Characterization and Genome Analysis of a Nicotine and Nicotinic Acid-Degrading Strain Pseudomonas putida JQ581 Isolated from Marine
Abstract
:1. Introduction
2. Results and Discussion
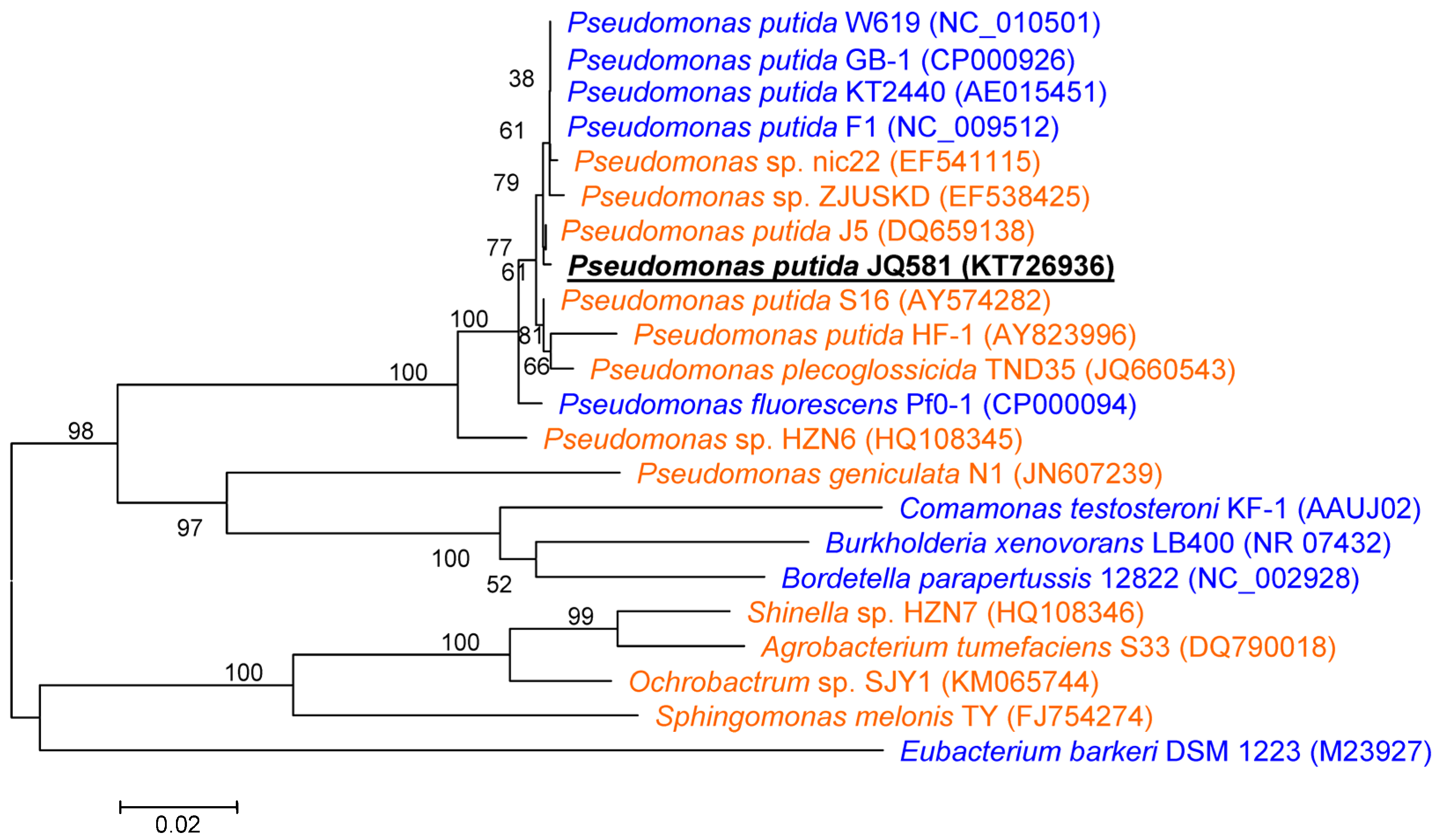
