Mycotoxin Identification and In Silico Toxicity Assessment Prediction in Atlantic Salmon
Abstract
1. Introduction
2. Results and Discussion
2.1. Mycotoxin Identification by Non-Target Screening
2.2. In Silico Toxicity Prediction
ProTox-II
3. Materials and Methods
3.1. Samples
3.2. Mycotoxin Extraction and LC-Q-TOF-MS Analysis
3.3. Non-Targeted Suspect Screening (TOF)
3.4. In Silico Prediction Methods
3.4.1. Acute Oral Toxicity Prediction
- Class I: fatal if swallowed (LD50 ≤ 5 mg/kg);
- Class II: fatal if swallowed (5 mg/kg < LD50 ≤ 50 mg/kg);
- Class III: toxic if swallowed (50 mg/kg < LD50 ≤ 300 mg/kg);
- Class IV: harmful if swallowed (300 mg/kg < LD50 ≤ 2000 mg/kg);
- Class V: may be harmful if swallowed (2000 mg/kg < LD50 ≤ 5000 mg/kg).
3.4.2. Toxicity Endpoint and Organ Toxicity Prediction
3.4.3. Toxicological Pathways
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nizza, A.; Piccolo, G. Chemical-nutritional characteristics of diets in aquaculture. Veter. Res. Commun. 2009, 33, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Binder, E.M. Managing the risk of mycotoxins in modern feed production. Anim. Feed. Sci. Technol. 2007, 133, 149–166. [Google Scholar] [CrossRef]
- Binder, E.; Tan, L.; Chin, L.; Handl, J.; Richard, J. Worldwide occurrence of mycotoxins in commodities, feeds and feed ingredients. Anim. Feed. Sci. Technol. 2007, 137, 265–282. [Google Scholar] [CrossRef]
- Zinedine, A.; Mañes, J. Occurrence and legislation of mycotoxins in food and feed from Morocco. Food Control. 2009, 20, 334–344. [Google Scholar] [CrossRef]
- Rodrigues, I.; Naehrer, K. A three-year survey on the worldwide occurrence of mycotoxins in feedstuffs and feed. Toxins 2012, 4, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Streit, E.; Schatzmayr, G.; Tassis, P.; Tzika, E.D.; Marin, D.E.; Taranu, I.; Tabuc, C.; Nicolau, A.I.; Aprodu, I.; Puel, O.; et al. Current situation of mycotoxin contamination and co-occurrence in animal feed—Focus on Europe. Toxins 2012, 4, 788–809. [Google Scholar] [CrossRef]
- Streit, E.; Schwab, C.; Sulyok, M.; Naehrer, K.; Krska, R.; Schatzmayr, G. Multi-mycotoxin screening reveals the occurrence of 139 different secondary metabolites in feed and feed ingredients. Toxins 2013, 5, 504–523. [Google Scholar] [CrossRef]
- Nácher-Mestre, J.; Serrano, R.; Beltran, E.; Pérez-Sánchez, J.; Silva, J.; Karalazos, V.; Hernández, F.; Berntssen, M. Occurrence and potential transfer of mycotoxins in gilthead sea bream and Atlantic salmon by use of novel alternative feed ingredients. Chemosphere 2015, 128, 314–320. [Google Scholar] [CrossRef]
- Tolosa, J.; Font, G.; Mañes, J.; Ferrer, E. Natural occurrence of emerging fusarium mycotoxins in feed and fish from aquaculture. J. Agric. Food Chem. 2014, 62, 12462–12470. [Google Scholar] [CrossRef]
- Nomura, H.; Ogiso, M.; Yamashita, M.; Takaku, H.; Kimura, A.; Chikasou, M.; Nakamura, Y.; Fujii, S.; Watai, M.; Yamada, H. Uptake by dietary exposure and elimination of aflatoxins in muscle and liver of rainbow trout (Oncorhynchus mykiss). J. Agric. Food Chem. 2011, 59, 5150–5158. [Google Scholar] [CrossRef]
- Bernhoft, A.; Høgåsen, H.R.; Rosenlund, G.; Moldal, T.; Grove, S.; Berntssen, M.; Thoresen, S.I.; Alexander, J. Effects of dietary deoxynivalenol or ochratoxin A on performance and selected health indices in Atlantic salmon (Salmo salar). Food Chem. Toxicol. 2018, 121, 374–386. [Google Scholar] [CrossRef] [PubMed]
- Bernhoft, A.; Høgåsen, H.R.; Rosenlund, G.; Ivanova, L.; Berntssen, M.H.G.; Alexander, J.; Eriksen, G.S.; Fæste, C.K. Tissue distribution and elimination of deoxynivalenol and ochratoxin A in dietary-exposed Atlantic salmon (Salmo salar). Food Addit. Contam. Part A 2017, 34, 1211–1224. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Zhou, T.; Young, J.C.; Boland, G.J.; Scott, P.M. Chemical and biological transformations for detoxification of trichothecene mycotoxins in human and animal food chains: A review. Trends Food Sci. Technol. 2010, 21, 67–76. [Google Scholar] [CrossRef]
- Caruso, D.; Talamond, P.; Moreau, Y. Mycotoxinesetpisciculture: Un risqueoublié? CAH Agric. 2013, 22, 165–173. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture 2016. Contributing to Food Security and Nutrition for All; FAO: Rome, Italy, 2016; p. 200. ISBN 978-92-5-109185-2. Available online: http://www.fao.org/3/a-i5555e.pdf (accessed on 2 March 2019).
