Abstract
Beyond their significant contribution to the dietary and industrial supplies, marine algae are considered to be a potential source of some unique metabolites with diverse health benefits. The pharmacological properties, such as antioxidant, anti-inflammatory, cholesterol homeostasis, protein clearance and anti-amyloidogenic potentials of algal metabolites endorse their protective efficacy against oxidative stress, neuroinflammation, mitochondrial dysfunction, and impaired proteostasis which are known to be implicated in the pathophysiology of neurodegenerative disorders and the associated complications after cerebral ischemia and brain injuries. As was evident in various preclinical studies, algal compounds conferred neuroprotection against a wide range of neurotoxic stressors, such as oxygen/glucose deprivation, hydrogen peroxide, glutamate, amyloid β, or 1-methyl-4-phenylpyridinium (MPP+) and, therefore, hold therapeutic promise for brain disorders. While a significant number of algal compounds with promising neuroprotective capacity have been identified over the last decades, a few of them have had access to clinical trials. However, the recent approval of an algal oligosaccharide, sodium oligomannate, for the treatment of Alzheimer’s disease enlightened the future of marine algae-based drug discovery. In this review, we briefly outline the pathophysiology of neurodegenerative diseases and brain injuries for identifying the targets of pharmacological intervention, and then review the literature on the neuroprotective potentials of algal compounds along with the underlying pharmacological mechanism, and present an appraisal on the recent therapeutic advances. We also propose a rational strategy to facilitate algal metabolites-based drug development.
1. Introduction
Neurons and supporting cells of the brain encounter degenerative changes during physiological or pathological aging, ischemic stroke, or other brain injuries [1]. The degenerative brain disorders such as Alzheimer’s disease (AD) and Parkinson’s diseases (PD) are the consequence of pathological brain aging, which are characterized by the region-specific loss of neurons [2]. Globally, these diseases account for the major causes of dementia among the elderly [3]. Although the exact etiologies of these brain disorders are not revealed yet, they share some common pathophysiology, such as oxidative stress (OS), neuroinflammation, mitochondrial dysfunction, protein misfolding, and defective protein clearance system that, in turn, make these diseases complicated [4,5], whereas, ischemic, traumatic, and other brain injuries, if not fatal, ensue secondary damage and constitute the appreciable causes of cognitive deficits among patients. Like neurodegenerative disorders, brain injuries also follow the same pathophysiology [6,7]. Whatever the forms of dementia disorder, the current therapeutic option can only alleviate symptoms, rather than halting the disease progression. Moreover, current drugs are associated with multiple side effects. Considering the tremendous social and economic impact of these diseases, scientists are, therefore, paying research efforts to discover the potential therapeutic agents that can target disease pathogenesis without causing undesirable effects in patient’s health. Although synthetic drugs have some advantages such as easy to develop, naturally-derived compounds have received priority as they are relatively well-tolerated. Natural compounds have been claimed to show anti-inflammatory, antioxidant, and immunomodulatory effects [8]. Compounds showing multiple pharmacological effects offer a better solution for the remedy of neurological disorders with complex pathomechanisms [9]. In the published literature, a significant quantity of natural products has been reported to show neuroprotective activity against a wide range of toxic insults [10,11]. Some of them have shown therapeutic promise in preclinical studies [12] and clinical trials [13,14].
Macroalgae, also known as seaweed, are among the highly abundant marine lives and potentially contribute to the renewable resources for food and industrial products [15,16,17]. Beyond this importance, algal metabolites, such as phenolics, alkaloids, terpenoids, carotenoids, phytosterols, and polysaccharides have attracted much attention to medicinal chemistry due to their structural uniqueness and functional diversity [17,18,19,20]. These biofunctional compounds have shown to provide neuroprotection in preclinical models of neurodegenerative diseases, ischemic stroke, brain trauma, diabetes, and obesity, among many others, owing to their antioxidant, anti-inflammatory, and immunomodulatory capacities [21,22,23,24,25,26,27,28]. Evidence suggests that algal metabolites, particularly fucoxanthin, fucosterol, and fucoidan could be potential leads for the development of therapy against CNS diseases [22,29,30,31]. Although the algal metabolite-based drug discovery progresses very slowly, the discovery of sodium oligomannate and its conditional approval as an anti-AD drug [32] raises hope for the future development of potential therapeutic agents from marine algae.
Over the last decade, some excellent works reviewed the neuroprotective effects of marine algae and their metabolites [21,22,23,29,33,34,35]. However, some of these reviews limited their scope either to a single pathogenic mechanism such as neuroinflammation [22] or to categorical brain disorders such as AD or PD [22,23,29,34,35]. Others have reviewed literature published a decade or half a decade ago [23,36]. Moreover, a few of them included reports that cover ischemic or other brain injuries. In the meantime, information on some potential algal compounds with neuroprotective activity has appeared in the scientific platform and there has also been significant progress in the clinical aspect. Addressing the knowledge gap and the possible limitations, offering a comprehensive review updating information on the neuroprotective effects of algal compounds and their therapeutic advances is timely. In this comprehensive review, we first briefly outline the pathobiology of neurodegenerative disorders, ischemic stroke, and traumatic brain injury and then provide pharmacological insights into the neuroprotective potentials of algal metabolites and highlight the recent progress in algae-based drug discovery. Finally, the rational strategy for algal compounds-based drug development has been discussed.
2. Pathophysiology of Brain Disorders
2.1. Neurodegenerative Disorders (AD and PD)
Neurodegenerative disorders, including AD and PD, are of major public health concern and contribute to the prime causes of dementia among elderly people. The pathological hallmarks of AD include extracellular deposition of amyloid plaque and intraneuronal aggregation of neurofibrillary tangles (NFT) [37]. On the other hand, PD is characterized by the degeneration of dopaminergic neurons in the substantia nigra [37] with the pathological hallmark of intraneuronal aggregation of α-synuclein [38]. Although the exact pathophysiology of these brain disorders remains elusive, it has been demonstrated that OS, neuroinflammation, mitochondrial dysfunction, and protein misfolding largely contribute to their development [37]. OS and neuroinflammation are two considerably diverse disease processes in many pathological events [39]. Conversely, they are interplayed with each other in the entire disease process. Thus, inhibition of neuroinflammation may reduce the OS and vice versa.
Oxidative stress (OS) is a pathological condition that develops when the production of reactive oxygen species (ROS) reaches an excessive level with lower efficiency of the cellular antioxidant defense system [40]. Factors contributing to OS in the brain include excitotoxicity, depletion of the cellular antioxidant system, high susceptibility to lipid peroxidation, and high oxygen demand [41]. OS may lead to mitochondrial dysfunction, which further results in the excessive ROS generation and establishes a vicious cycle of OS [42,43]. Moreover, the endoplasmic reticulum (ER), a site for protein folding, also takes part in ROS generation [44]. Protein misfolding in ER results in ER stress that is further responsible for ROS production [45]. ROS potentially contributes to the damage of cells through compromising the structure and function of biomolecules, including lipid peroxidation, protein oxidation, and deoxyribonucleic acid (DNA) damage, which eventually install neurodegeneration [38].
Neuroinflammation is another inevitable pathogenic factor of many neurodegenerative disorders [46]. Microglial activation is the major contributor to neuroinflammation [46]. A range of stimuli, including infection, trauma, toxic insults, and ischemia, may initiate microglial activation and disrupt the central nervous system (CNS) homeostasis [47,48]. Once activated, microglia released pro-inflammatory and neurotoxic elements, like chemokines, cytokines, proteases, eicosanoids, ROS, and excitatory amino acids [47]. All of these elements are documented as a key player in neuroinflammation-associated OS as well as chronic neurodegeneration [49]. The deposition of misfolded proteins, as evident in the major NDD, can also induce an inflammatory response, which further causes OS [50].
Dysregulation of cholesterol homeostasis is also a critical factor that could induce OS and inflammation, and thus may contribute to the pathogenesis of major brain disorders [51]. This disturbance in cholesterol metabolism in the brain is under the regulation of a cholesterol transport mechanism. Liver X receptor beta (LXR-β), once activated, promotes multiple genes that regulate reverse cholesterol transport and thus confers neuroprotection [52,53]. For instance, LXR-β agonist enhanced survival of dopaminergic neurons [54] and reduced the burden of mutant huntingtin [55] as well as promoted amyloid β (Aβ) clearance [56]. With the significant evidence of the implication of OS, neuroinflammation, and cholesterol dyshomeostasis in the pathobiology of neurodegenerative disorders, these pathological factors could be targeted for the development of potential therapeutics.
2.2. Ischemic Stroke
Ischemic stroke is responsible for the second-highest number of deaths and disability around the world [57]. It is a pathological condition resulting from sudden occlusion of blood supply to the brain. If the patient survives, the affected brain areas accompany the secondary damage due to the restoration of blood flow and reoxygenation. This ischemia/reperfusion (I/R) event initiates mitochondrial ROS generation [58] and subsequent inflammatory response [59].
Mitochondrial ROS is not only a crucial early driver of acute damage but is also considered an initiator of the consequence of a series of pathological features that develop over time following the reperfusion [60]. Initially, upon reperfusion, the burst of ROS production results in oxidative damage to mitochondria, and thereby disrupts ATP production [61], which ultimately initiates neuronal cell death cascades [62]. ROS-mediated mitochondrial damage further installs the inflammatory response via the activation of microglia and astrocytes as well as an influx of immune cells recruited by cytokines, adhesion molecules, and chemokines across the activated cerebral blood vessels [63]. This activation of the innate immunity triggers nuclear factor-kappa-B (NF-κB)-mediated production of numerous inflammatory cytokines that contribute to I / R injury [64]. Therefore, targeting OS and inflammatory response could be imperative to develop novel therapeutic strategies for the management of stroke.
2.3. Traumatic Brain Injury
Traumatic brain injury (TBI), an acquired brain injury caused by an external force or shock, is also considered to be a major cause of death globally, particularly in countries with a frequent incidence of traffic accidents [65]. Despite significant medical advances in recent times, the clinical outcomes of severely head-injured patients are not satisfactory.
As in ischemic stroke, mechanisms underlying the damages to the brain tissue with TBI are categorized into two classes: primary and secondary damages. Primary damage that irreversibly involves the mechanical damage of the skull and the brain has been complicated following the brain contusions, rupturing blood vessels, axonal injuries, and intracranial hemorrhages [66], whereas the secondary damage causes neuronal degeneration over time due to various biochemical changes such as OS, excitotoxicity, inflammation, and mitochondrial dysfunction [67]. Following TBI, various OS markers such as lipid peroxidation products, oxidized protein moieties, and DNA damage products accumulate in the brain while antioxidants and enzymes molecules such as glutathione (GSH), glutathione peroxidase (GPx), glutathione reductase (GR), glutathione S-transferases (GST), superoxide dismutase (SOD), and catalase (CAT) markedly decline [68]. It is suggested that treatment modalities associated with conferring neuroprotection on injured brain tissue and regeneration at the recovery stage of injured neurons have greater promise to restore at the site of brain injury following TBI.
3. Neuropharmacological Potentials of Marine Algae and Their Metabolites: Evidence from In Vitro Studies
Several compounds of diverse chemical classes have been reported from three major groups (brown, red, and green algae) of marine algae (Figure 1, Figure 2, Figure 3 and Figure 4). Neuropharmacological properties of these compounds reported in various in vitro models are compiled (Table 1) and discussed in the following subsections. Besides bioactive compounds, macroalgae that have shown promising neuroactive potentials, and thus demand further attention are also mentioned.
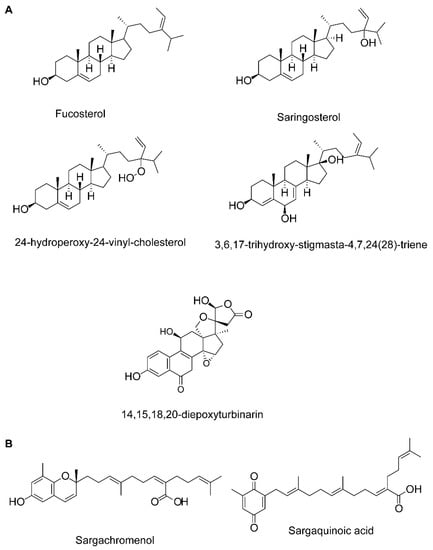
Figure 1.
Chemical structure of sterols (A) and plastoquinones (B) of marine algae.
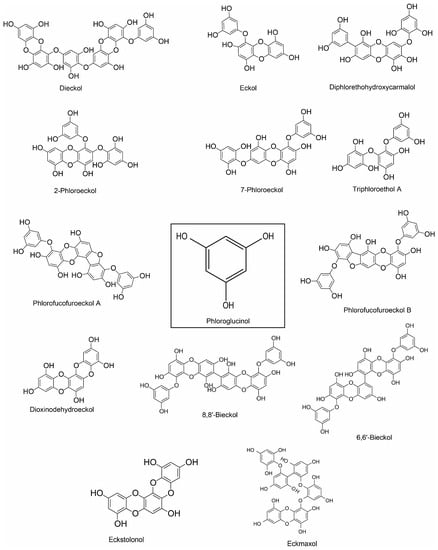
Figure 2.
Chemical structure of phlorotannin of marine algae.
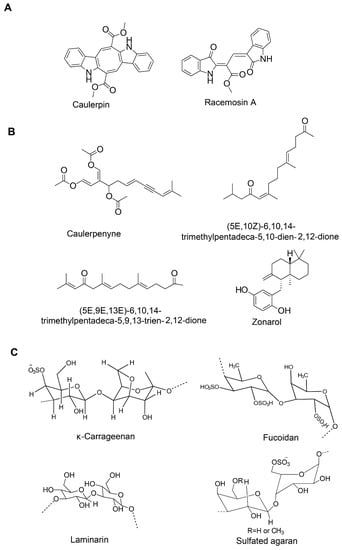
Figure 3.
Chemical structure of alkaloids (A), sesquiterpenes (B) and polysaccharides (C) of marine algae.
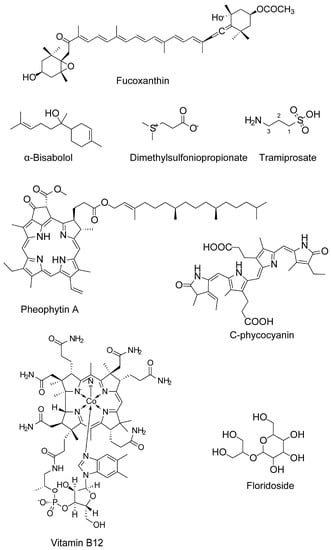
Figure 4.
Chemical structure of miscellaneous compounds from marine algae.

Table 1.
Summary on pharmacological effects, occurrence, effective dose, experimental model, cellular effects, potential pharmacological mechanism of algal metabolites.
3.1. Antioxidant Activity
Marine algae-derived compounds have been reported to exhibit strong antioxidant property (Table 1), and thus may protect against oxidative damage. For example, fucoxanthin, a carotenoid from Sargassum siliquastrum, attenuated OS-induced DNA damage [69]. Fucoxanthin also prevented H2O2-induced DNA damage, which was associated with increased production of GSH, and expression of SOD [36]. Moreover, fucoxanthin promoted antioxidant defense in lipopolysaccharide (LPS)-activated BV-2 microglia by activating nuclear factor erythroid 2-related factor 2 (Nrf2)/heme oxygenase-1 (HO-1) pathway and cell survival through activating cAMP-dependent protein kinase (PKA)/cAMP response element-binding (CREB) pathway and increasing BDNF secretion [70]. Fucosterol raised cellular antioxidant enzymes, such as SOD, GPx, and CAT in experimental rats [71]. Jung and colleagues demonstrated that fucosterol prevented ROS production in tert-butyl hydroperoxide (t-BHP)-induced RAW264.7 macrophages [72]. In addition, fucosterol conferred protection from oxidative damage in HepG2 cells by raising the GSH level [73] and in lung epithelial cells by increasing the expression of SOD, CAT, and HO-1, and nuclear translocation of Nrf2 [74]. Glycoprotein of U. pinnatifida improved SOD activity (53.45%) and inhibited xanthine oxidase (Xox) activity (82.05%) [75]. Diphlorethohydroxycarmalol and 6,6′-bieckol from Ishige okamurae exhibited antioxidant activity and reduced intracellular ROS level in RAW264.7 cells [76]. Sulfated polysaccharide fractions from Porphyra haitanesis showed antioxidant activity and inhibited Lipid peroxidation in rat liver microsome [77]. Porphyran from Porphyra yezoensis showed superoxide anion and hydroxyl radical scavenging activity [78].
In addition, a great number of marine algae have shown antioxidant activity, including Sargassum polycystum and Laurencia obtusa [79], Gelidium foliaceum, and Codium duthieae [80], to mention a few.
3.2. Anti-Inflammatory Activity
An appreciable number of algal compounds have been reported for anti-inflammatory activity (Table 1). Fucoxanthin, a common carotenoid of brown algae, attenuated inflammation, and OS in glial cells [36,70]. In Aβ42-induced BV2 cells, fucoxanthin attenuated inflammatory response, which was manifested by decreased secretion of proinflammatory mediators, such as tumor necrosis factor-alpha (TNF-α), interleukin (IL)-6, IL-1β and prostaglandin E2 (PGE2) and reduced expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2), and by lowering the phosphorylation of mitogen-activated protein kinase (MAPK) pathway [36]. In LPS-activated BV-2 microglia, fucoxanthin protected against neuroinflammation by lowering the expression of iNOS and COX-2 and reducing the secretion of inflammatory factors such as TNF-α, IL-6, PGE2, and nitric oxide (NO) that involved inhibition of protein kinase B (Akt)/NF-κB and MAPKs/ activating protein-1 (AP-1) pathways [70].
The anti-inflammatory activity of fucosterol has recently been reviewed [81]. In brief, fucosterol exhibited anti-inflammatory action [82] and attenuated LPS-induced inflammation in RAW 264.7 macrophage [72]; [83] and alveolar macrophage [84]. Fucosterol also protected against LPS- or Aβ-mediated neuroinflammation in activated microglial cells [85]. Several phlorotannins, such as dieckol [86], phlorofucofuroeckol A [87] and phlorofucofuroeckol B [88], 6,6’-bieckol [89], and 8,8’-bieckol [90] isolated from Ecklonia spp have been reported for their anti-inflammatory activities that involved suppression of NF-κB and MAPK pathways.
