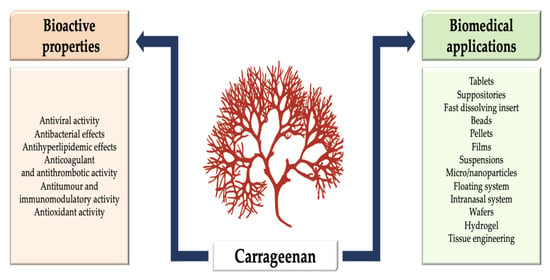
Carrageenan: Drug Delivery Systems and Other Biomedical Applications
Abstract
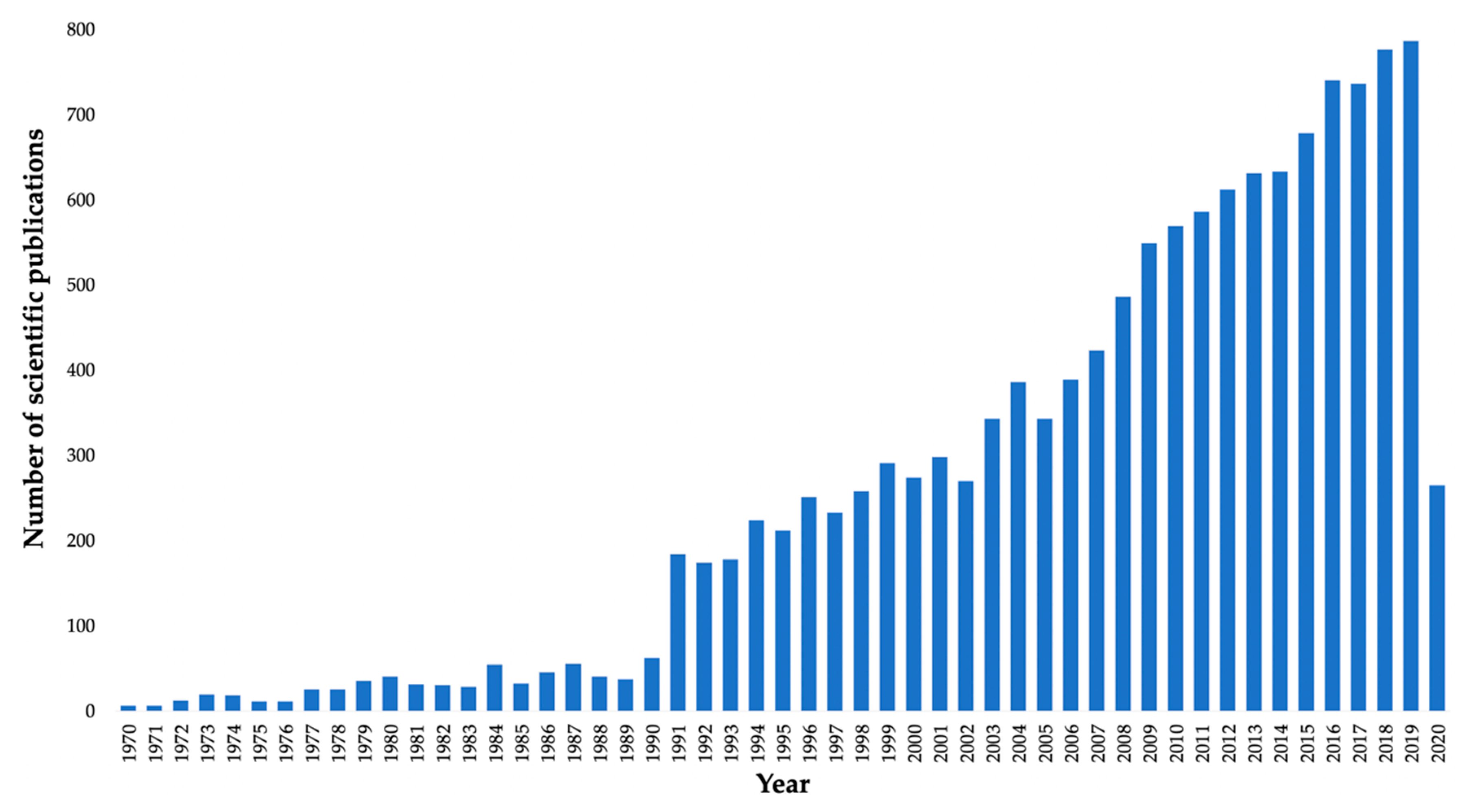
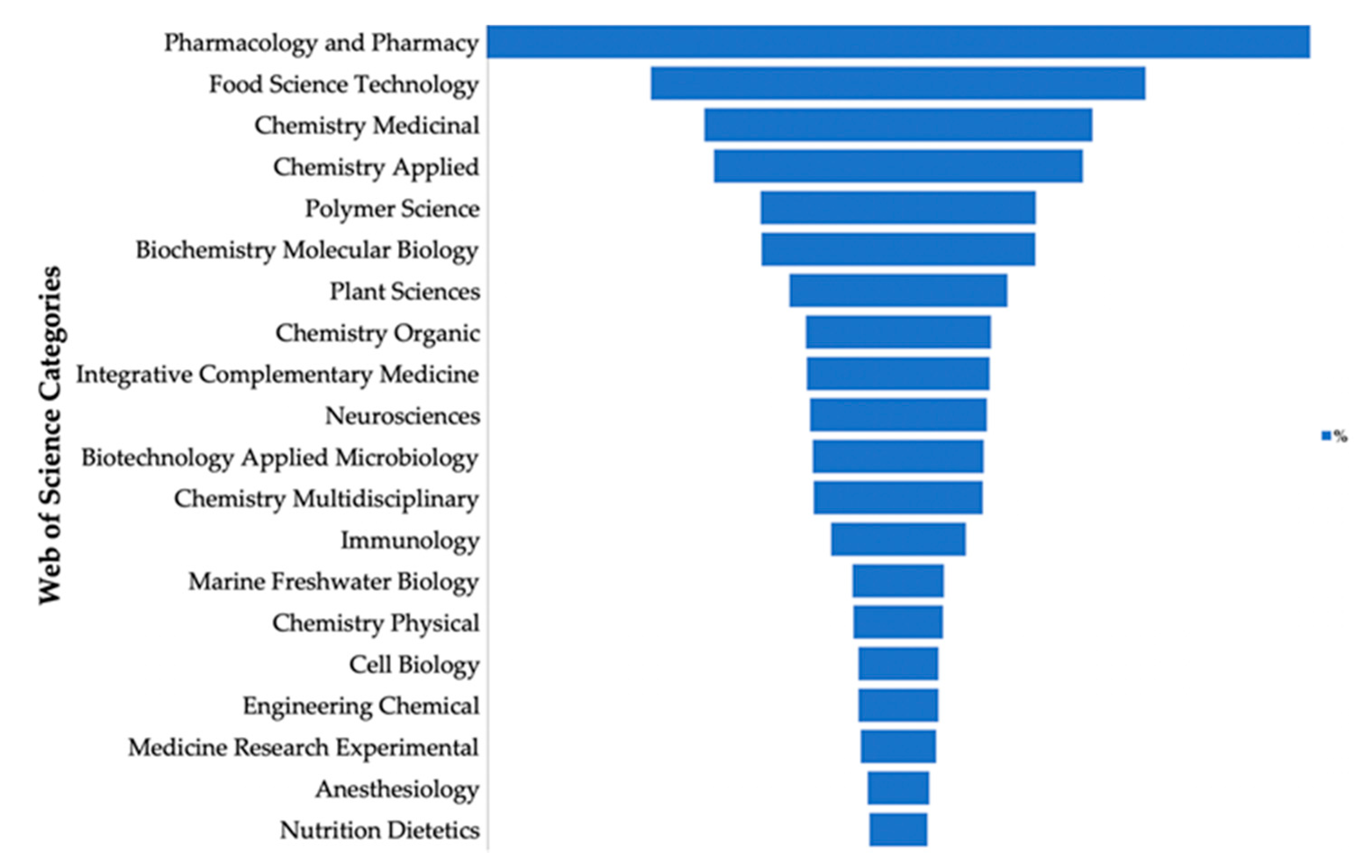
1. Introduction
2. General Properties of Carrageenan
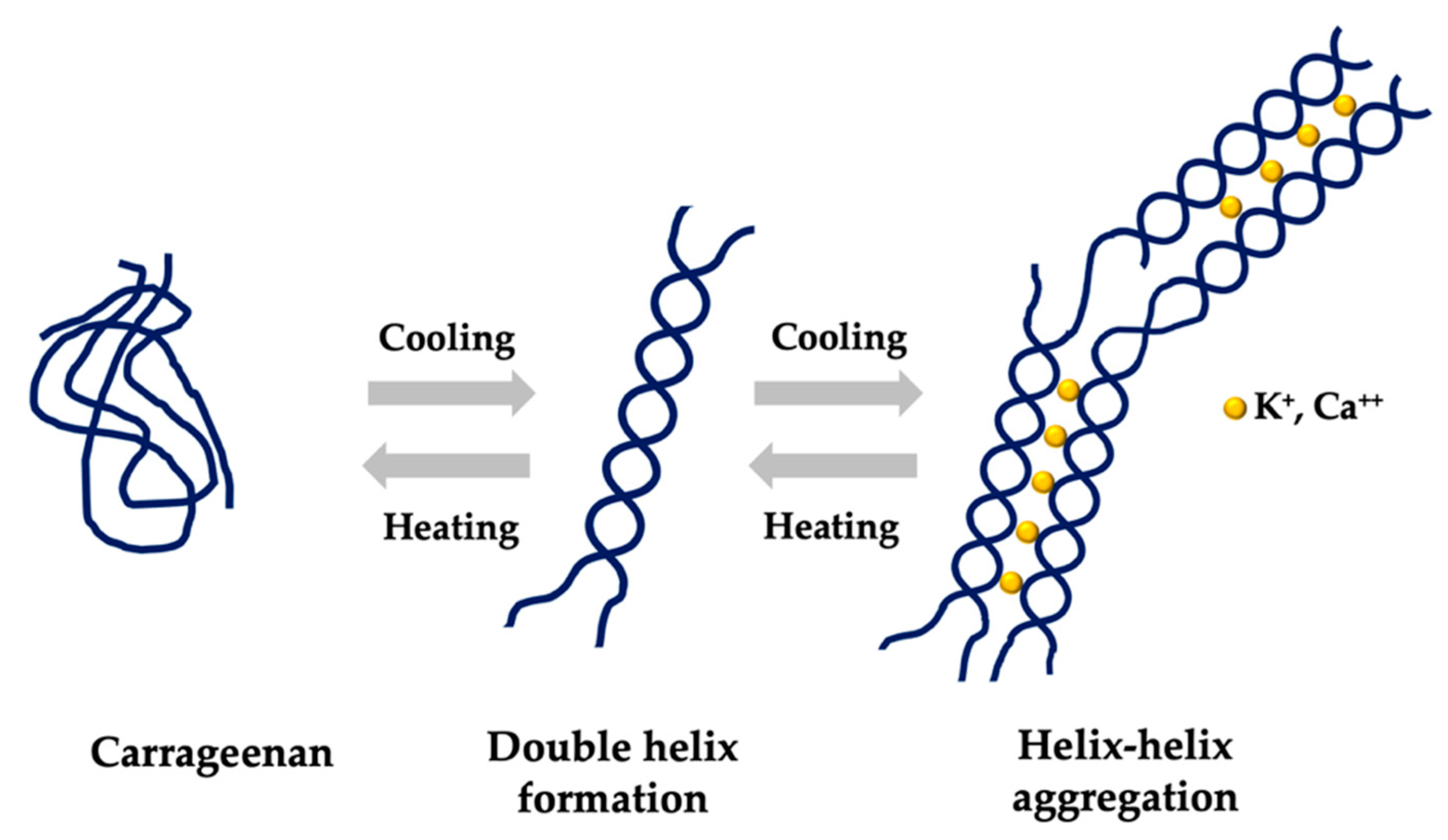
2.1. Chemical Structure and Properties
2.2. Source and Production of Carrageenan
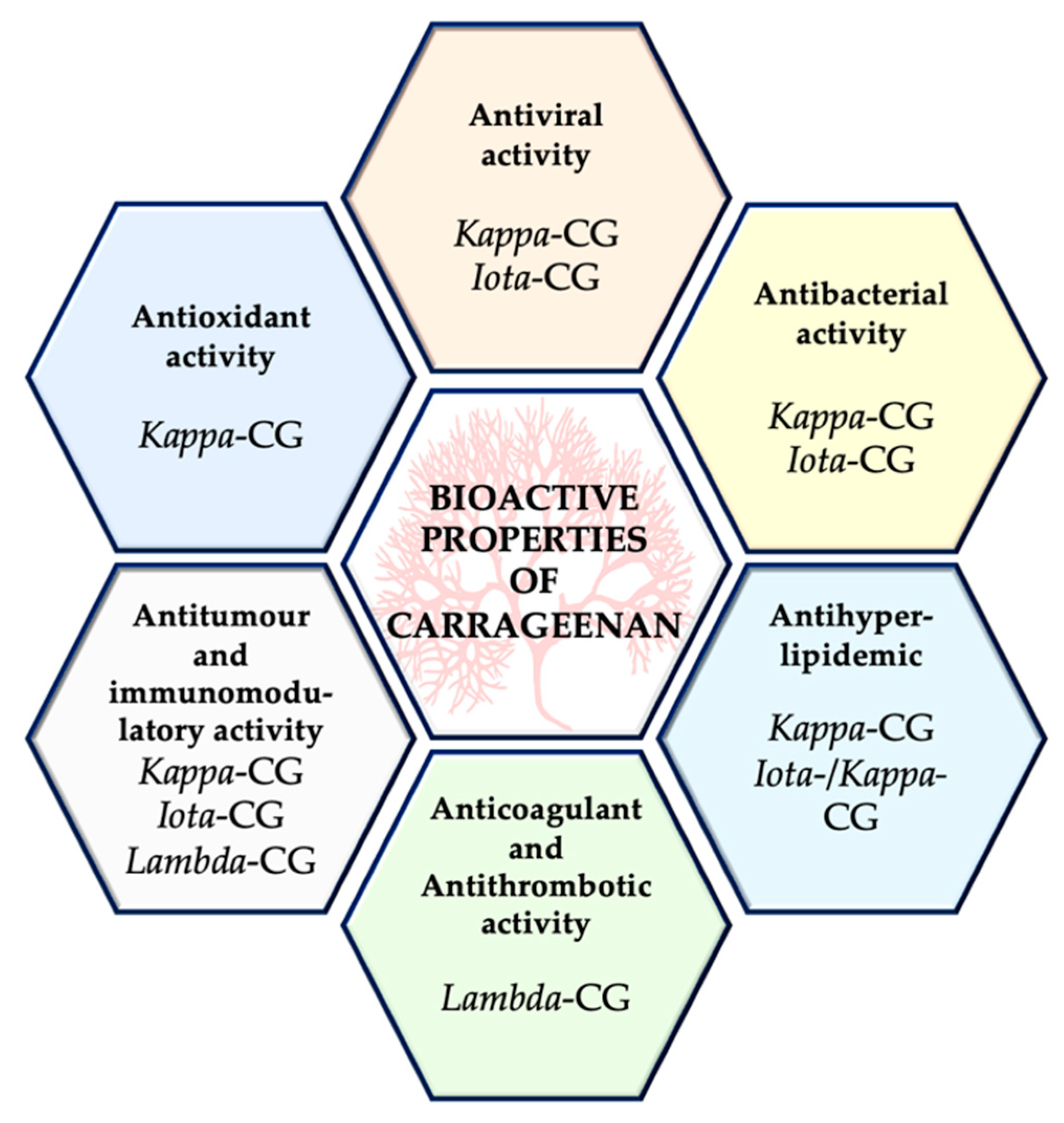
3. Bioactive Properties of Carrageenan
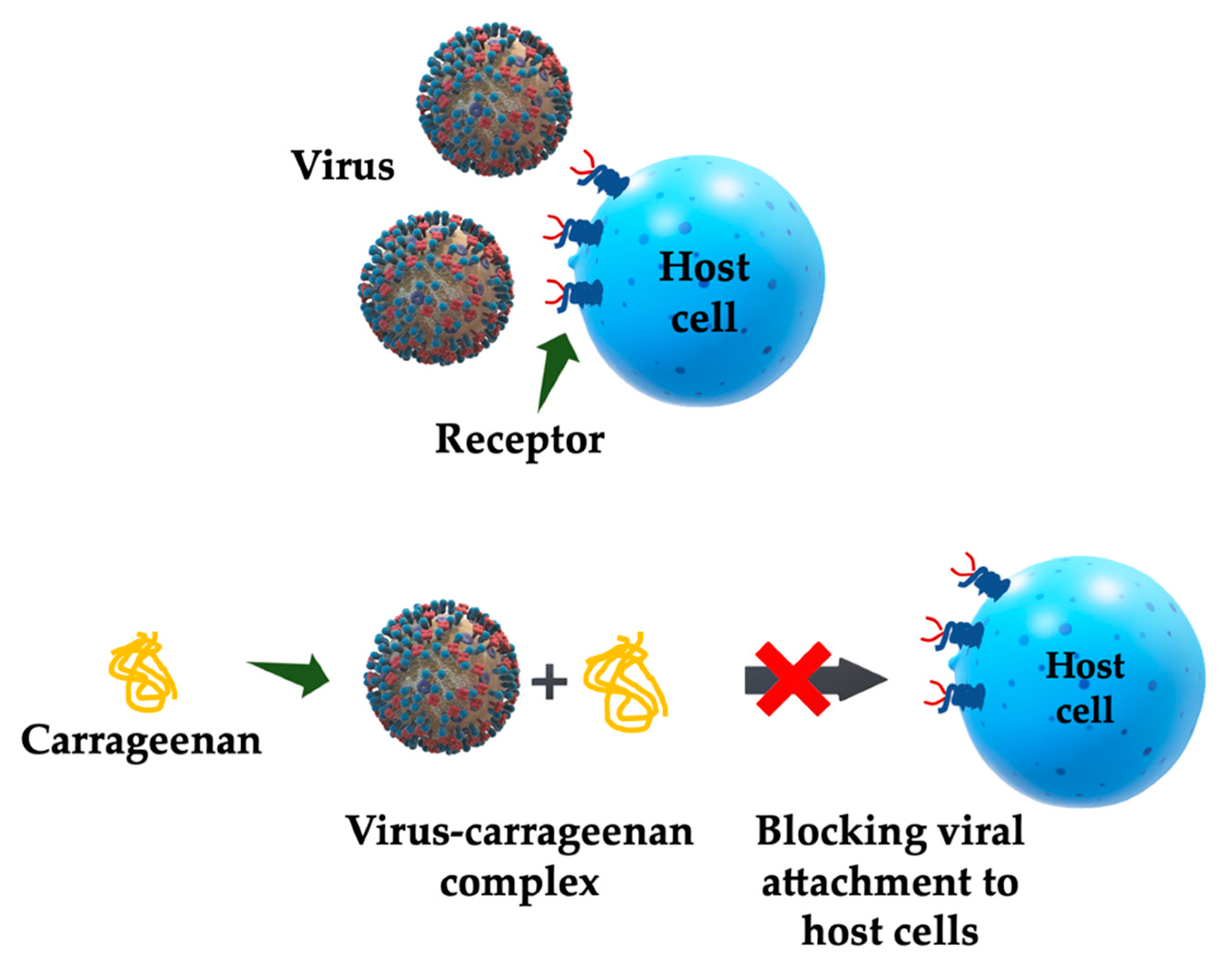
3.1. Antiviral Activity
3.2. Antibacterial Effects
3.3. Antihyperlipidemic Effects
3.4. Anticoagulant and Antithrombotic Activity
3.5. Antitumor and Immunomodulatory Activity
3.6. Antioxidant Activity
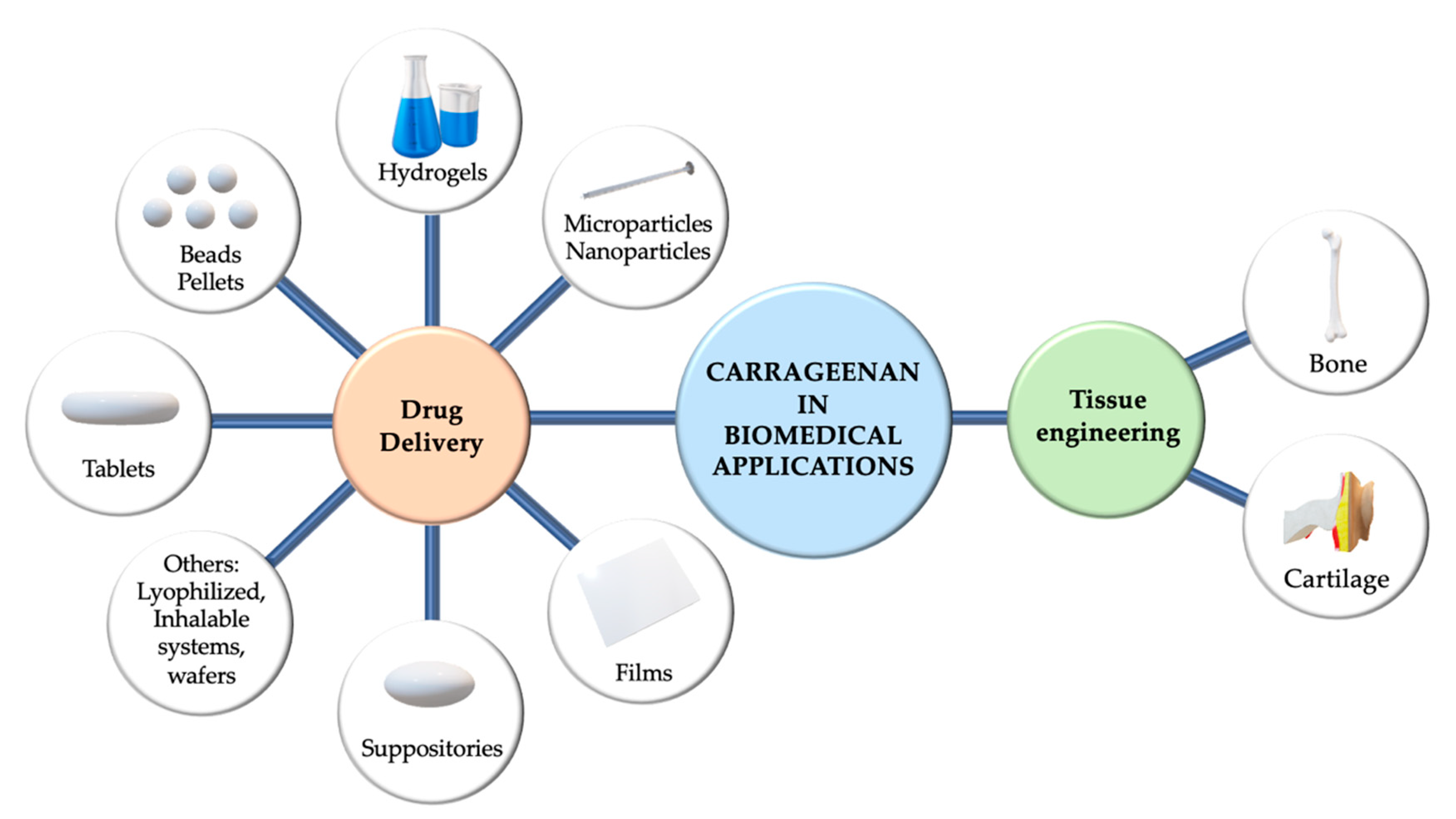
4. Carrageenan in Biomedical Applications
4.1. Applications of Carrageenan in Drug Delivery Systems
4.1.1. Carrageenan-Based Tablets
4.1.2. Carrageenan-Based Suppositories
4.1.3. Carrageenan-Based Fast-Dissolving Insert
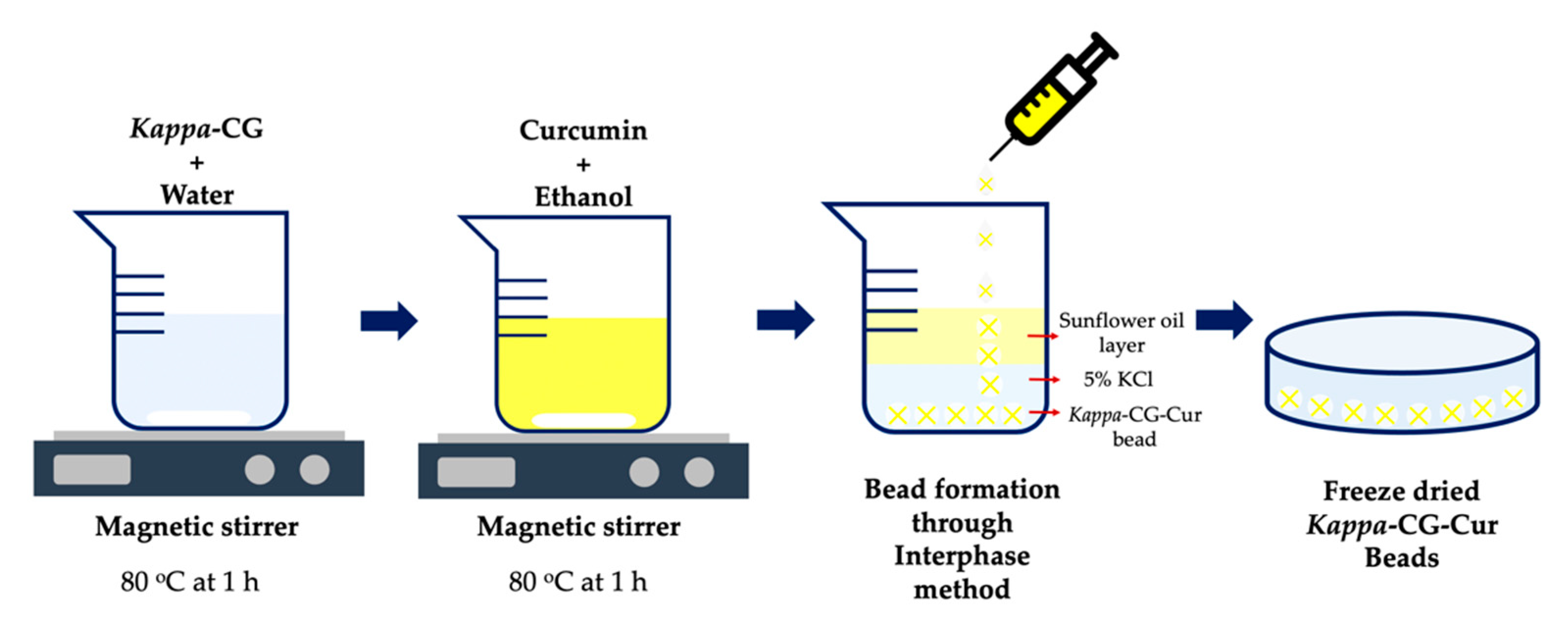
4.1.4. Carrageenan-Based Beads
4.1.5. Carrageenan-Based Pellets
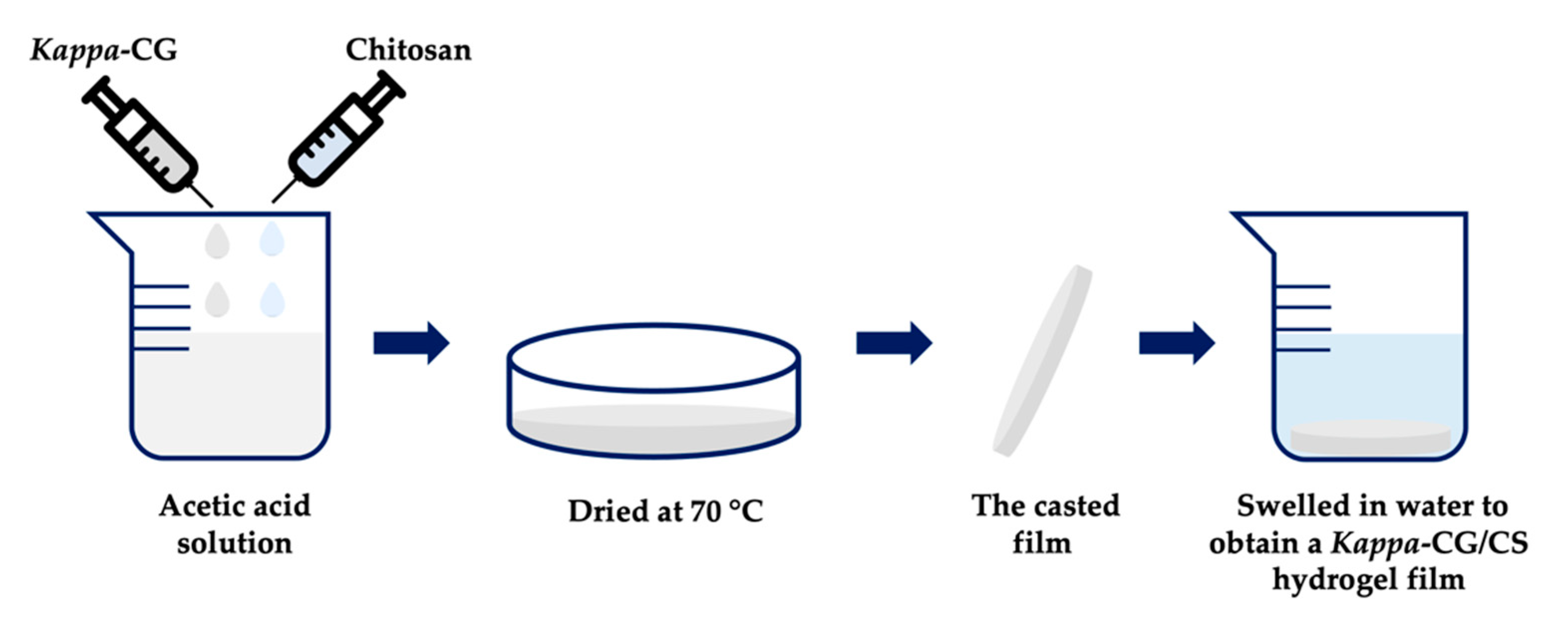
4.1.6. Carrageenan-Based Films
4.1.7. Carrageenan-Based Oral Suspensions
4.1.8. Carrageenan-Based Micro/Nanoparticles
4.1.9. Carrageenan-Based Floating System
4.1.10. Carrageenan-Based Intranasal System
4.1.11. Carrageenan-Based Wafers
4.1.12. Carrageenan-Based Hydrogel
4.2. Carrageenan-Based Tissue Engineering
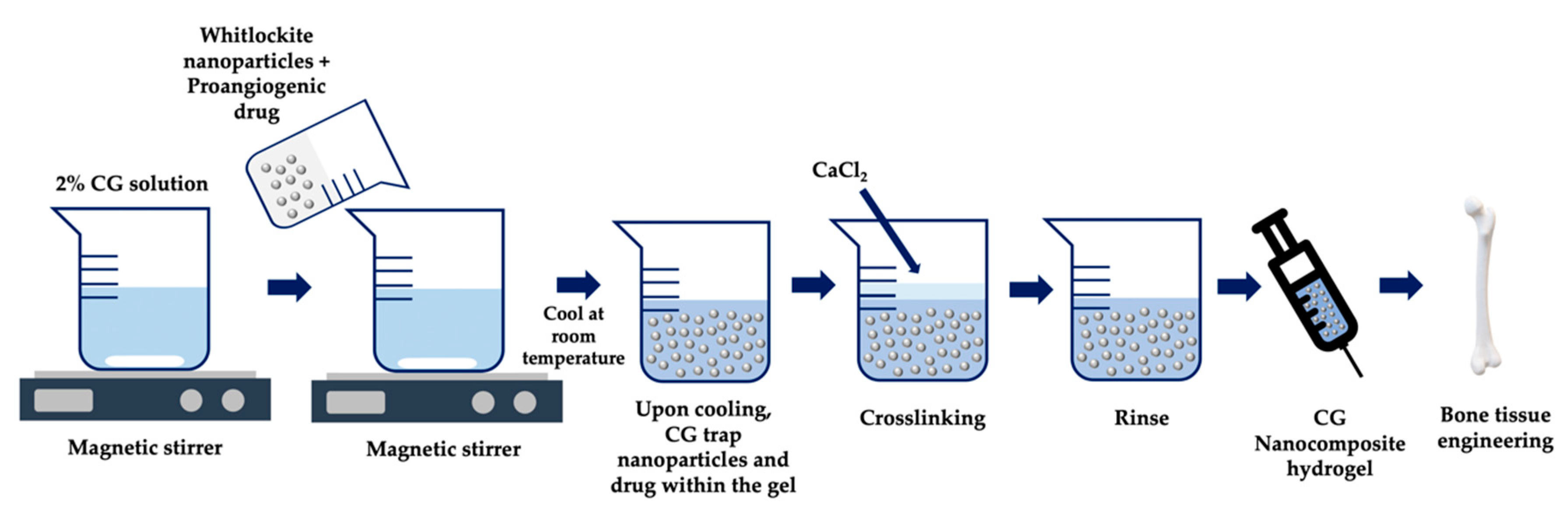
4.2.1. Bone or Cartilage
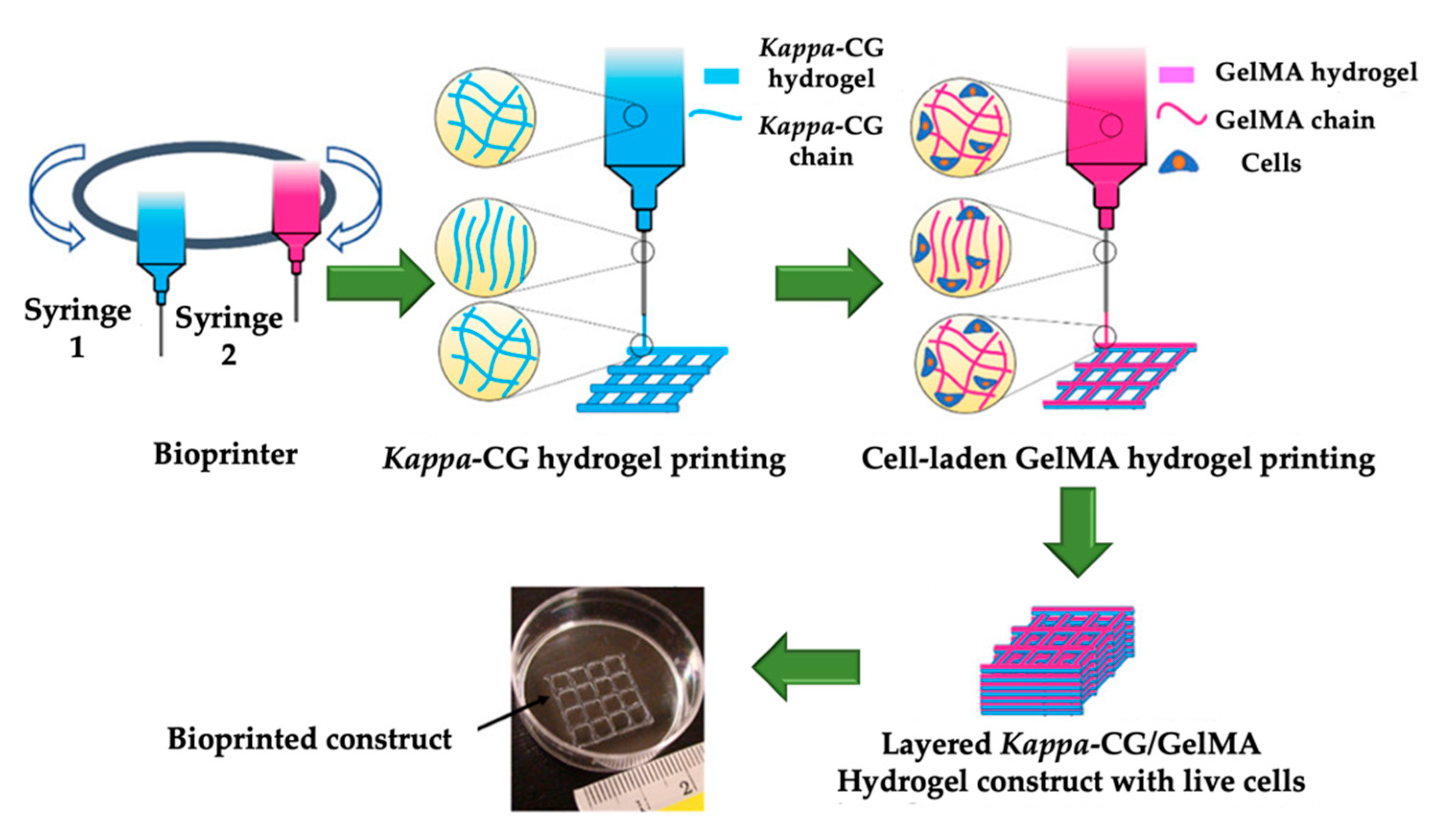
4.2.2. 3-D Bioprinting Applications
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xiong, Z.-Q.; Wang, J.-F.; Hao, Y.-Y.; Wang, Y. Recent Advances in the Discovery and Development of Marine Microbial Natural Products. Mar. Drugs 2013, 11, 700–717. [Google Scholar] [PubMed]
- Smith, A.M.; Moxon, S.; Morris, G.A. Biopolymers as wound healing materials. In Wound Healing Biomaterials; Elsevier: Amsterdam, The Netherlands, 2016; Volume 2, pp. 261–287. ISBN 9780081006061. [Google Scholar]
- De Jesus Raposo, M.; De Morais, A.; De Morais, R. Marine Polysaccharides from Algae with Potential Biomedical Applications. Mar. Drugs 2015, 13, 2967–3028. [Google Scholar] [PubMed]
- Necas, J.; Bartosikova, L. Carrageenan: A review. Vet. Med. (Praha.) 2013, 58, 187–205. [Google Scholar]
- Wijesekara, I.; Pangestuti, R.; Kim, S.-K. Biological activities and potential health benefits of sulfated polysaccharides derived from marine algae. Carbohydr. Polym. 2011, 84, 14–21. [Google Scholar]
- Abdul Khalil, H.P.S.; Saurabh, C.K.; Tye, Y.Y.; Lai, T.K.; Easa, A.M.; Rosamah, E.; Fazita, M.R.N.; Syakir, M.I.; Adnan, A.S.; Fizree, H.M.; et al. Seaweed based sustainable films and composites for food and pharmaceutical applications: A review. Renew. Sustain. Energy Rev. 2017, 77, 353–362. [Google Scholar]
- Cardoso, M.; Costa, R.; Mano, J. Marine Origin Polysaccharides in Drug Delivery Systems. Mar. Drugs 2016, 14, 34. [Google Scholar]
- Yadav, P. Biomedical Biopolymers, their Origin and Evolution in Biomedical Sciences: A Systematic Review. J. Clin. Diagn. Res. 2015, 9, 21–25. [Google Scholar]
- Jiao, G.; Yu, G.; Zhang, J.; Ewart, H. Chemical Structures and Bioactivities of Sulfated Polysaccharides from Marine Algae. Mar. Drugs 2011, 9, 196–223. [Google Scholar]
- Wang, W.; Wang, S.-X.; Guan, H.-S. The Antiviral Activities and Mechanisms of Marine Polysaccharides: An Overview. Mar. Drugs 2012, 10, 2795–2816. [Google Scholar]
- McCandless, E.L.; Craigie, J.S.; Walter, J.A. Carrageenans in the gametophytic and sporophytic stages of Chondrus crispus. Planta 1973, 112, 201–212. [Google Scholar]
- Lahaye, M. Developments on gelling algal galactans, their structure and physico-chemistry. J. Appl. Phycol. 2001, 13, 173–184. [Google Scholar]
- Campo, V.L.; Kawano, D.F.; da Silva, D.B.; Carvalho, I. Carrageenans: Biological properties, chemical modifications and structural analysis–A review. Carbohydr. Polym. 2009, 77, 167–180. [Google Scholar]
- McHugh, D.J. Carrageenan. In A Guide to the Seaweed Industry; FAO Fisheries Technical Paper: Rome, Italy, 2003; pp. 61–72. ISBN 9251049580. [Google Scholar]
- Zia, K.M.; Tabasum, S.; Nasif, M.; Sultan, N.; Aslam, N.; Noreen, A.; Zuber, M. A review on synthesis, properties and applications of natural polymer based carrageenan blends and composites. Int. J. Biol. Macromol. 2017, 96, 282–301. [Google Scholar] [PubMed]
- Mitchell, M.E.; Guiry, M.D. Carrageen: A local habitation or a name? J. Ethnopharmacol. 1983, 9, 347–351. [Google Scholar]
- Loureiro, R.R.; Cornish, M.L.; Neish, I.C. Applications of Carrageenan: With Special Reference to Iota and Kappa Forms as Derived from the Eucheumatoid Seaweeds. In Tropical Seaweed Farming Trends, Problems and Opportunities; Springer International Publishing: Cham, Swizerland, 2017; pp. 165–171. ISBN 978-3-319-63497-5. [Google Scholar]
- Liu, J.; Zhan, X.; Wan, J.; Wang, Y.; Wang, C. Review for carrageenan-based pharmaceutical biomaterials: Favourable physical features versus adverse biological effects. Carbohydr. Polym. 2015, 121, 27–36. [Google Scholar]
- Cohen, S.M.; Ito, N. A Critical Review of the Toxicological Effects of Carrageenan and Processed Eucheuma Seaweed on the Gastrointestinal Tract. Crit. Rev. Toxicol. 2002, 32, 413–444. [Google Scholar]
- Li, L.; Ni, R.; Shao, Y.; Mao, S. Carrageenan and its applications in drug delivery. Carbohydr. Polym. 2014, 103, 1–11. [Google Scholar]
- Guan, J.; Li, L.; Mao, S. Applications of Carrageenan in Advanced Drug Delivery. In Seaweed Polysaccharides; Venkatesan, J., Anil, S., Kim, S.-K., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 283–303. ISBN 9780128098172. [Google Scholar]
- Yegappan, R.; Selvaprithiviraj, V.; Amirthalingam, S.; Jayakumar, R. Carrageenan based hydrogels for drug delivery, tissue engineering and wound healing. Carbohydr. Polym. 2018, 198, 385–400. [Google Scholar]
- Yuan, H.; Zhang, W.; Li, X.; Lü, X.; Li, N.; Gao, X.; Song, J. Preparation and in vitro antioxidant activity of κ-carrageenan oligosaccharides and their oversulfated, acetylated, and phosphorylated derivatives. Carbohydr. Res. 2005, 340, 685–692. [Google Scholar]
- Sun, T.; Tao, H.; Xie, J.; Zhang, S.; Xu, X. Degradation and antioxidant activity of κ-Carrageenans. J. Appl. Polym. Sci. 2010, 117, 194–199. [Google Scholar]
- Gómez-Ordóñez, E.; Jiménez-Escrig, A.; Rupérez, P. Bioactivity of sulfated polysaccharides from the edible red seaweed Mastocarpus stellatus. Bioact. Carbohydr. Diet. Fibre 2014, 3, 29–40. [Google Scholar]
- Carlucci, M.J.; Pujol, C.A.; Ciancia, M.; Noseda, M.D.; Matulewicz, M.C.; Damonte, E.B.; Cerezo, A.S. Antiherpetic and anticoagulant properties of carrageenans from the red seaweed Gigartina skottsbergii and their cyclized derivatives: Correlation between structure and biological activity. Int. J. Biol. Macromol. 1997, 20, 97–105. [Google Scholar] [PubMed]