- European Commission. Commission Regulation (EC) No. 1881/2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs. Off. J. Eur. Union 2006, 364, 5–24. [Google Scholar]
- Zhao, Z.; Liu, N.; Yang, L.; Deng, Y.; Wang, J.; Song, S.; Lin, S.; Wu, A.; Zhou, Z.; Hou, J.-F. Multi-mycotoxin analysis of animal feed and animal-derived food using LC–MS/MS system with timed and highly selective reaction monitoring. Anal. Bioanal. Chem. 2015, 407, 7359–7368. [Google Scholar] [CrossRef]
- Abdallah, M.F.; Girgin, G.; Baydar, T.; Krska, R.; Sulyok, M. Occurrence of multiple mycotoxins and other fungal metabolites in animal feed and maize samples from Egypt using LC-MS/MS. J. Sci. Food Agric. 2017, 97, 4419–4428. [Google Scholar] [CrossRef]
- Åberg, A.T.; Solyakov, A.; Bondesson, U. Development and in-house validation of an LC-MS/MS method for the quantification of the mycotoxins deoxynivalenol, zearalenone, T-2 and HT-2 toxin, ochratoxin A and fumonisin B1 and B2 in vegetable animal feed. Food Addit. Contam. Part A 2013, 30, 541–549. [Google Scholar] [CrossRef]
- Mol, H.G.J.; Plaza-Bolanños, P.; Zomer, P.; De Rijk, T.C.; Stolker, A.A.M.; Mulder, P.P.J. Toward a generic extraction method for simultaneous determination of pesticides, mycotoxins, plant toxins, and veterinary drugs in feed and food matrixes. Anal. Chem. 2008, 80, 9450–9459. [Google Scholar] [CrossRef]
- Drwal, M.N.; Banerjee, P.; Dunkel, M.; Wettig, M.R.; Preissner, R. ProTox: A web server for the in silico prediction of rodent oral toxicity. Nucleic Acids Res. 2014, 42, W53–W58. [Google Scholar] [CrossRef]
- Quesada, S.P.; Paschoal, J.A.R.; Reyes, F.G.R. A simple method for the determination of fluoroquinolone residues in tilapia (Oreochromis niloticus) and pacu (Piaractus mesopotamicus) employing LC-MS/MS QToF. Food Addit. Contam. Part. A 2013, 30, 813–825. [Google Scholar] [CrossRef]
- Liao, C.-D.; Wong, J.W.; Zhang, K.; Yang, P.; Wittenberg, J.B.; Trucksess, M.W.; Hayward, D.G.; Lee, N.S.; Chang, J.S. Multi-mycotoxin analysis of finished grain and nut products using ultrahigh-performance liquid chromatography and positive electrospray ionization–quadrupole orbital ion trap high-resolution mass spectrometry. J. Agric. Food Chem. 2015, 63, 8314–8332. [Google Scholar] [CrossRef]
- Cirlini, M.; Dall’Asta, C.; Galaverna, G. Hyphenated chromatographic techniques for structural characterization and determination of masked mycotoxins. J. Chromatogr. A 2012, 1255, 145–152. [Google Scholar] [CrossRef]
- Nácher-Mestre, J.; Ibáñez, M.V.; Serrano, R.; Pérez-Sánchez, J.; Hernández, F. Qualitative screening of undesirable compounds from feeds to fish by liquid chromatography coupled to mass spectrometry. J. Agric. Food Chem. 2013, 61, 2077–2087. [Google Scholar] [CrossRef]
- Aresta, A.; Cioffi, N.; Palmisano, F.; Zambonin, C.G. Simultaneous determination of ochratoxin A and cyclopiazonic, mycophenolic, and tenuazonic acids in cornflakes by solid-phase microextraction coupled to high-performance liquid chromatography. J. Agric. Food Chem. 2003, 51, 5232–5237. [Google Scholar] [CrossRef]
- Sulyok, M.; Krska, R.; Schuhmacher, R. Application of an LC–MS/MS based multi-mycotoxin method for the semi-quantitative determination of mycotoxins occurring in different types of food infected by moulds. Food Chem. 2010, 119, 408–416. [Google Scholar] [CrossRef]
- Abdallah, M.F.; Krska, R.; Sulyok, M. Occurrence of ochratoxins, fumonisin B2, aflatoxins (B1and B2), and other secondary fungal metabolites in dried date palm fruits from Egypt: A Mini-Survey. J. Food Sci. 2018, 83, 559–564. [Google Scholar] [CrossRef]