2.1. Isolation and Identification of Strain JQ581
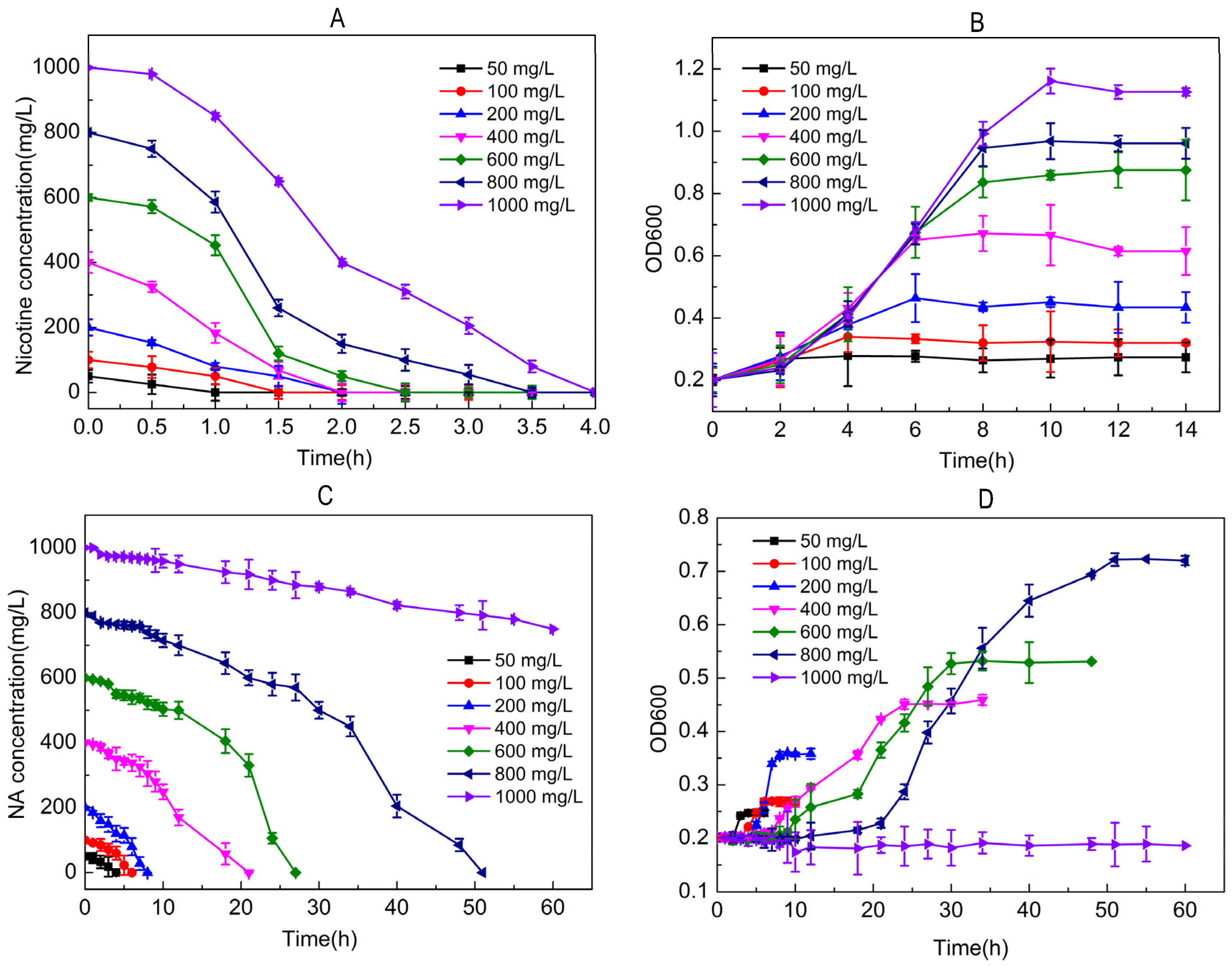
2.2. Utilization of Nicotine/NA