Nutraceutical Extract from Dulse (Palmaria palmata L.) Inhibits Primary Human Neutrophil Activation
Abstract
1. Introduction
2. Results
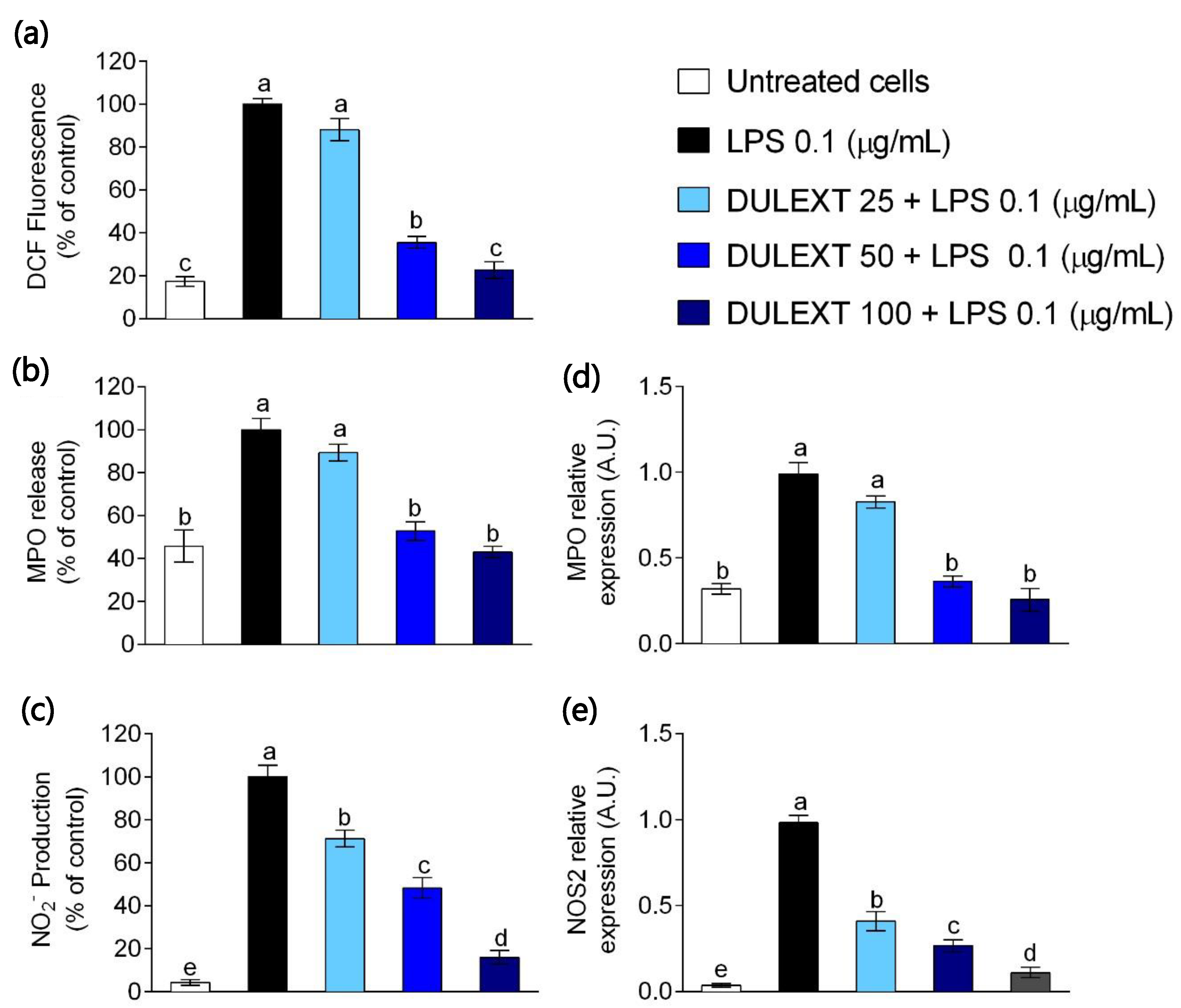
2.1. DULEXT Impairs Oxidant Conditions
2.2. DULEXT Down-Regulates TRL4 and Pro-Inflammatory Cytokine Gene Expression
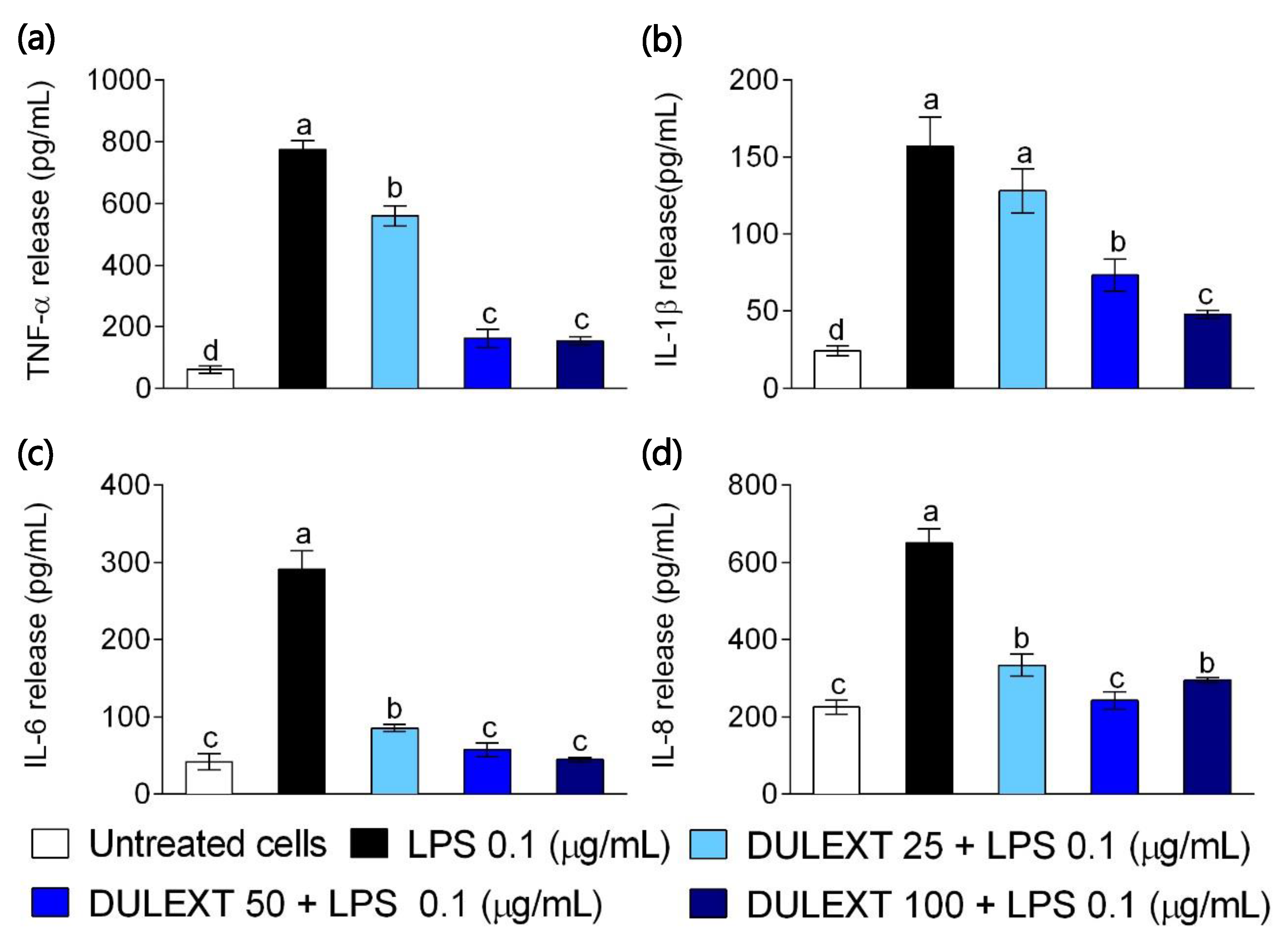
2.3. DULEXT Reduces the Release of Pro-Inflammatory Cytokines
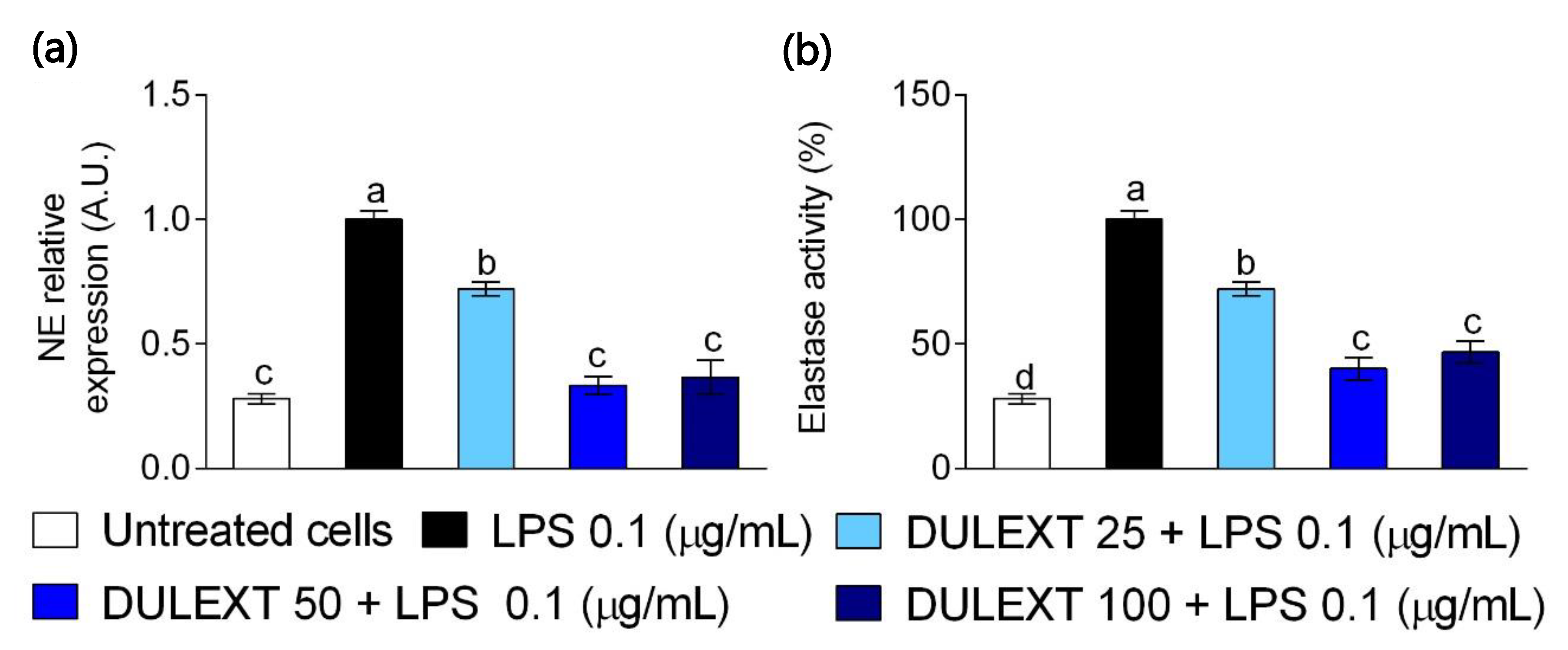
2.4. DULEXT Down-Regulates Neutrophil Elastase Gene Expression and Impairs its Enzymatic Activity
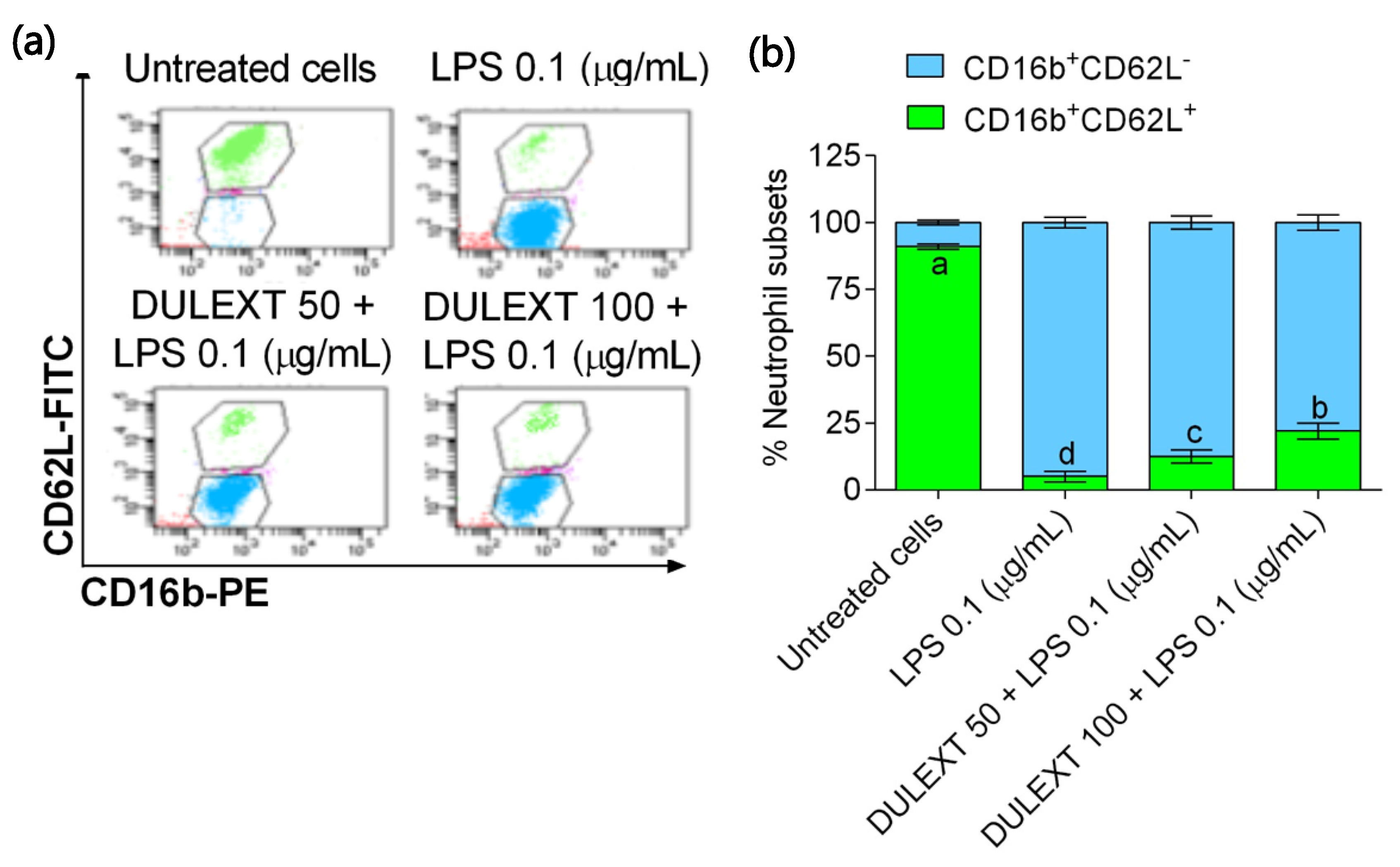
2.5. Effect of DULEXT on Neutrophil Subsets
3. Discussion
4. Materials and Methods
4.1. Preparation of Dulse (Palmaria palmata L.) Extract (DULEXT)
4.2. Blood Collection and Neutrophil Isolation
4.3. Cytotoxicity Assay
4.4. Intracellular Reactive Oxygen Species (ROS)
4.5. Nitrite Production
4.6. Cytokine Release.
4.7. RNA Isolation and Real-Time Quantitative PCR Analysis.
4.8. Human Neutrophil Elastase Inhibition Assay
4.9. Flow Cytometry
4.10. Statistical Evaluation.
5. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Robertson, R.C.; Guihéneuf, F.; Bahar, B.; Schmid, M.; Stengel, D.B.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C. The Anti-Inflammatory Effect of Algae-Derived Lipid Extracts on Lipopolysaccharide (LPS)-Stimulated Human THP-1 Macrophages. Mar. Drugs 2015, 13, 5402–5424. [Google Scholar] [CrossRef] [PubMed]
- Lordan, S.; Ross, R.P.; Stanton, C. Marine bioactives as functional food ingredients: Potential to reduce the incidence of chronic diseases. Mar. Drugs 2011, 9, 1056–1100. [Google Scholar] [CrossRef] [PubMed]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Fernando, S.; Misook, K.; Kwang-Tae, S.; Yoonhwa, J.; You-Jin, J. Antioxidant Activity of Marine Algal Polyphenolic Compounds: A Mechanistic Approach. J. Med. Food 2016, 19, 615–628. [Google Scholar] [CrossRef] [PubMed]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for analysis of plant phenolic compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef]
