Eckol as a Potential Therapeutic against Neurodegenerative Diseases Targeting Dopamine D3/D4 Receptors
Abstract
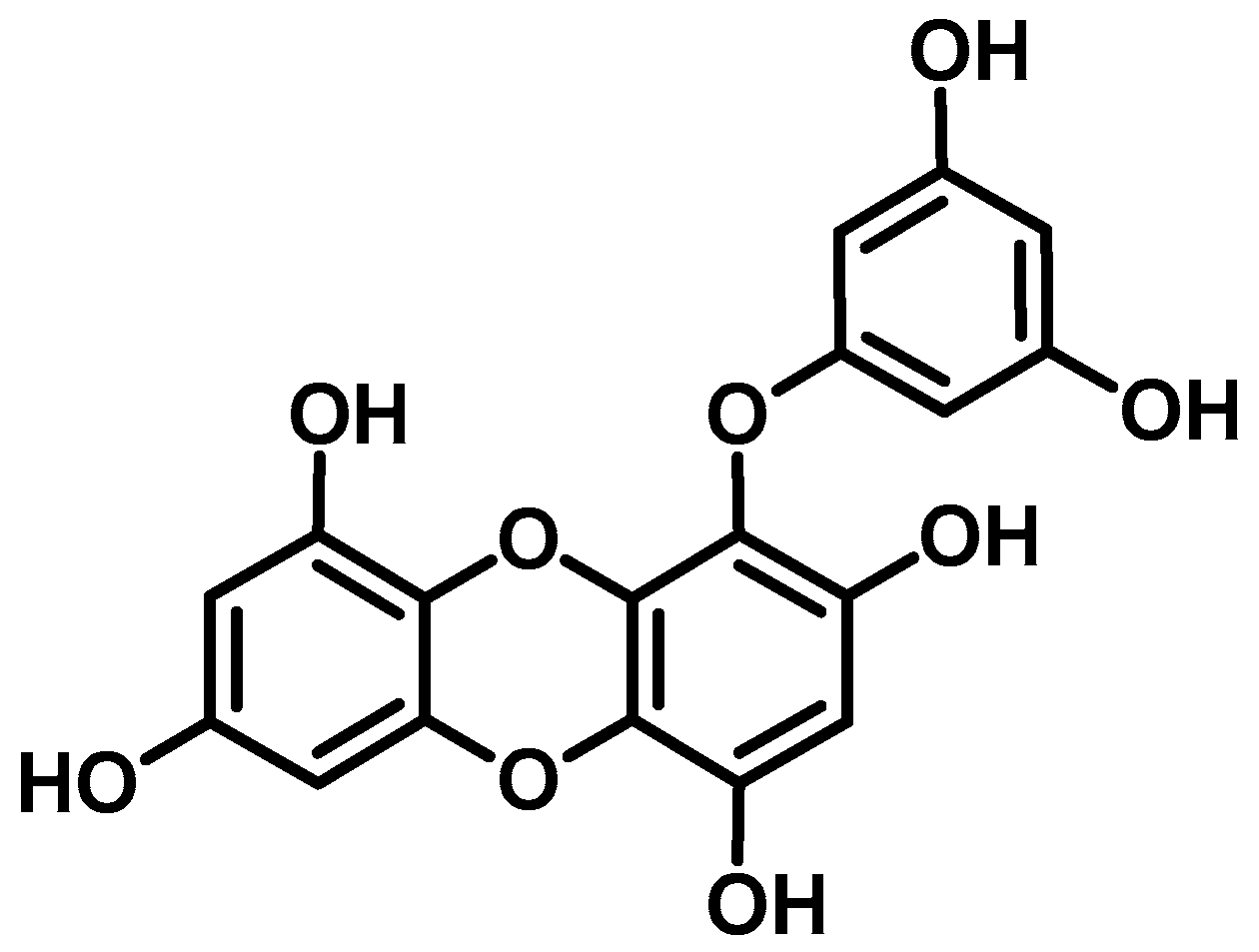
:1. Introduction
2. Results
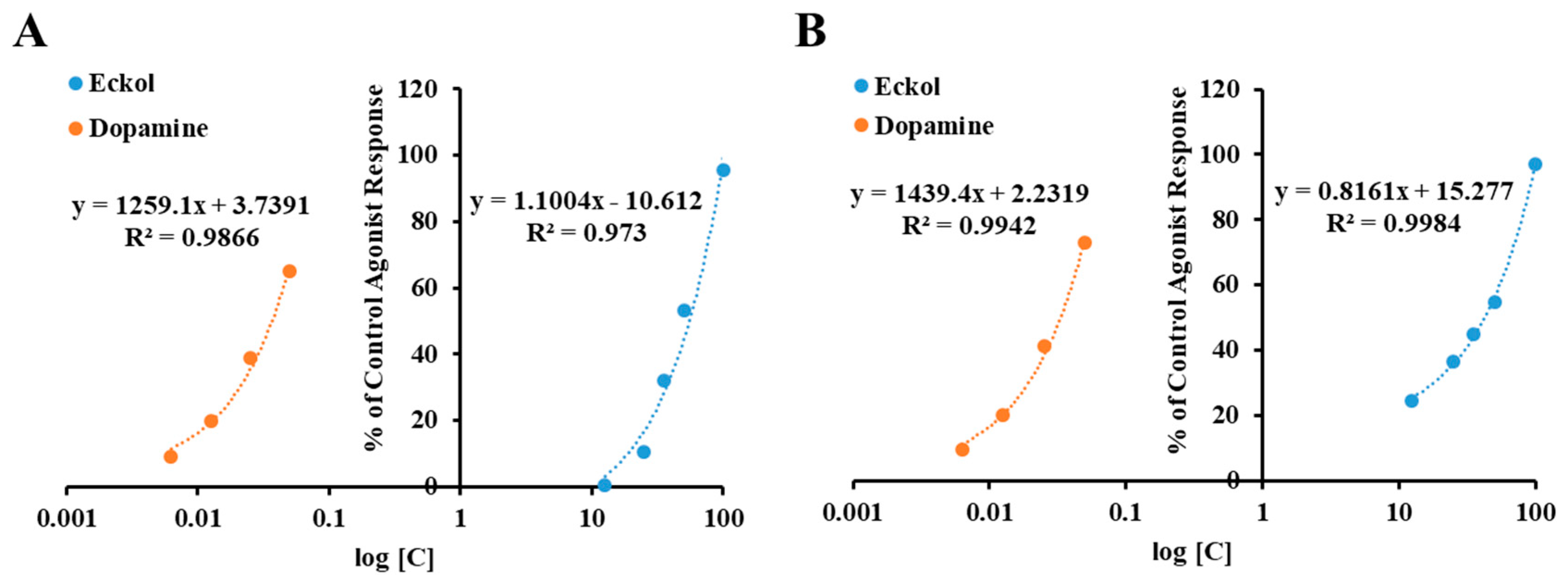
2.1. Functional G-Protein-Coupled Receptor (GPCR) Assay
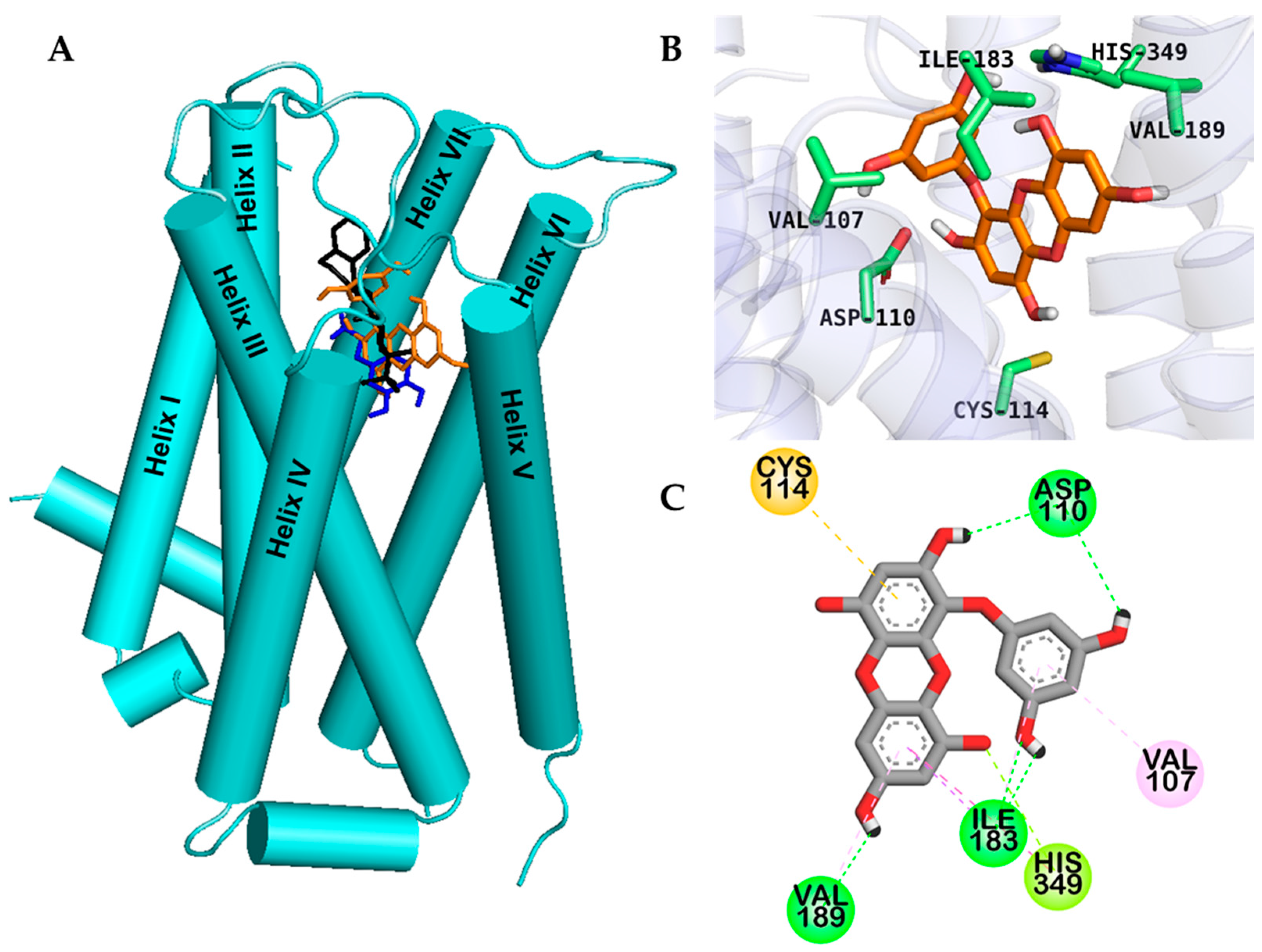
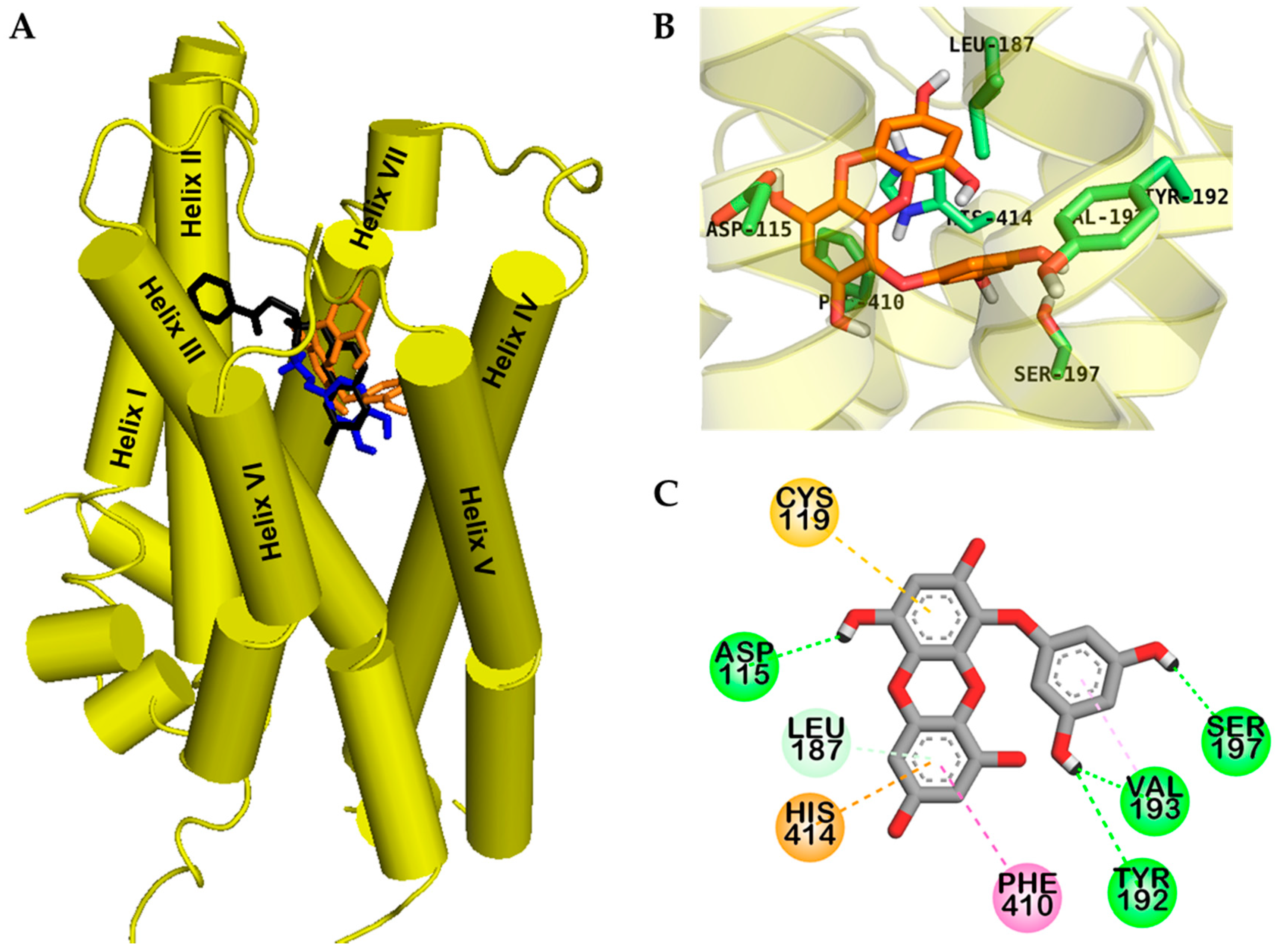
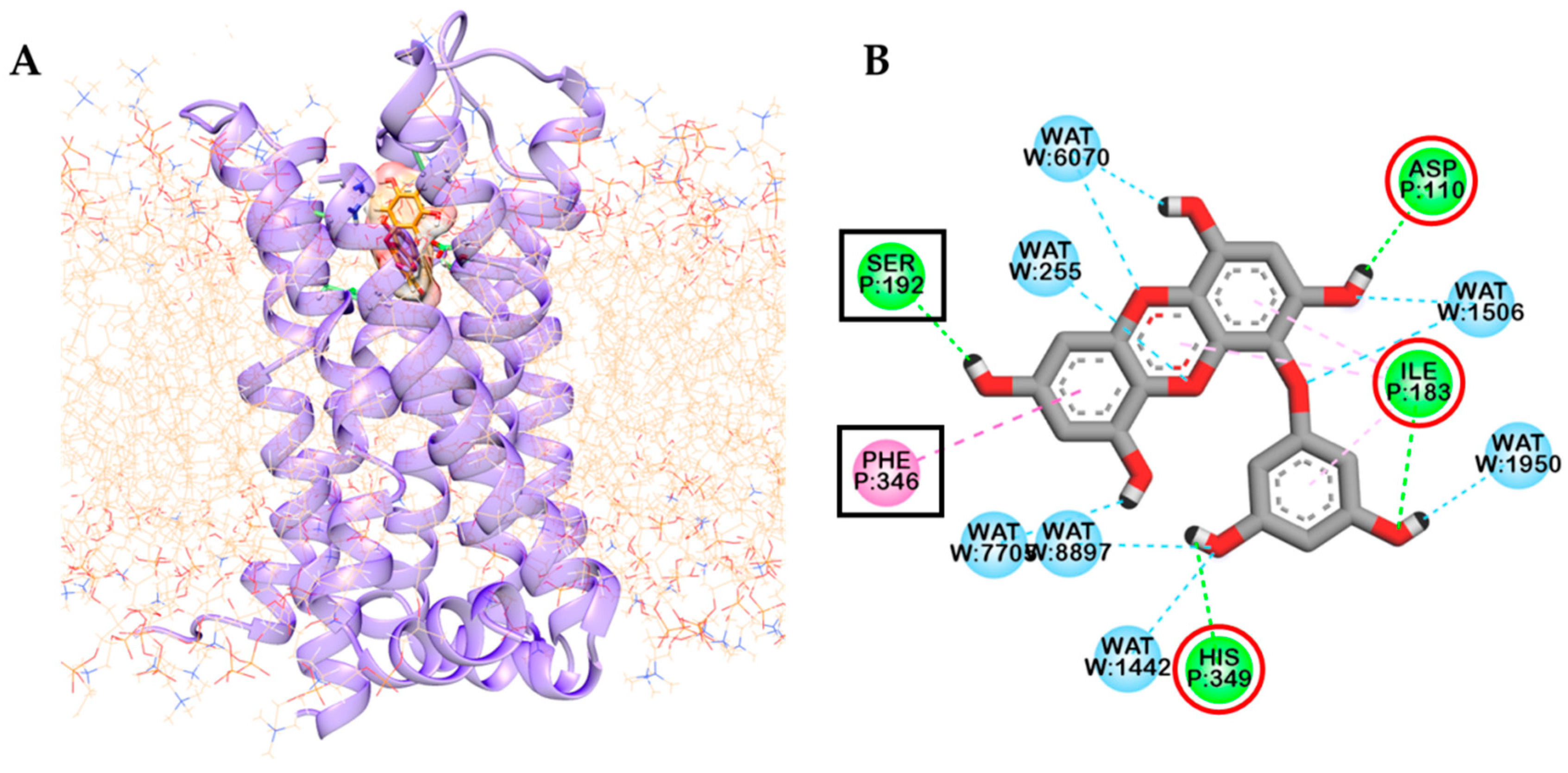
2.2. Molecular Docking Study
2.3. Absorption, Distribution, Metabolism, and Excretion (ADME) Prediction
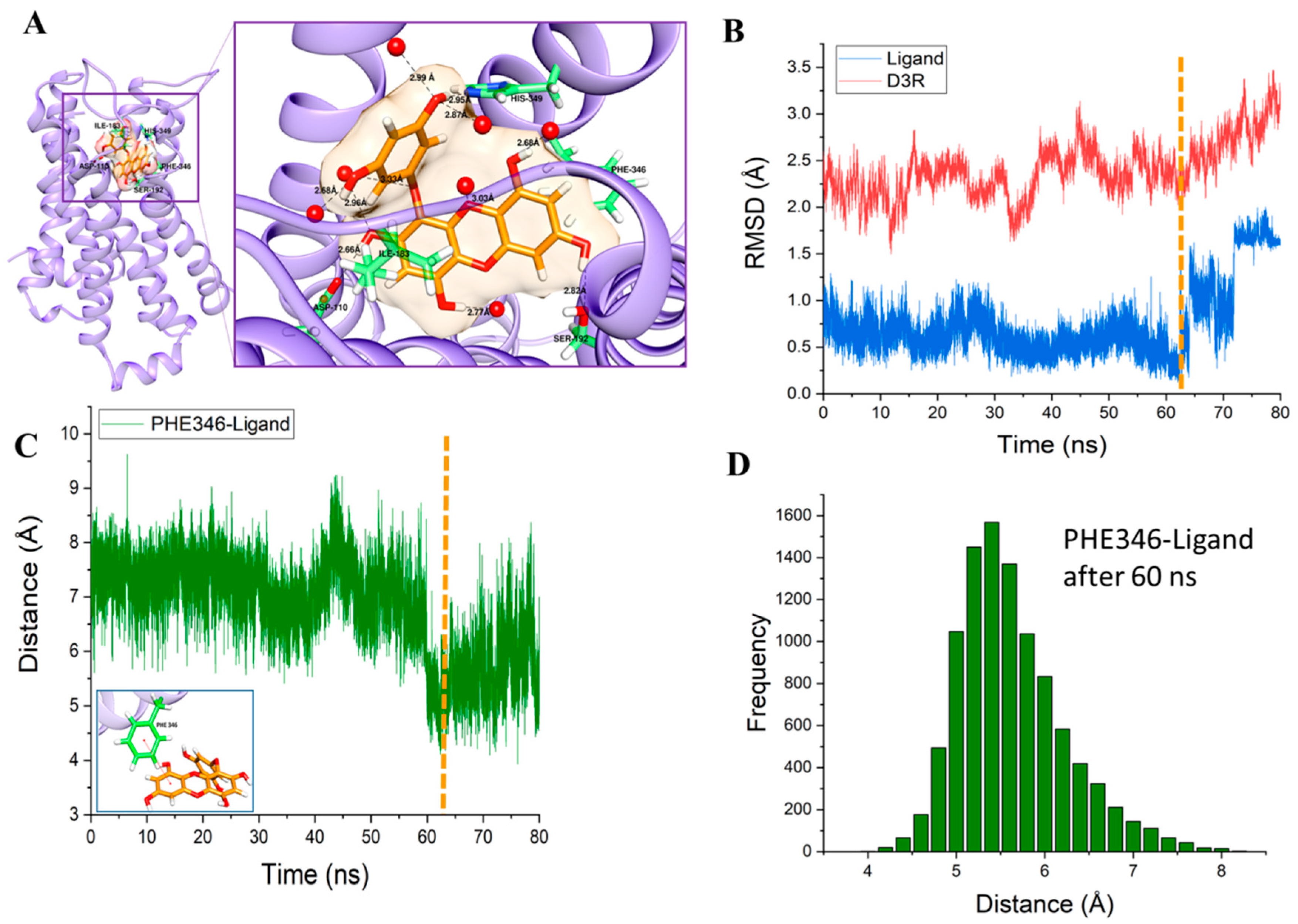
2.4. Molecular Dynamics Simulation Study
3. Discussion
4. Materials and Methods
4.1. Material
4.2. Isolation of Eckol
4.3. Functional GPCR Assay
4.4. Measurement of cAMP Level
4.5. Measurement of Intracellular [Ca2+] Level
4.6. Molecular Docking Study
4.7. ADME Prediction
4.8. Molecular Dynamics Simulation
4.9. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hauser, A.S.; Attwood, M.M.; Rask-Andersen, M.; Schiöth, H.B.; Gloriam, D.E. Trends in GPCR drug discovery: New agents, targets and indications. Nat. Rev. Drug Discov. 2017, 16, 829–842. [Google Scholar] [CrossRef] [PubMed]
- Hauser, A.S.; Chavali, S.; Masuho, I.; Jahn, L.J.; Martemyanov, K.A.; Gloriam, D.E.; Babu, M.M. Pharmacogenomics of GPCR drug targets. Cell 2018, 172, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wacker, D.; Levit, A.; Che, T.; Betz, R.M.; McCorvy, J.D.; Venkatakrishnan, A.; Huang, X.-P.; Dror, R.O.; Shoichet, B.K. D4 dopamine receptor high-resolution structures enable the discovery of selective agonists. Science 2017, 358, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Seeman, P.; Van Tol, H.H. Dopamine receptor pharmacology. Trends Pharmacol. Sci. 1994, 15, 264–270. [Google Scholar] [CrossRef]
- Zhang, W.; Yamada, M.; Gomeza, J.; Basile, A.S.; Wess, J. Multiple muscarinic acetylcholine receptor subtypes modulate striatal dopamine release, as studied with M1–M5 muscarinic receptor knock-out mice. J. Neurosci. 2002, 22, 6347–6352. [Google Scholar] [CrossRef] [PubMed]
