Liposomal Form of the Echinochrome-Carrageenan Complex
Abstract
1. Introduction
2. Results
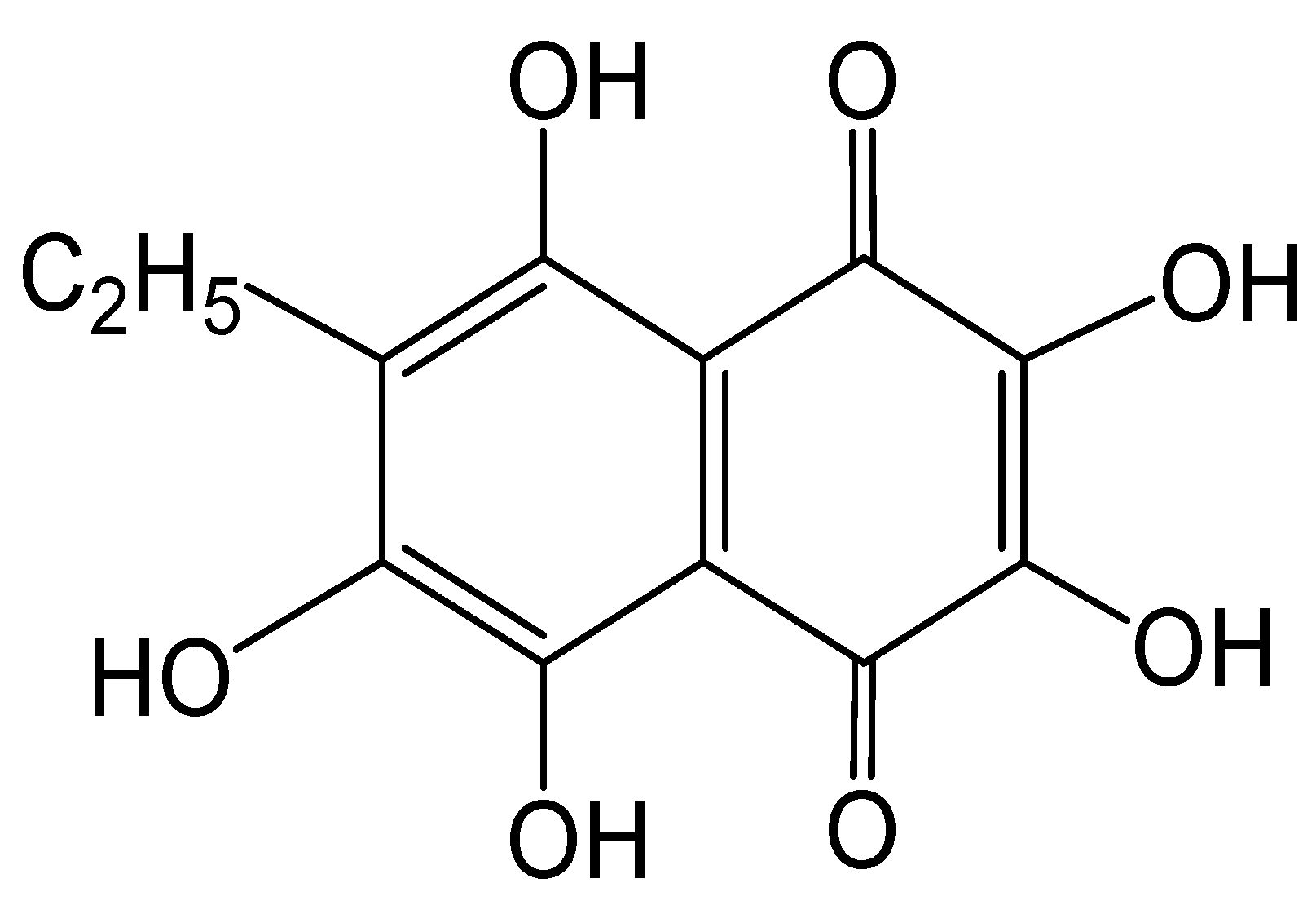
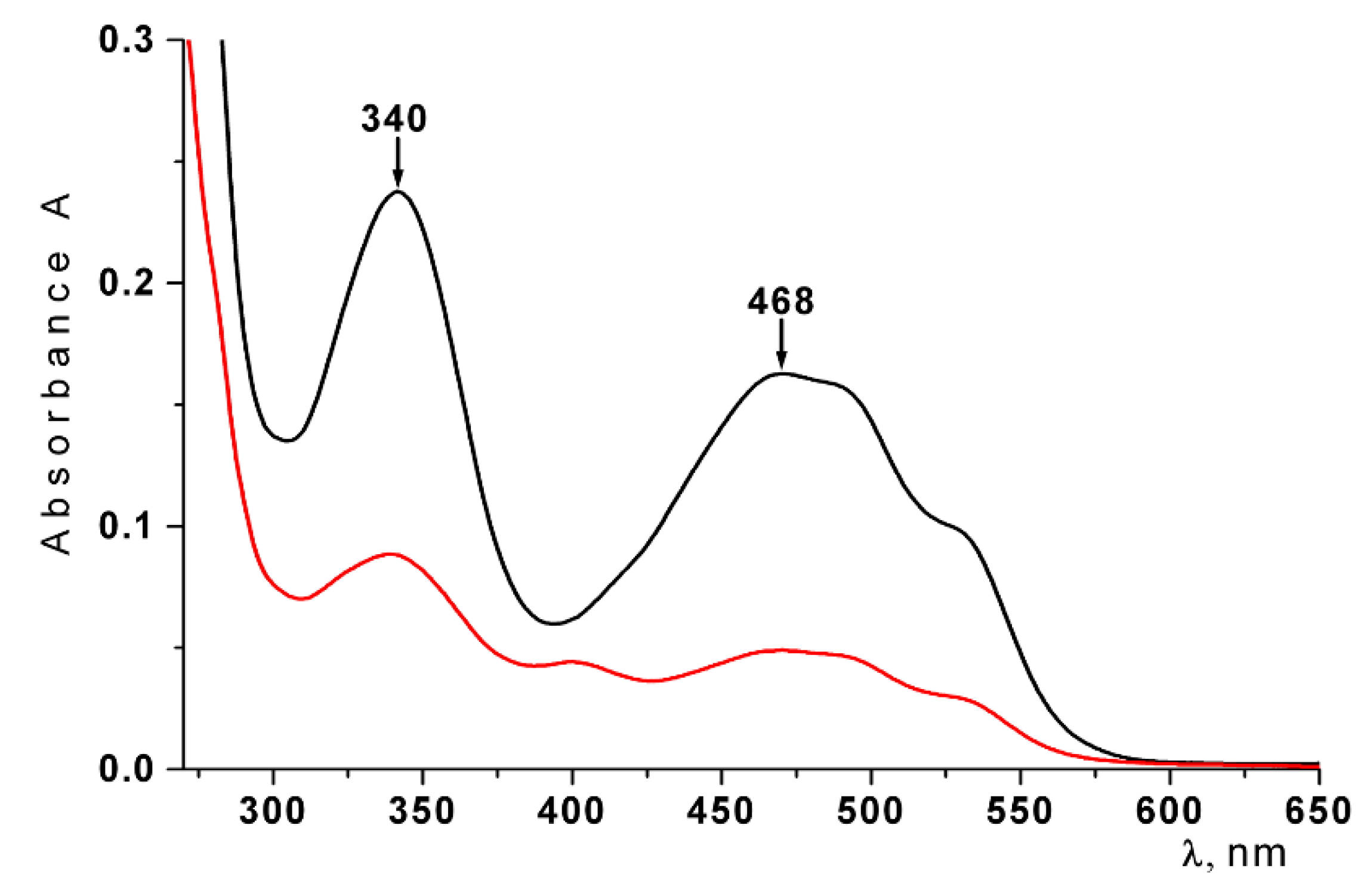
2.1. Characterization of CRG and Ech
2.2. Preparation and Characterization of Conventional Liposomes
2.3. The Entrapment Efficiency of Ech in Liposomes
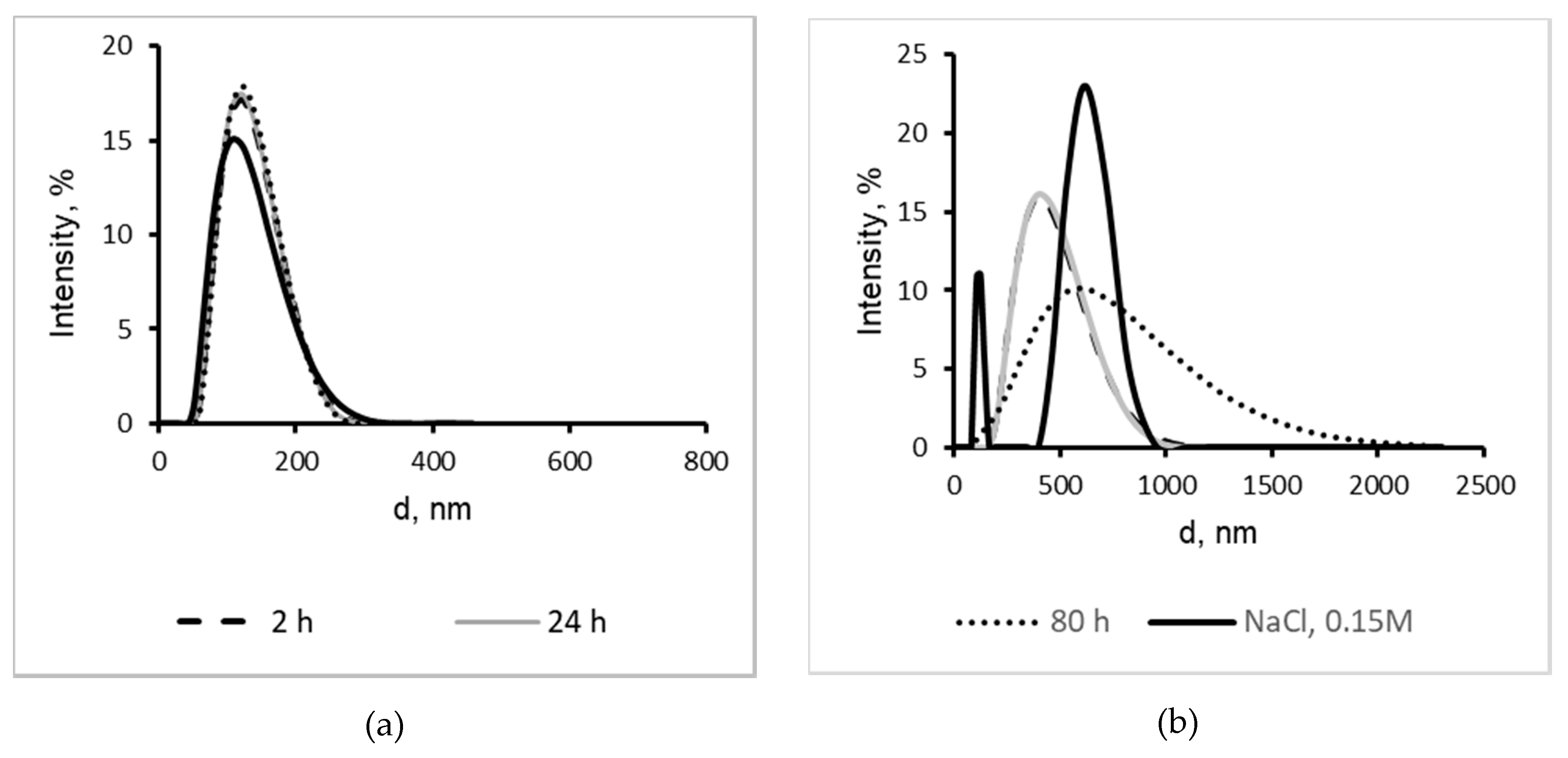
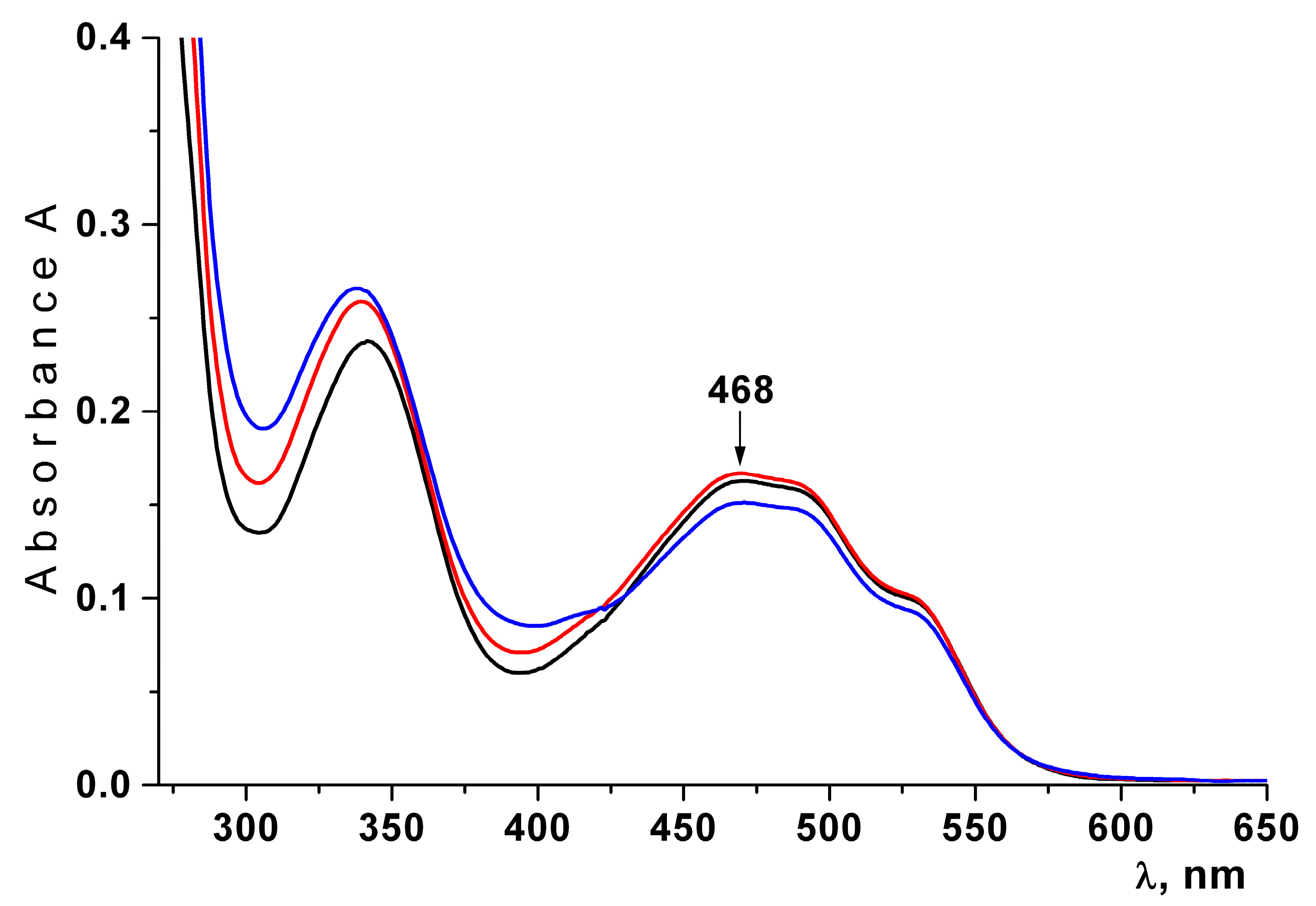
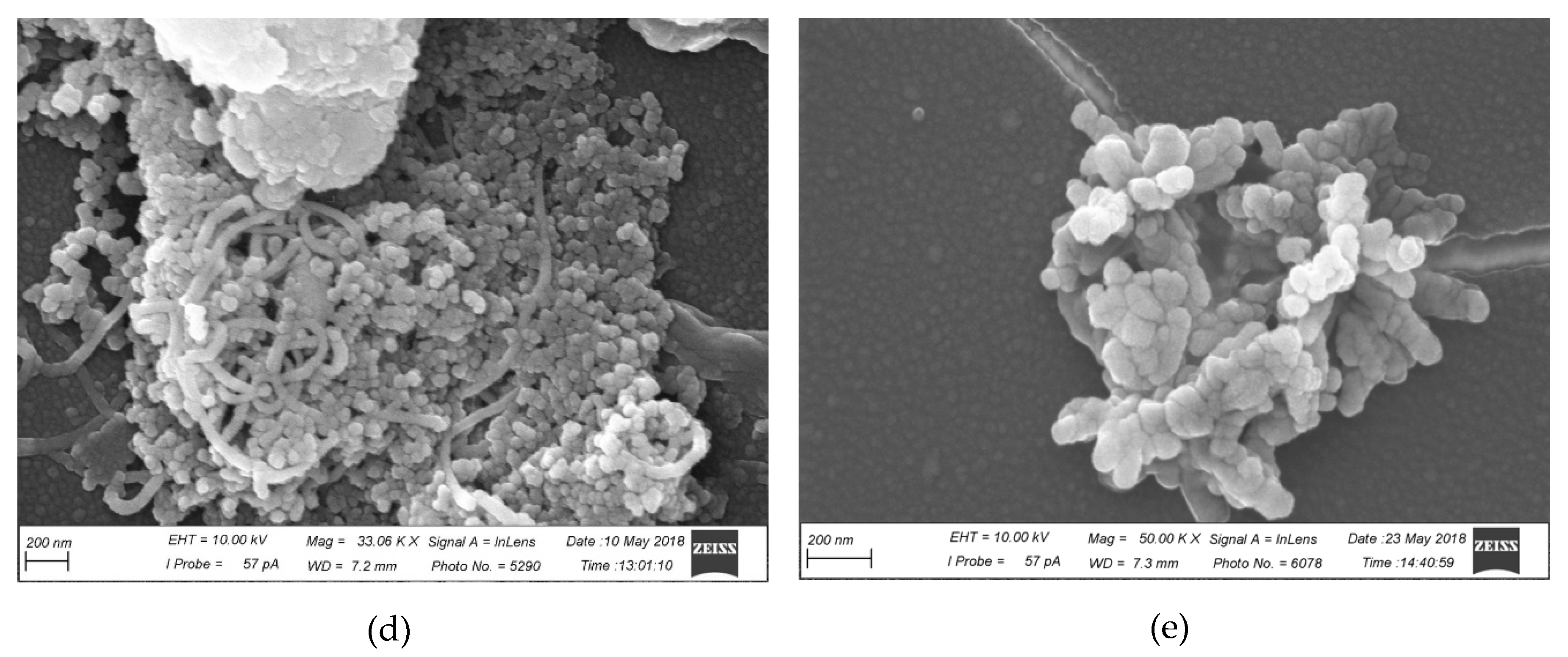
2.4. Characterization of CRG/Ech-Containing Liposomes
2.5. Interaction of Liposomes With Mucin
2.6. In Vitro Retention of Ech on Porcine Mucous Tissue
3. Discussion
4. Materials and Methods
4.1. Algal Material
4.2. Extraction of Polysaccharide
4.3. Molecular Weight Estimation
4.4. Fourier Transform-Infrared Spectroscopy (FT-IR)
4.5. Echinochrome
4.6. Preparation of Ech Water Solutions with Carrageenan
4.7. Preparation of Liposomes
4.8. Preparation of CRG/Ech-Containing Liposomes
4.9. Measurement of Encapsulation Efficiency
4.10. Lyophilization of Liposomes
4.11. Preparation of the Liposomes with Mucin
4.12. Dynamic Light Scattering (DLS) and Electrophoretic Properties of the CRG-Ech Complexes
4.13. Scanning Electron Microscopy Study
4.14. In Vitro Retention Studies of Liposome on Porcine Intestine
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Silva, T.H.; Duarte, A.R.; Moreira-Silva, J.; Mano, J.F.; Reis, R.L. Biomaterials from marine-origin biopolymers. In Biomimetic Approaches for Biomaterials Development; Mano, J.F., Ed.; Wiley-VCH Verlag: Weinheim, Germany, 2012; pp. 1–23. [Google Scholar]
- Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug Discov. 2009, 8, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, M.J.; Costa, R.R.; Mano, J.F. Marine Origin Polysaccharides in Drug Delivery Systems. Mar. Drugs 2016, 14, 34. [Google Scholar] [CrossRef] [PubMed]
- Kaldybekov, D.B.; Tonglairoum, P.; Opanasopit, P.; Khutoryanskiy, V.V. Mucoadhesive maleimide-functionalised liposomes for drug delivert to urinbary bladder. Eur. J. Pharm. Sci. 2018, 111, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.; Latifi, S.; Kahn, C.J.F.; Tamayol, A.; Habibey, R.; Passeri, E.; Linder, M.; Arab-Tehrany, E. The Positive Role of Curcumin-Loaded Salmon Nanoliposomes on the Culture of Primary Cortical Neurons. Mar. Drugs 2018, 16, 218. [Google Scholar] [CrossRef]
- Khan, I.; Elhissi, A.; Shah, M.; Alhnan, M.A.; Waqar, A. Liposome-based carrier systems and devices used for pulmonary drug delivery. In Biomaterial and Medical Tribology: Research and Development; Davim, J.P., Ed.; Woodhead Publishing Limited: Cambridge, UK, 2013; pp. 395–443. [Google Scholar]
- Vemuri, S.; Rhodes, C.T. Preparation and characterization of liposomes as therapeutic delivery systems: A review. Pharm. Acta Helv. 1995, 70, 95–111. [Google Scholar] [CrossRef]
