Distribution, Contents, and Types of Mycosporine-Like Amino Acids (MAAs) in Marine Macroalgae and a Database for MAAs Based on These Characteristics
Abstract
1. Introduction
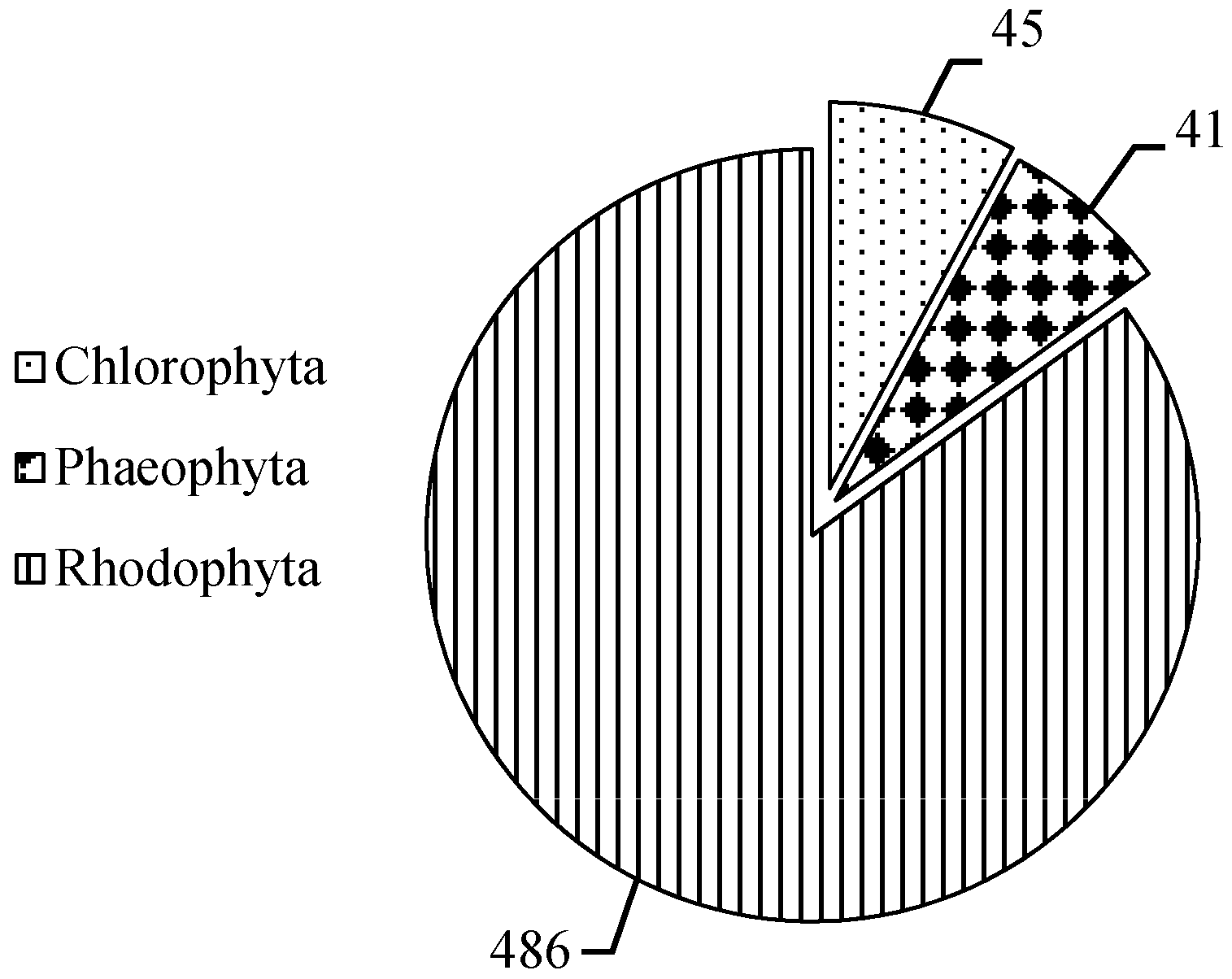
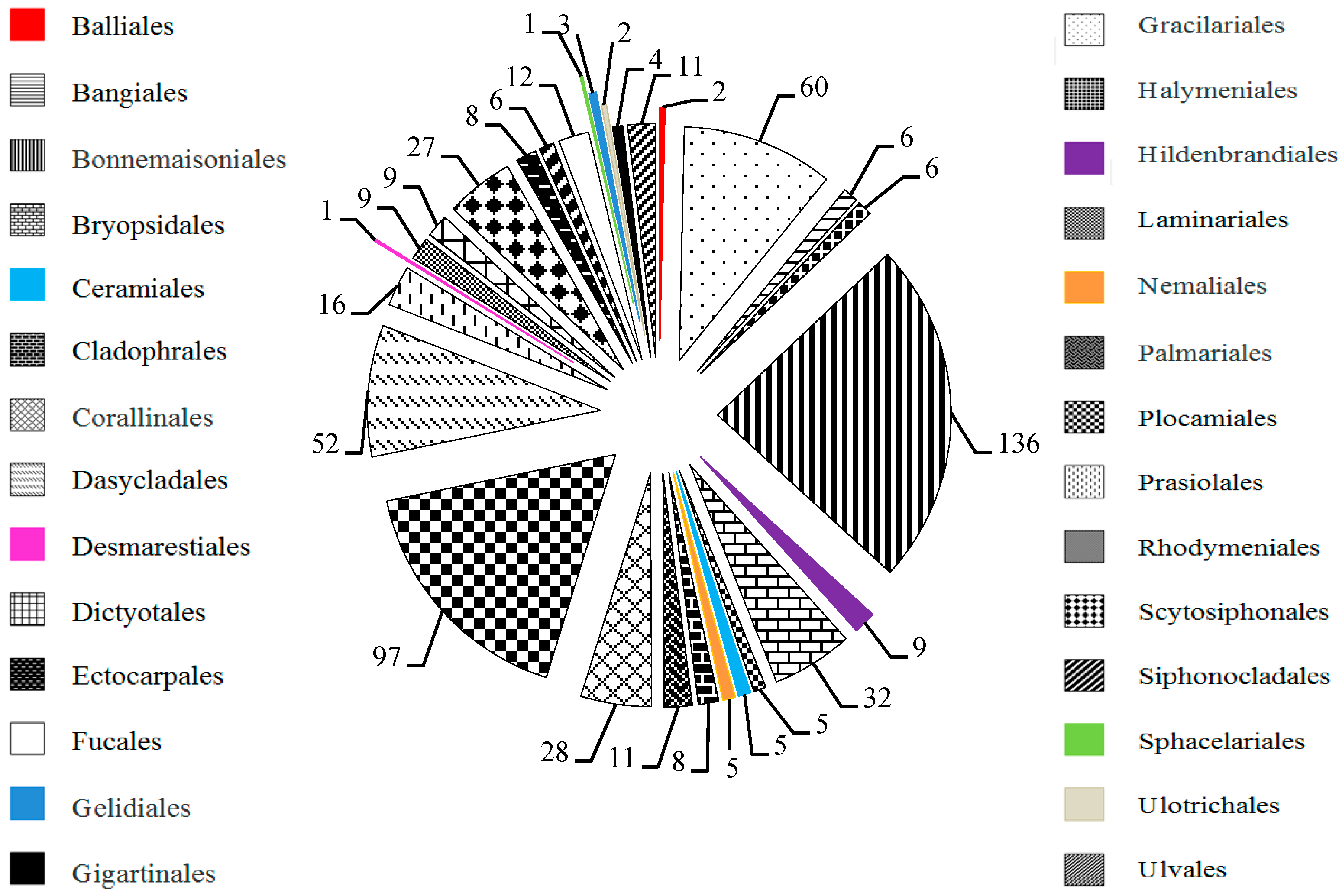
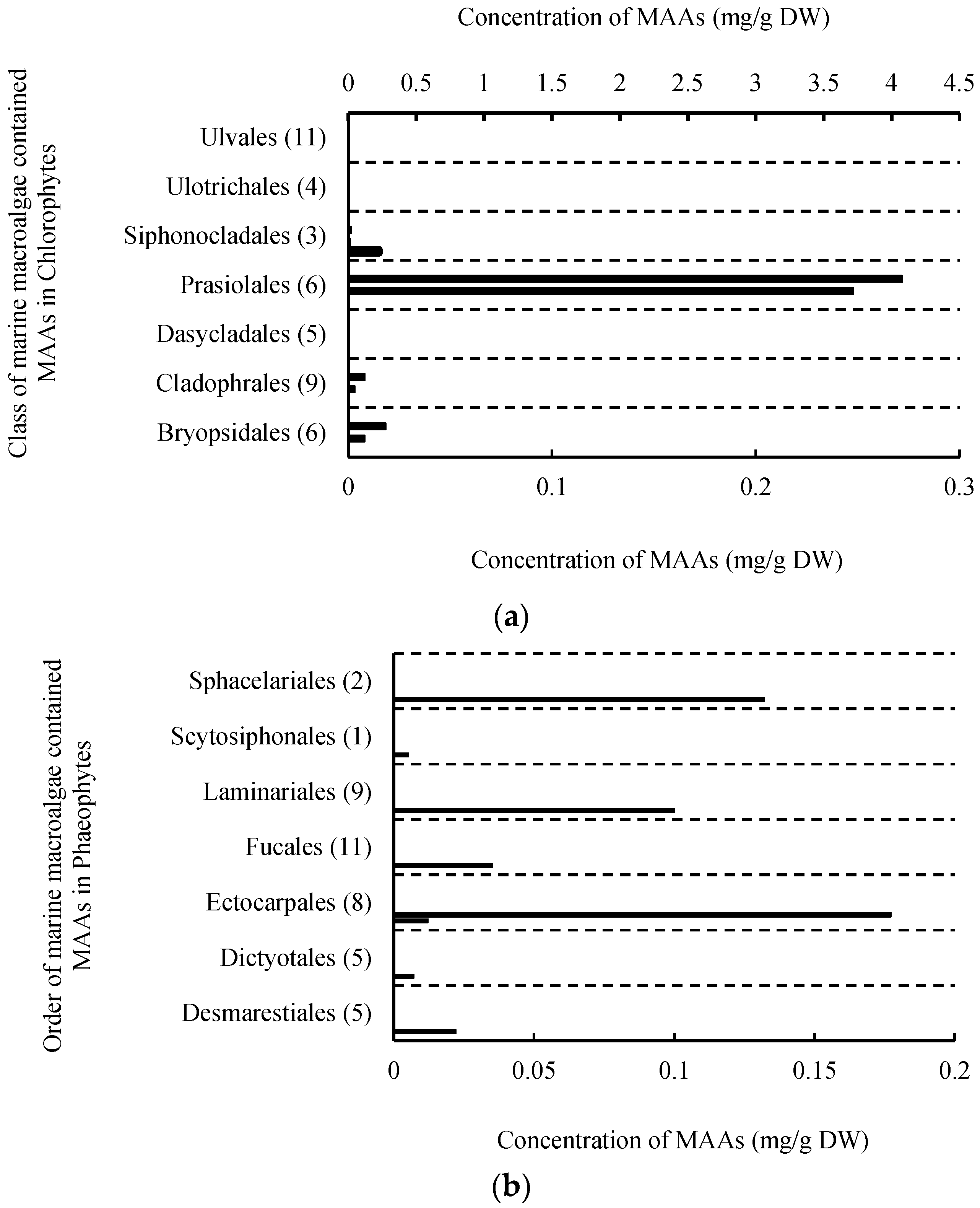
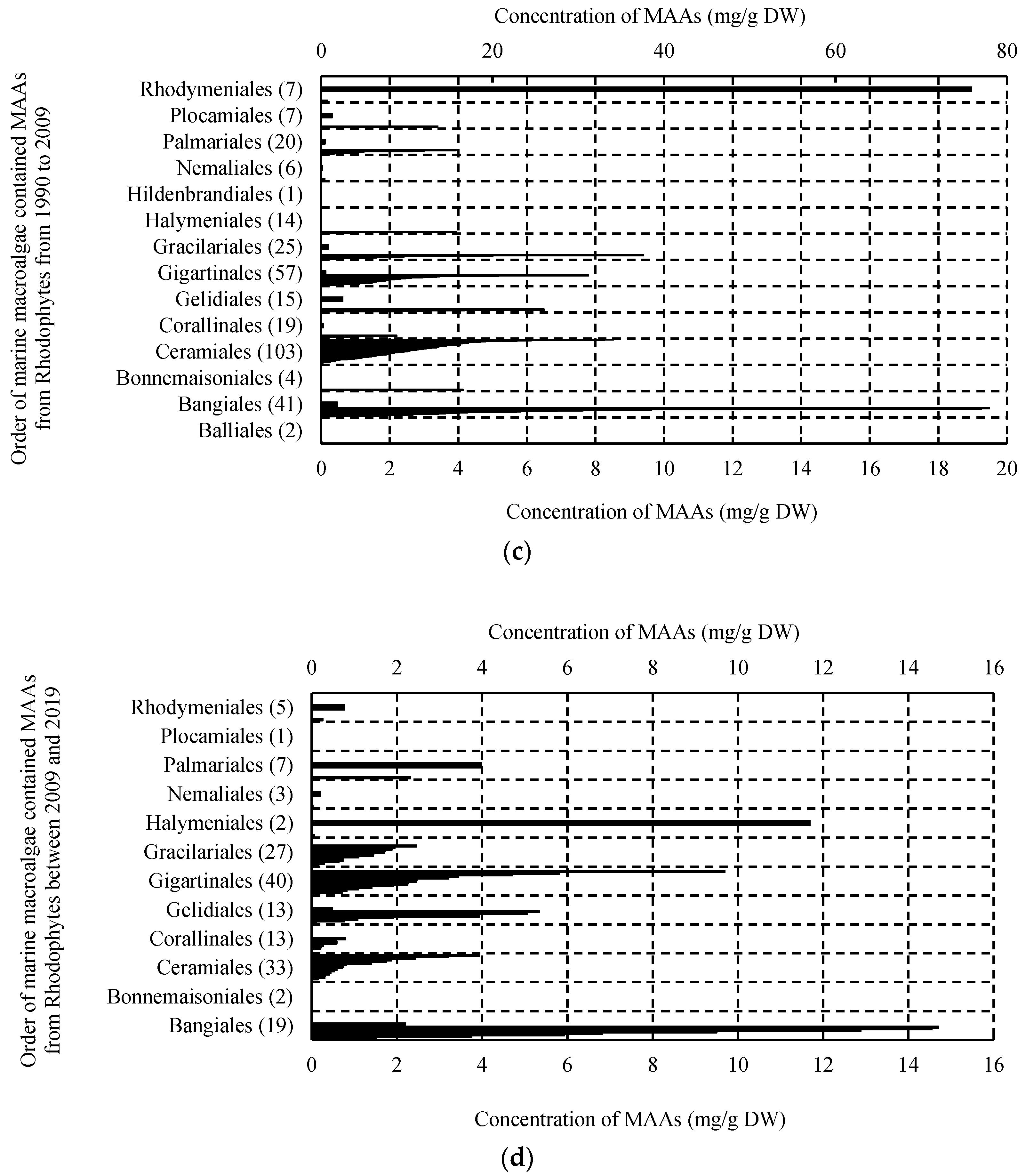
2. Results
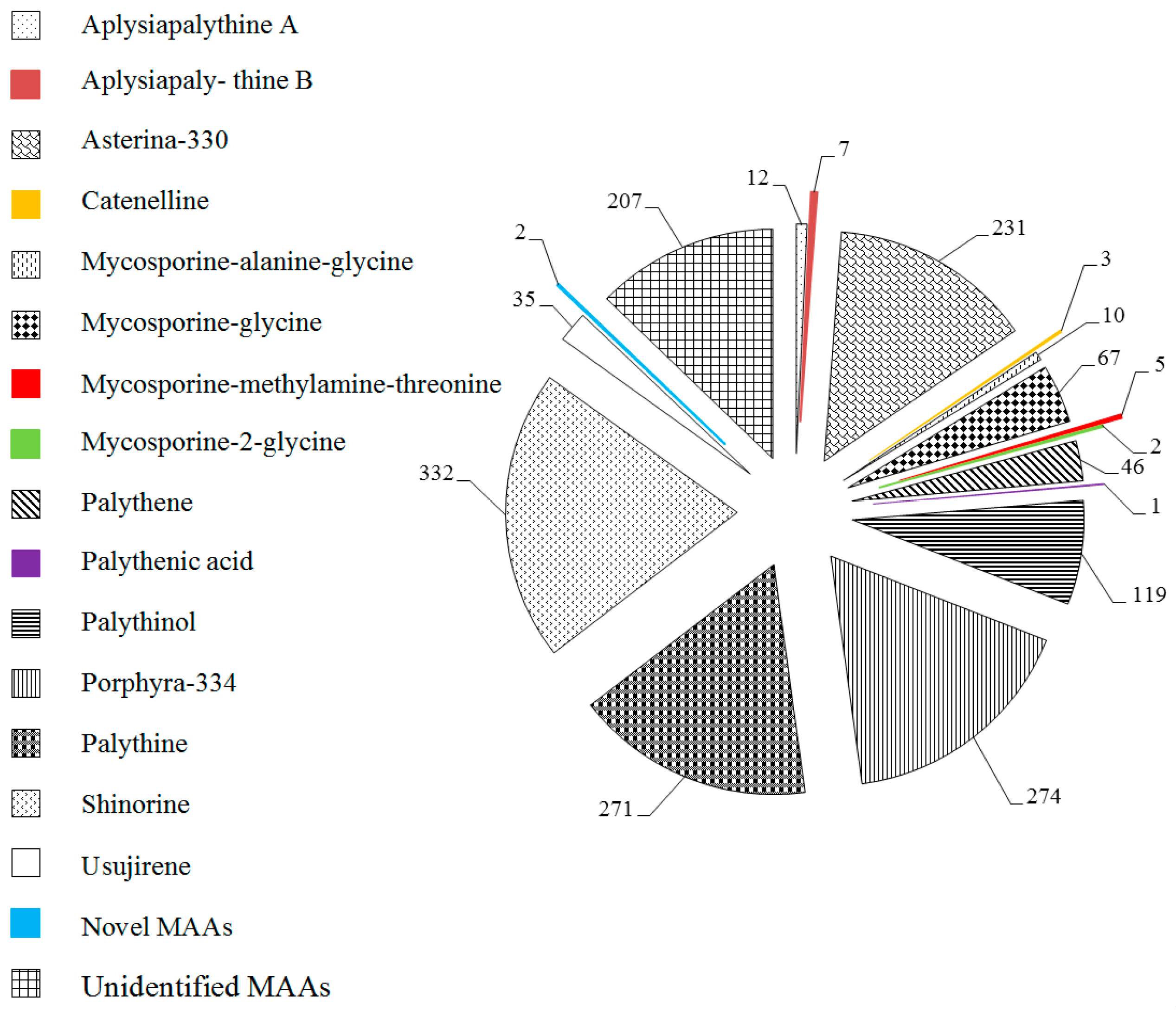
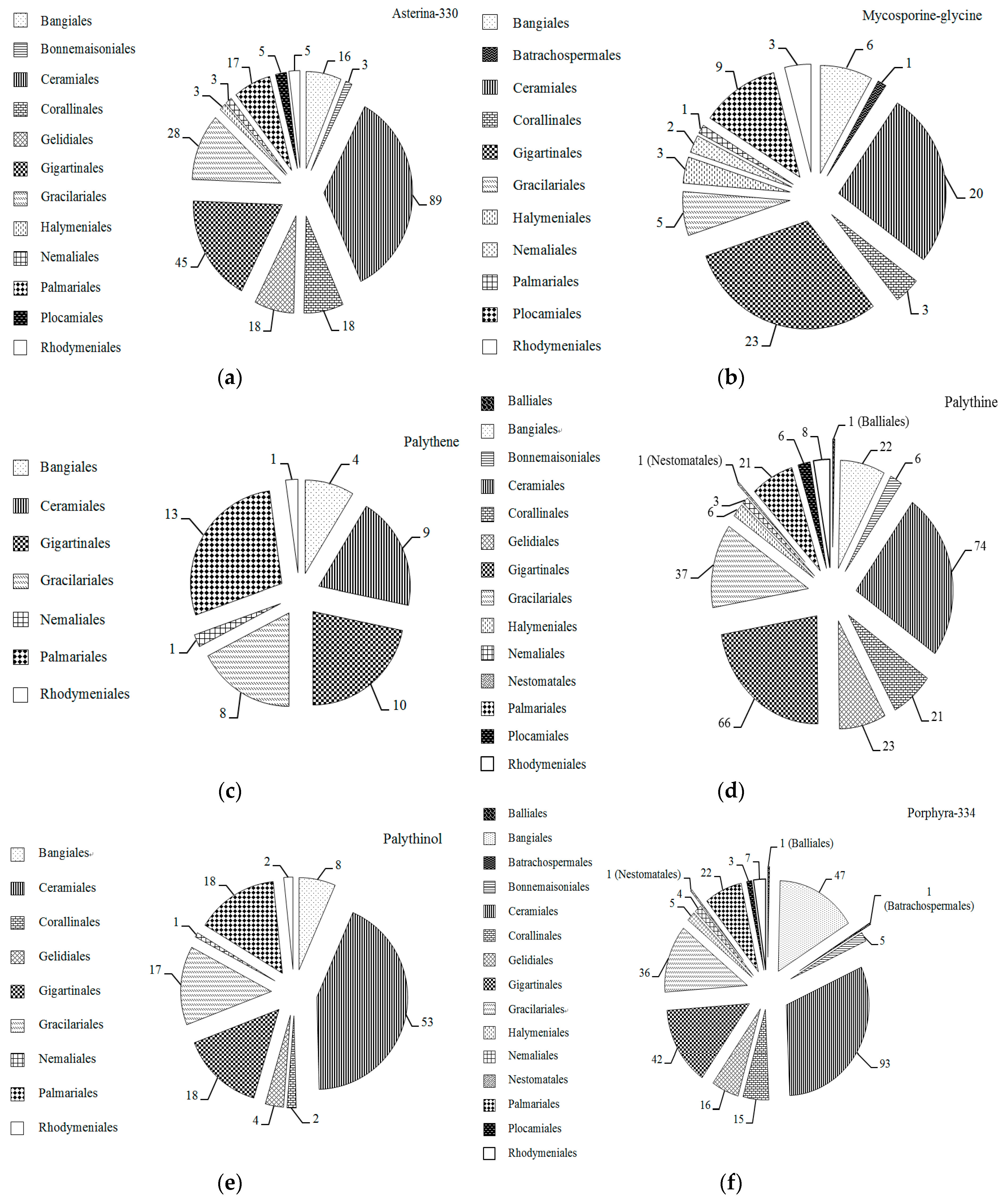
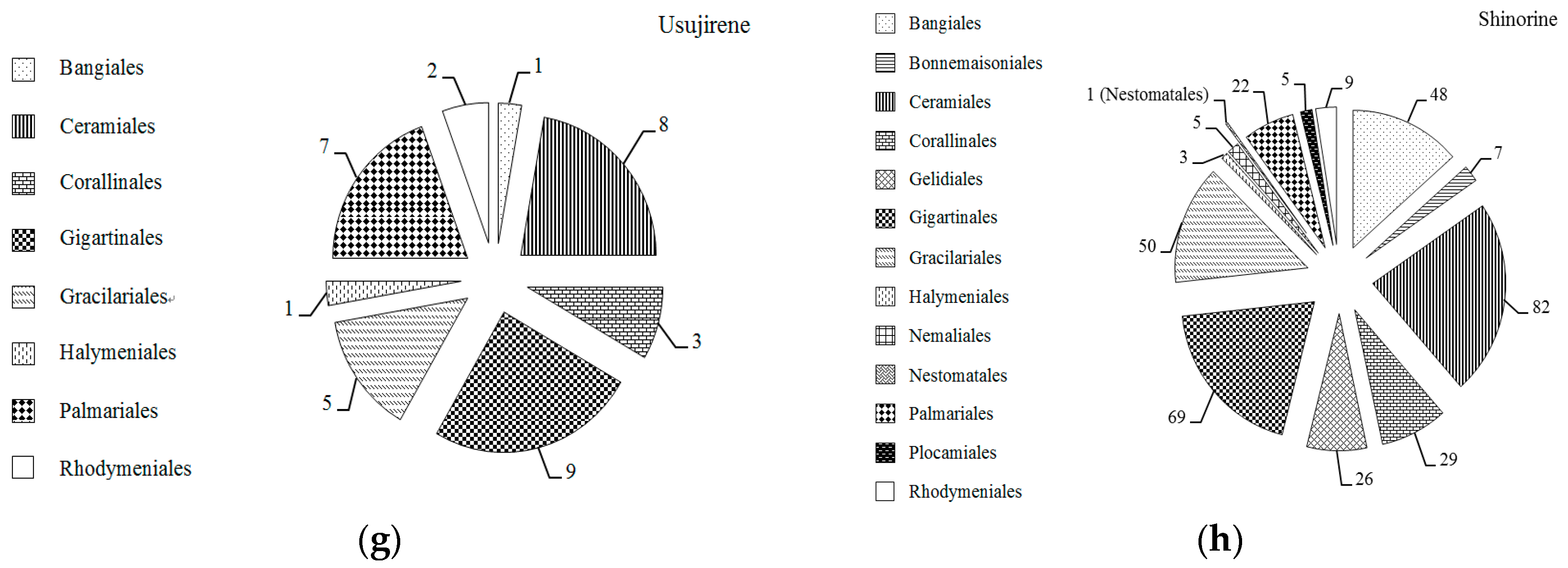
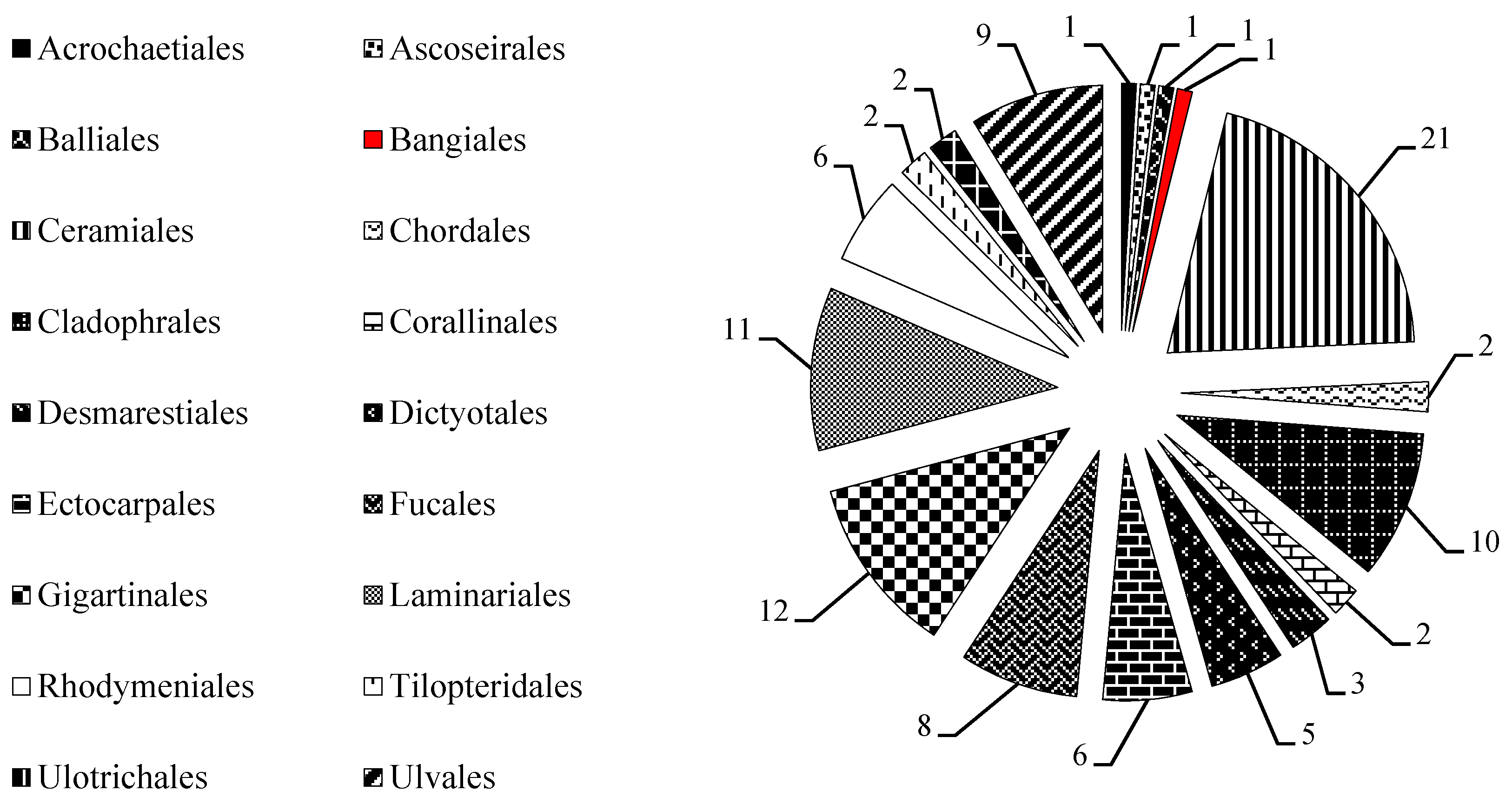
2.1. MAAs Distribution, Contents and Types in Marine Macroalgae
2.2. An Open Database for MAAs in Marine Macroalgae
2.3. Marine Macroalgae with No Detectable MAAs Concentrations
2.4. Trends in Research on MAAs in Marine Macroalgae
3. Materials and Methods
3.1. Methods
3.2. Total MAAs Contents and Specific MAA Concentration
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gröniger, A.; Hallier, C.; Häder, D.P. Influence of UV radiation and visible light on Porphyra umbilicalis: Photoinhibition and MAA concentration. J. Appl. Phycol. 1999, 11, 437–445. [Google Scholar] [CrossRef]
- Gröniger, A.; Sinha, R.P.; Klish, M.; Häder, D.P. Photoprotective compounds in cyanobacteria, phytoplankton and macroalgae-a database. J. Photochem. Photobiol. B Biol. 2000, 58, 115–122. [Google Scholar] [CrossRef]
- Hoyer, K.; Karsten, U.; Sawall, T.; Wiencke, C. Photoprotective substances in Antarctic macroalgae and their variation with respect to depth distribution, different tissues and developmental stages. Mar. Ecol. Prog. Ser. 2000, 211, 117–129. [Google Scholar] [CrossRef]
- Karsten, U.; Sawall, T.; Hanelt, D.; Bischof, K.; Figueroa, F.L.; Flores-Moya, A.; Wiencke, C. An inventory of UV-absorbing mycosporine-like amino acids in macroalgae from polar to warm-temperate regions. Bot. Mar. 1998, 41, 443–453. [Google Scholar] [CrossRef]
- Karsten, U.; Sawall, T.; Wiencke, C. A survey of the distribution of UV-absorbing substances in tropical macroalgae. Phycol. Res. 1998, 46, 271–279. [Google Scholar]
- Korbee Peinado, N.; Abdala Díaz, R.T.; Figueroa, F.L.; Helbling, E.W. Ammonium and UV radiation stimulate the accumulation of mycosporine-like amino acids in Porphyra columbina (Rhodophyta) from Patagonia, Argentina. J. Phycol. 2004, 40, 248–259. [Google Scholar] [CrossRef]
- Sinha, R.P.; Klisch, M.; Groniger, A.; Hader, D.P. Ultraviolet-absorbing/screening substances in cyanobacteria, phytoplankton and macroalgae. J. Photochem. Photobiol. B Biol. 1998, 47, 83–94. [Google Scholar] [CrossRef]
- Balskus, E.P.; Walsh, C.T. The genetic and molecular basis for sunscreen biosynthesis in cyanobacteria. Science 2010, 329, 1653–1656. [Google Scholar] [CrossRef]
- Carreto, J.I.; Carignan, M.O.; Daleo, G.; De Marco, S.G. Occurrence of mycosporine-like amino acids in the red-tide dinoflagellate Alexandrium excavatum: UV photoprotective compounds? J. Plankton Res. 1990, 12, 909–921. [Google Scholar] [CrossRef]
- Hannach, G.; Sigleo, A.C. Photoinduction of UV-absorbing compounds in six species of marine phytoplankton. Mar. Ecol. Prog. Ser. 1998, 174, 207–222. [Google Scholar] [CrossRef]
- Vernet, M.; Neori, A.; Haxo, F.T. Spectral properties and photosynthetic action in red-tide populations of Prorocentrum micans and Gonyaulax polyedra. Mar. Biol. 1989, 103, 365–371. [Google Scholar] [CrossRef]
- Yentsch, C.S.; Yentsch, C.M. The Attenuation of light by marine phytoplankton with specific reference to the absorption of near-UV radiation. In The Role of Solar Ultraviolet Radiation in Marine Ecosystems; Calkins, J., Ed.; Plenum Press: New York, NY, USA, 1982; Volume 1, pp. 691–700. [Google Scholar]
- Garcia-Pichel, F.; Castenholz, R.W. Occurrence of UV-absorbing, mycosporine-like compounds among cyanobacterial isolates and an estimate of their screening capacity. Appl. Environ. Microbiol. 1993, 59, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Karsten, U.; Garcia-Pichel, F. Carotenoids and mycosporine-like amino acid compounds in members of the genus Microcoleus (Cyanobacteria): A chemosystematic study. Syst. Appl. Microbiol. 1996, 19, 285–294. [Google Scholar] [CrossRef]
- Queseda, A.; Vincent, W.F. Strategies of adaptation by Antarctic cyanobacteria to ultraviolet radiation. Eur. J. Phycol. 1997, 32, 335–342. [Google Scholar] [CrossRef]
- Arai, T.; Nishijima, M.; Adachi, K.; Sano, H. Isolation and Structure of a UV Absorbing Substance from the Marine Bacterium Micrococcus sp. AK-334; Marine Biotechnology Institute: Tokyo, Japan, 1992; pp. 88–94. [Google Scholar]
- Favre-Bonvin, J.; Aprin, N.; Brevard, C. Structure of mycosporine. Chemistry 1976, 54, 1105–1113. [Google Scholar]
- Banaszak, A.T.; Trench, R.K. Effects of ultraviolet (UV) radiation on marine microalgal–invertebrate symbioses. Ⅱ. The synthesis of mycosporine-like amino acids in response to exposure to UV in Anthopleura elegantissima and Cassiopeia xamachana. J. Exp. Mar. Biol. Ecol. 1995, 194, 233–250. [Google Scholar] [CrossRef]
- Shick, J.M.; Dunlap, W.C.; Chalker, B.E.; Banaszak, A.T.; Rosenzweig, T.K. Survey of ultraviolet radiation-absorbing mycosporine-like amino acids in organs of coral reef holothuroids. Mar. Ecol. Prog. Ser. 1992, 90, 139–148. [Google Scholar] [CrossRef]
- Stochaj, W.R.; Dunlap, W.C.; Shick, J.M. Two new UV-absorbing mycosporine-like amino acids from the sea anem-one Anthopleura elegantissima and the effects of zooxanthellae and spectral irradiance on chemical composition and content. Mar. Biol. 1994, 118, 149–156. [Google Scholar] [CrossRef]
- Whitehead, K.; Karentz, D.; Hedges, J.I. Mycosporine-like amino acids (MAAs) in phytoplankton, a herbivorous pteropod (Limacina helicina), and its pteropod predator (Clione antarctica) in McMurdo Bay, Antarctica. Mar. Biol. 2001, 139, 1013–1019. [Google Scholar]
- Cockell, C.S.; Knowland, J. Ultraviolet radiation screening compounds. Biol. Rev. 1999, 74, 311–345. [Google Scholar] [CrossRef]
- Bandarangyake, W.M. Mycosporines: Are they nature’s sunscreens? Nat. Prod. Rep. 1998, 15, 159–172. [Google Scholar] [CrossRef]
- Gao, X.; He, Q.M.; Zhang, Z.H.; Xu, J.C.; Zhang, L. Study on antioxidant activity of mycosporine-like amino acids in two seaweeds. Mar. Environ. Sci. 2010, 29, 76–79. (In Chinese) [Google Scholar]
- Xu, Z.H. The Preparation Techniques of Mycosporine-Like Amino Acid from Porphyra Yezoensis. Master’s Thesis, Ocean University of China, Qiangdao, China, 2010. [Google Scholar]
- Nakayama, R.; Tamura, Y.; Kikuzaki, H.; Nakatani, N. Antioxidant effect of the constituent of susabinori (Porphyra yezoensis). J. Am. Oil Chem. Soc. 1999, 76, 649–653. [Google Scholar] [CrossRef]
- Maekawa, Y. Application of UV absorbance of algae. Ocean 1995, 27, 157–160. (In Japanese) [Google Scholar]
- Oren, A.; Gunde-Cimerman, N. Mycosporines and mycosproine-like amino acids: UV protectants or multipurpose secondary metabolites? FEMS Microbiol. Lett. 2007, 269, 1–10. [Google Scholar] [CrossRef]
- Dunlap, W.C.; Yamamoto, Y. Small molecule antioxidants in marine organisms: Antioxidant activity of mycosporine-glycine. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1995, 112, 105–114. [Google Scholar] [CrossRef]
- Adams, N.L.; Shick, J.M. Mycosporine-like amino acids provide protection against ultraviolet radiation in eggs of the green sea urchin Strongylocentrotus droebachiensis. Photochem. Photobiol. 1996, 64, 149–158. [Google Scholar] [CrossRef]
- Oren, A. Mycosporine-like amino acids as osmotic solutes in a community of halophilic cyanobactera. Geomicrobiol. J. 1997, 14, 231–240. [Google Scholar] [CrossRef]