- Bara-Jimenez, W.; Bibbiani, F.; Morris, M.J.; Dimitrova, T.; Sherzai, A.; Mouradian, M.M.; Chase, T.N. Effects of serotonin 5-HT1A agonist in advanced Parkinson’s disease. Mov. Disord. 2005, 20, 932–936. [Google Scholar] [CrossRef] [PubMed]
- Egashira, N.; Tanoue, A.; Matsuda, T.; Koushi, E.; Harada, S.; Takano, Y.; Tsujimoto, G.; Mishima, K.; Iwasaki, K.; Fujiwara, M. Impaired social interaction and reduced anxiety-related behavior in vasopressin V1a receptor knockout mice. Behav. Brain Res. 2007, 178, 123–127. [Google Scholar] [CrossRef]
- Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug Discov. 2009, 8, 69–85. [Google Scholar] [CrossRef]
- Mendola, D. Drugs from the Sea; Kargel: Basilea, Switzerland, 2000; pp. 120–133. [Google Scholar]
- Shin, H.-C.; Hwang, H.; Park, K.Y.; Kim, S.H.; Lee, H. Eckol Derivatives, Methods of Synthesis and Uses Thereof. U.S. Patent No. 20180127392A1, 10 May 2018. [Google Scholar]
- Yoon, N.Y.; Chung, H.Y.; Kim, H.R.; Choi, J.S. Acetyl- and butyrylcholinesterase inhibitory activities of sterols and phlorotannins from Ecklonia stolonifera. Fish. Sci. 2008, 74, 200–207. [Google Scholar] [CrossRef]
- Jung, H.A.; Oh, S.H.; Choi, J.S. Molecular docking studies of phlorotannins from Eisenia bicyclis with BACE1 inhibitory activity. Bioorg. Med. Chem. Lett. 2010, 20, 3211–3215. [Google Scholar] [CrossRef]
- Jung, H.A.; Roy, A.; Jung, J.H.; Choi, J.S. Evaluation of the inhibitory effects of eckol and dieckol isolated from edible brown alga Eisenia bicyclis on human monoamine oxidases A and B. Arch. Pharm. Res. 2017, 40, 480–491. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Yang, H.; Jeon, Y.-J.; Lee, C.J.; Jin, Y.-H.; Baek, N.-I.; Kim, D.; Kang, S.-M.; Yoon, M.; Yong, H.; et al. Phlorotannins of the edible brown seaweed Ecklonia cava Kjellman induce sleep via positive allosteric modulation of gamma-aminobutyric acid type A–benzodiazepine receptor: A novel neurological activity of seaweed polyphenols. Food Chem. 2012, 132, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.-M.; Cha, S.-H.; Ko, J.-Y.; Kang, M.-C.; Kim, D.; Heo, S.-J.; Kim, J.-S.; Heu, M.S.; Kim, Y.-T.; Jung, W.