Protective Effects of Fucoxanthin against Alcoholic Liver Injury by Activation of Nrf2-Mediated Antioxidant Defense and Inhibition of TLR4-Mediated Inflammation
Abstract
1. Introduction
2. Results and Discussion
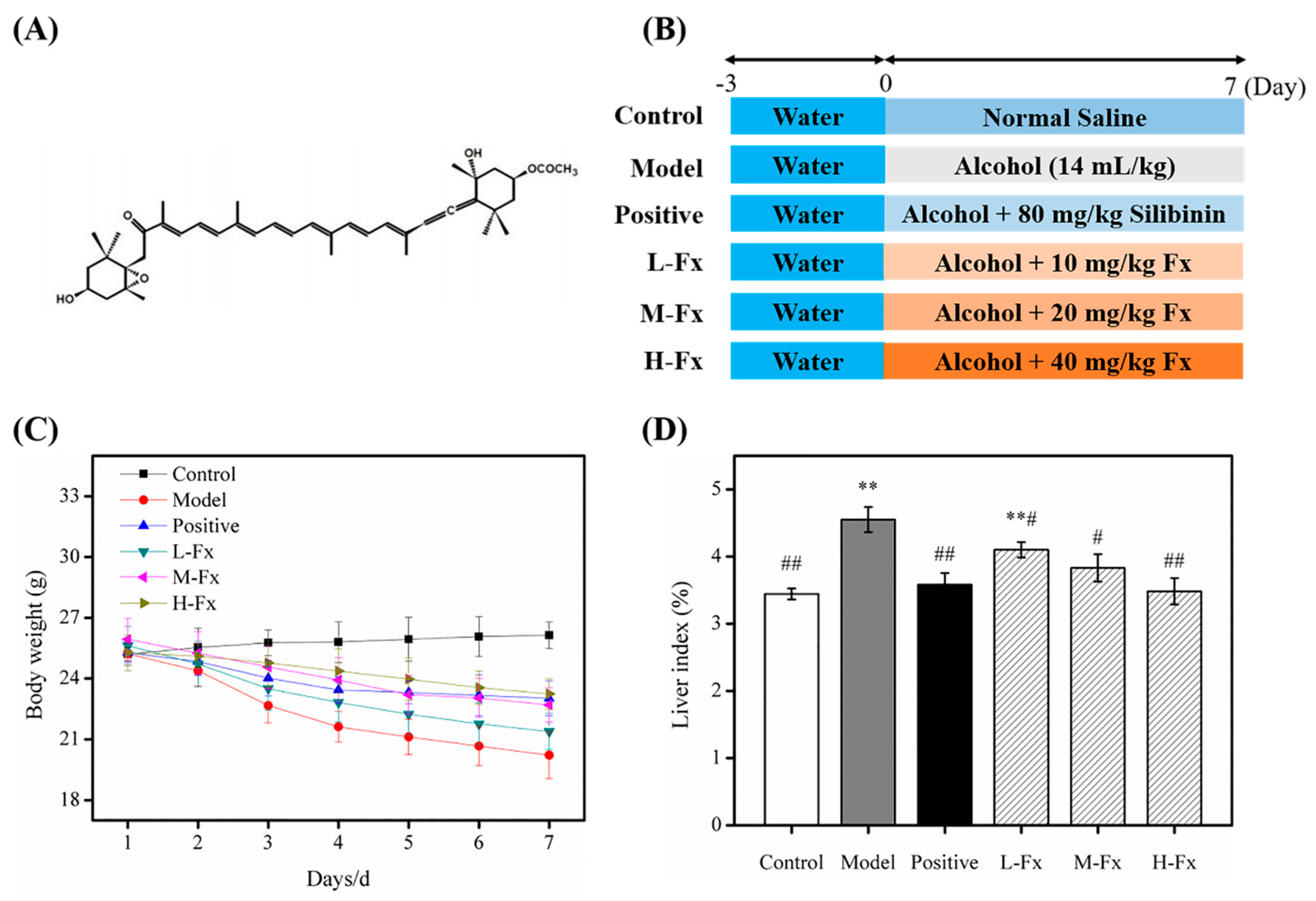
2.1. Effects of Fucoxanthin (Fx) on Bodyweight and Relative Liver Weight
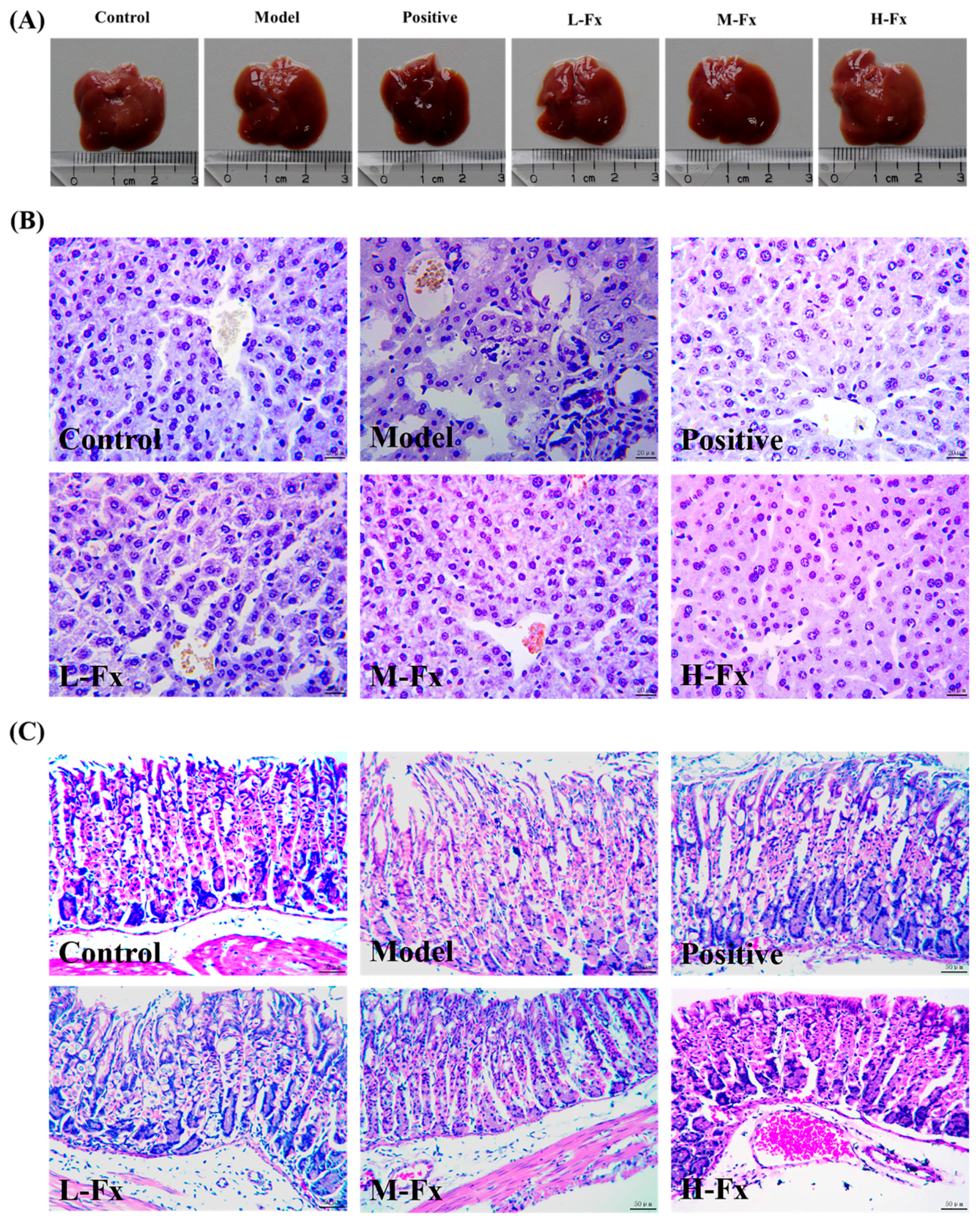
2.2. Effects of Fx on Alcohol-Induced Liver and Stomach Injury
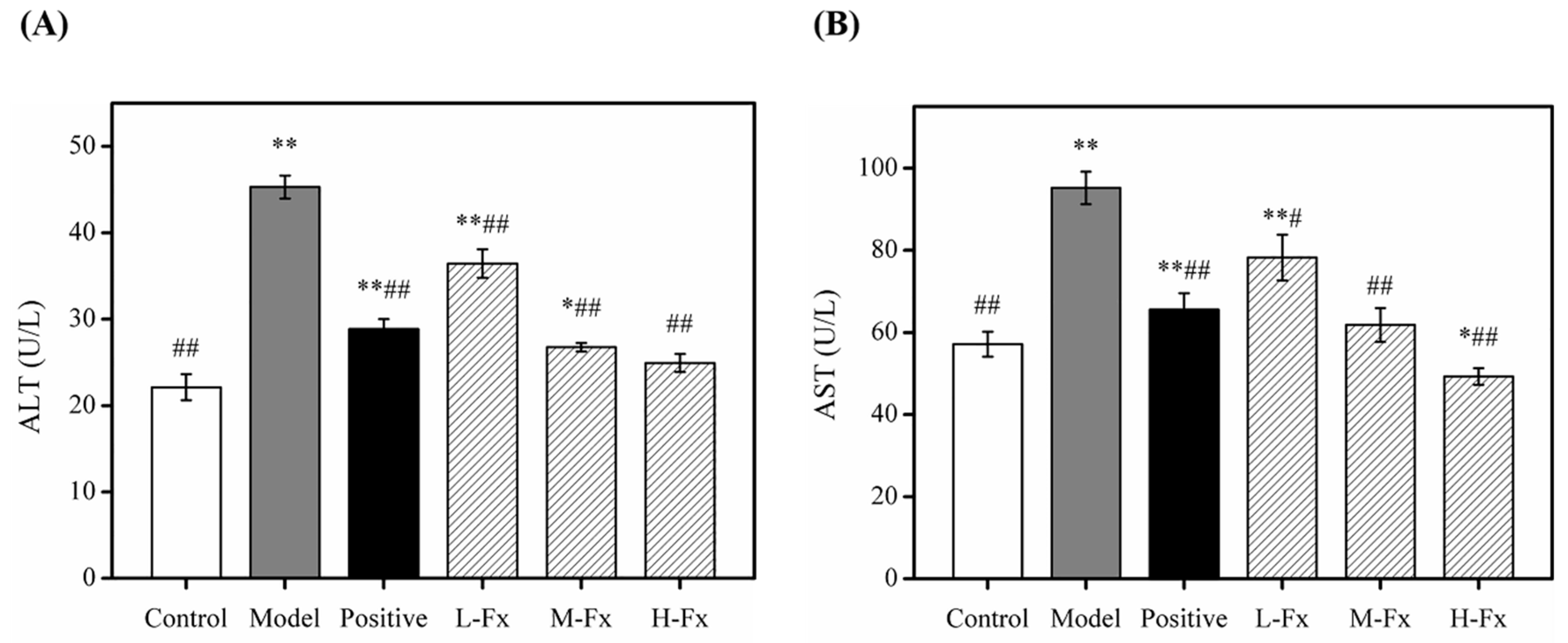
2.3. Effects of Fx on Serum Aspartate Transaminase (AST) and Alanine Transaminase (ALT) Activities
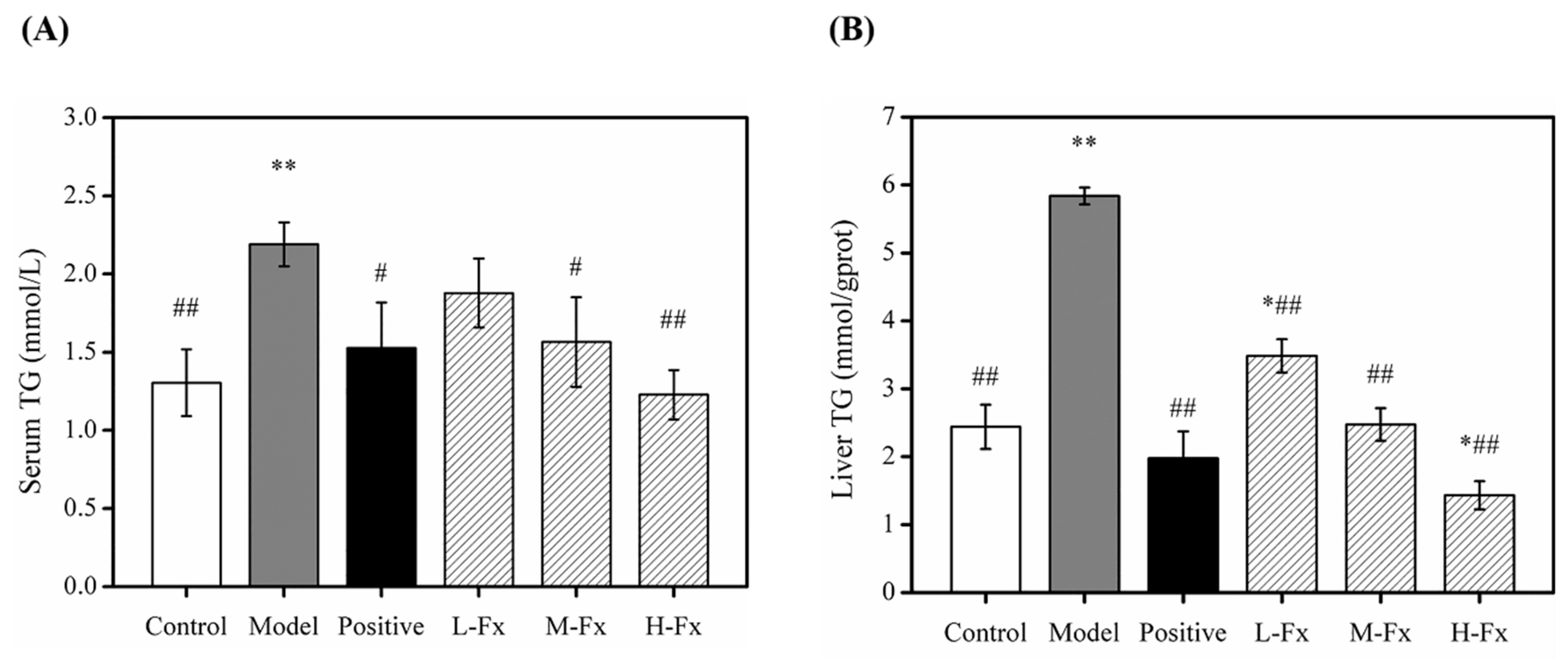
2.4. Effects of Fx on Serum and Hepatic Level of Triglyceride (TG)
2.5. Effects of Fx on Hepatic Levels of Malonaldehyde (MDA) and Antioxidant Enzymes
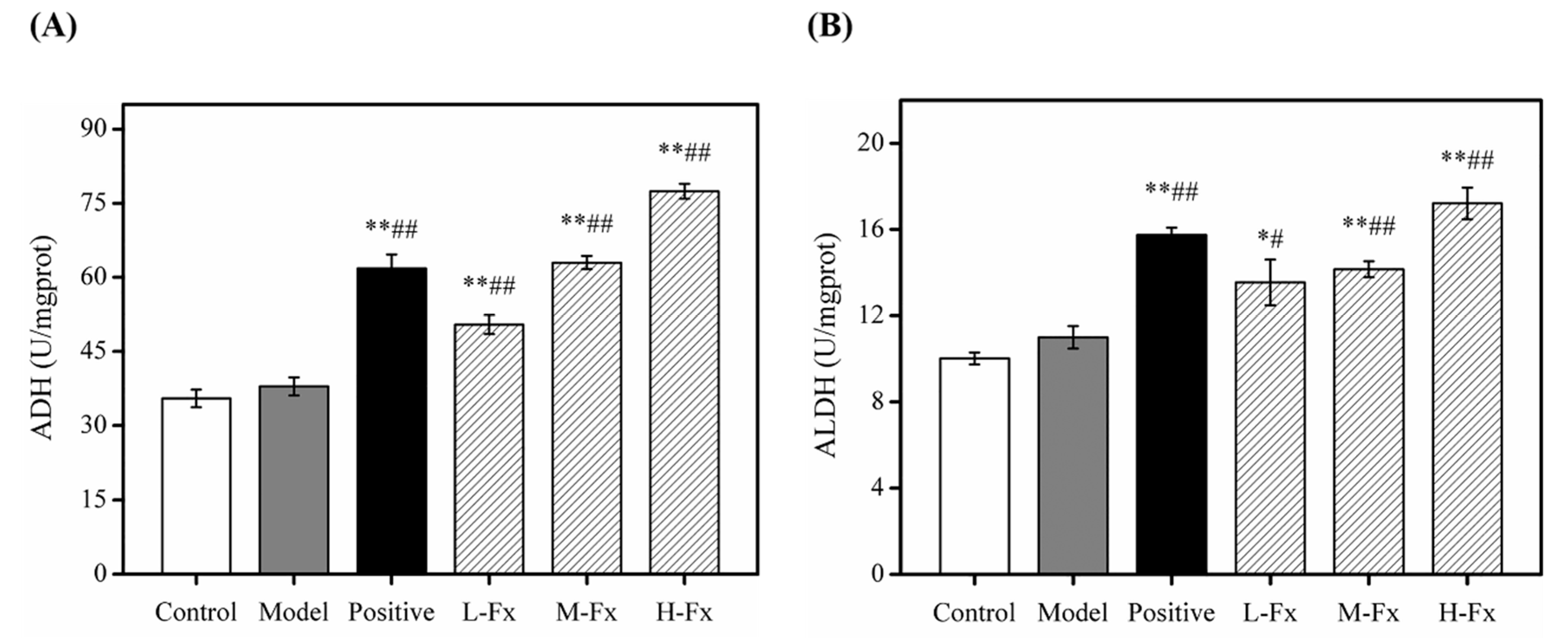
2.6. Effects of Fx on Hepatic Alcohol Dehydrogenase (ADH) and Acetaldehyde Dehydrogenase (ALDH) Activities
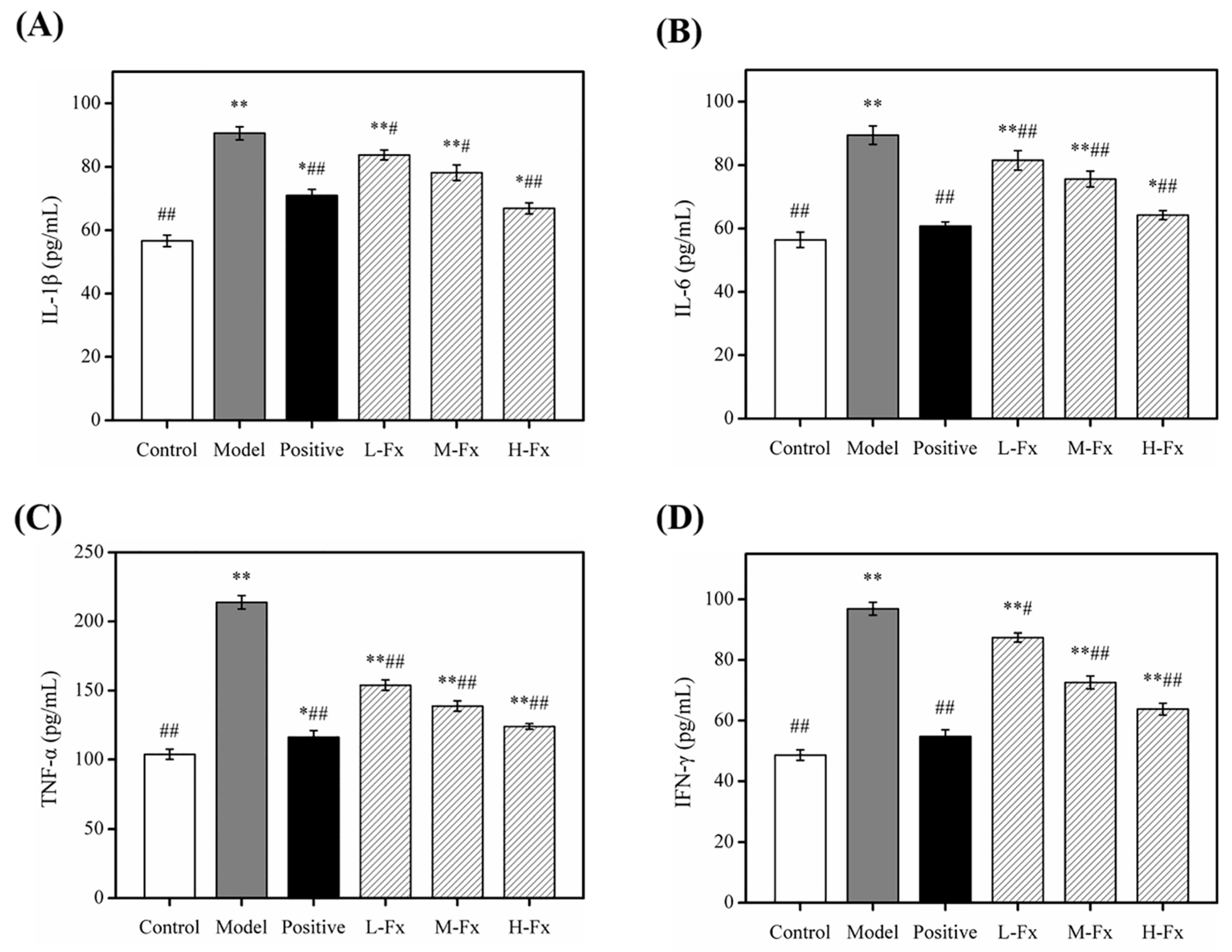
2.7. Effects of Fx on the Levels of Pro-Inflammatory Cytokines in Liver Tissue
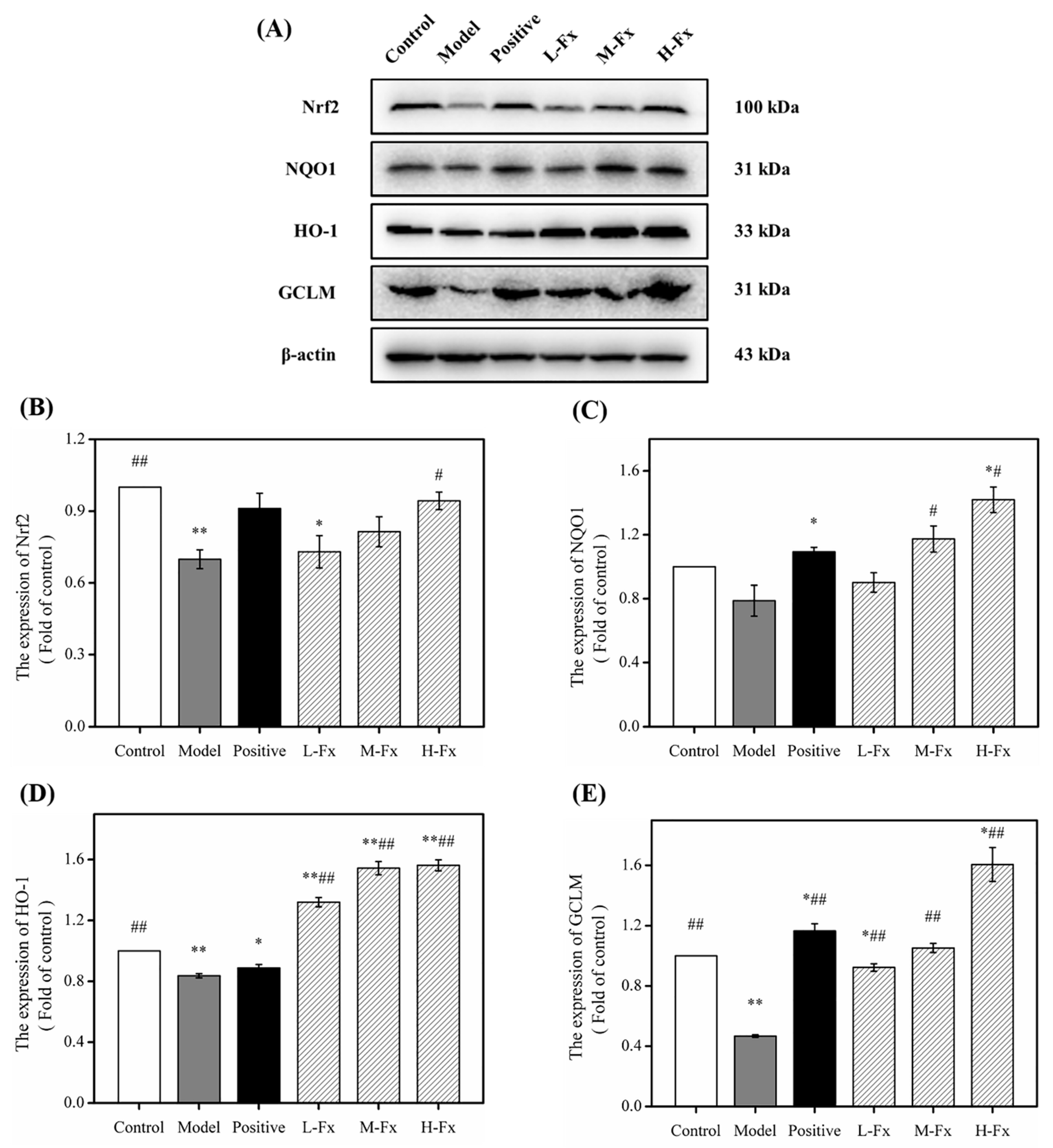
2.8. Effects of Fx on Nuclear Factor Erythrocyte-2-Related Factor 2 (Nrf2)-Mediated Antioxidant Response
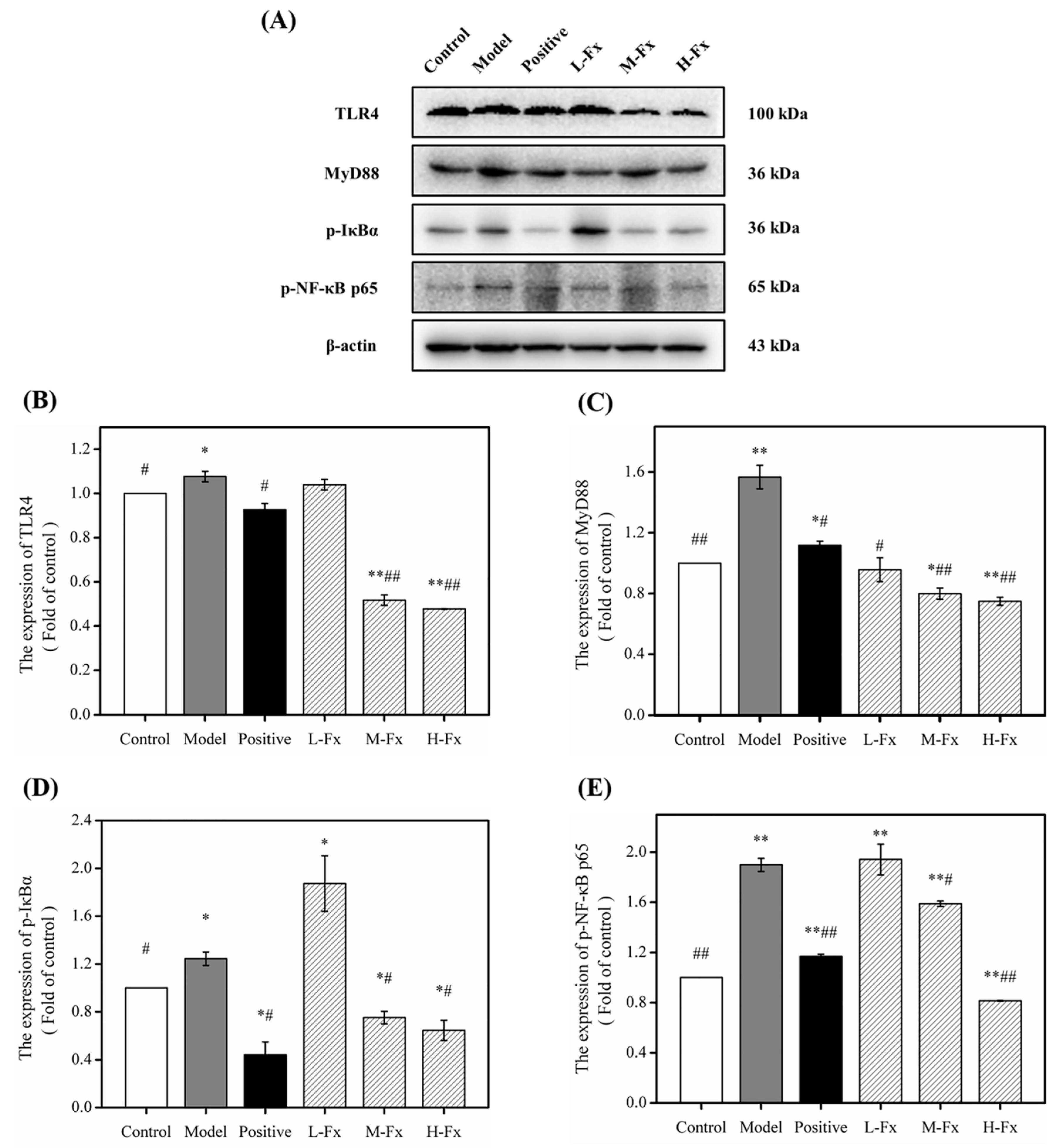
2.9. Effect of Fx on Toll-Like Receptor 4 (TLR4)-Induced Inflammatory Response
3. Materials and Methods
3.1. Materials and Chemicals
3.2. Animals and Treatment
3.3. Histopathological Analysis
3.4. Serum Biochemical Analysis
3.5. Determination of Hepatic MDA and Antioxidant Enzymes
3.6. Determination of Hepatic ADH and ALDH
3.7. Measurement of Pro-Inflammatory Cytokines in Liver
3.8. Western Blot Analysis
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Altamirano, J.; Bataller, R. Alcoholic liver disease: Pathogenesis and new targets for therapy. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Bataller, R. Alcoholic liver disease: Pathogenesis and new therapeutic targets. Gastroenterology 2011, 141, 1572–1585. [Google Scholar] [CrossRef] [PubMed]
