Oral Bioavailability of Omega-3 Fatty Acids and Carotenoids from the Microalgae Phaeodactylum tricornutum in Healthy Young Adults
Abstract
1. Introduction
2. Results
2.1. Adverse Effects during the Intervention
2.2. Laboratory Parameters
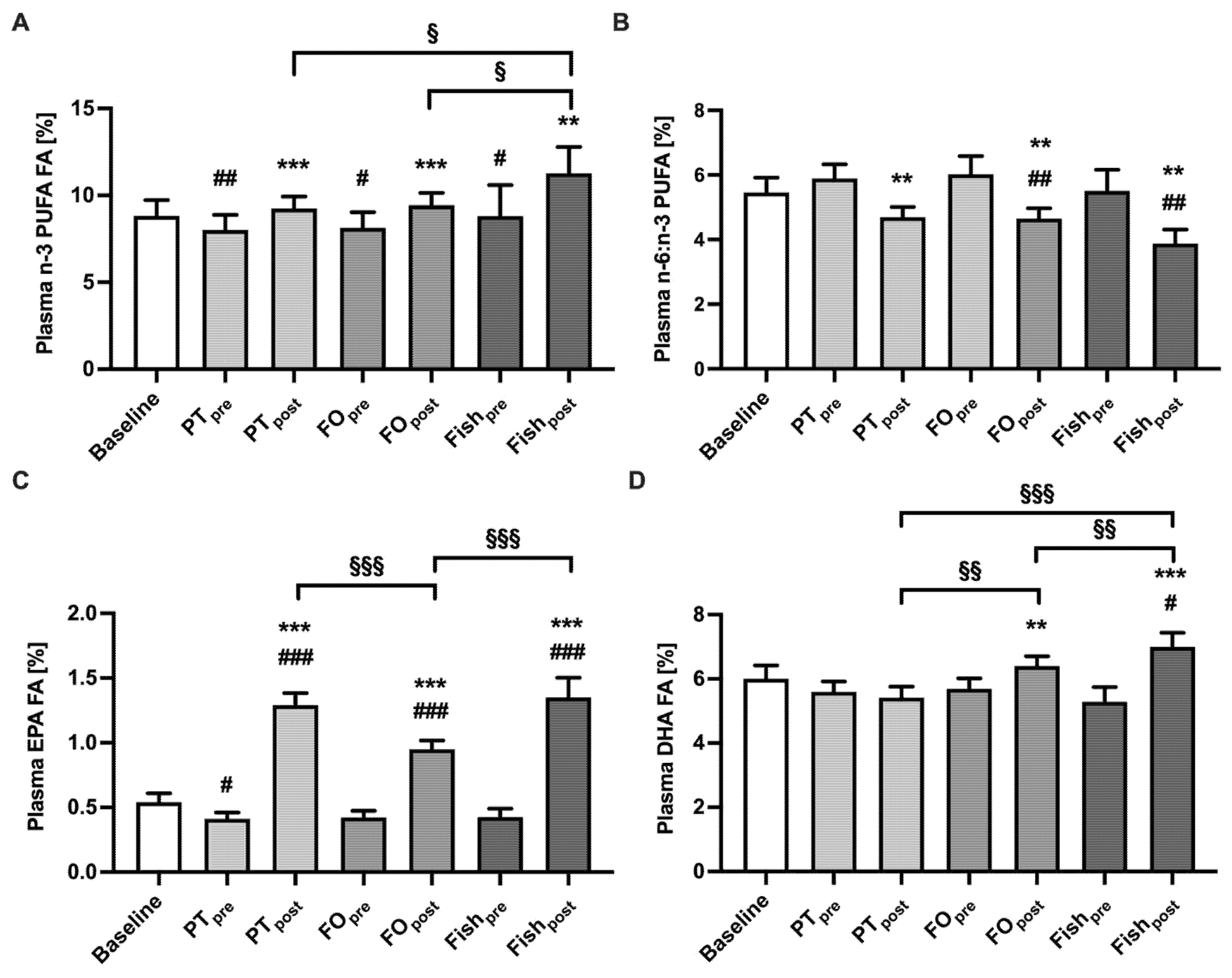
2.3. Change of Plasma Fatty Acids upon Intervention
2.4. Carotenoids and Tocopherol Concentrations before and after Intervention with PT
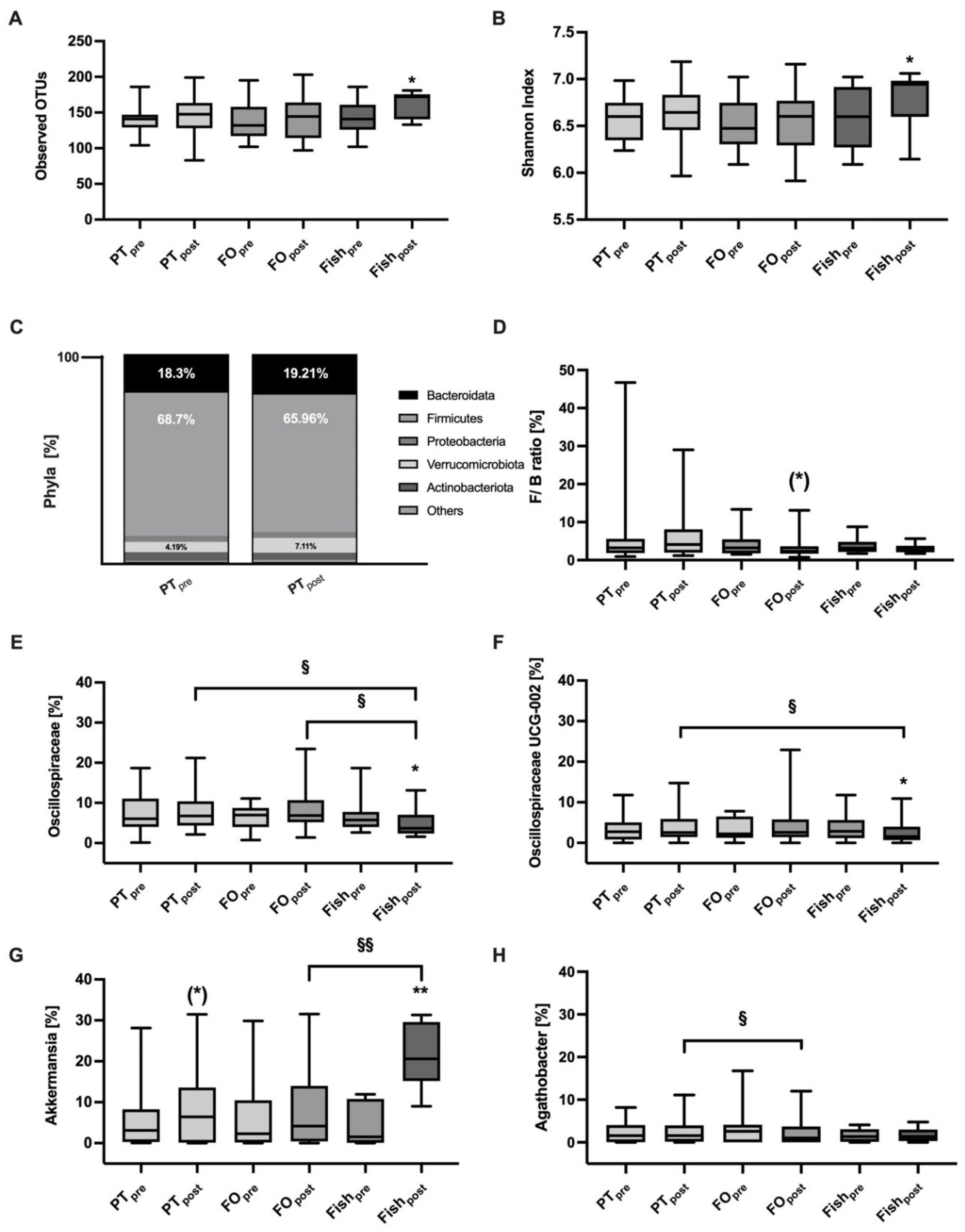
2.5. Gut Barrier Function and Gut Microbiome
3. Discussion
4. Materials and Methods
4.1. Participant Selection
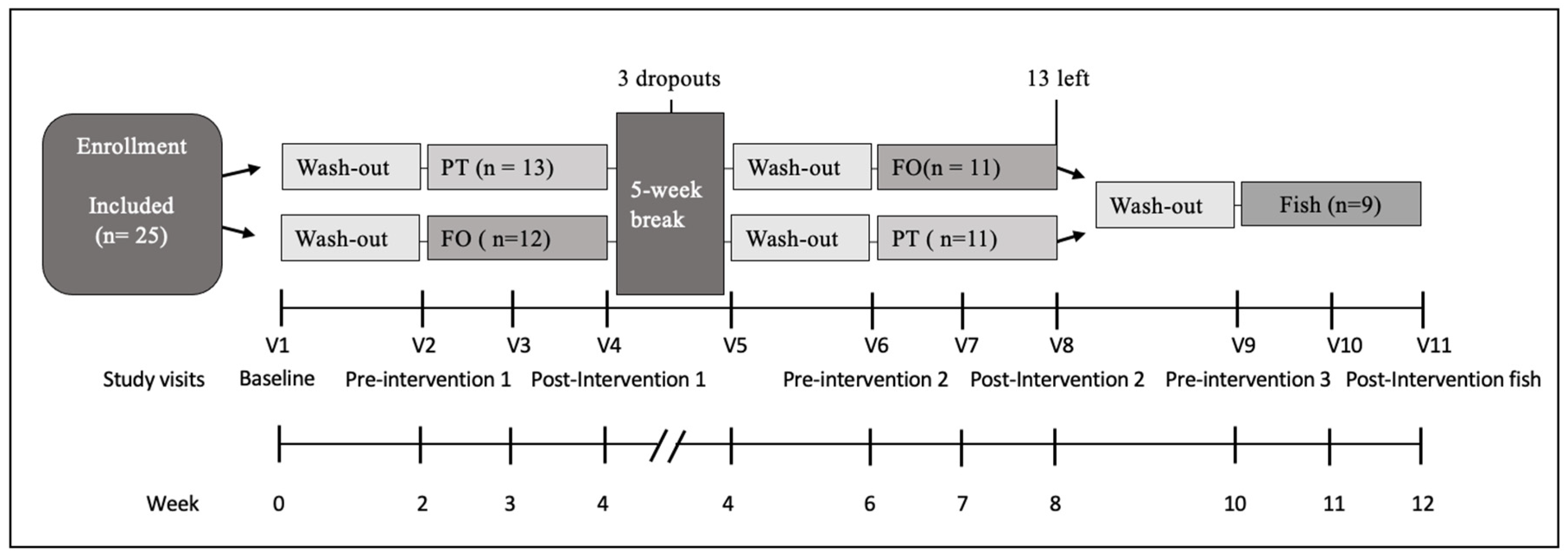
4.2. Study Design and Intervention
4.3. Subject Characterization
4.4. Study Products
4.5. Blood Plasma, Serum Measurements, and Fecal Samples
4.6. Quantification of Plasma Fatty Acids
4.7. Quantification of the Carotenoid FX and Its Metabolites FXOH and AxA in Plasma
4.8. Quantification of the Carotenoids Lutein/Zeaxanthin, Lycopenes, β-Cryptoxantins and α/β-Carotene, Retinol, and α/γ-Tocopherol in Plasma
4.9. Quantification of Tocopherol, Tocotrienol, and Carotenoids in PT and FO by HPLC
4.10. SCFA Analysis from Stool Samples
4.11. Analysis of Intestinal Permeability Markers Plasma Lipopolysaccharide-Binding Protein (LBP) and Fecal Zonulin
4.12. Gut Microbiome Analysis
4.13. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Myers, S.S.; Smith, M.R.; Guth, S.; Golden, C.D.; Vaitla, B.; Mueller, N.D.; Dangour, A.D.; Huybers, P. Climate Change and Global Food Systems: Potential Impacts on Food Security and Undernutrition. Annu. Rev. Public Health 2017, 38, 259–277. [Google Scholar] [CrossRef]
- Johnson, T.J.; Katuwal, S.; Anderson, G.A.; Gu, L.; Zhou, R.; Gibbons, W.R. Photobioreactor cultivation strategies for microalgae and cyanobacteria. Biotechnol. Prog. 2018, 34, 811–827. [Google Scholar] [CrossRef]
- Martins, D.A.; Custódio, L.; Barreira, L.; Pereira, H.; Ben-Hamadou, R.; Varela, J.; Abu-Salah, K.M. Alternative Sources of n-3 Long-Chain Polyunsaturated Fatty Acids in Marine Microalgae. Mar. Drugs 2013, 11, 2259–2281. [Google Scholar] [CrossRef]
- Ryckebosch, E.; Bruneel, C.; Muylaert, K.; Foubert, I. Microalgae as an alternative source of omega-3 long chain polyunsaturated fatty acids. Lipid Technol. 2012, 24, 128–130. [Google Scholar] [CrossRef]
