Extending the “One Strain Many Compounds” (OSMAC) Principle to Marine Microorganisms
Abstract
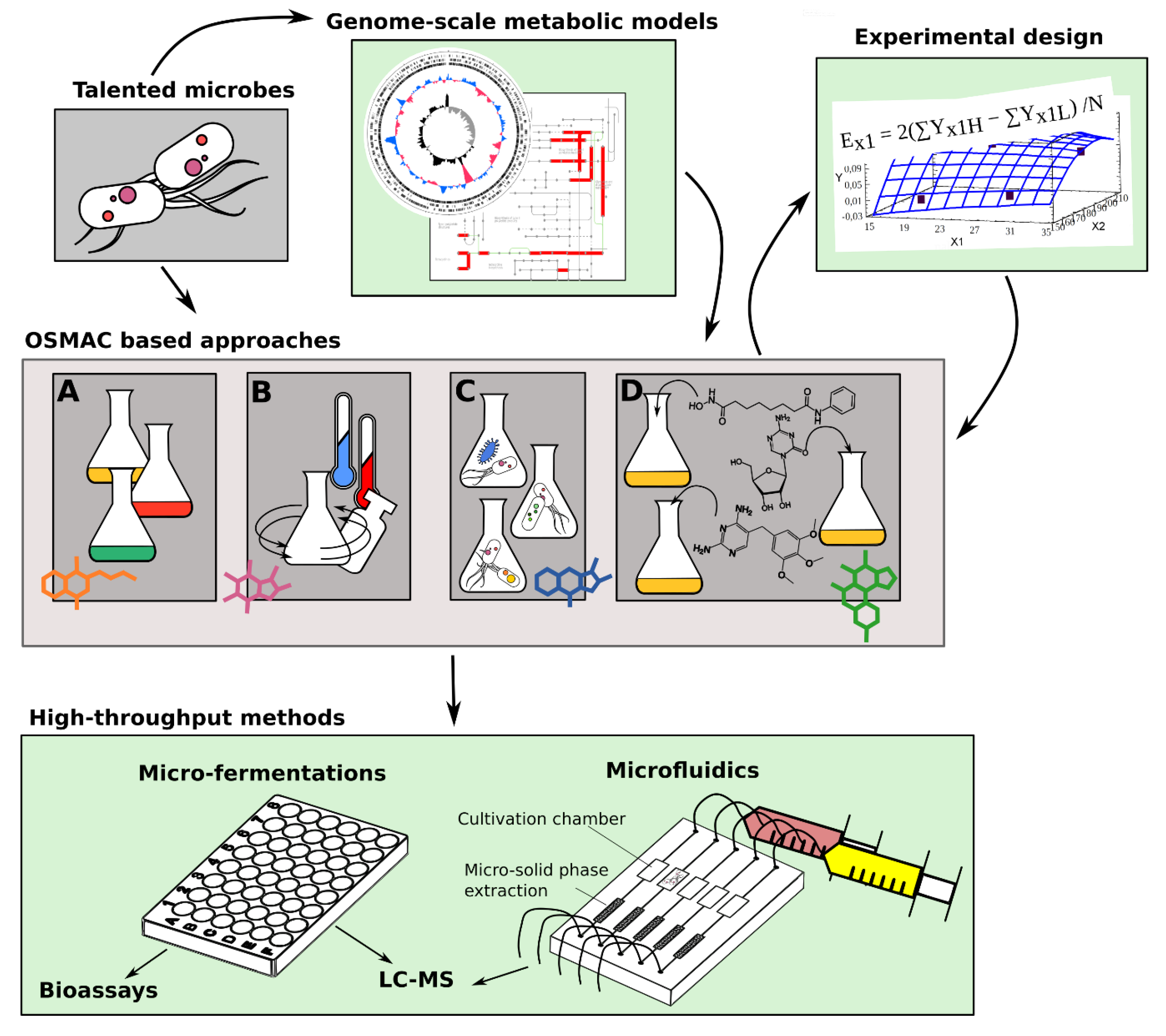
1. Introduction
2. Change in Nutrient Regimes
2.1. Carbon Source
2.2. Nitrogen Source
2.3. Sulfur and Phosphorus Sources
2.4. Trace-Elements
2.5. Comparison between Solid and Liquid Media
3. Change in Physical Parameters
3.1. Temperature
3.2. Vessel Types, Aeration, and Shaking Conditions
3.3. Osmotic Stress, Salinity, pH
4. Co-Cultivation and Other Environmental Cues
4.1. Prokaryote-Prokaryote Co-Cultivation
4.2. Prokaryote-Eukaryote Co-Cultivation
4.3. Eukaryote-Eukaryote Co-Cultivation
4.4. Addition of Chemical Elicitors
5. High Throughput Methods to Streamline Cultivation Based Biodiscovery
6. Concluding Remarks and Outlook
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- Demain, A.L.; Sanchez, S. Microbial drug discovery: 80 years of progress. J. Antibiot. 2009, 62, 5–16. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. Available online: https://amr-review.org/sites/default/files/160518_Final%20paper_with%20cover.pdf (accessed on 26 May 2016).