- Wruck, W.; Adjaye, J. Meta-analysis reveals up-regulation of cholesterol processes in non-alcoholic and down-regulation in alcoholic fatty liver disease. World J. Hepatol. 2017, 9, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Basra, S.; Anand, B.S. Definition, epidemiology and magnitude of alcoholic hepatitis. World J. Hepatol. 2011, 3, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Dugum, M.; McCullough, A. Diagnosis and management of alcoholic liver disease. J. Dig. Dis. 2015, 82, 226–236. [Google Scholar]
- Pavlov, C.S.; Casazza, G.; Semenistaia, M.; Nikolova, D.; Tsochatzis, E.; Liusina, E.; Ivashkin, V.T.; Gluud, C. Ultrasonography for diagnosis of alcoholic cirrhosis in people with alcoholic liver disease. Cochrane Database Syst. Rev. 2016, 3, CD011602. [Google Scholar] [CrossRef]
- Beier, J.I.; Arteel, G.E.; McClain, C.J. Advances in alcoholic liver disease. Curr. Gastroenterol. Rep. 2011, 13, 56–64. [Google Scholar] [CrossRef]
- Beier, J.I.; McClain, C.J. Mechanisms and cell signaling in alcoholic liver disease. Biol. Chem. 2010, 391, 1249–1264. [Google Scholar] [CrossRef]
- Dunn, W.; Shah, V.H. Pathogenesis of alcoholic liver disease. Clin. Liver Dis. 2016, 20, 445–456. [Google Scholar] [CrossRef]
- Kim, S.J.; Kim, D.G.; Lee, M.D. Effects of branched-chain amino acid infusions on liver regeneration and plasma amino acid patterns in partially hepatectomized rats. Hepatogastroenterology 2011, 58, 1280–1285. [Google Scholar] [CrossRef]
- Leung, T.M.; Nieto, N. CYP2E1 and oxidant stress in alcoholic and non-alcoholic fatty liver disease. J. Hepatol. 2013, 58, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Cederbaum, A.I. CYP2E1 and oxidative liver injury by alcohol. Free Radic. Biol. Med. 2008, 44, 723–738. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.C.; Lin, W.L.; Lee, Y.J.; Tang, Y.C.; Wang, C.J. Polyphenol-rich extract of Nelumbo nucifera leaves inhibits alcohol-induced steatohepatitis via reducing hepatic lipid accumulation and anti-inflammation in C57BL/6J mice. Food Funct. 2014, 5, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Sarumathi, A.; Sethupathy, S.; Saravanan, N. The protective efficacy of spirulina against bacterial endotoxin potentiated alcoholic liver disease. J. Funct. Foods 2014, 9, 254–263. [Google Scholar] [CrossRef]