- Carlucci, M.J.; Scolaro, L.A.; Damonte, E.B. Inhibitory Action of Natural Carrageenans on Herpes simplex Virus Infection of Mouse Astrocytes. Chemotherapy 1999, 45, 429–436. [Google Scholar] [PubMed]
- Buck, C.B.; Thompson, C.D.; Roberts, J.N.; Müller, M.; Lowy, D.R.; Schiller, J.T. Carrageenan Is a Potent Inhibitor of Papillomavirus Infection. PLoS Pathog. 2006, 2, e69. [Google Scholar]
- Grassauer, A.; Weinmuellner, R.; Meier, C.; Pretsch, A.; Prieschl-Grassauer, E.; Unger, H. Iota-Carrageenan is a potent inhibitor of rhinovirus infection. Virol. J. 2008, 5, 107. [Google Scholar] [PubMed]
- Inic-Kanada, A.; Stein, E.; Stojanovic, M.; Schuerer, N.; Ghasemian, E.; Filipovic, A.; Marinkovic, E.; Kosanovic, D.; Barisani-Asenbauer, T. Effects of iota-carrageenan on ocular Chlamydia trachomatis infection in vitro and in vivo. J. Appl. Phycol. 2018, 30, 2601–2610. [Google Scholar]
- Panlasigui, L.N.; Baello, O.Q.; Dimatangal, J.M.; Dumelod, B.D. Blood cholesterol and lipid-lowering effects of carrageenan on human volunteers. Asia Pac. J. Clin. Nutr. 2003, 12, 209–214. [Google Scholar]
- Zhou, G.; Sun, Y.P.; Xin, H.; Zhang, Y.; Li, Z.; Xu, Z. In vivo antitumor and immunomodulation activities of different molecular weight lambda-carrageenans from Chondrus ocellatus. Pharmacol. Res. 2004, 50, 47–53. [Google Scholar]
- Yuan, H.; Song, J.; Li, X.; Li, N.; Dai, J. Immunomodulation and antitumor activity of κ-carrageenan oligosaccharides. Cancer Lett. 2006, 243, 228–234. [Google Scholar]
- Liu, J.; Hafting, J.; Critchley, A.T.; Banskota, A.H.; Prithiviraj, B. Components of the Cultivated Red Seaweed Chondrus crispus Enhance the Immune Response of Caenorhabditis elegans to Pseudomonas aeruginosa through the pmk-1, daf-2/daf-16, and skn-1 Pathways. Appl. Environ. Microbiol. 2013, 79, 7343–7350. [Google Scholar]
- Usov, A.I. Polysaccharides of the red algae. In Advances in Carbohydrate Chemistry and Biochemistry; Academic Press Inc.: Cambridge, MA, USA, 2011; Volume 65, pp. 115–217. ISBN 978-0-12-385520-6. [Google Scholar]
- Therkelsen, G.H. Carrageenan. In Industrial Gums; Elsevier: Amsterdam, The Netherlands, 1993; pp. 145–180. ISBN 9780127462530. [Google Scholar]
- Kariduraganavar, M.Y.; Kittur, A.A.; Kamble, R.R. Polymer Synthesis and Processing. In Natural and Synthetic Biomedical Polymers; Elsevier: Amsterdam, The Netherlands, 2014; pp. 1–31. ISBN 9780123969835. [Google Scholar]
- BeMiller, J.N. Carrageenans. In Carbohydrate Chemistry for Food Scientists; Elsevier: Amsterdam, The Netherlands, 2019; pp. 279–291. ISBN 9780128120699. [Google Scholar]
- Nguyen, B.T.; Nicolai, T.; Benyahia, L.; Chassenieux, C. Synergistic effects of mixed salt on the gelation of κ-carrageenan. Carbohydr. Polym. 2014, 112, 10–15. [Google Scholar] [PubMed]
- Cunha, L.; Grenha, A. Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications. Mar. Drugs 2016, 14, 42. [Google Scholar]
- Running, C.A.; Falshaw, R.; Janaswamy, S. Trivalent iron induced gelation in lambda-carrageenan. Carbohydr. Polym. 2012, 87, 2735–2739. [Google Scholar] [PubMed]
- Rhein-Knudsen, N.; Ale, M.; Meyer, A. Seaweed Hydrocolloid Production: An Update on Enzyme Assisted Extraction and Modification Technologies. Mar. Drugs 2015, 13, 3340–3359. [Google Scholar] [PubMed]
- Collén, J.; Cornish, M.L.; Craigie, J.; Ficko-Blean, E.; Hervé, C.; Krueger-Hadfield, S.A.; Leblanc, C.; Michel, G.; Potin, P.; Tonon, T.; et al. Chondrus crispus-A present and historical model organism for red seaweeds. In Advances in Botanical Research; Academic Press Inc.: Cambridge, MA, USA, 2014; Volume 71, pp. 53–89. ISBN 9780124080621. [Google Scholar]
- Torres, M.D.; Flórez-Fernández, N.; Domínguez, H. Integral Utilization of Red Seaweed for Bioactive Production. Mar. Drugs 2019, 17, 314. [Google Scholar]
- Pangestuti, R.; Kim, S.-K. Biological Activities of Carrageenan. In Advances in Food and Nutrition Research; Elsevier Inc.: Amsterdam, The Netherlands, 2014; Volume 72, pp. 113–124. ISBN 9780128002698. [Google Scholar]
- Blanco-Pascual, N.; Alemán, A.; Gómez-Guillén, M.C.; Montero, M.P. Enzyme-assisted extraction of κ/ι-hybrid carrageenan from Mastocarpus stellatus for obtaining bioactive ingredients and their application for edible active film development. Food Funct. 2014, 5, 319–329. [Google Scholar]
- de Araújo, I.W.F.; Rodrigues, J.A.G.; Vanderlei, E.D.S.O.; De Paula, G.A.; Lima, T.D.B.; Benevides, N.M.B. Iota-carrageenans from Solieria filiformis (Rhodophyta) and their effects in the inflammation and coagulation. Acta Sci. Technol. 2012, 34, 127–135. [Google Scholar]
- Varadarajan, S.A.; Nazaruddin, R.; Arbakariya, A.; Mamot, S. Development of high yielding carragenan extraction method from Eucheuma Cotonii using cellulase and Aspergillus niger. In Proceedings of the Prosiding Seminar Kimia Bersama UKM-ITB VIII9, Bangi, Malaysia, 9–11 June 2009; pp. 461–469. [Google Scholar]
- Das, A.K.; Sharma, M.; Mondal, D.; Prasad, K. Deep eutectic solvents as efficient solvent system for the extraction of κ-carrageenan from Kappaphycus alvarezii. Carbohydr. Polym. 2016, 136, 930–935. [Google Scholar]
- Zainal-Abidin, M.H.; Hayyan, M.; Hayyan, A.; Jayakumar, N.S. New horizons in the extraction of bioactive compounds using deep eutectic solvents: A review. Anal. Chim. Acta 2017, 979, 1–23. [Google Scholar]
- Youssouf, L.; Lallemand, L.; Giraud, P.; Soulé, F.; Bhaw-Luximon, A.; Meilhac, O.; D’Hellencourt, C.L.; Jhurry, D.; Couprie, J. Ultrasound-assisted extraction and structural characterization by NMR of alginates and carrageenans from seaweeds. Carbohydr. Polym. 2017, 166, 55–63. [Google Scholar]
- Abdul Khalil, H.P.S.; Lai, T.K.; Tye, Y.Y.; Rizal, S.; Chong, E.W.N.; Yap, S.W.; Hamzah, A.A.; Nurul Fazita, M.R.; Paridah, M.T. A review of extractions of seaweed hydrocolloids: Properties and applications. Express Polym. Lett. 2018, 12, 296–317. [Google Scholar]
- Vázquez-Delfín, E.; Robledo, D.; Freile-Pelegrín, Y. Microwave-assisted extraction of the Carrageenan from Hypnea musciformis (Cystocloniaceae, Rhodophyta). J. Appl. Phycol. 2014, 26, 901–907. [Google Scholar]
- Boulho, R.; Marty, C.; Freile-Pelegrín, Y.; Robledo, D.; Bourgougnon, N.; Bedoux, G. Antiherpetic (HSV-1) activity of carrageenans from the red seaweed Solieria chordalis (Rhodophyta, Gigartinales) extracted by microwave-assisted extraction (MAE). J. Appl. Phycol. 2017, 29, 2219–2228. [Google Scholar]
- Hilliou, L.; Larotonda, F.D.S.; Abreu, P.; Ramos, A.M.; Sereno, A.M.; Gonçalves, M.P. Effect of extraction parameters on the chemical structure and gel properties of κ/ι-hybrid carrageenans obtained from Mastocarpus stellatus. Biomol. Eng. 2006, 23, 201–208. [Google Scholar]
- Gerber, P.; Dutcher, J.D.; Adams, E.V.; Sherman, J.H. Protective Effect of Seaweed Extracts for Chicken Embryos Infected with Influenza B or Mumps Virus. Exp. Biol. Med. 1958, 99, 590–593. [Google Scholar]