- Pye, C.R.; Bertin, M.J.; Lokey, R.S.; Gerwick, W.H.; Linington, R.G. Retrospective analysis of natural products provides insights for future discovery trends. Proc. Natl. Acad. Sci. USA 2017, 114, 5601–5606. [Google Scholar] [CrossRef] [PubMed]
- Jensen, P.R.; Fenical, W. Marine bacterial diversity as a resource for novel microbial products. J. Ind. Microbiol. Biotechnol. 1996, 17, 346–351. [Google Scholar] [CrossRef]
- Hughes, C.C.; Fenical, W. Antibacterials from the Sea. Chem. Eur. J. 2010, 16, 12512–12525. [Google Scholar] [CrossRef] [PubMed]
- Gribble, G. Biological Activity of Recently Discovered Halogenated Marine Natural Products. Mar. Drugs 2015, 13, 4044–4136. [Google Scholar] [CrossRef] [PubMed]
- Bentley, S.D.; Chater, K.F.; Cerdeño-Tárraga, A.M.; Challis, G.L.; Thomson, N.R.; James, K.D.; Harris, D.E.; Quail, M.A.; Kieser, H.; Harper, D.; et al. Complete genome sequence of the model actinomycete Streptomyces coelicolor A3(2). Nature 2002, 417, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Brakhage, A.A. Regulation of fungal secondary metabolism. Nat. Rev. Microbiol. 2013, 11, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Van Wezel, G.P.; McDowall, K.J. The regulation of the secondary metabolism of Streptomyces: New links and experimental advances. Nat. Prod. Rep. 2011, 28, 1311. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Soisson, S.M.; Young, K.; Shoop, W.; Kodali, S.; Galgoci, A.; Painter, R.; Parthasarathy, G.; Tang, Y.S.; Cummings, R.; et al. Platensimycin is a selective FabF inhibitor with potent antibiotic properties. Nature 2006, 441, 358–361. [Google Scholar] [CrossRef] [PubMed]
- Rutledge, P.J.; Challis, G.L. Discovery of microbial natural products by activation of silent biosynthetic gene clusters. Nat. Rev. Microbiol. 2015, 13, 509–523. [Google Scholar] [CrossRef] [PubMed]
- Reen, F.; Romano, S.; Dobson, A.; O’Gara, F. The Sound of Silence: Activating Silent Biosynthetic Gene Clusters in Marine Microorganisms. Mar. Drugs 2015, 13, 4754–4783. [Google Scholar] [CrossRef] [PubMed]
- Medema, M.H.; Breitling, R.; Bovenberg, R.; Takano, E. Exploiting plug-and-play synthetic biology for drug discovery and production in microorganisms. Nat. Rev. Microbiol. 2011, 9, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.U.; Blin, K.; Lee, S.Y.; Weber, T. Recent development of computational resources for new antibiotics discovery. Curr. Opin. Microbiol. 2017, 39, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Cimermancic, P.; Medema, M.H.; Claesen, J.; Kurita, K.; Wieland Brown, L.C.; Mavrommatis, K.; Pati, A.; Godfrey, P.A.; Koehrsen, M.; Clardy, J.; et al. Insights into Secondary Metabolism from a Global Analysis of Prokaryotic Biosynthetic Gene Clusters. Cell 2014, 158, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Naughton, L.M.; Romano, S.; O’Gara, F.; Dobson, A.D.W. Identification of secondary metabolite gene clusters in the Pseudovibrio genus reveals encouraging biosynthetic potential toward the production of novel bioactive compounds. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Romano, S. Ecology and Biotechnological Potential of Bacteria Belonging to the Genus Pseudovibrio. Appl. Environ. Microbiol. 2018, 84, e02516-17. [Google Scholar] [CrossRef] [PubMed]
- Rütschlin, S.; Gunesch, S.; Böttcher, T. One Enzyme, Three Metabolites: Shewanella algae Controls Siderophore Production via the Cellular Substrate Pool. Cell Chem. Biol. 2017, 24, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Fischbach, M.A.; Clardy, J. One pathway, many products. Nat. Chem. Biol. 2007, 3, 353–355. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.M.; Portmann, C.; Kontnik, R.; Walsh, C.T.; Clardy, J. NRPS Substrate Promiscuity Diversifies the Xenematides. Org. Lett. 2011, 13, 5144–5147. [Google Scholar] [CrossRef] [PubMed]
- Senges, C.H.R.; Al-Dilaimi, A.; Marchbank, D.H.; Wibberg, D.; Winkler, A.; Haltli, B.; Nowrousian, M.; Kalinowski, J.; Kerr, R.G.; Bandow, J.E. The secreted metabolome of Streptomyces chartreusis and implications for bacterial chemistry. Proc. Natl. Acad. Sci. USA 2018, 115, 2490–2495. [Google Scholar] [CrossRef] [PubMed]
- Bosello, M.; Robbel, L.; Linne, U.; Xie, X.; Marahiel, M.A. Biosynthesis of the Siderophore Rhodochelin Requires the Coordinated Expression of Three Independent Gene Clusters in Rhodococcus jostii RHA1. J. Am. Chem. Soc. 2011, 133, 4587–4595. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Philmus, B.; Chang, J.H.; Loper, J.E. Novel mechanism of metabolic co-regulation coordinates the biosynthesis of secondary metabolites in Pseudomonas protegens. eLife 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Vingadassalon, A.; Lorieux, F.; Juguet, M.; Le Goff, G.; Gerbaud, C.; Pernodet, J.L.; Lautru, S. Natural Combinatorial Biosynthesis Involving Two Clusters for the Synthesis of Three Pyrrolamides in Streptomyces netropsis. ACS Chem. Biol. 2015, 10, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Bode, H.B.; Bethe, B.; Höfs, R.; Zeeck, A. Big Effects from Small Changes: Possible Ways to Explore Nature’s Chemical Diversity. ChemBioChem 2002, 3, 619. [Google Scholar] [CrossRef]
- Adnani, N.; Vazquez-Rivera, E.; Adibhatla, S.; Ellis, G.; Braun, D.; Bugni, T. Investigation of Interspecies Interactions within Marine Micromonosporaceae Using an Improved Co-Culture Approach. Mar. Drugs 2015, 13, 6082–6098. [Google Scholar] [CrossRef] [PubMed]
