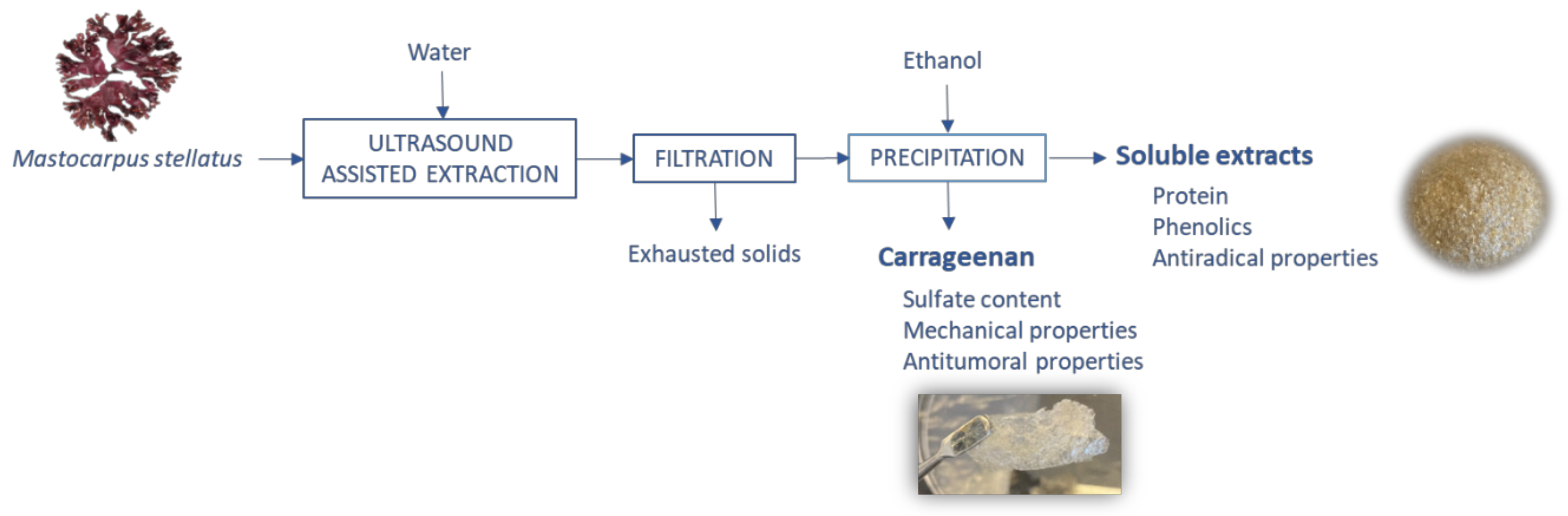
Ultrasound-Assisted Water Extraction of Mastocarpus stellatus Carrageenan with Adequate Mechanical and Antiproliferative Properties
Abstract
1. Introduction
2. Results and Discussion
2.1. Optimization of the Ultrasound-Assisted Water Extraction
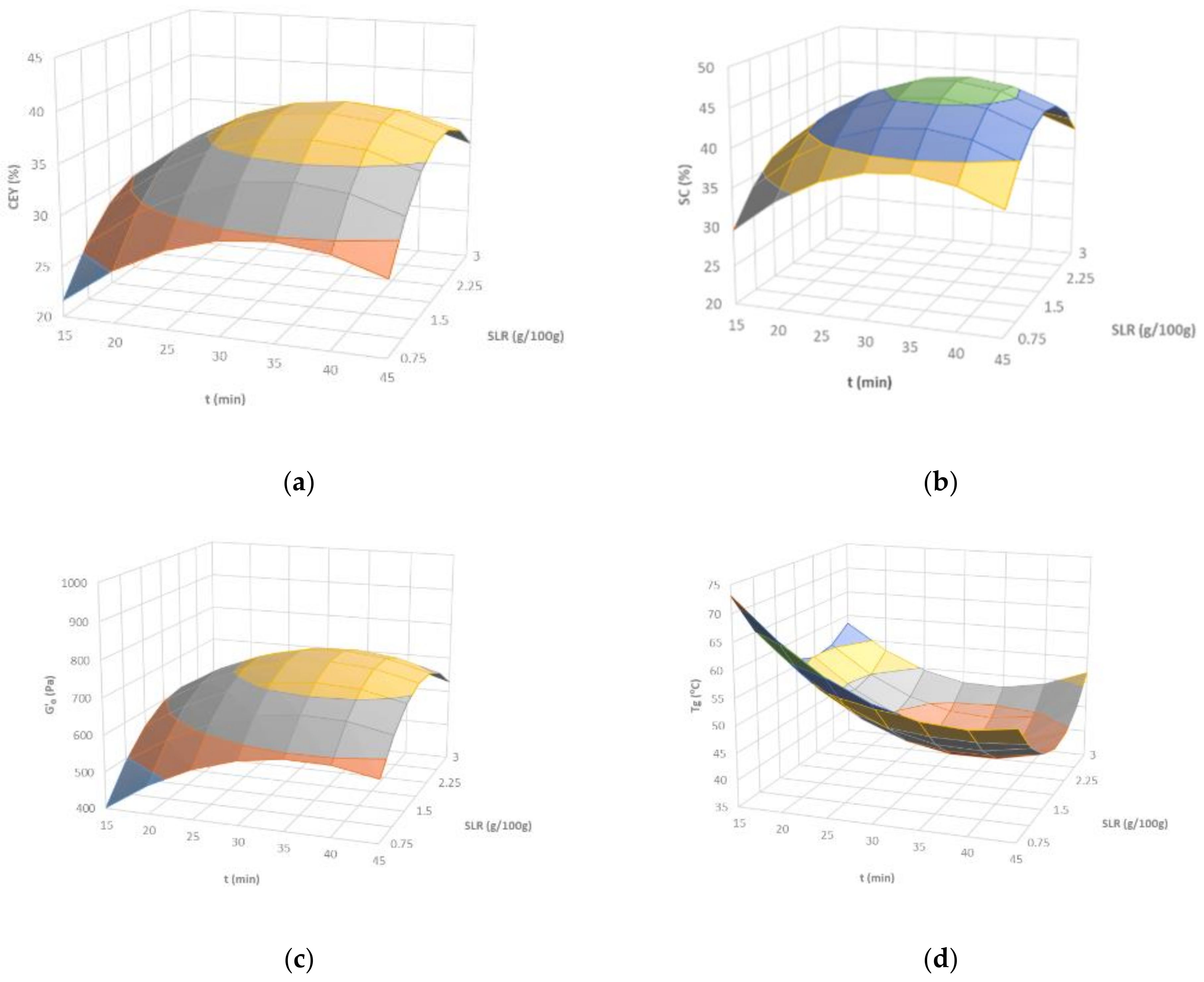
2.1.1. Carrageenan Yield and Properties
Carrageenan Extraction Yield (CEY)
Sulfate Content (SC)
Thermo-Rheological Features for the Biopolymer-Based Hydrogel Matrices
FTIR-ATR
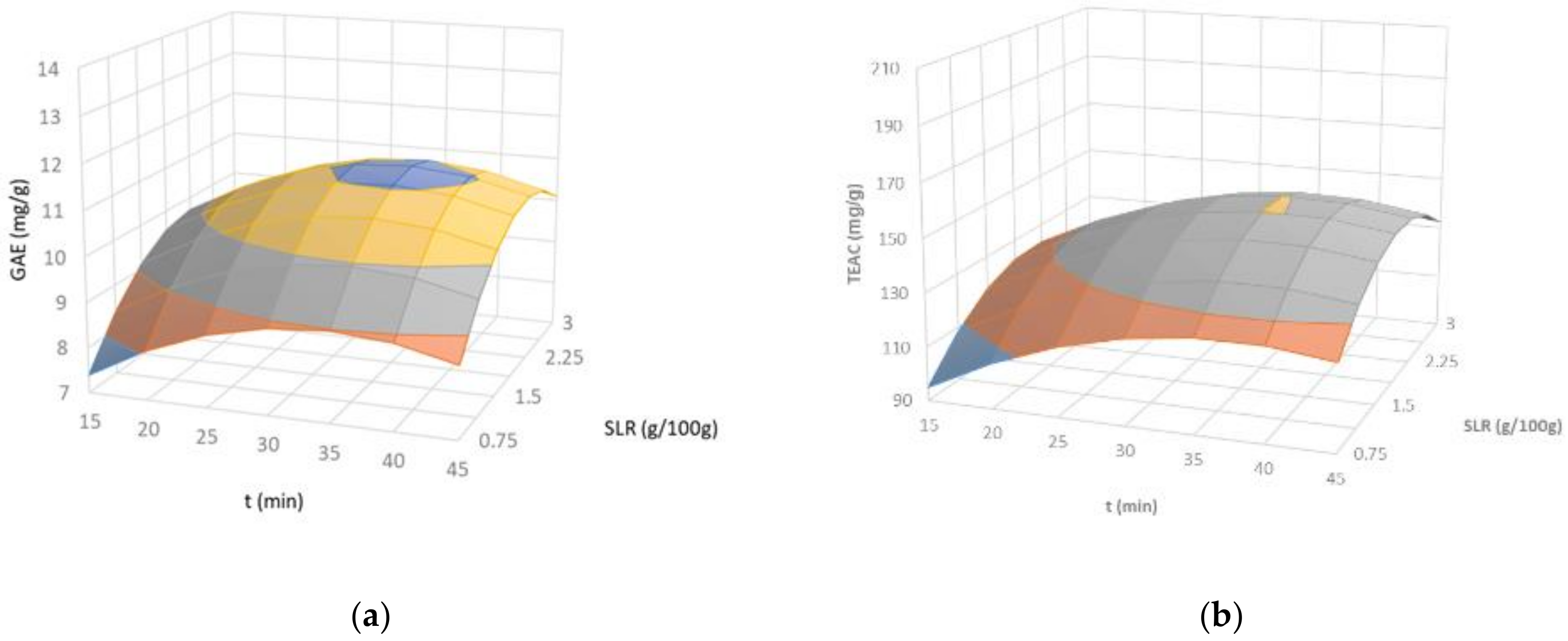
2.1.2. Composition and Antiradical Properties in the Soluble Extracts
Protein Content (PC)
Total Phenolic Content (TPC) and Antiradical Properties (TEAC)
2.2. Growth Inhibition of Human Cancer Cells
3. Materials and Methods
3.1. Materials
3.2. Ultrasound-Assisted Extraction
3.3. Analytical Methods
3.3.1. Extraction Yield
3.3.2. Soluble Sulphate Content
3.3.3. Protein Content
3.3.4. Total Phenolic Content
3.3.5. Trolox Equivalent Antioxidant Capacity
3.3.6. FTIR-ATR
Thermo-Rheological Testing
3.4. Antiproliferative Activity
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Azevedo, G.; Torres, M.D.; Sousa-Pinto, I.; Hilliou, L. Effect of pre-extraction alkali treatment on the chemical structure and gelling properties of extracted hybrid carrageenan from Chondrus crispus and Ahnfeltiopsis devoniensis. Food Hydrocoll. 2015, 50, 150–158. [Google Scholar] [CrossRef]
- Van De Velde, F.; Peppelman, H.A.; Rollema, H.S.; Tromp, R.H. On the structure of κ/ι-hybrid carrageenans. Carbohydr. Res. 2001, 331, 271–283. [Google Scholar] [CrossRef]
- Cotas, J.; Marques, V.; Afonso, M.B.; Rodrigues, C.M.P.; Pereira, L. Antitumour potential of Gigartina pistillata carrageenans against colorectal cancer stem cell-enriched tumourspheres. Mar. Drugs 2020, 18, 50. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Shao, B.; Nie, W.; Wei, X.W.; Li, Y.L.; Wang, B.L.; He, Z.Y.; Liang, X.; Ye, T.H.; Wei, Y.Q. Antitumor and adjuvant activity of λ-carrageenan by stimulating immune response in cancer immunotherapy. Sci. Rep. 2015, 5, 1–12. [Google Scholar]
