A Concise Route for the Synthesis of Tetracyclic Meroterpenoids: (±)-Aureol Preparation and Mechanistic Interpretation
Abstract
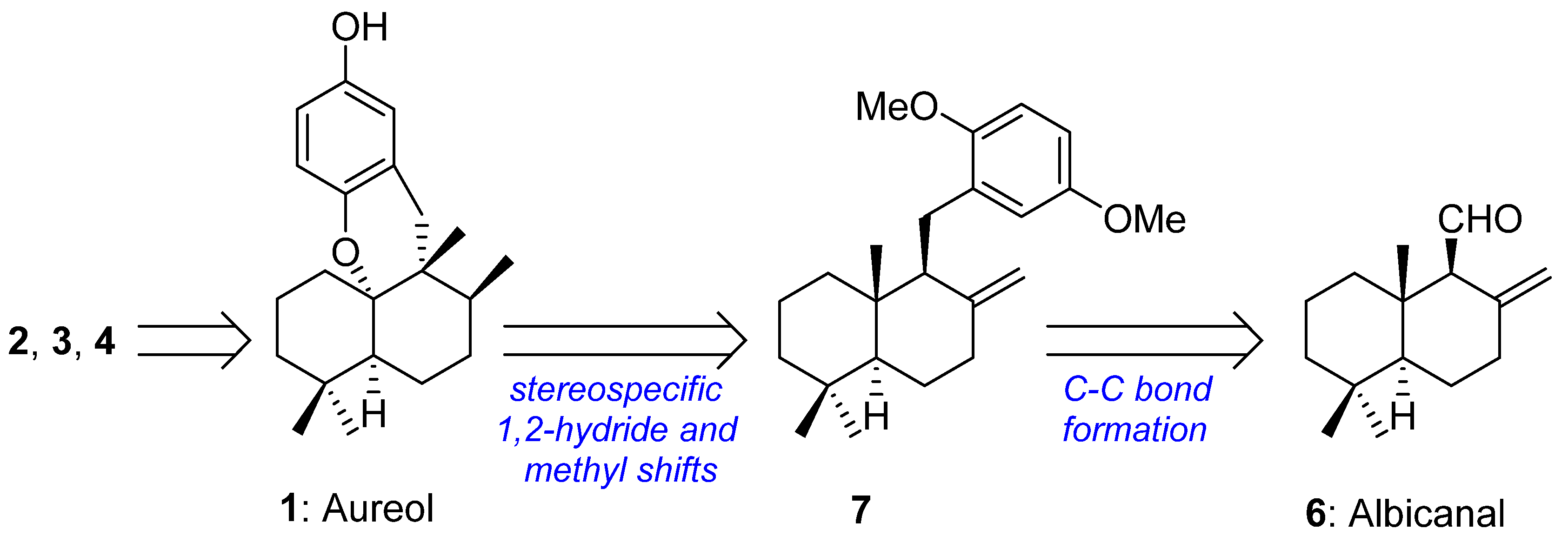
1. Introduction
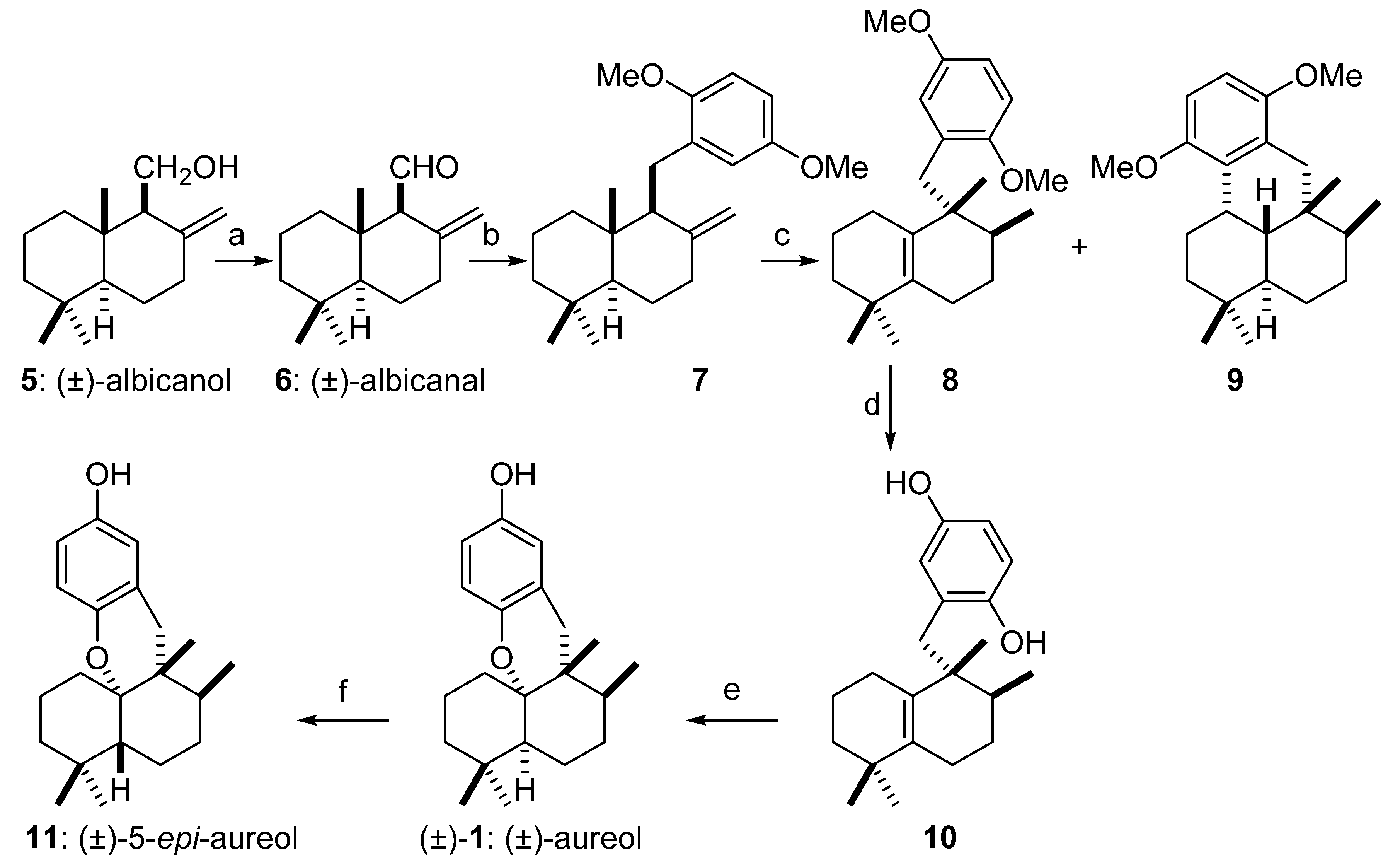
2. Results and Discussion
3. Experimental Section
3.1. General Methods
3.2. Dess–Martin Oxidation of (±)-Albicanol 5
3.3. Synthesis of Cis-Decaline 7
3.4. Synthesis of Tetrasubstituted Olefin 8
3.5. Preparation of 10 by Methyl Ether Deprotection of 8
3.6. Synthesis of (±)-Aureol ((±)-1)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Capon, R.J. Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier Science: New York, NY, USA, 1995; Volume 15, p. 89. [Google Scholar]
- Djura, P.; Stierle, D.B.; Sullivan, B.; Faulkner, D.J.; Arnold, E.; Clardy, J. Some metabolites of the marine sponges Smenospongia aurea and Smenospongia (polyfibrospongia) echina. J. Org. Chem. 1980, 45, 1435–1441. [Google Scholar] [CrossRef]
- Ciminiello, P.; Dell’Aversano, C.; Fattorusso, E.; Magno, S.; Pansini, M. Chemistry of verongida sponges. Secondary metabolite composition of the Caribbean sponge Verongula gigantea. J. Nat. Prod. 2000, 63, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.E.; Rueth, S.A.; Cross, S.S. An antiviral sesquiterpene hydroquinone from the marine sponge Strongylophora hartmani. J. Nat. Prod. 1991, 54, 1108–1111. [Google Scholar] [CrossRef] [PubMed]
- Utkina, N.K.; Denisenko, V.A.; Scholokova, O.V.; Virovaya, M.V.; Prokof’eva, N.G. Cyclosmenospongine, a new sesquiterpenoid aminoquinone from an Australian marine sponge Spongia sp. Tetrahedron Lett. 2003, 44, 101–102. [Google Scholar] [CrossRef]
- Bourguet-Kondracki, M.-L.; Martin, M.-T.; Guyot, M. Smenoqualone a novel sesquiterpenoid from the marine sponge Smenospongia sp. Tetrahedron Lett. 1992, 33, 8079–8080. [Google Scholar] [CrossRef]
- Shen, Y.-C.; Liaw, C.-C.; Ho, J.-R.; Khalil, A.T.; Kuo, Y.-H. Isolation of aureol from smenospongia sp. And cytotoxic activity of some aureol derivatives. Nat. Prod. Res. 2006, 20, 578–585. [Google Scholar] [CrossRef]