- Torres-Tiji, Y.; Fields, F.J.; Mayfield, S.P. Microalgae as a future food source. Biotechnol. Adv. 2020, 41, 107536. [Google Scholar] [CrossRef]
- Eggersdorfer, M.; Wyss, A. Carotenoids in human nutrition and health. Arch. Biochem. Biophys. 2018, 652, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Sommerburg, O.; Keunen, J.E.; Bird, A.C.; Van Kuijk, F.J.G.M. Fruits and vegetables that are sources for lutein and zeaxanthin: The macular pigment in human eyes. Br. J. Ophthalmol. 1998, 82, 907–910. [Google Scholar] [CrossRef]
- Schweiggert, R.M.; Carle, R. Carotenoid Deposition in Plant and Animal Foods and Its Impact on Bioavailability. Crit. Rev. Food Sci. Nutr. 2015, 57, 1807–1830. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-A.; Hayden, M.M.; Bannerman, S.; Jansen, J.; Crowe-White, K.M. Anti-Apoptotic Effects of Carotenoids in Neurodegeneration. Molecules 2020, 25, 3453. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Song, M.-H.; Oh, J.-W.; Keum, Y.-S.; Saini, R.K. Pro-oxidant Actions of Carotenoids in Triggering Apoptosis of Cancer Cells: A Review of Emerging Evidence. Antioxidants 2020, 9, 532. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Photoprotection by dietary carotenoids: Concept, mechanisms, evidence and future development. Mol. Nutr. Food Res. 2012, 56, 287–295. [Google Scholar] [CrossRef]
- Kim, S.M.; Jung, Y.-J.; Kwon, O.-N.; Cha, K.H.; Um, B.-H.; Chung, D.; Pan, C.-H. A Potential Commercial Source of Fucoxanthin Extracted from the Microalga Phaeodactylum tricornutum. Appl. Biochem. Biotechnol. 2012, 166, 1843–1855. [Google Scholar] [CrossRef]
- Fung, A.; Hamid, N.; Lu, J. Fucoxanthin content and antioxidant properties of Undaria pinnatifida. Food Chem. 2013, 136, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.-J.; Kim, S.M.; Jeong, S.-M.; Choi, H.-N.; Jang, Y.-H.; Kim, J.-I. Antioxidant effect of Phaeodactylum tricornutum in mice fed high-fat diet. Food Sci. Biotechnol. 2013, 22, 107–113. [Google Scholar] [CrossRef]
- Salem, N.; Eggersdorfer, M. Is the world supply of omega-3 fatty acids adequate for optimal human nutrition? Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Brenna, J.T.; Salem, N.; Sinclair, A.J.; Cunnane, S.C. α-Linolenic acid supplementation and conversion to n-3 long-chain polyunsaturated fatty acids in humans. Prostaglandins Leukot. Essent. Fat. Acids 2009, 80, 85–91. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA) Panel Members on Dietetic Products, Nutrition, and Allergies. Scientific Opinion on Dietary Reference Values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar] [CrossRef]