- Besednova, N.; Zaporozhets, T.; Kuznetsova, T.; Makarenkova, I.; Fedyanina, L.; Kryzhanovsky, S.; Malyarenko, O.; Ermakova, S. Metabolites of Seaweeds as Potential Agents for the Prevention and Therapy of Influenza Infection. Mar. Drugs 2019, 17, 373. [Google Scholar]
- Damonte, E.; Matulewicz, M.; Cerezo, A. Sulfated Seaweed Polysaccharides as Antiviral Agents. Curr. Med. Chem. 2012, 11, 2399–2419. [Google Scholar]
- Shi, Q.; Wang, A.; Lu, Z.; Qin, C.; Hu, J.; Yin, J. Overview on the antiviral activities and mechanisms of marine polysaccharides from seaweeds. Carbohydr. Res. 2017, 453–454, 1–9. [Google Scholar]
- Mahomoodally, M.F.; Lobine, D.; Rengasamy, K.R.R.; Gowrishankar, S.; Tewari, D.; Zengin, G.; Kim, D.H.; Sivanesan, I. Marine Algae: A Potential Resource of Anti-HSV Molecules. Processes 2019, 7, 887. [Google Scholar]
- Carlucci, M.; Ciancia, M.; Matulewicz, M.; Cerezo, A.; Damonte, E. Antiherpetic activity and mode of action of natural carrageenans of diverse structural types. Antivir. Res. 1999, 43, 93–102. [Google Scholar]
- Levendosky, K.; Mizenina, O.; Martinelli, E.; Jean-Pierre, N.; Kizima, L.; Rodriguez, A.; Kleinbeck, K.; Bonnaire, T.; Robbiani, M.; Zydowsky, T.M.; et al. Griffithsin and Carrageenan Combination To Target Herpes Simplex Virus 2 and Human Papillomavirus. Antimicrob. Agents Chemother. 2015, 59, 7290–7298. [Google Scholar] [PubMed]
- Derby, N.; Lal, M.; Aravantinou, M.; Kizima, L.; Barnable, P.; Rodriguez, A.; Lai, M.; Wesenberg, A.; Ugaonkar, S.; Levendosky, K.; et al. Griffithsin carrageenan fast dissolving inserts prevent SHIV HSV-2 and HPV infections in vivo. Nat. Commun. 2018, 9, 3881. [Google Scholar] [PubMed]
- Ugaonkar, S.R.; Wesenberg, A.; Wilk, J.; Seidor, S.; Mizenina, O.; Kizima, L.; Rodriguez, A.; Zhang, S.; Levendosky, K.; Kenney, J.; et al. A novel intravaginal ring to prevent HIV-1, HSV-2, HPV, and unintended pregnancy. J. Control. Release 2015, 213, 57–68. [Google Scholar] [PubMed]
- Perino, A.; Consiglio, P.; Maranto, M.; De Franciscis, P.; Marci, R.; Restivo, V.; Manzone, M.; Capra, G.; Cucinella, G.; Calagna, G. Impact of a new carrageenan-based vaginal microbicide in a female population with genital HPV-infection: First experimental results. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 6744–6752. [Google Scholar]
- Eccles, R.; Winther, B.; Johnston, S.L.; Robinson, P.; Trampisch, M.; Koelsch, S. Efficacy and safety of iota-carrageenan nasal spray versus placebo in early treatment of the common cold in adults: The ICICC trial. Respir. Res. 2015, 16, 121. [Google Scholar]
- Leibbrandt, A.; Meier, C.; König-Schuster, M.; Weinmüllner, R.; Kalthoff, D.; Pflugfelder, B.; Graf, P.; Frank-Gehrke, B.; Beer, M.; Fazekas, T.; et al. Iota-Carrageenan Is a Potent Inhibitor of Influenza A Virus Infection. PLoS ONE 2010, 5, e14320. [Google Scholar]
- Shao, Q.; Guo, Q.; Xu, W.P.; Li, Z.; Zhao, T.T. Specific Inhibitory Effect of κ-Carrageenan Polysaccharide on Swine Pandemic 2009 H1N1 Influenza Virus. PLoS ONE 2015, 10, e0126577. [Google Scholar]
- Morokutti-Kurz, M.; König-Schuster, M.; Koller, C.; Graf, C.; Graf, P.; Kirchoff, N.; Reutterer, B.; Seifert, J.-M.; Unger, H.; Grassauer, A.; et al. The Intranasal Application of Zanamivir and Carrageenan Is Synergistically Active against Influenza A Virus in the Murine Model. PLoS ONE 2015, 10, e0128794. [Google Scholar]
- Graf, C.; Bernkop-Schnürch, A.; Egyed, A.; Koller, C.; Prieschl-Grassauer, E.; Morokutti-Kurz, M. Development of a nasal spray containing xylometazoline hydrochloride and iota-carrageenan for the symptomatic relief of nasal congestion caused by rhinitis and sinusitis. Int. J. Gen. Med. 2018, 11, 275–283. [Google Scholar]
- Chiu, Y.-H.; Chan, Y.-L.; Tsai, L.-W.; Li, T.-L.; Wu, C.-J. Prevention of human enterovirus 71 infection by kappa carrageenan. Antivir. Res. 2012, 95, 128–134. [Google Scholar]
- Luo, Z.; Tian, D.; Zhou, M.; Xiao, W.; Zhang, Y.; Li, M.; Sui, B.; Wang, W.; Guan, H.; Chen, H.; et al. λ-Carrageenan P32 Is a Potent Inhibitor of Rabies Virus Infection. PLoS ONE 2015, 10, e0140586. [Google Scholar]
- Abu-Galiyun, E.; Huleihel, M.; Levy-Ontman, O. Antiviral bioactivity of renewable polysaccharides against Varicella Zoster. Cell Cycle 2019, 18, 3540–3549. [Google Scholar] [PubMed]
- Song, S.; Peng, H.; Wang, Q.; Liu, Z.; Dong, X.; Wen, C.; Ai, C.; Zhang, Y.; Wang, Z.; Zhu, B. Inhibitory activities of marine sulfated polysaccharides against SARS-CoV-2. Food Funct. 2020, 11, 7415–7420. [Google Scholar] [PubMed]
- Pereira, L.; Critchley, A.T. The COVID 19 novel coronavirus pandemic 2020: Seaweeds to the rescue? Why does substantial, supporting research about the antiviral properties of seaweed polysaccharides seem to go unrecognized by the pharmaceutical community in these desperate times? J. Appl. Phycol. 2020, 32, 1875–1877. [Google Scholar]
- Naidoo, D.; Roy, A.; Kar, P.; Mutanda, T.; Anandraj, A. Cyanobacterial metabolites as promising drug leads against the Mpro and PLpro of SARS-CoV-2: An in silico analysis. J. Biomol. Struct. Dyn. 2020, 1–13. [Google Scholar]
- Yamashita, S.; Sugita-Konishi, Y.; Shimizu, M. In vitro Bacteriostatic Effects on Dietary Polysaccharides. Food Sci. Technol. Res. 2001, 7, 262–264. [Google Scholar]
- Wang, F.F.; Yao, Z.; Wu, H.G.; Zhang, S.X.; Zhu, N.N.; Gai, X. Antibacterial Activities of Kappa-Carrageenan Oligosaccharides. Appl. Mech. Mater. 2011, 108, 194–199. [Google Scholar]
- Zhu, M.; Ge, L.; Lyu, Y.; Zi, Y.; Li, X.; Li, D.; Mu, C. Preparation, characterization and antibacterial activity of oxidized κ-carrageenan. Carbohydr. Polym. 2017, 174, 1051–1058. [Google Scholar]
- Bennett, C.; Ramezanpour, M.; Cooksley, C.; Vreugde, S.; Psaltis, A.J. Kappa-carrageenan sinus rinses reduce inflammation and intracellular Staphylococcus aureus infection in airway epithelial cells. Int. Forum Allergy Rhinol. 2019, 9, 918–925. [Google Scholar]
- Rasool, A.; Ata, S.; Islam, A.; Khan, R.U. Fabrication of novel carrageenan based stimuli responsive injectable hydrogels for controlled release of cephradine. RSC Adv. 2019, 9, 12282–12290. [Google Scholar]
- Madruga, L.Y.C.; Sabino, R.M.; Santos, E.C.G.; Popat, K.C.; de Balaban, R.C.; Kipper, M.J. Carboxymethyl-kappa-carrageenan: A study of biocompatibility, antioxidant and antibacterial activities. Int. J. Biol. Macromol. 2020, 152, 483–491. [Google Scholar] [PubMed]
- Matthan, N.R.; Zhu, L.; Pencina, M.; D’Agostino, R.B.; Schaefer, E.J.; Lichtenstein, A.H. Sex-Specific Differences in the Predictive Value of Cholesterol Homeostasis Markers and 10-Year Cardiovascular Disease Event Rate in Framingham Offspring Study Participants. J. Am. Heart Assoc. 2013, 2, e005066. [Google Scholar] [PubMed]