- Karentz, D.; McEuen, F.S.; Land, M.C.; Dunlap, W.C. Survey of mycosporine-like amino acid compounds in Antarctic marine organisms: Potential protection from ultraviolet exposure. Mar. Biol. 1991, 108, 157–166. [Google Scholar] [CrossRef]
- Maegawa, M.; Kunieda, M.; Kida, W. The influence of ultraviolet radiation on the photosynthetic activity of several algae from different depths. Jpn. J. Phycol. 1993, 41, 207–214. [Google Scholar]
- McClintock, J.B.; Karentz, D. Mycosporine-like amino acids in 38 species of subtidal marine organisms from McMurdo Sound, Antarctica. Antarct. Sci. 1997, 9, 392–398. [Google Scholar] [CrossRef]
- Karsten, U.; Escoubeyrou, K.; Charles, F. The effect of re-dissolution solvents and HPLC columns on the analysis of mycosporine-like amino acids in the eulittoral macroalgae Prasiola crispa and Porphyra umbilicalis. Helgol. Mar. Res. 2009, 63, 231–238. [Google Scholar] [CrossRef]
- Zacher, K.; Roleda, M.Y.; Wulff, A.; Hanelt, D.; Wiencke, C. Responses of Antarctic Iridaea cordata (Rhodophyta) tetraspores exposed to ultraviolet radiation. Phycol. Res. 2009, 57, 186–193. [Google Scholar] [CrossRef]
- Lamare, M.D.; Lesser, M.P.; Barker, M.F.; Barry, T.M.; Schimanski, K.B. Variation in sunscreen compounds (mycosporine-like amino acids) for marine species along a gradient of ultraviolet radiation transmission within doubtful sound, New Zealand. N. Z. J. Mar. Freshw. Res. 2004, 38, 775–793. [Google Scholar] [CrossRef][Green Version]
- Apprill, A.M.; Lesser, M.P. Effects of ultraviolet radiation on Laminaria saccharina in relation to depth and tidal height in the Gulf of Maine. Mar. Ecol. Prog. Ser. 2003, 256, 75–85. [Google Scholar] [CrossRef][Green Version]
- De la Coba, F.; Aguilera, J.; Figueroa, F.L.; Gálvez, M.V.; Herrera, E. Antioxidant activity of mycosporine-like amino acids isolated from three red macroalgae and one marine lichen. J. Appl. Phycol. 2009, 21, 161–169. [Google Scholar] [CrossRef]
- Karsten, U.; Sawall, T.; West, J.; Wiencke, C. Ultraviolet sunscreen compounds in epiphytic red algae from mangroves. Hydrobiologia 2000, 432, 159–171. [Google Scholar] [CrossRef]
- Karsten, U.; Franklin, L.A.; Lüning, K.; Wiencke, C. Natural ultraviolet radiation and photosynthetically active radiation induce formation of mycosporine-like amino acids in the marine macroalga Chondrus crispus (Rhodophyta). Planta 1998, 205, 257–262. [Google Scholar] [CrossRef]
- Huovinen, P.; Gómez, I.; Figueroa, F.L.; Ulloa, N.; Morales, V.; Lovengreen, C. Ultraviolet-absorbing mycosporine-like amino acids in red macroalgae from Chile. Bot. Mar. 2004, 47, 21–29. [Google Scholar] [CrossRef]
- Carreto, J.I.; Carignan, M.O.; Montoya, N.G. A high-resolution reverse-phase liquid chromatography method for the analysis of mycosporine-like amino acids (MAAs) in marine organisms. Mar. Biol. 2005, 146, 237–252. [Google Scholar] [CrossRef]
- Karsten, U.; Dummermuth, A.; Hoyer, K.; Wiencke, C. Interactive effects of ultraviolet radiation and salinity on the ecophysiology of two Arctic red algae from shallow waters. Polar Biol. 2003, 26, 249–258. [Google Scholar] [CrossRef]
- Karsten, U.; Bischof, K.; Hanelt, D.; Tüg, H.; Wiencke, C. The effect of ultraviolet radiation on photosynthesis and ultraviolet-absorbing substances in the endemic Arctic macroalga Devaleraea ramentacea (Rhodophyta). Physiol. Plant. 1999, 105, 58–66. [Google Scholar] [CrossRef]
- Karsten, U. Occurrence of photoprotective mycosporine-like amino acid compounds (MAAs) in marine red macroalgae from temperate Australian wates. Proc.-Linn. Soc. N. S. W. 2000, 122, 123–129. [Google Scholar]
- Chuang, L.F.; Chou, H.N.; Sung, P.J. Porphyra-334 isolated from the marine algae Bangia atropurpurea: Conformational performance for energy conversion. Mar. Drugs 2014, 12, 4732–4740. [Google Scholar] [CrossRef] [PubMed]
- Gravem, S.A.; Adams, N.L. Sex and microhabitat influence the uptake and allocation of mycosporine-like amino acids to tissues in the purple sea urchin, Strongylocentrotus purpuratus. Mar. Biol. 2012, 159, 2839–2852. [Google Scholar] [CrossRef]
- Hartmann, A.; Becker, K.; Karsten, U.; Remias, D.; Ganzera, M. Analysis of mycosporine-like amino acids in selected algae and cyanobacteria by hydrophilic interaction liquid chromatography and a novel MAAs from the red alga Catenella repens. Mar. Drugs 2015, 13, 6291–6305. [Google Scholar] [CrossRef] [PubMed]
- Véliz, K.; Chandía, N.; Karsten, U.; Lara, C.; Thiel, M. Geographic variation in biochemical and physiological traits of the red seaweeds Chondracanthus chamissoi and Gelidium lingulatum from the south east Pacific coast. J. Appl. Phycol. 2019, 31, 665–682. [Google Scholar] [CrossRef]
- Zhang, W. Study on Extraction, Antioxidation and Moisturizing Activities of MAAs from Gloiopeltis furcata. Master’s Thesis, Shanghai Ocean University, Shanghai, China, 2016. [Google Scholar]
- Yuan, Y.V.; Westcott, N.D.; Hu, C.; Kitt, D.D. Mycosporine-like amino acid composition of the edible red alga, Palmaria palmata (dulse) harvested from the west and east coasts of Grand Manan Island, New Brunswick. Food Chem. 2009, 112, 321–328. [Google Scholar] [CrossRef]
- Diehl, N.; Michalik, D.; Zuccarello, G.C.; Karsten, U. Stress metabolite pattern in the eulittoral red alga Pyropia plicata (Bangiales) in New Zealand-mycosporine-like amino acids and heterosides. J. Exp. Mar. Biol. Ecol. 2019, 510, 23–30. [Google Scholar] [CrossRef]
- Bedoux, G.; Hardouin, K.; Marty, C.; Taupin, L.; Vandanjon, L.; Bourgougnon, N. Chemical characterization and photoprotective activity measurement of extraxts from the red macroalgae Solieria chordalis. Bot. Mar. 2014, 57, 291–301. [Google Scholar] [CrossRef]
- Athukorala, Y.; Trang, S.; Kwok, C.; Yuan, Y.V. Antiproliferative and antioxidant activities and mycosporine-Like amino acid profiles of wild-harvested and cultivated edible Canadian marine red macroalgae. Molecules 2016, 21, 119. [Google Scholar] [CrossRef] [PubMed]
- Celis-Plá, P.S.M.; Martínez, B.; Quintano, E.; García-Sánchez, M.; Pedersen, A.; Navarro, N.P.; Copertino, M.S.; Mangaiyarkarasi, N.; Mariath, R.; Figueroa, F.L.; et al. Short-term ecophysiological and biochemical responses of Cystoseira tamariscifolia and Ellisolandia elongata to environmental changes. Aquat. Biol. 2014, 22, 227–243. [Google Scholar] [CrossRef]
- Guihéneuf, F.; Gietl, A.; Stengel, D.B. Temporal and spatial variability of mycosporine-like amino acids and pigments in three edible red seaweeds from western Ireland. J. Appl. Phycol. 2018, 30, 2573–2586. [Google Scholar] [CrossRef]
- Roleda, M.Y.; Zacher, K.; Wulff, A.; Hanelt, D.; Wiencke, C. Susceptibility of spores of different ploidy levels from Antarctic Gigartina skottsbergii (Gigartinales, Rhodophyta) to ultraviolet radiation. Phycologia 2008, 47, 361–370. [Google Scholar] [CrossRef]
- Sinha, R.P.; Singh, S.P.; Häder, D.P. Database on mycosporines and mycosporine-like amino acids (MAAs) in fungi, cyanobacteria, macroalgae, phytoplankton and animals. J. Photochem. Photobiol. B Biol. 2007, 89, 29–35. [Google Scholar] [CrossRef]
- Chen, C.M.; Chen, Y.; Hou, J.H.; Liang, Y.X., Translators; CiteSpace II: Detecting and visualizing emerging trends and transient patterns in scientific literature. J. China Soc. Sci. Tech. Inf. 2009, 28, 401–421. (In Chinese) [Google Scholar]
- Häder, D.-P.; Kumar, H.D.; Smith, R.C.; Worrest, R.C. Effects of solar UV radiation on aquatic ecosystems and interactions with climate change. Photochem. Photobiol. Sci. 2007, 6, 267–285. [Google Scholar] [CrossRef]