- AbuMweis, S.; Jew, S.; Tayyem, R.; Agraib, L. Eicosapentaenoic acid and docosahexaenoic acid containing supplements modulate risk factors for cardiovascular disease: A meta-analysis of randomised placebo-control human clinical trials. J. Hum. Nutr. Diet. 2017, 31, 67–84. [Google Scholar] [CrossRef] [PubMed]
- Péron, G.; Mittaine, J.F.; Le Gallic, B. Where do fishmeal and fish oil products come from? An analysis of the conversion ratios in the global fishmeal industry. Mar. Policy 2010, 34, 815–820. [Google Scholar] [CrossRef]
- Tocher, D.R. Omega-3 long-chain polyunsaturated fatty acids and aquaculture in perspective. Aquaculture 2015, 449, 94–107. [Google Scholar] [CrossRef]
- USA Food & Drug Administration. GRAS Notice Inventory. 2018. Available online: https://www.fda.gov/food/generally-recognized-safe-gras/gras-notice-inventory (accessed on 9 September 2020).
- European Commission. EUR-Lex-32017R2470-Durchführungsverordnung (EU) 2017/2470 (2017). Available online: https://eur-lex.europa.eu/eli/reg_impl/2017/2470/oj/deu (accessed on 8 December 2021).
- Mayer, C.; Côme, M.; Ulmann, L.; Zittelli, G.C.; Faraloni, C.; Nazih, H.; Ouguerram, K.; Chénais, B.; Mimouni, V. Preventive Effects of the Marine Microalga Phaeodactylum tricornutum, used as a Food Supplement, on Risk Factors Associated with Metabolic Syndrome in Wistar Rats. Nutrients 2019, 11, 1069. [Google Scholar] [CrossRef]
- Mayer, C.; Côme, M.; Blanckaert, V.; Zittelli, G.C.; Faraloni, C.; Nazih, H.; Ouguerram, K.; Mimouni, V.; Chénais, B. Effect of Carotenoids from Phaeodactylum tricornutum on Palmitate-Treated HepG2 Cells. Molecules 2020, 25, 2845. [Google Scholar] [CrossRef] [PubMed]
- Gille, A.; Neumann, U.; Louis, S.; Bischoff, S.C.; Briviba, K. Microalgae as a potential source of carotenoids: Comparative results of an in vitro digestion method and a feeding experiment with C57BL/6J mice. J. Funct. Foods 2018, 49, 285–294. [Google Scholar] [CrossRef]
- Simris Ingredients: 100% Plant-Based, Non-GMO & Toxin-Free. Available online: https://www.simris.com/pages/ingredients (accessed on 23 April 2021).