- Huyut, Z.; Beydemir, S.; İlhami Gülçin, I. Antioxidant and Antiradical Properties of Selected Flavonoids and Phenolic Compounds. Biochem. Res. Int. 2017, 2017, 7616791. [Google Scholar] [CrossRef]
- Allsopp, P.; Crowe, W.; Bahar, B.; Harnedy, P.A.; Brown, E.S.; Taylor, S.S.; Smith, T.J.; Soler-Vila, A.; Magee, P.J.; Gill, C.I.R.; et al. The effect of consuming Palmaria palmata enriched bread on inflammatory markers, antioxidant status, lipid profile and thyroid function in a randomised placebo controlled intervention trial in healthy adults. Eur. J. Nutr. 2016, 55, 1951–1962. [Google Scholar] [CrossRef]
- Hwang, P.A.; Chien, S.Y.; Chan, Y.L.; Lu, M.K.; Wu, C.H.; Kong, Z.L.; Wu, C.J. Inhibition of lipopolysaccharide (LPS)-induced inflammatory responses by Sargassum hemiphyllum sulfated polysaccharide extract in RAW 264.7 macrophage cells. J. Agric. Food Chem. 2011, 59, 2062–2068. [Google Scholar] [CrossRef]
- Lee, J.C.; Hou, M.F.; Huang, H.W.; Chang, F.R.; Yeh, C.C.; Tang, J.Y.; Chang, H.W. Marine algal natural products with anti-oxidative, anti-inflammatory, and anti-cancer properties. Cancer Cell Int. 2013, 13, 55. [Google Scholar] [CrossRef]
- Brown, E.M.; Allsopp, P.J.; Magee, P.J.; Gill, C.I.R.; Nitecki, S.; Strain, C.R.; McSorley, E.M. Seaweed and human health. Nutr. Rev. 2014, 72, 205–216. [Google Scholar] [CrossRef]
- Fitzgerald, C.; Mora-Soler, L.; Gallagher, E.; O’Connor, P.; Prieto, J.; Soler-Vila, A.; Hayes, M. Isolation and Characterization of Bioactive Pro-Peptides with in Vitro Renin Inhibitory Activities from the Macroalga Palmaria palmata. J. Agric. Food Chem. 2012, 60, 7421–7427. [Google Scholar] [CrossRef] [PubMed]