-K.; et al. Neuroprotective effects of phlorotannins isolated from a brown alga, Ecklonia cava, against H2O2-induced oxidative stress in murine hippocampal HT22 cells. Environ. Toxicol. Pharmacol. 2012, 34, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Ahn, B.R.; Moon, H.E.; Kim, H.R.; Jung, H.A.; Choi, J.S. Neuroprotective effect of edible brown alga Eisenia bicyclis on amyloid beta peptide-induced toxicity in PC12 cells. Arch. Pharm. Res. 2012, 35, 1989–1998. [Google Scholar] [CrossRef] [PubMed]
- Chien, E.Y.; Liu, W.; Zhao, Q.; Katritch, V.; Han, G.W.; Hanson, M.A.; Shi, L.; Newman, A.H.; Javitch, J.A.; Cherezov, V. Structure of the human dopamine D3 receptor in complex with a D2/D3 selective antagonist. Science 2010, 330, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Ajay; Bemis, G.W.; Murcko, M.A. Designing libraries with CNS activity. J. Med. Chem. 1999, 42, 4942–4951. [Google Scholar] [CrossRef]
- Huang, Y.; Thathiah, A. Regulation of neuronal communication by G protein-coupled receptors. FEBS Lett. 2015, 589, 1607–1619. [Google Scholar] [CrossRef]
- Gainetdinov, R.R.; Premont, R.T.; Bohn, L.M.; Lefkowitz, R.J.; Caron, M.G. Desensitization of G protein–coupled receptors and neuronal functions. Annu. Rev. Neurosci. 2004, 27, 107–144. [Google Scholar] [CrossRef]
- Löber, S.; Hübner, H.; Tschammer, N.; Gmeiner, P. Recent advances in the search for D3- and D4-selective drugs: Probes, models and candidates. Trends Pharmacol. Sci. 2011, 32, 148–157. [Google Scholar] [CrossRef]
- Levant, B. The D3 dopamine receptor: neurobiology and potential clinical relevance. Pharmacol. Rev. 1997, 49, 231–252. [Google Scholar]
- Prante, O.; Dörfler, M.; Gmeiner, P. Dopamine receptor subtype-selective drugs: D2-like receptors. In The Dopamine Receptors, 2nd ed.; Springer: Totowa, NJ, USA, 2010; pp. 101–135. [Google Scholar]
