Viridicatol Isolated from Deep-Sea Penicillium Griseofulvum Alleviates Anaphylaxis and Repairs the Intestinal Barrier in Mice by Suppressing Mast Cell Activation
Abstract
1. Introduction
2. Results
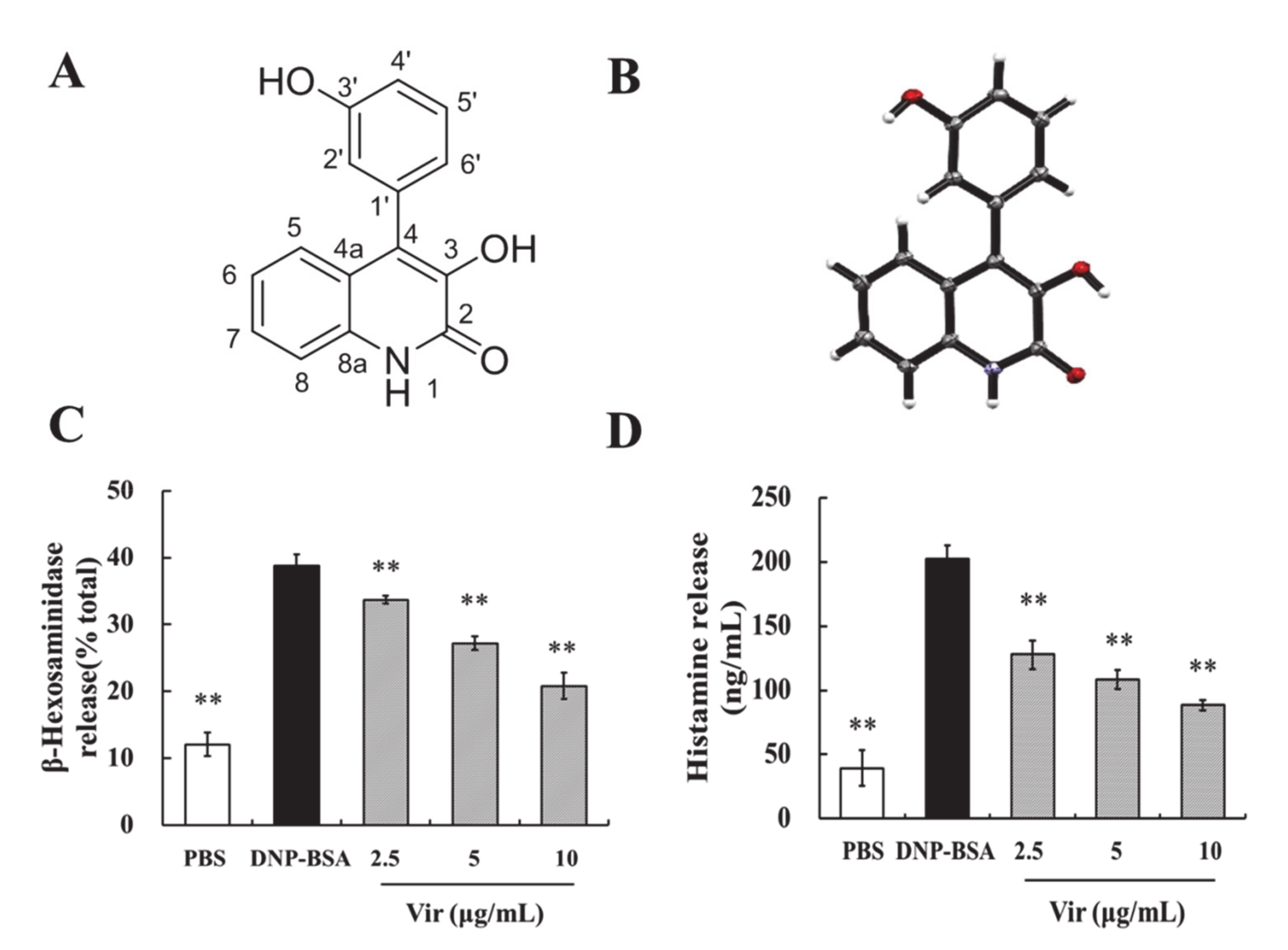
2.1. Structural Determination of Viridicatol
2.2. X-ray Crystallography of Viridicatol
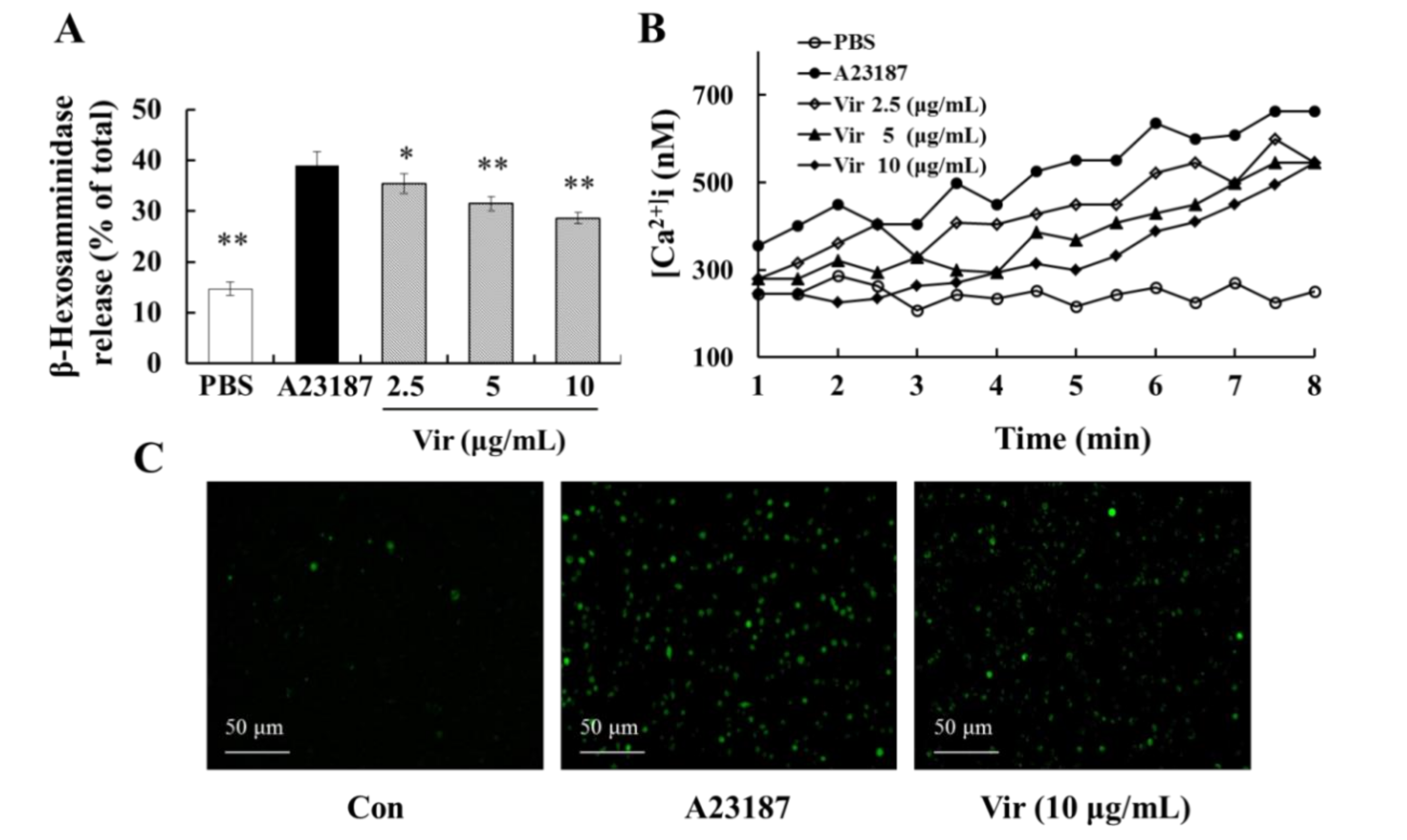
2.3. Viridicatol Decreased the Release of β-Hexosaminidase and Histamine in RBL-2H3 Cells
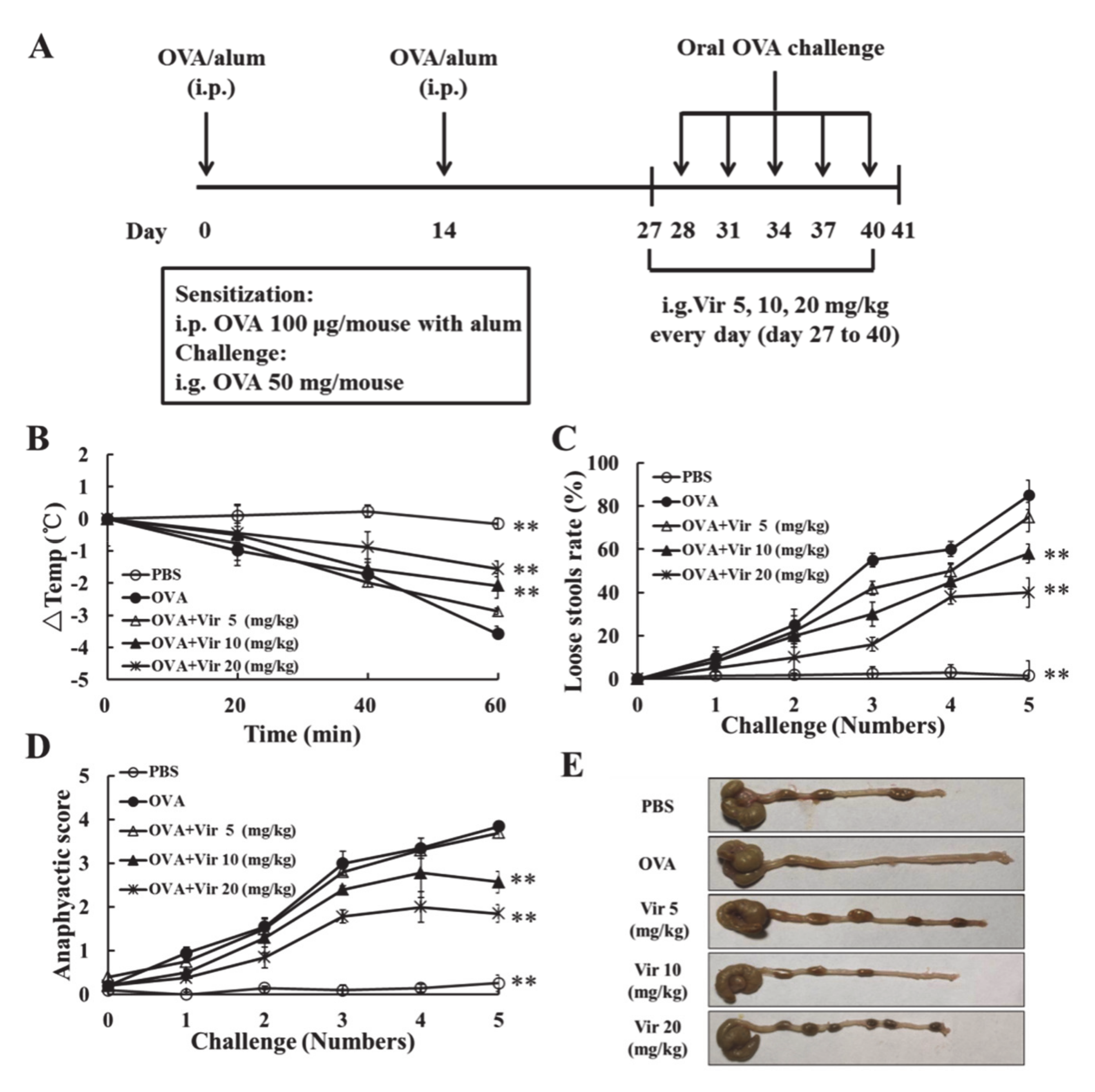
2.4. Viridicatol Relieved OVA-Induced Allergic Symptoms in Mice
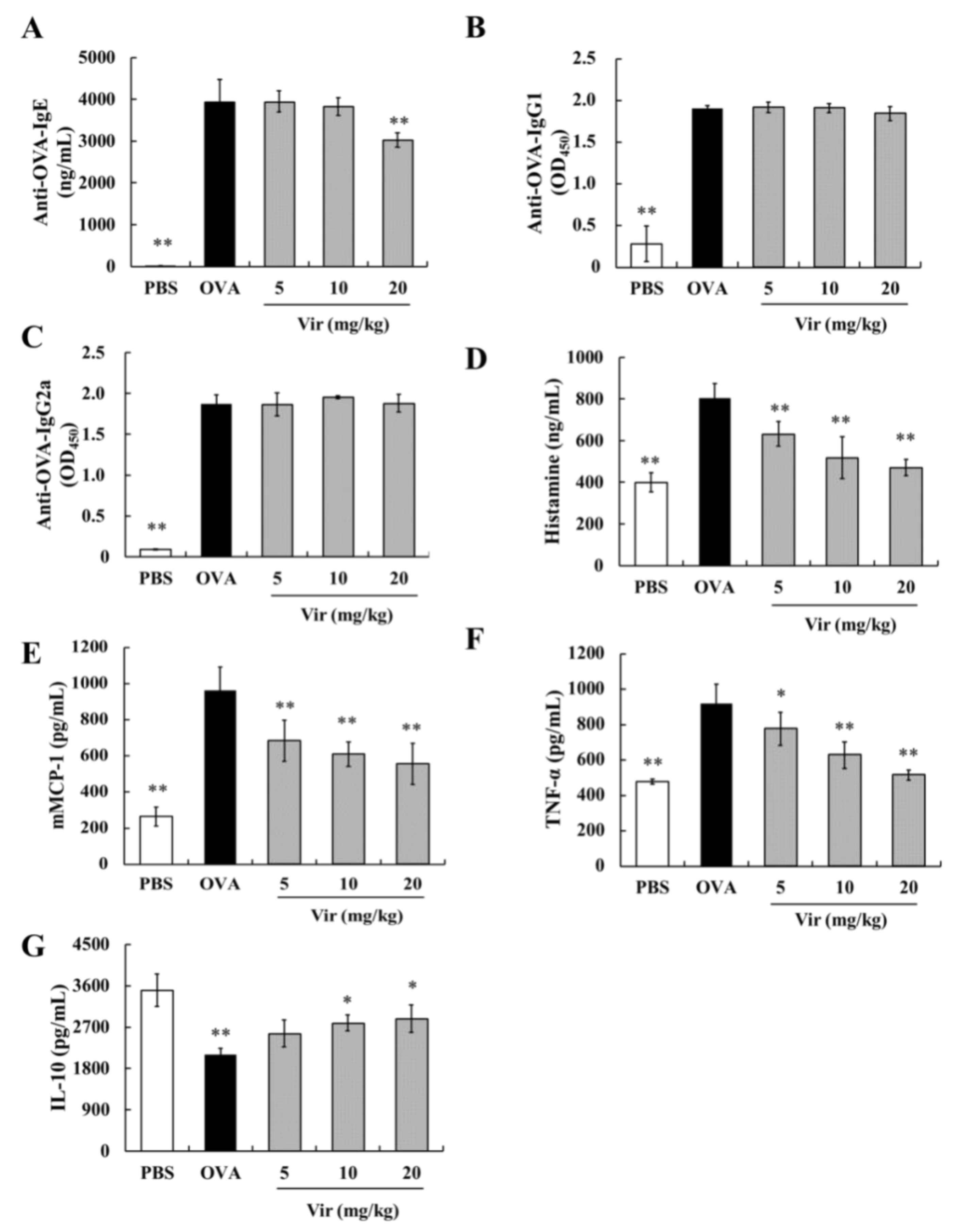
2.5. Effects of Viridicatol on Immunoglobulins, Anaphylactic Mediators, and Cytokines in the Mouse Serum
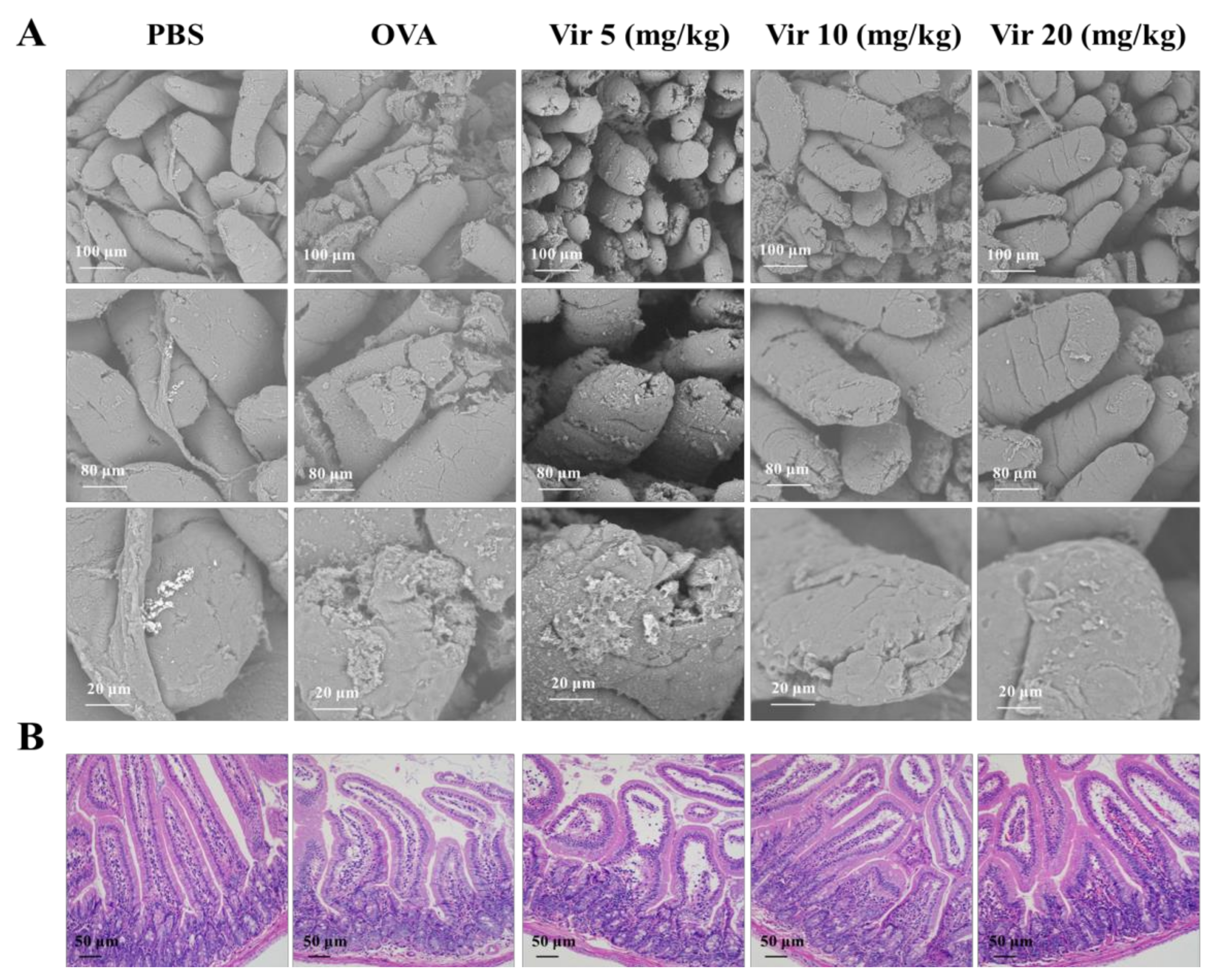
2.6. Viridicatol Alleviated Intestinal Injury and Inflammation of the Jejunum
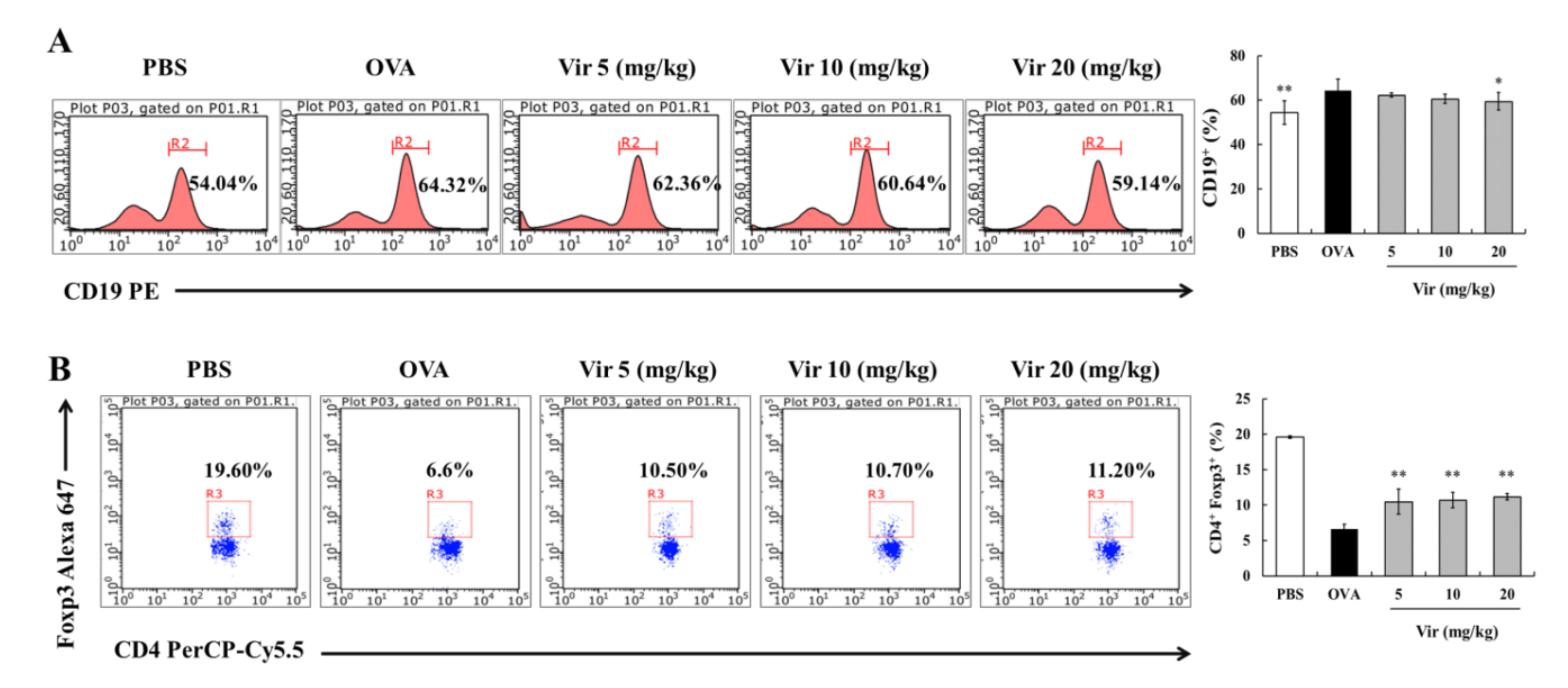
2.7. Effects of Viridicatol on the Subpopulation of B Cells and Tregs in the Spleen
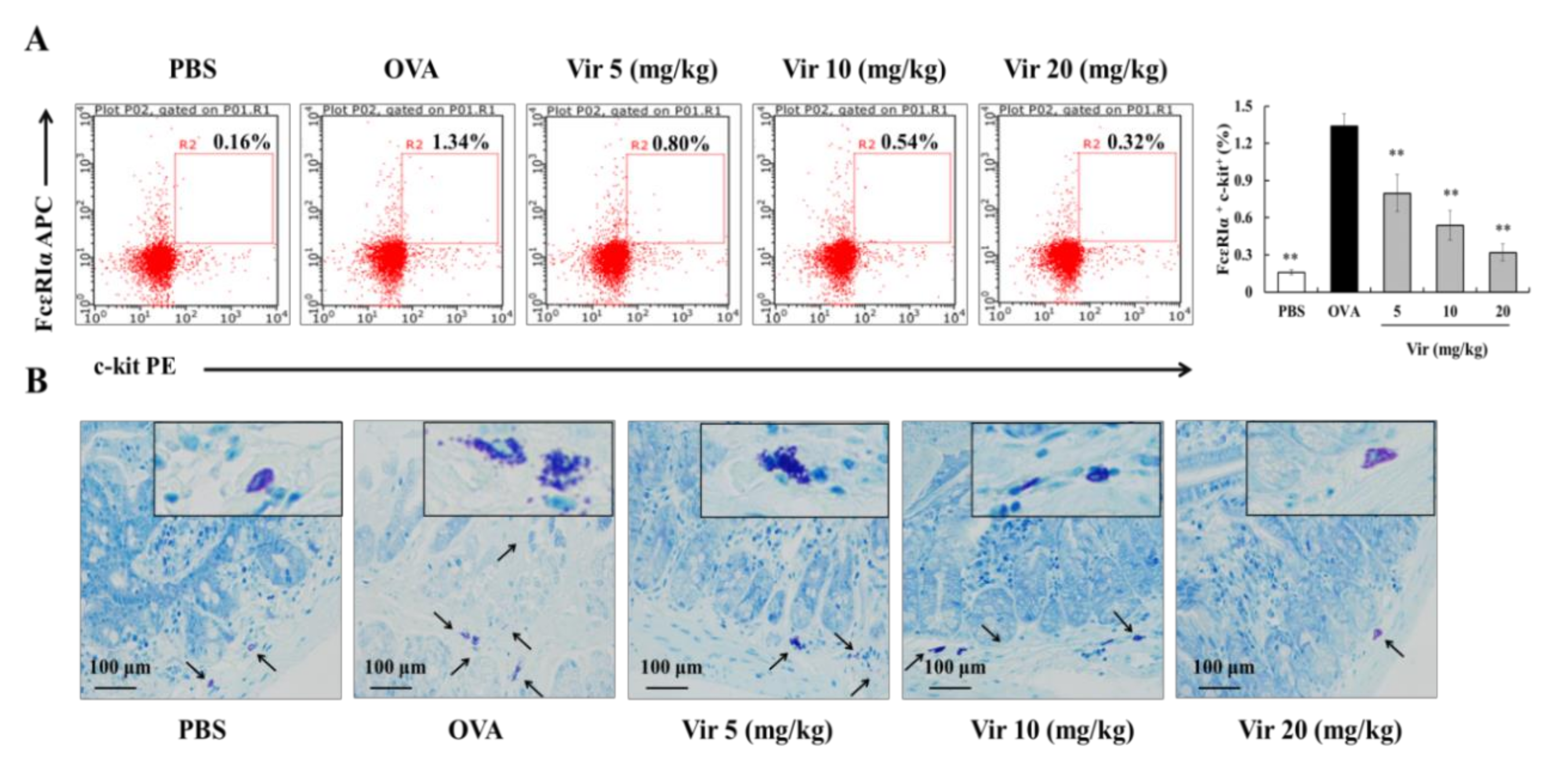
2.8. Effects of Viridicatol on MC Population and Degranulation
2.9. Viridicatol Decreased the Level of Intracellular Calcium
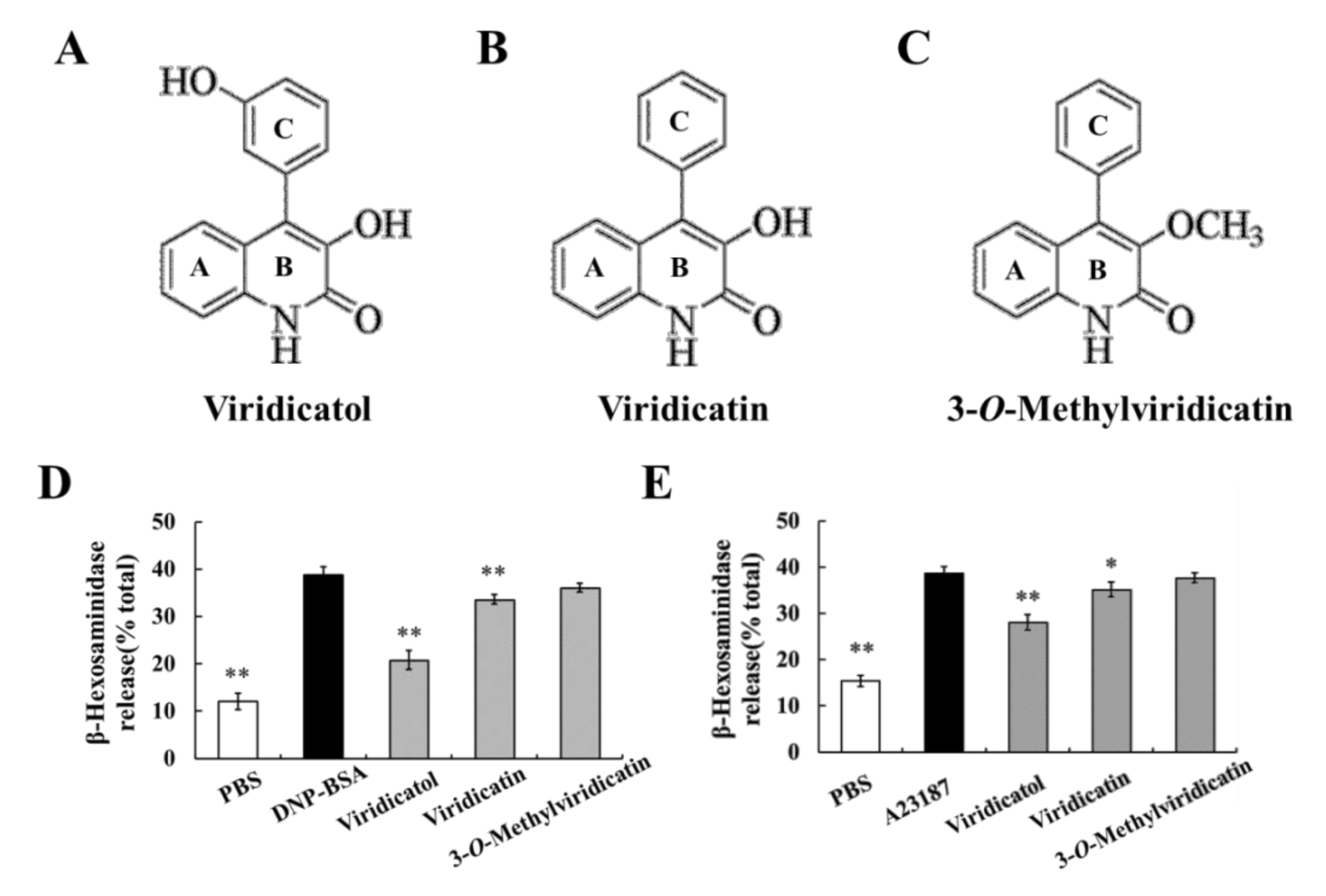
2.10. Relationship between the Chemical Structure and Inhibitory Effects on Cell Degranulation
3. Discussion
4. Materials and Methods
4.1. Reagents an General Experimental Procedures
4.2. Cell Culture
4.3. Animals
4.4. Viridicatol Extraction, Isolation, and Purification
4.5. Determination of Histamine and β-Hexosaminidase Release in RBL-2H3 Cells
4.6. OVA-Induced Model of Food Allergy
4.7. Measurement of Immunoglobulins, Anaphylactic Mediators, and Cytokines in the Mouse Serum
4.8. Morphology and Staining Analysis of the Intestinal Tissue
4.9. Splenic Lymphocyte Population Analysis by Flow Cytometry