- Banskota, A.H.; Stefanova, R.; Sperker, S.; Lall, S.P.; Craigie, J.S.; Hafting, J.T.; Critchley, A.T. Polar lipids from the marine macroalga Palmaria palmata inhibit lipopolysaccharide-induced nitric oxide production in RAW264 7 macrophage cells. Phytochemistry 2014, 101, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Ordóñez, E.; Jiménez-Escrig, A.; Rupérez, P. Effect of the red seaweed Mastocarpus stellatus intake on lipid metabolism and antioxidant status in healthy Wistar rats. Food Chem. 2012, 135, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Cian, R.E.; López-Posadas, R.; Drago, S.R.; De Medina, F.S.; Martínez-Augustín, O. A Porphyra columbina hydrolysate upregulates IL-10 production in rat macrophages and lymphocytes through an NF-κB, and p38 and JNK dependent mechanism. Food Chem. 2012, 134, 1982–1990. [Google Scholar] [CrossRef]
- Jung, W.K.; Choi, I.; Oh, S.; Park, S.G.; Seo, S.K.; Lee, S.W.; Lee, D.S.; Heo, S.J.; Jeon, I.J.; Je, J.Y.; et al. Anti-asthmatic effect of marine red alga Laurencia undulata polyphenolic extracts in a murine model of asthma. Food Chem. Toxicol. 2009, 47, 293–297. [Google Scholar] [CrossRef]
- Sánchez-Machado, D.I.; López-Cervantes, J.; López-Hernández, J.; Paseiro-Losada, P. Fatty acids, total lipid, protein and ash contents of processed edible seaweeds. Food Chem. 2004, 85, 439–444. [Google Scholar] [CrossRef]
- Price, R.K.; Wallace, J.M.W.; Hamill, L.L.; Keaveney, E.M.; Strain, J.J.; Parker, M.J.; Welch, R.W. Evaluation of the effect of wheat aleurone-rich foods on markers of antioxidant status, inflammation and endothelial function in apparently healthy men and women. Br. J. Nutr. 2012, 108, 1644–1651. [Google Scholar] [CrossRef]