- Rastogi, R.P.; Sinha, R.P.; Singh, S.P.; Häder, D.-P. Photoprotective compounds from marine organisms. J. Ind. Microbiol. Biotechnol. 2010, 37, 537–558. [Google Scholar] [CrossRef]
- Llewellyn, C.A.; Airs, R.L. Distribution and abundance of MAAs in 33 species of microalgae across 13 classes. Mar. Drugs 2010, 8, 1273–1291. [Google Scholar] [CrossRef]
- Karsten, U.; Friedl, T.; Schumann, R.; Hoyer, K.; Lembcke, S. Mycosporine-like amino acids and phylogenies in green algae: Prasiola and its relatives from the Trebouxiophyceae (Chlorophyta). J. Phycol. 2005, 41, 557. [Google Scholar] [CrossRef]
- Boedeker, C.; Karsten, U. The occurrence of mycosporine-like amino acids in the gametophytic and sporophytic stages of Bangia (Bangiales, Rhodophyta). Phycologia 2005, 44, 403–408. [Google Scholar] [CrossRef]
- Figueroa, F.L.; Israel, A.; Neori, A.; Martínez, B.; Malta, E.J.; Put, A.; Inken, S.; Marquardt, R.; Abdala-Díaz, R.; Korbee, N. Effect of nutrient supply on photosynthesis and pigmentation to short-term stress (UV radiation) in Gracilaria conferta (Rhodophyta). Mar. Pollut. Bull. 2010, 60, 1768–1778. [Google Scholar] [CrossRef]
- Álvarez-Gómez, F.; Korbee, N.; Figueroa, F.L. Analysis of antioxidant capacity and bioactive compounds in marine macroalgal and lichenic extracts using different solvents and evaluation methods. Cienc. Mar. 2016, 42, 271–288. [Google Scholar] [CrossRef]
- Quintano, E.; Celis-PláP, S.M.; Martínez, B.; Díez, I.; Muguerza, N.; Figueroa, F.L.; Gorostiaga, J.M. Ecophysiological responses of a threatened red alga to increased irradiance in an in situ transplant experiment. Mar. Environ. Res. 2019, 144, 166–177. [Google Scholar] [CrossRef] [PubMed]
- Briani, B.; Sissini, M.N.; Lucena, L.A.; Batista, M.B.; Costa, L.O.; Nunes, J.M.C.; Schmitz, C.; Ramlov, F.; Maraschin, M.; Korbee, N.; et al. The influence of environmental features in the content of mycosporine-like amino acids in red marine algae along the Brazilian coast. J. Phycol. 2018, 54, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Lalegerie, F.; Lajili, S.; Bedoux, G.; Taupin, L.; Stiger-Pouvreau, V.; Connan, S. Photo-protective compounds in red macroalgae from Brittany: Considerable diversity in mycosporine-like amino acids (MAAs). Mar. Environ. Res. 2019, 147, 37–48. [Google Scholar] [CrossRef]
- Orfanoudaki, M.; Hartmann, A.; Miladinovic, H.; Ngoc, H.N.; Karsten, U.; Ganzera, M. Bostrychines A–F, six novel mycosporine-like amino-acids and a novel betaine from the red alga Bostrychia scorpioides. Mar. Drugs 2019, 17, 356. [Google Scholar] [CrossRef]
- Franklin, L.; Kräbs, G.; Kuhlenkamp, R. Blue light and UV-A radiation control the synthesis of mycosporine-like amino acids in Chondrus crispus (Florideophyceae). J. Phycol. 2001, 37, 257–270. [Google Scholar] [CrossRef]
- Banaszak, A.T.; Lesser, M.P.; Kuffner, I.B.; Ondrusek, M. Relationship between ultraviolet (UV) and mycosporine-like amino acids (MAAs) in marine organisms. Bull. Mar. Sci. 1998, 63, 617–628. [Google Scholar]
- Navarro, N.P.; Mansilla, A.; Figueroa, F.; Korbee, N.; Jofre, J.; Plastino, E. Short-term effects of solar UV radiation and NO3− supply on the accumulation of mycosporine-like amino acids in Pyropia columbina (Bangiales, Rhodophyta) under spring ozone depletion in the sub-Antarctic region, Chile. Bot. Mar. 2014, 57, 9–20. [Google Scholar] [CrossRef]
- Aguilera, J.; Bischof, K.; Karsten, U.; Hanelt, D. Seasonal variation in ecophysiological patterns in macroalgae from an Arctic fjord. II. Pigment accumulation and biochemical defence systems against high light stress. Mar. Biol. 2002, 140, 1087–1095. [Google Scholar]
- Korbee, N.; Huovinen, P.; Figueroa, F.L.; Aguilera, J.; Karsten, U. Availability of ammonium influences photosynthesis and the accumulation of mycosporine-like amino acids in two Porphyra species (Bangiales, Rhodophyta). Mar. Biol. 2005, 146, 645–654. [Google Scholar] [CrossRef]
- Huovinen, P.; Matos, J.; Sousa-Pinto, I.; Figueroa, F.L. The role of nitrogen in photoprotection against high irradiance in the Mediterranean red alga Grateloupia lanceola. Aquat. Bot. 2006, 84, 208–316. [Google Scholar] [CrossRef]
- Bhatia, S.; Sharma, K.; Namdeo, A.G.; Chaugule, B.; Kavale, M.; Nanda, S. Broad-spectrum sun-protective action of Porphyra-334 derived from Porphyra vietnamensis. Pharmacogn. Res. 2010, 2, 45. [Google Scholar] [CrossRef]
- Kräbs, G.; Bischof, K.; Hanelt, D.; Karsten, U.; Wiencke, C. Wavelength-dependent induction of UV-absorbing mycosporine-like amino acids in the red alga Chondrus crispus under natural solar radiation. J. Exp. Mar. Biol. Ecol. 2002, 268, 69–82. [Google Scholar] [CrossRef]
- Torres, P.B.; Chow, F.; Ferreira, M.J.P.; dos Santos, D.Y.A.C. Mycosporine-like amino acids from Gracilariopsis tenuifrons (Gracilariales, Rhodophyta) and its variation under high light. J. Appl. Phycol. 2016, 28, 2035–2040. [Google Scholar] [CrossRef]
- Barufi, J.B.; Korbee, N.; Oliveira, M.C.; Figueroa, F.L. Effects of N supply on the accumulation of photosynthetic pigments and photoprotectors in Gracilaria tenuistipitata (Rhodophyta) cultured under UV radiation. J. Appl. Phycol. 2011, 23, 457–466. [Google Scholar] [CrossRef]
- Gómez, I.; Figueroa, F.L.; Huovinen, P.; Ulloa, N.; Morales, V. Photosynthesis of the red alga Gracilaria chilensis under natural solar radiation in an estuary in southern Chile. Aquaculture 2005, 244, 369–382. [Google Scholar] [CrossRef]
- Karsten, U.; West, J.A. Living in the intertidal zone-seasonal effects on heterosides and sun-screen compounds in the red alga Bangia atropurpurea (Bangiales). J. Exp. Mar. Biol. Ecol. 2000, 254, 221–234. [Google Scholar] [CrossRef]
- Cardozo, K.H.M.; Marques, L.G.; Carvalho, V.M.; Carignan, M.O.; Pinto, E.; Marinho-Soriano, E.; Colepicolo, P. Analyses of photoprotective compounds in red algae from the Brazilian coast. Braz. J. Pharmacogn. 2011, 21, 202–208. [Google Scholar] [CrossRef]
- Daniel, S.; Cornelia, S.; Fred, Z. UV-A sunscreen from red algae for protection against premature skin aging. Cosmetic and Toiletries Manufacture worldwide. Food Chem. 2004, 3, 139–143. [Google Scholar]
- Figueroa, F.L.; Escassi, L.; Perez-Rodriguez, E.; Korbee, N.; Giles, A.D.; Johnsen, G. Effects of short-term irradiation on photoinhibition and accumulation of mycosporine-like amino acids in sun and shade species of the red algal genus Porphyra. J. Photochem. Photobiol. B Biol. 2003, 69, 21–30. [Google Scholar] [CrossRef]
- Sinha, R.P.; Klisch, M.; Almut, G.; Häder, D.-P. Mycosporine-like amino acids in the marine red alga Gracilaria cornea-effects of UV and heat. Environ. Exp. Bot. 2000, 43, 33–43. [Google Scholar] [CrossRef]
- De la Coba, F.; Aguilera, J.; de Gálvez, M.V.; Alvarez, M.; Gallego, E.; Figueroa, F.L.; Herrera, E. Prevention of the ultraviolet effects on clinical and histopathological changes, as well as the heat shock protein-70 expression in mouse skin by topical application of algal UV-absorbing compounds. J. Dermatol. Sci. 2009, 55, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Berthon, J.Y.; Nachat-Kappes, R.; Bey, M.; Cadoret, J.P.; Renimela, I.; Filaire, E. Marine algae as attractive source to skin care. Free Radic. Res. 2017, 51, 555–567. [Google Scholar] [CrossRef]
- Torres, P.; Santos, J.P.; Chow, F.; Ferreira, M.J.P.; dos Santos, D.Y.A.C. Comparative analysis of in vitro antioxidant capacities of mycosporine-like amino acids (MAAs). Algal Res. 2018, 34, 57–67. [Google Scholar] [CrossRef]