- Rundberget, T.; Wilkins, A.L. Determination of Penicillium mycotoxins in foods and feeds using liquid chromatography-mass spectrometry. J. Chromatogr. A 2002, 964, 189–197. [Google Scholar] [CrossRef]
- Sulyok, M.; Berthiller, F.; Krska, R.; Schuhmacher, R. Development and validation of a liquid chromatography/tandem mass spectrometric method for the determination of 39 mycotoxins in wheat and maize. Rapid Commun. Mass Spectrom. 2006, 20, 2649–2659. [Google Scholar] [CrossRef]
- El-Sayed, Y.S.; Khalil, R.H. Toxicity, biochemical effects and residue of aflatoxin B1 in marine water-reared sea bass (Dicentrarchus labrax L.). Food Chem. Toxicol. 2009, 47, 1606–1609. [Google Scholar] [CrossRef]
- Guan, S.; He, J.; Young, J.C.; Zhu, H.; Li, X.-Z.; Ji, C.; Zhou, T. Transformation of trichothecene mycotoxins by microorganisms from fish digesta. Aquaculture 2009, 290, 290–295. [Google Scholar] [CrossRef]
- Deng, S.X.; Tian, L.X.; Liu, F.J.; Jin, S.J.; Liang, G.Y.; Yang, H.J.; Du, Z.Y.; Liu, Y.J. Toxic effects and residue of aflatoxin B1 in tilapia (Oreochromis niloticus × O. aureus) during long-term dietary exposure. Aquaculture 2010, 307, 233–240. [Google Scholar] [CrossRef]
- Pietsch, C.; Michel, C.; Kersten, S.; Valenta, H.; Dänicke, S.; Schulz, C.; Kloas, W.; Burkhardt-Holm, P. In vivo effects of deoxynivalenol (DON) on innate immune responses of carp (Cyprinus carpio L.). Food Chem. Toxicol. 2014, 68, 44–52. [Google Scholar] [CrossRef]
- Tola, S.; Bureau, D.P.; Hooft, J.M.; Beamish, F.W.; Sulyok, M.; Krska, R.; Encarnação, P.; Petkam, R. Effects of wheat naturally contaminated with fusarium mycotoxins on growth performance and selected health indices of red tilapia (Oreochromis niloticus × O. mossambicus). Toxins 2015, 7, 1929–1944. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the risks for animal and public health related to the presence of T-2 and HT-2 toxin in food and feed. EFSA J. 2011, 9, 2481. Available online: https://efsa.onlinelibrary.wiley.com/doi/epdf/ (accessed on 16 April 2019). [CrossRef]
- Prosperini, A.; Font, G.; Ruiz, M. Interaction effects of Fusarium enniatins (A, A1, B and B1) combinations on in vitro cytotoxicity of Caco-2 cells. Toxicol. Vitr. 2014, 28, 88–94. [Google Scholar] [CrossRef]
- Juan-García, A.; Manyes, L.; Ruiz, M.-J.; Font, G. Involvement of enniatins-induced cytotoxicity in human HepG2 cells. Toxicol. Lett. 2013, 218, 166–173. [Google Scholar] [CrossRef]
- Au, T.K.; Wallace, S.H.; Leung, P.C. The biology of ophiobolins. Life Sci. 2000, 67, 733–742. [Google Scholar] [CrossRef]
- Tolosa, J.; Font, G.; Mañes, J.; Ferrer, E. Mitigation of enniatins in edible fish tissues by thermal processes and identification of degradation products. Food Chem. Toxicol. 2017, 101, 67–74. [Google Scholar] [CrossRef]
- Banerjee, P.; Eckert, A.O.; Schrey, A.K.; Preissner, R. ProTox-II: A webserver for the prediction of toxicity of chemicals. Nucleic Acids Res. 2018, 46, W257–W263. [Google Scholar] [CrossRef]
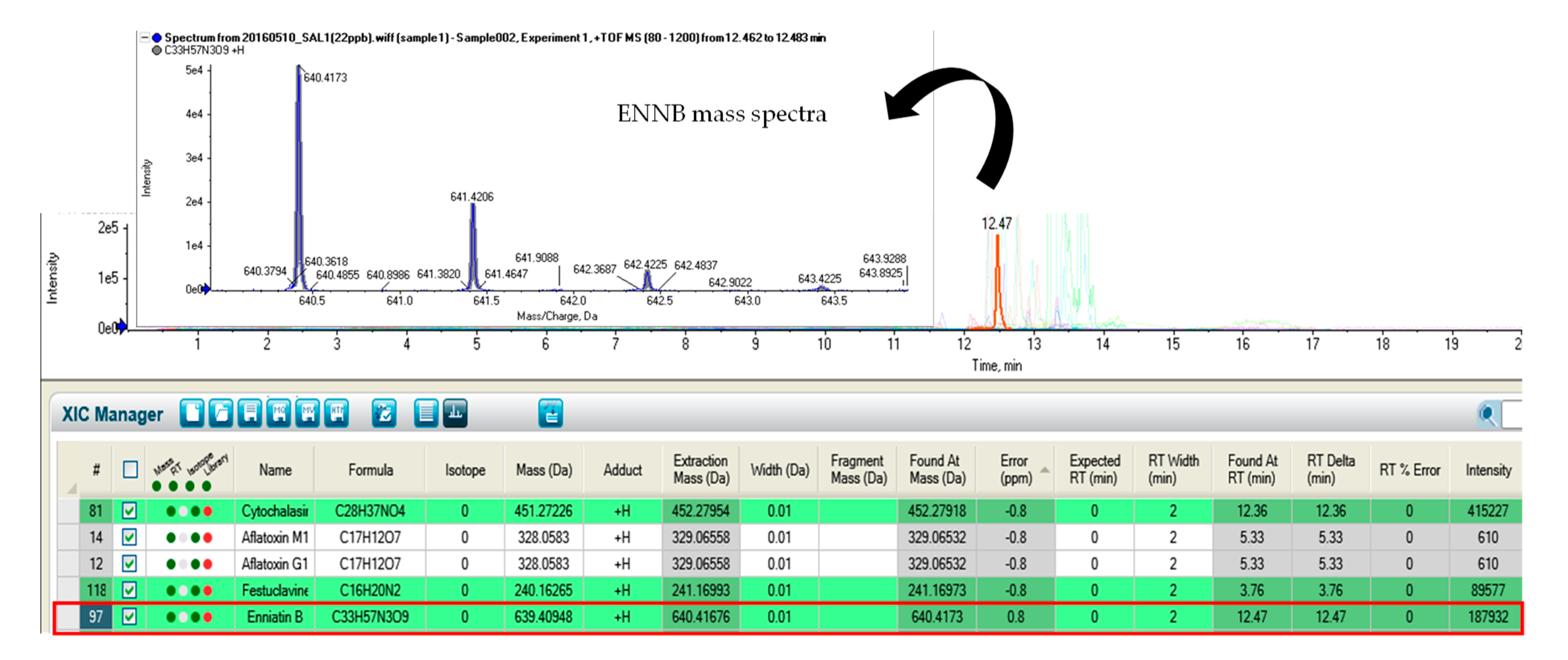
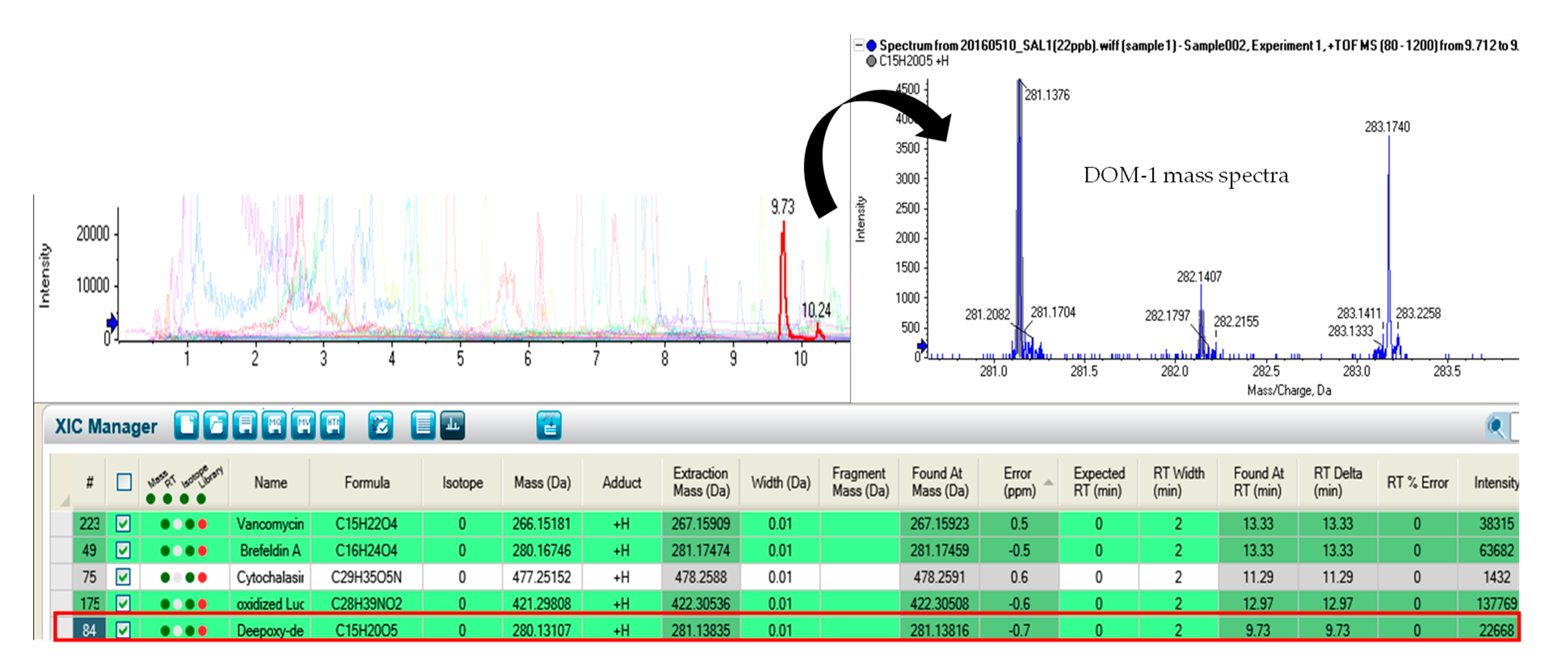
| Mycotoxins and Other Fungal Metabolites | Elemental Composition | Exact Mass (m/z) ** | RT * (min) |
|---|---|---|---|
| 2-amino-14,16-dimethyloctadecan-3-ol | C20H43NO | 314.3417 | 13.42 |
| Anisomycin | C14H19NO4 | 266.1387 | 1.42 |
| Chanoclavine | C16H20N20 | 257.1648 | 2.66 |
| Curvularin | C16H20O5 | 292.1310 | 8.29 |
| Cyclopenin | C17H14N2O3 | 295.1077 | 11.53 |
| Cyclopiazonic acid | C20H20N2O3 | 337.1547 | 6.19 |
| Cytochalasin J | C28H37NO4 | 452.2795 | 12.36 |
| Deoxybrevianamide E | C21H25N3O2 | 352.2020 | 4.86 |
| Deepoxy-deoxynivalenol | C15H20O5 | 281.1376 | 9.73 |
| Dihydrolysergol | C16H20N2O | 257.1648 | 2.66 |