Algal polysaccharides are known to act as anti-inflammatory agents [91]. Fucoidan, a sulfated polysaccharide attenuated inflammatory response in LPS-stimulated BV2 microglia by suppressing NF-κB and extracellular signal-regulated kinases (ERK)/MAPK/Akt pathways [92]. In another study, fucoidan decreased the generation ROS and TNF-α in LPS-induced primary microglia [93]. κ-Carrageenan oligosaccharides and its desulfated derivatives from red algae attenuated TNF-α production and showed anti-inflammatory activity in LPS-activated microglia [94]. Porphyran from Porphyra yezoensis attenuated nitric oxide (NO) generation in LPS-stimulated RAW264.7 cells by suppressing iNOS expression [78,95]. Treatment with sulfated oligosaccharides of Ulva lactuca and Enteromorpha prolifera reduced inflammatory factors and downregulated the expression of p53 and fork-head box protein O1 (FOXO1) genes and upregulated the expression of Sirt1 gene in SAMP8 mice [96]. Alginate-derived oligosaccharide inhibited the expression of inflammatory enzymes and secretion of proinflammatory cytokines in LPS/Aβ-induced BV2 microglia. This oligosaccharide also reduced the expression of toll-like receptor 4 (TLR4) and NF-κB [97]. Priming of LPS-stimulated primary microglia and astrocytes with seleno-polymannuronate (Se-PM) reduced the expression of inflammatory enzymes and the production of inflammatory mediators by suppressing NF-κB and MAPK signaling [98]. Sargachromenol isolated from Sargassum micracanthum attenuated inflammatory response in LPS-induced RAW 264.7 macrophages [99]. Kang and colleagues reported that sargaquinoic acid of Sargassum siliquastrum suppressed inflammatory response in LPS-stimulated RAW 264.7 macrophages by downregulating NF-κB and c-JNK pathways [100]. Pretreatment of LPS-stimulated BV-2 microglial cells with floridoside inhibited inflammation by blocking p38/ERK phosphorylation [101]. Glycoprotein from U. pinnatifida (UPGP) reduced the expression of inflammatory enzymes and NO synthesis in LPS-stimulated RAW 264.7 macrophage [75]. Moreover, several algal alkaloids such as caulerpin, racemosin A-C, and caulersin were shown to have anti-inflammatory activity [102].
In addition, several marine algae have been reported to show anti-inflammatory properties in various experimental models, for instance, Ecklonia cava [103], Myagropsis myagroides [104,105], Sargassum serratifolium [106], and three Malaysian seaweeds (Padina australis, Sargassum polycystum, and Caulerpa racemosa) [107] in LPS-stimulated murine BV2 microglia; Ulva conglobata in interferon gamma-induced BV2 cells [108]; Sargassum fulvellum [109], Sargassum horneri [110], Myagropsis myagroides [111,112] in LPS-stimulated RAW 264.7 macrophage cells and Sargassum serratifolium in LPS-stimulated mouse peritoneal macrophages [113]. Owing to their capacity to modulate various inflammatory pathways, these algae and their respective compounds have shown encouraging effects in protecting various cell types from the inflammatory response.
3.3. Anticholinesterase Activity
Currently prescribed anti-AD drugs are mostly based on the inhibition of cholinesterase activity. Several algal metabolites have been reported to inhibit cholinesterase activity (Table 1). For example, fucosterol and 24-hydroperoxy 24-vinylcholesterol isolated from E. stolonifera showed inhibitory activity against butyrylcholinesterase (BChE) [114]. Another study also demonstrated anticholinesterase activity of fucosterol [85]. Enzyme kinetics and computational analysis indicated a non-competitive mode of acetylcholinesterase (AChE) inhibition of fucosterol [115].
Fucoxanthin exhibited anti-BChE activity which was of mixed inhibition type [116], whereas Lin and colleagues demonstrated that fucoxanthin showed non-competitive inhibition against AChE [117]. α-Bisabolol from Padina gymnospora showed inhibition against cholinesterase activity [118]. U. pinnatifida-derived glycoprotein showed AChE and BChE inhibitory activities [75].
The IC50 values for phloroglucinol, dibenzo [1,4] dioxine-2,4,7,9-tetraol and eckol from Ecklonia maxima range from 76.70 to 579.32 μM, with later two compounds possessing the highest AChE inhibitory activity [119]. Dieckol and phlorofucofuroeckol exhibited a similar anti-AChE activity [120]. Sargaquinoic acid and sargachromenol from Sargassum sagamianum have shown reasonable AChE inhibitory activity while the BChE inhibitory activity of sargaquinoic acid is 1000-fold higher than for AChE [121]. Tyrosol and its derivative, 4-(1,2-dihydroxyethyl) phenol from Macrocystis angustifolia showed anti-AChE activity [122]. Meroterpenoids, such as sargahydroquinoic acid, sargachromenol, and sargaquinoic acid of S. serratifolium exhibited potent anti-AChE activity [123]. Among the phlorotannins tested 8,8′-bieckol showed potent anti-AChE activity [124].
In addition, the extracts from some marine algae have shown anti-cholinesterase properties. These include Halimeda cuneata [80], Padina australis [125], Botryococcus braunii and Nannochloropsis oculata [126], Cystoseira tamariscifolia and Cystoseira nodicaulis [127], Ishige foliacea [128], and Asparagopsis taxiformis [129].
3.4. Anti-Amyloidogenic and Aggregation Inhibition Activity
As amyloid-β deposition is one of the hallmarks of AD, compounds that interfere with the generation of pathogenic Aβ and/or that inhibit its aggregation are of therapeutic importance. Several metabolites of marine algae have shown anti-amyloidogenic potentials (Table 1). For example, fucoxanthin at variant concentrations reduced the formation of Aβ1–42 fibril and Aβ1–42 oligomers, when co-incubated with Aβ1–42 monomers [135,136]. Both studies also demonstrated that fucoxanthin has been shown to inhibit Aβ aggregation [135,136]. Inhibition of β-site amyloid precursor protein cleaving enzyme 1 (BACE1) with fucoxanthin was of a mixed-type [134]. In addition, molecular docking analysis revealed a differential pattern of interaction [134]. Fucosterol showed a potential anti-BACE1 activity, which was a noncompetitive type [134]. Supporting these findings, a recent in silico study also explained the binding and interaction pattern of fucosterol with BACE1 [142]. α-Bisabolol from Padina gymnospora prevented oligomers formation as well as disaggregated the matured fibrils [118]. Glycoprotein from U. pinnatifida exhibited anti-BACE1 activities with IC50 values of 73.35 ± 2.54 μg/mL [75]. Meroterpenoids, such as sargahydroquinoic acid, sargachromenol, and sargaquinoic acid of S. serratifolium, exhibited potent anti-BACE1 activity [123]. Phlorotannins, such as eckol, dieckol, and 8,8′-bieckol from Ecklonia cava showed anti-BACE1 activity [124]. Olasehinde et al. reported that four South African macroalgae such as Gracilaria gracilis, Ulva lactuca, Ecklonia maxima, and Gelidium pristoides exhibited anti-cholinesterase, anti-BACE1, and Aβ aggregation inhibitory activities, indicating that these types of seaweed could be potential sources of anti-AD agents [35]. Ishige foliacea extract showed β-secretase inhibition property [128].
3.5. Cholesterol Homeostasis and Aβ Clearance Activity
Some algal metabolites are known to activate LXR-β (Table 1), and thus help regulate cholesterol homeostasis and enhance Aβ clearance [56]. Fucosterol is a selective LXR-β agonist that upregulated several LXR target genes, such as ATP-binding cassette transporter A1 (ABCA1), ABCG1, and apolipoprotein E (ApoE) [138,139], suggesting that fucosterol could play a significant role in brain cholesterol homeostasis. Saringasterol, another selective LXR-β agonist isolated from S. fusiforme, activated the expression of similar LXR target genes in multiple cell lines [139]. Alginate-derived oligosaccharide isolated from marine brown algae promoted the microglial phagocytosis of Aβ, which is connected to the activation of toll-like receptor signaling [97]. As cholesterol imbalance and impaired protein clearance system significantly contribute to the pathogenesis of major neurological disorders, more efforts should, therefore, be paid to explore similar compounds that may help regulate cholesterol homeostasis and proteostasis.
3.6. Monoamine Oxidase Inhibition and Affinity to Dopaminergic Receptors
Inhibition of MAO-A (monoamine oxidase-A), an enzyme that catalyzes oxidative deamination of neuroamines, such as dopamine, norepinephrine, and serotonin (5-HT), is a putative approach to raise the brain 5-HT level, thus alleviating the symptoms of Parkinsonism [143]. Seong and team screened the multi-target effects of three phlorotannins, i.e., phloroglucinol, phlorofucofuroeckol-A (PFF-A), and dieckol against human MAO-A and -B and various neuronal G-protein-coupled receptors (GPCRs). Of these, PFF-A exhibited a relatively higher inhibition against both hMAO isoforms, with greater selectivity toward hMAO-B (Table 1). Enzyme kinetics and computational findings indicated that PFF-A noncompetitively interacted with hMAOs and acted allosterically. In a functional assay for GPCR screening, dieckol and PFF-A showed a multi-target combination of D3R/D4R agonism and D1/5HT1A/NK1 antagonism [140].
3.7. Anti-Aging
Algal compounds that exhibited anti-aging effects (Table 1) could have therapeutic value for physiological as well as pathological brain aging. Sulfated oligosaccharides of Ulva lactuca and Enteromorpha prolifera, when treated in SAMP8 mice, increased the serum level of antioxidant molecules and total antioxidant capacity, and decreased the levels of malondialdehyde (MDA) and advanced glycation end products in the serum of experimental mice [96]. It has also been observed that these oligosaccharides decreased inflammatory factors, increased BDNF and choline acetyltransferase (ChAT) levels, and promoted the survival of hippocampal neurons. The underlying mechanisms involved the downregulation of p53 and FOXO1 genes and the upregulation of Sirt1 gene [96]. Caenorhabditis elegans, when treated with fucosterol (at 50 µg/mL), survived longer compared to control, indicating that this algal compound might help extend life-span and thus might protect against premature aging [141]. Antioxidant, anti-inflammatory, and immunostimulatory properties of fucosterol were supposed to be involved in its pro-survival effect [144].
3.8. Neurotrophic Activity
Compounds with neuritogenic potentials are promising to reconstruct a damaged neuronal network, which is a characteristic feature of neurodegeneration. Several algal metabolites have shown a promising neurite outgrowth promoting potentials in cell culture conditions (Table 2). Sargachromenol from Sargassum macrocarpum promoted nerve growth factor (NGF)-dependent neuronal differentiation of PC12D cells by activating cyclic AMP-mediated protein kinase and MAPK1/2 and supported their survival by activating phosphatidylinositol-3 kinase (PI3K) [145]. Sargaquinoic acid, another metabolite from S. macrocarpum, promoted neuritogenesis in PC12D cells, which involved cooperation between two independent pathways, i.e., the TrkA-MAPK pathway and adenylate cyclase-PKA pathway [146]. Ina and colleagues demonstrated that the neurodifferentiation of PC12 cells by pheophytin a of Sargassum fulvellum required the presence of NGF and involved the activation of an MAPK signaling pathway [147]. Vitamin B12, a chlorophyll-related analog to pheophytin a, also stimulated NGF-dependent PC12 cell differentiation by an MAPK signaling pathway [148].

Table 2.
Neurotrophic activity of algal phytochemicals in vitro.
Dimethylsulfoniopropionate (DMSP) promoted neurite outgrowth and protected against TDA-induced cytotoxicity, involving the upregulation of Hsp32 and activation of the extracellular signal-regulated kinases 1/2 (ERK1/2) [149]. Fucoxanthin has shown to exhibit neurite outgrowth activity (15.7–31% of cells to develop neurite outgrowth) at much lower concentrations (0.1–2 μM), in the absence of NGF support, indicating that this marine carotenoid could a potential neurotrophic molecule [136]. Gracilariopsis chorda and its active compound arachidonic acid modulated spine dynamics, and potentiated functional synaptic plasticity of hippocampal neurons [150].
In addition, several marine algae have shown to promote neurite outgrowth. For example, Sargassum macrocarpum and Jania adharens showed neuritogenic potentials and promoted neuron-specific dendrites and axons from PC12D cells [151]. Two compounds, namely sargachromenol [145] and sargaquinoic acid [148], having neurite outgrowth potential were already isolated. Porphyra yezoensis and its compound taurine facilitated neuronal development and maturation of primary hippocampal neurons [152]. Gelidium amansii [153,154,155,156], Sargassum fulvellum [157], Undaria pinnatifida and Saccharina japonica [158], Gracilariopsis chorda [150,159], and carrageenophyte Kappaphycus alvarezii [160,161,162,163] promoted neuronal morphology and functions. Of these, G. amansii that exhibited neuromodulatory potentials in several studies [153,154,155,156] could be the most promising candidate for further isolation of neurotrophic agents and thus expects special attention of natural product chemists.
3.9. Neuroprotective Activity
Compounds that possess antioxidant, anti-inflammatory, anti-amyloidogenic, and anti-aggregation, cholesterol homeostasis, and protein clearance activities are expected to show potential neuroprotective effects. Congruently, the following metabolites isolated from marine algae have been reported to confer neuroprotection against a range of toxic stimuli (Table 3).

Table 3.
Neuroprotective activity of algal compounds in vitro and in vivo.
Several studies reported the neuroprotective activity of fucoxanthin. For example, fucoxanthin attenuated β-amyloid oligomer-induced [164] and H2O2-induced [165] apoptosis and OS in SH-SY5Y cells through activating a pro-survival PI3K/Akt pathway and suppressing the proapoptotic ERK pathway. Fucoxanthin-mediated protection against H2O2-induced apoptosis in primary cerebellar granule neurons also involved a similar neuroprotective mechanism [165]. Co-incubation of fucoxanthin with Aβ1–42 oligomers formed modified Aβ1–42 oligomers, which were relatively less toxic to SH-SY5Y cells compared to Aβ1–42 oligomers, indicating that fucoxanthin-triggered structural modification of Aβ1–42 oligomers reduced their neurotoxicity [135]. Fucoxanthin, isolated from Undaria pinnatifida, also attenuated hypoxia/reoxygenation (H/R)-induced cellular injury in primary cortical [166] and hippocampal neurons [167]. Likewise, fucoxanthin suppressed oxygen-glucose deprivation/ reperfusion (OGD/R)-induced neuronal apoptosis, via activating the Nrf2/HO-1 signaling [168]. In the TBI model of mouse primary cortical neurons, fucoxanthin promoted neuronal survival against secondary injury and enhanced antioxidant enzymes such as HO-1 and NAD(P)H dehydrogenase [quinone] 1 (NQO-1) via activating Nrf2-ARE and Nrf2-autophagy pathways [169]. Fucoxanthin also attenuated both Aβ1-42- and H2O2-induced toxicity in PC12 cells [136].
Zonarol (ZO), a para-hydroquinone-type molecule from Dictyopteris undulata protected against OS in HT22 hippocampal and cerebrocortical neurons by activating the Nrf2/ARE pathway [170]. It induced the expression of NQO-1, HO-1, and peroxiredoxin 4 (PRDX4) and thus helps regulate intracellular redox state [170]. α-Bisabolol, an active compound of Padina gymnospora, protected against Aβ25-35-induced neurotoxicity in PC12 cells [137] and also in Neuro2a cells and transgenic C. elegans [171]. In PC12 cells, the rescuing effects of α-bisabolol against Aβ induced neurotoxicity were similar to donepezil, which is a currently prescribed anti-AD drug [137]. In Neuro2a cells, α-bisabolol exhibited inhibition against cholinesterase and β-secretase activity. In addition, α-bisabolol prevented apoptosis in Neuro2a cells by inhibiting the production of ROS and reactive nitrogen species (RNS) and reducing the expression of bcl-2-like protein (Bax) and caspase-3 [171]. In a transgenic C. elegans Alzheimer’s model, α-bisabolol attenuated Aβ-induced proteotoxicity by decreasing the expression of angiotensin-converting enzyme 1 (ace-1), hsp-4, and Aβ [171]. The neuroprotective roles of fucosterol have been reviewed in our recent article [81]. In brief, fucosterol attenuated Aβ-induced neurotoxicity in hippocampal neurons [172] and SH-SY5Y cells [173]. In addition, three isolated compounds including α-tocospirone, (23E)-3β-hydroxy-stigmasta-5,23-dien-28-one and (22E)-3β-hydroxy-cholesta-5,22-dien-24-one from Caulerpa racemose attenuated Aβ25-35-induced toxicity in SHSY5Y cells [174].
Phlorotannins, a specialized group of tannins, particularly rich in brown algae, have shown significant neuroprotective effects in several neurotoxicity models. Liu and colleagues evaluated three phlorotannins, including 8,8’-bieckol, dieckol, and eckol for their neuroprotection against Aβ25-35-mediated cytotoxicity in PC12 cells [96]. Of these, dieckol showed maximum protection, although all were shown to suppress inflammatory response by inactivating the NF-κB pathway [96]. A similar study by Ahn and teams demonstrated that six phlorotannins, such as phloroglucinol, dioxinodehydroeckol, eckol, dieckol, phlorofucofuroeckol A, and 7-phloroeckol from Eisenia bicyclis protected against Aβ-induced cytotoxicity by inhibiting ROS generation and Ca2+ release [175]. Dieckol attenuated glutamate-induced excitotoxicity in primary cortical neurons and HT22 neurons by scavenging ROS and nuclear factor-like 2/heme oxygenase-1 pathway [176]. In addition, in another study, phloroglucinol from E. cava suppressed Aβ1-42 -provoked ROS accumulation in an HT-22 hippocampal cell line [177].
Phloroglucinol also rescued the Aβ1-42-induced reduction of dendritic spine density and synaptic protein (synaptophysin and postsynaptic density) levels in primary cultures of rat hippocampal neuronal [177]. Kang and co-investigators isolated five phlorotannins, such as phloroglucinol, eckol, triphloroethol A, eckstolonol, and dieckol from E. cava that attenuated H2O2-induced oxidative damage in HT22 hippocampus neurons by lowering ROS production, lipid peroxidation and Ca2+ release [178]. Phlorofucofuroeckol attenuated glutamate-induced cytotoxicity and improved mitochondrial dysfunction in PC12 cells [180]. Preconditioned HT22 hippocampal neurons with diphlorethohydroxycarmalol (DPHC), a phlorotannin of Ishige okamurae, was able to escape H2O2-induced oxidative damage due to antiapoptotic, pro-survival, and antioxidant potentials of DPHC [179]. Eckmaxol, a phlorotannin of Ecklonia maxima, reduced Aβ-oligomer-induced neuronal apoptosis in SH-SY5Y cells by inhibiting GSK-3β and ERK pathways [181,203].