- Machu, L.; Misurcova, L.; Ambrozova, J.V.; Orsavova, J.; Mlcek, J.; Sochor, J.; Jurikova, T. Phenolic Content and Antioxidant Capacity in Algal Food Products. Molecules 2015, 20, 1118–1133. [Google Scholar] [CrossRef]
- Suwal, S.; Perreault, V.; Marciniak, A.; Tamigneaux, E.; Deslandes, E.; Bazinet, L.; Jacques, H.; Beaulieu, L.; Doyen, A. Effects of high hydrostatic pressure and polysaccharidases on the extraction of antioxidant compounds from red macroalgae, Palmaria palmata and Solieria chordalis. J. Food Eng. 2019, 252, 53–59. [Google Scholar] [CrossRef]
- Yuan, Y.V.; Westcott, N.D.; Hu, C.; Kitts, D.D. Mycosporine- like amino acid composition of the edible red alga, Palmaria palmata (dulse) harvested from the west and east coasts of Grand Manan Island, New Brunswick. Food Chem. 2009, 112, 321–328. [Google Scholar] [CrossRef]
- Wang, T.; Jónsdóttir, R.; Kristinsson, H.G.; Hreggvidsson, G.O.; Jónsson, J.O.; Thorkelsson, G.; Ólafsdóttir, G. Enzyme-enhanced extraction of antioxidant ingredients from red algae Palmaria palmata. LWT Food Sci. Technol. 2010, 43, 1387–1393. [Google Scholar] [CrossRef]
- Park, E.J.; Pezzuto, J.M. Antioxidant marine products in cancer chemoprevention. Antioxid. Redox Signal. 2013, 19, 115–138. [Google Scholar] [CrossRef] [PubMed]
- Mouritsen, O.G.; Dawczynski, C.; Duelund, L.; Jahreis, G.; Vetter, W.; Schröder, M. On the human consumption of the red seaweed dulse (Palmaria palmata (L.) Weber & Mohr. J. Appl. Phycol. 2013, 25, 1777–1791. [Google Scholar]
- Coussens, L.M.; Werb, Z. Inflammation and Cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Cassatella, M.A.; Costantini, C.; Jaillon, S. Neutrophils in the activation and regulation of innate and adaptative immunity. Nat. Rev. Immunol. 2011, 11, 519–531. [Google Scholar] [CrossRef]