4.10. Release of β-Hexosaminidase and Ca2+ Influx of RBL-2H3 Cells Induced by A23187
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Renz, H.; Allen, K.J.; Sicherer, S.H.; Sampson, H.A.; Lack, G.; Beyer, K.; Oettgen, H.C. Food allergy. Nat. Rev. Dis. Primers. 2018, 4, 17098. [Google Scholar] [CrossRef] [PubMed]
- Sicherer, S.H.; Sampson, H.A. Food allergy: Epidemiology, pathogenesis, diagnosis, and treatment. J. Allergy Clin. Immunol. 2014, 133, 291–307. [Google Scholar] [CrossRef] [PubMed]
- Begin, P.; Filion, C.; Graham, F.; Lacombe-Barrios, J.; Paradis, J.; Paradis, L.; Des Roches, A. Consultation with registered dietitian to prevent accidental reactions to food: Insight from an egg allergy influenza vaccination cohort. Eur. J. Clin. Nutr. 2017, 71, 287–289. [Google Scholar] [CrossRef] [PubMed]
- Dhanapala, P.; De Silva, C.; Doran, T.; Suphioglu, C. Cracking the egg: An insight into egg hypersensitivity. Mol. Immunol. 2015, 66, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.T.; Hu, Z.Y.; Cheng, Y.W.; Xu, H.X.; Velickovic, T.C.; He, K.; Sun, F.; He, Z.D.; Liu, Z.G.; Wu, X.L. Changes in Allergenicity of Ovalbumin in Vitro and in Vivo on Conjugation with Quercetin. J. Agr. Food Chem. 2020, 68, 4027–4035. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Courtade, L.; Han, S.; Lee, S.; Mian, F.M.; Buck, R.; Forsythe, P. Attenuation of food allergy symptoms following treatment with human milk oligosaccharides in a mouse model. Allergy 2015, 70, 1091–1102. [Google Scholar] [CrossRef]
- Galli, S.J.; Tsai, M.; Piliponsky, A.M. The development of allergic inflammation. Nature 2008, 454, 445–454. [Google Scholar] [CrossRef]
- Konig, J.; Wells, J.; Cani, P.D.; Garcia-Rodenas, C.L.; MacDonald, T.; Mercenier, A.; Whyte, J.; Troost, F.; Brummer, R.J. Human Intestinal Barrier Function in Health and Disease. Clin. Transl. Gastroenterol. 2016, 7, e196. [Google Scholar] [CrossRef]