- Calvo, G.H.; Cosenza, V.A.; Sáenz, D.A.; Navarro, D.A.; Stortz, C.A.; Céspedes, M.A.; Mamone, L.A.; Casas, A.G.; Di Venosa, G.M. Disaccharides obtained from carrageenans as potential antitumor agents. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Tecson, M.G.; Abad, L.V.; Ebajo, V.D.; Camacho, D.H. Ultrasound-assisted depolymerization of kappa-carrageenan and characterization of degradation product. Ultrason. Sonochem. 2021, 73, 105540. [Google Scholar] [CrossRef] [PubMed]
- Khotimchenko, M.; Tiasto, V.; Kalitnik, A.; Begun, M.; Khotimchenko, R.; Leonteva, E.; Bryukhovetskiy, I.; Khotimchenko, Y. Antitumor potential of carrageenans from marine red algae. Carbohydr. Polym. 2020, 246, 116568. [Google Scholar] [CrossRef]
- Prasetyaningrum, A.; Praptyana, I.R.; Nurfiningsih; Ratnawati. Carrageenan: Nutraceutical and functional food as future food. IOP Conf. Ser. Earth Environ. Sci. 2019, 292, 012068. [Google Scholar] [CrossRef]
- Cicinskas, E.; Begun, M.A.; Tiasto, V.A.; Belousov, A.S.; Vikhareva, V.V.; Mikhailova, V.A.; Kalitnik, A.A. In vitro antitumor and immunotropic activity of carrageenans from red algae Chondrus armatus and their low-molecular weight degradation products. J. Biomed. Mater. Res. Part A 2020, 108, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Groult, H.; Cousin, R.; Chot-Plassot, C.; Maura, M.; Bridiau, N.; Piot, J.M.; Maugard, T.; Fruitier-Arnaudin, I. Λ-carrageenan oligosaccharides of distinct anti-heparanase and anticoagulant activities inhibit MDA-MB-231 breast cancer cell migration. Mar. Drugs 2019, 17, 140. [Google Scholar] [CrossRef]
- Ghannam, A.; Murad, H.; Jazzara, M.; Odeh, A.; Allaf, A.W. Isolation, Structural characterization, and antiproliferative activity of phycocolloids from the red seaweed Laurencia papillosa on MCF-7 human breast cancer cells. Int. J. Biol. Macromol. 2018, 108, 916–926. [Google Scholar] [CrossRef] [PubMed]
- Abdul Khalil, H.P.S.; Lai, T.K.; Tye, Y.Y.; Rizal, S.; Chong, E.W.N.; Yap, S.W.; Hamzah, A.A.; Nurul Fazita, M.R.; Paridah, M.T. A review of extractions of seaweed hydrocolloids: Properties and applications. Express Polym. Lett. 2018, 12, 296–317. [Google Scholar] [CrossRef]