- Jie, M.; Mao, S.; Li, H.; Lin, J.M. Multi-channel microfluidic chip-mass spectrometry platform for cell analysis. Chin. Chem. Lett. 2017, 28, 1625–1630. [Google Scholar] [CrossRef]
- Okazaki, T.; Kitahara, T.; Okami, Y. Studies on marine microorganisms. IV. A new antibiotic SS-228 Y produced by Chainia isolated from shallow sea mud. J. Antibiot. 1975, 28, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, S.; Chávez, A.; Forero, A.; García-Huante, Y.; Romero, A.; Sánchez, M.; Rocha, D.; Sánchez, B.; Ávalos, M.; Guzmán-Trampe, S.; et al. Carbon source regulation of antibiotic production. J. Antibiot. 2010, 63, 442–459. [Google Scholar] [CrossRef] [PubMed]
- Martín, J.F.; Sola-Landa, A.; Santos-Beneit, F.; Fernández-Martínez, L.T.; Prieto, C.; Rodríguez-García, A. Cross-talk of global nutritional regulators in the control of primary and secondary metabolism in Streptomyces: Cross-talk of global regulators. Microb. Biotechnol. 2011, 4, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Shang, Z.; Li, X.M.; Li, C.S.; Wang, B.G. Diverse Secondary Metabolites Produced by Marine-Derived Fungus Nigrospora sp. MA75 on Various Culture Media. Chem. Biodivers. 2012, 9, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Smetanina, O.F.; Yurchenko, A.N.; Ivanets, E.V.; Kalinovsky, A.I.; Khudyakova, Y.V.; Dyshlovoy, S.A.; von Amsberg, G.; Yurchenko, E.A.; Afiyatullov, S.S. Unique prostate cancer-toxic polyketides from marine sediment-derived fungus Isaria felina. J. Antibiot. 2017, 70, 856–858. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos Dias, A.C.; Couzinet-Mossion, A.; Ruiz, N.; Bellec, M.L.; Gentil, E.; Wielgosz-Collin, G.; Bertrand, S. Sugar Induced Modification in Glycolipid Production in Acremonium sp. Revealed by LC-MS Lipidomic Approach. Curr. Biotechnol. 2017, 6. [Google Scholar] [CrossRef]
- Wietz, M.; Månsson, M.; Gram, L. Chitin stimulates production of the antibiotic andrimid in a Vibrio coralliilyticus strain: Andrimid production in V. coralliilyticus. Environ. Microbiol. Rep. 2011, 3, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Giubergia, S.; Phippen, C.; Nielsen, K.F.; Gram, L. Growth on Chitin Impacts the Transcriptome and Metabolite Profiles of Antibiotic-Producing Vibrio coralliilyticus S2052 and Photobacterium galatheae S2753. mSystems 2017, 2, e00141-16. [Google Scholar] [CrossRef] [PubMed]
- Giubergia, S.; Phippen, C.; Gotfredsen, C.H.; Nielsen, K.F.; Gram, L. Influence of Niche-Specific Nutrients on Secondary Metabolism in Vibrionaceae. Appl. Environ. Microbiol. 2016, 82, 4035–4044. [Google Scholar] [CrossRef] [PubMed]
- Sujatha, P.; Bapi Raju, K.V.V.S.N.; Ramana, T. Studies on a new marine streptomycete BT-408 producing polyketide antibiotic SBR-22 effective against methicillin resistant Staphylococcus aureus. Microbiol. Res. 2005, 160, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Saha, M.; Ghosh, D.; Ghosh, D.; Garai, D.; Jaisankar, P.; Sarkar, K.K.; Dutta, P.K.; Das, S.; Jha, T.; Mukherjee, J. Studies on the production and purification of an antimicrobial compound and taxonomy of the producer isolated from the marine environment of the Sundarbans. Appl. Microbiol. Biotechnol. 2005, 66, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Darabpour, E.; Roayaei Ardakani, M.; Motamedi, H.; Ronagh, M.T.; Najafzadeh, H. Purification and optimization of production conditions of a marine-derived antibiotic and ultra-structural study on the effect of this antibiotic against MRSA. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 157–165. [Google Scholar] [PubMed]
- Preetha, R.; Jayaprakash, N.S.; Philip, R.; Bright Singh, I.S. Optimization of carbon and nitrogen sources and growth factors for the production of an aquaculture probiotic (Pseudomonas MCCB 103) using response surface methodology. J. Appl. Microbiol. 2006, 102, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Zhu, T.; Wei, H.; Zhang, G.; Wang, H.; Gu, Q. Spicochalasin A and New Aspochalasins from the Marine-Derived Fungus Spicaria elegans. Eur. J. Org. Chem. 2009, 2009, 3045–3051. [Google Scholar] [CrossRef]
- Miao, L.; Kwong, T.F.N.; Qian, P.Y. Effect of culture conditions on mycelial growth, antibacterial activity, and metabolite profiles of the marine-derived fungus Arthrinium c.f. saccharicola. Appl. Microbiol. Biotechnol. 2006, 72, 1063–1073. [Google Scholar] [CrossRef] [PubMed]
- Lauritano, C.; Andersen, J.H.; Hansen, E.; Albrigtsen, M.; Escalera, L.; Esposito, F.; Helland, K.; Hanssen, K.Ø.; Romano, G.; Ianora, A. Bioactivity Screening of Microalgae for Antioxidant, Anti-Inflammatory, Anticancer, Anti-Diabetes, and Antibacterial Activities. Front. Mar. Sci. 2016, 3. [Google Scholar] [CrossRef]
- Wang, F.; Huang, L.; Gao, B.; Zhang, C. Optimum Production Conditions, Purification, Identification, and Antioxidant Activity of Violaxanthin from Microalga Eustigmatos cf. polyphem (Eustigmatophyceae). Mar. Drugs 2018, 16, 190. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lu, Z.; Sun, K.; Zhu, W. Effects of High Salt Stress on Secondary Metabolite Production in the Marine-Derived Fungus Spicaria elegans. Mar. Drugs 2011, 9, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Luan, Y.; Wei, H.; Zhang, Z.; Che, Q.; Liu, Y.; Zhu, T.; Mándi, A.; Kurtán, T.; Gu, Q.; Li, D. Eleganketal A, a Highly Oxygenated Dibenzospiroketal from the Marine-Derived Fungus Spicaria elegans KLA03. J. Nat. Prod. 2014, 77, 1718–1723. [Google Scholar] [CrossRef] [PubMed]
- Nigan, V.K.; Verma, R.; Kumar, A.; Kundu, S.; Ghosh, P. Influence of medium constituents on the biosynthesis of cephalosporin-C. Electron. J. Biotechnol. 2007, 10, 230–239. [Google Scholar] [CrossRef]