Several studies have confirmed the neuroprotective capacity of algal polysaccharides, including fucoidan [204] and carrageenan. Fucoidan, a sulfated polysaccharide, attenuated Aβ1−42-induced neurotoxicity in rat cholinergic basal forebrain neurons [182]. It restored Aβ-induced decline in whole-cell currents, increased phosphorylation of protein kinase C (PKC), and showed antioxidant and anti-apoptotic effects [182]. Fucoidan protected H2O2-induced cell death in PC-12 cells by activating the PI3K/Akt signaling pathway. The antioxidant, antiapoptotic, and prosurvival effects of fucoidan could explain its neuroprotection capacity [184]. Fucoidan protected against Aβ25-35 and d-Gal-induced neurotoxicity in PC12 cells by reducing OS, suppressing apoptosis pathway, and promoting antioxidant defense [185]. Wu and colleagues reported that fucoidan suppressed intracellular Ca2+ responses by selective inhibition of N-methyl-D-aspartate (NMDA) receptors in cortical neurons and L-type Ca2+ channels in hippocampal neurons [187]. Three fucoidan extracts from Sargassum crassifolium attenuated H2O2-induced cytotoxicity in rat pheochromocytoma PC-12 cells [205]. In the MPP+ PD model, fucoidan attenuated cytotoxicity in a dopaminergic neuronal precursor cell line (MN9D) [183,186] by protecting lysosomes, reducing the expression of light chain 3-II (LC3-II), inhibiting the expression of cathepsin D (CatD)-Bax and the OS response [186]. Fucoidan of Sargassum hemiphyllum attenuated 6-hydroxydopamine-induced apoptosis in SH-SY5Y cells [206]. The acidic oligosaccharide sugar chain attenuated Aβ-stimulated astrocytes conditioned medium-induced cytotoxicity in SH-SY5Y cells by mitigating oxidative damage, reducing inflammatory response, and preventing Ca2+ influx [189]. In addition, κ-carrageenan-derived pentasaccharide (KCP) protected against Aβ25-35-induced neurotoxicity in SH-SY5Y cells by regulating the c-Jun N-terminal kinase (JNK) signaling pathway [192]. Moreover, κ-carrageenan from Hypnea musciformis attenuated 6-hydroxydopamine-induced neurotoxicity on SH-SY5Y cells by modulation of the mitochondria transmembrane potential and reducing caspase 3 activity [207]. Oligo-porphyran (OP), an acid hydrolytic product of porphyran (a polysaccharide of Pyropia yezoensis) attenuated 6-OHDA-induced cytotoxicity in PC12 cells by activating PI3K/ Akt/PKC pathway that involved anti-apoptotic, antioxidant and anti-inflammatory signals [188].
Sargaquinoic acid identified from Sargassum macrocarpum promoted cell survival and neurite regeneration and attenuated H2O2-induced OS in PC12D cells [208]. Racemosin A, a bisindole alkaloid from Caulerpa racemose, attenuated Aβ25-35-induced damage in SH-SY5Y cells [190]. Tramiprosate, a small aminosulphonate compound of red marine algae, attenuated OGD- or NMDA-induced injury in PC12 cells and primary cortical neurons [191] by disrupting the interaction between PSD95 and nNOS and inhibition of nNOS translocation [191]. Potentials of tramiprosate against AD and PD have also been reviewed elsewhere [28,209,210]. Dimethylsulfoniopropionate protected against tropodithietic acid-induced cytotoxicity in OLN-93 and N2a cells by lowering the activation of ERK1/2 and induction of HSP32 [149]. Phycoerythrin-derived peptide isolated from Pyropia yezoensis promoted survivability of frontal cortical neuron by activating TrkB receptor-ERK1/2 signaling and attenuating ER stress in rat prefrontal cortex [211] and attenuated glutamate-induced ER stress and senescence of rat primary hippocampal neurons [212]. Stearic acid from Caulerpa racemosa protected against OGD-induced SH-SY5Y cell damage while (8E)-heptadec-8-en-7-one showed moderate neuroprotective activity against Aβ25-35-induced SHSY5Y cell damage [213].
In addition, extracts from several marine algae have shown neuroprotective activity in various in vitro models. The neuroprotective algae include Ulva conglobata that protected against glutamate-induced neurotoxicity in murine hippocampal HT22 cell line [108], Botryococcus braunii, and Nannochloropsis oculata against H2O2-induced cytotoxicity in dopaminergic SH-SY5Y cells [126], Padina pavonica, Sargassum muticum, Saccorhiza polyschides, Codium tomentosum, and Ulva compressa [214], and Bifurcaria bifurcata [215] against 6-hydroxidopamine-induced cytotoxicity in neuroblastoma cells, Cystoseira tamariscifolia and Cystoseira nodicaulis against H2O2-induced cytotoxicity in SH-SY5Y cells [127], Gracilaria corticata against aluminium-induced neurotoxicity in the hippocampus, and cerebral cortexes of rat brains [216], Australian macroalgae against Aβ 1-42-induced neurotoxicity in PC-12 cells [217], Ishige foliacea against H2O2- or Aβ-induced cell death in human neuroblastoma SH-SY5Y cells [128], Undaria pinnatifida against endoplasmic reticulum stress in hypothalamic neurons [218] and Gracilariopsis corda [219] and Gelidium amansii [153] against H/R-induced oxidative damage in primary hippocampal neurons, indicating that these algae could offer some potential compounds with encouraging neuroprotective activity, and, therefore, demand further investigation.
4. Neuropharmacological Potentials of Marine Algae and Their Metabolites: Evidence from In Vivo Studies
The neuroprotective effects of some potential algal compounds that were reported in the in vitro conditions have successfully been translated into animal models (Table 3), suggesting that these compounds could be potential candidates for further evaluation in the clinical trials.
Fucoidan is one of the algal compounds that has shown strong neuroprotection in several animal models. In the PD model of C57 / BL mice, fucoidan ameliorated MPTP-induced behavioral deficits, probably by elevating dopamine and its metabolite levels and increasing tyrosine hydroxylase expression [183]. In addition, fucoidan inhibited MPTP-induced lipid peroxidation and restored antioxidant capacity [183]. Similarly, fucoidan also improved behavioral capacity, by attenuating the loss of dopaminergic neurons and inhibited the deleterious activation of microglia in the substantia nigra pars compacta in LPS-induced neurotoxicity [93]. In an Aβ-induced rodent AD model, fucoidan ameliorated impaired memory, by reversing the decreased activity of ChAT, SOD, and GPx, increased activity of AChE, and rectifying the imbalance between apoptosis and pro-survival signals [193]. Fucoidan improved d-Gal-induced cognitive impairment in mice by mitigating OS and attenuating the caspase-dependent apoptosis pathway [185]. Wang and colleagues demonstrated that the supplementation of fucoidan alleviated Aβ-induced paralyzed phenotype in a transgenic C. elegans AD model [194]. Fucoidan reduced Aβ accumulation, probably by promoting proteasomal activity [194]. In another study, fucoidan-rich substances from Ecklonia cava improved trimethyltin-induced cognitive dysfunction by inhibiting Aβ production and Tau hyperphosphorylation [195]. Fucoidan also attenuated transient global cerebral ischemic injury in the gerbil hippocampal CA1 area through mitigating glial activation and oxidative stress [196].
Laminarin, another polysaccharide of brown algae, has shown to protect I/R injury in gerbil models. Intraperitoneal injection of laminarin (50 mg/kg) following 5 min I/R attenuated reactive gliosis (anti-inflammatory) in the hippocampal CA1 of young gerbils [197]. A similar study following the same experimental protocol, but with aged gerbils, showed that laminarin (50 mg/kg) attenuated ischemia-induced death of pyramidal neurons in the hippocampal CA1 of aged gerbils [198]. This neuroprotective effect of laminarin is attributed to its antioxidant and anti-inflammatory properties [198]. Oligo-porphyran, a synthetic product of porphyran (Pyropia yezoensis) ameliorated behavioral deficits in 6-OHDA-induced Parkinsonian mice model by protecting dopaminergic loss and activating the PI3K/Akt/Bcl-2 pathway that involved cellular signaling of anti-apoptosis and antioxidation [199]. Zhang and colleagues demonstrated that porphyran from Pyropia haitanensis improved the Aβ1-40-induced learning and memory deficits probably by elevating cerebral acetylcholine level [200].
Fucoxanthin is another significant algal metabolite that was found to be effective in a wide range of brain dysfunction (such as AD, ischemic stroke, and traumatic brain injury). Fucoxanthin ameliorated scopolamine-induced [135] and Aβ oligomer-induced [117] cognitive impairments in mice, possibly by inhibiting AChE activity and OS, modulating ChAT activity, and increasing BDNF expression. Fucoxanthin alleviated cerebral ischemic/reperfusion (I/R) injury, improved the neurologic deficit score, and downregulated the expression of apoptosis-linked proteins in brain samples [168]. Fucoxanthin also attenuated traumatic brain injury that involved the Nrf2-ARE and Nrf2-autophagy pathways-dependent neuroprotective mechanism [169].
Fucosterol co-infusion ameliorated sAβ1-42-induced cognitive deficits in aging rats by modulating BDNF signaling [172]. Dieckol and phlorofucofuroeckol raised the brain level of acetylcholine by inhibiting AChE and reduced the inhibition of latency in ethanol-intoxicated memory-impaired mice [120]. Yang and co-investigators demonstrated that stereotaxic injection of phloroglucinol promoted synaptic plasticity and improved memory impairment in 5XFAD (Tg6799) mice [177]. In a later study, the same group reported phloroglucinol (orally administered)-mediated amelioration of cognitive dysfunction that involved a reduction in the amyloid β peptide burden and pro-inflammatory mediators and restoration of reduction in the dendritic spine density in the hippocampus of 5XFAD mice [220]. Phlorofucofuroeckol improved ischemic brain damage in the rat MCAO model [180]. C-Phycocyanin improved the functional outcome and survival of gerbils on global cerebral I/R injury [201]. The in vitro neuroprotective effect of tramiprosate has been translated into in MCAO rat model in which it improved functional recovery following ischemic stroke [191]. Sulfated agaran, a sulfated polysaccharide from Gracilaria cornea, attenuated oxidative/nitrosative stress and ameliorates behavioral deficits in rat 6-hydroxydopamine Parkinson’s disease model [202]. It raised levels of dopamine, 3,4-Dihydroxyphenylacetic acid (DOPAC), GSH, and BDNF, decreased serotonin (5-HT) and thiobarbituric acid reactive substances (TBARS) levels, and decreased the expression of p65, IL-1β, and iNOS [202]. Glycoproteins isolated from Capsosiphon fulvescens ameliorated aging-induced spatial memory deficits by attenuating GSK-3β-mediated ER stress in rat dorsal hippocampus [221] and promoted probiotics-induced cognitive improvement in aged rat model [222]. Gracilariopsis chorda and its active compound arachidonic acid, given independently through oral route for 10 days, improved scopolamine-induced memory impairment in mice [150].
In addition, extracts from several marine algae have shown to either ameliorate memory impairment or enhance cognition in various in vivo models. For instance, Gelidiella acerosa attenuated Aβ25-35-induced cytotoxicity and memory deficits in mice [223], Sargassum swartzii improved memory functions in rats [224], Ishige foliacea [128], Undaria pinnatifida [225] ameliorated scopolamine-induced memory deficits in mice, Haematococcus pluvialis recovered Alzheimer’s disease in rats [226], and fermented Spirulina maxima prevented memory impairment in mice [227]. In addition, some marine algae have shown to attenuate ischemic injury in stroke models. For example, Ecklonia cava ameliorated transient focal ischemia in the rat MCAO model [228].
5. Recent Progress on the Development of Marine Algae-Based Neurotherapeutics
An algal oligosaccharide, sodium oligomannate, recently received conditional approval in China for improving cognitive function in patients with mild to moderate AD [32]. In preclinical studies, sodium oligomannate conferred neuroprotection against Aβ-induced neurotoxicity in human neuroblastoma cells [229] and ameliorated memory dysfunction in the 5XFAD transgenic mouse model [230]. Sodium oligomannate can cross the blood–brain barrier through glucose transporter (GLUT1) and inhibits Aβ fibril formation and destabilizes the preformed fibrils into nontoxic monomers [230]. Although the complete mechanism of pharmacological actions remains unclear, sodium oligomannate harnessed neuroinflammation and thus ameliorated memory impairment by suppressing gut dysbiosis and the associated phenylalanine/isoleucine accumulation [230]. In a phase IIa pilot study in patients with AD, there was an elevation of Aβ1–42 levels in the cerebrospinal fluid (CSF) following sodium oligomannate treatment, suggesting a significant role in Aβ clearance into CSF [231]. There was a differential reduction in the cerebral glucose metabolic rate (CMRglu) in various brain regions following sodium oligomannate in clinical trials [231]. While in a phase IIa trial, the CMRglu in left orbitofrontal gyrus, left precuneus, right posterior cingulate gyrus, and right hippocampus were found to be low, in a phase III trial, the lower rate was reported in superior parietal gyrus, inferior parietal gyrus, angular gyrus, and anterior wedge [232]. However, this newly approved drug lacks some advanced information like global data of effectivity and thus requires a large-scale global trial before it receives approval from the Food and Drug Administration (FDA).
6. Algal Metabolites-Based Drug Discovery and Design
While a significant quantity of active compounds has been isolated from marine algae and added to the compound databases [233,234,235,236,237,238] every year, it is disappointing that very few of them have access to clinical trial and the success rate is also not very satisfactory. In this context, the current strategy of drug development requires a reformation with the inclusion of some modern approaches, such as virtual screening and network pharmacology. The system biology approach along with an in silico study constitutes a potential computation tool that can better explain how a biologically effective compound interacts with the signal molecules of various cellular pathways.
Recent multitarget drugs have been designed by analyzing the 3D structure of already characterized compounds and crystal structure of target protein molecules. This information is focused on the virtual design of new chemical entities that include more than one biological function in a single molecule [239]. This approach is also known as target fishing, which identifies not only interacting proteins but also potential off-targets, and thus helps to understand polypharmacology, pharmacokinetics, and toxicity in the early stages of drug discovery [240]. For example, using in silico target fishing approach, Hannan and colleagues elucidated pharmacological mechanism of fucosterol-mediated neuroprotection and demonstrated that fucosterol showed interaction with potential targets, including LXR, TrkB, GR, Toll-like receptor (TLR) 2/4, and BACE1 [142]. Computational methods involving target screening are classified based on their principle including pharmacophore screening, shape screening, and reverse docking. When the target is available in the crystal structure, target fishing can be accomplished by a reverse docking approach, while, in the target’s absence, pharmacophore or shape screening can be used to find the relevant target by comparing pharmacophoric feature or shape of the compound, taking information from protein–ligand binding databases [241]. In this effort, several natural product databases containing compound target interactomes are available nowadays including, SuperNatural [242], TCMID [243], TCMSP [244], and many others [245,246]; however, not many are dedicated to marine algae [233,234,235]. Although algal metabolites show structural diversity and redundancy, the mentioned databases could still be available for network pharmacology to get insight into the disease-modifying mechanisms. Following this in silico approach, Vitale et al. identified caulerpin as a PPAR agonist which was confirmed by both in vitro and in vivo assays [247]. In a reverse way, virtual screening through molecular docking analysis could be an alternative to find out potent hits from a large chemical library for a single target.
Compared to experimental high throughput screening, virtual screening, either by ligand or structure-based approach, can deliver the shorten cycle of hit discovery, with higher success hit rates. Furthermore, a structure-based approach consisting of molecular docking, receptor-based pharmacophore modeling together with molecular dynamics simulations and MM/PB(GB)SA approaches not only predict protein–ligand interaction but also provide a detailed binding mechanism, protein dynamics, and also highlight structure–activity relationship (SAR) for future drug design [248]. Several recent studies have been adopting molecular docking techniques to analyze detailed protein–ligand interaction for marine bioactive compounds. For example, Jung et al. employed molecular docking studies to predict comparative binding interaction of monoamine oxidase (MAO) with fucoxanthin, a carotenoid from Eisenia bicyclis, where they revealed fucoxanthin as a reversible competitive hMAO inhibitor, binding strongly to the enzyme, following hydrogen bonding and hydrophobic interactions [249]. A similar approach has been applied to elucidate the interaction of fucosterol and fucoxanthin with BACE1 while analyzing BACE1 enzyme inhibition by fucosterol and fucoxanthin. Here, binding interaction analysis by molecular docking identified that the presence of hydroxyl group in fucosterol and fucoxanthin is important for BACE1 inhibition, by which both compounds interacted with Lys224 residue, Gly11, and Ala127 of the active site, respectively [134]. Interestingly, fucoxanthin was also identified as a dopamine agonist, where a molecular docking study suggested that it formed H-bonding with Ser196 and Asp115 of the D4 receptor, and Ser196 and Thr115 residues of D3 receptors [250]. The same group also identified some bromophenols derivatives as D3R and hD4R antagonists and studied the interaction and binding pattern by molecular docking [251].
In addition, several studies employed virtual screening to identify potent lead molecules from the database of seaweed metabolites. For instance, Florest et al. identified sigma-2 (σ2) receptor binding ligand by using both structure and ligand-based screening [252]. However, less effort has been deployed to develop marine natural product libraries, although significant studies so far have reported many compounds isolated from marine sources by large populations in the world. In this exertion, Davis et al. developed a chemical library of the natural compounds from marine algae, SWMD, comprised of 1110 metabolites, isolated from brown algae (266), green algae (33), and red algae (811) along with their physical and chemical properties [233]. Nevertheless, the information including experimentally-determined quantitative activity data and source information for more marine algal metabolites is still needed to facilitate computational based approaches in the exploration of marine compounds for future drug discovery.