- Patil, Y.P.; Jadhav, S. Novel methods for liposome preparation. Chem. Phys. Lipids 2014, 177, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Yousaf, S.; Subramanian, S.; Korale, O.; Alhnan, M.A.; Ahmed, W.; Taylor, K.M.G.; Elhissi, A. Proliposome powders prepared using a slurry method for the generation of beclometasone dipropionate liposomes. Int. J. Pharm. 2015, 496, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Misshra, G.P.; Bagui, M.; Tamboli, V.; Mitra, A.K. Recent applications of liposomes in ophthalmic drug delivery. J. Drug Deliv. 2011, 2011, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P. Recent advances with liposomes as pharmaceutical carriers. Nat. Rev. Drug Discov. 2005, 4, 145–160. [Google Scholar] [CrossRef] [PubMed]
- Mayer, L.D.; Bally, M.B.; Loughrey, H.; Masin, D.; Cullis, P.R. Liposomal vincistine preparations which exhibit decreased drug toxicity and increased activity against murine L1210 and P388 tumors. Cancer Res. 1990, 50, 575–579. [Google Scholar] [PubMed]
- de Marie, S.; Janknegt, R.; Bakker-Woudenberg, I.A.J.M. Clinical use of liposomal and lipid complexes amphotericin B. J. Antimicrob. Chemother. 1994, 33, 907–916. [Google Scholar] [PubMed]
- Adamczak, M.I.; Martinsen, Ø.G.; Smistad, G.; Hiorth, M. Polymer coated mucoadhesive liposomes intended for the management of xerostomia. Int. J. Pharm. 2017, 527, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Matsui, Y.; Yamamoto, H.; Kawashima, Y. Mucoadhesive properties of carbopol or chitosan-coated liposomes and their effectiveness in the oral administration of calcitonin to rats. J. Control Release 2003, 86, 235–242. [Google Scholar] [CrossRef]