- González-Ballesteros, N.; Torres, M.D.; Flórez-Fernández, N.; Diego-González, L.; Simón-Vázquez, R.; Rodríguez-Argüelles, M.C.; Domínguez, H. Eco-friendly extraction of Mastocarpus stellatus carrageenan for the synthesis of gold nanoparticles with improved biological activity. Int. J. Biol. Macromol. 2021, in press. [Google Scholar]
- Gereniu, C.R.N.; Saravana, P.S.; Chun, B.S. Recovery of carrageenan from Solomon Islands red seaweed using ionic liquid-assisted subcritical water extraction. Sep. Purif. Technol. 2018, 196, 309–317. [Google Scholar] [CrossRef]
- Ponthier, E.; Domínguez, H.; Torres, M.D. The microwave assisted extraction sway on the features of antioxidant compounds and gelling biopolymers from Mastocarpus stellatus. Algal Res. 2020, 51, 102081. [Google Scholar] [CrossRef]
- Youssouf, L.; Lallemand, L.; Giraud, P.; Soulé, F.; Bhaw-Luximon, A.; Meilhac, O.; D’Hellencourt, C.L.; Jhurry, D.; Couprie, J. Ultrasound-assisted extraction and structural characterization by NMR of alginates and carrageenans from seaweeds. Carbohydr. Polym. 2017, 166, 55–63. [Google Scholar] [CrossRef]
- Tasende, M.G.; Cid, M.; Fraga, M.I. Qualitative and quantitative analysis of carrageenan content in gametophytes of Mastocarpus stellatus (Stackhouse) Guiry along Galician coast (NW Spain). J. Appl. Phycol. 2013, 25, 587–596. [Google Scholar] [CrossRef]
- Blanco-Pascual, N.; Gómez-Guillén, M.C.; Montero, M.P. Integral Mastocarpus stellatus use for antioxidant edible film development. Food Hydrocoll. 2014, 40, 128–137. [Google Scholar] [CrossRef]
- Alemán, A.; Blanco-Pascual, N.; Montero, M.P.; Gómez-Guillén, M.C. Simple and efficient hydrolysis procedure for full utilization of the seaweed Mastocarpus stellatus to produce antioxidant films. Food Hydrocoll. 2016, 56, 277–284. [Google Scholar] [CrossRef]
- Barral-Martínez, M.; Flórez-Fernández, N.; Domínguez, H.; Torres, M.D. Tailoring hybrid carrageenans from Mastocarpus stellatus red seaweed using microwave hydrodiffusion and gravity. Carbohydr. Polym. 2020, 248, 116830. [Google Scholar] [CrossRef]