- Bouras, N.; Mathieu, F.; Sabaou, N.; Lebrihi, A. Effect of amino acids containing sulfur on dithiolopyrrolone antibiotic productions by Saccharothrix algeriensis NRRL B-24137. J. Appl. Microbiol. 2006, 100, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Demain, A.L.; Zhang, J. Cephalosporin C Production by: The Methionine Story. Crit. Rev. Biotechnol. 2008, 18, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Brock, N.L.; Menke, M.; Klapschinski, T.A.; Dickschat, J.S. Marine bacteria from the Roseobacter clade produce sulfur volatiles via amino acid and dimethylsulfoniopropionate catabolism. Org. Biomol. Chem. 2014, 12, 4318–4323. [Google Scholar] [CrossRef] [PubMed]
- Geng, H.; Belas, R. Expression of Tropodithietic Acid Biosynthesis Is Controlled by a Novel Autoinducer. J. Bacteriol. 2010, 192, 4377–4387. [Google Scholar] [CrossRef] [PubMed]
- Romano, S.; Schulz-Vogt, H.N.; González, J.M.; Bondarev, V. Phosphate Limitation Induces Drastic Physiological Changes, Virulence-Related Gene Expression, and Secondary Metabolite Production in Pseudovibrio sp. Strain FO-BEG1. Appl. Environ. Microbiol. 2015, 81, 3518–3528. [Google Scholar] [CrossRef] [PubMed]
- Romano, S.; Dittmar, T.; Bondarev, V.; Weber, R.J.M.; Viant, M.R.; Schulz-Vogt, H.N. Exo-Metabolome of Pseudovibrio sp. FO-BEG1 Analyzed by Ultra-High Resolution Mass Spectrometry and the Effect of Phosphate Limitation. PLoS ONE 2014, 9, e96038. [Google Scholar] [CrossRef] [PubMed]
- Martín, J.F.; Rodríguez-García, A.; Liras, P. The master regulator PhoP coordinates phosphate and nitrogen metabolism, respiration, cell differentiation and antibiotic biosynthesis: Comparison in Streptomyces coelicolor and Streptomyces avermitilis. J. Antibiot. 2017, 70, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Santos-Beneit, F. The Pho regulon: A huge regulatory network in bacteria. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Lauritano, C.; Martín, J.; de la Cruz, M.; Reyes, F.; Romano, G.; Ianora, A. First identification of marine diatoms with anti-tuberculosis activity. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Hii, K.S.; Lim, P.T.; Kon, N.F.; Takata, Y.; Usup, G.; Leaw, C.P. Physiological and transcriptional responses to inorganic nutrition in a tropical Pacific strain of Alexandrium minutum: Implications for the saxitoxin genes and toxin production. Harmful Algae 2016, 56, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Elshahawi, S.I.; Trindade-Silva, A.E.; Hanora, A.; Han, A.W.; Flores, M.S.; Vizzoni, V.; Schrago, C.G.; Soares, C.A.; Concepcion, G.P.; Distel, D.L.; et al. Boronated tartrolon antibiotic produced by symbiotic cellulose-degrading bacteria in shipworm gills. Proc. Natl. Acad. Sci. USA 2013, 110, E295–E304. [Google Scholar] [CrossRef] [PubMed]
- Romano, S.; Bondarev, V.; Kölling, M.; Dittmar, T.; Schulz-Vogt, H.N. Phosphate Limitation Triggers the Dissolution of Precipitated Iron by the Marine Bacterium Pseudovibrio sp. FO-BEG1. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Gribble, G.W. Natural Organohalogens: A New Frontier for Medicinal Agents? J. Chem. Educ. 2004, 81, 1441. [Google Scholar] [CrossRef]
- Si, Y.; Tang, M.; Lin, S.; Chen, G.; Feng, Q.; Wang, Y.; Hua, H.; Bai, J.; Wang, H.; Pei, Y. Cytotoxic cytochalasans from Aspergillus flavipes PJ03-11 by OSMAC method. Tetrahedron Lett. 2018. [Google Scholar] [CrossRef]
- Huang, H.; Wang, F.; Luo, M.; Chen, Y.; Song, Y.; Zhang, W.; Zhang, S.; Ju, J. Halogenated Anthraquinones from the Marine-Derived Fungus Aspergillus sp. SCSIO F063. J. Nat. Prod. 2012, 75, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, H.; Rotinsulu, H.; Narita, R.; Takahashi, R.; Namikoshi, M. Induced Production of Halogenated Epidithiodiketopiperazines by a Marine-Derived Trichoderma cf. brevicompactum with Sodium Halides. J. Nat. Prod. 2015, 78, 2319–2321. [Google Scholar] [CrossRef] [PubMed]
- Fil, T.P.; Pallini, H.F.; Amaral, L.D.S.; Silva, J.V.D.; Bidóia, D.L.; Peron, F.; Garcia, F.P.; Nakamura, C.V.; Rodrigues-Filho, E. Copper and Manganese Cations Alter Secondary Metabolism in the Fungus Penicillium brasilianum. J. Braz. Chem. Soc. 2016. [Google Scholar] [CrossRef]
- Ochi, K.; Tanaka, Y.; Tojo, S. Activating the expression of bacterial cryptic genes by rpoB mutations in RNA polymerase or by rare earth elements. J. Ind. Microbiol. Biotechnol. 2014, 41, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Palmer, D.A.; Bender, C.L. Effects of Environmental and Nutritional Factors on Production of the Polyketide Phytotoxin Coronatine by Pseudomonas syringae pv. Glycinea. Appl. Environ. Microbiol. 1993, 59, 1619–1626. [Google Scholar] [PubMed]
- Wang, W.J.; Li, D.Y.; Li, Y.C.; Hua, H.M.; Ma, E.L.; Li, Z.L. Caryophyllene Sesquiterpenes from the Marine-Derived Fungus Ascotricha sp. ZJ-M-5 by the One Strain–Many Compounds Strategy. J. Nat. Prod. 2014, 77, 1367–1371. [Google Scholar] [CrossRef] [PubMed]
- Ye, P.; Shen, L.; Jiang, W.; Ye, Y.; Chen, C.T.; Wu, X.; Wang, K.; Wu, B. Zn-Driven Discovery of a Hydrothermal Vent Fungal Metabolite Clavatustide C, and an Experimental Study of the Anti-Cancer Mechanism of Clavatustide B. Mar. Drugs 2014, 12, 3203–3217. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Pan, C.; Auckloo, B.N.; Chen, X.; Chen, C.T.A.; Wang, K.; Wu, X.; Ye, Y.; Wu, B. Stress-driven discovery of a cryptic antibiotic produced by Streptomyces sp. WU20 from Kueishantao hydrothermal vent with an integrated metabolomics strategy. Appl. Microbiol. Biotechnol. 2017, 101, 1395–1408. [Google Scholar] [CrossRef] [PubMed]