- Sun, H.; Mu, T.; Liu, X.; Zhang, M.; Chen, J. Purple sweet potato (Ipomoea batatas L.) anthocyanins: Preventive effect on acute and subacute alcoholic liver damage and dealcoholic effect. J. Agric. Food Chem. 2014, 62, 2364–2373. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhang, X.; Liu, F.; Hu, Y.; He, C.; Li, P.; Su, H.; Wan, J. Saponins isolated from the leaves of Panax notoginseng protect against alcoholic liver injury via inhibiting ethanol-induced oxidative stress and gut-derived endotoxin-mediated inflammation. J. Funct. Foods 2015, 19, 214–224. [Google Scholar] [CrossRef]
- Shin, S.M.; Yang, J.H.; Ki, S.H. Role of the Nrf2-ARE pathway in liver diseases. Oxid. Med. Cell. Longev. 2013, 2013, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.J.; Song, D.; Yoo, H.; Chung, K.H.; Lee, K.; An, J. Hederagenin supplementation alleviates the pro-inflammatory and apoptotic response to alcohol in rats. Nutrients 2017, 9, 41. [Google Scholar] [CrossRef]
- Ambade, A.; Mandrekar, P. Oxidative stress and inflammation: Essential partners in alcoholic liver disease. Int. J. Hepatol. 2012, 2012, 853175. [Google Scholar] [CrossRef]
- Kiuru, P.; Auria, M.; Muller, C. Exploring marine resources for bioactive compounds. Planta Med. 2014, 80, 1234–1246. [Google Scholar] [CrossRef]
- Arumugam, V.; Venkatesan, M.; Ramachandran, S.; Sundaresan, U. Bioactive Peptides from Marine Ascidians and Future Drug Development–A Review. Int. J. Pept. Res. Ther. 2017, 24, 13–18. [Google Scholar] [CrossRef]
- Liu, H.; Liu, H.; Zhu, L.; Zhang, Z.; Zheng, X.; Liu, J. Comparative Transcriptome Analyses Provide Potential Insights into the Molecular Mechanisms of Astaxanthin in the Protection against Alcoholic Liver Disease in Mice. Mar. Drugs 2019, 17, 181. [Google Scholar] [CrossRef] [PubMed]
- Ge, N.; Liang, H.; Zhao, Y.; Liu, Y.; Gong, A.; Zhang, W. Aplysin Protects Against Alcohol-Induced Liver Injury Via Alleviating Oxidative Damage and Modulating Endogenous Apoptosis-Related Genes Expression in Rats. J. Food Sci. 2018, 83, 2612–2621. [Google Scholar] [CrossRef] [PubMed]