- Kortagere, S.; Cheng, S.-Y.; Antonio, T.; Zhen, J.; Reith, M.E.; Dutta, A.K. Interaction of novel hybrid compounds with the D3 dopamine receptor: Site-directed mutagenesis and homology modeling studies. Biochem. Pharmacol. 2011, 81, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Wiens, B.L.; Nelson, C.S.; Neve, K.A. Contribution of serine residues to constitutive and agonist-induced signaling via the D2S dopamine receptor: Evidence for multiple, agonist-specific active conformations. Mol. Pharmacol. 1998, 54, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Javitch, J.A.; Ballesteros, J.A.; Weinstein, H.; Chen, J. A cluster of aromatic residues in the sixth membrane-spanning segment of the dopamine D2 receptor is accessible in the binding-site crevice. Biochemistry 1998, 37, 998–1006. [Google Scholar] [CrossRef] [PubMed]
- Hemmings, H.C., Jr.; Greengard, P.; Tung, H.L.; Cohen, P. DARPP-32, a dopamine-regulated neuronal phosphoprotein, is a potent inhibitor of protein phosphatase-1. Nature 1984, 310, 503. [Google Scholar] [CrossRef] [PubMed]
- Vallar, L.; Meldolesi, J. Mechanisms of signal transduction at the dopamine D2 receptor. Trends Pharmacol. Sci. 1989, 10, 74–77. [Google Scholar] [CrossRef]
- Beaulieu, J.-M.; Gainetdinov, R.R. The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol. Rev. 2011, 63, 182–217. [Google Scholar] [CrossRef] [PubMed]
- Cussac, D.; Newman-Tancredi, A.; Pasteau, V.; Millan, M.J. Human dopamine D3 receptors mediate mitogen-activated protein kinase activation via a phosphatidylinositol 3-kinase and an atypical protein kinase C-dependent mechanism. Mol. Pharmacol. 1999, 56, 1025–1030. [Google Scholar] [CrossRef]
- Kim, K.C.; Kang, K.A.; Zhang, R.; Piao, M.J.; Kim, G.Y.; Kang, M.Y.; Lee, S.J.; Lee, N.H.; Surh, Y.-J.; Hyun, J.W. Up-regulation of Nrf2-mediated heme oxygenase-1 expression by eckol, a phlorotannin compound, through activation of Erk and PI3K/Akt. Int. J. Biochem. Cell Biol. 2010, 42, 297–305. [Google Scholar] [CrossRef]
- Hyun, K.-H.; Yoon, C.-H.; Kim, R.-K.; Lim, E.-J.; An, S.; Park, M.-J.; Hyun, J.-W.; Suh, Y.; Kim, M.-J.; Lee, S.-J. Eckol suppresses maintenance of stemness and malignancies in glioma stem-like cells. Toxicol. Appl. Pharmacol. 2011, 254, 32–40. [Google Scholar] [CrossRef]