- Groschwitz, K.R.; Hogan, S.P. Intestinal barrier function: Molecular regulation and disease pathogenesis. J. Allergy Clin. Immu. 2009, 124, 3–20. [Google Scholar] [CrossRef]
- Takhar, P.; Smurthwaite, L.; Coker, H.A.; Fear, D.J.; Banfield, G.K.; Carr, V.A.; Durham, S.R.; Gould, H.J. Allergen drives class switching to IgE in the nasal mucosa in allergic rhinitis. J. Immunol. 2005, 174, 5024–5032. [Google Scholar] [CrossRef]
- Rivas, M.N.; Chatila, T.A. Regulatory T cells in allergic diseases. J. Allergy Clin. Immu. 2016, 138, 639–652. [Google Scholar] [CrossRef]
- Tordesillas, L.; Mondoulet, L.; Blazquez, A.B.; Benhamou, P.H.; Sampson, H.A.; Berin, M.C. Epicutaneous immunotherapy induces gastrointestinal LAP(+) regulatory T cells and prevents food-induced anaphylaxis. J. Allergy Clin. Immu. 2017, 139, 189. [Google Scholar] [CrossRef] [PubMed]
- Harvima, I.T.; Levi-Schaffer, F.; Draber, P.; Friedman, S.; Polakovicova, I.; Gibbs, B.F.; Blank, U.; Nilsson, G.; Maurer, M. Molecular targets on mast cells and basophils for novel therapies. J. Allergy Clin. Immunol. 2014, 134, 530–544. [Google Scholar] [CrossRef] [PubMed]
- Gilfillan, A.M.; Beaven, M.A. Regulation of mast cell responses in health and disease. Crit. Rev. Immunol. 2011, 31, 475–529. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Park, S.B.; Kang, S.M.; Jeon, H.; Lim, J.P.; Kwon, T.K.; Park, W.H.; Kim, H.M.; Shin, T.Y. Anti-allergic effects of Teucrium japonicum on mast cell-mediated allergy model. Food Chem. Toxicol. 2009, 47, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, N.; Yamada, K.; Shoji, K.; Mori, M.; Sugano, M. Effect of tea polyphenols on histamine release from rat basophilic leukemia (RBL-2H3) cells: The structure-inhibitory activity relationship. Allergy 1997, 52, 58–64. [Google Scholar] [CrossRef]