| Enniatin A | C36H63N3O9 | 682.4637 | 13.28 |
| Enniatin A1 | C35H61N3O9 | 668.4481 | 13.04 |
| Enniatin B | C33H57N3O9 | 640.4168 | 12.47 |
| Enniatin B1 | C34H59N3O9 | 654.4324 | 12.78 |
| Enniatin B2 | C32H55N3O9 | 626.4011 | 12.50 |
| Festuclavine | C16H20N2 | 241.1699 | 3.80 |
| FK 506 | C44H69NO12 | 804.4893 | 6.33 |
| Fumigaclavine A | C18H22N2O2 | 299.1754 | 11.49 |
| Fumitremorgin C | C22H25N3O3 | 380.1969 | 4.33 |
| Fumonisin B2 | C34H59NO14 | 706.4008 | 13.20 |
| Fusaproliferin | C27H40O5 | 445.2949 | 13.19 |
| Fusidic acid | C31H48O6 | 517.3524 | 14.40 |
| Methysergide | C21H27N3O2 | 354.2176 | 12.20 |
| Mycophenolic acid | C17H20O6 | 321.1333 | 2.97 |
| Myriocin | C21H39NO6 | 402.2850 | 7.68 |
| Ophiobolin A | C25H36O4 | 401.2686 | 13.28 |
| Ophiobolin B | C25H38O4 | 403.2843 | 13.33 |
| Oxidized luol | C28H39NO2 | 422.3054 | 12.97 |
| Paspaline | C32H39NO4 | 502.2952 | 12.57 |
| Penicillic acid | C16H18N2O5S | 351.1009 | 11.26 |
| Penicillin G | C27H33NO6 | 468.2381 | 11.06 |
| Penicillin V | C37H44O6NCl | 634.2930 | 9.79 |
| Phomopsin A | C22H25NO8 | 432.1653 | 10.55 |
| Pseurotin A | C16H24O6 | 313.1646 | 8.54 |
| Radicicol | C22H23N5O2 | 390.1925 | 8.59 |
| Rugulosin | C29H38O8 | 515.2639 | 13.66 |
| Sulochrin | C24H34O9 | 467.2276 | 4.35 |
| T-2 Toxin | C22H30N4O4 | 415.2340 | 7.74 |
| Tetracycline | C17H24O4 | 293.1747 | 13.35 |
| Vancomycin | C15H22O4 | 267.1591 | 12.92 |
| Mycotoxin | Oral Toxicity Prediction Results | |||
|---|---|---|---|---|
| Predicted LD50 (mg/kg) | Predicted Toxicity Class | Average Similarity (%) | Prediction Accuracy (%) | |
| 2-amino-14,16-dimethyloctadecan-3-ol | 3500 | 5 | 100 | 100 |
| Alamethicin F30 | 80 | 3 | 100 | 100 |
| Anisomycin | 72 | 3 | 100 | 100 |
| Chanoclavine | 110 | 3 | 66 | 68 |
| Curvularin | 450 | 4 | 62 | 68 |
| Cyclopenin | 2200 | 5 | 58 | 67 |
| Cyclopiazonic acid | 93 | 3 | 67 | 68 |
| Cytochalasin J | 400 | 4 | 66 | 68 |
| Deepoxy-deoxynivalenol | 34 | 2 | 89 | 71 |
| Deoxybrevianamide E | 1000 | 4 | 67 | 68 |
| Dihydrolysergol | 110 | 3 | 90 | 73 |
| Enniatin A | 1600 | 4 | 76 | 69 |
| Enniatin A1 | 1600 | 4 | 76 | 69 |
| Enniatin B | 3 | 1 | 100 | 100 |
| Enniatin B1 | 1600 | 4 | 76 | 69 |
| Enniatin B2 | 3 | 1 | 100 | 100 |
| Festuclavine | 110 | 3 | 95 | 73 |
| FK 506 | 134 | 3 | 100 | 100 |
| Fumigaclavine A | 800 | 4 | 82 | 71 |
| Fumitremorgin C | 72 | 3 | 100 | 100 |
| Fumonisin B2 | 4280 | 5 | 68 | 68 |
| Fusaproliferin | 5000 | 5 | 71 | 69 |
| Fusidic acid | 841 | 4 | 100 | 100 |
| Methysergide | 200 | 3 | 100 | 100 |
| Mycophenolic acid | 352 | 4 | 100 | 100 |
| Myriocin | 300 | 3 | 100 | 100 |
| Ophiobolin A | 238 | 3 | 100 | 100 |
| Ophiobolin B | 238 | 3 | 72 | 69 |
| Oxidized luol | - | - | - | - |
| Paspaline | 374 | 4 | 73 | 69 |
| Penicillic acid | 600 | 4 | 100 | 100 |
| Penicillin G | 1000 | 4 | 100 | 100 |
| Penicillin V | 1040 | 4 | 100 | 100 |
| Phomopsin A | 400 | 4 | 56 | 67 |
| Pseurotin A | 134 | 3 | 49 | 54 |
| Radicicol | 300 | 3 | 100 | 100 |
| Rugulosin | 220 | 3 | 53 | 67 |
| Sulochrin | 690 | 4 | 61 | 68 |
| T-2 Toxin | 3 | 1 | 100 | 100 |
| Tetracycline | 678 | 4 | 100 | 100 |
| Vancomycin | 300 | 3 | 100 | 100 |
| Mycotoxin | Classification | ||||
|---|---|---|---|---|---|
| Organ Toxicity (% Probability) | Toxicity Endpoint (% Probability) | ||||
| Hepatotoxicity | Carcinogenicity | Immunotoxicity | Mutagenicity | Cytotoxicity | |
| 2-amino-14,16-dimethyloctadecan-3-ol | Inactive (74) | Inactive (50) | Inactive (97) | Inactive (94) | Inactive (71) |
| Alamethicin F30 | Inactive (97) | Inactive (57) | Inactive (99) | Inactive (85) | Inactive (78) |
