Evaluation of the Immunomodulatory Effects of Fucoidan Derived from Cladosiphon Okamuranus Tokida in Mice
Abstract
1. Introduction
2. Results
2.1. Proliferative Activity of Splenic Immune Cells
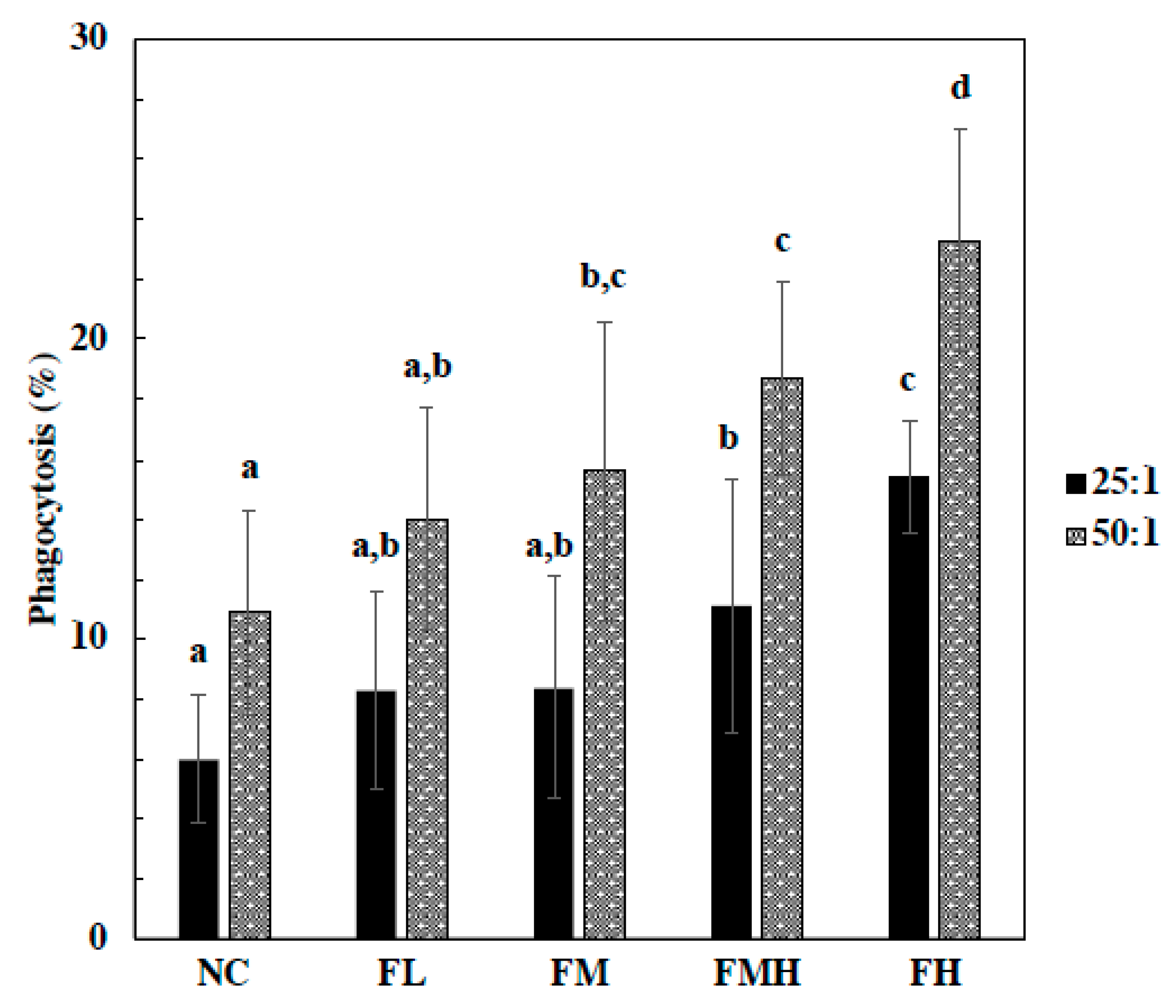
2.2. Effect of Fucoidan on Phagocytes Activity
2.3. Effect of Fucoidan on Cytokines Production
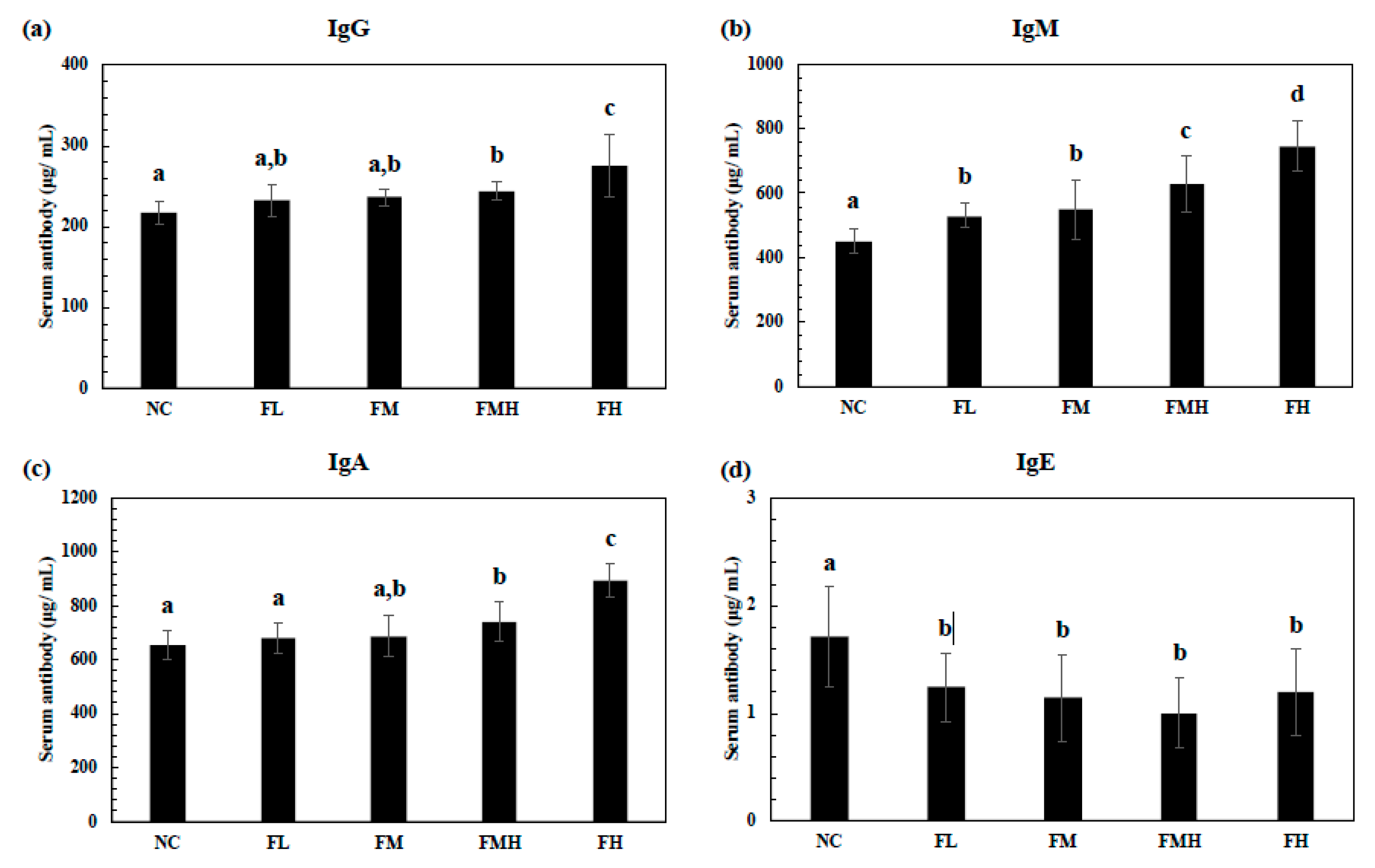
2.4. Effect of Fucoidan on Serum Antibody Production
3. Discussion
4. Materials and Methods
4.1. Fucoidan
4.2. Animals and Treatments
4.3. Preparation of Splenocytes Suspension, Peritoneal Macrophages, and Serum
4.4. Immune Cell Proliferation
4.5. Phagocytic Activity
4.6. Cytokine Production
4.7. Determination of Serum Immunoglobulin
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Blilan, M.I.; et al. Consorzio Interuniversitario Nazionale per la Bio-Oncologia, Italy. A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, S.; Nagaoka, M.; Hara, T.; Kimura-Takagi, I.; Mitsuyama, K.; Ueyama, S. Fucoidan derived from Cladosiphon okamuranus Tokita ameliorates murine chronic colitis through the down-regulation of interleukin-6 production on colonic epithelial cells. Clin. Exp. Immunol. 2004, 136, 432–439. [Google Scholar] [CrossRef] [PubMed]
- McClure, M.O.; Whitby, D.; Patience, N.; Gooderham, N.J.; Bradshaw, A.; Popov, R.C.; Weber, N.; Davies, D.S.; Cook, G.M.W.; Keynes, R.J.; et al. Dextrin sulfate and fucoidan are potent inhibitors of HIV infection in vitro. Antivir. Chem. Chemother. 1991, 2, 149–156. [Google Scholar] [CrossRef]
- Atashrazm, F.; Lowenthal, R.M.; Woods, G.M.; Holloway, A.F.; Dickinson, J.L. Fucoidan and cancer: A multifunctional molecule with anti-tumor potential. Mar. Dugs 2015, 13, 2327–2346. [Google Scholar] [CrossRef] [PubMed]