- Figueroa, F.L.; Korbee, N.; Abdala-Díaz, R.; Jerez, C.G.; López de la Torre, M.; Güenaga, L.; Larrubia, M.A.; Gómez-Pinchetti, J.L. Biofiltration of fishpond effluents and accumulation of N-compounds (phycobiliproteins and mycosporine-like amino acids) versus compounds (polysaccharides) in Hydropuntia cornea (Rhodophyta). Mar. Pollut. Bull. 2012, 64, 310–318. [Google Scholar] [CrossRef]
- Hartmann, A.; Murauer, A.; Ganzera, M. Quantitative analysis of mycosporine-like amino acids in marine algae by capillary electrophoresis with diode-array detection. J. Pharm. Biomed. Anal. 2017, 138, 153–157. [Google Scholar] [CrossRef]
- Bischof, K.; Kräbs, G.; Hanelt, D.; Wiencke, C. Photosynthetic characteristics and mycosporine-like amino acids under UVradiation: A competitive advantage of Mastocarpus stellatus over Chondrus crispus at the Helgoland shoreline? Helgol. Mar. Res. 2000, 54, 47–52. [Google Scholar] [CrossRef]
- Ito, S.; Hirata, Y. Isolation and structure of a mycosporine from the zoanthid Palythoa tuberculosa. Tetrahedron Lett. 1977, 28, 2429–2430. [Google Scholar] [CrossRef]
- Barceló-Villalobos, M.; Figueroa, F.L.; Korbee, N.; Álvarez-Gómez, F.; Abreu, M.H. Production of mycosporine-like amino acids from Gracilaria vermiculophylla (Rhodophyta) cultured through one year in an integrated multi-trophic aquaculture (IMTA) system. Mar. Biotechnol. 2017, 19, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Carreto, J.; Carignan, M.O. Mycosporine-like amino acids: Relevant secondary metabolites. Chemical and ecological aspects. Mar. Drugs 2011, 9, 387–446. [Google Scholar] [CrossRef] [PubMed]
- Cardozo, K.H.M.; Carvalho, V.M.; Pinto, E.; Colepicolo, P. Fragmentation of mycosporine-like amino acids by hydrogen/deuterium exchange and electrospray ionisation tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2006, 20, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, F.L.; Barufi, J.B.; Malta, E.J.; Conde-Álvarez, R.; Nitschke, U.; Arenas, F.; Mata, M.; Connan, S.; Abreu, M.H.; Marquardt, R.; et al. Cystoseira tamariscifolia (Heterokontophyta), Ulva rigida (Chlorophyta) and Ellisolandia elongata (Rhodophyta). Aquat. Biol. 2014, 22, 177–193. [Google Scholar] [CrossRef]
- Hoyer, K.; Karsten, U.; Wiencke, C. Induction of sunscreen compounds in Antarctic macroalgae by different radiation conditions. Mar. Biol. 2002, 141, 619–627. [Google Scholar]
- Ju, Q.; Tang, X.X.; Zhao, X.W.; Ren, X.Q.; Li, Y.F. Effects of UV-B radiation and different light repair conditions on the early development of the tetraspores of Chondrus ocellatus Holm. Acta Oceanol. Sin. 2011, 30, 100–111. [Google Scholar] [CrossRef]
- Ju, Q.; Xiao, H.; Wang, Y.; Tang, X.X. Effects of UV-B radiation on tetraspores of Chondrus ocellatus Holm (Rhodophyta), and effects of red and blue light on repair of UV-B-induced damage. Chin. J. Oceanol. Limnol. 2015, 33, 650–663. [Google Scholar] [CrossRef]
- Korbee, N.; Figueroa, F.L.; Aguilera, J. Effect of light quality on the accumulation of photosynthetic pigments, proteins and mycosporine-like amino acids in the red alga Porphyra leucosticta (Bangiales, Rhodophyta). J. Photochem. Photobiol. B Biol. 2005, 80, 71–78. [Google Scholar] [CrossRef]
- Pliego-Cortés, H.; Bedoux, G.; Boulho, R.; Taupin, L.; Freile-Pelegrín, Y.; Bourgougnon, N.; Robledo, D. Stress tolerance and photoadaptation to solar radiation in Rhodymenia pseudopalmata (Rhodophyta) through mycosporine-like amino acids, phenolic compounds, and pigments in an Integrated Multi-Trophic Aquaculture system. Algal Res. 2019, 41, 101542. [Google Scholar] [CrossRef]
- Orfanoudaki, M.; Hartmann, A.; Karsten, U.; Ganzera, M. Chemical profiling of mycosporine-like amino acids in twenty-three red algal species. J. Phycol. 2019, 55, 393–403. [Google Scholar] [CrossRef]
- Jin, N.N.; Zhang, Z.H.; Li, B.F. The constitutes and extraction analysis of mycos-porine-like amino acids (MAAs) in the Gracilariaceae. Mar. Sci. 2012, 36, 74–80. (In Chinese) [Google Scholar]
- Volkmann, M.; Gorbushina, A.A. A broadly applicable method for extraction and characterization of mycosporines and mycosporine-like amino acids of terrestrial, marine and fresh water origin. FEMS Microbiol. Lett. 2006, 255, 286–295. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Gao, K. Impacts of solar UV radiation on the photosynthesis, growth, and UV-absorbing compounds in Gracilaria lemaneiformis (Rhodophyta) grown at different nitrate concentrations. J. Phycol. 2009, 45, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Jin, N.N. Study on the Isolation, Purification and Application of Mycosporine-Like Amino Acids (MAAs) in Gracilaria changii. Master’s Thesis, Ocean University of China, Qiangdao, China, 2012. [Google Scholar]
- Navarro, N.P. Sunscreens of red algae from Patagonia: A biotechnological perspective. Pure Appl. Chem. 2015, 87, 953–960. [Google Scholar] [CrossRef]
- Roleda, M.Y.; Nyberg, C.D.; Wulff, A. UVR defense mechanisms in eurytopic and invasive Gracilaria vermiculophylla (Gracilariales, Rhodophyta). Physiol. Plant. 2012, 146, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Gacesa, R.; Lawrence, K.P.; Georgakopoulos, N.D.; Yabe, K.; Dunlap, W.C.; Barlow, D.J.; Wells, G.; Young, A.R.; Lon, P.F. The mycosporine-like amino acids porphyra-334 and shinorine are antioxidants and direct antagonists of Keap1-Nrf2 binding. Biochimie 2018, 154, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Navarro, N.P.; Figueroa, F.L.; Korbee, N.; Mansilla, A.; Matsuhiro, B.; Barahona, T.; Plastino, E.M. The Effects of NO3− Supply on Mazzaella laminarioides (Rhodophyta, Gigartinales) from Southern Chile. Photochem. Photobiol. 2014, 90, 1299–1307. [Google Scholar] [CrossRef]
- Jiang, H.X.; Gao, K.S.; Helbling, E.W. UV-absorbing compounds in Porphyra haitanensis (Rhodophyta) with special reference to effects of desiccation. J. Appl. Phycol. 2008, 20, 387–395. [Google Scholar] [CrossRef]
- Velasco-Charpentier, C.; Pizarro-Mora, F.; Navarro, N.P. Variation in mycosporine-like amino acids concentrations in seaweeds from Valparaiso and Magellan Regions, Chile. Rev. Biol. Mar. Oceanogr. 2016, 51, 703–708. [Google Scholar] [CrossRef]
- Karsten, U.; Hoyer, K. UV-absorbing mycosporine-like amino acids in marine macroalgae and their role in UV protection. Ber. Polarforsch. Meeresforsch. 2004, 492, 175–185. [Google Scholar]
- Hoyer, K.; Sabine, S.; Karsten, U.; Wiencke, C. Interactive effects of temperature and radiation on the mycosporine-like arnino acid contents in polar macroalgae. Ber. Polarforsch. Meeresforsch. 2003, 440, 70–89. [Google Scholar]
- Karsten, U.; Wiencke, C. Factors controlling the formation of UV-absorbing mycosporine-like amino acids in the marine red alga Palmaria palmata from Spitsbergen (Norway). J. Plant Physiol. 1999, 155, 407–415. [Google Scholar] [CrossRef]
- Wada, N.; Sakamoto, T.; Matsugo, S. Mycosporine-like amino acids and their derivatives as natural antioxidants. Antioxidants 2015, 4, 603–646. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Pandey, S.; Rajneesh, J.P.; Ahmed, H.; Vidya Singh Shailendra, P.S.; Sinha, R.P. Mycosporine-like amino acids (MAAs) profile of two marine red macroalgae, Gelidium sp. and Ceramium sp. Int. J. Appl. Sci. Biotechnol. 2017, 5, 12–21. [Google Scholar] [CrossRef]
- Bhatia, S.; Sharma, K.; Sharma, A.; Purohit, A.P. Mycosporine and mycosporine-like amino acids: A paramount tool against ultra violet irradiation. Pharmacogn. Rev. 2011, 5, 138–146. [Google Scholar] [CrossRef]