7. Safety Issues on Marine Algae-Derived Compounds
As a popular food material in East Asian countries, including Japan, Korea and China, seaweed is consumed without reported toxicity. However, the concern is that seaweed may sometimes accumulate a considerable amount of heavy metals, such as cadmium, arsenic, mercury, and lead, and even some essential microelements such as iodine and iron [253]. It is, therefore, essential to conduct appropriate safety evaluations for seaweed. More importantly, while there are safety concerns during therapeutic development, the toxicity profile of seaweed-derived compounds needs to be thoroughly investigated. Safety information on algal metabolites is limited. However, toxicity profiles of algal polysaccharides have been reported by several studies. Observations from both in vitro and in vivo studies satisfied the non-toxic behavior of fucoidan irrespective of algal sources [254]. Fucoidan isolated from Undaria pinnatifida and Laminaria japonica was found to be safe in animal models given at very high oral doses [255,256,257,258]. Clinical studies also demonstrated the non-toxic health benefits of fucoidan in humans [259,260]. Safety evaluation studies on carrageenan suggest that sub-chronic or chronic feeding of this food-grade polysaccharide did not induce any toxic effects [261]. Moreover, dietary supplementation of carrageenan was not associated with carcinogenicity, genotoxicity, or reproductive defects [261]. Another study reported that no toxicological response was induced when iota-carrageenan was administered through the intranasal route [262]. Several studies also investigated toxicity of fucoxanthin and suggested that this carotenoid was safe and caused no visible toxicity in experimental subjects [263,264,265]. The toxicity profiles of some other marine metabolites have recently been reviewed [25]. As sufficient toxicological profiles of other potentially bioactive metabolites are lacking, they should be investigated with appropriate experimental models.
8. Conclusions and Future Perspectives
The current review highlights several neuropharmacological attributes, such as antioxidant, anti-inflammatory, anti-cholinesterase, anti-amyloidogenic, antiaging, protein clearance, cholesterol homeostasis, and neuritogenic capacity of algae-derived metabolites that underlie their neuroprotective functions against a wide range of neurotoxic stimuli (Figure 5). The neuroprotective effects of marine algae and their metabolites do not necessarily depend on a single attribute, rather on the synergism of multiple of these pharmacological properties. As neurodegenerative disorders involve complex pathogenic mechanisms, they could be better managed with a single compound targeting two or more of the pathogenic mechanisms or multiple compounds with the complementary mechanism of action. In this context, algal compounds, such as fucoxanthin, fucosterol, and fucoidan that are known to target multiple pathogenic mechanisms could be potential candidates for future drug development. In addition, several metabolites, including laminarin, porphyran, saringasterol, α-bisabolol, and phlorotannins that exhibited encouraging neuroprotective roles, also deserve further attention.
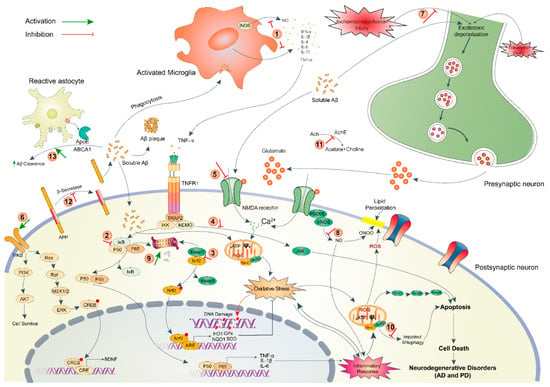
Figure 5.
A scheme highlighting the pathophysiology of neurodegenerative disorders and post-ischemic consequences along with indicating the underlying mechanism of neuroprotective action of algal compounds. The numeric symbols indicate the points of pharmacological action that include (1) inhibition of cytokine secretion from activated microglia by fucoxanthin, fucosterol, fucoidan, dieckol, phlorofucofuroeckol and bieckol, κ-carrageenan, floridoside and seleno-polymannarate, (2) attenuation of inflammatory response via inhibition of NF-κB pathway by eckol, dieckol and 8,8-bieckol, (3) priming of antioxidant defense system via activation of Nrf2/ARE pathway (blocking interaction between Nrf2 and Keap1) by fucoxanthin, fucoidan and zonarol, (4) Reduction of apoptosis via inhibiting pro-apoptotic JNK/Erk pathway by dimethylsulfoniopropionate and κ-carrageenan-derived pentasaccharide, (5) Inhibition of glutamate-induced Ca2+ influx via blocking extrasynaptic GluN2B by fucoidan and tramiprosate, (6) Activation of BDNF-dependent pro-survival pathway via inducing PI3K/Akt or TrkB/ERK signaling by fucoxanthin and fucosterol, (7) Attenuation of I/R-injury via preventing excitotoxic depolarization by C-phycocyanin, (8) Inhibition of nNOS sequestration by tramiprosate, (9) proteasomal degradation by fucoidan, (10) Induction of autophagy/mitophagy by fucoxanthin, (11) anticholinesterase activity by fucoidan, fucoxanthin, dieckol and phlorofucofuroeckol, (12) anti-amyloidogenesis via blocking β-secretase activity by fucoxanthin, fucosterol and glycoprotein, and (13) Aβ-clearance via enhancing the transcription of ApoE and ABC transporters genes by fucosterol, saringasterol, and alginate-derived oligosaccharide. NF-κB/p50-pp65, nuclear factor kappa-light-chain-enhancer of activated B cells; Nrf2, nuclear factor erythroid 2-related factor 2; ARE, antioxidant response element; IkB, inhibitor of NF-κB; Keap1, Kelch-like ECH-associated protein 1; JNK, c-Jun N-terminal kinases; GluN2B, N-methyl D-aspartate receptor subtype 2B; PI3K, phosphoinositide 3-kinases; Akt, protein kinase B; MEK1/2, mitogen-activated protein kinase kinase; ERK, extracellular signal-regulated kinases; TrkB, tropomyosin receptor kinase B; CREB, cAMP-response element binding protein; CRE, cAMP response elements; BDNF, Brain-derived neurotrophic factor; AChE, acetylcholinesterase; Ach, acetylcholine; ABCA1, ATP-binding cassette transporter A1; nNOS, neuronal nitric oxide synthase; ROS, reactive oxygen species; ψ, mitochondrial membrane potential.
Although neuroactive compounds were isolated from a range of algae, seaweed species under Phaeophyceae yield the highest number of compounds. However, species from other groups, for example, Gelidium amansii under Rhodophyceae that exhibited significant neuromodulatory effects, also could offer some promising metabolites. Moreover, a large number of species remain unexplored. While degenerating brains experience disruption of synaptic connectivity, compounds with neuritogenic capacity may potentially enhance the regeneration of damaged processes. Therefore, compounds, both neuroprotective and neurotrophic, are equally important. However, in contrast to neuroprotective compounds that potentially support neuronal survival, a few compounds showing neurite outgrowth potential have been discovered in marine algae. Compounds, including those that have already shown neuroprotective ability as well as those that have not yet been explored, therefore, need to be screened for their ability to promote neurite extension.
Despite a sizable collection of algae-based natural products with distinct neuroprotective functions, only sodium oligomannate has emerged as a successful drug for AD. This review, therefore, calls for intensive research on other potential compounds to translate the preclinical findings into clinical models. In addition, the factors that are responsible for the failure of a clinical trial need to be carefully reviewed. For example, the bioavailability of a candidate drug in the brain, including its ability to cross BBB, remains one of the barriers to therapeutic success. If the ADME (absorption, distribution, metabolism, and excretion) properties of a preclinically effective compound sufficiently guarantee its drug-likeliness, it is highly likely that the compound may succeed in clinical trials. This is why the ongoing strategy requires a rational reformation incorporating modern approaches, such as virtual screening and system biology, to strengthen the algae-based drug development process. The computational study will provide some crucial information on the ADME properties of potential leads and its interaction and binding affinity to molecular targets while system biology knowledge will identify the potential interaction of target molecules and cellular signaling pathways at the systemic level. With the constant discovery of new compounds, all these strategies will accelerate the designing and development of algae-based future drugs.
Author Contributions
Conceptualization, M.A.H. and I.S.M.; Resources, M.A.H., R.D., A.A.M.S., M.N.H., M.M., M.A.R., M.A.; Writing—Original Draft Preparation, M.A.H., R.D., A.A.M.S., M.N.H., M.M., M.A.R.; Writing—Review and Editing, M.A.H., A.A.M.S., M.N.H., M.M., M.J.U.; Visualization, M.A.H., R.D. and M.A.; Supervision, I.S.M. All authors have read and agreed to the published version of the manuscript.
Funding
Our research and publications have been supported by the Basic Science Research Program (#2018R1A2B6002232 to I.S.M.) through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning. M.A.H. and M.A.R. wish to acknowledge the NRF for Korea Research Fellowship (#2018H1D3A1A01074712 to M.A.H. and #2016H1D3A1908615 to M.A.R) funded by the Ministry of Science, ICT and Future Planning. M.J.U. is supported by the RP-Grant 2020 of Ewha Womans University, Republic of Korea
Acknowledgments
This article is dedicated to Yong-Ki Hong, Professor of Biotechnology, Pukyong National University, Korea on the occasion of his retirement. Yong-Ki Hong has spent most of his academic and research career working on marine biotechnology, including the enrichment of macroalgae-based functional metabolites and the development of value-added products.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chi, H.; Chang, H.Y.; Sang, T.K. Neuronal Cell Death Mechanisms in Major Neurodegenerative Diseases. Int. J. Mol. Sci. 2018, 19, 3082. [Google Scholar] [CrossRef]
- Vasili, E.; Dominguez-Meijide, A.; Outeiro, T.F. Spreading of α-Synuclein and Tau: A Systematic Comparison of the Mechanisms Involved. Front. Mol. Neurosci. 2019, 12, 107. [Google Scholar] [CrossRef]
- Zhong, X.; Wang, J.; Carlsson, C.; Okonkwo, O.; Zetterberg, H.; Li, L. A Strategy for Discovery and Verification of Candidate Biomarkers in Cerebrospinal Fluid of Preclinical Alzheimer’s Disease. Front. Mol. Neurosci. 2018, 11, 483. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, G.; Chakrabarti, S.; Chatterjee, U.; Saso, L. Proteinopathy, oxidative stress and mitochondrial dysfunction: Cross talk in Alzheimer’s disease and Parkinson’s disease. Drug Des. Dev. Ther. 2017, 11, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Verri, M.; Pastoris, O.; Dossena, M.; Aquilani, R.; Guerriero, F.; Cuzzoni, G.; Venturini, L.; Ricevuti, G.; Bongiorno, A.I. Mitochondrial Alterations, Oxidative Stress and Neuroinflammation in Alzheimer’s Disease. Int. J. Immunopathol. Pharmacol. 2012, 25, 345–353. [Google Scholar] [CrossRef]
- Jayaraj, R.L.; Azimullah, S.; Beiram, R.; Jalal, F.Y.; Rosenberg, G.A. Neuroinflammation: Friend and foe for ischemic stroke. J. Neuroinflamm. 2019, 16, 142. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.T. Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol. Res. 2017, 39, 73–82. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef] [PubMed]
- Leon, R.; Garcia, A.G.; Marco-Contelles, J. Recent advances in the multitarget-directed ligands approach for the treatment of Alzheimer’s disease. Med. Res. Rev. 2013, 33, 139–189. [Google Scholar] [CrossRef]
- Angeloni, C.; Vauzour, D. Natural Products and Neuroprotection. Int. J. Mol. Sci. 2019, 20, 5570. [Google Scholar] [CrossRef] [PubMed]
- Rehman, M.U.; Wali, A.F.; Ahmad, A.; Shakeel, S.; Rasool, S.; Ali, R.; Rashid, S.M.; Madkhali, H.; Ganaie, M.A.; Khan, R. Neuroprotective Strategies for Neurological Disorders by Natural Products: An update. Curr. Neuropharmacol. 2019, 17, 247–267. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, H.J.; Lee, K.W. Naturally occurring phytochemicals for the prevention of Alzheimer’s disease. J. Neurochem. 2010, 112, 1415–1430. [Google Scholar] [CrossRef]
- Turner, R.S.; Thomas, R.G.; Craft, S.; van Dyck, C.H.; Mintzer, J.; Reynolds, B.A.; Brewer, J.B.; Rissman, R.A.; Raman, R.; Aisen, P.S. A randomized, double-blind, placebo-controlled trial of resveratrol for Alzheimer disease. Neurology 2015, 85, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Stojanović-Radić, Z.; Matejić, J.; Sharifi-Rad, M.; Anil Kumar, N.V.; Martins, N.; Sharifi-Rad, J. The therapeutic potential of curcumin: A review of clinical trials. Eur. J. Med. Chem. 2019, 163, 527–545. [Google Scholar] [CrossRef] [PubMed]
- Leandro, A.; Pereira, L.; Gonçalves, A.M.M. Diverse Applications of Marine Macroalgae. Mar. Drugs 2020, 18, 17. [Google Scholar] [CrossRef] [PubMed]
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Peng, J.; Yuan, J.P.; Wu, C.F.; Wang, J.H. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar] [CrossRef]
- Salehi, B.; Sharifi-Rad, J.; Seca, A.M.L.; Pinto, D.C.G.A.; Michalak, I.; Trincone, A.; Mishra, A.P.; Nigam, M.; Zam, W.; Martins, N. Current Trends on Seaweeds: Looking at Chemical Composition, Phytopharmacology, and Cosmetic Applications. Molecules 2019, 24, 4182. [Google Scholar] [CrossRef]
- Pangestuti, R.; Kim, S.K. Biological activities and health benefit effects of natural pigments derived from marine algae. J. Funct. Foods 2011, 3, 255–266. [Google Scholar] [CrossRef]
- Alghazwi, M.; Kan, Y.Q.; Zhang, W.; Gai, W.P.; Garson, M.J.; Smid, S. Neuroprotective activities of natural products from marine macroalgae during 1999–2015. J. Appl. Phycol. 2016, 28, 3599–3616. [Google Scholar] [CrossRef]
- Barbalace, M.C.; Malaguti, M.; Giusti, L.; Lucacchini, A.; Hrelia, S.; Angeloni, C. Anti-Inflammatory Activities of Marine Algae in Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 3061. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, M.; Valentão, P.; Andrade, P.B. Bioactive compounds from macroalgae in the new millennium: Implications for neurodegenerative diseases. Mar. Drugs 2014, 12, 4934–4972. [Google Scholar] [CrossRef]
- Cornish, M.L.; Critchley, A.T.; Mouritsen, O.G. Consumption of seaweeds and the human brain. J. Appl. Phycol. 2017, 29, 2377–2398. [Google Scholar] [CrossRef]
- Rengasamy, K.R.R.; Mahomoodally, M.F.; Aumeeruddy, M.Z.; Zengin, G.; Xiao, J.; Kim, D.H. Bioactive compounds in seaweeds: An overview of their biological properties and safety. Food Chem. Toxicol. 2020, 135, 111013. [Google Scholar] [CrossRef]
- Rajeswari, R.; Jeyaprakash, K. Biopotential effects of seaweeds for neurological disorders mini review. J. Pharm. Pharm. Sci. 2017, 6, 427–436. [Google Scholar]
- Lauritzen, L.; Brambilla, P.; Mazzocchi, A.; Harsløf, L.B.S.; Ciappolino, V.; Agostoni, C. DHA Effects in Brain Development and Function. Nutrients 2016, 8, 6. [Google Scholar] [CrossRef]
- Caltagirone, C.; Ferrannini, L.; Marchionni, N.; Nappi, G.; Scapagnini, G.; Trabucchi, M. The potential protective effect of tramiprosate (homotaurine) against Alzheimer’s disease: A review. Aging Clin. Exp. Res. 2012, 24, 580–587. [Google Scholar]
- Schepers, M.; Martens, N.; Tiane, A.; Vanbrabant, K.; Liu, H.B.; Lütjohann, D.; Mulder, M.; Vanmierlo, T. Edible seaweed-derived constituents: An undisclosed source of neuroprotective compounds. Neural Regen. Res. 2020, 15, 790–795. [Google Scholar]