- Izawa, K.; Yamanishi, Y.; Maehara, A.; Takahashi, M.; Isobe, M.; Ito, S.; Kaitani, A.; Matsukawa, T.; Matsuoka, T.; Nakahara, F.; et al. The receptor LMIR3 negatively regulates mast cell activation and allergic responses by binding to extracellular ceramide. Immunity 2012, 37, 827–839. [Google Scholar] [CrossRef]
- Nishida, K.; Yamasaki, S.; Ito, Y.; Kabu, K.; Hattori, K.; Tezuka, T.; Nishizumi, H.; Kitamura, D.; Goitsuka, R.; Geha, R.S.; et al. FcεRI-mediated mast cell degranulation requires calcium-independent microtubule-dependent translocation of granules to the plasma membrane. J. Cell Biol. 2005, 170, 115–126. [Google Scholar] [CrossRef]
- Han, S.; Sun, L.; He, F.; Che, H. Anti-allergic activity of glycyrrhizic acid on IgE-mediated allergic reaction by regulation of allergy-related immune cells. Sci. Rep. 2017, 7, 7222. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2012, 29, 144–222. [Google Scholar] [CrossRef]
- Imhoff, J.F. Natural Products from Marine Fungi--Still an Underrepresented Resource. Mar. Drugs 2016, 14, 19. [Google Scholar] [CrossRef] [PubMed]
- Nicoletti, R.; Trincone, A. Bioactive Compounds Produced by Strains of Penicillium and Talaromyces of Marine Origin. Mar. Drugs 2016, 14, 37. [Google Scholar] [CrossRef]
- Zhang, P.; Wei, Q.; Yuan, X.; Xu, K. Newly reported alkaloids produced by marine-derived Penicillium species (covering 2014-2018). Bioorg. Chem. 2020, 99, 103840. [Google Scholar] [CrossRef] [PubMed]
- Paget, G.E.; Walpole, A.L. Some cytological effects of griseofulvin. Nature 1958, 182, 1320–1321. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Leoni, M.E.; Cercenado, E.; Rodeno, P.; Bernaldo de Quiros, J.C.; Martinez-Hernandez, D.; Bouza, E. Susceptibility of Streptococcus pneumoniae to penicillin: A prospective microbiological and clinical study. Clin. Infect. Dis. 1992, 14, 427–435. [Google Scholar] [CrossRef]