- Wright, A.E.; Cross, S.S.; Burres, N.S.; Koehn, F. Antiviral and Antitumor Terpene Hydroquinones from Marine Sponge and Methods of Use. Harbor Branch Oceanographic Institution, Inc., Fort Pierce, FL, USA. U.S. Patent PCT WO 9112250 A1, 22 August 1991. [Google Scholar]
- Longley, R.E.; McConnell, O.J.; Essich, E.; Harmody, D. Evaluation of marine sponge metabolites for cytotoxicity and signal transduction activity. J. Nat. Prod. 1993, 56, 915–920. [Google Scholar] [CrossRef]
- Wildermuth, R.; Speck, K.; Haut, F.-L.; Mayer, P.; Karge, B.; Brönstrup, M.; Magauer, T. A modular synthesis of tetracyclic meroterpenoid antibiotics. Nat. Commun. 2017, 8, 2083. [Google Scholar] [CrossRef]
- Taishi, T.; Takechi, S.; Mori, S. First total synthesis of (±)-stachyflin. Tetrahedron Lett. 1998, 39, 4347–4350. [Google Scholar] [CrossRef]
- Nakamura, M.; Suzuki, A.; Nakatani, M.; Fuchikami, T.; Inoue, M.; Katoh, T. An efficient synthesis of (+)-aureol via boron trifluoride etherate-promoted rearrangement of (+)-arenarol. Tetrahedron Lett. 2002, 43, 6929–6932. [Google Scholar] [CrossRef]
- Watanabe, K.; Sakurai, J.; Abe, H.; Katoh, T. Total synthesis of (+)-stachyflin: A potential anti-influenza A virus agent. Chem. Commun. 2010, 46, 4055–4057. [Google Scholar] [CrossRef] [PubMed]
- Marcos, I.S.; Conde, A.; Moro, R.F.; Basabe, P.; Diez, D.; Urones, J.G. Synthesis of quinone/hydroquinone sesquiterpenes. Tetrahedron 2010, 66, 8280–8290. [Google Scholar] [CrossRef]
- Sakurai, J.; Kikudhi, T.; Takahasi, O.; Watanabe, K.; Katoh, T. Enantioselective total synthesis of (+)-stachyflin; a potential anti-influenza A virus agente isolated from a microorganism. Eur. J. Org. Chem. 2011, 16, 2948–2957. [Google Scholar] [CrossRef]
- Kuan, K.K.W.; Pepper, H.P.; Bloch, W.M.; George, J.H. Total synthesis of (+)-aureol. Org. Lett. 2012, 14, 4710–4713. [Google Scholar] [CrossRef] [PubMed]
- Kamishima, T.; Kikuchi, T.; Katoh, T. Total synthesis of (+)-strongylin A, a rearranged sesquiterpenoid hydroquinone from a marine sponge. Eur. J. Org. Chem. 2013, 21, 4558–4563. [Google Scholar] [CrossRef]