- Microphyt—USA Food and Drug Administration Search Results. Available online: https://search.usa.gov/search?query=Microphyt&affiliate=fda1 (accessed on 3 December 2021).
- Neumann, U.; Derwenskus, F.; Gille, A.; Louis, S.; Schmid-Staiger, U.; Briviba, K.; Bischoff, S.C. Bioavailability and Safety of Nutrients from the Microalgae Chlorella vulgaris, Nannochloropsis oceanica and Phaeodactylum tricornutum in C57BL/6 Mice. Molecules 2018, 10, 965. [Google Scholar] [CrossRef] [PubMed]
- Neumann, U.; Louis, S.; Gille, A.; Derwenskus, F.; Schmid-Staiger, U.; Briviba, K.; Bischoff, S.C. Anti-inflammatory effects of Phaeodactylum tricornutum extracts on human blood mononuclear cells and murine macrophages. Environ. Boil. Fishes 2018, 30, 2837–2846. [Google Scholar] [CrossRef]
- Neumann, U.; Derwenskus, F.; Flaiz Flister, V.; Schmid-Staiger, U.; Hirth, T.; Bischoff, S.C. Fucoxanthin, A Carotenoid Derived from Phaeodactylum tricornutum Exerts Antiproliferative and Antioxidant Activities In Vitro. Antioxidants 2019, 8, 183. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tang, Y.; Zhang, Y.; Zhang, S.; Qu, J.; Wang, X.; Kong, R.; Han, C.; Liu, Z. Fucoxanthin: A Promising Medicinal and Nutritional Ingredient. Evid.-Based Complement. Altern. Med. 2015, 2015, 723515. [Google Scholar] [CrossRef]
- Seethaler, B.; Basrai, M.; Neyrinck, A.M.; Nazare, J.-A.; Walter, J.; Delzenne, N.M.; Bischoff, S.C. Biomarkers for assessment of intestinal permeability in clinical practice. Am. J. Physiol. Liver Physiol. 2021, 321, G11–G17. [Google Scholar] [CrossRef]
- Sørensen, M.; Berge, G.M.; Reitan, K.I.; Ruyter, B. Microalga Phaeodactylum tricornutum in feed for Atlantic salmon (Salmo salar)—Effect on nutrient digestibility, growth and utilization of feed. Aquaculture 2016, 460, 116–123. [Google Scholar] [CrossRef]
- Ryan, L.; Fraser, A.; Symington, A. Algal-oil supplements are a viable alternative to fish-oil supplements in terms of docosahexaenoic acid (22:6n-3; DHA). J. Funct. Foods 2015, 19, 852–858. [Google Scholar] [CrossRef]
- Arterburn, L.M.; Oken, H.A.; Hall, E.B.; Hamersley, J.; Kuratko, C.N.; Hoffman, J.P. Algal-Oil Capsules and Cooked Salmon: Nutritionally Equivalent Sources of Docosahexaenoic Acid. J. Am. Diet. Assoc. 2008, 108, 1204–1209. [Google Scholar] [CrossRef]
- Conquer, J.A.; Holub, B.J. Dietary docosahexaenoic acid as a source of eicosapentaenoic acid in vegetarians and omnivores. Lipids 1997, 32, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Wang, K.; Wan, L.; Li, A.; Hu, Q.; Zhang, C. Production, Characterization, and Antioxidant Activity of Fucoxanthin from the Marine Diatom Odontella aurita. Mar. Drugs 2013, 11, 2667–2681. [Google Scholar] [CrossRef]
- Haimeur, A.; Ulmann, L.; Mimouni, V.; Guéno, F.; Pineau-Vincent, F.; Meskini, N.; Tremblin, G. The role of Odontella aurita, a marine diatom rich in EPA, as a dietary supplement in dyslipidemia, platelet function and oxidative stress in high-fat fed rats. Lipids Health Dis. 2012, 11, 147. [Google Scholar] [CrossRef]