- Khutoryanskiy, V.V. Advances in Mucoadhesion and Mucoadhesive Polymers. Macromol. Biosci. 2011, 11, 748–764. [Google Scholar] [CrossRef] [PubMed]
- Maderuelo, C.; Zarzuelo, A.; Lanao, J.M. Critical factors in the release of drugs from sustained release hydrophilic matrices. J. Control. Release 2011, 154, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Laurienzo, P. Marine polysaccharides in pharmaceutical applications: An overview. Mar. Drugs 2010, 8, 2435–2465. [Google Scholar] [CrossRef] [PubMed]
- Klementsrud, T.; Jonassen, H.; Hiorth, M.; Kjøniksen, A.L.; Smistad, G. Studies on pectin-coated liposomes and their interaction with mucin. Colloids Surf. B Biointerfaces 2013, 103, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Andrews, G.P.; Laverty, T.P.; Jones, D.S. Mucoadhesive polymeric platforms for controlled drug delivery. Eur. J. Pharm. Biopharm. 2009, 71, 505–518. [Google Scholar] [CrossRef] [PubMed]
- Smistad, G.; Bøyum, S.; Alund, S.J.; Samuelsen, A.B.C.; Hiorth, M. The potential of pectin as a stabilizer for liposomal drug delivery systems. Carbohydr. Polym. 2012, 90, 1337–1344. [Google Scholar] [CrossRef] [PubMed]
- Leong, K.H.; Chung, L.V.; Noordin, M.I.; Mohama, K.; Nichikawa, M.; Onuki, Y. Carboxymethylation of kappa-carrageenan for intestinal-targeted delivery of bioactive macromolecules. Carbohydr. Polym. 2011, 83, 1507–1515. [Google Scholar] [CrossRef]