- Hambardzumyan, D.; Becher, O.J.; Rosenblum, M.K.; Pandolfi, P.P.; Manova-Todorova, K.; Holland, E.C. PI3K pathway regulates survival of cancer stem cells residing in the perivascular niche following radiation in medulloblastoma in vivo. Genes Dev. 2008, 22, 436–448. [Google Scholar] [CrossRef]
- Bleau, A.-M.; Hambardzumyan, D.; Ozawa, T.; Fomchenko, E.I.; Huse, J.T.; Brennan, C.W.; Holland, E.C. PTEN/PI3K/Akt pathway regulates the side population phenotype and ABCG2 activity in glioma tumor stem-like cells. Cell Stem Cell 2009, 4, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Jha, S.K.; Jha, N.K.; Kar, R.; Ambasta, R.K.; Kumar, P. p38 MAPK and PI3K/AKT signalling cascades in Parkinson’s disease. Int. J. Mol. Cell. Med. 2015, 4, 67–86. [Google Scholar]
- Oak, J.N.; Lavine, N.; Van Tol, H.H. Dopamine D4 and D2L receptor etimulation of the mitogen-activated protein kinase pathway is dependent ontrans-activation of the platelet-derived growth factor receptor. Mol. Pharmacol. 2001, 60, 92–103. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Buck, D.C.; Yang, R.; Macey, T.A.; Neve, K.A. Dopamine D2 receptor stimulation of mitogen-activated protein kinases mediated by cell type-dependent transactivation of receptor tyrosine kinases. J. Neurochem. 2005, 93, 899–909. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, S.S. Receptor tyrosine kinase transactivation: fine-tuning synaptic transmission. Trends Neurosci. 2003, 26, 119–122. [Google Scholar] [CrossRef]
- Jung, H.A.; Jin, S.E.; Ahn, B.R.; Lee, C.M.; Choi, J.S. Anti-inflammatory activity of edible brown alga Eisenia bicyclis and its constituents fucosterol and phlorotannins in LPS-stimulated RAW264.7 macrophages. Food Chem. Toxicol. 2013, 59, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Goodsell, D.S.; Morris, G.M.; Olson, A.J. Automated docking of flexible ligands: applications of AutoDock. J. Mol. Recognit. 1996, 9, 1–5. [Google Scholar] [CrossRef]
- Kumar, A.; Srivastava, G.; Srivastava, S.; Verma, S.; Negi, A.S.; Sharma, A. Investigation of naphthofuran moiety as potential dual inhibitor against BACE-1 and GSK-3β: Molecular dynamics simulations, binding energy, and network analysis to identify first-in-class dual inhibitors against Alzheimer’s disease. J. Mol. Model. 2017, 23, 239. [Google Scholar] [CrossRef]