- Rosales, A.; Muñoz-Bascón, J.; Roldan-Molina, E.; Rivas-Bascón, N.; Padial, N.M.; Rodríguez-Maecker, R.; Rodríguez-García, I.; Oltra, J.E. Synthesis of (±)-aureol by bionspired rearrangements. J. Org. Chem. 2015, 80, 1866–1870. [Google Scholar] [CrossRef]
- Katoh, T.; Atsumi, S.; Saito, R.; Narita, K.; Katoh, T. Unified synthesis of the marine sesquiterpene quinones (+)-smenoqualone, (-)-ilimaquinone, (+)-smenospongine, and (+)-sospongiaquinone. Eur. J. Org. Chem. 2017, 26, 3837–3849. [Google Scholar] [CrossRef]
- Wang, J.-L.; Li, H.-J.; Wang, M.; Wang, J.-H.; Wu, Y.-C. A six-step approach to marine natural product (+)-aureol. Tetrahedron Lett. 2018, 59, 945–948. [Google Scholar] [CrossRef]
- Gansäuer, A.; Rosales, A.; Justicia, J. Catalytic epoxypolyene cyclization via radicals: Highly diastereoselective formal synthesis of puupehedione and 8-epi-puupehedione. Synlett 2006, 6, 927–929. [Google Scholar] [CrossRef]
- Rosales, A.; López-Sánchez, C.; Álvarez-Corral, M.; Muñoz-Dorado, M.; Rodríguez-García, I. Total synthesis of (±)-euryfuran through Ti(III) catalyzed radical cyclization. Lett. Org. Chem. 2007, 4, 553–555. [Google Scholar] [CrossRef]
- Rosales, A.; Muñoz-Bascón, J.; Moráles-Álcazar, V.M.; Castilla-Alcalá, J.A.; Oltra, J.E. Ti(III)-catalyzed, concise synthesis of marine furanospongian diterpenes. RSC Adv. 2012, 2, 12922–12925. [Google Scholar] [CrossRef]
- Rosales Martinez, A.; Pozo Morales, L.; Diaz Ojeda, E. Cp2TiCl-catalyzed, concise synthetic approach to marine natural product (±)-cyclozonarone. Synth. Commun. 2019, 49, 2554–2560. [Google Scholar] [CrossRef]
- Goehl, M.; Seifert, K. Synthesis of the sesquiterpenes albicanol, drimanol, and drimanic acid, and the marine sesquiterpene hydroquinone deoxyspongiaquinol. Eur. J. Org. Chem. 2014, 31, 6975–6982. [Google Scholar] [CrossRef]
- Laube, T.; Schröder, J.; Stehle, R.; Seifert, K. Total synthesis of yahazunol, zonarone and isozonarone. Tetrahedron 2002, 58, 4299–4309. [Google Scholar] [CrossRef]
- Oblak, E.Z.; VanHyst, M.D.; Li, J.; Wiemer, A.J.; Wright, D.L. Cyclopropene cycloadditions with annulated furans: Total synthesis of (+)- and (−)-frondosin B and (+)-frondosin A. J. Am. Chem. Soc. 2014, 136, 4309–4315. [Google Scholar] [CrossRef] [PubMed]