| Anisomycin | Inactive (89) | Inactive (73) | Inactive (66) | Inactive (78) | Inactive (63) |
| Chanoclavine | Inactive (67) | Inactive (74) | Inactive (99) | Active (56) | Inactive (59) |
| Curvularin | Inactive (82) | Active (65) | Active (57) | Inactive (97) | Inactive (75) |
| Cyclopenin | Active (56) | Inactive (58) | Inactive (96) | Active (52) | Inactive (53) |
| Cyclopiazonic acid | Inactive (63) | Inactive (62) | Inactive (73) | Inactive (52) | Inactive (58) |
| Cytochalasin J | Inactive (68) | Inactive (54) | Active (98) | Inactive (72) | Inactive (75) |
| Deepoxy-deoxynivalenol | Inactive (80) | Inactive (77) | Inactive (64) | Active (50) | Inactive (70) |
| Deoxybrevianamide E | Inactive (81) | Inactive (61) | Active (82) | Inactive (63) | Inactive (70) |
| Dihydrolysergol | Inactive (92) | Inactive (68) | Inactive (95) | Active (74) | Inactive (69) |
| Enniatin A | Inactive (70) | Inactive (63) | Inactive (88) | Inactive (67) | Inactive (51) |
| Enniatin A1 | Inactive (70) | Inactive (63) | Inactive (88) | Inactive (67) | Inactive (51) |
| Enniatin B | Inactive (73) | Inactive (66) | Inactive (97) | Inactive (64) | Active (56) |
| Enniatin B1 | Inactive (70) | Inactive (63) | Inactive (88) | Inactive (67) | Inactive (51) |
| Enniatin B2 | Inactive (72) | Inactive (66) | Inactive (81) | Inactive (65) | Active (59) |
| Festuclavine | Inactive (89) | Inactive (74) | Inactive (92) | Active (93) | Inactive (64) |
| FK 506 | Inactive (87) | Inactive (50) | Active (99) | Inactive (70) | Inactive (64) |
| Fumigaclavine A | Inactive (83) | Active (51) | Active (63) | Active (50) | Inactive (65) |
| Fumitremorgin C | Inactive (89) | Inactive (73) | Inactive (66) | Inactive (78) | Inactive (63) |
| Fumonisin B2 | Inactive (78) | Active (74) | Active (52) | Inactive (100) | Inactive (71) |
| Fusaproliferin | Inactive (90) | Inactive (62) | Inactive (95) | Inactive (87) | Inactive (71) |
| Fusidic acid | Inactive (73) | Inactive (52) | Active (99) | Inactive (87) | Inactive (63) |
| Methysergide | Inactive (96) | Inactive (61) | Inactive (73) | Active (52) | Inactive (89) |
| Mycophenolic acid | Inactive (85) | Inactive (58) | Active (80) | Inactive (93) | Inactive (88) |
| Myriocin | Inactive (85) | Inactive (59) | Inactive (99) | Inactive (90) | Inactive (71) |
| Ophiobolin A | Inactive (76) | Inactive (52) | Active (98) | Inactive (75) | Inactive (73) |
| Ophiobolin B | Inactive (80) | Active (53) | Active (92) | Inactive (72) | Inactive (72) |
| Oxidizedluol | - | - | - | - | - |
| Paspaline | Inactive (68) | Inactive (73) | Active (95) | Inactive (73) | Inactive (78) |
| Penicillic acid | Inactive (69) | Inactive (75) | Inactive (99) | Inactive (52) | Inactive (67) |
| Penicillin G | Inactive (87) | Inactive (83) | Inactive (99) | Inactive (97) | Inactive (60) |
| Penicillin V | Inactive (91) | Inactive (81) | Inactive (99) | Inactive (95) | Inactive (55) |
| Phomopsin A | Active (55) | Active (53) | Inactive (96) | Inactive (53) | Inactive (82) |
| Pseurotin A | Inactive (65) | Inactive (63) | Active (62) | Inactive (63) | Inactive (57) |
| Radicicol | Inactive (58) | Active (53) | Active (96) | Inactive (52) | Inactive (57) |
| Rugulosin | Inactive (64) | Active (70) | Active (73) | Inactive (94) | Inactive (69) |
| Sulochrin | Inactive (59) | Inactive (76) | Inactive (57) | Inactive (71) | Inactive (82) |
| T-2 Toxin | Inactive (85) | Active (77) | Active (99) | Active (71) | Inactive (64) |
| Tetracycline | Active (58) | Inactive (75) | Active (99) | Inactive (95) | Inactive (91) |
| Vancomycin | Inactive (58) | Active (53) | Active (96) | Inactive (52) | Inactive (57) |