- Álvarez-Gómez, F.; Korbee, N.; Casas-Arrojo, V.; Abdala-Díaz, R.T.; Figueroa, F.L. UV photoprotection, cytotoxicity and immunology capacity of red algae extracts. Molecules 2019, 24, 341. [Google Scholar] [CrossRef]
- Ryu, J.; Park, S.J.; Kim, I.H.; Choi, Y.H.; Nam, T.J. Protective effect of porphyra-334 on UVA-induced photoaging in human skin fibroblasts. Int. J. Mol. Med. 2014, 34, 796–803. [Google Scholar] [CrossRef]
- Kulkarni, A.; Lee, J.H.; Seo, H.H.; Kim, H.S.; Cho, M.J.; Shin, D.S.; Kim, T.; Moh, S.H. Photoinduced conductivity in mycosporine-like amino acids. Mater. Chem. Phys. 2015, 151, 1–4. [Google Scholar] [CrossRef]
- Ying, R.; Zhang, Z.H.; Song, W.S.; Li, B.F.; Hou, H. Protective effect of MAAs extracted from Porphyra tenera against UV irradiation-dinduced photoaging in mouse skin. J. Photochem. Photobiol. B Biol. 2019, 192, 26–33. [Google Scholar]
- Navarro, N.P.; Figueroa, F.; Korbee, N.; Mansilla, A.; Jofre, J.; Plastino, E.M. Differential responses of tetrasporophytes and gametophytes of Mazzaella laminarioides (Gigartinales, Rhodophyta) under solar UV radiation. J. Phycol. 2016, 52, 451–462. [Google Scholar] [CrossRef]
- Navarr, N.P.; Figueroa, F.L.; Korbee, N. Mycosporine-like amino acids vs carrageenan yield in Mazzaella laminarioides (Gigartinales; Rhodophyta) under high and low UV solar irradiance. Phycologia 2017, 56, 570–578. [Google Scholar] [CrossRef]
- Ni, M.Y. The study on the Isolation, Purification, Identification and Antioxidant Activity of Mycosporine-Like Amino Acids (MAAs) in Eucheuma. Master’s Thesis, Ocean University of China, Qiangdao, China, 2014. [Google Scholar]
- Zhang, M.M. The Preparation Techniques of Mycosporine-Like Amino Acid from Porphyra yezoensis. Master’s Thesis, Ocean University of China, Qiangdao, China, 2015. [Google Scholar]
- De la Coba Francisca, A.J.; Korbee, N.; María Victoria de Gálvez, H.-C.E.; Álvarez-Gómez, F.; Figueroa, F.L. UVA and UVB photoprotective capabilities of topical formulations containing mycosporine-like amino acids (MAAs) through different biological effective protection factors (BEPFs). Mar. Drugs 2019, 17, 55. [Google Scholar] [CrossRef]
- Jin, N.N.; Zhang, Z.H.; Li, B.F.; Yan, F.F.; Sun, J.S. Study on the isolation, purification and composition analysis of mycosporine-like amino acids (MAAs) in Gracilaria changii. J. Fish. China 2011, 35, 1829–1836. (In Chinese) [Google Scholar]
- Hartmann, A.; Gostner, J.; Fuchs, J.E.; Chaita, E.; Aligiannis, N.; Skaltsounis, L.; Ganzera, M. Inhibition of collagenase by mycosporine-like amino acids from marine sources. Planta Med. 2015, 81, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Tashiro, Y.; Matsukawa, S.; Ogawa, H. Influence of pH and temperature on the ultraviolet-absorbing properties of porphyra-334. Fish. Sci. 2005, 71, 1382–1384. [Google Scholar] [CrossRef]
- Niu, M.Y.; Zhang, Z.H.; Gao, M.; Bu, L.; Zhang, M.M. Optimization the extraction process of mycosporine-like amino acids from Eucheuma. Acad. Period. Farm Prod. Process. 2014, 7, 42–46. (In Chinese) [Google Scholar]
- He, Q.M. Study on Preparation and Character of UV-Absorbing Compound in Seaweed. Master’s Thesis, Ocean University of China, Qiangdao, China, 2008. [Google Scholar]
- Ying, R.; Zhang, Z.H.; Duan, X.S.; Zhao, X.; Hou, H.; Li, B.F. Optimization of purification process of mycosporine-like amino acid from Porphyra haitanensis and study on its antiultraviolet activity. Mar. Sci. 2017, 41, 71–80. (In Chinese) [Google Scholar]
- Tartarotti, B.; Sommaruga, R. The effect of different methanol concentrations and temperatures on the extraction of mycosporine-like amino acids (MAAs) in algae and zooplankton. Arch. Hydrobiol. 2002, 154, 691–703. [Google Scholar] [CrossRef]
| MAA | Structure | Extinction Coefficient ε (M−1 cm−1) | Maximum Absorption Wavelength (nm) |
|---|---|---|---|
| Asterina-330 | 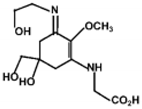 | 43800 | 330 |
| Mycosporine-2-glycine | 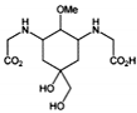 | — | 334 |
| Mycosporine-glycine | 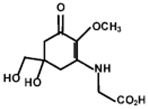 | 28100 | 310 |
| Palythene | 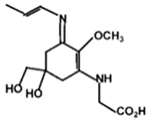 | 50000 | 360 |
| Palythine | 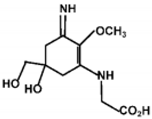 | 35500~36200 | 320 |
| Palythenic acid | 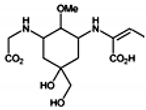 | 29200 | 337 |
| Palythinol | 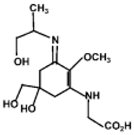 | 43500 | 332 |
| Porphyra-334 | 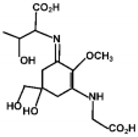 | 42300 | 334 |
| Shinorine | 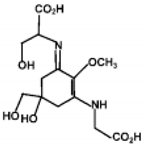 | 43700 | 334 |
| Usujirene | 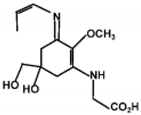 | — | 357 |
| MAAs | Green Macroalgae | Ref. | Brown Macroalgae | Ref. |
|---|---|---|---|---|
| AS | Codium adhaerens, Codium fragile | [7] | Padina crassa | [5] |
| MG | Boodlea composite, Chaetomorpha tortuosa, Codium adhaerens, Prasiola crispa ssp. antarctica | [4,7,34] | Desmarestia menziesii | [5] |
| PE | Caulerpa weberiana, Chaetomorpha tortuosa | [7,34] | ||
| PL | Codium adhaerens | [7] | Padina crassa | [5] |
| PR | Acetabularia mediterranea, Acrosiphonia arcta, Acrosiphonia penicilliformis, Caulerpa racemosa, Cladophora rupestris, Codium adhaerens, Codium fragile, Dictyosphaeria cavernosa, Enteromorpha bulbosa, Ulva lactuca | [2,4,5,7,75] | Ascophyllum nodosum, Chorda tomentosa, Desmarestia aculeata, Desmarestia menziesii, Dictyosiphon foeniculaceus, Dictyota bartayresii, Halopteris scoparia, Himantothallus grandifolius, Hydroclathrus clathratus, Padina crassa, Pilayella littoralis, Sargassum oligocystum | [3,4,5,7,34,75] |
| PI | Acetabularia mediterranea, Cladophora rupestris, Codium adhaerens, Codium fragile | [7,75] | Desmarestia menziesii, Halopteris scoparia, Himantothallus grandifolius, Padina crassa, Sargassum oligocystum | [3,4,5,7,48] |
| SH | Acetabularia mediterranea, Caulerpa weberiana, Cladophora rupestris, Codium adhaerens, Codium fragile, Dictyosphaeria cavernosa, Monostroma hariotii, Valonia aegagropila | [2,5,7,34,75] | Ascophyllum nodosum, Desmarestia aculeata, Fucus spiralis, Halopteris scoparia, Padina crassa, Sargassum oligocystum | [4,5,7,34,75] |
| UN | Enteromorpha intestinalis, Prasiola stipata, Prasiola crispa ssp. antarctica | [2,75] | Prasiola crispa | [49] |
| MAAs | Red Macroalgae | Ref. |
|---|---|---|
| APA | Agarophyton chilense, Bostrychina arbusculaa, Ceramium sp. a, Euptilota formosissima a, Gigartina macrocarpa a, Hymenena affinis, Mastocarpus stellatus a, Porphyra umbilicalis a, Pyropia columbina a, Pyropia plicata a, Rhodophyllis membranecea a, Sarcothalia atropurpurea a, Spongoclonium pastorale a | [71] |
| APB | Agarophyton chilense, Champia novae-zelandiae, Gigartina macrocarpa, Porphyra umbilicalis, Pyropia columbina, Pyropia plicata, Sarcothalia atropurpurea | [71] |