- Phillips, J.C.; Braun, R.; Wang, W.; Gumbart, J.; Tajkhorshid, E.; Villa, E.; Chipot, C.; Skeel, R.D.; Kale, L.; Schulten, K. Scalable molecular dynamics with NAMD. J. Comput. Chem. 2005, 26, 1781–1802. [Google Scholar] [CrossRef]
- MacKerell, A.D., Jr.; Banavali, N.; Foloppe, N. Development and current status of the CHARMM force field for nucleic acids. Biopolymers 2000, 56, 257–265. [Google Scholar] [CrossRef]
- Mackerell, A.D., Jr.; Feig, M.; Brooks, C.L., III. Extending the treatment of backbone energetics in protein force fields: Limitations of gas-phase quantum mechanics in reproducing protein conformational distributions in molecular dynamics simulations. J. Comput. Chem. 2004, 25, 1400–1415. [Google Scholar] [CrossRef] [PubMed]
- Zoete, V.; Cuendet, M.A.; Grosdidier, A.; Michielin, O. SwissParam: A fast force field generation tool for small organic molecules. J. Comput. Chem. 2011, 32, 2359–2368. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, W.L.; Chandrasekhar, J.; Madura, J.D.; Impey, R.W.; Klein, M.L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 1983, 79, 926–935. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Feller, S.E.; Zhang, Y.; Pastor, R.W.; Brooks, B.R. Constant pressure molecular dynamics simulation: The Langevin piston method. J. Chem. Phys. 1995, 103, 4613–4621. [Google Scholar] [CrossRef]
- Darden, T.; York, D.; Pedersen, L. Particle mesh Ewald: An N log (N) method for Ewald sums in large systems. J. Chem. Phys. 1993, 98, 10089–10092. [Google Scholar] [CrossRef]
| Receptor | % of Control Agonist Response | EC50 a (μM) | Reference Agonists of Respective Target Receptors EC50 (nM) b | ||
|---|---|---|---|---|---|
| 25 μM | 50 μM | ||||
| D1 (h) Dopamine | −0.58 ± 2.46 e,f | −2.77 ± 1.27 d | ‒ | dopamine | 36 |
| D3 (h) Dopamine | 10.60 ± 4.16 d | 53.10 ± 6.67 c | 48.62 ± 3.21 | dopamine | 2.9 |
| D4 (h) Dopamine | 36.55 ± 10.94 c | 54.66 ± 7.05 c | 42.55 ± 2.54 | dopamine | 3.3 |