- Amine, H.; Benomar, Y.; Haimeur, A.; Messaouri, H.; Meskini, N.; Taouis, M. Odontella aurita-enriched diet prevents high fat diet-induced liver insulin resistance. J. Endocrinol. 2016, 228, 1–12. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Walker, R.E.; Jackson, K.H.; Tintle, N.L.; Shearer, G.C.; Bernasconi, A.; Masson, S.; Latini, R.; Heydari, B.; Kwong, R.Y.; Flock, M.; et al. Predicting the effects of supplemental EPA and DHA on the omega-3 index. Am. J. Clin. Nutr. 2019, 110, 1034–1040. [Google Scholar] [CrossRef]
- Carrera-Bastos, P.; Fontes-Villalba, M.; O’Keefe, J.H.; Lindeberg, S.; Cordain, L. The western diet and lifestyle and diseases of civilization. Res. Rep. Clin. Cardiol. 2011, 2, 15–35. [Google Scholar] [CrossRef]
- Barden, A.E.; Mas, E.; Mori, T.A. n-3 Fatty acid supplementation and proresolving mediators of inflammation. Curr. Opin. Lipidol. 2016, 27, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhuang, P.; He, W.; Chen, J.N.; Wang, W.Q.; Freedman, N.D.; Abnet, C.; Wang, J.B.; Jiao, J.J. Association of fish and long-chain omega-3 fatty acids intakes with total and cause-specific mortality: Prospective analysis of 421,309 individuals. J. Intern. Med. 2018, 284, 399–417. [Google Scholar] [CrossRef]
- Asher, A.; Tintle, N.L.; Myers, M.; Lockshon, L.; Bacareza, H.; Harris, W.S. Blood omega-3 fatty acids and death from COVID-19: A pilot study. Prostaglandins Leukot. Essent. Fat. Acids 2021, 166, 102250. [Google Scholar] [CrossRef]
- Kawee-Ai, A.; Kuntiya, A.; Kim, S.M. Anticholinesterase and Antioxidant Activities of Fucoxanthin Purified from the Microalga Phaeodactylum Tricornutum. Nat. Prod. Commun. 2013, 8, 1381–1386. [Google Scholar] [CrossRef]
- Xiao, H.; Zhao, J.; Fang, C.; Cao, Q.; Xing, M.; Li, X.; Hou, J.; Ji, A.; Song, S. Advances in Studies on the Pharmacological Activities of Fucoxanthin. Mar. Drugs 2020, 18, 634. [Google Scholar] [CrossRef]
- Foo, S.C.; Yusoff, F.M.; Ismail, M.; Basri, M.; Yau, S.K.; Khong, N.M.; Chan, K.W.; Ebrahimi, M. Antioxidant capacities of fucoxanthin-producing algae as influenced by their carotenoid and phenolic contents. J. Biotechnol. 2017, 241, 175–183. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, H.; Wen, H.; Fang, H.; Hong, Z.; Yi, R.; Liu, R. Simultaneous Determination of Fucoxanthin and Its Deacetylated Metabolite Fucoxanthinol in Rat Plasma by Liquid Chromatography-Tandem Mass Spectrometry. Mar. Drugs 2015, 13, 6521–6536. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Ozaki, Y.; Taminato, M.; Das, S.K.; Mizuno, M.; Yoshimura, K.; Maoka, T.; Kanazawa, K. The distribution and accumulation of fucoxanthin and its metabolites after oral administration in mice. Br. J. Nutr. 2009, 102, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Ozaki, Y.; Mizuno, M.; Yoshida, M.; Nishitani, Y.; Azuma, T.; Komoto, A.; Maoka, T.; Tanino, Y.; Kanazawa, K. Pharmacokinetics of fucoxanthinol in human plasma after the oral administration of kombu extract. Br. J. Nutr. 2012, 107, 1566–1569. [Google Scholar] [CrossRef]