| Mycotoxin | Tox21 Nuclear Receptor Signaling Pathways (% Probability) | ||||||
|---|---|---|---|---|---|---|---|
| Aryl Hydrocarbon Receptor (AhR) | Androgen Receptor (AR) | Androgen Receptor Ligand Binding Domain (AR-LBD) | Aromatase | Estrogen Receptor Alpha (ER) | Estrogen Receptor Ligand Binding Domain (ER-LBD) | Peroxisome Proliferator Activated Receptor Gamma (PPAR-Gamma) | |
| 2-amino-14,16-dimethyloctadecan-3-ol | Inactive (98) | Inactive (99) | Inactive (99) | Inactive (99) | Inactive (88) | Inactive (99) | Inactive (98) |
| Alamethicin F30 | Inactive (96) | Inactive (97) | Inactive (99) | Inactive (97) | Inactive (93) | Inactive (98) | Inactive (95) |
| Anisomycin | Inactive (93) | Inactive (95) | Inactive (99) | Inactive (94) | Inactive (90) | Inactive (94) | Inactive (99) |
| Chanoclavine | Active (60) | Inactive (90) | Inactive (88) | Inactive (79) | Inactive (78) | Inactive (96) | Inactive (92) |
| Curvularin | Inactive (85) | Inactive (97) | Inactive (98) | Inactive (82) | Active (95) | Active (94) | Inactive (94) |
| Cyclopenin | Inactive (79) | Inactive (91) | Inactive (99) | Inactive (80) | Inactive (90) | Inactive (97) | Inactive (94) |
| Cyclopiazonic acid | Inactive (80) | Inactive (92) | Inactive (97) | Inactive (79) | Inactive (86) | Inactive (94) | Inactive (94) |
| Cytochalasin J | Inactive (88) | Inactive (91) | Inactive (95) | Inactive (81) | Inactive (78) | Inactive (91) | Inactive (94) |
| Deepoxy-deoxynivalenol | Inactive (94) | Inactive (92) | Inactive (85) | Inactive (81) | Inactive (83) | Inactive (97) | Inactive (95) |
| Deoxybrevianamide E | Inactive (82) | Inactive (94) | Inactive (97) | Inactive (82) | Inactive (89) | Inactive (98) | Inactive (84) |
| Dihydrolysergol | Inactive (50) | Inactive (93) | Inactive (95) | Inactive (87) | Inactive (88) | Inactive (98) | Inactive (99) |
| Enniatin A | Inactive (96) | Inactive (94) | Inactive (97) | Inactive (97) | Inactive (90) | Inactive (97) | Inactive (97) |
| Enniatin A1 | Inactive (96) | Inactive (94) | Inactive (97) | Inactive (97) | Inactive (90) | Inactive (97) | Inactive (97) |
| Enniatin B | Inactive (97) | Inactive (94) | Inactive (96) | Inactive (97) | Inactive (88) | Inactive (96) | Inactive (98) |
| Enniatin B1 | Inactive (96) | Inactive (94) | Inactive (97) | Inactive (97) | Inactive (90) | Inactive (97) | Inactive (97) |
| Enniatin B2 | Inactive (97) | Inactive (95) | Inactive (97) | Inactive (97) | Inactive (89) | Inactive (96) | Inactive (98) |
| Festuclavine | Inactive (52) | Inactive (97) | Inactive (96) | Inactive (91) | Inactive (88) | Inactive (98) | Inactive (96) |
| FK 506 | Inactive (99) | Inactive (99) | Inactive (99) | Active (79) | Inactive (82) | Inactive (91) | Inactive (95) |
| Fumigaclavine A | Inactive (50) | Inactive (94) | Inactive (96) | Inactive (90) | Inactive (91) | Inactive (97) | Inactive (97) |
| Fumitremorgin C | Inactive (93) | Inactive (95) | Inactive (99) | Inactive (94) | Inactive (90) | Inactive (94) | Inactive (99) |
| Fumonisin B2 | Inactive (98) | Inactive (96) | Inactive (99) | Inactive (99) | Inactive (85) | Inactive (96) | Inactive (97) |
| Fusaproliferin | Inactive (97) | Inactive (89) | Inactive (81) | Inactive (95) | Inactive (77) | Inactive (98) | Inactive (97) |
| Fusidic acid | Inactive (99) | Inactive (65) | Inactive (63) | Inactive (96) | Inactive (71) | Inactive (81) | Inactive (99) |
| Methysergide | Active (100) | Inactive (91) | Inactive (90) | Inactive (98) | Inactive (98) | Inactive (99) | Inactive (99) |
| Mycophenolic acid | Inactive (87) | Inactive (96) | Inactive (90) | Inactive (71) | Inactive (64) | Inactive (89) | Inactive (91) |