- Nauseef, W.M. How neutrophils kill and degrade microbes: An integrated view. Immunol. Rev. 2007, 219, 88–102. [Google Scholar] [CrossRef]
- Michel, P.; Granica, S.; Magiera, A.; Rosińska, K.; Jurek, M.; Poraj, L.; Olszewska, M.A. Salicylate and Procyanidin-Rich Stem Extracts of Gaultheria procumbens L. Inhibit Pro-Inflammatory Enzymes and Suppress Pro-Inflammatory and Pro-Oxidant Functions of Human Neutrophils Ex Vivo. Int. J. Mol. Sci. 2019, 20, 1753. [Google Scholar] [CrossRef]
- Roberts, K.J.; Cross, A.; Vasieva, O.; Moots, R.J.; Edwards, S.W. Inhibition of pre-B cell colony-enhancing factor (PBEF/NAMPT/visfatin) decreases the ability of human neutrophils to generate reactive oxidants but does not impair bacterial killing. J. Leukoc. Biol. 2013, 94, 481–492. [Google Scholar] [CrossRef]
- Berry, M.P.; Graham, C.M.; McNab, F.W.; Xu, Z.; Bloch, S.A.; Oni, T.; Wilkinson, K.A.; Banchereau, R.; Skinner, J.; Wilkinson, R.J.; et al. An interferon-inducible neutrophil-driven blood transcriptional signature in human tuberculosis. Nature 2010, 466, 973–977. [Google Scholar] [CrossRef]
- Nauseef, W.M. Neutrophils, from cradle to grave and beyond. Immunol. Rev. 2016, 273, 5–10. [Google Scholar] [CrossRef]
- Geng, S.; Zhang, Y.; Lee, C.; Li, L. Novel reprogramming of neutrophils modulates inflammation resolution during atherosclerosis. Sci. Adv. 2019, 5, eaav2309. [Google Scholar] [CrossRef] [PubMed]
- Soehnlein, O.; Steffens, S.; Hidalgo, A.; Weber, C. Neutrophils as protagonists and targets in chronic inflammation. Nat. Rev. Immunol. 2017, 17, 248–261. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, R.; Miranda, A.; Vergara, L. Modulation of endogenous antioxidant system by wine polyphenols in human disease. Clin. Chim. Acta 2011, 412, 410–424. [Google Scholar] [CrossRef] [PubMed]