- Barenholz, Y. Relevancy of drug loading to liposomal formulation therapeutic efficacy. J. Liposome Res. 2003, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Yermak, I.M.; Khotimchenko, Y.S. Chemical properties, biological activities and applications of carrageenan from red algae. Recent Adv. Mar. Biotechnol. 2003, 9, 207–255. [Google Scholar]
- Pereira, L. Biological and therapeutic properties of the seaweed polysaccharides. Int. Biol. Rev. 2018, 2, 1–50. [Google Scholar]
- Knutsen, S.H.; Myslabodsky, D.E.; Larsen, B.; Usov, A.I. A modified system of nomenclature for red algae galactans. Bot. Mar. 1994, 37, 163–169. [Google Scholar] [CrossRef]
- Yermak, I.M.; Mischchenko, N.P.; Davydova, V.N.; Glazunov, V.P.; Tarbeeva, D.V.; Kravchenko, A.O.; Pimenova, E.A.; Sorokina, I.V. Carrageenans-Sulfated Polysaccharides from Red Seaweeds as Matrices for the Inclusion of Echinochrome. Mar. Drugs 2017, 15, 337. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.H.; Kim, H.K.; Song, I.S.; Noh, S.J.; Marquez, J.; Ko, K.S.; Rhee, B.D.; Kim, N.; Mishchenko, N.P.; Fedoreyev, S.A.; et al. Echinochrome A increases mitochondrial mass and function by modulating mitochondrial biogenesis regulatory genes. Mar. Drugs 2014, 12, 4602–4615. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.Y.; McGregor, R.A.; Noh, S.J.; Choi, S.J.; Mishchenko, N.P.; Fedoreyev, S.A.; Stonik, V.A.; Han, J. Echinochrome A Improves Exercise Capacity during Short-Term Endurance Training in Rats. Mar. Drugs 2015, 13, 5722–5731. [Google Scholar] [CrossRef] [PubMed]
- Mishchenko, N.P.; Fedoreev, S.A.; Bagirova, V.L. Histochrome: A new original domestic drug. Pharm. Chem. J. 2003, 37, 48–52. [Google Scholar] [CrossRef]