| Myriocin | Inactive (99) | Inactive (98) | Inactive (97) | Inactive (99) | Inactive (93) | Inactive (97) | Inactive (99) |
| Ophiobolin A | Inactive (97) | Inactive (83) | Inactive (75) | Inactive (63) | Inactive (85) | Inactive (94) | Inactive (95) |
| Ophiobolin B | Inactive (97) | Inactive (85) | Inactive (82) | Inactive (87) | Inactive (81) | Inactive (92) | Inactive (98) |
| Oxidizedluol | - | - | - | - | - | - | - |
| Paspaline | Inactive (80) | Inactive (89) | Inactive (85) | Inactive (78) | Inactive (81) | Inactive (89) | Inactive (88) |
| Penicillic acid | Inactive (99) | Inactive (99) | Inactive (98) | Inactive (96) | Inactive (95) | Inactive (97) | Inactive (93) |
| Penicillin G | Inactive (96) | Inactive (97) | Inactive (99) | Inactive (99) | Inactive (97) | Inactive (99) | Inactive (98) |
| Penicillin V | Inactive (97) | Inactive (96) | Inactive (99) | Inactive (98) | Inactive (96) | Inactive (98) | Inactive (97) |
| Phomopsin A | Inactive (53) | Inactive (95) | Inactive (96) | Inactive (85) | Inactive (80) | Inactive (89) | Inactive (92) |
| Pseurotin A | Inactive (88) | Inactive (97) | Inactive (99) | Inactive (88) | Inactive (88) | Inactive (95) | Inactive (94) |
| Radicicol | Inactive (75) | Inactive (94) | Inactive (94) | Inactive (84) | Inactive (81) | Inactive (85) | Inactive (85) |
| Rugulosin | Inactive (74) | Inactive (92) | Inactive (91) | Inactive (79) | Inactive (54) | Inactive (62) | Inactive (85) |
| Sulochrin | Inactive (60) | Inactive (93) | Inactive (100) | Inactive (92) | Active (74) | Inactive (68) | Inactive (95) |
| T-2 Toxin | Inactive (96) | Inactive (87) | Inactive (86) | Inactive (85) | Inactive (74) | Inactive (97) | Inactive (91) |
| Tetracycline | Inactive (87) | Inactive (99) | Inactive (98) | Inactive (98) | Inactive (98) | Inactive (99) | Inactive (99) |
| Vancomycin | Inactive (75) | Inactive (94) | Inactive (94) | Inactive (84) | Inactive (81) | Inactive (85) | Inactive (85) |
| Mycotoxin | Nuclear Factor (Erythroid-Derived 2-Like 2/Antioxidant Responsive Element) (nrf2/ARE) | Heat Shock Factor Response Element (HSE) | Mitochondrial Membrane Potential (MMP) | Phosphoprotein (Tumor Supressor) p53 | ATPase Family AAA Domain Containing Protein 5 (ATAD5) |
|---|---|---|---|---|---|
| 2-amino-14,16-dimethyloctadecan-3-ol | Inactive (96) | Inactive (96) | Inactive (95) | Inactive (99) | Inactive (99) |
| Alamethicin F30 | Inactive (98) | Inactive (98) | Inactive (94) | Inactive (94) | Inactive (97) |
| Anisomycin | Inactive (97) | Inactive (97) | Inactive (93) | Inactive (96) | Inactive (98) |
| Chanoclavine | Inactive (92) | Inactive (92) | Inactive (64) | Inactive (85) | Inactive (88) |
| Curvularin | Active (79) | Active (79) | Active (94) | Active (64) | Inactive (97) |
| Cyclopenin | Inactive (91) | Inactive (91) | Inactive (66) | Inactive (73) | Inactive (85) |
| Cyclopiazonic acid | Inactive (85) | Inactive (85) | Inactive (55) | Inactive (74) | Inactive (96) |
| Cytochalasin J | Inactive (86) | Inactive (86) | Inactive (69) | Inactive (74) | Inactive (90) |
| Deepoxy-deoxynivalenol | Inactive (90) | Inactive (90) | Inactive (78) | Inactive (87) | Inactive (90) |
| Deoxybrevianamide E | Inactive (93) | Inactive (93) | Inactive (77) | Inactive (77) | Inactive (96) |
| Dihydrolysergol | Inactive (95) | Inactive (95) | Inactive (87) | Inactive (96) | Inactive (98) |
| Enniatin A | Inactive (96) | Inactive (94) | Inactive (97) | Inactive (97) | Inactive (90) |
| Enniatin A1 | Inactive (96) | Inactive (94) | Inactive (97) | Inactive (97) | Inactive (90) |
| Enniatin B | Inactive (97) | Inactive (94) | Inactive (96) | Inactive (97) | Inactive (88) |