- Pangestuti, R.; Kim, S.K. Biological activities and health benefit effects of natural pigments derived from marine algae. J. Funct. Foods 2011, 3, 255–266. [Google Scholar] [CrossRef]
- Yuan, Y.V.; Carrington, M.F.; Walsh, N.A. Extracts from dulse (Palmaria palmata) are effective antioxidants and inhibitors of cell proliferation in vitro. Food Chem. Toxicol. 2005, 43, 1073–1081. [Google Scholar] [CrossRef]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. BioMed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef]
- Jiménez-Escrig, A.; Jiménez-Jiménez, I.; Pulido, R.; Saura-Calixto, F. Antioxidant activity of fresh and processed edible seaweeds. J. Sci. Food Agric. 2001, 81, 530–534. [Google Scholar] [CrossRef]
- Lee, D.; Nishizawa, M.; Shimizu, Y.; Saeki, H. Anti-inflammatory effects of dulse (Palmaria palmata) resulting from the simultaneous water-extraction of phycobiliproteins and chlorophyll α. Food Res. Int. 2017, 100, 514–521. [Google Scholar] [CrossRef]
- Rosales, C. Neutrophil: A Cell with Many Roles in Inflammation or Several Cell Types? Front. Physiol. 2018, 9, 113. [Google Scholar] [CrossRef]
- Mortaz, E.; Alipoor, S.D.; Adcock, I.M.; Mumby, S.; Koenderman, L. Update on Neutrophil Function in Severe Inflammation. Front. Immunol. 2018, 9, 2171. [Google Scholar] [CrossRef]
- Kettritz, R. Neutral serine proteases of neutrophils. Immunol. Rev. 2016, 273, 232–248. [Google Scholar] [CrossRef] [PubMed]
- Colom, B.; Bodkin, J.V.; Beyrau, M.; Woodfin, A.; Ody, C.; Rourke, C.; Chavakis, T.; Brohi, K.; Imhof, B.A.; Nourshargh, S. Leukotriene B4-neutrophil elastase axis drives neutrophil reverse transendothelial cell migration in vivo. Immunity 2015, 42, 1075–1086. [Google Scholar] [CrossRef] [PubMed]