- Mikami, N.; Hosokawa, M.; Miyashita, K.; Sohma, H.; Ito, Y.M.; Kokai, Y. Reduction of HbA1c levels by fucoxanthin-enriched akamoku oil possibly involves the thrifty allele of uncoupling protein 1 (UCP1): A randomised controlled trial in normal-weight and obese Japanese adults. J. Nutr. Sci. 2017, 6, e5. [Google Scholar] [CrossRef] [PubMed]
- Asai, A.; Yonekura, L.; Nagao, A. Low bioavailability of dietary epoxyxanthophylls in humans. Br. J. Nutr. 2008, 100, 273–277. [Google Scholar] [CrossRef]
- Ravi, H.; Arunkumar, R.; Baskaran, V. Chitosan-glycolipid nanogels loaded with anti-obese marine carotenoid fucoxanthin: Acute and sub-acute toxicity evaluation in rodent model. J. Biomater. Appl. 2015, 30, 420–434. [Google Scholar] [CrossRef]
- Perry, A.; Rasmussen, H.; Johnson, E.J. Xanthophyll (lutein, zeaxanthin) content in fruits, vegetables and corn and egg products. J. Food Compos. Anal. 2009, 22, 9–15. [Google Scholar] [CrossRef]
- Tang, G. Bioconversion of dietary provitamin A carotenoids to vitamin A in humans. Am. J. Clin. Nutr. 2010, 91, 1468S–1473S. [Google Scholar] [CrossRef]
- Lietz, G.; Lange, J.; Rimbach, G. Molecular and dietary regulation of β,β-carotene 15,15′-monooxygenase 1 (BCMO1). Arch. Biochem. Biophys. 2010, 502, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Harvey, P.J.; Ben-Amotz, A. Towards a sustainable Dunaliella salina microalgal biorefinery for 9-cis β-carotene production. Algal Res. 2020, 50, 102002. [Google Scholar] [CrossRef]
- Häubner, N.; Sylvander, P.; Vuori, K.; Snoeijs, P. Abiotic stress modifies the synthesis of alpha-tocopherol and beta-carotene in phytoplankton species. J. Phycol. 2014, 50, 753–759. [Google Scholar] [CrossRef]
- Costantini, L.; Molinari, R.; Farinon, B.; Merendino, N. Impact of Omega-3 Fatty Acids on the Gut Microbiota. Int. J. Mol. Sci. 2017, 18, 2645. [Google Scholar] [CrossRef]
- Derwenskus, F.; Metz, F.; Gille, A.; Schmid-Staiger, U.; Briviba, K.; Schließmann, U.; Hirth, T. Pressurized extraction of unsaturated fatty acids and carotenoids from wet Chlorella vulgaris and Phaeodactylum tricornutum biomass using subcritical liquids. GCB Bioenergy 2019, 11, 335–344. [Google Scholar] [CrossRef]
- Thurnhofer, S.; Lehnert, K.; Vetter, W. Exclusive quantification of methyl-branched fatty acids and minor 18:1-isomers in foodstuff by GC/MS in the SIM mode using 10,11-dichloroundecanoic acid and fatty acid ethyl esters as internal standards. Eur. Food Res. Technol. 2008, 226, 975–983. [Google Scholar] [CrossRef]
- Thurnhofer, S.; Vetter, W. A Gas Chromatography/Electron Ionization—Mass Spectrometry—Selected Ion Monitoring Method for Determining the Fatty Acid Pattern in Food after Formation of Fatty Acid Methyl Esters. J. Agric. Food Chem. 2005, 53, 8896–8903. [Google Scholar] [CrossRef]
- Stuetz, W.; Mcgready, R.; Cho, T.; Prapamontol, T.; Biesalski, H.; Stepniewska, K.; Nosten, F. Relation of DDT residues to plasma retinol, α-tocopherol, and β-carotene during pregnancy and malaria infection: A case–control study in Karen women in northern Thailand. Sci. Total Environ. 2006, 363, 78–86. [Google Scholar] [CrossRef]