- Elyakov, G.B.; Maximov, O.B.; Mischenko, N.P.; Koltsova, E.A.; Fedoreev, S.A.; Glebko, L.I.; Krasovskaya, N.P.; Artjukov, A.A. Drug Preparation “Histochrome” for Treating Acute Myocardial Infarction and Ischaemic Heart Diseases. European Patent 1,121,930, 14 November 2007. [Google Scholar]
- Yermak, I.M.; Kim, Y.H.; Titlyanov, E.A.; Isakov, V.V.; Solov’eva, T.F. Chemical structure and gel properties of carrageenan from algae belonging to the Gigartinaceae and Tichocapaceae, collected from the Russian Pacific coast. J. Appl. Phycol. 1999, 11, 41–48. [Google Scholar] [CrossRef]
- Kalitnik, A.A.; Marcov, P.A.; Anastyuk, S.D.; Barabanova, A.O.; Glazunov, V.P.; Popov, S.V.; Ovodov, Y.S.; Yermak, I.M. Gelling polysaccharide from Chondrus armatus and its oligosaccharides: The structural peculiarities and anti-inflammatory activity. Carbohydr. Polym. 2015, 115, 768–775. [Google Scholar] [CrossRef] [PubMed]
- Novikov, V.L.; Shestak, O.P.; Mishchenko, N.P.; Fedoreev, S.A.; Vasileva, E.A.; Glazunov, V.P.; Artyukov, A.A. Oxidation of 7-ethyl-2,3,5,6,8-pentahydroxy-1,4-naphthoquinone (echinochrome A) by atmospheric oxygen. 1. Structure of dehydroechinochrome. Rus. Chem. Bull. 2018, 67, 282–290. [Google Scholar] [CrossRef]
- Kulkami, S.B.; Betageri, G.V.; Singh, M. Factors affecting microencapsulation of drugs in liposomes. J. Microencapsul. 1995, 12, 229–246. [Google Scholar] [CrossRef]