- Aparicio-Soto, M.; Montserrat-de la Paz, S.; Sanchez-Hidalgo, M.; Cardeno, A.; Bermúdez, B.; Muriana, F.J.G.; Alarcón-de la Lastra, C. Virgin olive oil and its phenol fraction modulate monocyte/macrophage functionality: A potential therapeutic strategy in the treatment of systemic lupus erythematosus. Br. J. Nutr. 2018, 120, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Bernaldo de Quiros, A.; Lage-Yusty, M.A.; López-Hernández, J. Determination of phenolic compounds in macroalgae for human consumption. Food Chem. 2010, 121, 634–638. [Google Scholar] [CrossRef]
- Montserrat-de la Paz, S.; Fernandez-Arche, M.A.; Ángel-Martín, M.; García-Giménez, M.D. The sterols isolated from Evening Primrose oil modulate the release of proinflammatory mediators. Phytomedicine 2012, 19, 1072–1076. [Google Scholar] [CrossRef]
- Cardeno, A.; Aparicio-Soto, M.; Montserrat-de la Paz, S.; Bermúdez, B.; Muriana, F.J.G.; Alarcón-de-la-Lastra, C. Squalene targets pro- and anti-inflammatory mediators and pathways to modulate over-activation of neutrophils, monocytes and macrophages. J. Funct. Foods 2015, 14, 779–790. [Google Scholar] [CrossRef]
- Montserrat-de la Paz, S.; García-Giménez, M.D.; Ángel-Martín, M.; Pérez-Camino, M.C.; Fernández-Arche, A. Long-chain fatty alcohols from evening primrose oil inhibit the inflammatory response in murine peritoneal macrophages. J. Ethnopharmacol. 2014, 151, 131–136. [Google Scholar] [CrossRef]
- Schepetkin, I.A.; Khlebnikov, A.I.; Quinn, M.T. N-benzoylpyrazoles are novel small-molecule inhibitors of human neutrophil elastase. J. Med. Chem. 2007, 50, 4928–4938. [Google Scholar] [CrossRef]

| Polyphenol | Chemical Structure | Concentration μg/mg of Extract |
|---|---|---|
| 4-hydroxybenzoic acid (HBA) |  | 6.3 ± 0.7 |
| Epicatechin (EC) | 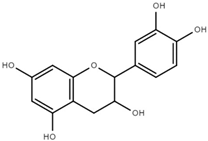 | 3.6 ± 0.5 |