- Montoya-Arroyo, A.; Alfaro-Solís, J.D.; Esquivel, P.; Jiménez, V.M.; Frank, J. Vitamin E profiles in Acrocomia aculeata from three regions in Costa Rica. J. Food Compos. Anal. 2021, 100, 103936. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
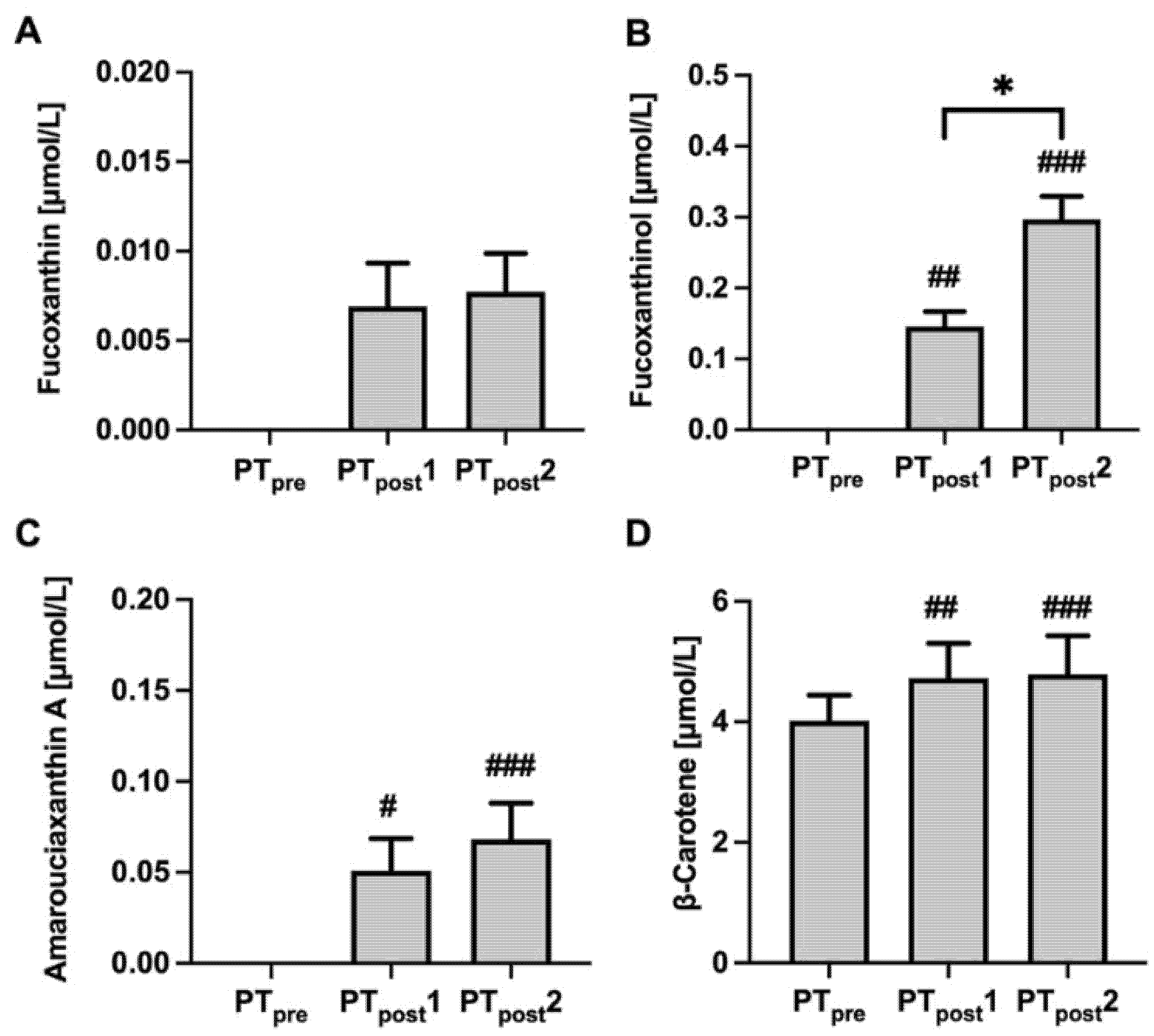
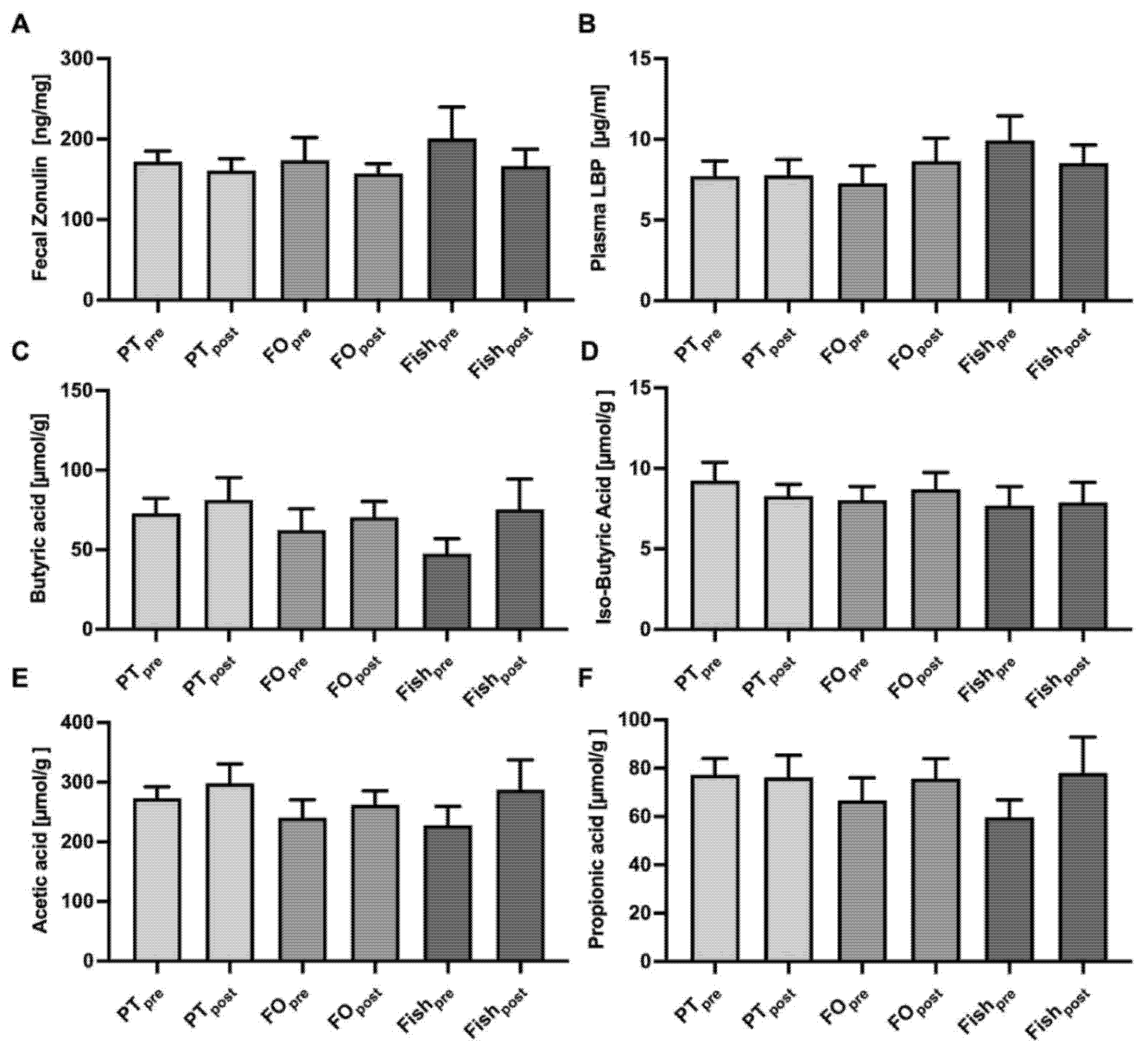
| Side Effects | PT (n = 22) Diary|Protocol | FO (n = 22) Diary|Protocol | Fish (n = 9) Diary|Protocol | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Minimal | Mild | Severe | Minimal | Mild | Severe | Minimal | Mild | Severe | |
| Bowel problems | 2|6 | - | - | 1|6 | - | - | - | - | - |
| Bloating | 1|6 | . | - | 1|1 | - | - | - | - | |
| Stomach pain | 2|2 | - | - | - | - | - | - | - | - |
| Constipation | 1|2 | - | - | - | - | - | - | - | - |
| Stool discoloration | 0|2 | - | - | - | - | - | - | - | - |
| Increased bowel movements | 1|2 | - | - | - | 1|2 | - | - | - | - |
| Belching (at least 1×) | 1|2 | - | - | - | 7|7 | - | - | - | - |
| Headache | 0|2 | - | - | 1|0 | - | - | - | - | - |
| Increased skin impurities | 0|1 | - | - | - | - | - | - | - | - |
| Increased feeling of thirst | 0|1 | - | - | - | - | - | - | - | - |
| Reduced appetite | 0|1 | - | - | - | - | - | - | - | - |
| Blood Count | Baseline (V1) | PTpre (V2/V6) | PTpost (V4/V8) | FOpre (V2/V6) | FOpost (V4/V8) | Fishpre (V9) | Fishpost (V11) |
|---|---|---|---|---|---|---|---|
| γ-GT [mg/L] | 14.4 ± 4.9 | 14.1 ± 4.4 | 13.6 ± 4.0 | 14.0 ± 4.4 | 14.4 ± 5.2 | 11.4 ± 3.2 | 12.4 ± 2.7 * |
| AST [mg/L] | 21.7 ± 5.0 | 21.5 ± 3.9 | 25.0 ± 12.5 | 21.6 ± 4.8 | 24.0 ± 10.3 | 23.6 ± 4.2 | 23.3 ± 7.8 |
| ALT [mg/L] | 19.3 ± 12.8 | 16.8 ± 5.5 | 17.0 ± 7.8 | 15.5 ± 5.8 | 21.1 ± 21 | 14.8 ± 5.9 | 12.9 ± 3.3 # |
| CRP [mg/L] | 1.1 ± 1.4 | 1.0 ± 0.9 | 1.1 ± 1.0 | 0.8 ± 0.8 | 0.8 ± 0.8 | 0.8 ± 0.7 | 1.1 ± 1.0 |
| Glucose [mg/dL] | 81.5 ± 8.9 | 83.0 ± 7.0 | 81.1 ± 6.0 | 83.8 ± 6.0 | 81.1 ± 5.7 | 77.8 ± 3.7 | 81.9 ± 3.1 |
| Uric acid [mg/dL] | 4.5 ± 0.9 | 4.4 ± 0.8 | 4.8 ± 1.1 ** | 4.6 ± 1.1 | 4.7 ± 1.1 | 4.4 ± 0.5 | 4.3 ± 0.6 |
| Chol [mg/dL] | 187 ± 37 | 186 ± 40 | 195 ± 31 | 188 ± 37 | 180 ± 30 §§ | 191 ± 38 | 192 ± 36 |
| TAG [mg/dL] | 78.2 ± 23.3 | 91.2 ± 49.5 | 94.8 ± 43.7 | 84.2 ± 27.5 | 75.0 ± 28.6 §§ | 91.3 ± 69.9 | 78.3 ± 36.7 |
| HDL Chol [mg/dL] | 58.7 ± 12.3 | 59.3 ± 11.1 | 62.3 ± 12.0 # | 61.5 ± 13.7 | 58.9 ± 11.9 §§ | 63.9 ± 10.6 | 66.3 ± 8.7 |
| LDL Chol [mg/dL] | 115 ± 26.2 | 106 ± 27.2 | 115 ± 22.7 | 110 ± 26.6 | 104 ± 21.3 #§§ | 116 ± 26.4 | 115 ± 24.0 |
| LDL/HDL- ratio | 2.1 ± 0.7 | 1.9 ± 0.6 # | 2.0 ± 0.6 | 1.9 ± 0.6 ## | 1.9 ± 0.5 ##§ | 1.9 ± 0.3 | 1.8 ± 0.3 * |
| Chol/HDL- ratio | 3.3 ± 0.8 | 3.3 ± 0.8 | 3.2 ± 0.8 | 3.2 ± 0.7 | 3.2 ± 0.7 | 3.1 ± 0.4 | 2.9 ± 0.4 ** |
| Parameter | All (n = 22) | Group 1 (n = 11) | Group 2 (n = 11) |
|---|---|---|---|
| Age [years] | 25.7 ± 5.7 | 26.5 ± 7 | 24.8 ± 3.3 |