- Kandzija, N.; Khutoryansky, V.V. Delivery of riboflavin-5-monophosphate into the cornea:can liposomes provide any enhancement effects? J. Pharm. Sci. 2017, 106, 3041–3049. [Google Scholar] [CrossRef] [PubMed]
- Varma, D.D.; Verma, S.; Blume, G.; Fahr, A. Particle size of liposomes influences dermal delivery of substances into skin. Int. J. Pharm. 2003, 258, 141–151. [Google Scholar] [CrossRef]
- Tang, B.C.; Dawson, M.; Lai, S.K.; Wang, Y.Y.; Suk, J.S.; Yang, M.; Zeitlin, P.; Boyle, M.P.; Fu, J.; et al. Biodegradable polymer nanoparticles that rapidly penetrate the human mucus barrier. Proc. Natl. Acad. Sci. USA 2009, 106, 19268–19273. [Google Scholar] [CrossRef] [PubMed]
- Marriott, C.; Gregory, N.P. Mucus physiology and pathology. In Bioadhesive Drug Delivery Systems; Lenaerts, V.M., Gurny, R., Eds.; CRC Press: Boca Raton, FL, USA, 1990; pp. 1–24. [Google Scholar]
- Lai, S.K.; Wang, Y.Y.; Hanes, J. Mucus-penetrating nanoparticles for drug and gene delivery to mucosal tissues. Adv. Drug Deliv. Rev. 2009, 61, 158–171. [Google Scholar] [CrossRef] [PubMed]
- Cone, R.A. Barrier properties of mucus. Adv. Drug Deliv. Rev. 2008, 61, 75–85. [Google Scholar] [CrossRef] [PubMed]

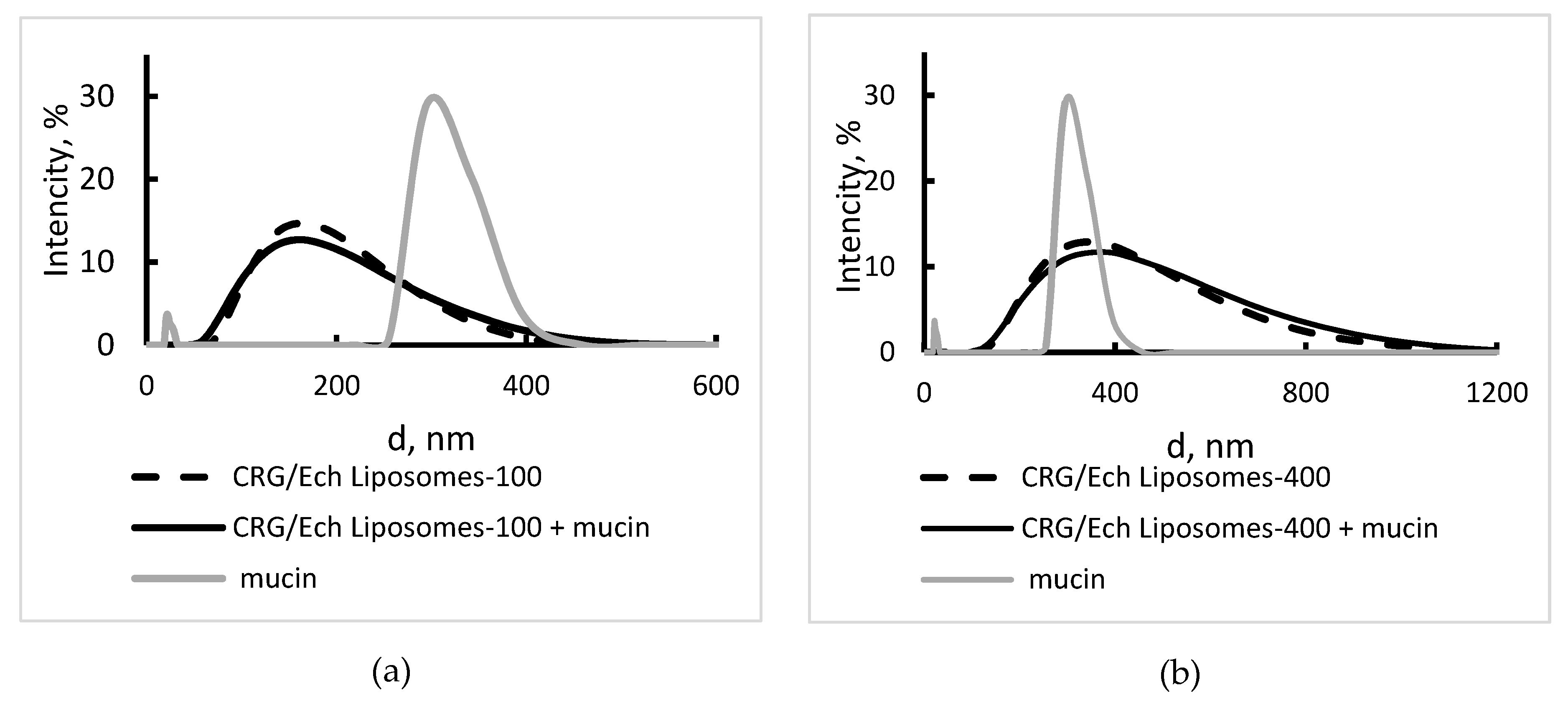
| Sample | PDI | Z-Average, nm | Hydrodinamic Diameter | ζ-Potential, mV | |
|---|---|---|---|---|---|
| d, nm | Contents, % | ||||
| Conventional liposomes-100 | 0.084 ± 0.015 | 130.2 ± 4.4 | 125.6 ± 2.5 | 100 | −14.6 ± 1.5 |
| Conventional liposomes-400 | 0.135 ± 0.023 | 402.6 ± 12.4 | 430.3 ± 29.8 | 100 | −8.2 ± 0.3 |