- Nagamine, T.; Hayakawa, K.; Kusakabe, T.; Takada, H.; Nakazato, K.; Hisanaga, E.; Iha, M. Inhibitory effect of fucoidan on Huh7 hepatoma cells through downregulation of CXCL12. Nutr. Cancer 2009, 61, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Ikeguchi, M.; Saito, H.; Miki, Y.; Kimura, T. Effect of fucoidan dietary supplement on the chemotherapy treatment of patients with unresectable advanced gastric cancer. Oncol. Lett. 2011, 2, 319–322. [Google Scholar] [CrossRef]
- Azuma, K.; Ishihara, T.; Nakamoto, H.; Amaha, T.; Osaki, T.; Tsuka, T.; Imagawa, T.; Minami, S.; Takashima, O.; Ifuku, S.; et al. Effect of oral administration of fucoidan extracted from Cladosiphon okamuranus on tumor growth and survival time in a tumor-bearing mouse model. Mar. Drugs 2012, 10, 2337–2348. [Google Scholar] [CrossRef]
- Ohnogi, H.; Naito, Y.; Higashimaru, Y.; Uno, K.; Yoshikawa, T. Immune efficacy and safety of fucoidan extracted from gagome kombu (Kjellmaniella crassifolia) in healthy japanese subjects. Jpn. J. Complement. Altern. Med. 2015, 12, 87–93. [Google Scholar] [CrossRef][Green Version]
- Murayama, H.; Tamauchi, H.; Hashimoto, M.; Nakano, T. Suppression of Th2 immune responses by mekabu fucoidan from Undaria pinnatifida sporophylls. Int. Arch. Allergy Immunol. 2005, 137, 289–294. [Google Scholar] [CrossRef]
- Nishitsuji, K.; Arimoto, A.; Iwai, K.; Sudo, Y.; Hisata, K.; Fujie, M.; Arakaki, N.; Kushiro, T.; Konishi, T.; Shinzato, C.; et al. A draft genome of the brown alga, Cladosiphon okamuranus, S-strain: A platform for future studies of ‘mozuku’ biology. DNA Res. 2016, 23, 561–570. [Google Scholar] [CrossRef]
- Nagaoka, M.; Shibata, H.; Kimura-Takagi, I.; Hashimoto, S.; Kimura, K.; Makino, T.; Aiyama, R.; Ueyama, S.; Yokokura, T. Structure study of fucoidan from Cladosiphon okamuranus Tokida. Glycoconj. J. 2009, 16, 19–26. [Google Scholar] [CrossRef]