- Youssef, D.T.A.; Alahdal, A.M. Cytotoxic and Antimicrobial Compounds from the Marine-Derived Fungus, Penicillium Species. Molecules 2018, 23, 394. [Google Scholar] [CrossRef]
- Liu, Q.M.; Xie, C.L.; Gao, Y.Y.; Liu, B.; Lin, W.X.; Liu, H.; Cao, M.J.; Su, W.J.; Yang, X.W.; Liu, G.M. Deep-Sea-Derived Butyrolactone I Suppresses Ovalbumin-Induced Anaphylaxis by Regulating Mast Cell Function in a Murine Model. J. Agric. Food Chem. 2018, 66, 5581–5592. [Google Scholar] [CrossRef]
- Gao, Y.Y.; Liu, Q.M.; Liu, B.; Xie, C.L.; Cao, M.J.; Yang, X.W.; Liu, G.M. Inhibitory Activities of Compounds from the Marine Actinomycete Williamsia sp MCCC 1A11233 Variant on IgE-Mediated Mast Cells and Passive Cutaneous Anaphylaxis. J. Agr. Food. Chem. 2017, 65, 10749–10756. [Google Scholar] [CrossRef]
- Xing, C.P.; Xie, C.L.; Xia, J.M.; Liu, Q.M.; Lin, W.X.; Ye, D.Z.; Liu, G.M.; Yang, X.W. Penigrisacids A-D, Four New Sesquiterpenes from the Deep-Sea-Derived Penicillium griseofulvum. Mar. Drugs 2019, 17, 507. [Google Scholar] [CrossRef]
- Liang, P.; Zhang, Y.Y.; Yang, P.; Grond, S.; Zhang, Y.; Qian, Z.J. Viridicatol and viridicatin isolated from a shark-gill-derived fungus Penicillium polonicum AP2T1 as MMP-2 and MMP-9 inhibitors in HT1080 cells by MAPKs signaling pathway and docking studies. Med. Chem. Res. 2019, 28, 1039–1048. [Google Scholar] [CrossRef]
- Yamaki, K.; Yoshino, S. Tyrosine kinase inhibitor sunitinib relieves systemic and oral antigen-induced anaphylaxes in mice. Allergy 2012, 67, 114–122. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, Y.; Luo, H.; Sun, L.; Xu, M.; Yu, J.; Zhou, Q.; Meng, G.; Yang, S. Recent Update on the Pharmacological Effects and Mechanisms of Dihydromyricetin. Front. Pharmacol. 2018, 9, 1204. [Google Scholar] [CrossRef]