- Blanco-Pascual, N.; Alemán, A.; Gómez-Guillén, M.C.; Montero, M.P. Enzyme-assisted extraction of κ/ι-hybrid carrageenan from Mastocarpus stellatus for obtaining bioactive ingredients and their application for edible active film development. Food Funct. 2014, 5, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Azizi, R.; Farahnaky, A. Ultrasound assisted-viscosifying of kappa carrageenan without heating. Food Hydrocoll. 2016, 61, 85–91. [Google Scholar] [CrossRef]
- Cui, R.; Zhu, F. Ultrasound modified polysaccharides: A review of structure, physicochemical properties, biological activities and food applications. Trends Food Sci. Technol. 2021, 107, 491–508. [Google Scholar] [CrossRef]
- Shirsath, S.R.; Sonawane, S.H.; Gogate, P.R. Intensification of extraction of natural products using ultrasonic irradiations—A review of current status. Chem. Eng. Process. Process Intensif. 2012, 53, 10–23. [Google Scholar] [CrossRef]
- Zendeboodi, F.; Farahnaky, A.; Gholian, M.M. Structural changes and stress relaxation behavior of κ-carrageenan cold-processed gels: Effects of ultrasonication time and power. J. Texture Stud. 2019, 50, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Bixler, H.J.; Porse, H. A decade of change in the seaweed hydrocolloids industry. J. Appl. Phycol. 2011, 23, 321–335. [Google Scholar] [CrossRef]
- Geonzon, L.C.; Zhuang, X.; Santoya, A.M.; Bacabac, R.G.; Xie, J.; Matsukawa, S. Gelation mechanism and network structure of mixed kappa carrageenan/lambda carrageenan gels studied by macroscopic and microscopic observation methods. Food Hydrocoll. 2020, 105, 105759. [Google Scholar] [CrossRef]
- Hilliou, L. Chapter Two—Hybrid Carrageenans: Isolation, Chemical Structure, and Gel Properties. In Marine Carbohydrates: Fundamentals and Applications, Part A; Kim, S.-K., Ed.; Academic Press: Cambridge, MA, USA, 2014; Volumn 72, pp. 17–43. [Google Scholar]
- Torres, M.D.; Chenlo, F.; Moreira, R. Thermal reversibility of kappa/iota-hybrid carrageenan gels extracted from Mastocarpus stellatus at different ionic strengths. J. Taiwan Inst. Chem. Eng. 2017, 71, 414–420. [Google Scholar] [CrossRef]
- Souza, H.K.S.; Hilliou, L.; Bastos, M.; Gonalves, M.P. Effect of molecular weight and chemical structure on thermal and rheological properties of gelling κ/ι-hybrid carrageenan solutions. Carbohydr. Polym. 2011, 85, 429–438. [Google Scholar] [CrossRef]