- Shibata, H.; Kimura-Takagi, I.; Nagaoka, M.; Hashimoto, S.; Sawada, H.; Ueyama, S.; Yokokura, T. Inhibitor effect of Cladosiphon fucoidan on the adhesion of Helicobacter pylori to human gastric cells. J. Nutr. Sci. Vitaminol. 1999, 45, 325–336. [Google Scholar] [CrossRef]
- Shibata, H.; Iimuro, M.; Uchiya, N.; Kawamori, T.; Nagaoka, M.; Ueyama, S.; Hashimoto, S.; Yokokura, T.; Sugimura, T.; Wakabayashi, K. Prevent effects of Cladosiphon fucoidan against Helicobacter pylori infect in Mongolian gerbils. Helicobacter 2003, 8, 59–65. [Google Scholar] [CrossRef]
- Nagaoka, M.; Shibata, H.; Kimura-Takagi, I.; Hashimoto, S.; Aiyama, R.; Ueyama, S.; Yokokura, T. Anti-ulcer effects and biological activities of polysaccharides from marine algae. Biofactors 2000, 12, 267–274. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Suzuki, T.; Hirano, M.; Nagaoka, M.; Hashimoto, S.; Shibata, H.; Kimura-Takagi, I.; Ueyama, S.; Kudo, T.; Yoshikawa, M.; et al. Effect of fucoidan and fucoidan containing tea on gastric ulcer and non-ulcer dyspepsia. Jpn. Pharmacol. Ther. 2000, 28, 63–70. (In Japanese) [Google Scholar]
- Chen, Y.M.; Tsai, Y.H.; Tsai, T.Y.; Chiu, Y.S.; Wei, L.; Chen, W.C.; Huang, C.C. Fucoidan supplementation improves exercise performance and exhibits anti-fatigue action in mice. Nutrients 2015, 7, 239–252. [Google Scholar] [CrossRef]
- Abe, S.; Hiramatsu, K.; Ichikawa, O.; Ikeda, T.; Kawamoto, H.; Kasagi, T.; Miki, Y. Effects of mozuku fucoidan on defecation and fecal condition in human. J. Yonago Med. Assoc. 2010, 61, 122–128. (In Japanese) [Google Scholar]
- Tomori, M.; Matsuda, K.; Nakamura, Y.; Kadena, K.; Shimoji, S.; Nagamine, T.; Iha, M. Effect of Cladosiphon okamuranus fucoidan containing drink on improvement of defecation in healthy subjects with mild constipation. Jpn. Pharmacol. Ther. 2016, 44, 1621–1626. (In Japanese) [Google Scholar]