- Williams, L.L. Marine algae as a source of prevention and relief in those with depression and dementia. World J. Pharm. Pharm. Sci. 2017, 6, 26–38. [Google Scholar] [CrossRef]
- Kim, S.K.; Pangestuti, R. Biological activities and potential health benefits of fucoxanthin derived from marine brown algae. Adv. Food Nutr. Res. 2011, 64, 111–128. [Google Scholar]
- Syed, Y.Y. Sodium Oligomannate: First Approval. Drugs 2020, 80, 441–444. [Google Scholar] [CrossRef] [PubMed]
- Pangestuti, R.; Kim, S.K. Neuroprotective effects of marine algae. Mar. Drugs 2011, 9, 803–818. [Google Scholar] [CrossRef]
- Huang, C.; Zhang, Z.; Cui, W. Marine-Derived Natural Compounds for the Treatment of Parkinson’s Disease. Mar. Drugs 2019, 17, 221. [Google Scholar] [CrossRef]
- Olasehinde, T.A.; Olaniran, A.O.; Okoh, A.I. Macroalgae as a Valuable Source of Naturally Occurring Bioactive Compounds for the Treatment of Alzheimer’s Disease. Mar. Drugs 2019, 17, 609. [Google Scholar] [CrossRef] [PubMed]
- Pangestuti, R.; Vo, T.S.; Ngo, D.H.; Kim, S.K. Fucoxanthin Ameliorates Inflammation and Oxidative Reponses in Microglia. J. Agric. Food Chem. 2013, 61, 3876–3883. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Dan, X.; Babbar, M.; Wei, Y.; Hasselbalch, S.G.; Croteau, D.L.; Bohr, V.A. Ageing as a risk factor for neurodegenerative disease. Nat. Rev. Neurol. 2019, 15, 565–581. [Google Scholar] [CrossRef]
- Singh, A.; Kukreti, R.; Saso, L.; Kukreti, S. Oxidative Stress: A Key Modulator in Neurodegenerative Diseases. Molecules 2019, 24, 1583. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.S.; Kabir, M.T.; Mamun, A.A.; Barreto, G.E.; Rashid, M.; Perveen, A.; Ashraf, G.M. Pharmacological approaches to mitigate neuroinflammation in Alzheimer’s disease. Int. Immunopharmacol. 2020, 84, 106479. [Google Scholar] [CrossRef]
- Hannan, M.A.; Dash, R.; Sohag, A.A.M.; Haque, M.N.; Moon, I.S. Neuroprotection against oxidative stress: Phytochemicals targeting TrkB signaling and the Nrf2-ARE antioxidant system. Front. Mol. Neurosci. 2020, 13, 116. [Google Scholar]
- Sivandzade, F.; Prasad, S.; Bhalerao, A.; Cucullo, L. NRF2 and NF-κB interplay in cerebrovascular and neurodegenerative disorders: Molecular mechanisms and possible therapeutic approaches. Redox Biol. 2019, 21, 101059. [Google Scholar] [CrossRef] [PubMed]
- Castelli, V.; Benedetti, E.; Antonosante, A.; Catanesi, M.; Pitari, G.; Ippoliti, R.; Cimini, A.; d’Angelo, M. Neuronal Cells Rearrangement During Aging and Neurodegenerative Disease: Metabolism, Oxidative Stress and Organelles Dynamic. Front. Mol. Neurosci. 2019, 12, 132. [Google Scholar] [CrossRef] [PubMed]
- Kandlur, A.; Satyamoorthy, K.; Gangadharan, G. Oxidative Stress in Cognitive and Epigenetic Aging: A Retrospective Glance. Front. Mol. Neurosci. 2020, 13, 41. [Google Scholar] [CrossRef]
- Chaudhari, N.; Talwar, P.; Parimisetty, A.; Lefebvre d’Hellencourt, C.; Ravanan, P. A Molecular Web: Endoplasmic Reticulum Stress, Inflammation, and Oxidative Stress. Front. Mol. Neurosci. 2014, 8, 213. [Google Scholar] [CrossRef]
- Gerakis, Y.; Hetz, C. Emerging roles of ER stress in the etiology and pathogenesis of Alzheimer’s disease. FEBS J. 2018, 285, 995–1011. [Google Scholar] [CrossRef] [PubMed]
- Guzman-Martinez, L.; Maccioni, R.B.; Andrade, V.; Navarrete, L.P.; Pastor, M.G.; Ramos-Escobar, N. Neuroinflammation as a Common Feature of Neurodegenerative Disorders. Front. Pharmacol. 2019, 10, 1008. [Google Scholar] [CrossRef]
- Yanuck, S.F. Microglial Phagocytosis of Neurons: Diminishing Neuronal Loss in Traumatic, Infectious, Inflammatory, and Autoimmune CNS Disorders. Front. Psychiatry 2019, 10, 712. [Google Scholar] [CrossRef]
- Sofroniew, M.V. Astrocyte barriers to neurotoxic inflammation. Nat. Rev. Neurosci. 2015, 16, 249–263. [Google Scholar] [CrossRef]
- Dash, R.; Mitra, S.; Ali, M.C.; Oktaviani, D.F.O.; Hannan, M.A.; Choi, S.M.; Moon, I.S. Phytosterols: Targeting Neuroinflammation in Neurodegeneration. Curr. Pharm. Des. 2020, 26, 1–23. [Google Scholar]
- Liu, Z.; Zhou, T.; Ziegler, A.C.; Dimitrion, P.; Zuo, L. Oxidative Stress in Neurodegenerative Diseases: From Molecular Mechanisms to Clinical Applications. Oxid. Med. Cell. Longev. 2017, 2017, 2525967. [Google Scholar] [CrossRef]
- Mouzat, K.; Chudinova, A.; Polge, A.; Kantar, J.; Camu, W.; Raoul, C.; Lumbroso, S. Regulation of Brain Cholesterol: What Role Do Liver X Receptors Play in Neurodegenerative Diseases? Int. J. Mol. Sci. 2019, 20, 3858. [Google Scholar] [CrossRef] [PubMed]
- Ito, A.; Hong, C.; Rong, X.; Zhu, X.; Tarling, E.J.; Hedde, P.N.; Gratton, E.; Parks, J.; Tontonoz, P. LXRs link metabolism to inflammation through Abca1-dependent regulation of membrane composition and TLR signaling. eLife 2015, 4, e08009. [Google Scholar] [CrossRef]
- Xu, P.; Li, D.; Tang, X.; Bao, X.; Huang, J.; Tang, Y.; Yang, Y.; Xu, H.; Fan, X. LXR agonists: New potential therapeutic drug for neurodegenerative diseases. Mol. Neurobiol. 2013, 48, 715–728. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.B.; Tan, X.J.; Wu, W.F.; Warner, M.; Gustafsson, J.A. Liver X receptor beta protects dopaminergic neurons in a mouse model of Parkinson disease. Proc. Natl. Acad. Sci. USA 2012, 109, 13112–13117. [Google Scholar] [CrossRef] [PubMed]
- Futter, M.; Diekmann, H.; Schoenmakers, E.; Sadiq, O.; Chatterjee, K.; Rubinsztein, D.C. Wild-type but not mutant huntingtin modulates the transcriptional activity of liver X receptors. J. Med. Genet. 2009, 46, 438–446. [Google Scholar] [CrossRef]
- Wolf, A.; Bauer, B.; Hartz, A.M. ABC Transporters and the Alzheimer’s Disease Enigma. Front. Psychiatry 2012, 3, 54. [Google Scholar] [CrossRef]
- Campbell, B.C.V.; De Silva, D.A.; Macleod, M.R.; Coutts, S.B.; Schwamm, L.H.; Davis, S.M.; Donnan, G.A. Ischaemic stroke. Nat. Rev. Dis. Primers 2019, 5, 70. [Google Scholar] [CrossRef] [PubMed]
- Soares, R.O.S.; Losada, D.M.; Jordani, M.C.; Évora, P.; Castroesilva, O. Ischemia/Reperfusion Injury Revisited: An Overview of the Latest Pharmacological Strategies. Int. J. Mol. Sci. 2019, 20, 5034. [Google Scholar] [CrossRef]
- Wu, L.; Xiong, X.; Wu, X.; Ye, Y.; Jian, Z.; Zhi, Z.; Gu, L. Targeting Oxidative Stress and Inflammation to Prevent Ischemia-Reperfusion Injury. Front. Mol. Neurosci. 2020, 13, 28. [Google Scholar] [CrossRef]
- Tschoe, C.; Bushnell, C.D.; Duncan, P.W.; Alexander-Miller, M.A.; Wolfe, S.Q. Neuroinflammation after Intracerebral Hemorrhage and Potential Therapeutic Targets. J. Stroke 2020, 22, 29–46. [Google Scholar] [CrossRef]
- Liu, F.; Lu, J.; Manaenko, A.; Tang, J.; Hu, Q. Mitochondria in Ischemic Stroke: New Insight and Implications. Aging Dis. 2018, 9, 924–937. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.L.; Mukda, S.; Chen, S.D. Diverse roles of mitochondria in ischemic stroke. Redox Biol. 2018, 16, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Gadani, S.P.; Walsh, J.T.; Lukens, J.R.; Kipnis, J. Dealing with Danger in the CNS: The Response of the Immune System to Injury. Neuron 2015, 87, 47–62. [Google Scholar] [CrossRef] [PubMed]
- Mracsko, E.; Veltkamp, R. Neuroinflammation after intracerebral hemorrhage. Front. Cell. Neurosci. 2014, 8, 388. [Google Scholar] [CrossRef] [PubMed]
- Malekahmadi, M.; Moradi Moghaddam, O.; Islam, S.M.S.; Tanha, K.; Nematy, M.; Pahlavani, N.; Firouzi, S.; Zali, M.R.; Norouzy, A. Evaluation of the effects of pycnogenol (French maritime pine bark extract) supplementation on inflammatory biomarkers and nutritional and clinical status in traumatic brain injury patients in an intensive care unit: A randomized clinical trial protocol. Trials 2020, 21, 162. [Google Scholar] [CrossRef]
- Beauchamp, K.; Mutlak, H.; Smith, W.R.; Shohami, E.; Stahel, P.F. Pharmacology of traumatic brain injury: Where is the “golden bullet”? Mol. Med. 2008, 14, 731–740. [Google Scholar] [CrossRef]
- Khatri, N.; Thakur, M.; Pareek, V.; Kumar, S.; Sharma, S.; Datusalia, A.K.J.C.; Targets, N.D.-D. Oxidative stress: Major threat in traumatic brain injury. CNS Neurol. Disord. Drug Targets 2018, 17, 689–695. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rodriguez, A.; Jose Egea-Guerrero, J.; Murillo-Cabezas, F.; Carrillo-Vico, A. Oxidative stress in traumatic brain injury. Curr. Med. Chem. 2014, 21, 1201–1211. [Google Scholar] [CrossRef]
- Heo, S.J.; Ko, S.C.; Kang, S.M.; Kang, H.S.; Kim, J.P.; Kim, S.H.; Lee, K.W.; Cho, M.G.; Jeon, Y.J. Cytoprotective effect of fucoxanthin isolated from brown algae Sargassum siliquastrum against H2O2-induced cell damage. Eur. Food Res. Technol. 2008, 228, 145–151. [Google Scholar] [CrossRef]
- Zhao, D.; Kwon, S.H.; Chun, Y.S.; Gu, M.Y.; Yang, H.O. Anti-Neuroinflammatory Effects of Fucoxanthin via Inhibition of Akt/NF-κB and MAPKs/AP-1 Pathways and Activation of PKA/CREB Pathway in Lipopolysaccharide-Activated BV-2 Microglial Cells. Neurochem. Res. 2017, 42, 667–677. [Google Scholar] [CrossRef]
- Lee, S.; Lee, Y.S.; Jung, S.H.; Kang, S.S.; Shin, K.H. Anti-oxidant activities of fucosterol from the marine algae Pelvetia siliquosa. Arch. Pharm. Res. 2003, 26, 719–722. [Google Scholar] [CrossRef]
- Jung, H.A.; Jin, S.E.; Ahn, B.R.; Lee, C.M.; Choi, J.S. Anti-inflammatory activity of edible brown alga Eisenia bicyclis and its constituents fucosterol and phlorotannins in LPS-stimulated RAW264.7 macrophages. Food Chem. Toxicol. 2013, 59, 199–206. [Google Scholar] [CrossRef]
- Choi, J.S.; Han, Y.R.; Byeon, J.S.; Choung, S.Y.; Sohn, H.S.; Jung, H.A. Protective effect of fucosterol isolated from the edible brown algae, Ecklonia stolonifera and Eisenia bicyclis, on tert-butyl hydroperoxide- and tacrine-induced HepG2 cell injury. J. Pharm. Pharmacol. 2015, 67, 1170–1178. [Google Scholar] [CrossRef]
- Fernando, I.P.S.; Jayawardena, T.U.; Kim, H.S.; Lee, W.W.; Vaas, A.P.J.P.; De Silva, H.I.C.; Abayaweera, G.S.; Nanayakkara, C.M.; Abeytunga, D.T.U.; Lee, D.S.; et al. Beijing urban particulate matter-induced injury and inflammation in human lung epithelial cells and the protective effects of fucosterol from Sargassum binderi (Sonder ex J. Agardh). Environ. Res. 2019, 172, 150–158. [Google Scholar] [CrossRef]
- Rafiquzzaman, S.M.; Kim, E.Y.; Lee, J.M.; Mohibbullah, M.; Alam, M.B.; Soo Moon, I.; Kim, J.M.; Kong, I.S. Anti-Alzheimers and anti-inflammatory activities of a glycoprotein purified from the edible brown alga Undaria pinnatifida. Food Res. Int. 2015, 77, 118–124. [Google Scholar] [CrossRef]
- Zou, Y.; Qian, Z.J.; Li, Y.; Kim, M.M.; Lee, S.H.; Kim, S.K. Antioxidant Effects of Phlorotannins Isolated from Ishige okamurae in Free Radical Mediated Oxidative Systems. J. Agric. Food Chem. 2008, 56, 7001–7009. [Google Scholar] [CrossRef]
- Zhang, Q.; Yu, P.; Li, Z.; Zhang, H.; Xu, Z.; Li, P. Antioxidant activities of sulfated polysaccharide fractions from Porphyra haitanesis. J. Appl. Phycol. 2003, 15, 305–310. [Google Scholar] [CrossRef]
- Isaka, S.; Cho, K.; Nakazono, S.; Abu, R.; Ueno, M.; Kim, D.; Oda, T. Antioxidant and anti-inflammatory activities of porphyran isolated from discolored nori (Porphyra yezoensis). Int. J. Biol. Macromol. 2015, 74, 68–75. [Google Scholar] [CrossRef]
- Anggadiredja, J.; Andyani, R.; Hayati, M. Antioxidant activity of Sargassum polycystum (Phaeophyta) and Laurencia obtusa (Rhodophyta) from Seribu Islands. J. Appl. Phycol. 1997, 9, 477. [Google Scholar] [CrossRef]
- Rengasamy, K.R.R.; Amoo, S.O.; Aremu, A.O.; Stirk, W.A.; Gruz, J.; Šubrtová, M.; Doležal, K.; Van Staden, J. Phenolic profiles, antioxidant capacity, and acetylcholinesterase inhibitory activity of eight South African seaweeds. J. Appl. Phycol. 2015, 27, 1599–1605. [Google Scholar] [CrossRef]
- Hannan, M.A.; Sohag, A.A.M.; Dash, R.; Haque, M.N.; Mohibbullah, M.; Oktaviani, D.F.; Hossain, M.T.; Choi, H.J.; Moon, I.S. Phytosterols of marine algae: Insights into the potential health benefits and molecular pharmacology. Phytomedicine 2020, 69, 153201. [Google Scholar] [CrossRef]
- Sun, Z.; Mohamed, M.A.A.; Park, S.Y.; Yi, T.H. Fucosterol protects cobalt chloride induced inflammation by the inhibition of hypoxia-inducible factor through PI3K/Akt pathway. Int. Immunopharmacol. 2015, 29, 642–647. [Google Scholar] [CrossRef] [PubMed]
- Yoo, M.S.; Shin, J.S.; Choi, H.E.; Cho, Y.W.; Bang, M.H.; Baek, N.I.; Lee, K.T. Fucosterol isolated from Undaria pinnatifida inhibits lipopolysaccharide-induced production of nitric oxide and pro-inflammatory cytokines via the inactivation of nuclear factor-kappaB and p38 mitogen-activated protein kinase in RAW264.7 macrophages. Food Chem. 2012, 135, 967–975. [Google Scholar] [CrossRef] [PubMed]
- Brandhorst, S.; Choi, I.Y.; Wei, M.; Cheng, C.W.; Sedrakyan, S.; Navarrete, G.; Dubeau, L.; Yap, L.P.; Park, R.; Vinciguerra, M.; et al. A Periodic Diet that Mimics Fasting Promotes Multi-System Regeneration, Enhanced Cognitive Performance, and Healthspan. Cell Metab. 2015, 22, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.H.; Gan, S.Y.; Tan, S.C.; Gany, S.A.; Ying, T.; Gray, A.I.; Igoli, J.; Chan, E.W.L.; Phang, S.M. Fucosterol inhibits the cholinesterase activities and reduces the release of pro-inflammatory mediators in lipopolysaccharide and amyloid-induced microglial cells. J. Appl. Phycol. 2018, 30, 3261–3270. [Google Scholar] [CrossRef]
- Jung, W.K.; Heo, S.J.; Jeon, Y.J.; Lee, C.M.; Park, Y.M.; Byun, H.G.; Choi, Y.H.; Park, S.G.; Choi, I.W. Inhibitory effects and molecular mechanism of dieckol isolated from marine brown alga on COX-2 and iNOS in microglial cells. J. Agric. Food Chem. 2009, 57, 4439–4446. [Google Scholar] [CrossRef]
- Kim, A.R.; Lee, M.S.; Choi, J.W.; Utsuki, T.; Kim, J.I.; Jang, B.C.; Kim, H.R. Phlorofucofuroeckol A suppresses expression of inducible nitric oxide synthase, cyclooxygenase-2, and pro-inflammatory cytokines via inhibition of nuclear factor-κB, c-Jun NH2-terminal kinases, and Akt in microglial cells. Inflammation 2013, 36, 259–271. [Google Scholar] [CrossRef]
- Yu, D.K.; Lee, B.; Kwon, M.; Yoon, N.; Shin, T.; Kim, N.G.; Choi, J.S.; Kim, H.R. Phlorofucofuroeckol B suppresses inflammatory responses by down-regulating nuclear factor κB activation via Akt, ERK, and JNK in LPS-stimulated microglial cells. Int. Immunopharmacol. 2015, 28, 1068–1075. [Google Scholar] [CrossRef]
- Kim, A.R.; Lee, B.; Joung, E.J.; Gwon, W.G.; Utsuki, T.; Kim, N.G.; Kim, H.R. 6,6′-Bieckol suppresses inflammatory responses by down-regulating nuclear factor-κB activation via Akt, JNK, and p38 MAPK in LPS-stimulated microglial cells. Immunopharmacol. Immunotoxicol. 2016, 38, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.I.; Jung, S.H.; Lee, K.T.; Choi, J.H. 8,8′-Bieckol, isolated from edible brown algae, exerts its anti-inflammatory effects through inhibition of NF-κB signaling and ROS production in LPS-stimulated macrophages. Int. Immunopharmacol. 2014, 23, 460–468. [Google Scholar] [CrossRef]