- Kamei, R.; Fujimura, T.; Matsuda, M.; Kakihara, K.; Hirakawa, N.; Baba, K.; Ono, K.; Arakawa, K.; Kawamoto, S. A flavanone derivative from the Asian medicinal herb (Perilla frutescens) potently suppresses IgE-mediated immediate hypersensitivity reactions. Biochem. Biophys. Res. Commun. 2017, 483, 674–679. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, M.; Lopes, G.; Valentao, P.; Ferreres, F.; Gil-Izquierdo, A.; Pereira, D.M.; Andrade, P.B. Edible seaweeds’ phlorotannins in allergy: A natural multi-target approach. Food Chem. 2018, 265, 233–241. [Google Scholar] [CrossRef]
- Lee, D.; Kim, H.S.; Shin, E.; Do, S.G.; Lee, C.K.; Kim, Y.M.; Lee, M.B.; Min, K.Y.; Koo, J.; Kim, S.J.; et al. Polysaccharide isolated from Aloe vera gel suppresses ovalbumin-induced food allergy through inhibition of Th2 immunity in mice. Biomed. Pharmacother. 2018, 101, 201–210. [Google Scholar] [CrossRef]
- Veldhoen, M.; Hocking, R.J.; Atkins, C.J.; Locksley, R.M.; Stockinger, B. TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 2006, 24, 179–189. [Google Scholar] [CrossRef]
- Forbes, E.E.; Groschwitz, K.; Abonia, J.P.; Brandt, E.B.; Cohen, E.; Blanchard, C.; Ahrens, R.; Seidu, L.; McKenzie, A.; Strait, R.; et al. IL-9- and mast cell-mediated intestinal permeability predisposes to oral antigen hypersensitivity. J. Exp. Med. 2008, 205, 897–913. [Google Scholar] [CrossRef]
- Bischoff, S.C. Physiological and pathophysiological functions of intestinal mast cells. Semin. Immunopathol. 2009, 31, 185–205. [Google Scholar] [CrossRef] [PubMed]