| M5 (h) Acetylcholine (muscarinic) | 0.38 ± 0.10 e | 0.39 ± 0.46 d | ‒ | acetylcholine | 3.8 |
| NK1 (h) Tachykinin | −2.32 ± 0.26 e,f | −2.99 ± 0.21 d | ‒ | [Sar9, Met(O2)11]-SP | 0.094 |
| V1A (h) Vasopressin/Oxytocin | −8.91 ± 3.20 f | −12.46 ± 0.51 e | ‒ | AVP | 0.11 |
| 5-HT1A (h) Serotonin | −1.78 ± 0.45 e,f | −3.04 ± 1.23 d | ‒ | serotonin | 3.1 |
| Receptor | % Inhibition of Control Agonist Response | IC50 a (μM) | Reference Antagonists of Respective Target Receptors IC50 (nM) b | ||
|---|---|---|---|---|---|
| 25 μM | 50 μM | ||||
| D1 (h) Dopamine | 11.55 ± 2.15 | 1.89 ± 1.62 | ‒ | SCH 23390 | 0.5 |
| D3 (h) Dopamine | −9.0 ± 6.98 | −15.1 ± 2.51 | ‒ | (+)-butaclamol | 16 |
| D4 (h) Dopamine | 1.33 ± 1.70 | −3.33 ± 4.70 | ‒ | clozapine | 49 |
| M5 (h) Acetylcholine (muscarinic) | 1.0 ± 0.4 | −4.8 ± 2.25 | ‒ | atropine | 0.33 |
| NK1 (h) Tachykinin | −11.35 ± 6.37 | −4.32 ± 3.33 | ‒ | L 733,060 | 0.21 |
| V1A (h) Vasopressin/Oxytocin | −13.46 ± 6.93 | −4.26 ± 11.06 | ‒ | [d(CH2)5 1,Tyr(Me)2]-AVP | 0.05 |
| 5-HT1A (h) Serotonin | 6.17 ± 10.31 | 3.49 ± 4.33 | ‒ | (S)-WAY-100635 | 0.77 |
| Target | Compounds | Binding Energy (kcal/mol) | No. of H-Bonds | H-Bond Interaction Residues | Hydrophobic Interacting Residues | Others |
|---|---|---|---|---|---|---|
| hD3R | Dopamine a (Agonist) | −5.84 | 5 | Salt bridge: Asp110, O–H bond: Val111, Thr115, Ser196 | Alkyl: Val111, Cys114, π–Alkyl: Phe346 | |
| Rotigotine a (Agonist) | −9.23 | 2 | Salt bridge: Asp110, C–H bond: Ser192 | Alkyl: Val111, π–Alkyl: Phe345, His349, Val107, Cys181, Val111, Cys114 | ||
| Eticlopride a (Antagonist) | −8.50 b | 3 | Salt bridge, O–H bond: Asp110, C–H bond: His349 | Alkyl: Val111, Cys114, Val189, π–Alkyl: Phe346, His349, Val111, Ile183, π–π T shaped: Phe345 | ||
| (+)-Butaclamol a (Antagonist) | −8.50 | 1 | Salt bridge: Asp110 | Alkyl: Val86, Val111, Cys114, π–Alkyl: Trp342, Phe346, π–π stacked: Phe345, π–Sigma: Thr369 | ||
| Eckol | −6.41 | 5 | O–H bond: Ile183, His349, Asp110, Val189 | π–Alkyl: Val189, Val107, Ile183, π–π T shaped: His349, π–Sigma: Thr369 | π–Sulfur: Cys114, π–lone pair: His349 | |