| Female/male [n] | 15|7 | 10|3 | 5|4 |
| Body Weight [Kg] | 65.9 ± 10.7 | 65.8 ± 12.8 | 68.2 ± 9 |
| Height [m] | 1.74 ± 1 | 1.72 ± 0.09 | 1.78 ± 0.09 |
| BMI [kg/m2] | 21.5 ± 2.0 | 22.0 ± 2.6 | 21.5 ± 1.9 |
| Vegetarian|Vegan| | 2|2| | 2|0 | 0|2 |
| Hemoglobin [g/dL] | 14.4 ± 1.3 | 14.3 ± 1.1 | 14.4 ± 1.6 |
| Hematocrit [%] | 42.5 ± 2.9 | 42.2 ± 2.2 | 42.5 ± 3.8 |
| Erythrocytes [cells/pL] | 4.9 ± 0.3 | 4.9 ± 0.2 | 4.9 ± 0.4 |
| Leucocyte [cells/nL] | 5.6 ± 0.4 | 6.2 ± 1.2 | 5.3 ± 2.3 |
| Platelet count [cells/nL] | 240.8 ± 59.6 | 250.1 ± 54.5 | 245.5 ± 68 |
| TSH [mU/L] | 1.6 ± 0.6 | 1.8 ± 0.7 | 1.5 ± 0.4 |
| Insulin [µE/mL] | 7.1 ± 3.3 | 8.4 ± 3.6 | 6.0 ± 2.9 |
| HOMA-IR | 1.4 ± 0.7 | 1.7 ± 0.8 | 1.2 ± 0.7 |
| Dietary intake (FFQ) [g/day] | |||
| Fat | 49.2 ± 23.7 | 44.8 ± 18.9 | 55.5 ± 29.4 |
| Saturated fatty acids | 22.23 ± 11.0 | 19.7 ± 7.9 | 25.9 ± 14.11 |
| Unsaturated fatty acids | 7.6 ± 4.1 | 7.0 ± 3.4 | 8.5 ± 5.0 |
| Short chain fatty acids | 1.2 ± 0.8 | 0.9 ± 0.4 | 1.4 ± 1.1 |
| Long chain fatty acids | 43.40 ± 20.9 | 39.6 ± 17.13 | 48.9 ± 25.5 |
| Cholesterol | 191.8 ± 109.2 | 177.2 ± 104.4 | 212.9 ± 118.7 |
| Fatty Acids [mg/day] | PT | FO | Fish |
|---|---|---|---|
| n-3 PUFA | 305 | 310 | 554 * |
| EPA + DHA | 286 | 300 | 299 * |
| 20: 5 n-3 (EPA) | 282 | 178 | 198 * |
| 22:6 n-3 (DHA) | 5 | 122 | 101 * |
| 18:3 n-3 (ALA) | 0.9 | 1.8 | 94.1 * |
| 18:4 n-3 | 0.7 | 0.2 | 58.9 * |
| 22:5 n-3 | 9.5 | 7 | 100 * |
| 16:2 n-6 | 13.4 | 2.8 | 0.6 * |
| 18:2 n-6 | 3.8 | 5.3 | 34.9 * |
| 18:3 n-6 | 1.2 | 0.9 | 0.1 * |
| 20:3 n-6 | 0.1 | 0.3 | 0.2 * |
| 20:4 n-6 | 3.7 | 0 | 50.2 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stiefvatter, L.; Lehnert, K.; Frick, K.; Montoya-Arroyo, A.; Frank, J.; Vetter, W.; Schmid-Staiger, U.; Bischoff, S.C. Oral Bioavailability of Omega-3 Fatty Acids and Carotenoids from the Microalgae Phaeodactylum tricornutum in Healthy Young Adults. Mar. Drugs 2021, 19, 700. https://doi.org/10.3390/md19120700
Stiefvatter L, Lehnert K, Frick K, Montoya-Arroyo A, Frank J, Vetter W, Schmid-Staiger U, Bischoff SC. Oral Bioavailability of Omega-3 Fatty Acids and Carotenoids from the Microalgae Phaeodactylum tricornutum in Healthy Young Adults. Marine Drugs. 2021; 19(12):700. https://doi.org/10.3390/md19120700
Chicago/Turabian StyleStiefvatter, Lena, Katja Lehnert, Konstantin Frick, Alexander Montoya-Arroyo, Jan Frank, Walter Vetter, Ulrike Schmid-Staiger, and Stephan C. Bischoff. 2021. "Oral Bioavailability of Omega-3 Fatty Acids and Carotenoids from the Microalgae Phaeodactylum tricornutum in Healthy Young Adults" Marine Drugs 19, no. 12: 700. https://doi.org/10.3390/md19120700
APA StyleStiefvatter, L., Lehnert, K., Frick, K., Montoya-Arroyo, A., Frank, J., Vetter, W., Schmid-Staiger, U., & Bischoff, S. C. (2021). Oral Bioavailability of Omega-3 Fatty Acids and Carotenoids from the Microalgae Phaeodactylum tricornutum in Healthy Young Adults. Marine Drugs, 19(12), 700. https://doi.org/10.3390/md19120700