- Florez, N.; Gonzalez-Munoz, M.J.; Ribeiro, D.; Fernandes, E.; Dominguez, H.; Freitas, M. Algae Polysaccharides’ Chemical Characterization and their Role in the Inflammatory Process. Curr. Med. Chem. 2017, 24, 149–175. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Han, M.H.; Park, C.; Jin, C.Y.; Kim, G.Y.; Choi, I.W.; Kim, N.D.; Nam, T.J.; Kwon, T.K.; Choi, Y.H. Anti-inflammatory effects of fucoidan through inhibition of NF-κB, MAPK and Akt activation in lipopolysaccharide-induced BV2 microglia cells. Food Chem. Toxicol. 2011, 49, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.Q.; Jia, Y.J.; Zhang, T.; Zhang, Q.B.; Wang, X.M. Fucoidan Protects against Lipopolysaccharide-Induced Rat Neuronal Damage and Inhibits the Production of Proinflammatory Mediators in Primary Microglia. CNS Neurosci. Ther. 2012, 18, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.A.; Xu, L.; Wu, H.G. Immunomodulatory function of κ-carrageenan oligosaccharides acting on LPS-activated microglial cells. Neurochem. Res. 2014, 39, 333–343. [Google Scholar] [CrossRef]
- Jiang, Z.; Hama, Y.; Yamaguchi, K.; Oda, T. Inhibitory effect of sulphated polysaccharide porphyran on nitric oxide production in lipopolysaccharide-stimulated RAW264.7 macrophages. J. Biochem. 2012, 151, 65–74. [Google Scholar] [CrossRef]
- Liu, X.Y.; Liu, D.; Lin, G.P.; Wu, Y.J.; Gao, L.Y.; Ai, C.; Huang, Y.F.; Wang, M.F.; El-Seedi, H.R.; Chen, X.H.; et al. Anti-ageing and antioxidant effects of sulfate oligosaccharides from green algae Ulva lactuca and Enteromorpha prolifera in SAMP8 mice. Int. J. Biol. Macromol. 2019, 139, 342–351. [Google Scholar] [CrossRef]
- Zhou, R.; Shi, X.Y.; Bi, D.C.; Fang, W.S.; Wei, G.B.; Xu, X. Alginate-Derived Oligosaccharide Inhibits Neuroinflammation and Promotes Microglial Phagocytosis of β-Amyloid. Mar. Drugs 2015, 13, 5828–5846. [Google Scholar] [CrossRef]
- Bi, D.; Lai, Q.; Han, Q.; Cai, N.; He, H.; Fang, W.; Yi, J.; Li, X.; Xu, H.; Li, X.; et al. Seleno-polymannuronate attenuates neuroinflammation by suppressing microglial and astrocytic activation. J. Funct. Foods 2018, 51, 113–120. [Google Scholar] [CrossRef]
- Yang, E.J.; Ham, Y.M.; Yang, K.W.; Lee, N.H.; Hyun, C.G. Sargachromenol from Sargassum micracanthum inhibits the lipopolysaccharide-induced production of inflammatory mediators in RAW 264.7 macrophages. Sci. World J. 2013, 2013, 712303. [Google Scholar] [CrossRef]
- Kang, G.J.; Han, S.C.; Yoon, W.J.; Koh, Y.S.; Hyun, J.W.; Kang, H.K.; Youl Cho, J.; Yoo, E.S. Sargaquinoic acid isolated from Sargassum siliquastrum inhibits lipopolysaccharide-induced nitric oxide production in macrophages via modulation of nuclear factor-κB and c-Jun N-terminal kinase pathways. Immunopharm. Immunot. 2013, 35, 80–87. [Google Scholar] [CrossRef]
- Kim, M.; Li, Y.X.; Dewapriya, P.; Ryu, B.; Kim, S.K. Floridoside suppresses pro-inflammatory responses by blocking MAPK signaling in activated microglia. BMB Rep. 2013, 46, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Souza, C.R.M.; Bezerra, W.P.; Souto, J.T. Marine Alkaloids with Anti-Inflammatory Activity: Current Knowledge and Future Perspectives. Mar. Drugs 2020, 18, 147. [Google Scholar] [CrossRef] [PubMed]
- Jung, W.K.; Ahn, Y.W.; Lee, S.H.; Choi, Y.H.; Kim, S.K.; Yea, S.S.; Choi, I.; Park, S.G.; Seo, S.K.; Lee, S.W.; et al. Ecklonia cava ethanolic extracts inhibit lipopolysaccharide-induced cyclooxygenase-2 and inducible nitric oxide synthase expression in BV2 microglia via the MAP kinase and NF-kappaB pathways. Food Chem. Toxicol. 2009, 47, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, J.I.; Choi, J.W.; Kim, M.; Yoon, N.Y.; Choi, C.G.; Choi, J.S.; Kim, H.R. Anti-inflammatory effect of hexane fraction from Myagropsis myagroides ethanolic extract in lipopolysaccharide-stimulated BV-2 microglial cells. J. Pharm. Pharmacol. 2013, 65, 895–906. [Google Scholar] [CrossRef]
- Kim, S.; Lee, M.S.; Lee, B.; Gwon, W.G.; Joung, E.J.; Yoon, N.Y.; Kim, H.R. Anti-inflammatory effects of sargachromenol-rich ethanolic extract of Myagropsis myagroides on lipopolysaccharide-stimulated BV-2 cells. BMC Complement. Altern. Med. 2014, 14, 231. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.J.; Joung, E.J.; Kwon, M.S.; Lee, B.; Utsuki, T.; Oh, C.W.; Kim, H.R. Anti-Inflammatory Effect of Ethanolic Extract of Sargassum serratifolium in Lipopolysaccharide-Stimulated BV2 Microglial Cells. J. Med. Food 2016, 19, 1023–1031. [Google Scholar] [CrossRef]
- Gany, S.A.; Tan, S.C.; Gan, S.Y. Antioxidative, anticholinesterase and anti-neuroinflammatory properties of Malaysian brown and green seaweeds. World Acad. Sci. Eng. Technol. 2015, 8, 1269–1275. [Google Scholar]
- Jin, D.Q.; Lim, C.S.; Sung, J.Y.; Choi, H.G.; Ha, I.; Han, J.S. Ulva conglobata, a marine algae, has neuroprotective and anti-inflammatory effects in murine hippocampal and microglial cells. Neurosci. Lett. 2006, 402, 154–158. [Google Scholar] [CrossRef]
- Gwon, W.G.; Lee, M.S.; Kim, J.S.; Kim, J.I.; Lim, C.W.; Kim, N.G.; Kim, H.R. Hexane fraction from Sargassum fulvellum inhibits lipopolysaccharide-induced inducible nitric oxide synthase expression in RAW 264.7 cells via NF-κB pathways. Am. J. Chin. Med. 2013, 41, 565–584. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.E.; Jung, Y.C.; Jung, I.; Lee, H.W.; Youn, H.Y.; Lee, J.S. Anti-inflammatory effects of ethanolic extract from Sargassum horneri (Turner) C. Agardh on lipopolysaccharide-stimulated macrophage activation via NF-κB pathway regulation. Immunol. Investig. 2015, 44, 137–146. [Google Scholar] [CrossRef]
- Joung, E.J.; Lee, M.S.; Choi, J.W.; Kim, J.S.; Shin, T.; Jung, B.M.; Yoon, N.Y.; Lim, C.W.; Kim, J.I.; Kim, H.R. Anti-inflammatory effect of ethanolic extract from Myagropsis myagroides on murine macrophages and mouse ear edema. BMC Complement. Altern. Med. 2012, 12, 171. [Google Scholar] [CrossRef] [PubMed]
- Joung, E.J.; Lee, M.S.; Choi, J.W.; Kim, J.S.; Shin, T.; Jung, B.M.; Kim, J.I.; Kim, H.R. Anti-inflammatory effects of phlorofucofuroeckol B-rich ethyl acetate fraction obtained from Myagropsis myagroides on lipopolysaccharide-stimulated RAW 264.7 cells and mouse edema. Int. Immunopharmacol. 2012, 14, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Joung, E.J.; Gwon, W.G.; Shin, T.; Jung, B.M.; Choi, J.; Kim, H.R. Anti-inflammatory action of the ethanolic extract from Sargassum serratifolium on lipopolysaccharide-stimulated mouse peritoneal macrophages and identification of active components. J. Appl. Phycol. 2017, 29, 563–573. [Google Scholar] [CrossRef]
- Yoon, N.Y.; Chung, H.Y.; Kim, H.R.; Choi, J.S. Acetyl- and butyrylcholinesterase inhibitory activities of sterols and phlorotannins from Ecklonia stolonifera. Fish. Res. 2008, 74, 200–207. [Google Scholar] [CrossRef]
- Castro-Silva, E.S.; Bello, M.; Hernandez-Rodriguez, M.; Correa-Basurto, J.; Murillo-Alvarez, J.I.; Rosales-Hernandez, M.C.; Munoz-Ochoa, M. In vitro and in silico evaluation of fucosterol from Sargassum horridum as potential human acetylcholinesterase inhibitor. J. Biomol. Struct. Dyn. 2019, 37, 3259–3268. [Google Scholar] [CrossRef]
- Kawee-ai, A.; Kuntiya, A.; Kim, S.M. Anticholinesterase and antioxidant activities of fucoxanthin purified from the microalga Phaeodactylum tricornutum. Nat. Prod. Commun. 2013, 8, 1381–1386. [Google Scholar] [CrossRef]
- Lin, J.; Huang, L.; Yu, J.; Xiang, S.; Wang, J.; Zhang, J.; Yan, X.; Cui, W.; He, S.; Wang, Q. Fucoxanthin, a Marine Carotenoid, Reverses Scopolamine-Induced Cognitive Impairments in Mice and Inhibits Acetylcholinesterase in vitro. Mar. Drugs 2016, 14, 67. [Google Scholar] [CrossRef]
- Shanmuganathan, B.; Sheeja Malar, D.; Sathya, S.; Pandima Devi, K. Antiaggregation Potential of Padina gymnospora against the Toxic Alzheimer’s Beta-Amyloid Peptide 25-35 and Cholinesterase Inhibitory Property of Its Bioactive Compounds. PLoS ONE 2015, 10, e0141708. [Google Scholar] [CrossRef]
- Kannan, R.R.R.; Aderogba, M.A.; Ndhlala, A.R.; Stirk, W.A.; Van Staden, J. Acetylcholinesterase inhibitory activity of phlorotannins isolated from the brown alga, Ecklonia maxima (Osbeck) Papenfuss. Food Res. Int. 2013, 54, 1250–1254. [Google Scholar] [CrossRef]
- Myung, C.S.; Shin, H.C.; Bao, H.Y.; Yeo, S.J.; Lee, B.H.; Kang, J.S. Improvement of memory by dieckol and phlorofucofuroeckol in ethanol-treated mice: Possible involvement of the inhibition of acetylcholinesterase. Arch. Pharm. Res. 2005, 28, 691–698. [Google Scholar] [CrossRef]
- Choi, B.W.; Ryu, G.; Park, S.H.; Kim, E.S.; Shin, J.; Roh, S.S.; Shin, H.C.; Lee, B.H. Anticholinesterase activity of plastoquinones from Sargassum sagamianum: Lead compounds for Alzheimer’s disease therapy. Phytother. Res. 2007, 21, 423–426. [Google Scholar] [CrossRef]
- Rengasamy, K.R.R.; Aderogba, M.A.; Amoo, S.O.; Stirk, W.A.; Van Staden, J. Macrocystis angustifolia is a potential source of enzyme inhibitors linked to type 2 diabetes and dementia. J. Appl. Phycol. 2014, 26, 1557–1563. [Google Scholar] [CrossRef]
- Seong, S.H.; Ali, M.Y.; Kim, H.R.; Jung, H.A.; Choi, J.S. BACE1 inhibitory activity and molecular docking analysis of meroterpenoids from Sargassum serratifolium. Bioorg. Med. Chem. 2017, 25, 3964–3970. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Jun, M. Dual BACE1 and Cholinesterase Inhibitory Effects of Phlorotannins from Ecklonia cava-An In Vitro and in Silico Study. Mar. Drugs 2019, 17, 91. [Google Scholar] [CrossRef]
- Murugan, A.C.; Vallal, D.; Karim, M.R.; Govindan, N.; Yusoff, M.; Rahman, M.J. In vitro antiradical and neuroprotective activity of polyphenolic extract from marine algae Padina australis H. J. Chem. Pharm. Res. 2015, 7, 355–362. [Google Scholar]
- Custódio, L.; Soares, F.; Pereira, H.; Rodrigues, M.J.; Barreira, L.; Rauter, A.P.; Alberício, F.; Varela, J. Botryococcus braunii and Nannochloropsis oculata extracts inhibit cholinesterases and protect human dopaminergic SH-SY5Y cells from H2O2-induced cytotoxicity. J. Appl. Phycol. 2015, 27, 839–848. [Google Scholar] [CrossRef]
- Custódio, L.; Silvestre, L.; Rocha, M.I.; Rodrigues, M.J.; Vizetto-Duarte, C.; Pereira, H.; Barreira, L.; Varela, J. Methanol extracts from Cystoseira tamariscifolia and Cystoseira nodicaulis are able to inhibit cholinesterases and protect a human dopaminergic cell line from hydrogen peroxide-induced cytotoxicity. Pharm. Biol. 2016, 54, 1687–1696. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.E.; Son, H.J.; Lim, D.W.; Yoon, M.; Lee, J.; Kim, Y.T.; Han, D.; Lee, C.; Um, M.Y. Memory-enhancing effects of Ishige foliacea extract: In vitro and in vivo study. J. Food Biochem. 2020, 44, e13162. [Google Scholar] [CrossRef]
- Nunes, N.; Rosa, G.P.; Ferraz, S.; Barreto, M.C.; de Carvalho, M.A.A.P. Fatty acid composition, TLC screening, ATR-FTIR analysis, anti-cholinesterase activity, and in vitro cytotoxicity to A549 tumor cell line of extracts of 3 macroalgae collected in Madeira. J. Appl. Phycol. 2019. [Google Scholar] [CrossRef]
- De Souza, E.T.; de Lira, D.P.; de Queiroz, A.C.; da Silva, D.J.C.; de Aquino, A.B.; Mella, E.A.C.; Lorenzo, V.P.; de Miranda, G.E.C.; de Araújo-Júnior, J.X.; de Oliveira Chaves, M.C.; et al. The antinociceptive and anti-inflammatory activities of caulerpin, a bisindole alkaloid isolated from seaweeds of the genus Caulerpa. Mar. Drugs 2009, 7, 689–704. [Google Scholar] [CrossRef] [PubMed]
- Cengiz, S.; Cavas, L.; Yurdakoc, K.; Pohnert, G. The Sesquiterpene Caulerpenyne from Caulerpa spp. is a Lipoxygenase Inhibitor. Mar. Biotechnol. 2011, 13, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Ryan, S.; O’Gorman, D.M.; Nolan, Y.M. Evidence that the marine-derived multi-mineral Aquamin has anti-inflammatory effects on cortical glial-enriched cultures. Phytother. Res. 2011, 25, 765–767. [Google Scholar] [CrossRef] [PubMed]
- Ryu, G.; Park, S.H.; Kim, E.S.; Choi, B.W.; Ryu, S.Y.; Lee, B.H. Cholinesterase inhibitory activity of two farnesylacetone derivatives from the brown alga Sargassum sagamianum. Arch. Pharm. Res. 2003, 26, 796–799. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.A.; Ali, M.Y.; Choi, R.J.; Jeong, H.O.; Chung, H.Y.; Choi, J.S. Kinetics and molecular docking studies of fucosterol and fucoxanthin, BACE1 inhibitors from brown algae Undaria pinnatifida and Ecklonia stolonifera. Food Chem. Toxicol. 2016, 89, 104–111. [Google Scholar] [CrossRef]
- Xiang, S.; Liu, F.; Lin, J.; Chen, H.; Huang, C.; Chen, L.; Zhou, Y.; Ye, L.; Zhang, K.; Jin, J.; et al. Fucoxanthin Inhibits β-Amyloid Assembly and Attenuates β-Amyloid Oligomer-Induced Cognitive Impairments. J. Agric. Food Chem. 2017, 65, 4092–4102. [Google Scholar] [CrossRef]
- Alghazwi, M.; Smid, S.; Musgrave, I.; Zhang, W. In vitro studies of the neuroprotective activities of astaxanthin and fucoxanthin against amyloid beta (Aβ(1-42)) toxicity and aggregation. Neurochem. Int. 2019, 124, 215–224. [Google Scholar] [CrossRef]
- Shanmuganathan, B.; Suryanarayanan, V.; Sathya, S.; Narenkumar, M.; Singh, S.K.; Ruckmani, K.; Pandima Devi, K. Anti-amyloidogenic and anti-apoptotic effect of α-bisabolol against Aβ induced neurotoxicity in PC12 cells. Eur. J. Med. Chem. 2018, 143, 1196–1207. [Google Scholar] [CrossRef]
- Hoang, M.H.; Jia, Y.; Jun, H.J.; Lee, J.H.; Lee, B.Y.; Lee, S.J. Fucosterol Is a Selective Liver X Receptor Modulator That Regulates the Expression of Key Genes in Cholesterol Homeostasis in Macrophages, Hepatocytes, and Intestinal Cells. J. Agric. Food Chem. 2012, 60, 11567–11575. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, J.; Fu, Z.; Ye, C.; Zhang, R.; Song, Y.; Zhang, Y.; Li, H.; Ying, H.; Liu, H. 24(S)-Saringosterol from edible marine seaweed Sargassum fusiforme is a novel selective LXRbeta agonist. J. Agric. Food Chem. 2014, 62, 6130–6137. [Google Scholar] [CrossRef]
- Seong, S.H.; Paudel, P.; Choi, J.W.; Ahn, D.H.; Nam, T.J.; Jung, H.A.; Choi, J.S. Probing multi-target action of phlorotannins as new monoamine oxidase inhibitors and dopaminergic receptor modulators with the potential for treatment of neuronal disorders. Mar. Drugs 2019, 17, 377. [Google Scholar] [CrossRef]
- Oktaviani, D.F.; Bae, Y.S.; Meinita, M.D.N.; Moon, I.S.; Hong, Y.K. An ethanol extract of the brown seaweed Hizikia fusiformis and its active constituent, fucosterol, extend the lifespan of the nematode Caenorhabditis elegans. J. Life Sci. 2019, 29, 1120–1125. [Google Scholar]
- Hannan, M.A.; Dash, R.; Sohag, A.A.M.; Moon, I.S. Deciphering molecular mechanism of the neuropharmacological action of fucosterol through integrated system pharmacology and in silico analysis. Mar. Drug 2019, 17, 639. [Google Scholar] [CrossRef]
- Dezsi, L.; Vecsei, L. Monoamine Oxidase B Inhibitors in Parkinson’s Disease. CNS Neurol. Disord. DrugTargets 2017, 16, 425–439. [Google Scholar] [CrossRef] [PubMed]
- Abdul, Q.A.; Choi, R.J.; Jung, H.A.; Choi, J.S. Health benefit of fucosterol from marine algae: A review. J. Sci. Food Agric. 2016, 96, 1856–1866. [Google Scholar] [CrossRef] [PubMed]
- Tsang, C.K.; Ina, A.; Goto, T.; Kamei, Y. Sargachromenol, a novel nerve growth factor-potentiating substance isolated from Sargassum macrocarpum, promotes neurite outgrowth and survival via distinct signaling pathways in PC12D cells. Neuroscience 2005, 132, 633–643. [Google Scholar] [CrossRef]
- Kamei, Y.; Tsang, C.K. Sargaquinoic acid promotes neurite outgrowth via protein kinase A and MAP kinases-mediated signaling pathways in PC12D cells. Int. J. Dev. Neurosci. 2003, 21, 255–262. [Google Scholar] [CrossRef]
- Ina, A.; Hayashi, K.I.; Nozaki, H.; Kamei, Y. Pheophytin a, a low molecular weight compound found in the marine brown alga Sargassum fulvellum, promotes the differentiation of PC12 cells. Int. J. Dev. Neurosci. 2007, 25, 63–68. [Google Scholar] [CrossRef]