- English, A.L.; Boufridi, A.; Quinn, R.J.; Kurtböke, D.I. Evaluation of fermentation conditions triggering increased antibacterial activity from a near-shore marine intertidal environment-associated Streptomyces species. Synth. Syst. Biotechnol. 2017, 2, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.C.; Yang, F.; Zhang, R.; Shi, X.; Lu, X.H.; Luan, Y.S.; Xiu, Z.L.; Dong, Y.S. Production of polyketides with anthelmintic activity by the fungus Talaromyces wortmannii using one strain-many compounds (OSMAC) method. Phytochem. Lett. 2016, 18, 157–161. [Google Scholar] [CrossRef]
- Guo, W.; Zhang, Z.; Zhu, T.; Gu, Q.; Li, D. Penicyclones A–E, Antibacterial Polyketides from the Deep-Sea-Derived Fungus Penicillium sp. F23-2. J. Nat. Prod. 2015, 78, 2699–2703. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Zhang, G.; Wang, B.; Li, X.; Yue, S.; Chen, J.; Zhang, H.; Wang, H. Production and Identification of Inthomycin B Produced by a Deep-Sea Sediment-Derived Streptomyces sp. YB104 Based on Cultivation-Dependent Approach. Curr. Microbiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Myouga, H.; Yoshimizu, M.; Tajima, K.; Ezura, Y. Purification of an Antiviral Substance Produced by Alteromonas sp. and Its Virucidal Activity against Fish Viruses. Fish Pathol. 1995, 30, 15–22. [Google Scholar] [CrossRef]
- Ingebrigtsen, R.A.; Hansen, E.; Andersen, J.H.; Eilertsen, H.C. Light and temperature effects on bioactivity in diatoms. J. Appl. Phycol. 2016, 28, 939–950. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.S.; Rong, X.G.; Kang, H.H.; Ma, L.Y.; Hamann, M.; Liu, W.Z. Raistrickiones A−E from a Highly Productive Strain of Penicillium raistrickii Generated through Thermo Change. Mar. Drugs 2018, 16, 213. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, L.A.; Sierra, M.G.; Siñeriz, F.; Delgado, O. Andrimid production at low temperature by a psychrotolerant Serratia proteamaculans strain. World J. Microbiol. Biotechnol. 2013, 29, 1773–1781. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.; Gachhui, R.; Mukherjee, J. Enhanced biofilm formation and melanin synthesis by the oyster settlement-promoting is related to hydrophobic surface and simulated intertidal environment. Biofouling 2015, 31, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.; Sarkar, S.; Gachhui, R.; Mukherjee, J. A novel conico-cylindrical flask aids easy identification of critical process parameters for cultivation of marine bacteria. Appl. Microbiol. Biotechnol. 2011, 90, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Veerabadhran, M.; Chakraborty, S.; Mitra, S.; Karmakar, S.; Mukherjee, J. Effects of flask configuration on biofilm growth and metabolites of intertidal Cyanobacteria isolated from a mangrove forest. J. Appl. Microbiol. 2018, 125, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Burja, A.M.; Abou-Mansour, E.; Banaigs, B.; Payri, C.; Burgess, J.G.; Wright, P.C. Culture of the marine cyanobacterium, Lyngbya majuscula (Oscillatoriaceae), for bioprocess intensified production of cyclic and linear lipopeptides. J. Microbiol. Methods 2002, 48, 207–219. [Google Scholar] [CrossRef]
- Gallagher, K.A.; Wanger, G.; Henderson, J.; Llorente, M.; Hughes, C.C.; Jensen, P.R. Ecological implications of hypoxia-triggered shifts in secondary metabolism: Hypoxia-triggered secondary metabolism. Environ. Microbiol. 2017, 19, 2182–2191. [Google Scholar] [CrossRef] [PubMed]
- Kebede, B.; Wrigley, S.; Prashar, A.; Rahlff, J.; Wolf, M.; Reinshagen, J.; Gribbon, P.; Imhoff, J.; Silber, J.; Labes, A.; et al. Establishing the Secondary Metabolite Profile of the Marine Fungus: Tolypocladium geodes sp. MF458 and Subsequent Optimisation of Bioactive Secondary Metabolite Production. Mar. Drugs 2017, 15, 84. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Peng, J.; Zhu, T.; Gu, Q.; Keyzers, R.A.; Li, D. Sorbicillamines A–E, Nitrogen-Containing Sorbicillinoids from the Deep-Sea-Derived Fungus Penicillium sp. F23–2. J. Nat. Prod. 2013, 76, 2106–2112. [Google Scholar] [CrossRef] [PubMed]
- Overy, D.; Correa, H.; Roullier, C.; Chi, W.C.; Pang, K.L.; Rateb, M.; Ebel, R.; Shang, Z.; Capon, R.; Bills, G.; et al. Does Osmotic Stress Affect Natural Product Expression in Fungi? Mar. Drugs 2017, 15, 254. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zheng, J.; Liu, P.; Wang, W.; Zhu, W. Three New Compounds from Aspergillus terreus PT06-2 Grown in a High Salt Medium. Mar. Drugs 2011, 9, 1368–1378. [Google Scholar] [CrossRef] [PubMed]
- Bose, U.; Hewavitharana, A.; Ng, Y.; Shaw, P.; Fuerst, J.; Hodson, M. LC-MS-Based Metabolomics Study of Marine Bacterial Secondary Metabolite and Antibiotic Production in Salinispora arenicola. Mar. Drugs 2015, 13, 249–266. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.K.; Hodson, M.P.; Hewavitharana, A.K.; Bose, U.; Shaw, P.N.; Fuerst, J.A. Effects of salinity on antibiotic production in sponge-derived Salinispora actinobacteria. J. Appl. Microbiol. 2014, 117, 109–125. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.; Roy, D.; Mukherjee, J. Production of a potentially novel antimicrobial compound by a biofilm-forming marine Streptomyces sp. in a niche-mimic rotating disk bioreactor. Bioprocess Biosyst. Eng. 2010, 33, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Holdenrieder, O. Kristallbildung bei Heterobasidion annosum (Fr.) Bref. (Fomes annosus P. Karst.) und anderen holzbewohnenden Pilzen. For. Pathol. 1982, 12, 41–58. [Google Scholar] [CrossRef]