- Haneji, K.; Matsuda, T.; Tomita, M.; Kawakami, H.; Ohshiro, K.; Uchihara, J.N.; Masuda, M.; Takasu, N.; Tanaka, Y.; Ohta, T.; et al. Fucoidan extracted from cladosiphon okamuranus Tokida induces apoptosis of human T-cell leukemia virus type 1-–infected T-cell lines and primary adult T-cell leukemia cells. Nutr. Cancer 2005, 52, 189–201. [Google Scholar] [CrossRef]
- Araya, N.; Takahashi, K.; Sato, T.; Nakamura, T.; Sawa, C.; Hasegawa, D.; Ando, H.; Aratani, S.; Yagishita, N.; Fujii, R.; et al. Fucoidan therapy decreases the proviral load in patients with human T-lymphotropic virus type-I-associated neurological disease. Antivir. Ther. 2011, 16, 89–98. [Google Scholar] [CrossRef]
- Irhimeh, M.R.; Fitton, J.H.; Lowenthal, R.M.; Kongtawelert, P. A quantitative method to detec fucoidan in human plasma using a novel antibody. Methods Find. Exp. Clin. Pharmacol. 2005, 27, 705–710. [Google Scholar] [CrossRef]
- Tokita, Y.; Nakajima, K.; Mochida, H.; Iha, M.; Nagamine, T. Development of fucoidan-specific antibody and measurement of fucoidan in serum and urine by sandwich ELISA. Biosci. Biotechnol. Biochem. 2010, 74, 350–357. [Google Scholar] [CrossRef]
- Murayama, H.; Tamauchi, H.; Iizuka, M.; Nakano, T. The role of NK cells in antitumor activity of dietary fucoidan from Undaria pinnatifida Sporophylls (Mekabu). Planta Med. 2006, 72, 1415–1417. [Google Scholar] [CrossRef]
- Li, B.; Lu, F.; Wei, X.; Zhao, R. Fucoidan: Structure and bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar] [CrossRef]
- Nagamine, T.; Nakazato, K.; Tomioka, S.; Iha, M.; Nakajima, K. Intestinal absorption of fucoidan extracted from the brown seaweed, Cladosiphon okamuranus. Mar. Drugs 2015, 13, 48–64. [Google Scholar] [CrossRef]
- Tokita, Y.; Hirayama, M.; Nakajima, K.; Tamaki, K.; Iha, M.; Nagamine, T. Detection of fucoidan in urine after oral intake of traditional japanese seaweed, Okinawa mozuku (Cladosiphon okamuranus Tokida). J. Nutr. Sci. Vitaminol. 2017, 63, 419–421. [Google Scholar] [CrossRef]