- Pereira, L.; Amado, A.M.; Critchley, A.T.; van de Velde, F.; Ribeiro-Claro, P.J.A. Identification of selected seaweed polysaccharides (phycocolloids) by vibrational spectroscopy (FTIR-ATR and FT-Raman). Food Hydrocoll. 2009, 23, 1903–1909. [Google Scholar] [CrossRef]
- Pereira, L.; Van De Velde, F. Portuguese carrageenophytes: Carrageenan composition and geographic distribution of eight species (Gigartinales, Rhodophyta). Carbohydr. Polym. 2011, 84, 614–623. [Google Scholar] [CrossRef]
- Veith, P.D.; Reynolds, E.C. Production of a high gel strength whey protein concentrate from cheese whey. J. Dairy Sci. 2004, 87, 831–840. [Google Scholar] [CrossRef]
- Pereira, L. A Review of the Nutrient Composition of Selected Edible Seaweeds; Pomin, V.H., Ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2011; ISBN 978-161470878-0. [Google Scholar]
- Gómez-Ordóñez, E.; Jiménez-Escrig, A.; Rupérez, P. Bioactivity of sulfated polysaccharides from the edible red seaweed Mastocarpus stellatus. Bioact. Carbohydrates Diet. Fibre 2014, 3, 29–40. [Google Scholar] [CrossRef]
- Uribe, E.; Vega-Gálvez, A.; García, V.; Pastén, A.; Rodríguez, K.; López, J.; Scala, K. Di Evaluation of physicochemical composition and bioactivity of a red seaweed (Pyropia orbicularis) as affected by different drying technologies. Dry. Technol. 2020, 38, 1218–1230. [Google Scholar] [CrossRef]
- Souza, R.B.; Frota, A.F.; Silva, J.; Alves, C.; Neugebauer, A.Z.; Pinteus, S.; Rodrigues, J.A.G.; Cordeiro, E.M.S.; de Almeida, R.R.; Pedrosa, R.; et al. In vitro activities of kappa-carrageenan isolated from red marine alga Hypnea musciformis: Antimicrobial, anticancer and neuroprotective potential. Int. J. Biol. Macromol. 2018, 112, 1248–1256. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Song, J. Preparation, structural characterization and in vitro antitumor activity of kappa-carrageenan oligosaccharide fraction from Kappaphycus striatum. J. Appl. Phycol. 2005, 17, 7–13. [Google Scholar] [CrossRef]