- Speck, K.; Wildermuth, R.; Magauer, T. Convergent assembly of the tetracyclic meroterpenoid (−)-cyclosmenospongine by a non-biomimetic polyene cyclization. Angew. Chem. Int. Ed. 2016, 55, 14131–14135. [Google Scholar] [CrossRef]
- Carraher, C.E., Jr. Ionic Chain-Reaction and Complex Coordinative Polymerization (Addition Polymerization). In Polymer Chemistry, 6th ed.; Marcel Dekker: New York, NY, USA, 2003. [Google Scholar]
- Poigny, S.; Huor, T.; Guyot, M.; Samadi, M. Synthesis of (−)-Hyatellaquinone and Revision of Absolute Configuration of Naturally Occurring (+)-Hyatellaquinone. J. Org. Chem. 1999, 64, 9318–9320. [Google Scholar] [CrossRef]
- Urban, S.; Capon, R.J. Marine sesquiterpene quinones and hydroquinones: Acid-catalyzed rearrangements and stereochemical investigations. Aust. J. Chem. 1994, 47, 1023–1029. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosales Martínez, A.; Enríquez, L.; Jaraíz, M.; Pozo Morales, L.; Rodríguez-García, I.; Díaz Ojeda, E. A Concise Route for the Synthesis of Tetracyclic Meroterpenoids: (±)-Aureol Preparation and Mechanistic Interpretation. Mar. Drugs 2020, 18, 441. https://doi.org/10.3390/md18090441
Rosales Martínez A, Enríquez L, Jaraíz M, Pozo Morales L, Rodríguez-García I, Díaz Ojeda E. A Concise Route for the Synthesis of Tetracyclic Meroterpenoids: (±)-Aureol Preparation and Mechanistic Interpretation. Marine Drugs. 2020; 18(9):441. https://doi.org/10.3390/md18090441
Chicago/Turabian StyleRosales Martínez, Antonio, Lourdes Enríquez, Martín Jaraíz, Laura Pozo Morales, Ignacio Rodríguez-García, and Emilio Díaz Ojeda. 2020. "A Concise Route for the Synthesis of Tetracyclic Meroterpenoids: (±)-Aureol Preparation and Mechanistic Interpretation" Marine Drugs 18, no. 9: 441. https://doi.org/10.3390/md18090441
APA StyleRosales Martínez, A., Enríquez, L., Jaraíz, M., Pozo Morales, L., Rodríguez-García, I., & Díaz Ojeda, E. (2020). A Concise Route for the Synthesis of Tetracyclic Meroterpenoids: (±)-Aureol Preparation and Mechanistic Interpretation. Marine Drugs, 18(9), 441. https://doi.org/10.3390/md18090441