- D’Orazio, N.; Gammone, M.A.; Gemello, E.; Girolamo, M.D.; Cusenza, S.; Riccioni, G. Marine Bioactives: Pharmacological Properties and Potential Applications against Inflammatory Diseases. Mar. Drugs 2012, 10, 812–833. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Yuan, J.P.; Wu, C.F.; Wang, J.H. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.; Riccioni, G.; D’Orazio, N. Marine carotenoids against oxidative stress: Effects on human health. Mar. Drugs 2015, 13, 6226–6246. [Google Scholar] [CrossRef] [PubMed]
- Nishino, H.; Murakoshi, M.; Tokuda, H.; Satomi, Y. Cancer prevention by carotenoids. Arch. Biochem. Biophys. 2009, 483, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.T.; Tsou, Y.C.; Chen, Y.T.; Lu, W.J.; Hwang, P.A. Effects of low-molecular-weight fucoidan and high stability fucoxanthin on glucose homeostasis, lipid metabolism, and liver function in a mouse model of type II diabetes. Mar. Drugs 2017, 15, 113. [Google Scholar] [CrossRef]
- Liu, H.; Liu, M.; Fu, X.; Zhang, Z.; Zhu, L.; Zheng, X.; Liu, J. Astaxanthin Prevents Alcoholic Fatty Liver Disease by Modulating Mouse Gut Microbiota. Nutrients 2018, 10, 1298. [Google Scholar] [CrossRef]
- Li, X.; Jin, Q.; Zhang, Y.; Wu, Y.L.; Jin, C.M.; Cui, B.W.; Li, Y.; Jin, M.J.; Shang, Y.; Jiang, M. Inhibition of P2 × 7R–NLRP3 Inflammasome Activation by Pleurotus citrinopileatus: A Possible Protective Role in Alcoholic Hepatosteatosis. J. Agric. Food Chem. 2018, 66, 13183–13190. [Google Scholar] [CrossRef]
- Huang, Q.; Zhang, S.; Zheng, L.; Liao, M.; He, M.; Huang, R.; Zhuo, L.; Lin, X. Protective effect of isoorientin-2″-O-α-L-arabinopyranosyl isolated from Gypsophila elegans on alcohol induced hepatic fibrosis in rats. Food Chem. Toxicol. 2012, 50, 1992–2001. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Ju, J.; Lee, Y.; Park, J.; Yeo, I.; Park, M.; Roh, Y.; Han, S.; Hong, J. Astaxanthin alleviated ethanol-induced liver injury by inhibition of oxidative stress and inflammatory responses via blocking of STAT3 activity. Sci. Rep. 2018, 8, 14090–14099. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Ye, P. Research Progress of Alcohol Intake and Dyslipidemia. Adv. Cardiovasc. Dis. 2010, 31, 370–373. [Google Scholar]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 2012, 5, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.M. Regulation and function of AMPK in physiology and diseases. Exp. Mol. Med. 2016, 48, e245. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Wei, R.; Deng, A.; Lei, T. Protective Effects of Ethanolic Extracts from Artichoke, an Edible Herbal Medicine, against Acute Alcohol-Induced Liver Injury in Mice. Nutrients 2017, 9, 1000. [Google Scholar] [CrossRef]
- Yuan, R.; Tao, X.; Liang, S.; Pan, Y.; He, L.; Sun, J.; Wenbo, J.; Li, X.; Chen, J.; Wang, C. Protective effect of acidic polysaccharide from Schisandra chinensis on acute ethanol-induced liver injury through reducing CYP2E1-dependent oxidative stress. Biomed. Pharmacother. 2018, 99, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Qi, L.; Hu, M.; Liu, Y.; Yu, K.; Liu, Q.; Liu, X. Salmonella proteomics under oxidative stress reveals coordinated regulation of antioxidant defense with iron metabolism and bacterial virulence. J. Proteom. 2017, 157, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhu, P.; Jiang, C.; Ma, L.; Zhang, Z.; Zeng, X. Preliminary characterization, antioxidant activity in vitro and hepatoprotective effect on acute alcohol-induced liver injury in mice of polysaccharides from the peduncles of Hovenia dulcis. Food Chem. Toxicol. 2012, 50, 2964–2970. [Google Scholar] [CrossRef]
- Ding, R.; Tian, K.; Cao, Y.; Bao, J.; Wang, M.; He, C.; Hu, Y.; Su, H.; Wan, J. Protective Effect of Panax notoginseng Saponins on Acute Ethanol-Induced Liver Injury Is Associated with Ameliorating Hepatic Lipid Accumulation and Reducing Ethanol-Mediated Oxidative Stress. J. Agric. Food Chem. 2015, 63, 2413–2422. [Google Scholar] [CrossRef]
- Qu, L.; Zhu, Y.; Liu, Y.; Yang, H.; Zhu, C.; Ma, P.; Deng, J.; Fan, D. Protective effects of ginsenoside Rk3 against chronic alcohol-induced liver injury in mice through inhibition of inflammation, oxidative stress, and apoptosis. Food Chem. Toxicol. 2019, 126, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Becker, U.; Grønbaek, M.; Johansen, D.; Sørensen, T.I. Lower risk for alcohol-induced cirrhosis in wine drinkers. Hepatology 2002, 35, 868–875. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Gao, F.; Li, Q.; Lv, J.; Wang, Y.; Hu, P.; Xiang, Q.; Wei, L. Protective Effect of Astragalus polysaccharides on Liver Injury Induced by Several Different Chemotherapeutics in Mice. Asian Pac. J. Cancer Prev. 2014, 15, 10413–10420. [Google Scholar] [CrossRef]
- Tang, C.; Huang, H.; Lee, Y.; Tang, Y.; Wang, C. Hepatoprotective effect of mulberry water extracts on ethanol-induced liver injury via anti-inflammation and inhibition of lipogenesis in C57BL/6J mice. Food Chem. Toxicol. 2013, 62, 786–796. [Google Scholar] [CrossRef] [PubMed]
- Shu, L.; Lei, T.; Chai, G.; Bo, W.; Wang, B. Targeting heme oxygenase-1 by quercetin ameliorates alcohol-induced acute liver injury via inhibiting NLRP3 inflammasome activation. Food Funct. 2018, 9, 4184–4193. [Google Scholar]
- Wang, M.; Shen, G.; Xu, L.; Liu, X.; Brown, J.; Feng, D.; Ross, R.; Gao, B.; Liangpunsakul, S.; Ju, C. IL-1 receptor like 1 protects against alcoholic liver injury by limiting NF-κB activation in hepatic macrophages. J. Hepatol. 2018, 68, S0168827817322638. [Google Scholar] [CrossRef] [PubMed]
- Neyrinck, A.M.; Etxeberria, U.; Taminiau, B.; Daube, G.; Hul, M.V.; Everard, A.; Cani, P.D.; Bindels, L.B.; Delzenne, N.M. Rhubarb extract prevents hepatic inflammation induced by acute alcohol intake, an effect related to the modulation of the gut microbiota. Mol. Nutr. Food Res. 2017, 61, 1500899. [Google Scholar] [CrossRef]
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-Antioxidant Response Element Signaling Pathway and Its Activation by Oxidative Stress. J. Biol. Chem. 2009, 284, 13291–13295. [Google Scholar] [CrossRef]
- Wang, Z.; Dou, X.; Li, S.; Zhang, X.; Sun, X.; Zhou, Z.; Song, Z. Nrf2 activation-induced hepatic VLDLR overexpression in response to oxidative stress contributes to alcoholic liver disease in mice. Hepatology 2014, 59, 1381–1392. [Google Scholar] [CrossRef]
- Klaassen, C.D.; Reisman, S.A. Nrf2 the rescue: Effects of the antioxidative/electrophilic response on the liver. Toxicol. Appl. Pharmacol. 2010, 244, 57–65. [Google Scholar] [CrossRef]