- Amano, H.; Kakinuma, M.; Coury, D.A.; Ohno, H.; Hara, T. Effect of a seaweed mixture on serum lipid level and platelet aggregation in rats. Fish. Sci. 2005, 71, 1160–1166. [Google Scholar]
- Chen, F.; Deng, Z.; Zhang, Z.; Zhang, R.; Xu, Q.; Fan, G.; Luo, T.; McClements, D.J. Controlling lipid digestion profiles using mixtures of different types of microgel: Alginate beads and carrageenan beads. J. Food Eng. 2018, 238, 156–163. [Google Scholar]
- Sokolova, E.V.; Kravchenko, A.O.; Sergeeva, N.V.; Davydova, V.N.; Bogdanovich, L.N.; Yermak, I.M. Effect of carrageenans on some lipid metabolism components in vitro. Carbohydr. Polym. 2020, 230, 115629. [Google Scholar]
- Valado, A.; Pereira, M.; Caseiro, A.; Figueiredo, J.P.; Loureiro, H.; Almeida, C.; Cotas, J.; Pereira, L. Effect of Carrageenans on Vegetable Jelly in Humans with Hypercholesterolemia. Mar. Drugs 2019, 18, 19. [Google Scholar]
- Wanyonyi, S.; du Preez, R.; Brown, L.; Paul, N.; Panchal, S. Kappaphycus alvarezii as a Food Supplement Prevents Diet-Induced Metabolic Syndrome in Rats. Nutrients 2017, 9, 1261. [Google Scholar]
- du Preez, R.; Paul, N.; Mouatt, P.; Majzoub, M.E.; Thomas, T.; Panchal, S.K.; Brown, L. Carrageenans from the Red Seaweed Sarconema filiforme Attenuate Symptoms of Diet-Induced Metabolic Syndrome in Rats. Mar. Drugs 2020, 18, 97. [Google Scholar]
- Chaidedgumjorn, A.; Toyoda, H.; Woo, E.R.; Lee, K.B.; Kim, Y.S.; Toida, T.; Imanari, T. Effect of (1→3)- and (1→4)-linkages of fully sulfated polysaccharides on their anticoagulant activity. Carbohydr. Res. 2002, 337, 925–933. [Google Scholar]
- Liang, W.; Mao, X.; Peng, X.; Tang, S. Effects of sulfate group in red seaweed polysaccharides on anticoagulant activity and cytotoxicity. Carbohydr. Polym. 2014, 101, 776–785. [Google Scholar]
- Dos Santos-Fidencio, G.C.; Gonçalves, A.G.; Noseda, M.D.; Duarte, M.E.R.; Ducatti, D.R.B. Effects of carboxyl group on the anticoagulant activity of oxidized carrageenans. Carbohydr. Polym. 2019, 214, 286–293. [Google Scholar] [PubMed]
- Zhou, G.; Xin, H.; Sheng, W.; Sun, Y.; Li, Z.; Xu, Z. In vivo growth-inhibition of S180 tumor by mixture of 5-Fu and low molecular λ-carrageenan from Chondrus ocellatus. Pharmacol. Res. 2005, 51, 153–157. [Google Scholar] [PubMed]
- Zhou, G.; Sheng, W.; Yao, W.; Wang, C. Effect of low molecular λ-carrageenan from Chondrus ocellatus on antitumor H-22 activity of 5-Fu. Pharmacol. Res. 2006, 53, 129–134. [Google Scholar] [PubMed]
- Liu, Z.; Gao, T.; Yang, Y.; Meng, F.; Zhan, F.; Jiang, Q.; Sun, X. Anti-Cancer Activity of Porphyran and Carrageenan from Red Seaweeds. Molecules 2019, 24, 4286. [Google Scholar]
- Haijin, M.; Xiaolu, J.; Huashi, G. A κ-carrageenan derived oligosaccharide prepared by enzymatic degradation containing anti-tumor activity. J. Appl. Phycol. 2003, 15, 297–303. [Google Scholar]
- Yuan, H.; Song, J.; Li, X.; Li, N.; Liu, S. Enhanced immunostimulatory and antitumor activity of different derivatives of κ-carrageenan oligosaccharides from Kappaphycus striatum. J. Appl. Phycol. 2011, 23, 59–65. [Google Scholar]
- Luo, M.; Shao, B.; Nie, W.; Wei, X.-W.; Li, Y.-L.; Wang, B.-L.; He, Z.-Y.; Liang, X.; Ye, T.-H.; Wei, Y.-Q. Antitumor and Adjuvant Activity of λ-carrageenan by Stimulating Immune Response in Cancer Immunotherapy. Sci. Rep. 2015, 5, 11062. [Google Scholar]
- Prasedya, E.S.; Miyake, M.; Kobayashi, D.; Hazama, A. Carrageenan delays cell cycle progression in human cancer cells in vitro demonstrated by FUCCI imaging. BMC Complement. Altern. Med. 2016, 16, 270. [Google Scholar]
- Jin, Z.; Han, Y.-X.; Han, X.-R. Degraded Iota-Carrageenan Can Induce Apoptosis in Human Osteosarcoma Cells Via the Wnt/β-Catenin Signaling Pathway. Nutr. Cancer 2013, 65, 126–131. [Google Scholar]
- Calvo, G.H.; Cosenza, V.A.; Sáenz, D.A.; Navarro, D.A.; Stortz, C.A.; Céspedes, M.A.; Mamone, L.A.; Casas, A.G.; Di Venosa, G.M. Disaccharides obtained from carrageenans as potential antitumor agents. Sci. Rep. 2019, 9, 6654. [Google Scholar]
- Cotas, J.; Marques, V.; Afonso, M.B.; Rodrigues, C.M.P.; Pereira, L. Antitumour Potential of Gigartina pistillata Carrageenans against Colorectal Cancer Stem Cell-Enriched Tumourspheres. Mar. Drugs 2020, 18, 50. [Google Scholar]
- Cicinskas, E.; Begun, M.A.; Tiasto, V.A.; Belousov, A.S.; Vikhareva, V.V.; Mikhailova, V.A.; Kalitnik, A.A. In vitro antitumor and immunotropic activity of carrageenans from red algae Chondrus armatus and their low-molecular weight degradation products. J. Biomed. Mater. Res. Part A 2020, 108, 254–266. [Google Scholar]
- Rocha de Souza, M.C.; Marques, C.T.; Guerra Dore, C.M.; Ferreira da Silva, F.R.; Oliveira Rocha, H.A.; Leite, E.L. Antioxidant activities of sulfated polysaccharides from brown and red seaweeds. J. Appl. Phycol. 2007, 19, 153–160. [Google Scholar] [PubMed]
- Souza, M.P.; Vaz, A.F.M.; Costa, T.B.; Cerqueira, M.A.; De Castro, C.M.M.B.; Vicente, A.A.; Carneiro-da-Cunha, M.G. Construction of a Biocompatible and Antioxidant Multilayer Coating by Layer-by-Layer Assembly of κ-Carrageenan and Quercetin Nanoparticles. Food Bioprocess Technol. 2018, 11, 1050–1060. [Google Scholar]
- Pacheco-Quito, E.-M.; Ruiz-Caro, R.; Rubio, J.; Tamayo, A.; Veiga, M.-D. Carrageenan-Based Acyclovir Mucoadhesive Vaginal Tablets for Prevention of Genital Herpes. Mar. Drugs 2020, 18, 249. [Google Scholar]
- Raveendran, S.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Pharmaceutically versatile sulfated polysaccharide based bionano platforms. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 605–626. [Google Scholar]
- Sánchez-Sánchez, M.-P.; Martín-Illana, A.; Ruiz-Caro, R.; Bermejo, P.; Abad, M.-J.; Carro, R.; Bedoya, L.-M.; Tamayo, A.; Rubio, J.; Fernández-Ferreiro, A.; et al. Chitosan and Kappa-Carrageenan Vaginal Acyclovir Formulations for Prevention of Genital Herpes. In Vitro and Ex Vivo Evaluation. Mar. Drugs 2015, 13, 5976–5992. [Google Scholar]
- Zaveri, T.; Hayes, J.; Ziegler, G. Release of Tenofovir from Carrageenan-Based Vaginal Suppositories. Pharmaceutics 2014, 6, 366–377. [Google Scholar]
- Zaveri, T.; Running, C.A.; Surapaneni, L.; Ziegler, G.R.; Hayes, J.E. Innovative sensory methods to access acceptability of mixed polymer semisoft ovules for microbicide applications. Drug Deliv. Transl. Res. 2016, 6, 551–564. [Google Scholar]
- Gu, J.; Yang, S.; Ho, E.A. Biodegradable Film for the Targeted Delivery of siRNA-Loaded Nanoparticles to Vaginal Immune Cells. Mol. Pharm. 2015, 12, 2889–2903. [Google Scholar]
- Mahdavinia, G.R.; Rahmani, Z.; Karami, S.; Pourjavadi, A. Magnetic/pH-sensitive κ-carrageenan/sodium alginate hydrogel nanocomposite beads: Preparation, swelling behavior, and drug delivery. J. Biomater. Sci. Polym. Ed. 2014, 25, 1891–1906. [Google Scholar] [PubMed]
- Kulkarni, R.V.; Boppana, R.; Krishna Mohan, G.; Mutalik, S.; Kalyane, N.V. pH-responsive interpenetrating network hydrogel beads of poly(acrylamide)-g-carrageenan and sodium alginate for intestinal targeted drug delivery: Synthesis, in vitro and in vivo evaluation. J. Colloid Interface Sci. 2012, 367, 509–517. [Google Scholar] [PubMed]