| Enniatin B1 | Inactive (96) | Inactive (94) | Inactive (97) | Inactive (97) | Inactive (90) |
| Enniatin B2 | Inactive (97) | Inactive (95) | Inactive (97) | Inactive (97) | Inactive (89) |
| Festuclavine | Inactive (52) | Inactive (97) | Inactive (96) | Inactive (91) | Inactive (88) |
| FK 506 | Inactive (99) | Inactive (99) | Inactive (99) | Active (79) | Inactive (82) |
| Fumigaclavine A | Inactive (50) | Inactive (94) | Inactive (96) | Inactive (90) | Inactive (91) |
| Fumitremorgin C | Inactive (93) | Inactive (95) | Inactive (99) | Inactive (94) | Inactive (90) |
| Fumonisin B2 | Inactive (98) | Inactive (96) | Inactive (99) | Inactive (99) | Inactive (85) |
| Fusaproliferin | Inactive (97) | Inactive (89) | Inactive (81) | Inactive (95) | Inactive (77) |
| Fusidic acid | Inactive (99) | Inactive (65) | Inactive (63) | Inactive (96) | Inactive (71) |
| Methysergide | Active (100) | Inactive (91) | Inactive (90) | Inactive (98) | Inactive (98) |
| Mycophenolic acid | Inactive (87) | Inactive (96) | Inactive (90) | Inactive (71) | Inactive (64) |
| Myriocin | Inactive (99) | Inactive (98) | Inactive (97) | Inactive (99) | Inactive (93) |
| Ophiobolin A | Inactive (97) | Inactive (83) | Inactive (75) | Inactive (63) | Inactive (85) |
| Ophiobolin B | Inactive (97) | Inactive (85) | Inactive (82) | Inactive (87) | Inactive (81) |
| Oxidizedluol | - | - | - | - | - |
| Paspaline | Inactive (80) | Inactive (89) | Inactive (85) | Inactive (78) | Inactive (81) |
| Penicillic acid | Inactive (99) | Inactive (99) | Inactive (98) | Inactive (96) | Inactive (95) |
| Penicillin G | Inactive (96) | Inactive (97) | Inactive (99) | Inactive (99) | Inactive (97) |
| Penicillin V | Inactive (97) | Inactive (96) | Inactive (99) | Inactive (98) | Inactive (96) |
| Phomopsin A | Inactive (53) | Inactive (95) | Inactive (96) | Inactive (85) | Inactive (80) |
| Pseurotin A | Inactive (88) | Inactive (97) | Inactive (99) | Inactive (88) | Inactive (88) |
| Radicicol | Inactive (75) | Inactive (94) | Inactive (94) | Inactive (84) | Inactive (81) |
| Rugulosin | Inactive (74) | Inactive (92) | Inactive (91) | Inactive (79) | Inactive (54) |
| Sulochrin | Inactive (60) | Inactive (93) | Inactive (100) | Inactive (92) | Active (74) |
| T-2 Toxin | Inactive (96) | Inactive (87) | Inactive (86) | Inactive (85) | Inactive (74) |
| Tetracycline | Inactive (87) | Inactive (99) | Inactive (98) | Inactive (98) | Inactive (98) |
| Vancomycin | Inactive (75) | Inactive (94) | Inactive (94) | Inactive (84) | Inactive (81) |
Sample Availability: Samples are available from the authors in the laboratory of food chemistry in the Preventive Medicine Department, Faculty of Pharmacy, University of Valencia. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tolosa, J.; Barba, F.J.; Pallarés, N.; Ferrer, E. Mycotoxin Identification and In Silico Toxicity Assessment Prediction in Atlantic Salmon. Mar. Drugs 2020, 18, 629. https://doi.org/10.3390/md18120629
Tolosa J, Barba FJ, Pallarés N, Ferrer E. Mycotoxin Identification and In Silico Toxicity Assessment Prediction in Atlantic Salmon. Marine Drugs. 2020; 18(12):629. https://doi.org/10.3390/md18120629
Chicago/Turabian StyleTolosa, Josefa, Francisco J. Barba, Noelia Pallarés, and Emilia Ferrer. 2020. "Mycotoxin Identification and In Silico Toxicity Assessment Prediction in Atlantic Salmon" Marine Drugs 18, no. 12: 629. https://doi.org/10.3390/md18120629
APA StyleTolosa, J., Barba, F. J., Pallarés, N., & Ferrer, E. (2020). Mycotoxin Identification and In Silico Toxicity Assessment Prediction in Atlantic Salmon. Marine Drugs, 18(12), 629. https://doi.org/10.3390/md18120629