- Ina, A.; Kamei, Y. Vitamin B(12), a chlorophyll-related analog to pheophytin a from marine brown algae, promotes neurite outgrowth and stimulates differentiation in PC12 cells. Cytotechnology 2006, 52, 181–187. [Google Scholar] [CrossRef]
- Wichmann, H.; Brinkhoff, T.; Simon, M.; Richter-Landsberg, C. Dimethylsulfoniopropionate promotes process outgrowth in neural cells and exerts protective effects against tropodithietic acid. Mar. Drugs 2016, 14, 89. [Google Scholar] [CrossRef]
- Mohibbullah, M.; Choi, J.S.; Bhuiyan, M.M.H.; Haque, M.N.; Rahman, M.K.; Moon, I.S.; Hong, Y.K. The red alga Gracilariopsis chorda and its active constituent arachidonic acid promote spine dynamics via dendritic filopodia and potentiate functional synaptic plasticity in hippocampal neurons. J. Med. Food 2018, 21, 481–488. [Google Scholar] [CrossRef]
- Kamei, Y.; Sagara, A. Neurite outgrowth promoting activity of marine algae from Japan against rat adrenal medulla pheochromocytoma cell line, PC12D. Cytotechnology 2002, 40, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Mohibbullah, M.; Bhuiyan, M.M.; Hannan, M.A.; Getachew, P.; Hong, Y.K.; Choi, J.S.; Choi, I.S.; Moon, I.S. The Edible Red alga Porphyra yezoensis promotes neuronal survival and cytoarchitecture in primary hippocampal neurons. Cell. Mol. Neurobiol. 2016, 36, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Hannan, M.A.; Haque, M.N.; Mohibbullah, M.; Dash, R.; Hong, Y.K.; Moon, I.S. Gelidium amansii Attenuates hypoxia/reoxygenation-induced oxidative injury in primary hippocampal neurons through suppressing glun2b expression. Antioxidants 2020, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Hannan, M.A.; Mohibbullah, M.; Hong, Y.K.; Moon, I.S. Proteomic analysis of the neurotrophic effect of Gelidium amansii in primary cultured neurons. J. Med. Food 2017, 20, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Hannan, M.A.; Kang, J.Y.; Hong, Y.K.; Lee, H.; Choi, J.S.; Choi, I.S.; Moon, I.S. The marine alga Gelidium amansii promotes the development and complexity of neuronal cytoarchitecture. Phytother. Res. 2013, 27, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Hannan, M.A.; Mohibbullah, M.; Hong, Y.K.; Nam, J.H.; Moon, I.S. Gelidium amansii promotes dendritic spine morphology and synaptogenesis, and modulates NMDA receptor-mediated postsynaptic current. In Vitro Cell. Dev. Biol. Anim. 2014, 50, 445–452. [Google Scholar] [CrossRef]
- Hannan, M.A.; Kang, J.Y.; Hong, Y.K.; Lee, H.; Chowdhury, M.T.; Choi, J.S.; Choi, I.S.; Moon, I.S. A brown alga Sargassum fulvellum facilitates neuronal maturation and synaptogenesis. In Vitro Cell. Dev. Biol. Anim. 2012, 48, 535–544. [Google Scholar] [CrossRef]
- Hannan, M.A.; Mohibbullah, M.; Hwang, S.Y.; Lee, K.; Kim, Y.C.; Hong, Y.K.; Moon, I.S. Differential neuritogenic activities of two edible brown macroalgae, Undaria pinnatifida and Saccharina japonica. Am. J. Chin. Med. 2014, 42, 1371–1384. [Google Scholar] [CrossRef]
- Mohibbullah, M.; Hannan, M.A.; Park, I.S.; Moon, I.S.; Hong, Y.K. The edible red seaweed Gracilariopsis chorda promotes axodendritic architectural complexity in hippocampal neurons. J. Med. Food 2016, 19, 638–644. [Google Scholar] [CrossRef]
- Tirtawijaya, G.; Mohibbullah, M.; Meinita, M.D.N.; Moon, I.S.; Hong, Y.K. The tropical carrageenophyte Kappaphycus alvarezii extract promotes axodendritic maturation of hippocampal neurons in primary culture. J. Appl. Phycol. 2018, 30, 3233–3241. [Google Scholar] [CrossRef]
- Tirtawijaya, G.; Mohibbullah, M.; Meinita, M.D.N.; Moon, I.S.; Hong, Y.K. The ethanol extract of the rhodophyte Kappaphycus alvarezii promotes neurite outgrowth in hippocampal neurons. J. Appl. Phycol. 2016, 28, 2515–2522. [Google Scholar] [CrossRef]
- Tirtawijaya, G.; Haque, M.N.; Choi, J.S.; Moon, I.S.; Meinita, M.D.N.; Choi, J.S.; Hong, Y.K.; Science, F. Spinogenesis and synaptogenesis effects of the red seaweed Kappaphycus alvarezii and its isolated cholesterol on hippocampal neuron cultures. Prev. Nutr. Food Sci. 2019, 24, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Tirtawijaya, G.; Meinita, M.D.N.; Marhaeni, B.; Haque, M.N.; Moon, I.S.; Hong, Y.K. Neurotrophic activity of the Carrageenophyte Kappaphycus alvarezii cultivated at different depths and for different growth periods in various areas of indonesia. Evid. Based Complement. Alternat. Med. 2018, 2018, 1098076. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Yu, J.; Zhao, J.; Zhang, K.; Zheng, J.; Wang, J.; Huang, C.; Zhang, J.; Yan, X.; Gerwick, W.H.; et al. Fucoxanthin, a marine carotenoid, attenuates β-amyloid oligomer-induced neurotoxicity possibly via regulating the PI3K/Akt and the ERK pathways in SH-SY5Y cells. Oxid. Med. Cell. Longev. 2017, 2017, 6792543. [Google Scholar] [CrossRef]
- Yu, J.; Lin, J.J.; Yu, R.; He, S.; Wang, Q.W.; Cui, W.; Zhang, J.R. Fucoxanthin prevents H(2)O(2)-induced neuronal apoptosis via concurrently activating the PI3-K/Akt cascade and inhibiting the ERK pathway. Food Nutr. Res. 2017, 61, 1304678. [Google Scholar] [CrossRef]
- Ikeda, K.; Kitamura, A.; Machida, H.; Watanabe, M.; Negishi, H.; Hiraoka, J.; Nakano, T. Effect of Undaria pinnatifida (Wakame) on the development of cerebrovascular diseases in stroke-prone spontaneously hypertensive rats. Clin. Exp. Pharmacol. Physiol. 2003, 30, 44–48. [Google Scholar] [CrossRef]
- Mohibbullah, M.; Haque, M.N.; Khan, M.N.A.; Park, I.S.; Moon, I.S.; Hong, Y.K. Neuroprotective effects of fucoxanthin and its derivative fucoxanthinol from the phaeophyte Undaria pinnatifida attenuate oxidative stress in hippocampal neurons. J. Appl. Phycol. 2018, 30, 3243–3252. [Google Scholar] [CrossRef]
- Hu, L.; Chen, W.; Tian, F.; Yuan, C.; Wang, H.; Yue, H. Neuroprotective role of fucoxanthin against cerebral ischemic/reperfusion injury through activation of Nrf2/HO-1 signaling. Biomed. Pharmacother. 2018, 106, 1484–1489. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, H.; Fan, Y.; Gao, Y.; Li, X.; Hu, Z.; Ding, K.; Wang, Y.; Wang, X. Fucoxanthin provides neuroprotection in models of traumatic brain injury via the Nrf2-ARE and Nrf2-autophagy pathways. Sci. Rep. 2017, 7, 46763. [Google Scholar] [CrossRef]
- Shimizu, H.; Koyama, T.; Yamada, S.; Lipton, S.A.; Satoh, T. Zonarol, a sesquiterpene from the brown algae Dictyopteris undulata, provides neuroprotection by activating the Nrf2/ARE pathway. Biochem. Biophys. Res. Commun. 2015, 457, 718–722. [Google Scholar] [CrossRef]
- Shanmuganathan, B.; Sathya, S.; Balasubramaniam, B.; Balamurugan, K.; Devi, K.P. Amyloid-β induced neuropathological actions are suppressed by Padina gymnospora (Phaeophyceae) and its active constituent α-bisabolol in Neuro2a cells and transgenic Caenorhabditis elegans Alzheimer’s model. Nitric Oxide 2019, 91, 52–66. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.H.; Choi, J.S.; Nam, T.J. Fucosterol from an edible brown alga Ecklonia stolonifera prevents soluble amyloid beta-induced cognitive dysfunction in aging rats. Mar. Drugs 2018, 16, 368. [Google Scholar] [CrossRef] [PubMed]
- Gan, S.Y.; Wong, L.Z.; Wong, J.W.; Tan, E.L. Fucosterol exerts protection against amyloid β-induced neurotoxicity, reduces intracellular levels of amyloid β and enhances the mRNA expression of neuroglobin in amyloid β-induced SH-SY5Y cells. Int. J. Biol. Macromol. 2019, 121, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Liu, D.Q.; Liang, T.J.; Li, J.; Zhang, H.Y.; Liu, A.H.; Guo, Y.W.; Mao, S.C. Bioactive constituents from the green alga Caulerpa racemosa. Bioorg. Med. Chem. 2015, 23, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Ahn, B.R.; Moon, H.E.; Kim, H.R.; Jung, H.A.; Choi, J.S. Neuroprotective effect of edible brown alga Eisenia bicyclis on amyloid beta peptide-induced toxicity in PC12 cells. Arch. Pharm. Res. 2012, 35, 1989–1998. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Amarsanaa, K.; Lee, J.H.; Rhim, J.K.; Kwon, J.M.; Kim, S.H.; Park, J.M.; Jung, S.C.; Eun, S.Y. Neuroprotective mechanisms of dieckol against glutamate toxicity through reactive oxygen species scavenging and nuclear factor-like 2/heme oxygenase-1 pathway. Korean J. Physiol. Pharmacol. 2019, 23, 121–130. [Google Scholar] [CrossRef]
- Yang, E.J.; Ahn, S.; Ryu, J.; Choi, M.S.; Choi, S.; Chong, Y.H.; Hyun, J.W.; Chang, M.J.; Kim, H.S. Phloroglucinol Attenuates the Cognitive Deficits of the 5XFAD Mouse Model of Alzheimer’s Disease. PLoS ONE 2015, 10, e0135686. [Google Scholar] [CrossRef]
- Kang, S.M.; Cha, S.H.; Ko, J.Y.; Kang, M.C.; Kim, D.; Heo, S.J.; Kim, J.S.; Heu, M.S.; Kim, Y.T.; Jung, W.K.; et al. Neuroprotective effects of phlorotannins isolated from a brown alga, Ecklonia cava, against H2O2-induced oxidative stress in murine hippocampal HT22 cells. Environ. Toxicol. Pharmacol. 2012, 34, 96–105. [Google Scholar] [CrossRef]
- Heo, S.J.; Cha, S.H.; Kim, K.N.; Lee, S.H.; Ahn, G.; Kang, D.H.; Oh, C.; Choi, Y.U.; Affan, A.; Kim, D.; et al. Neuroprotective effect of phlorotannin isolated from Ishige okamurae against H2O2 -induced oxidative stress in murine hippocampal neuronal cells, HT22. Appl. Biochem. Biotechnol. 2012, 166, 1520–1532. [Google Scholar] [CrossRef]
- Kim, J.J.; Kang, Y.J.; Shin, S.A.; Bak, D.H.; Lee, J.W.; Lee, K.B.; Yoo, Y.C.; Kim, D.K.; Lee, B.H.; Kim, D.W.; et al. Phlorofucofuroeckol improves glutamate-induced neurotoxicity through modulation of oxidative stress-mediated mitochondrial dysfunction in PC12 cells. PLoS ONE 2016, 11, e0163433. [Google Scholar] [CrossRef]
- Wang, J.; Zheng, J.; Huang, C.; Zhao, J.; Lin, J.; Zhou, X.; Naman, C.B.; Wang, N.; Gerwick, W.H.; Wang, Q.; et al. Eckmaxol, a phlorotannin extracted from Ecklonia maxima, Produces anti-beta-amyloid oligomer neuroprotective effects possibly via directly acting on glycogen synthase kinase 3 beta. ACS. Chem. Neurosci. 2018, 9, 1349–1356. [Google Scholar] [CrossRef]
- Jhamandas, J.H.; Wie, M.B.; Harris, K.; MacTavish, D.; Kar, S. Fucoidan inhibits cellular and neurotoxic effects of beta-amyloid (A beta) in rat cholinergic basal forebrain neurons. Eur. J. Neurosci. 2005, 21, 2649–2659. [Google Scholar] [CrossRef] [PubMed]
- Luo, D.; Zhang, Q.; Wang, H.; Cui, Y.; Sun, Z.; Yang, J.; Zheng, Y.; Jia, J.; Yu, F.; Wang, X.; et al. Fucoidan protects against dopaminergic neuron death in vivo and in vitro. Eur. J. Pharmacol. 2009, 617, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Dong, C.; Yin, J.; Shen, J.; Tian, J.; Li, C. Neuroprotective effect of fucoidan on H2O2-induced apoptosis in PC12 cells via activation of PI3K/Akt pathway. Cell. Mol. Neurobiol. 2012, 32, 523–529. [Google Scholar] [CrossRef]
- Wei, H.; Gao, Z.; Zheng, L.; Zhang, C.; Liu, Z.; Yang, Y.; Teng, H.; Hou, L.; Yin, Y.; Zou, X. Protective effects of fucoidan on Aβ25-35 and d-Gal-induced neurotoxicity in PC12 cells and d-Gal-induced cognitive dysfunction in mice. Mar. Drugs 2017, 15, 77. [Google Scholar] [CrossRef]
- Liang, Z.; Liu, Z.; Sun, X.; Tao, M.; Xiao, X.; Yu, G.; Wang, X. The Effect of Fucoidan on cellular oxidative stress and the CatD-Bax signaling axis in MN9D cells damaged by 1-Methyl-4-Phenypyridinium. Front. Aging Neurosci. 2019, 10, 429. [Google Scholar] [CrossRef]
- Wu, H.; Gao, S.; Terakawa, S. Inhibitory effects of fucoidan on NMDA receptors and l-type Ca(2+) channels regulating the Ca(2+) responses in rat neurons. Pharm. Biol. 2019, 57, 1–7. [Google Scholar] [CrossRef]
- Liu, Y.; Deng, Z.; Geng, L.; Wang, J.; Zhang, Q. In vitro evaluation of the neuroprotective effect of oligo-porphyran from Porphyra yezoensis in PC12 cells. J. Appl. Phycol. 2019, 31, 2559–2571. [Google Scholar] [CrossRef]
- Wang, S.; Li, J.; Xia, W.; Geng, M. A marine-derived acidic oligosaccharide sugar chain specifically inhibits neuronal cell injury mediated by beta-amyloid-induced astrocyte activation in vitro. Neurol. Res. 2007, 29, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.Q.; Mao, S.C.; Zhang, H.Y.; Yu, X.Q.; Feng, M.T.; Wang, B.; Feng, L.H.; Guo, Y.W. Racemosins A and B, two novel bisindole alkaloids from the green alga Caulerpa racemosa. Fitoterapia 2013, 91, 15–20. [Google Scholar] [CrossRef]
- Wu, S.; Yue, Y.; Tian, H.; Tao, L.; Wang, Y.; Xiang, J.; Wang, S.; Ding, H. Tramiprosate protects neurons against ischemic stroke by disrupting the interaction between PSD95 and nNOS. Neuropharmacology 2014, 83, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jiang, L.; Li, X. κ-carrageenan-derived pentasaccharide attenuates Aβ25-35-induced apoptosis in SH-SY5Y cells via suppression of the JNK signaling pathway. Mol. Med. Rep. 2017, 15, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Li, C.; Yin, J.; Shen, J.; Wang, H.; Wu, Y.; Jin, H. Fucoidan, a sulfated polysaccharide from brown algae, improves cognitive impairment induced by infusion of Aβ peptide in rats. Environ. Toxicol. Pharmacol. 2012, 33, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yi, K.; Zhao, Y. Fucoidan inhibits amyloid-β-induced toxicity in transgenic Caenorhabditis elegans by reducing the accumulation of amyloid-β and decreasing the production of reactive oxygen species. Food Funct. 2018, 9, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Park, S.K.; Kang, J.Y.; Kim, J.M.; Yoo, S.K.; Han, H.J.; Chung, D.H.; Kim, D.O.; Kim, G.H.; Heo, H.J. Fucoidan-rich substances from ecklonia cava improve trimethyltin-induced cognitive dysfunction via down-regulation of amyloid β production/Tau hyperphosphorylation. Mar. Drugs 2019, 17, 591. [Google Scholar] [CrossRef]
- Kim, H.; Ahn, J.H.; Song, M.; Kim, D.W.; Lee, T.K.; Lee, J.C.; Kim, Y.M.; Kim, J.D.; Cho, J.H.; Hwang, I.K.; et al. Pretreated fucoidan confers neuroprotection against transient global cerebral ischemic injury in the gerbil hippocampal CA1 area via reducing of glial cell activation and oxidative stress. Biomed. Pharmacother. 2019, 109, 1718–1727. [Google Scholar] [CrossRef]
- Lee, T.K.; Ahn, J.H.; Park, C.W.; Kim, B.; Park, Y.E.; Lee, J.C.; Park, J.H.; Yang, G.E.; Shin, M.C.; Cho, J.H.; et al. Pre-treatment with laminarin protects hippocampal ca1 pyramidal neurons and attenuates reactive gliosis following transient forebrain ischemia in gerbils. Mar. Drugs 2020, 18, 52. [Google Scholar] [CrossRef]
- Park, J.H.; Ahn, J.H.; Lee, T.K.; Park, C.W.; Kim, B.; Lee, J.C.; Kim, D.W.; Shin, M.C.; Cho, J.H.; Lee, C.H.; et al. Laminarin pretreatment provides neuroprotection against forebrain ischemia/reperfusion injury by reducing oxidative stress and neuroinflammation in aged gerbils. Mar. Drugs 2020, 18, 213. [Google Scholar] [CrossRef]
- Liu, Y.; Geng, L.; Zhang, J.; Wang, J.; Zhang, Q.; Duan, D.; Zhang, Q. Oligo-Porphyran ameliorates neurobehavioral deficits in parkinsonian mice by regulating the PI3K/Akt/Bcl-2 pathway. Mar. Drugs 2018, 16, 82. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, X.; Pan, Y.; Wang, G.; Mao, G. The degraded polysaccharide from Pyropia haitanensis represses amyloid beta peptide-induced neurotoxicity and memory in vivo. Int. J. Biol. Macromol. 2020, 146, 725–729. [Google Scholar] [CrossRef]