- Ariffin, S.H.Z.; Yeen, W.W.; Abidin, I.Z.Z.; Wahab, R.M.A.; Ariffin, Z.Z.; Senafi, S. Cytotoxicity effect of degraded and undegraded kappa and iota carrageenan in human intestine and liver cell lines. BMC Complement. Altern. Med. 2014, 14, 1–16. [Google Scholar]
- Suganya, A.M.; Sanjivkumar, M.; Chandran, M.N.; Palavesam, A.; Immanuel, G. Pharmacological importance of sulphated polysaccharide carrageenan from red seaweed Kappaphycus alvarezii in comparison with commercial carrageenan. Biomed. Pharmacother. 2016, 84, 1300–1312. [Google Scholar] [CrossRef] [PubMed]
- Dodgson, K.S. Determination of inorganic sulphate in studies on the enzymic and non-enzymic hydrolysis of carbohydrate and other sulphate esters. Biochem. J. 1961, 78, 312–319. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Koivikko, R.; Loponen, J.; Honkanen, T.; Jormalainen, V. Contents of solubre, cell-wall-bound and exuded phlorotannins in the brown alga Fucus vesiculosus, with implications on their ecological functions. J. Chem. Ecol. 2005, 31, 195–212. [Google Scholar] [CrossRef] [PubMed]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Azevedo, G.; Bernardo, G.; Hilliou, L. NaCl and KCl phase diagrams of kappa/iota-hybrid carrageenans extracted from Mastocarpus stellatus. Food Hydrocoll. 2014, 37, 116–123. [Google Scholar] [CrossRef]


| Exp. | Variables (Coded Levels) | Response Functions | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SLR, g/100 g (X1) | A, % (X2) | t, min (X3) | CEY, % | SC, % | G′0 (1 Hz), Pa | Tgel, °C | PC, % | TPC, mg GAE/g | TEAC, mg/g | |
| 1 | 0.75 (−1) | 75 (0) | 15 (−1) | 23.7 | 29.4 | 392 | 72 | 14.1 | 7.5 | 101 |
| 2 | 0.75 (−1) | 75 (0) | 45 (1) | 30.1 | 35.8 | 578 | 52 | 17.7 | 9.7 | 134 |
| 3 | 3.00 (1) | 75 (0) | 15 (−1) | 26.9 | 30.5 | 498 | 59 | 15.9 | 8.2 | 114 |
| 4 | 3.00 (1) | 75 (0) | 45 (1) | 34.1 | 37.9 | 604 | 50 | 18.1 | 10.1 | 141 |
| 5 | 1.88 (0) | 50 (−1) | 15 (−1) | 28.1 | 36.8 | 525 | 58 | 16.3 | 8.6 | 120 |
| 6 | 1.88 (0) | 50 (−1) | 45 (1) | 35.2 | 44.7 | 650 | 47 | 19.1 | 10.7 | 149 |
| 7 | 1.88 (0) | 100 (1) | 15 (−1) | 30.3 | 29.6 | 570 | 53 | 17.5 | 9.4 | 131 |
| 8 | 1.88 (0) | 100 (1) | 45 (1) | 36.1 | 34.3 | 672 | 45 | 19.3 | 10.8 | 150 |
| 9 | 0.75 (−1) | 50 (−1) | 30 (0) | 27.5 | 38.6 | 481 | 59 | 15.8 | 8.1 | 112 |