| AS | Acanthophora muscoides, Acanthophora specifera, Agarophyton chilense, Amansia multifida, Ahnfeltiopsis devoniensis, Actinotrichia fragilis, Arthrocardia gardneri, Asparagopsis armata, Bangia atropurpurea, Bonnemaisonia hamifera, Bostrychia arbuscula, Bostrychia calliptera, Bostrychia montagnei, Bostrychia moritziana, Bostrychia radicans, Bostrychia scorpioides, Bostrychia simpliciuscula, Bryothamnion seaforthii, Bostrychia tenella, Bryothamnion triquetrum, Caloglossa apomeiotica, Caloglossa leprieurii, Calliarthron tuberculosum, Caloglossa stipitata, Caloglossa ogasawaraensis, Centroceras clavulatum, Ceramium nodulosum, Ceramium sp., Ceramium secundatum, Champia novae-zelandiae, Chondracanthus acicularis, Chondracanthus elegans, Chondracanthus teedei, Chondrus crispus, Chondrus ocellatus, Corallina officianalis var. chilensisa, Corallina officinalis, Corallina sp., Corallina vancouveriensis, Craspedocarpus erosus, Cryptonemia crenulata, Curdiea racovitzae, Devaleraea ramentacea, Dichotomaria marginata, Digenea simplex, Ellisolandia elongata, Euptilota formosissima, Gastroclonium ovatum, Gelidiella acerosa, Gelidiopsis variabilis, Gelidium amansii, Gelidium crinale, Gelidium corneum, Gelidium floridanum, Gelidium pusillum, Gelidium sesquipedale, Gloiopeltis furcata, Gigartina macrocarpa, Gigartina pistillata, Gigartina skottsbergii, Gracilaria caudata, Gracilaria changii, Gracilaria chilensis, Gracilaria cornea, Gracilaria conferta, Gracilaria eucheumoides, Gracilaria domingensis, Gracilaria saliconia, Gracilaria tenuistipitata, Gracilaria vermiculophylla, Gracilariopsis longissima, Gracilariopsis tenuifrons, Gymnogongrus antarctica, Gymnogongrus antarcticus, Gymnogongrus griffithsiae, Hypnea musciformis, Hypnea spinella, Iridaea chordata, Jania adhaerens, Jania crassa, Jania cubensis, Jania rubens, Jania subulata, Kallymenia antarctica, Laurencia caraibica, Laurencia cartilaginea, Laurencia changii, Laurencia dendroidea, Laurencia filiformis, Laurencia obtusa, Lithophyllum incrustans, Lithophyllum expansum, Mastocarpus stellatus, Mazzaella flaccida, Mazzaella laminarioides, Myriogramme manginii, Notophycus fimbriatus, Osmundea hybrid, Osmundea pinnatitida, Osmundea spectabilis, Pachymenia laciniata, Palmaria decipiens, Palmaria palmata, Palisada flagellifera, Palisada perforate, Phyllophora appendiculata, Plocamium cartilagineum, Polysiphonia arctica, Porphyra endiviifolium, Porphyra columbina, Porphyra leucosticta, Porphyra rosengurttii, Porphyra ssp., Prionitis lanceolata, Pterocladiella capillacea, Pyropia acanthophora, Pyropia columbina, Pyropia plicata, Rhodophyllis membranecea, Rhodymenia pseudopalmata, Sarcothalia atropurpurea, Solieria filiformis, Spongoclonium pastorale, Spyridia clavata, Stictosiphonia arbuscula, Stictosiphonia hookeri, Stictosiphonia intricate, Stictosiphonia tangatensis, Tricleocarpa cylindrica, Vertebrata lanosa | [3,4,5,6,7,32,35,37,39,40,41,42,44,48,49,51,52,55,66,67,70,71,74,75,78,79,80,81,95,96,97,98,99,100,101,102,103,104] |
| CL | Catenella caespitosa, Catenella repens, Catenella nipae | [49,72] |
| MAG | Champia novae-zelandiae, Ceramium sp., Gigartina macrocarpa, Mastocarpus stellatus, Porphyra umbilicalis, Pyropia columbina, Pyropia plicata, Rhodophyllis membranecea, Sarcothalia atropurpurea, Spongoclonium pastorale | [78] |
| MG | Acanthophora muscoides, Acanthophora specifera, Agarophyton chilense, Apophlaea lyallii, Blastophyllis calliblepharoides, Bostrychia moritziana, Bostrychia radicans, Bostrychia scorpioides, Centroceras clavulatum, Ceramium rubrum, Ceramium sp., Champia novae-zelandiae, Chondria arinata, Curdiea racovitzae, Devaleraea ramentacea, Dumontia incrassata a, Gracilaria caudata, Gracilaria cornea, Grateloupia lanceola, Gymnogongrus turquetii, Hypnea spinella, Iridaea chordata, Jania subulata, Kallymenia antarctica, Laurencia caraibica, Laminaria saccharina, Mazzaella laminarioides, Notophycus fimbriatus, Osmundaria obtusiloba, Pachymenia orbicularis, Palmaria decipiens, Palmaria palmata, Phyllophora antarctica, Phyllophora appendiculata, Porphyra columbina, Porphyra endiviifolium, Porphyra purpurea-violacea, Pyropia plicata, Rhodymenia spp., Sarcothalia atropurpurea, Schizymenia apoda, Spongoclonium pastorale | [2,3,4,5,6,7,32,34,35,37,40,42,43,44,45,55,69,71,74,75,76,77,78] |
| MMT | Agarophyton chilense, Ceramium sp., Porphyra umbilicalis, Pyropia columbina, Pyropia plicata, Sarcothalia atropurpurea | [77] |
| M2G | Gloiopeltis furcata | [40] |
| PE | Acanthophora specifera, Actinotrichia fragilis, Agarophyton chilense, Bangia atropurpurea, Bostrychia simpliciuscula, Ceramium nodulosum, Chondrus crispus, Curdiea racovitzae, Devaleraea ramentacea, Gigartina macrocarpa, Gracilaria changii, Gracilaria chilensis, Gracilaria tenuistipitata, Gracilariopsis tenuifrons, Gracilaria vermiculophylla, Iridaea chordata, Osmundea hybrid, Osmundea pinnatitida, Palmaria decipiens, Palmaria palmata, Phyllophora antarctica, Phyllophora appendiculata, Porphyra purpurea-violacea, Pyropia plicata, Rhodymenia pseudopalmata, Sarcothalia atropurpurea, Spongoclonium pastorale, Stictosiphonia hookeri, Vertebrata lanosa | [3,4,5,32,34,40,41,42,43,44,70,75,77,96,99,102,104] |
| PA | Solieria chordalis | [54] |
| PL | Acanthophora specifera, Actinotrichia fragilis, Bangia atropurpurea, Bostrychia calliptera, Bostrychia montagnei, Bostrychia moritziana, Bostrychia radicans, Bostrychia simpliciuscula, Bostrychia tenella, Caloglossa leprieurii, Chondria arinata, Chondrus crispus, Corallina vancouveriensis, Curdiea racovitzae, Devaleraea ramentacea, Ellisolandia elongata, Gelidium corneum, Gelidium pusillum, Gelidium sesquipedale, Gracilaria changii, Gracilaria chilensis, Gracilaria cornea, Gracilaria domingensis, Gracilaria eucheumoides, Gracilaria saliconia, Gracilaria tenuistipitata, Gracilariopsis longissima, Gracilariopsis tenuifrons, Halopythis incurve, Iridaea chordata, Kallymenia antarctica, Laurencia cartilaginea, Laurencia changii, Laurencia obtusa, Mazzaella flaccida, Mastocarpus stellatus, Osmundea spectabilis, Palmaria decipiens, Palmaria palmata, Plocamium cartilagineum, Polysiphonia arctica, Porphyra endiviifolium, Rhodymenia pseudopalmata, Rhodymenia spp., Stictosiphonia intricate, Stictosiphonia tangatensis | [3,4,5,32,35,37,39,40,41,42,44,45,48,52,55,65,67,75,79,96,97,98,99,102,104,105] |
| PR | Acanthophora muscoides, Acanthophora specifera, Actinotrichia fragilis, Agarophyton chilense, Amansia multifida, Arthrocardia gardneri, Asparagopsis armata, Asparagopsis taxiformis, Bangia atropurpurea, Bangia fuscopurpurea, Blastophyllis calliblepharoides, Bostrychia arbuscula, Bostrychia calliptera, Bostrychia harveyi, Bostrychia montagnei, Bostrychia moritziana, Bostrychia radicans, Bostrychia scorpioides, Bostrychia simpliciuscula, Bostrychia tenella, Bryothamnion seaforthii, Calliarthron tuberculosum, Caloglossa apomeiotica, Caloglossa leprieurii, Caloglossa ogasawaraensis, Caloglossa stipitata, Calliarthron tuberculosum, Catenella nipae, Ceramium nodulosum, Ceramium sp., Champia novae-zelandiae, Chondracanthus acicularis, Chondria arinata, Chondrus crispus, Corallina officinalis, Corallina officianalis var. chilensisa, Corallina vancouveriensis, Craspedocarpus erosus, Curdiea racovitzae, Cystoclonium purpureum, Devaleraea ramentacea, Dumontia incrassata, Endocladia muricata, Euptilota formosissima, Galaxaura oblongata, Ganonema farinosa, Gastroclonium ovatum, Gelidiella acerosa, Gelidiopsis variabilis, Gelidium amansii, Gelidium crinale, Gelidium corneum, Gelidium floridanum, Gelidium pusillum, Gelidium sesquipedale, Georgiella confluens, Gigartina macrocarpa, Gigartina skottsbergii, Gloiopeltis furcata, Gracilaria birdiae, Gracilaria caudata, Gracilaria changii, Gracilaria chilensis, Gracilaria conferta, Gracilaria cornea, Gracilaria domingensis, Gracilaria eucheumoides, Gracilaria saliconia, Gracilaria vermiculophylla, Gracilaria tenuistipitata, Grateloupia lanceola, Gymnogongrus griffithsiae, Halopythis incurve, Hymenena affinis, Hydropuntia cornea, Hypnea musciformis, Hypnea spinella, Iridaea sp., Iridaea chordata, Jania adhaerens, Jania rubens, Kallymenia antarctica, Laurencia caraibica, Laurencia cartilaginea, Laurencia changii, Laurencia dendroidea, Laurencia filiformis, Laurencia