- Kadena, K.; Tomori, M.; Iha, M.; Nagamine, T. Absorption study of mozuku fucoidan in japanese volunteers. Mar. Drugs 2018, 16, 254. [Google Scholar] [CrossRef]
- Shimizu, J.; Wada-Funada, U.; Mano, H.; Matahira, Y.; Kawaguchi, M.; Wada, M. Proportion of murine cytotoxic T cells is increased by high molecular-weight fucoidan extracted from okinawa mozuku (Cladosipohn okamuranus). J. Health Sci. 2005, 51, 394–397. [Google Scholar] [CrossRef]
- Jang, J.Y.; Moon, S.Y.; Joo, H.G. Differential effects of fucoidans with low and high molecular weight on the viability and function of spleen cells. Food Chem. Toxicol. 2014, 68, 234–238. [Google Scholar] [CrossRef]
- Doi, T.; Higashino, K.; Kurihara, Y.; Wada, Y.; Miyazaki, T.; Nakamura, H.; Uesugi, S.; Imanishi, T.; Kawabe, Y.; Itakura, H.; et al. Charge collagen structure mediate recognition of negatively charged macromolecules by macrophage scavenger receptors. J. Biol. Chem. 1993, 268, 2126–2133. [Google Scholar]
- Takai, M.; Miyazaki, Y.; Tachibana, H.; Yamada, K. The enhancing effect of fucoidan derived from Undaria pinnatifida on immunoglobulin production by mouse spleen lymphocytes. Biosci. Biotechnol. Biochem. 2014, 78, 1743–1747. [Google Scholar] [CrossRef]
| Stimulation Index (S.I.) | |||
|---|---|---|---|
| Group | Dose (mg/kg) | Con A (5.0 μg/mL) | LPS (10.0 μg/mL) |
| NC | - | 2.59 ± 0.26 a | 2.10 ± 0.14 a |
| FL | 102.5 | 2.94 ± 0.29 b | 2.42 ± 0.15 b |
| FM | 205.0 | 3.16 ± 0.21 b,c | 2.45 ± 0.19 b |
| FMH | 410.0 | 3.40 ± 0.28 c,d | 2.74 ± 0.28 c |
| FH | 1025.0 | 3.54 ± 0.39 d | 2.82 ± 0.23 c |
| Group | Dose (mg/kg) | Con A (5.0 μg/ mL) | LPS (10.0 μg/ mL) |
|---|---|---|---|
| IL-2 (pg/mL) | |||
| NC | - | 2625.1 ± 526.1 a | 34.1 ± 5.6 a |
| FL | 102.5 | 2944.2 ± 874.0 a,b | 38.9 ± 6.8 a,b |