- Sonnenbichler, J.; Dietrich, J.; Peipp, H. Secondary Fungal Metabolites and Their Biological Activities, V. Investigations Concerning the Induction of the Biosynthesis of Toxic Secondary Metabolites in Basidiomycetes. Biol. Chem. Hoppe Seyler 1994, 375, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Sonnenbichler, J.; Lamm, V.; Gieren, A.; Holdenrieder, O.; Lotter, H. A cyclopentabenzopyranone produced by the fungus heterobasidion annosum in dual cultures. Phytochemistry 1983, 22, 1489–1491. [Google Scholar] [CrossRef]
- Cueto, M.; Jensen, P.R.; Kauffman, C.; Fenical, W.; Lobkovsky, E.; Clardy, J. Pestalone, a New Antibiotic Produced by a Marine Fungus in Response to Bacterial Challenge. J. Nat. Prod. 2001, 64, 1444–1446. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Sun, X.; Jin, M.; Zhang, X. A Novel Benzoquinone Compound Isolated from Deep-Sea Hydrothermal Vent Triggers Apoptosis of Tumor Cells. Mar. Drugs 2017, 15, 200. [Google Scholar] [CrossRef]
- Briand, E.; Bormans, M.; Gugger, M.; Dorrestein, P.C.; Gerwick, W.H. Changes in secondary metabolic profiles of Microcystis aeruginosa strains in response to intraspecific interactions: Cyanopeptides production under mono and co-culture. Environ. Microbiol. 2016, 18, 384–400. [Google Scholar] [CrossRef] [PubMed]
- Burgess, J.G.; Jordan, E.M.; Bregu, M.; Mearns-Spragg, A.; Boyd, K.G. Microbial antagonism: A neglected avenue of natural products research. J. Biotechnol. 1999, 70, 27–32. [Google Scholar] [CrossRef]
- Sung, A.; Gromek, S.; Balunas, M. Upregulation and Identification of Antibiotic Activity of a Marine-Derived Streptomyces sp. via Co-Cultures with Human Pathogens. Mar. Drugs 2017, 15, 250. [Google Scholar] [CrossRef] [PubMed]
- Dusane, D.H.; Matkar, P.; Venugopalan, V.P.; Kumar, A.R.; Zinjarde, S.S. Cross-Species Induction of Antimicrobial Compounds, Biosurfactants and Quorum-Sensing Inhibitors in Tropical Marine Epibiotic Bacteria by Pathogens and Biofouling Microorganisms. Curr. Microbiol. 2011, 62, 974–980. [Google Scholar] [CrossRef] [PubMed]
- Fdhila, F.; Vázquez, V.; Sánchez, J.L.; Riguera, R. dd-Diketopiperazines: Antibiotics Active against Vibrio anguillarum Isolated from Marine Bacteria Associated with Cultures of Pecten m aximus. J. Nat. Prod. 2003, 66, 1299–1301. [Google Scholar] [CrossRef] [PubMed]
- Dashti, Y.; Grkovic, T.; Abdelmohsen, U.; Hentschel, U.; Quinn, R. Production of Induced Secondary Metabolites by a Co-Culture of Sponge-Associated Actinomycetes, Actinokineospora sp. EG49 and Nocardiopsis sp. RV163. Mar. Drugs 2014, 12, 3046–3059. [Google Scholar] [CrossRef] [PubMed]
- Trischman, J.A.; Oeffner, R.E.; de Luna, M.G.; Kazaoka, M. Competitive Induction and Enhancement of Indole and a Diketopiperazine in Marine Bacteria. Mar. Biotechnol. 2004, 6, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Onaka, H. Novel antibiotic screening methods to awaken silent or cryptic secondary metabolic pathways in actinomycetes. J. Antibiot. 2017, 70, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, S.; Wong, C.P.; Ozeki, M.; Zhang, H.; Hayashi, F.; Awakawa, T.; Asamizu, S.; Onaka, H.; Abe, I. Umezawamides, new bioactive polycyclic tetramate macrolactams isolated from a combined-culture of Umezawaea sp. and mycolic acid-containing bacterium. J. Antibiot. 2018. [Google Scholar] [CrossRef] [PubMed]
- Conde-Martínez, N.; Acosta-González, A.; Díaz, L.E.; Tello, E. Use of a mixed culture strategy to isolate halophilic bacteria with antibacterial and cytotoxic activity from the Manaure solar saltern in Colombia. BMC Microbiol. 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.C.; Jensen, P.R.; Kauffman, C.A.; Fenical, W. Libertellenones A–D: Induction of cytotoxic diterpenoid biosynthesis by marine microbial competition. Bioorg. Med. Chem. 2005, 13, 5267–5273. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.C.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Induced Production of Emericellamides A and B from the Marine-Derived Fungus Emericella sp. in Competing Co-culture. J. Nat. Prod. 2007, 70, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Ding, W.; Wang, Q.; Ma, Z.; Xu, X.; Zhao, X.; Chen, Z. Induction of cryptic bioactive 2,5-diketopiperazines in fungus Penicillium sp. DT-F29 by microbial co-culture. Tetrahedron 2017, 73, 907–914. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Zhuang, Y.; Kong, F.; Zhang, C.; Zhu, W. Phenolic Polyketides from the Co-Cultivation of Marine-Derived Penicillium sp. WC-29-5 and Streptomyces fradiae 007. Mar. Drugs 2014, 12, 2079–2088. [Google Scholar] [CrossRef] [PubMed]
- Wakefield, J.; Hassan, H.M.; Jaspars, M.; Ebel, R.; Rateb, M.E. Dual Induction of New Microbial Secondary Metabolites by Fungal Bacterial Co-cultivation. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Moree, W.J.; Yang, J.Y.; Zhao, X.; Liu, W.T.; Aparicio, M.; Atencio, L.; Ballesteros, J.; Sánchez, J.; Gavilán, R.G.; Gutiérrez, M.; et al. Imaging Mass Spectrometry of a Coral Microbe Interaction with Fungi. J. Chem. Ecol. 2013, 39, 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- Netzker, T.; Fischer, J.; Weber, J.; Mattern, D.J.; König, C.C.; Valiante, V.; Schroeckh, V.; Brakhage, A.A. Microbial communication leading to the activation of silent fungal secondary metabolite gene clusters. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Ding, W.; Ma, Z. Induced production of cytochalasans in co-culture of marine fungus and actinomycete sp. Nat. Prod. Res. 2016, 30, 1718–1723. [Google Scholar] [CrossRef] [PubMed]