| hD4R | Dopamine c (Agonist) | −5.68 | 3 | Salt bridge: Asp115, O–H bond: Ser196 | π–Alkyl: Cys119, π–π T shaped: Phe410, π–Sigma: Val116 | |
| Nemonapride c (Agonist) | −11.82 d | 5 | Salt bridge, O–N bond: Asp115, C–H bond: Ser196, O–H bond: Tyr438 | Alkyl: Val193, π–Alkyl: Leu111, Cys185, π–π T shaped: Phe91, Phe410, π–Sigma: Val116 | π–Sulfur: Cys119, Amide– π stacked: Leu90, Phe91 | |
| CHEMBL332154 c (Antagonist) | −9.42 | 5 | Salt bridge: Asp115, O–H bond: Asp115, Thr120, C–O bond: Cys185 | π–Alkyl: Val87, Cys185, Val116, Leu187, Cys119, π–π T shaped: Phe410, Phe411, His414, π–Sigma: Leu111, Val116 | ||
| Eckol | −6.46 | 4 | O–H bond: Tyr192, Asp115, Val193, Ser197 | π–Alkyl: Leu187, Val193, π–π T shaped: Phe410, His414 | π–Sulfur: Cys119, π–Cation: His414 |
| Compound | Molecular Weight (g/mol) | Log Po/w a | Plasma Protein Binding b | Human Intestinal Absorption c | In Vivo Blood–Brain Barrier Penetration ([brain]/[blood]) d |
|---|---|---|---|---|---|
| Eckol | 372.285 | 2.99 | 100% | 55.60% | 0.25 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paudel, P.; Seong, S.H.; Wu, S.; Park, S.; Jung, H.A.; Choi, J.S. Eckol as a Potential Therapeutic against Neurodegenerative Diseases Targeting Dopamine D3/D4 Receptors. Mar. Drugs 2019, 17, 108. https://doi.org/10.3390/md17020108
Paudel P, Seong SH, Wu S, Park S, Jung HA, Choi JS. Eckol as a Potential Therapeutic against Neurodegenerative Diseases Targeting Dopamine D3/D4 Receptors. Marine Drugs. 2019; 17(2):108. https://doi.org/10.3390/md17020108
Chicago/Turabian StylePaudel, Pradeep, Su Hui Seong, Sangwook Wu, Suhyun Park, Hyun Ah Jung, and Jae Sue Choi. 2019. "Eckol as a Potential Therapeutic against Neurodegenerative Diseases Targeting Dopamine D3/D4 Receptors" Marine Drugs 17, no. 2: 108. https://doi.org/10.3390/md17020108
APA StylePaudel, P., Seong, S. H., Wu, S., Park, S., Jung, H. A., & Choi, J. S. (2019). Eckol as a Potential Therapeutic against Neurodegenerative Diseases Targeting Dopamine D3/D4 Receptors. Marine Drugs, 17(2), 108. https://doi.org/10.3390/md17020108