| FM | 205.0 | 3184.5 ± 519.1 a,b | 39.3 ± 3.5 a,b |
| FMH | 410.0 | 3223.4 ± 539.6 b | 39.9 ± 5.6 a,b |
| FH | 1025.0 | 3249.2 ± 452.4 b | 41.9 ± 10.1 b |
| IL-4 (pg/mL) | |||
| NC | - | 51.9 ± 11.2 a | 14.3 ± 2.8 a |
| FL | 102.5 | 43.9 ± 13.2 a | 10.6 ± 0.9 b |
| FM | 205.0 | 41.4 ± 15.0 a,b | 10.7 ± 1.7 b |
| FMH | 410.0 | 42.1 ± 14.7 a,b | 10.2 ± 1.2 b |
| FH | 1025.0 | 31.2 ± 8.8 b | 9.7 ± 0.8 b |
| IL-5 (pg/mL) | |||
| NC | - | 121.9 ± 41.3 a | 10.0 ± 1.8 a |
| FL | 102.5 | 78.9 ± 14.5 b | 7.6 ± 1.0 b |
| FM | 205.0 | 71.6 ± 17.6 b | 7.4 ± 1.0 b |
| FMH | 410.0 | 68.4 ± 15.7 b | 7.1 ± 0.7 b |
| FH | 1025.0 | 65.8 ± 19.2 b | 7.0 ± 0.8 b |
| IFN-γ (ng/mL) | |||
| NC | - | 19.3 ± 4.8 a | 4.0 ± 0.9 a |
| FL | 102.5 | 20.4 ± 5.3 a | 5.1 ± 1.3 a,b |
| FM | 205.0 | 24.3 ± 6.8 a | 5.9 ± 2.4 b |
| FMH | 410.0 | 24.4 ± 7.7 a | 6.6 ± 1.9 b |
| FH | 1025.0 | 24.5 ± 5.2 a | 8.8 ± 2.8 c |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomori, M.; Nagamine, T.; Miyamoto, T.; Iha, M. Evaluation of the Immunomodulatory Effects of Fucoidan Derived from Cladosiphon Okamuranus Tokida in Mice. Mar. Drugs 2019, 17, 547. https://doi.org/10.3390/md17100547
Tomori M, Nagamine T, Miyamoto T, Iha M. Evaluation of the Immunomodulatory Effects of Fucoidan Derived from Cladosiphon Okamuranus Tokida in Mice. Marine Drugs. 2019; 17(10):547. https://doi.org/10.3390/md17100547
Chicago/Turabian StyleTomori, Makoto, Takeaki Nagamine, Tomofumi Miyamoto, and Masahiko Iha. 2019. "Evaluation of the Immunomodulatory Effects of Fucoidan Derived from Cladosiphon Okamuranus Tokida in Mice" Marine Drugs 17, no. 10: 547. https://doi.org/10.3390/md17100547
APA StyleTomori, M., Nagamine, T., Miyamoto, T., & Iha, M. (2019). Evaluation of the Immunomodulatory Effects of Fucoidan Derived from Cladosiphon Okamuranus Tokida in Mice. Marine Drugs, 17(10), 547. https://doi.org/10.3390/md17100547