| 10 | 3.00 (1) | 50 (−1) | 30 (0) | 33.4 | 41.2 | 623 | 49 | 18.4 | 10.2 | 143 |
| 11 | 0.75 (−1) | 100 (1) | 30 (0) | 29.2 | 30.3 | 543 | 56 | 16.7 | 8.9 | 125 |
| 12 | 3.00 (1) | 100 (1) | 30 (0) | 34.2 | 31.5 | 654 | 47 | 18.9 | 10.6 | 146 |
| 13 | 1.88 (0) | 75 (0) | 30 (0) | 39.5 | 46.2 | 727 | 41 | 20.0 | 11.1 | 155 |
| 14 | 1.88 (0) | 75 (0) | 30 (0) | 39.7 | 46.3 | 725 | 42 | 20.1 | 11.2 | 155 |
| 15 | 1.88 (0) | 75 (0) | 30 (0) | 39.6 | 46.1 | 725 | 41 | 20.1 | 11.0 | 156 |
| Coefficients | CEY, % | SC, % | G′o (1 Hz), Pa | Tgel, °C | PC, mg/g | GAE, mg/g | TEAC, mg/g |
|---|---|---|---|---|---|---|---|
| a0 | −32.3 a | −29.8 a | −663.4 a | 156.9 a | −4.75 a | −3.84 a | −74.4 a |
| a1 | 20.3 a | 21.6 a | 454.9 a | −38.1 a | 7.91 a | 4.94 a | 72.3 a |
| a2 | 0.63 | 0.8 | 9.70 a | −0.68 a | 0.17 | 0.097 | 1.53 a |
| a3 | 1.52 a | 1.9 a | 31.0 a | −2.88 a | 0.56 a | 0.32 a | 5.03 a |
| a4 | −0.001 | −0.002 | −0.015 | 0.002 | −0.001 | −0.0005 | −0.007 |
| a5 | −0.008 | −0.009 | −0.28 | 0.009 | −0.004 | −0.004 | −0.089 |
| a6 | 0.012 | 0.014 | −1.33 a | 0.18 a | −0.028 a | −0.004 | −0.089 |
| a7 | −4.80 a | −5.50 a | −92.3 a | 7.51 a | −1.59 a | −1.01 a | −15.2 a |
| a8 | −0.004 | −0.006 | −0.05 a | 0.004 | −0.001 | −0.0004 | −0.007 |
| a9 | −0.021 a | −0.026 a | −0.39 a | 0.033 a | −0.006 a | −0.004 a | −0.059 a |
| F | 15.6 | 6.1 | 12.1 | 8.5 | 5.2 | 4.3 | 3.9 |
| R2 | 98.5 | 95.8 | 98.4 | 96.5 | 96.1 | 95.3 | 94.9 |
| Seaweed | Extraction, Degradation Conditions SOLVENT, SLR, Temp, Time | Antiproliferative Activity Cell Line, IC50 (μg/mL) | Reference |
|---|---|---|---|
| κ-carrageenan | Commercial, treated with NaOH and KCl, SLR 1 g/100 mL + 0.1 M HCl, 60 °C, 4 h | HeLa, 500 | [38] |
| BGC, 500 | |||
| Eucheuma denticulatum κ-carrageenan | Commercial, degraded 0.1 M HCl, 60 °C, 4 h | Caco-2, 280 | [39] |
| HepG2, 450 | |||
| Eucheuma denticulatum ι-carrageenan | Commercial, degraded 0.1 M HCl, 60 °C, 4 h | Caco-2, 1000 | [39] |
| Kappaphycus alvarezii κ-carrageenan | Distilled water SLR 1 g/35 mL, 2 h + boiling, 30 min + NaOH, boiling, 4 h | HT-29, 73.87 | [40] |
| HepG2 56.71 | |||
| MG63, 47.85 | |||