- Ghanam, D.; Kleinebudde, P. Suitability of κ-carrageenan pellets for the formulation of multiparticulate tablets with modified release. Int. J. Pharm. 2011, 409, 9–18. [Google Scholar] [PubMed]
- Valle, B.L.; Omwancha, W.S.; Neau, S.H.; Wigent, R.J. Use of к-carrageenan, chitosan and Carbopol 974P in extruded and spheronized pellets that are devoid of MCC. Drug Dev. Ind. Pharm. 2016, 42, 1903–1916. [Google Scholar]
- Kilor, V.A.; Sapkal, N.P.; Awari, J.G.; Shewale, B.D. Development and Characterization of Enteric-Coated Immediate-Release Pellets of Aceclofenac by Extrusion/Spheronization Technique Using κ-Carrageenan as a Pelletizing Agent. AAPS PharmSciTech 2010, 11, 336–343. [Google Scholar]
- Pettinelli, N.; Rodríguez-Llamazares, S.; Farrag, Y.; Bouza, R.; Barral, L.; Feijoo-Bandín, S.; Lago, F. Poly(hydroxybutyrate-co-hydroxyvalerate) microparticles embedded in κ-carrageenan/locust bean gum hydrogel as a dual drug delivery carrier. Int. J. Biol. Macromol. 2020, 146, 110–118. [Google Scholar]
- Obaidat, R.M.; Alnaief, M.; Mashaqbeh, H. Investigation of Carrageenan Aerogel Microparticles as a Potential Drug Carrier. AAPS PharmSciTech 2018, 19, 2226–2236. [Google Scholar]
- Chan, S.W.; Mirhosseini, H.; Taip, F.S.; Ling, T.C.; Nehdi, I.A.; Tan, C.P. Emulsion formulation optimization and characterization of spray-dried κ-carrageenan microparticles for the encapsulation of CoQ10. Food Sci. Biotechnol. 2016, 25, 53–62. [Google Scholar]
- Huang, W.; Wang, L.; Wei, Y.; Cao, M.; Xie, H.; Wu, D. Fabrication of lysozyme/κ-carrageenan complex nanoparticles as a novel carrier to enhance the stability and in vitro release of curcumin. Int. J. Biol. Macromol. 2020, 146, 444–452. [Google Scholar]
- Sun, X.; Pan, C.; Ying, Z.; Yu, D.; Duan, X.; Huang, F.; Ling, J.; Ouyang, X. Stabilization of zein nanoparticles with k-carrageenan and tween 80 for encapsulation of curcumin. Int. J. Biol. Macromol. 2020, 146, 549–559. [Google Scholar]
- Vijayakumar, S.; Saravanakumar, K.; Malaikozhundan, B.; Divya, M.; Vaseeharan, B.; Durán-Lara, E.F.; Wang, M.H. Biopolymer K-carrageenan wrapped ZnO nanoparticles as drug delivery vehicles for anti MRSA therapy. Int. J. Biol. Macromol. 2020, 144, 9–18. [Google Scholar] [PubMed]
- Zhu, M.; Li, X.; Ge, L.; Zi, Y.; Qi, M.; Li, Y.; Li, D.; Mu, C. Green synthesis of κ-carrageenan@Ag submicron-particles with high aqueous stability, robust antibacterial activity and low cytotoxicity. Mater. Sci. Eng. C 2020, 106, 110185. [Google Scholar]
- Shanmugapriya, K.; Kim, H.; Kang, H.W. A new alternative insight of nanoemulsion conjugated with κ-carrageenan for wound healing study in diabetic mice: In vitro and in vivo evaluation. Eur. J. Pharm. Sci. 2019, 133, 236–250. [Google Scholar] [PubMed]
- Mahdavinia, G.R.; Karimi, M.H.; Soltaniniya, M.; Massoumi, B. In vitro evaluation of sustained ciprofloxacin release from κ-carrageenan-crosslinked chitosan/hydroxyapatite hydrogel nanocomposites. Int. J. Biol. Macromol. 2019, 126, 443–453. [Google Scholar]
- González Ocampo, J.I.; Machado de Paula, M.M.; Bassous, N.J.; Lobo, A.O.; Ossa Orozco, C.P.; Webster, T.J. Osteoblast responses to injectable bone substitutes of kappa-carrageenan and nano hydroxyapatite. Acta Biomater. 2019, 83, 425–434. [Google Scholar]
- Pourjavadi, A.; Doroudian, M.; Ahadpour, A.; Azari, S. Injectable chitosan/κ-carrageenan hydrogel designed with au nanoparticles: A conductive scaffold for tissue engineering demands. Int. J. Biol. Macromol. 2019, 126, 310–317. [Google Scholar]
- Yegappan, R.; Selvaprithiviraj, V.; Amirthalingam, S.; Mohandas, A.; Hwang, N.S.; Jayakumar, R. Injectable angiogenic and osteogenic carrageenan nanocomposite hydrogel for bone tissue engineering. Int. J. Biol. Macromol. 2019, 122, 320–328. [Google Scholar]
- Padhi, J.R.; Nayak, D.; Nanda, A.; Rauta, P.R.; Ashe, S.; Nayak, B. Development of highly biocompatible Gelatin & i-Carrageenan based composite hydrogels: In depth physiochemical analysis for biomedical applications. Carbohydr. Polym. 2016, 153, 292–301. [Google Scholar]
- Amarnath Praphakar, R.; Sumathra, M.; Sam Ebenezer, R.; Vignesh, S.; Shakila, H.; Rajan, M. Fabrication of bioactive rifampicin loaded κ-Car-MA-INH/Nano hydroxyapatite composite for tuberculosis osteomyelitis infected tissue regeneration. Int. J. Pharm. 2019, 565, 543–556. [Google Scholar]
- González Ocampo, J.I.; Bassous, N.; Ossa Orozco, C.P.; Webster, T.J. Evaluation of cytotoxicity and antimicrobial activity of an injectable bone substitute of carrageenan and nano hydroxyapatite. J. Biomed. Mater. Res. Part A 2018, 106, 2984–2993. [Google Scholar]
- Li, B.; Zaveri, T.; Ziegler, G.R.; Hayes, J.E. User Preferences in a Carrageenan-Based Vaginal Drug Delivery System. PLoS ONE 2013, 8, e54975. [Google Scholar]
- Gupta, V.K.; Hariharan, M.; Wheatley, T.A.; Price, J.C. Controlled-release tablets from carrageenans: Effect of formulation, storage and dissolution factors. Eur. J. Pharm. Biopharm. 2001, 51, 241–248. [Google Scholar] [PubMed]
- Buchholcz, G.; Kelemen, A.; Sovány, T.; Pintye-Hódi, K. Matrix tablets based on a carrageenan with the modified-release of sodium riboflavin 5′-phosphate. Pharm. Dev. Technol. 2015, 20, 676–683. [Google Scholar]
- Bettini, R.; Bonferoni, M.C.; Colombo, P.; Zanelotti, L.; Caramella, C. Drug Release Kinetics and Front Movement in Matrix Tablets Containing Diltiazem or Metoprolol/ λ-Carrageenan Complexes. Biomed Res. Int. 2014, 2014, 1–8. [Google Scholar]
- Kos, P.; Pavli, M.; Baumgartner, S.; Kogej, K. Release mechanism of doxazosin from carrageenan matrix tablets: Effect of ionic strength and addition of sodium dodecyl sulphate. Int. J. Pharm. 2017, 529, 557–567. [Google Scholar] [PubMed]
- Yermak, I.; Mischchenko, N.; Davydova, V.; Glazunov, V.; Tarbeeva, D.; Kravchenko, A.; Pimenova, E.; Sorokina, I. Carrageenans-Sulfated Polysaccharides from Red Seaweeds as Matrices for the Inclusion of Echinochrome. Mar. Drugs 2017, 15, 337. [Google Scholar]
- Ozsoy, Y.; Bergişadi, N. Preparation of mefenamic acid sustained release beads based on kappa-carrageenan. Boll. Chim. Farm. 2000, 139, 120–123. [Google Scholar] [PubMed]
- Piyakulawat, P.; Praphairaksit, N.; Chantarasiri, N.; Muangsin, N. Preparation and evaluation of chitosan/carrageenan beads for controlled release of sodium diclofenac. AAPS PharmSciTech 2007, 8, 120. [Google Scholar]
- Mahdavinia, G.R.; Etemadi, H.; Soleymani, F. Magnetic/pH-responsive beads based on caboxymethyl chitosan and κ-carrageenan and controlled drug release. Carbohydr. Polym. 2015, 128, 112–121. [Google Scholar]
- Kim, E.-H.; Choi, H.-K. Preparation of Various Solid-Lipid Beads for Drug Delivery of Enrofloxacin. Drug Deliv. 2004, 11, 365–370. [Google Scholar]