- Pentón-Rol, G.; Marín-Prida, J.; Pardo-Andreu, G.; Martínez-Sánchez, G.; Acosta-Medina, E.F.; Valdivia-Acosta, A.; Lagumersindez-Denis, N.; Rodríguez-Jiménez, E.; Llópiz-Arzuaga, A.; López-Saura, P.A.; et al. C-Phycocyanin is neuroprotective against global cerebral ischemia/reperfusion injury in gerbils. Brain. Res. Bull. 2011, 86, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Souza, R.B.; Frota, A.F.; Sousa, R.S.; Cezario, N.A.; Santos, T.B.; Souza, L.M.; Coura, C.O.; Monteiro, V.S.; Cristino Filho, G.; Vasconcelos, S.M.; et al. Neuroprotective effects of sulphated agaran from marine alga Gracilaria cornea in Rat 6-Hydroxydopamine parkinson’s disease model: Behavioural, neurochemical and transcriptional alterations. Basic Clin. Pharmacol. Toxicol. 2017, 120, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Yi, M.; Ding, L.; He, S.; Yan, X. Isolation and purification of a neuroprotective phlorotannin from the marine algae Ecklonia maxima by size exclusion and high-speed counter-current chromatography. Mar. Drugs 2019, 17, 212. [Google Scholar] [CrossRef] [PubMed]
- Dimitrova-Shumkovska, J.; Krstanoski, L.; Veenman, L. Potential beneficial actions of fucoidan in brain and liver injury, disease, and intoxication—Potential implication of sirtuins. Mar. Drugs 2020, 18, 242. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.N.; Chen, P.W.; Huang, C.Y. Compositional characteristics and in vitro evaluations of antioxidant and neuroprotective properties of crude extracts of fucoidan prepared from compressional puffing-pretreated Sargassum crassifolium. Mar. Drugs 2017, 15, 183. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Kuo, C.H.; Chen, P.W. Compressional-puffing pretreatment enhances neuroprotective effects of fucoidans from the brown seaweed Sargassum hemiphyllum on 6-Hydroxydopamine-induced apoptosis in SH-SY5Y cells. Molecules 2017, 23, 78. [Google Scholar] [CrossRef]
- Souza, R.B.; Frota, A.F.; Silva, J.; Alves, C.; Neugebauer, A.Z.; Pinteus, S.; Rodrigues, J.A.G.; Cordeiro, E.M.S.; de Almeida, R.R.; Pedrosa, R.; et al. In vitro activities of kappa-carrageenan isolated from red marine alga Hypnea musciformis: Antimicrobial, anticancer and neuroprotective potential. Int. J. Biol. Macromol. 2018, 112, 1248–1256. [Google Scholar] [CrossRef]
- Tsang, C.K.; Kamei, Y. Sargaquinoic acid supports the survival of neuronal PC12D cells in a nerve growth factor-independent manner. Eur. J. Pharmacol. 2004, 488, 11–18. [Google Scholar] [CrossRef]
- Tsolaki, M. Future strategies of management of Alzheimer’s Disease. The role of homotaurine. Hell. J. Nucl. Med. 2019, 22, 82–94. [Google Scholar]
- Ricciardi, L.; De Nigris, F.; Specchia, A.; Fasano, A. Homotaurine in Parkinson’s disease. Neurol. Sci. 2015, 36, 1581–1587. [Google Scholar] [CrossRef]
- Oh, J.H.; Kim, E.Y.; Nam, T.J. Phycoerythrin peptide from Pyropia yezoensis alleviates endoplasmic reticulum stress caused by perfluorooctane sulfonate-induced calcium dysregulation. Mar. Drugs 2018, 16, 44. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.H.; Kim, E.Y.; Nam, T.J. Phycoerythrin-derived tryptic peptide of a red alga Pyropia yezoensis attenuates glutamate-induced er stress and neuronal senescence in primary rat hippocampal neurons. Mol. Nutr. Food Res. 2018, 62, e1700469. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.L.; Zhang, H.; Feng, M.; Yang, H.; Yang, P.; Lin, K.; Guo, Y.; Mao, S. The fatty acids of green alga Caulerpa racemosa and their bioactivities. Zhongguo Haiyang Yaowu 2013, 32, 13–20. [Google Scholar]
- Silva, J.; Alves, C.; Pinteus, S.; Mendes, S.; Pedrosa, R. Neuroprotective effects of seaweeds against 6-hydroxidopamine-induced cell death on an in vitro human neuroblastoma model. BMC Complement. Altern. Med. 2018, 18, 58. [Google Scholar] [CrossRef]
- Silva, J.; Alves, C.; Freitas, R.; Martins, A.; Pinteus, S.; Ribeiro, J.; Gaspar, H.; Alfonso, A.; Pedrosa, R. Antioxidant and neuroprotective potential of the brown seaweed Bifurcaria bifurcata in an in vitro Parkinson’s Disease model. Mar. Drugs 2019, 17, 85. [Google Scholar] [CrossRef]
- Chacko, A.; Ittiyavirah, S.P. Pharmacology, neuroprotective effect of against aluminium-induced Gracilaria corticata neurotoxicity in the hippocampus and cerebral cortex of rat brain: Biochemical and histological approach. J. Pharm. Pharmacol. 2019, 5, 604–613. [Google Scholar]
- Alghazwi, M.; Smid, S.; Zhang, W. In vitro protective activity of South Australian marine sponge and macroalgae extracts against amyloid beta (Aβ(1-42)) induced neurotoxicity in PC-12 cells. Neurotoxicol. Teratol. 2018, 68, 72–83. [Google Scholar] [CrossRef]
- Kim, J.; Moon, I.S.; Goo, T.W.; Moon, S.S.; Seo, M. Algae Undaria pinnatifida protects hypothalamic neurons against endoplasmic reticulum stress through Akt/mTOR signaling. Molecules 2015, 20, 20998–21009. [Google Scholar] [CrossRef]
- Mohibbullah, M.; Hannan, M.A.; Choi, J.Y.; Bhuiyan, M.M.H.; Hong, Y.K.; Choi, J.S.; Choi, I.S.; Moon, I.S. The edible marine alga Gracilariopsis chorda alleviates hypoxia/reoxygenation-induced oxidative stress in cultured hippocampal neurons. J. Med. Food 2015, 18, 960–971. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, I.; Cao, M.; Su, Z.Y.; Wu, R.; Guo, Y.; Fang, M.; Kong, A.N. Fucoxanthin elicits epigenetic modifications, nrf2 activation and blocking transformation in mouse skin JB6 P+ Cells. AAPS J. 2018, 20, 32. [Google Scholar] [CrossRef]
- Oh, J.H.; Nam, T.J. Hydrophilic glycoproteins of an edible green alga Capsosiphon fulvescens prevent aging-induced spatial memory impairment by suppressing gsk-3beta-mediated er stress in dorsal hippocampus. Mar. Drugs 2019, 17, 168. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.H.; Nam, T.J.; Choi, Y.H. Capsosiphon fulvescens Glycoproteins Enhance Probiotics-Induced Cognitive Improvement in Aged Rats. Nutrients 2020, 12, 837. [Google Scholar] [CrossRef] [PubMed]
- Nisha, S.A.; Devi, K.P. Gelidiella acerosa protects against Aβ 25-35-induced toxicity and memory impairment in Swiss Albino mice: An in vivo report. Pharm. Biol. 2017, 55, 1423–1435. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, P.J.A.; Khan, A.; Uddin, N.; Khaliq, S.; Rasheed, M.; Nawaz, S.; Dar, A.; Hanif, M. Sargassum swartzii extracts ameliorate memory functions by neurochemical modulation in a rat model. Food Sci. Biotechnol. 2017, 26, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.Y.; Mohibbullah, M.; Park, I.S.; Moon, I.S.; Hong, Y.K. An ethanol extract from the phaeophyte Undaria pinnatifida improves learning and memory impairment and dendritic spine morphology in hippocampal neurons. J. Appl. Phycol. 2018, 30, 129–136. [Google Scholar] [CrossRef]
- El-Baz, F.K.; Aly, H.F.; Ali, G.H. Haematococcus pluvialis modulating effect on neurotransmitters, hormones and oxidative damage-associated with alzheimers disease in experimental rats model. Int. J. Pharm. Pharm. Sci. 2017, 9, 198–206. [Google Scholar] [CrossRef]
- Choi, W.Y.; Kang, D.H.; Heo, S.J.; Lee, H.Y. Enhancement of the neuroprotective effect of fermented Spirulina maxima associated with antioxidant activities by ultrasonic extraction. Appl. Sci. 2018, 8, 2469. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, N.S.; Jeong, Y.G.; Lee, J.H.; Kim, E.J.; Han, S.Y. Protective efficacy of an Ecklonia cava extract used to treat transient focal ischemia of the rat brain. Anat. Cell. Biol. 2012, 45, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Jiang, R.W.; Du, X.G.; Zhang, X.; Wang, X.; Hu, D.Y.; Meng, T.; Chen, Y.L.; Geng, M.Y.; Shen, J.K. Synthesis and bioassay of β-(1,4)-D-mannans as potential agents against Alzheimer’s disease. Acta Pharmacol. Sin. 2013, 34, 1585–1591. [Google Scholar] [CrossRef]
- Wang, X.; Sun, G.; Feng, T.; Zhang, J.; Huang, X.; Wang, T.; Xie, Z.; Chu, X.; Yang, J.; Wang, H.; et al. Sodium oligomannate therapeutically remodels gut microbiota and suppresses gut bacterial amino acids-shaped neuroinflammation to inhibit Alzheimer’s disease progression. Cell Res. 2019, 29, 787–803. [Google Scholar] [CrossRef]
- Xiao, S. A phase II clinical trial on GV-971 in patients with Alzheimer’s [abstract no. OC 3]. J. Prev. Alz. Dis. 2014, 1, 214–296. [Google Scholar]
- Xiao, S.; Zhang, Z.; Geng, M. Phase 3 Clinical trial of a novel and multi-targeted oligosaccharide in patients with mildmoderate ad in china. China J. Prev. Alzheimers Dis. 2018, 5, S10. [Google Scholar]
- Davis, G.D.; Vasanthi, A.H. Seaweed metabolite database (SWMD): A database of natural compounds from marine algae. Bioinformation 2011, 5, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Lei, J.; Zhou, J. A marine natural product database. J. Chem. Inf. Comput. Sci. 2002, 42, 742–748. [Google Scholar] [CrossRef]
- Babu, P.A.; Puppala, S.S.; Aswini, S.L.; Vani, M.R.; Kumar, C.N.; Prasanna, T. A database of natural products and chemical entities from marine habitat. Bioinformation 2008, 3, 142–143. [Google Scholar] [CrossRef][Green Version]
- Barbosa, A.J.M.; Roque, A.C.A. Free marine natural products databases for biotechnology and bioengineering. Biotechnol. J. 2019, 14, 1800607. [Google Scholar] [CrossRef]
- Dictionary of Marine Natural Products. Available online: http://dmnp.chemnetbase.com/faces/chemical/ChemicalSearch.xhtml (accessed on 20 May 2020).
- MarinLit, A Database of the Marine Natural Products Literature. Available online: http://pubs.rsc.org/marinlit (accessed on 20 May 2020).
- Keller, T.H.; Pichota, A.; Yin, Z. A practical view of ‘druggability’. Curr. Opin. Chem. Biol. 2006, 10, 357–361. [Google Scholar] [CrossRef]
- Wale, N.; Karypis, G. Target fishing for chemical compounds using target-ligand activity data and ranking based methods. J. Chem. Inf. Model. 2009, 49, 2190–2201. [Google Scholar] [CrossRef]
- Huang, H.; Zhang, G.; Zhou, Y.; Lin, C.; Chen, S.; Lin, Y.; Mai, S.; Huang, Z. Reverse screening methods to search for the protein targets of chemopreventive compounds. Front. Chem. 2018, 6, 138. [Google Scholar] [CrossRef]
- Dunkel, M.; Fullbeck, M.; Neumann, S.; Preissner, R. SuperNatural: A searchable database of available natural compounds. Nucleic Acids Res. 2006, 34, D678–D683. [Google Scholar] [CrossRef]
- Huang, L.; Xie, D.; Yu, Y.; Liu, H.; Shi, Y.; Shi, T.; Wen, C. TCMID 2.0: A comprehensive resource for TCM. Nucleic Acids Res. 2017, 46, D1117–D1120. [Google Scholar] [CrossRef] [PubMed]
- Ru, J.; Li, P.; Wang, J.; Zhou, W.; Li, B.; Huang, C.; Li, P.; Guo, Z.; Tao, W.; Yang, Y.; et al. TCMSP: A database of systems pharmacology for drug discovery from herbal medicines. J. Cheminform. 2014, 6, 13. [Google Scholar] [CrossRef] [PubMed]
- Pereira, F.; Aires-de-Sousa, J. Computational methodologies in the exploration of marine natural product leads. Mar. Drugs 2018, 16, 236. [Google Scholar] [CrossRef] [PubMed]
- Sorokina, M.; Steinbeck, C. Review on natural products databases: Where to find data in 2020. J. Cheminform. 2020, 12, 20. [Google Scholar] [CrossRef]
- Vitale, R.M.; D’Aniello, E.; Gorbi, S.; Martella, A.; Silvestri, C.; Giuliani, M.E.; Fellous, T.; Gentile, A.; Carbone, M.; Cutignano, A.; et al. Fishing for targets of alien metabolites: A novel peroxisome proliferator-activated receptor (PPAR) agonist from a marine pest. Mar. Drugs 2018, 16, 431. [Google Scholar] [CrossRef]
- Chamberlin, S.R.; Blucher, A.; Wu, G.; Shinto, L.; Choonoo, G.; Kulesz-Martin, M.; McWeeney, S. Natural product target network reveals potential for cancer combination therapies. Front. Pharmacol. 2019, 10, 557. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.A.; Roy, A.; Choi, J.S. In vitro monoamine oxidase A and B inhibitory activity and molecular docking simulations of fucoxanthin. Fish. Sci. 2017, 83, 123–132. [Google Scholar] [CrossRef]
- Paudel, P.; Seong, S.H.; Jung, H.A.; Choi, J.S. Characterizing fucoxanthin as a selective dopamine D(3)/D(4) receptor agonist: Relevance to Parkinson’s disease. Chem. Biol. Interact. 2019, 310, 108757. [Google Scholar] [CrossRef]
- Paudel, P.; Park, S.E.; Seong, S.H.; Jung, H.A.; Choi, J.S. Bromophenols from symphyocladia latiuscula target human monoamine oxidase and dopaminergic receptors for the management of neurodegenerative diseases. J. Agric. Food Chem. 2020, 68, 2426–2436. [Google Scholar] [CrossRef]
- Floresta, G.; Amata, E.; Barbaraci, C.; Gentile, D.; Turnaturi, R.; Marrazzo, A.; Rescifina, A. A Structure- and Ligand-Based Virtual Screening of a Database of “Small” Marine Natural Products for the Identification of “Blue” Sigma-2 Receptor Ligands. Mar. Drugs 2018, 16, 10. [Google Scholar] [CrossRef]
- Sá Monteiro, M.; Sloth, J.J.; Holdt, S.L.; Hansen, M. Analysis and risk assessment of seaweed. EFSA J. 2019, 17, e170915. [Google Scholar]
- Hwang, P.A.; Yan, M.D.; Lin, H.T.; Li, K.L.; Lin, Y.C. Toxicological evaluation of low molecular weight fucoidan in vitro and in vivo. Mar. Drugs 2016, 14, 7. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zhang, Q.; Song, J. Toxicological evaluation of fucoidan extracted from Laminaria japonica in Wistar rats. Food Chem. Toxicol. 2005, 43, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.J.; Jeun, J.; Houng, S.J.; Jun, H.J.; Kweon, D.K.; Lee, S.J. Toxicological evaluation of fucoidan from Undaria pinnatifida in vitro and in vivo. Phytother. Res. 2010, 24, 1078–1083. [Google Scholar] [PubMed]
- Kim, K.J.; Lee, O.H.; Lee, B.Y. Genotoxicity studies on fucoidan from Sporophyll of Undaria pinnatifida. Food Chem. Toxicol. 2010, 48, 1101–1104. [Google Scholar] [CrossRef]
- Kim, K.J.; Lee, O.H.; Lee, H.H.; Lee, B.Y. A 4-week repeated oral dose toxicity study of fucoidan from the Sporophyll of Undaria pinnatifida in Sprague-Dawley rats. Toxicology 2010, 267, 154–158. [Google Scholar] [CrossRef]
- Myers, S.P.; O’Connor, J.; Fitton, J.H.; Brooks, L.; Rolfe, M.; Connellan, P.; Wohlmuth, H.; Cheras, P.A.; Morris, C. A combined phase I and II open label study on the effects of a seaweed extract nutrient complex on osteoarthritis. Biologics 2010, 4, 33–44. [Google Scholar] [CrossRef]
- Myers, S.P.; Mulder, A.M.; Baker, D.G.; Robinson, S.R.; Rolfe, M.I.; Brooks, L.; Fitton, J.H. Effects of fucoidan from fucus vesiculosus in reducing symptoms of osteoarthritis: A randomized placebo-controlled trial. Biologics 2016, 10, 81–88. [Google Scholar]
- Weiner, M.L. Food additive carrageenan: Part II: A critical review of carrageenan in vivo safety studies. Crit. Rev. Toxicol. 2014, 44, 244–269. [Google Scholar] [CrossRef]
- Hebar, A.; Koller, C.; Seifert, J.M.; Chabicovsky, M.; Bodenteich, A.; Bernkop-Schnurch, A.; Grassauer, A.; Prieschl-Grassauer, E. Non-clinical safety evaluation of intranasal iota-carrageenan. PLoS ONE 2015, 10, e0122911. [Google Scholar] [CrossRef]
- Beppu, F.; Niwano, Y.; Tsukui, T.; Hosokawa, M.; Miyashita, K. Single and repeated oral dose toxicity study of fucoxanthin (FX), a marine carotenoid, in mice. J. Toxicol. Sci. 2009, 34, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Kadekaru, T.; Toyama, H.; Yasumoto, T. Safety Evaluation of Fucoxanthin purified from Undaria pinnatifida. Nippon Shokuhin Kagaku Kogaku Kaishi 2008, 55, 304–308. [Google Scholar] [CrossRef]
- Beppu, F.; Niwano, Y.; Sato, E.; Kohno, M.; Tsukui, T.; Hosokawa, M.; Miyashita, K. In vitro and in vivo evaluation of mutagenicity of fucoxanthin (FX) and its metabolite fucoxanthinol (FXOH). J. Toxicol. Sci. 2009, 34, 693–698. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).