| Epigallocatechin (ECG) | 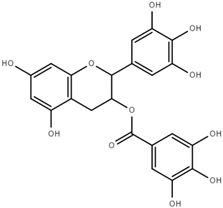 | 2.8 ± 0.5 |
| Gallic acid (GA) | 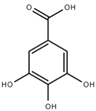 | 1.6 ± 0.2 |
| Epigallocatechin gallate (ECGg) | 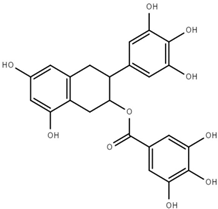 | 1.0 ± 0.2 |
| Catechin (C) | 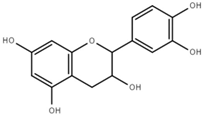 | 0.7 ± 0.1 |
| Catechin gallate (CG) | 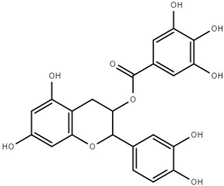 | 0.2 ± 0.1 |
| Total phenolic content | 18.9 ± 2.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Millan-Linares, M.C.; Martin, M.E.; Rodriguez, N.M.; Toscano, R.; Claro, C.; Bermudez, B.; Pedroche, J.; Millan, F.; Montserrat-de la Paz, S. Nutraceutical Extract from Dulse (Palmaria palmata L.) Inhibits Primary Human Neutrophil Activation. Mar. Drugs 2019, 17, 610. https://doi.org/10.3390/md17110610
Millan-Linares MC, Martin ME, Rodriguez NM, Toscano R, Claro C, Bermudez B, Pedroche J, Millan F, Montserrat-de la Paz S. Nutraceutical Extract from Dulse (Palmaria palmata L.) Inhibits Primary Human Neutrophil Activation. Marine Drugs. 2019; 17(11):610. https://doi.org/10.3390/md17110610
Chicago/Turabian StyleMillan-Linares, Maria C, Maria E Martin, Noelia M Rodriguez, Rocio Toscano, Carmen Claro, Beatriz Bermudez, Justo Pedroche, Francisco Millan, and Sergio Montserrat-de la Paz. 2019. "Nutraceutical Extract from Dulse (Palmaria palmata L.) Inhibits Primary Human Neutrophil Activation" Marine Drugs 17, no. 11: 610. https://doi.org/10.3390/md17110610
APA StyleMillan-Linares, M. C., Martin, M. E., Rodriguez, N. M., Toscano, R., Claro, C., Bermudez, B., Pedroche, J., Millan, F., & Montserrat-de la Paz, S. (2019). Nutraceutical Extract from Dulse (Palmaria palmata L.) Inhibits Primary Human Neutrophil Activation. Marine Drugs, 17(11), 610. https://doi.org/10.3390/md17110610