- Belščak-Cvitanović, A.; Komes, D.; Karlović, S.; Djaković, S.; Špoljarić, I.; Mršić, G.; Ježek, D. Improving the controlled delivery formulations of caffeine in alginate hydrogel beads combined with pectin, carrageenan, chitosan and psyllium. Food Chem. 2015, 167, 378–386. [Google Scholar] [PubMed]
- Azizi, S.; Mohamad, R.; Abdul Rahim, R.; Mohammadinejad, R.; Bin Ariff, A. Hydrogel beads bio-nanocomposite based on Kappa-Carrageenan and green synthesized silver nanoparticles for biomedical applications. Int. J. Biol. Macromol. 2017, 104, 423–431. [Google Scholar] [PubMed]
- Sathuvan, M.; Thangam, R.; Gajendiran, M.; Vivek, R.; Balasubramanian, S.; Nagaraj, S.; Gunasekaran, P.; Madhan, B.; Rengasamy, R. κ-Carrageenan: An effective drug carrier to deliver curcumin in cancer cells and to induce apoptosis. Carbohydr. Polym. 2017, 160, 184–193. [Google Scholar] [PubMed]
- Bornhöft, M.; Thommes, M.; Kleinebudde, P. Preliminary assessment of carrageenan as excipient for extrusion/spheronisation. Eur. J. Pharm. Biopharm. 2005, 59, 127–131. [Google Scholar] [PubMed]
- Thommes, M.; Kleinebudde, P. Use of κ-carrageenan as alternative pelletisation aid to microcrystalline cellulose in extrusion/spheronisation. I. Influence of type and fraction of filler. Eur. J. Pharm. Biopharm. 2006, 63, 59–67. [Google Scholar]
- Thommes, M.; Kleinebudde, P. Properties of pellets manufactured by wet extrusion/spheronization process using κ-carrageenan: Effect of process parameters. AAPS PharmSciTech 2007, 8, 101–108. [Google Scholar]
- Ige, P.P.; Rajput, P.; Pardeshi, C.; Kawade, R.; Swami, B.; Mahajan, H.; Nerkar, P.; Belgamwar, V.; Surana, S.; Gattani, S. Development of pellets of nifedipine using HPMC K15 M and κ-carrageenan as mucoadhesive sustained delivery system and in vitro evaluation. Iran. Polym. J. 2013, 22, 911–921. [Google Scholar]
- Tran, T.T.D.; Tran, P.H.L. Controlled Release Film Forming Systems in Drug Delivery: The Potential for Efficient Drug Delivery. Pharmaceutics 2019, 11, 290. [Google Scholar]
- Sedayu, B.B.; Cran, M.J.; Bigger, S.W. A Review of Property Enhancement Techniques for Carrageenan-based Films and Coatings. Carbohydr. Polym. 2019, 216, 287–302. [Google Scholar]
- Boateng, J.S.; Pawar, H.V.; Tetteh, J. Polyox and carrageenan based composite film dressing containing anti-microbial and anti-inflammatory drugs for effective wound healing. Int. J. Pharm. 2013, 441, 181–191. [Google Scholar]
- Shojaee-Aliabadi, S.; Hosseini, H.; Mohammadifar, M.A.; Mohammadi, A.; Ghasemlou, M.; Hosseini, S.M.; Khaksar, R. Characterization of κ-carrageenan films incorporated plant essential oils with improved antimicrobial activity. Carbohydr. Polym. 2014, 101, 582–591. [Google Scholar] [PubMed]
- Fouda, M.M.G.; El-Aassar, M.R.; El Fawal, G.F.; Hafez, E.E.; Masry, S.H.D.; Abdel-Megeed, A. k-Carrageenan/poly vinyl pyrollidone/polyethylene glycol/silver nanoparticles film for biomedical application. Int. J. Biol. Macromol. 2015, 74, 179–184. [Google Scholar] [PubMed]
- Oun, A.A.; Rhim, J.-W. Carrageenan-based hydrogels and films: Effect of ZnO and CuO nanoparticles on the physical, mechanical, and antimicrobial properties. Food Hydrocoll. 2017, 67, 45–53. [Google Scholar]
- Yu, H.C.; Zhang, H.; Ren, K.; Ying, Z.; Zhu, F.; Qian, J.; Ji, J.; Wu, Z.L.; Zheng, Q. Ultrathin κ-Carrageenan/Chitosan Hydrogel Films with High Toughness and Antiadhesion Property. ACS Appl. Mater. Interfaces 2018, 10, 9002–9009. [Google Scholar] [PubMed]
- Jaiswal, L.; Shankar, S.; Rhim, J.-W. Carrageenan-based functional hydrogel film reinforced with sulfur nanoparticles and grapefruit seed extract for wound healing application. Carbohydr. Polym. 2019, 224, 115191. [Google Scholar] [PubMed]
- Bani-Jaber, A.; Abdullah, S. Development and characterization of novel ambroxol sustained-release oral suspensions based on drug-polymeric complexation and polymeric raft formation. Pharm. Dev. Technol. 2020, 25, 666–675. [Google Scholar]
- Venkatesan, J.; Anil, S.; Kim, S.-K.; Shim, M. Seaweed Polysaccharide-Based Nanoparticles: Preparation and Applications for Drug Delivery. Polymers (Basel) 2016, 8, 30. [Google Scholar]
- Luo, Y.; Wang, Q. Recent development of chitosan-based polyelectrolyte complexes with natural polysaccharides for drug delivery. Int. J. Biol. Macromol. 2014, 64, 353–367. [Google Scholar]
- Briones, A.V.; Sato, T. Encapsulation of glucose oxidase (GOD) in polyelectrolyte complexes of chitosan–carrageenan. React. Funct. Polym. 2010, 70, 19–27. [Google Scholar]
- Grenha, A.; Gomes, M.E.; Rodrigues, M.; Santo, V.E.; Mano, J.F.; Neves, N.M.; Reis, R.L. Development of new chitosan/carrageenan nanoparticles for drug delivery applications. J. Biomed. Mater. Res. A 2010, 92, 1265–1272. [Google Scholar]
- Rodrigues, S.; da Costa, A.M.R.; Grenha, A. Chitosan/carrageenan nanoparticles: Effect of cross-linking with tripolyphosphate and charge ratios. Carbohydr. Polym. 2012, 89, 282–289. [Google Scholar] [PubMed]
- Rodrigues, S.; Cordeiro, C.; Seijo, B.; Remuñán-López, C.; Grenha, A. Hybrid nanosystems based on natural polymers as protein carriers for respiratory delivery: Stability and toxicological evaluation. Carbohydr. Polym. 2015, 123, 369–380. [Google Scholar] [PubMed]
- Rosas-Durazo, A.; Lizardi, J.; Higuera-Ciapara, I.; Argüelles-Monal, W.; Goycoolea, F.M. Development and characterization of nanocapsules comprising dodecyltrimethylammonium chloride and κ-carrageenan. Colloids Surf. B Biointerfaces 2011, 86, 242–246. [Google Scholar] [PubMed]
- Guzman-Villanueva, D.; El-Sherbiny, I.M.; Herrera-Ruiz, D.; Smyth, H.D.C. Design and In Vitro Evaluation of a New Nano-Microparticulate System for Enhanced Aqueous-Phase Solubility of Curcumin. Biomed Res. Int. 2013, 2013, 1–9. [Google Scholar]
- Leong, K.H.; Chung, L.Y.; Noordin, M.I.; Onuki, Y.; Morishita, M.; Takayama, K. Lectin-functionalized carboxymethylated kappa-carrageenan microparticles for oral insulin delivery. Carbohydr. Polym. 2011, 86, 555–565. [Google Scholar]
- Devi, N.; Maji, T.K. Genipin Crosslinked Chitosan-κ-carrageenan Polyelectrolyte Nanocapsules for the Controlled Delivery of Isoniazid. Int. J. Polym. Mater. 2010, 59, 828–841. [Google Scholar]
- Bosio, V.E.; Cacicedo, M.L.; Calvignac, B.; León, I.; Beuvier, T.; Boury, F.; Castro, G.R. Synthesis and characterization of CaCO 3 –biopolymer hybrid nanoporous microparticles for controlled release of doxorubicin. Colloids Surf. B Biointerfaces 2014, 123, 158–169. [Google Scholar]
- Raman, M.; Devi, V.; Doble, M. Biocompatible ι-carrageenan-γ-maghemite nanocomposite for biomedical applications–synthesis, characterization and in vitro anticancer efficacy. J. Nanobiotechnol. 2015, 13, 18. [Google Scholar]
- Karimi, M.H.; Mahdavinia, G.R.; Massoumi, B. pH-controlled sunitinib anticancer release from magnetic chitosan nanoparticles crosslinked with κ-carrageenan. Mater. Sci. Eng. C 2018, 91, 705–714. [Google Scholar]