- Nioi, P.; Mcmahon, M.; Itoh, K.; Yamamoto, M.; Hayes, J.D. Identification of a novel Nrf2-regulated antioxidant response element (ARE) in the mouse NAD(P)H:quinone oxidoreductase 1 gene: Reassessment of the ARE consensus sequence. Biochem. J. 2003, 374, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Xia, T.; Yao, J.; Jin, Z.; Yu, Z.; Jia, S.; Min, W. Protective effects of Shanxi aged vinegar against hydrogen peroxide-induced oxidative damage in LO2 cells through Nrf2-mediated antioxidant responses. RSC Adv. 2017, 7, 17377–17386. [Google Scholar] [CrossRef]
- Mandrekar, P.; Szabo, G. Signalling pathways in alcohol-induced liver inflammation. J. Hepatol. 2009, 50, 1258–1266. [Google Scholar] [CrossRef] [PubMed]
- Hritz, I.; Velayudham, A.; Dolganiuc, A.; Kodys, K.; Mandrekar, P.; Kurt-Jones, E.; Szabo, G. Bone marrow-derived immune cells mediate sensitization to liver injury in a myeloid differentiation factor 88-dependent fashion. Hepatology 2008, 48, 1342–1347. [Google Scholar] [CrossRef] [PubMed]
- Petrasek, J.; Mandrekar, P.; Szabo, G. Toll-Like Receptors in the Pathogenesis of Alcoholic Liver Disease. Gastroenterol. Res. Pract. 2010, 2010, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhuang, T.; Si, Y.; Qi, K.; Zhao, J. Coriolus versicolor mushroom polysaccharides exert immunoregulatory effects on mouse B cells via membrane Ig and TLR-4 to activate the MAPK and NF-κB signaling pathways. Mol. Immunol. 2015, 64, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Tai, S.; Chen, J.; Chou, C.; Fu, S.; Chen, Y. Ameliorative effects of pepsin-digested chicken liver hydrolysates on development of alcoholic fatty livers in mice. Food Funct. 2017, 8, 1763–1771. [Google Scholar] [CrossRef] [PubMed]
- Hong, K.S.; Yun, S.M.; Cho, J.M.; Lee, D.Y.; Ji, S.D.; Son, J.-G.; Kim, E.H. Silkworm (Bombyx mori) powder supplementation alleviates alcoholic fatty liver disease in rats. J. Funct. Foods 2018, 43, 29–36. [Google Scholar] [CrossRef]
- Tang, Y.; Yu, F.; Zhang, G.; Yang, Z.; Huang, F.; Ding, G. A purified serine protease from Nereis virens and its impaction of apoptosis on human lung cancer cells. Molecules 2017, 22, 1123. [Google Scholar] [CrossRef]