- Scherlach, K.; Busch, B.; Lackner, G.; Paszkowski, U.; Hertweck, C. Symbiotic Cooperation in the Biosynthesis of a Phytotoxin. Angew. Chem. Int. Ed. 2012, 51, 9615–9618. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Aktas, N.; Konuklugil, B.; Mándi, A.; Daletos, G.; Lin, W.; Dai, H.; Kurtán, T.; Proksch, P. A new fusarielin analogue from Penicillium sp. isolated from the Mediterranean sponge Ircinia oros. Tetrahedron Lett. 2015, 56, 5317–5320. [Google Scholar] [CrossRef]
- Zhuravleva, O.I.; Kirichuk, N.N.; Denisenko, V.A.; Dmitrenok, P.S.; Yurchenko, E.A.; Min’ko, E.M.; Ivanets, E.V.; Afiyatullov, S.S. New Diorcinol J Produced by Co-Cultivation of Marine Fungi Aspergillus sulphureus and Isaria felina. Chem. Nat. Compd. 2016, 52, 227–230. [Google Scholar] [CrossRef]
- Shang, Z.; Salim, A.A.; Capon, R.J. Chaunopyran A: Co-Cultivation of Marine Mollusk-Derived Fungi Activates a Rare Class of 2-Alkenyl-Tetrahydropyran. J. Nat. Prod. 2017, 80, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; He, X.; Zhang, G.; Che, Q.; Zhu, T.; Gu, Q.; Li, D. Inducing Secondary Metabolite Production by Combined Culture of Talaromyces aculeatus and Penicillium variabile. J. Nat. Prod. 2017, 80, 3167–3171. [Google Scholar] [CrossRef] [PubMed]
- Mandelare, P.E.; Adpressa, D.A.; Kaweesa, E.N.; Zakharov, L.N.; Loesgen, S. Coculture of Two Developmental Stages of a Marine-Derived Aspergillus alliaceus Results in the Production of the Cytotoxic Bianthrone Allianthrone A. J. Nat. Prod. 2018. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.B.; Bassler, B.L. Quorum Sensing in Bacteria. Annu. Rev. Microbiol. 2001, 55, 165–199. [Google Scholar] [CrossRef] [PubMed]
- Buchan, A.; Mitchell, A.; Cude, W.N.; Campagna, S. Acyl-Homoserine Lactone-Based Quorum Sensing in Members of the Marine Bacterial Roseobacter Clade: Complex Cell-to-Cell Communication Controls Multiple Physiologies. In Stress and Environmental Regulation of Gene Expression and Adaptation in Bacteria; de Bruijn, F.J., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016; pp. 225–233. ISBN 978-1-119-00481-3. [Google Scholar]
- Ishida, K.; Lincke, T.; Behnken, S.; Hertweck, C. Induced Biosynthesis of Cryptic Polyketide Metabolites in a Burkholderia thailandensis Quorum Sensing Mutant. J. Am. Chem. Soc. 2010, 132, 13966–13968. [Google Scholar] [CrossRef] [PubMed]
- Boyer, M.; Wisniewski-Dyé, F. Cell–cell signalling in bacteria: Not simply a matter of quorum: Cell–cell signalling in bacteria. FEMS Microbiol. Ecol. 2009, 70, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Zheng, L.; Zhou, W.; Cui, Z.; Han, P.; Tian, L.; Wang, X. A case study on chemical defense based on quorum sensing: Antibacterial activity of sponge-associated bacterium Pseudoalteromonas sp. NJ6-3-1 induced by quorum sensing mechanisms. Ann. Microbiol. 2011, 61, 247–255. [Google Scholar] [CrossRef]
- Brotherton, C.A.; Medema, M.H.; Greenberg, E.P. luxR Homolog-Linked Biosynthetic Gene Clusters in Proteobacteria. mSystems 2018, 3, e00208-17. [Google Scholar] [CrossRef] [PubMed]
- Subramoni, S.; Florez Salcedo, D.V.; Suarez-Moreno, Z.R. A bioinformatic survey of distribution, conservation, and probable functions of LuxR solo regulators in bacteria. Front. Cell. Infect. Microbiol. 2015, 5. [Google Scholar] [CrossRef]
- Bondarev, V.; Richter, M.; Romano, S.; Piel, J.; Schwedt, A.; Schulz-Vogt, H.N. The genus Pseudovibrio contains metabolically versatile bacteria adapted for symbiosis: The genus Pseudovibrio. Environ. Microbiol. 2013, 15, 2095–2113. [Google Scholar] [CrossRef] [PubMed]
- Okada, B.K.; Seyedsayamdost, M.R. Antibiotic dialogues: Induction of silent biosynthetic gene clusters by exogenous small molecules. FEMS Microbiol. Rev. 2017, 41, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Pimentel-Elardo, S.M.; Sørensen, D.; Ho, L.; Ziko, M.; Bueler, S.A.; Lu, S.; Tao, J.; Moser, A.; Lee, R.; Agard, D.; et al. Activity-Independent Discovery of Secondary Metabolites Using Chemical Elicitation and Cheminformatic Inference. ACS Chem. Biol. 2015, 10, 2616–2623. [Google Scholar] [CrossRef] [PubMed]
- Seyedsayamdost, M.R. High-throughput platform for the discovery of elicitors of silent bacterial gene clusters. Proc. Natl. Acad. Sci. USA 2014, 111, 7266–7271. [Google Scholar] [CrossRef] [PubMed]
- Beyersmann, P.G.; Tomasch, J.; Son, K.; Stocker, R.; Göker, M.; Wagner-Döbler, I.; Simon, M.; Brinkhoff, T. Dual function of tropodithietic acid as antibiotic and signaling molecule in global gene regulation of the probiotic bacterium Phaeobacter inhibens. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Cichewicz, R.H. Epigenome manipulation as a pathway to new natural product scaffolds and their congeners. Nat. Prod. Rep. 2010, 27, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.H.; Keller, N. Regulation of Secondary Metabolism in Filamentous Fungi. Annu. Rev. Phytopathol. 2005, 43, 437–458. [Google Scholar] [CrossRef] [PubMed]