obtusa, Lithophyllum expansum, Lithophyllum incrustans, Lithothamnion antarcticum, Lithophyllum expansum, Mazzaella flaccida, Mastocarpus jardinii, Mastocarpus papillatus, Mastocarpus stellatus, Myriogramme manginii, Neuroglossum ligulatum, Nodularia spumigena, Notophycus fimbriatus, Osmundea hybrid, Osmundaria obtusiloba, Osmundea spectabilis, Pachymenia laciniata, Pachymenia orbicularis, Palmaria decipiens, Palmaria palmata, Palisada flagellifera, Palisada perforate, Pantoneura plocamioides, Plocamium cartilagineum, Phyllophora antarctica, Polysiphonia arctica, Porphyra dioica, Porphyra endiviifolium, Porphyra leucosticta, Porphyra plocamiestris, Porphyra purpurea, Porphyra purpurea-violacea, Porphyra rosengurttii, Porphyra sp., Porphyra tenera, Porphyra umbilicalis, Porphyra yezoensis, Porphyra vietnamensis, Prionitis lanceolata, Pseudolithophyllum expansum, Pterocladiella capillacea, Pterocladia sp., Ptilota plumosa, Pyropia acanthophora, Pyropia columbina, Pyropia plicata, Rhodophyllis membranecea, Rhodymenia pseudopalmata, Rhodymenia spp., Rhodymenia subantarctica, Sarcothalia atropurpurea, Schizymenia apoda, Scinaia boergesenii, Spongoclonium pastorale, Spyridia clavata, Spyridia filamentosa, Stictosiphonia arbuscula, Stictosiphonia hookeri, Stictosiphonia intricate, Stictosiphonia tangatensis, Tricleocarpa cylindrical, Vertebrata lanosa | [1,3,4,5,6,7,32,34,35,37,40,41,42,44,45,47,48,51,52,55,57,58,65,66,67,69,71,74,75,76,77,78,79,84,85,86,87,88,90,91,92,95,100,102,104,105,106,107] |
| PI | Acanthophora muscoides, Acanthophora specifera, Agarophyton chilense, Amansia multifida, Amphiroa rigida, Ahnfeltiopsis devoniensis, Arthrocardia gardneri, Asparagopsis armata, Asparagopsis taxiformis, Bangia atropurpurea, Blastophyllis calliblepharoides, Bonnemaisonia hamifera, Bostrychia arbuscula, Bostrychia calliptera, Bostrychia harveyi, Bostrychia moritziana, Bostrychia montagnei, Bostrychia pinnata, Bostrychia radicans, Bostrychia scorpioides, Bryothamnion seaforthii, Bostrychia tenella, Bryothamnion triquetrum, Calliarthron tuberculosum, Caloglossa ogasawaraensis, Centroceras clavulatum, Ceramium nodulosum, Ceramium secundatum, Ceramium sp., Champia novae-zelandiae, Chondracanthus acicularis, Chondracanthus chamissoi, Chondracanthus elegans, Chondracanthus teedei, Chondria arinata, Chondrus crispus, Chondrus ocellatus, Chondrus yendoi, Corallina elongata, Corallina officinalis, Corallina officianalis var. chilensisa, Corallina sp., Corallina vancouveriensis, Craspedocarpus erosus, Cryptonemia crenulata, Curdiea racovitzae, Devaleraea ramentacea, Dichotomaria marginata, Digenea simplex, Dumontia incrassata a, Ellisolandia elongata, Endocladia muricata, Euptilota formosissima, Gastroclonium ovatum, Gelidiella acerosa, Gelidiopsis variabilis, Gelidium amansii, Gelidium corneum, Gelidium crinale, Gelidium floridanum, Gelidium lingulatum, Gelidium pusillum, Gelidium sesquipedale, Georgiella confluens, Gigartina macrocarpa, Gigartina pistillata, Gigartina skottsbergii, Gracilaria asiatica, Gracilaria birdiae, Gracilaria caudata, Gracilaria changii, Gracilaria chilensis, Gracilaria conferta, Gracilaria cornea, Gracilaria domingensis, Gracilaria eucheumoides, Gracilaria saliconia, Gracilaria tenuistipitata, Gracilaria vermiculophylla, Gracilariopsis longissima, Grateloupia lanceola, Gymnogongrus Antarctica, Gymnogongrus antarcticus, Gymnogongrus griffithsiae, Halopythis incurve, Hymenena affinis, Hypnea musciformis, Hypnea spinella, Hypoglossum hypoglossoides, Iridaea chordata, Iridaea sp., Jania adhaerens, Jania crassa, Jania cubensis, Jania rubens, Jania subulata, Kallymenia antarctica, Laurencia caraibica, Laurencia cartilaginea, Laurencia changii, Laurencia dendroidea, Laurencia filiformis, Laurencia obtusa, Lithophyllum incrustans, Lithophyllum expansum, Lithothamnion antarcticum, Lomentaria articulate, Mastocarpus jardinii, Mastocarpus stellatus, Mazzaella flaccida, Mazzaella laminarioides, Myriogramme manginii, Neuroglossum ligulatum, Notophycus fimbriatus, Osmundea hybrid, Osmundaria obtusiloba, Osmundea pinnatitida, Osmundea spectabilis, Pachymenia laciniata, Pachymenia orbicularis, Palmaria decipiens, Palmaria palmata, Palisada flagellifera, Palisada perforate, Phyllophora antarctica, Phyllophora appendiculata, Plocamium cartilagineum, Polysiphonia arctica, Polysiphonia urceolata, Porphyra leucosticta, Porphyra purpurea-violacea, Porphyra rosengurttii, Porphyra sp., Porphyra yezoensis, Porphyra umbilicalis, Prionitis lanceolata, Pterocladiella capillacea, Pterocladia sp., Pyropia columbina, Pyropia acanthophora, Pyropia plicata, Rhodophyllis membranecea, Rhodymenia pseudopalmata, Rhodymenia spp., Sarcothalia atropurpurea, Sarcothalia papillosa, Schizymenia apoda, Scinaia boergesenii, Solieria filiformis, Solieria pacifica, Spongoclonium pastorale, Stictosiphonia arbuscula, Stictosiphonia intricate, Stictosiphonia tangatensis, Tricleocarpa cylindrical, Vertebrata lanosa | [3,4,5,6,7,32,34,35,36,37,39,40,41,42,43,44,45,46,48,50,52,55,56,58,65,66,67,68,69,70,71,75,76,77,78,79,82,83,84,90,91,95,97,100,101,102,108,109,110] |
| SH | Acanthophora muscoides, Acanthophora spicifera, Actinotrichia fragilis, Agarophyton chilense, Ahnfeltiopsis devoniensis, Amansia multifida, Amphiroa rigida, Amphiroa sp., Ahnfeltiopsis devoniensis, Arthrocardia gardneri, Asparagopsis armata, Asparagopsis taxiformis, Bangia atropurpurea, Bangia fuscopurpurea, Bangia sp., Bonnemaisonia hamifera, Bostrychina arbuscula, Bostrychia calliptera, Bostrychia moritziana, Bostrychia montagnei, Bostrychia moritziana, Bostrychia scorpioides, Bostrychia simpliciuscula, Bostrychia tenella, Bryothamnion seaforthii, Bryothamnion triquetrum, Callithamnion tetragonum, Caloglossa apomeiotica, Caloglossa leprieurii, Caloglossa ogasawaraensis, Caloglossa stipitata, Calliarthron tuberculosum, Centroceras clavulatum, Ceramium echionotum, Ceramium nodulosum, Ceramium rubrum, Ceramium secundatum, Ceramium sp., Champia novae-zelandiae, Chondracanthus acicularis, Chondracanthus chamissoi, Chondracanthus elegans, Chondracanthus teedei, Chondria arinata, Chondrus crispus, Chondrus yendoi, Corallina elongata, Corallina officinalis, Corallina officianalis var. chilensisa, Corallina sp., Corallina vancouveriensis, Craspedocarpus erosus, Cryptonemia crenulata, Curdiea racovitzae, Cystoclonium purpureum a, Devaleraea ramentacea, Dichotomaria marginata, Digenea simplex, Dumontia incrassata a, Ellisolandia elongata, Endocladia muricata, Euptilota formosissima, Galaxaura oblongata, Ganonema farinosa, Gastroclonium ovatum, Gelidiella acerosa, Gelidiopsis variabilis, Gelidium amansii, Gelidium corneum, Gelidium crinale, Gelidium floridanum, Gelidium latifolium, Gelidium lingulatum, Gelidium pusillum, Gelidium sp., Gelidium sesquipedale, Georgiella confluens, Gloiopeltis furcata, Gigartina macrocarpa, Gigartina pistillata, Gigartina skottsbergii, Gracilaria birdiae, Gracilaria caudata, Gracilaria cornea, Gracilaria changii, Gracilaria chilensis, Gracilaria conferta, Gracilaria cornea, Gracilaria domingensis, Gracilaria eucheumoides, Gracilaria gracilis, Gracilaria saliconia, Gracilaria tenuistipitata, Gracilaria vermiculophylla, Gracilariopsis longissima, Grateloupia doryphora, Gymnogongrus Antarctica, Gymnogongrus antarcticus, Gymnogongrus griffithsiae, Gymnogongrus turquetii, Halopythis incurve, Hypnea musciformis, Hypnea spinella, Hypoglossum hypoglossoides, Iridaea chordata, Iridaea sp., Jania adhaerens, Jania crassa, Jania