| Group | T-AOC (U/mg prot) | MDA (nmol/mg prot) | SOD (U/mg prot) | GSH-Px (U/mg prot) | CAT (U/mg prot) |
|---|---|---|---|---|---|
| Control | 5.72 ± 0.05 ## | 3.07 ± 0.21 ## | 168.52 ± 8.73 ## | 129.57 ± 5.03 ## | 89.61 ± 2.13 ## |
| Model | 1.32 ± 0.12 ** | 5.90 ± 0.46 ** | 88.05 ± 14.71 ** | 69.60 ± 4.41 ** | 64.11 ± 2.54 ** |
| Positive | 9.34 ± 0.52 ** ## | 3.20 ± 0.22 ## | 156.57 ± 3.81 ## | 129.28 ± 6.87 ## | 96.93 ± 2.14 * ## |
| L-Fx | 3.39 ± 0.17 ** ## | 4.14 ± 0.22 ** ## | 121.36 ± 6.23 ** # | 86.69 ± 3.43 ** ## | 85.60 ± 2.09 ## |
| M-Fx | 5.22 ± 0.23 * ## | 3.79 ± 0.30 ## | 152.34 ± 8.24 ## | 100.75 ± 6.89 ** ## | 105.41 ± 1.29 ** ## |
| H-Fx | 9.64 ± 0.14 ** ## | 2.91 ± 0.26 ## | 175.74 ± 12.68 ## | 128.79 ± 7.73 ## | 134.00 ± 1.36 ** ## |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, J.; Tian, X.; Zhang, W.; Zheng, P.; Huang, F.; Ding, G.; Yang, Z. Protective Effects of Fucoxanthin against Alcoholic Liver Injury by Activation of Nrf2-Mediated Antioxidant Defense and Inhibition of TLR4-Mediated Inflammation. Mar. Drugs 2019, 17, 552. https://doi.org/10.3390/md17100552
Zheng J, Tian X, Zhang W, Zheng P, Huang F, Ding G, Yang Z. Protective Effects of Fucoxanthin against Alcoholic Liver Injury by Activation of Nrf2-Mediated Antioxidant Defense and Inhibition of TLR4-Mediated Inflammation. Marine Drugs. 2019; 17(10):552. https://doi.org/10.3390/md17100552
Chicago/Turabian StyleZheng, Jiawen, Xiaoxiao Tian, Wen Zhang, Pingan Zheng, Fangfang Huang, Guofang Ding, and Zuisu Yang. 2019. "Protective Effects of Fucoxanthin against Alcoholic Liver Injury by Activation of Nrf2-Mediated Antioxidant Defense and Inhibition of TLR4-Mediated Inflammation" Marine Drugs 17, no. 10: 552. https://doi.org/10.3390/md17100552
APA StyleZheng, J., Tian, X., Zhang, W., Zheng, P., Huang, F., Ding, G., & Yang, Z. (2019). Protective Effects of Fucoxanthin against Alcoholic Liver Injury by Activation of Nrf2-Mediated Antioxidant Defense and Inhibition of TLR4-Mediated Inflammation. Marine Drugs, 17(10), 552. https://doi.org/10.3390/md17100552