| κ-carrageenan | Commercial | HT-29, 123.8 Hep G2, 125.0 | [40] |
| MG63, 55.48 | |||
| Laurencia papillosa κ-, ι-, λ-carrageenan | Water, 90 °C, 12 h, solvent purification | MCF-7, 200, 50, 25 µM | [11] |
| Eucheuma spinosum ι-carrageenan | Commercial | LM2, 200 | [5] |
| Hypnea muscifomis κ-carrageenan | Water, room temp., 24 h + Water, 90 °C, 8 h 0.1 M trifluoroacetic acid, 80 °C, 0.5–3 h | LM2, 100–250 | [5] |
| Iridaea undulosa κ/ι-hybrid carrageenan | Water, SLR 2 g/100 mL, room temp., 16 h, alkaline 0.1 M trifluoroacetic acid, 80 °C, 0.5–3 h | LM2, 150–200 | [5] |
| Gigartina pistillata λ/ξ hybrid carrageenan | Acetone:methanol (1:1), 16 h, 4 °C, 1 M NaOH, SLR: 1 g/150 mL, 85–90 °C, 3 h | HT-29, 0.705 | [3] |
| Gigartina pistillata κ/ι hybrid carrageenan | Acetone:methanol (1:1), 16 h, 4 °C, 1 M NaOH, SLR: 1 g/150 mL, 85–90 °C, 3 h | HT-29, 0.657 | [3] |
| Mastocarpus stellatus κ/ι hybrid carrageenan | UAE (80 kHz, 79% amplitude): Water, 2.1 g/100 g, 80 °C, 36.5 min | A-549, 7.5 | This work |
| A-2780, 7.2 | This work | ||
| HeLa-229, 51.9 | This work | ||
| HT-29, 36.5 | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres, M.D.; Flórez-Fernández, N.; Dominguez, H. Ultrasound-Assisted Water Extraction of Mastocarpus stellatus Carrageenan with Adequate Mechanical and Antiproliferative Properties. Mar. Drugs 2021, 19, 280. https://doi.org/10.3390/md19050280
Torres MD, Flórez-Fernández N, Dominguez H. Ultrasound-Assisted Water Extraction of Mastocarpus stellatus Carrageenan with Adequate Mechanical and Antiproliferative Properties. Marine Drugs. 2021; 19(5):280. https://doi.org/10.3390/md19050280
Chicago/Turabian StyleTorres, Maria Dolores, Noelia Flórez-Fernández, and Herminia Dominguez. 2021. "Ultrasound-Assisted Water Extraction of Mastocarpus stellatus Carrageenan with Adequate Mechanical and Antiproliferative Properties" Marine Drugs 19, no. 5: 280. https://doi.org/10.3390/md19050280
APA StyleTorres, M. D., Flórez-Fernández, N., & Dominguez, H. (2021). Ultrasound-Assisted Water Extraction of Mastocarpus stellatus Carrageenan with Adequate Mechanical and Antiproliferative Properties. Marine Drugs, 19(5), 280. https://doi.org/10.3390/md19050280