cubensis, Jania rubens, Jania subulata, Kallymenia antarctica, Laurencia caraibica, Laurencia changii, Laurencia dendroidea, Laurencia filiformis, Laurencia obtusa, Lithophyllum expansum, Lithophyllum incrustans, Lithophyllum expansum, Lithothamnion antarcticum, Lomentaria articulate, Mazzaella flaccida, Mazzaella laminarioides, Mastocarpus jardinii, Mastocarpus papillatus, Mastocarpus stellatus, Myriogramme manginii, Neuroglossum ligulatum, Notophycus fimbriatus, Osmundaria obtusiloba, Osmundea pinnatitida, Osmundea spectabilis, Pachymenia laciniata, Palmaria decipiens, Palmaria palmata, Palisada flagellifera, Palisada perforate, Pantoneura plocamioides, Phyllophora antarctica, Phyllophora appendiculata, Plocamium cartilagineum, Polysiphonia arctica, Porphyra dioica, Porphyra endiviifolium, Polysiphonia urceolata, Porphyra haitanensis, Porphyra leucosticta, Porphyra plocamiestris, Porphyra purpurea, Porphyra rosengurttii, Porphyra sp., Porphyra umbilicalis, Porphyra yezoensis, Prionitis lanceolata, Pseudolithophyllum expansum a, Pterocladia lucida, Pterocladiella capillacea, Pterocladia sp., Pyropia acanthophora, Pyropia columbina, Pyropia plicata, Rhodomela virgata, Rhodophyllis membranecea, Rhodymenia pseudopalmata, Rhodymenia spp., Rissoella verruculosa, Sarcothalia atropurpurea, Sarcothalia papillosa, Schizymenia apoda, Scinaia boergesenii, Spongoclonium pastorale, Spyridia clavata, Stictosiphonia arbuscula, Stictosiphonia hookeri, Stictosiphonia intricate, Stictosiphonia tangatensis, Trichocarpus crinitus, Tricleocarpa cylindrica | [1,3,4,5,6,7,32,34,35,36,37,38,39,40,42,43,44,45,48,50,51,52,53,55,56,57,66,67,68,70,71,75,76,77,78,79,82,84,85,86,90,91,92,96,98,101,103,105,108,110,111,112,113] |
| US | Agarophyton chilense, Bostrychia scorpioides, Calliarthron tuberculosum, Ceramium nodulosum, Ceramium sp., Chondracanthus acicularis, Corallina officianalis var. chilensisa, Corallina vancouveriensis, Gigartina macrocarpa, Gracilariopsis tenuifrons, Gracilaria vermiculophylla, Mastocarpus papillatus, Mastocarpus stellatus, Mazzaella flaccida, Osmundea hybrid, Osmundea pinnatitida, Palmaria decipiens, Palmaria palmata, Phyllophora Antarctica, Prionitis lanceolata, Pterocladiella capillacea, Pyropia plicata, Rhodophyllis membranecea, Rhodymenia pseudopalmata, Sarcothalia atropurpurea, Spongoclonium pastorale, Vertebrata lanosa | [4,6,37,39,52,55,70,76,77,78,104] |
| NM | Bostrychia scorpioides, Gracilaria vermiculophylla | [70,71] |
| UN | Ahnfeltiopsis devoniensis I~II, Asparagopsis armata, Bonnemaisonia hamifera, Bostrychia calliptera, Bostrychia scorpioides I~II, Bostrychia sp., Callithamnion tetragonum, Callophyllis variegata, Catenella caespitosa I~II, Catenella nipae I~IX, Catenella impudica, Catenella repens I~II, Ceramium nodulosum I~II, Ceraminum rubrum, Ceramium secundatum I~II, Chondracanthus acicularis I~II, Chondria arinata, Chondrus crispus I~II, Chondrus ocellatus, Corallina officinalis, Corallina sp. I~II, Curdiea racovitzae I~II, Devaleraea ramentacea, Gastroclonium ovatum, Gelidium corneum I~II, Gigartina pistillata I~II, Gigartina skottsbergii, Gigartina skottsbergii, Gracilaria changii, Gracilaria conferta, Gracilaria gracilis, Gracilaria lemaneiformis I~II, Gracilaria vermiculophylla I~III, Gracilariopsis longissima, Gracilariopsis tenuifrons, Hypoglossum hypoglossoides, Iridaea chordata, Iridaea tuberculosa, Jridaea cordata, Kallymenia antarctica, Lophurella hoockeriana, Mazzaella laminarioides, Mastocarpus stellatus I~II, Myriogramme manginii, Neuroglossum ligulatum, Nothogenia fastigiata, Notophycus fimbriatus, Nothogenia sp., Osmundea hybrid, Osmundea pinnatitida, Palmaria decipiens, Palmaria palmata I~III, Phyllophora truncata, Polysiphonia arctica, Polysiphonia sp., Porphyra dioica, Porphyra endiviifolium, Porphyra rosengurttii, Porphyria spec., Porphyra yezoensis, Pseudolithophyllum expansum, Ptilota gunneri, Ptilonia magellanica, Pyropia columbina, Rhodomela confervoides, Sarcothalia crispata, Sarcothalia papillosa, Schizoseris sp., Solieria chordalis, Vertebrata lanosa | [1,3,4,6,7,37,40,44,47,54,57,69,70,75,86,88,90,102,105,114,115] |
| Marine Macroalgae | |
|---|---|
| Chlorophyta | Acrosiphonia arcta [3], Acrosiphonia penicilliformis [3], Anadyomene wrightii [3], Boergesenia forbesii [3], Chaetomorpha linum [3], Chaetomorpha melagonium [3], Cladophora rupestris [3], Enteromorpha compressa [3], Enteromorpha intestinalis [3], Enteromorpha sp. [3], Enteromorpha spp. [3], Monostroma nitidum [3], Monostoma arcticum [3], Ulva conglohata [3], Ulva fasciata [3], Ulva lactuca [3], Ulva olivascens [3], Ulva rotundata [3], Valoniopsis pachynema [3] |
| Phaeophyta | Adenocystis utricularis [3], Alaria esculenta [3], Ascoseira mirabilis [3], Chnoospora implexa [3], Chorda filum [3], Chordaria flagelliformis [3], Colpomenia sinuosa [3], Cystoseira usneoides [3], Desmarestia aculeata [3], Desmarestia menziesii [3], Dictyota dichotoma [3], Fucus distichus [3], Fucus serratus [3], Fucus spiralis [3], Fucus vesiculosus [3], Kjellmaniella crassifolia [3], Laminaria digitata [3], Laminaria hyperborean [3], Laminaria japonica [3], Laminaria ochroleuca [3], Laminaria saccharina [3], Laminaria solidungula [3], Padina boryana [3], Padina pavonica [3], Phaeurus antarcticus [3], Saccorhiza dermatodea [3], Saccorhiza polyschides [3], Sargassum muticum [3] |
| Rhodophyta | Antarcticothamnion polysporum [3], Audouinella purpurea [3], Ballia callitricha [3], Bornetia secundiflora [70], Calliblepharis jubata [70], Callithamnion tetragonum [70], Champia parvula [70], Chylocladia verticillata [70], Delesseria lancifolia [3], Delesseria sanguinea [3], Dilsea carnosa [76], Furcellaria lumbricalis [3,76], Griffithsia corallinoides [70], Hymenocladiopsis crustigena [3], Heterosiphonia plumosa [70], Membranoptera alata [70], Metacallophyllis laciniata [70], Myriogramme smithii [3], Odonthalia dentate [4], Pantoneura plocamioides [3], Phyllophora ahnfeltioides [3], Phycodrys austrogeorgica [3], Phycodrys rubens [3,71], Phycodrys quercifolia [3], Phyllophora truncata [3], Picconiella plumosa [3], Plocamium cartilagineum [3], Plumaria plumosa [70], Porphyra plocamiestris [3], Ptilota serrate [3], Polysiphonia elongata [3], Polyides rotundus [3], Sphaerococcus coronopifolius [70] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Y.; Zhang, N.; Zhou, J.; Dong, S.; Zhang, X.; Guo, L.; Guo, G. Distribution, Contents, and Types of Mycosporine-Like Amino Acids (MAAs) in Marine Macroalgae and a Database for MAAs Based on These Characteristics. Mar. Drugs 2020, 18, 43. https://doi.org/10.3390/md18010043
Sun Y, Zhang N, Zhou J, Dong S, Zhang X, Guo L, Guo G. Distribution, Contents, and Types of Mycosporine-Like Amino Acids (MAAs) in Marine Macroalgae and a Database for MAAs Based on These Characteristics. Marine Drugs. 2020; 18(1):43. https://doi.org/10.3390/md18010043
Chicago/Turabian StyleSun, Yingying, Naisheng Zhang, Jing Zhou, Shasha Dong, Xin Zhang, Lei Guo, and Ganlin Guo. 2020. "Distribution, Contents, and Types of Mycosporine-Like Amino Acids (MAAs) in Marine Macroalgae and a Database for MAAs Based on These Characteristics" Marine Drugs 18, no. 1: 43. https://doi.org/10.3390/md18010043
APA StyleSun, Y., Zhang, N., Zhou, J., Dong, S., Zhang, X., Guo, L., & Guo, G. (2020). Distribution, Contents, and Types of Mycosporine-Like Amino Acids (MAAs) in Marine Macroalgae and a Database for MAAs Based on These Characteristics. Marine Drugs, 18(1), 43. https://doi.org/10.3390/md18010043