- Alnaief, M.; Obaidat, R.; Mashaqbeh, H. Effect of processing parameters on preparation of carrageenan aerogel microparticles. Carbohydr. Polym. 2018, 180, 264–275. [Google Scholar]
- Selvakumaran, S.; Muhamad, I.I. Evaluation of kappa carrageenan as potential carrier for floating drug delivery system: Effect of cross linker. Int. J. Pharm. 2015, 496, 323–331. [Google Scholar] [PubMed]
- Selvakumaran, S.; Muhamad, I.I.; Abd Razak, S.I. Evaluation of kappa carrageenan as potential carrier for floating drug delivery system: Effect of pore forming agents. Carbohydr. Polym. 2016, 135, 207–214. [Google Scholar] [PubMed]
- Hebar, A.; Koller, C.; Seifert, J.-M.; Chabicovsky, M.; Bodenteich, A.; Bernkop-Schnürch, A.; Grassauer, A.; Prieschl-Grassauer, E. Non-Clinical Safety Evaluation of Intranasal Iota-Carrageenan. PLoS ONE 2015, 10, e0122911. [Google Scholar]
- Alavi, S.; Mortazavi, S.A. Freeze-Dried K-Carrageenan/Chitosan Polyelectrolyte Complex-Based Insert: A Novel Intranasal Delivery System for Sumatriptan Succinate. Iran. J. Pharm. Res. IJPR 2018, 17, 1172–1181. [Google Scholar]
- Kianfar, F.; Antonijevic, M.; Chowdhry, B.; Boateng, J.S. Lyophilized wafers comprising carrageenan and pluronic acid for buccal drug delivery using model soluble and insoluble drugs. Colloids Surf. B Biointerfaces 2013, 103, 99–106. [Google Scholar]
- Pawar, H.V.; Boateng, J.S.; Ayensu, I.; Tetteh, J. Multifunctional Medicated Lyophilised Wafer Dressing for Effective Chronic Wound Healing. J. Pharm. Sci. 2014, 103, 1720–1733. [Google Scholar]
- Chai, Q.; Jiao, Y.; Yu, X. Hydrogels for Biomedical Applications: Their Characteristics and the Mechanisms behind Them. Gels 2017, 3, 6. [Google Scholar]
- Farris, S.; Schaich, K.M.; Liu, L.; Piergiovanni, L.; Yam, K.L. Development of polyion-complex hydrogels as an alternative approach for the production of bio-based polymers for food packaging applications: A review. Trends Food Sci. Technol. 2009, 20, 316–332. [Google Scholar]
- Song, R.; Murphy, M.; Li, C.; Ting, K.; Soo, C.; Zheng, Z. Current development of biodegradable polymeric materials for biomedical applications. Drug Des. Devel. Ther. 2018, 12, 3117–3145. [Google Scholar]
- Salgueiro, A.M.; Daniel-da-Silva, A.L.; Fateixa, S.; Trindade, T. κ-Carrageenan hydrogel nanocomposites with release behavior mediated by morphological distinct Au nanofillers. Carbohydr. Polym. 2013, 91, 100–109. [Google Scholar]
- Jayaramudu, T.; Raghavendra, G.M.; Varaprasad, K.; Sadiku, R.; Ramam, K.; Raju, K.M. Iota-Carrageenan-based biodegradable Ag0 nanocomposite hydrogels for the inactivation of bacteria. Carbohydr. Polym. 2013, 95, 188–194. [Google Scholar] [PubMed]
- Hezaveh, H.; Muhamad, I.I. Modification and swelling kinetic study of kappa-carrageenan-based hydrogel for controlled release study. J. Taiwan Inst. Chem. Eng. 2013, 44, 182–191. [Google Scholar]
- Hezaveh, H.; Muhamad, I.I. Controlled drug release via minimization of burst release in pH-response kappa-carrageenan/polyvinyl alcohol hydrogels. Chem. Eng. Res. Des. 2013, 91, 508–519. [Google Scholar]
- El-Aassar, M.R.; El Fawal, G.F.; Kamoun, E.A.; Fouda, M.M.G. Controlled drug release from cross-linked κ-carrageenan/hyaluronic acid membranes. Int. J. Biol. Macromol. 2015, 77, 322–329. [Google Scholar] [PubMed]
- Rasool, A.; Ata, S.; Islam, A.; Rizwan, M.; Azeem, M.K.; Mehmood, A.; Khan, R.U.; Qureshi, A. ur R.; Mahmood, H.A. Kinetics and controlled release of lidocaine from novel carrageenan and alginate-based blend hydrogels. Int. J. Biol. Macromol. 2020, 147, 67–78. [Google Scholar] [PubMed]
- Mawazi, S.M.; Al-Mahmood, S.M.A.; Chatterjee, B.; Hadi, H.A.B.; Doolaanea, A.A. Carbamazepine gel formulation as a sustained release epilepsy medication for pediatric use. Pharmaceutics 2019, 11, 488. [Google Scholar]
- Zhao, J.; Sun, C.; Li, H.; Dong, X.; Zhang, X. Studies on the physicochemical properties, gelling behavior and drug release performance of agar/κ-carrageenan mixed hydrogels. Int. J. Biol. Macromol. 2020, 154, 878–887. [Google Scholar]
- Daniel-Da-Silva, A.L.; Lopes, A.B.; Gil, A.M.; Correia, R.N. Synthesis and characterization of porous κ-carrageenan/calcium phosphate nanocomposite scaffolds. J. Mater. Sci. 2007, 42, 8581–8591. [Google Scholar]
- Kim, I.Y.; Iwatsuki, R.; Kikuta, K.; Morita, Y.; Miyazaki, T.; Ohtsuki, C. Thermoreversible behavior of κ-carrageenan and its apatite-forming ability in simulated body fluid. Mater. Sci. Eng. C 2011, 31, 1472–1476. [Google Scholar]
- Feng, W.; Feng, S.; Tang, K.; He, X.; Jing, A.; Liang, G. A novel composite of collagen-hydroxyapatite/kappa-carrageenan. J. Alloys Compd. 2017, 693, 482–489. [Google Scholar]
- Mirza, S.; Jolly, R.; Zia, I.; Saad Umar, M.; Owais, M.; Shakir, M. Bioactive Gum Arabic/κ-Carrageenan-Incorporated Nano-Hydroxyapatite Nanocomposites and Their Relative Biological Functionalities in Bone Tissue Engineering. ACS Omega 2020, 5, 11279–11290. [Google Scholar] [PubMed]
- González, J.I.; Ossa, C.P.O. Injectability Evaluation of Bone-Graft Substitutes Based on Carrageenan and Hydroxyapatite Nanorods. In Minerals, Metals and Materials Series; Springer International Publishing: Berlin/Heidelberg, Germany, 2017; pp. 33–46. ISBN 9783319521312. [Google Scholar]
- Santo, V.E.; Frias, A.M.; Carida, M.; Cancedda, R.; Gomes, M.E.; Mano, J.F.; Reis, R.L. Carrageenan-Based Hydrogels for the Controlled Delivery of PDGF-BB in Bone Tissue Engineering Applications. Biomacromolecules 2009, 10, 1392–1401. [Google Scholar] [PubMed]
- Rocha, P.M.; Santo, V.E.; Gomes, M.E.; Reis, R.L.; Mano, J.F. Encapsulation of adipose-derived stem cells and transforming growth factor-β1 in carrageenan-based hydrogels for cartilage tissue engineering. J. Bioact. Compat. Polym. 2011, 26, 493–507. [Google Scholar]
- Soares, P.A.G.; C de Seixas, J.R.P.; Albuquerque, P.B.S.; Santos, G.R.C.; Mourão, P.A.S.; Barros, W.; Correia, M.T.S.; Carneiro-da-Cunha, M.G. Development and characterization of a new hydrogel based on galactomannan and κ-carrageenan. Carbohydr. Polym. 2015, 134, 673–679. [Google Scholar]
- Popa, E.G.; Carvalho, P.P.; Dias, A.F.; Santos, T.C.; Santo, V.E.; Marques, A.P.; Viegas, C.A.; Dias, I.R.; Gomes, M.E.; Reis, R.L. Evaluation of the in vitro and in vivo biocompatibility of carrageenan-based hydrogels. J. Biomed. Mater. Res. Part A 2014, 102, 4087–4097. [Google Scholar]
- Rnjak-Kovacina, J.; Wray, L.S.; Burke, K.A.; Torregrosa, T.; Golinski, J.M.; Huang, W.; Kaplan, D.L. Lyophilized Silk Sponges: A Versatile Biomaterial Platform for Soft Tissue Engineering. ACS Biomater. Sci. Eng. 2015, 1, 260–270. [Google Scholar]
- Kretlow, J.D.; Klouda, L.; Mikos, A.G. Injectable matrices and scaffolds for drug delivery in tissue engineering. Adv. Drug Deliv. Rev. 2007, 59, 263–273. [Google Scholar]