| CRG/Ech Liposomes-100 | 0.132 ± 0.15 | 140.8 ± 1.0 | 159.3 ± 5.8 | 100 | −24.4 ± 2.7 |
| CRG/Ech Liposomes-400 | 0.219 ± 0.023 | 334.5 ± 1.4 | 419.5 ± 12.0 | 100 | −15.6 ± 0.2 |
| Mucin, 0.15 mg/mL | 0.692 ± 0.070 | 732.2 ± 80.3 | 16.6 ± 1.2 | 6 | −17.5 ± 1.6 |
| 237.9 ± 14.0 | 94 | ||||
| CRG/Ech Liposomes-100 + mucin | 0.116 ± 0.014 | 140.0 ± 1.1 | 158.1 ± 3.3 | 100 | −20.8 ± 2.9 |
| CRG/Ech Liposomes-400 + mucin | 0.227 ± 0.016 | 343.7 ± 2.3 | 408.1 ± 9.9 | 100 | −22.6 ± 0.1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yermak, I.M.; Gorbach, V.I.; Glazunov, V.P.; Kravchenko, A.O.; Mishchenko, N.P.; Pimenova, E.A.; Davydova, V.N. Liposomal Form of the Echinochrome-Carrageenan Complex. Mar. Drugs 2018, 16, 324. https://doi.org/10.3390/md16090324
Yermak IM, Gorbach VI, Glazunov VP, Kravchenko AO, Mishchenko NP, Pimenova EA, Davydova VN. Liposomal Form of the Echinochrome-Carrageenan Complex. Marine Drugs. 2018; 16(9):324. https://doi.org/10.3390/md16090324
Chicago/Turabian StyleYermak, Irina M., Vladimir I. Gorbach, Valery P. Glazunov, Anna O. Kravchenko, Natalya P. Mishchenko, Evgeniya A. Pimenova, and Viktoria N. Davydova. 2018. "Liposomal Form of the Echinochrome-Carrageenan Complex" Marine Drugs 16, no. 9: 324. https://doi.org/10.3390/md16090324
APA StyleYermak, I. M., Gorbach, V. I., Glazunov, V. P., Kravchenko, A. O., Mishchenko, N. P., Pimenova, E. A., & Davydova, V. N. (2018). Liposomal Form of the Echinochrome-Carrageenan Complex. Marine Drugs, 16(9), 324. https://doi.org/10.3390/md16090324