- Vervoort, H.C.; Drašković, M.; Crews, P. Histone Deacetylase Inhibitors as a Tool to Up-Regulate New Fungal Biosynthetic Products: Isolation of EGM-556, a Cyclodepsipeptide, from Microascus sp. Org. Lett. 2011, 13, 410–413. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Zhang, Z.; Chen, Y.; Che, Q.; Zhu, T.; Gu, Q.; Li, D. Varitatin A, a Highly Modified Fatty Acid Amide from Penicillium variabile Cultured with a DNA Methyltransferase Inhibitor. J. Nat. Prod. 2015, 78, 2841–2845. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Zhang, Z.; Che, Q.; Zhu, T.; Gu, Q.; Li, D. Varilactones and wortmannilactones produced by Penicillium variabile cultured with histone deacetylase inhibitor. Arch. Pharm. Res. 2018, 41, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Shao, C.L.; Chen, M.; Liu, Q.A.; Wang, C.Y. Brominated resorcylic acid lactones from the marine-derived fungus Cochliobolus lunatus induced by histone deacetylase inhibitors. Tetrahedron Lett. 2014, 55, 4888–4891. [Google Scholar] [CrossRef]
- Christian, O.E.; Compton, J.; Christian, K.R.; Mooberry, S.L.; Valeriote, F.A.; Crews, P. Using Jasplakinolide to Turn on Pathways that Enable the Isolation of New Chaetoglobosins from Phomospis asparagi. J. Nat. Prod. 2005, 68, 1592–1597. [Google Scholar] [CrossRef] [PubMed]
- Adnani, N.; Rajski, S.R.; Bugni, T.S. Symbiosis-inspired approaches to antibiotic discovery. Nat. Prod. Rep. 2017, 34, 784–814. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, C.; Marker, A.; Kurtböke, D. An Overview on Marine Sponge-Symbiotic Bacteria as Unexhausted Sources for Natural Product Discovery. Diversity 2017, 9, 40. [Google Scholar] [CrossRef]
- Indraningrat, A.; Smidt, H.; Sipkema, D. Bioprospecting Sponge-Associated Microbes for Antimicrobial Compounds. Mar. Drugs 2016, 14, 87. [Google Scholar] [CrossRef] [PubMed]
- Flórez, L.V.; Biedermann, P.H.W.; Engl, T.; Kaltenpoth, M. Defensive symbioses of animals with prokaryotic and eukaryotic microorganisms. Nat. Prod. Rep. 2015, 32, 904–936. [Google Scholar] [CrossRef] [PubMed]
- Palazzotto, E.; Weber, T. Omics and multi-omics approaches to study the biosynthesis of secondary metabolites in microorganisms. Curr. Opin. Microbiol. 2018, 45, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Hemmerich, J.; Noack, S.; Wiechert, W.; Oldiges, M. Microbioreactor Systems for Accelerated Bioprocess Development. Biotechnol. J. 2018, 13, 1700141. [Google Scholar] [CrossRef] [PubMed]
- Chase, M.; Duetz, W.A.; Bills, G. Miniaturization of Fermentations. In Manual of Industrial Microbiology and Biotechnology, 3rd ed.; Lynd, L.R., Zhao, H., Katz, L., Baltz, R.H., Bull, A.T., Junker, B., Masurekar, P., Davies, J.E., Reeves, C.D., Demain, A.L., Eds.; American Society of Microbiology: Washington, DC, USA, 2010; pp. 99–116. ISBN 978-1-55581-512-7. [Google Scholar]
- Palomo, S.; González, I.; de la Cruz, M.; Martín, J.; Tormo, J.; Anderson, M.; Hill, R.; Vicente, F.; Reyes, F.; Genilloud, O. Sponge-Derived Kocuria and Micrococcus spp. as Sources of the New Thiazolyl Peptide Antibiotic Kocurin. Mar. Drugs 2013, 11, 1071–1086. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Haque, S.; Niwas, R.; Srivastava, A.; Pasupuleti, M.; Tripathi, C.K.M. Strategies for Fermentation Medium Optimization: An In-Depth Review. Front. Microbiol. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Managamuri, U.; Vijayalakshmi, M.; Ganduri, V.S.R.K.; Rajulapati, S.B.; Bonigala, B.; Kalyani, B.S.; Poda, S. Isolation, identification, optimization, and metabolite profiling of Streptomyces sparsus VSM-30. 3 Biotech 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, M.; Li, Y.; Cui, Y.; Zhang, Y.; Wang, Z.; Wang, M.; Huang, Y. Application of response surface methodology to optimize the production of antimicrobial metabolites by Micromonospora Y15. Biotechnol. Biotechnol. Equip. 2017, 31, 1016–1025. [Google Scholar] [CrossRef]
- Zhao, H.G.; Wang, M.; Lin, Y.Y.; Zhou, S.L. Optimization of culture conditions for penicilazaphilone C production by a marine-derived fungus Penicillium sclerotiorum M-22. Lett. Appl. Microbiol. 2018, 66, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Ven, K.; Vanspauwen, B.; Pérez-Ruiz, E.; Leirs, K.; Decrop, D.; Gerstmans, H.; Spasic, D.; Lammertyn, J. Target Confinement in Small Reaction Volumes Using Microfluidic Technologies: A Smart Approach for Single-Entity Detection and Analysis. ACS Sens. 2018, 3, 264–284. [Google Scholar] [CrossRef] [PubMed]
- Eribol, P.; Uguz, A.K.; Ulgen, K.O. Screening applications in drug discovery based on microfluidic technology. Biomicrofluidics 2016, 10, 011502. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Ye, C.; Liu, L. Genome-scale biological models for industrial microbial systems. Appl. Microbiol. Biotechnol. 2018, 102, 3439–3451. [Google Scholar] [CrossRef] [PubMed]
- Toro, L.; Pinilla, L.; Avignone-Rossa, C.; Ríos-Estepa, R. An enhanced genome-scale metabolic reconstruction of Streptomyces clavuligerus identifies novel strain improvement strategies. Bioprocess Biosyst. Eng. 2018, 41, 657–669. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romano, S.; Jackson, S.A.; Patry, S.; Dobson, A.D.W. Extending the “One Strain Many Compounds” (OSMAC) Principle to Marine Microorganisms. Mar. Drugs 2018, 16, 244. https://doi.org/10.3390/md16070244
Romano S, Jackson SA, Patry S, Dobson ADW. Extending the “One Strain Many Compounds” (OSMAC) Principle to Marine Microorganisms. Marine Drugs. 2018; 16(7):244. https://doi.org/10.3390/md16070244
Chicago/Turabian StyleRomano, Stefano, Stephen A. Jackson, Sloane Patry, and Alan D. W. Dobson. 2018. "Extending the “One Strain Many Compounds” (OSMAC) Principle to Marine Microorganisms" Marine Drugs 16, no. 7: 244. https://doi.org/10.3390/md16070244
APA StyleRomano, S., Jackson, S. A., Patry, S., & Dobson, A. D. W. (2018). Extending the “One Strain Many Compounds” (OSMAC) Principle to Marine Microorganisms. Marine Drugs, 16(7), 244. https://doi.org/10.3390/md16070244