- Mokhtari, H.; Kharaziha, M.; Karimzadeh, F.; Tavakoli, S. An injectable mechanically robust hydrogel of Kappa-carrageenan-dopamine functionalized graphene oxide for promoting cell growth. Carbohydr. Polym. 2019, 214, 234–249. [Google Scholar]
- Balasubramanian, R.; Kim, S.S.; Lee, J. Novel synergistic transparent k -Carrageenan/Xanthan gum/Gellan gum hydrogel film: Mechanical, thermal and water barrier properties. Int. J. Biol. Macromol. 2018, 118, 561–568. [Google Scholar]
- Popa, E.G.; Caridade, S.G.; Mano, J.F.; Reis, R.L.; Gomes, M.E. Chondrogenic potential of injectable κ -carrageenan hydrogel with encapsulated adipose stem cells for cartilage tissue-engineering applications. J. Tissue Eng. Regen. Med. 2015, 9, 550–563. [Google Scholar]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [PubMed]
- Zhang, Y.; Zhou, D.; Chen, J.; Zhang, X.; Li, X.; Zhao, W.; Xu, T. Biomaterials Based on Marine Resources for 3D Bioprinting Applications. Mar. Drugs 2019, 17, 555. [Google Scholar]
- Kelder, C.; Bakker, A.; Klein-Nulend, J.; Wismeijer, D. The 3D Printing of Calcium Phosphate with K-Carrageenan under Conditions Permitting the Incorporation of Biological Components—A Method. J. Funct. Biomater. 2018, 9, 57. [Google Scholar]
- Li, H.; Tan, Y.J.; Li, L. A strategy for strong interface bonding by 3D bioprinting of oppositely charged κ-carrageenan and gelatin hydrogels. Carbohydr. Polym. 2018, 198, 261–269. [Google Scholar]
- Kim, M.H.; Lee, Y.W.; Jung, W.-K.; Oh, J.; Nam, S.Y. Enhanced rheological behaviors of alginate hydrogels with carrageenan for extrusion-based bioprinting. J. Mech. Behav. Biomed. Mater. 2019, 98, 187–194. [Google Scholar] [PubMed]
- Li, H.; Tan, Y.J.; Liu, S.; Li, L. Three-Dimensional Bioprinting of Oppositely Charged Hydrogels with Super Strong Interface Bonding. ACS Appl. Mater. Interfaces 2018, 10, 11164–11174. [Google Scholar] [PubMed]
- Tytgat, L.; Van Damme, L.; del P. Ortega Arevalo, M.; Declercq, H.; Thienpont, H.; Otteveare, H.; Blondeel, P.; Dubruel, P.; Van Vlierberghe, S. Extrusion-based 3D printing of photo-crosslinkable gelatin and κ-carrageenan hydrogel blends for adipose tissue regeneration. Int. J. Biol. Macromol. 2019, 140, 929–938. [Google Scholar]
| Sulphated Polysaccharide | Marine Algae Group | Main Genera | Type of Carrageenan | Chemical Structure | Ester Sulphate Content (%) | 3,6-AG Content (%) | Reference |
|---|---|---|---|---|---|---|---|
| Carrageenan | Rhodophyceae | Chondrus, Euchema, Furcellaria, Fucus, Gigartina, Hypnea, Iridae, Kappaphycus | Kappa-CG | 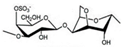 | 25–30 | 28–35 | [35] |
| Iota-CG | 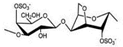 | 28–30 | 25–30 | ||||
| Lambda-CG | 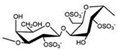 | 32–39 | – |
| Bioactive Properties | Type of CG | Applications | Reference |
|---|---|---|---|
| Antiviral activity | Kappa-CG Iota-CG | Inhibits Herpes Simplex Virus (HSV), Human Papillomavirus (HPV), Varicella Zoster Virus (VZV) and Human Rhinoviruses | [4,5,20,45,59,60] |
| Lambda-CG/kappa-CG | Bioactivity against HPV and HSV-2 | [64] | |
| CG | Genital HPV infection | [65] | |
| Iota-CG | Reduction in cold symptoms and reduces the growth of Human Rhinoviruses (HRV) | [66] | |
| Iota-CG | Potential inhibitor of the Influenza A Virus infection | [67] | |
| Kappa-CG | H1N1/2009 and other similar viruses | [68] | |
| Iota-CG Kappa-CG | Influenza A Virus strains (pandemic H1N1/09, H3N2, H5N1, H7N7) | [69] | |
| Iota-CG | Human Rhinovirus (HRV) 1a, hRV8 and Human Coronavirus OC43 | [70] | |
| Kappa-CG | Enterovirus 71 (EV 71) | [71] | |
| Lambda-CG | Rabies Virus (RABV) | [72] | |
| Kappa-CG Iota-CG Lambda-CG | Varicella Zoster Virus (VZV) | [73] | |
| Iota-CG | Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) | [74] | |
| Antibacterial effects | Iota-CG | Inhibits the growth of the bacterial strains | [77] |
| Iota-CG | Ocular Chlamydia trachomatis infection | [30] | |
| Kappa-CG | Activity against Saccharomyces cerevisiae | [78] | |
| Kappa-CG | Activity against Gram-positive and Gram-negative bacteria | [79] | |
| Kappa-CG | Reduced the production of interleukin-6 in cells treated with kappa-CG | [80] | |
| Kappa-CG | Activity against S. aureus and E. coli | [81] | |
| Kappa-CG | Activity against S. aureus, Bacillus cereus, E. coli and Pseudomonas aeruginosa | [82] | |
| Antihyperlipidemic effects | CG | Hypocholesterolemic effect | [45] |
| Kappa-CG | Reduces serum levels of total cholesterol, triglycerides and low-density lipoprotein cholesterol (LDL-C), and increasing high-density lipoprotein cholesterol (HDL-C) | [84,85] | |
| Kappa-CG kappa/β-CG Iota/kappa-CG | Modulate prostaglandin E2 synthesis and stimulate IL-1β and IL-6 synthesis | [86] | |
| Kappa-CG/iota-CG | Reduces in serum levels of total cholesterol | [87] | |
| CG | Metabolic syndrome | [88] | |
| Iota-CG | Metabolic syndrome | [89] | |
| Anticoagulant and antithrombotic activity | Lambda-CG | Highest anticoagulant activity in the rabbit whole blood test | [91] |
| Lambda-CG | Antithrombotic activity | [4,21] | |
| Kappa-CG Iota-CG Iota/nu-CG Theta-CG Lambda-CG | Anticoagulant activity | [92] | |
| Antitumor and immunomodulatory activity | Lambda-CG | Anticancer effects, immunomodulation | [95,97] |
| Lambda-CG | Improve the antitumor activity of 5-Fluorouracil | [93] | |
| Lambda-CG | Inhibits tumour growth in mice with murine melanoma cell lines | [98] | |
| Kappa-CG Lambda-CG | Kappa-CG delays the cell cycle in the G2/M phase Lambda-CG stalled the cell cycle in both the G1 and G2/M phase | [99] | |
| Iota-CG | Suppressed tumour growth, induced apoptosis, and halted the G1 phase | [100] | |
| Kappa-CG Iota-CG | Cytotoxic effect on LM2 tumour cells | [101] | |
| Lambda-CG/epsilon-CG | Inhibits colorectal cancer stem-like cells | [102] | |
| Kappa-CG Lambda-CG | Antitumour and immunotropic effects | [103] | |
| Antioxidant activity | Kappa-CG | Antioxidant activity in the multilayer coating | [105] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacheco-Quito, E.-M.; Ruiz-Caro, R.; Veiga, M.-D. Carrageenan: Drug Delivery Systems and Other Biomedical Applications. Mar. Drugs 2020, 18, 583. https://doi.org/10.3390/md18110583
Pacheco-Quito E-M, Ruiz-Caro R, Veiga M-D. Carrageenan: Drug Delivery Systems and Other Biomedical Applications. Marine Drugs. 2020; 18(11):583. https://doi.org/10.3390/md18110583
Chicago/Turabian StylePacheco-Quito, Edisson-Mauricio, Roberto Ruiz-Caro, and María-Dolores Veiga. 2020. "Carrageenan: Drug Delivery Systems and Other Biomedical Applications" Marine Drugs 18, no. 11: 583. https://doi.org/10.3390/md18110583
APA StylePacheco-Quito, E.-M., Ruiz-Caro, R., & Veiga, M.-D. (2020). Carrageenan: Drug Delivery Systems and Other Biomedical Applications. Marine Drugs, 18(11), 583. https://doi.org/10.3390/md18110583