- Jacob, C.; Yang, P.C.; Darmoul, D.; Amadesi, S.; Saito, T.; Cottrell, G.S.; Coelho, A.M.; Singh, P.; Grady, E.F.; Perdue, M.; et al. Mast cell tryptase controls paracellular permeability of the intestine. Role of protease-activated receptor 2 and beta-arrestins. J. Biol. Chem. 2005, 280, 31936–31948. [Google Scholar] [CrossRef]
- Nguyet, T.M.N.; Lomunova, M.; Le, B.V.; Lee, J.S.; Park, S.K.; Kang, J.S.; Kim, Y.H.; Hwang, I. The mast cell stabilizing activity of Chaga mushroom critical for its therapeutic effect on food allergy is derived from inotodiol. Int. Immunopharmacol. 2018, 54, 286–295. [Google Scholar] [CrossRef]
- Trebak, M.; Kinet, J.P. Calcium signalling in T cells. Nat. Rev. Immunol. 2019, 19, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.S. Calcium signaling mechanisms in T lymphocytes. Annu. Rev. Immunol. 2001, 19, 497–521. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.; Hogan, P.G. Calcium signaling in cells of the immune and hematopoietic systems. Immunol. Rev. 2009, 231, 5–9. [Google Scholar] [CrossRef]
- Di Capite, J.; Parekh, A.B. CRAC channels and Ca2+ signaling in mast cells. Immunol. Rev. 2009, 231, 45–58. [Google Scholar] [CrossRef]
- Zhang, Y.F.; Liu, Q.M.; Liu, B.; Shu, Z.D.; Han, J.; Liu, H.; Cao, M.J.; Yang, X.W.; Gu, W.; Liu, G.M. Dihydromyricetin inhibited ovalbumin-induced mice allergic responses by suppressing the activation of mast cells. Food. Funct. 2019, 10, 7131–7141. [Google Scholar] [CrossRef] [PubMed]
- Costa, H.F.; Leite, F.C.; Alves, A.F.; Barbosa, J.M.; dos Santos, C.R.B.; Piuvezam, M.R. Managing murine food allergy with Cissampelos sympodialis Eichl (Menispermaceae) and its alkaloids. Int. Immunopharmacol. 2013, 17, 300–308. [Google Scholar] [CrossRef][Green Version]
