and Thyroid Hormone Levels in Healthy Japanese Adults: A Randomized, Double-Blind Study" Marine Drugs 19, no. 7: 352. https://doi.org/10.3390/md19070352
APA StyleAoe, S., Yamanaka, C., Ohtoshi, H., Nakamura, F., & Fujiwara, S. (2021). Effects of Daily Kelp (Laminaria japonica) Intake on Body Composition, Serum Lipid Levels, and Thyroid Hormone Levels in Healthy Japanese Adults: A Randomized, Double-Blind Study. Marine Drugs, 19(7), 352. https://doi.org/10.3390/md19070352