- Xin, M.L.; Ma, Y.J.; Xu, K.; Chen, M.C. Dihydromyricetin An effective non-hindered phenol antioxidant for linear low-density polyethylene stabilisation. J. Therm. Anal. Calorim. 2013, 114, 1167–1175. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, Y.; Shu, Z.; Liu, M.; Zeng, R.; Wang, Y.; Liu, H.; Cao, M.; Su, W.; Liu, G. Sulfated oligosaccharide of Gracilaria lemaneiformis protect against food allergic response in mice by up-regulating immunosuppression. Carbohydr. Polym. 2020, 230, 115567. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shu, Z.; Liu, Q.; Xing, C.; Zhang, Y.; Zhou, Y.; Zhang, J.; Liu, H.; Cao, M.; Yang, X.; Liu, G. Viridicatol Isolated from Deep-Sea Penicillium Griseofulvum Alleviates Anaphylaxis and Repairs the Intestinal Barrier in Mice by Suppressing Mast Cell Activation. Mar. Drugs 2020, 18, 517. https://doi.org/10.3390/md18100517
Shu Z, Liu Q, Xing C, Zhang Y, Zhou Y, Zhang J, Liu H, Cao M, Yang X, Liu G. Viridicatol Isolated from Deep-Sea Penicillium Griseofulvum Alleviates Anaphylaxis and Repairs the Intestinal Barrier in Mice by Suppressing Mast Cell Activation. Marine Drugs. 2020; 18(10):517. https://doi.org/10.3390/md18100517
Chicago/Turabian StyleShu, Zhendan, Qingmei Liu, Cuiping Xing, Yafen Zhang, Yu Zhou, Jun Zhang, Hong Liu, Minjie Cao, Xianwen Yang, and Guangming Liu. 2020. "Viridicatol Isolated from Deep-Sea Penicillium Griseofulvum Alleviates Anaphylaxis and Repairs the Intestinal Barrier in Mice by Suppressing Mast Cell Activation" Marine Drugs 18, no. 10: 517. https://doi.org/10.3390/md18100517
APA StyleShu, Z., Liu, Q., Xing, C., Zhang, Y., Zhou, Y., Zhang, J., Liu, H., Cao, M., Yang, X., & Liu, G. (2020). Viridicatol Isolated from Deep-Sea Penicillium Griseofulvum Alleviates Anaphylaxis and Repairs the Intestinal Barrier in Mice by Suppressing Mast Cell Activation. Marine Drugs, 18(10), 517. https://doi.org/10.3390/md18100517