for Growth by Strain JQ581
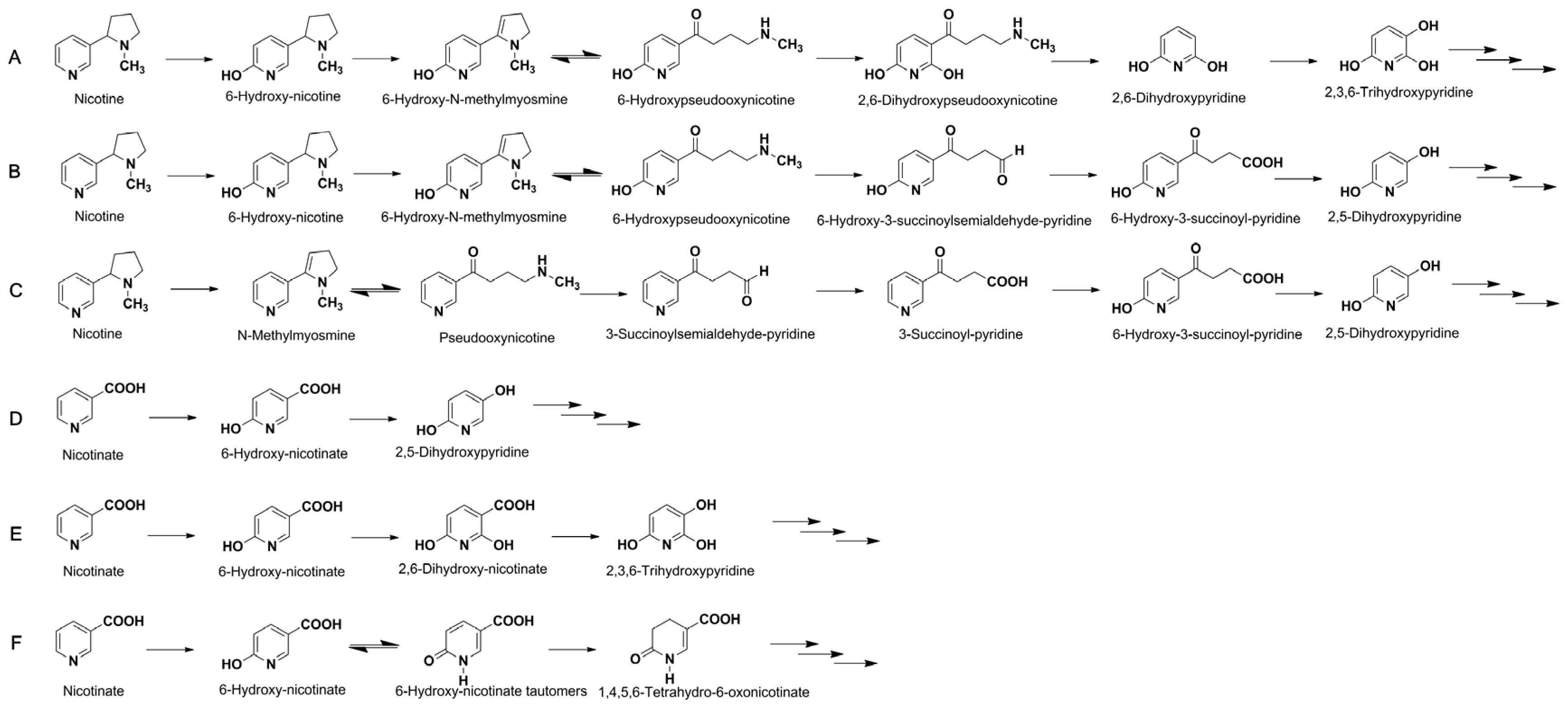
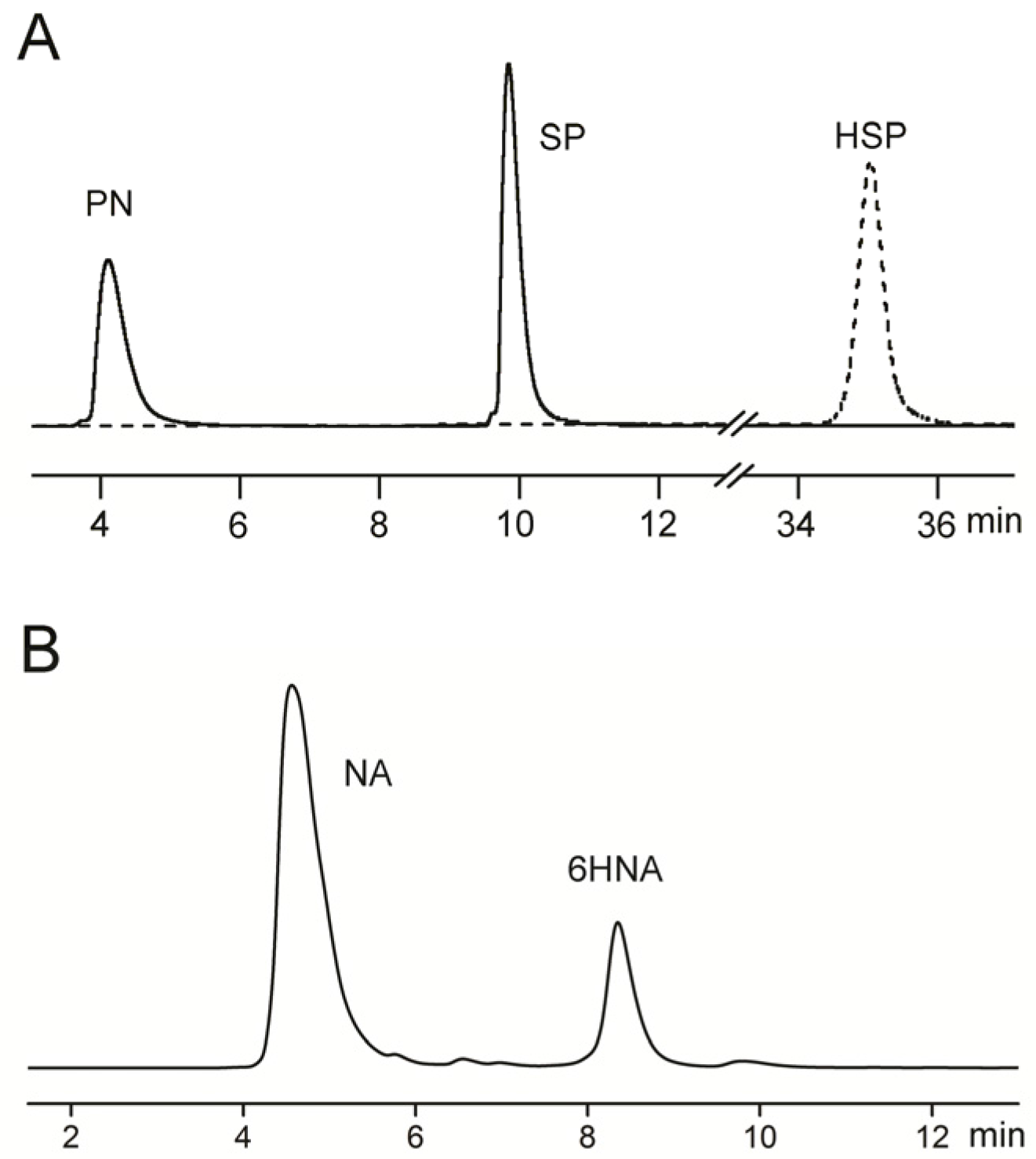
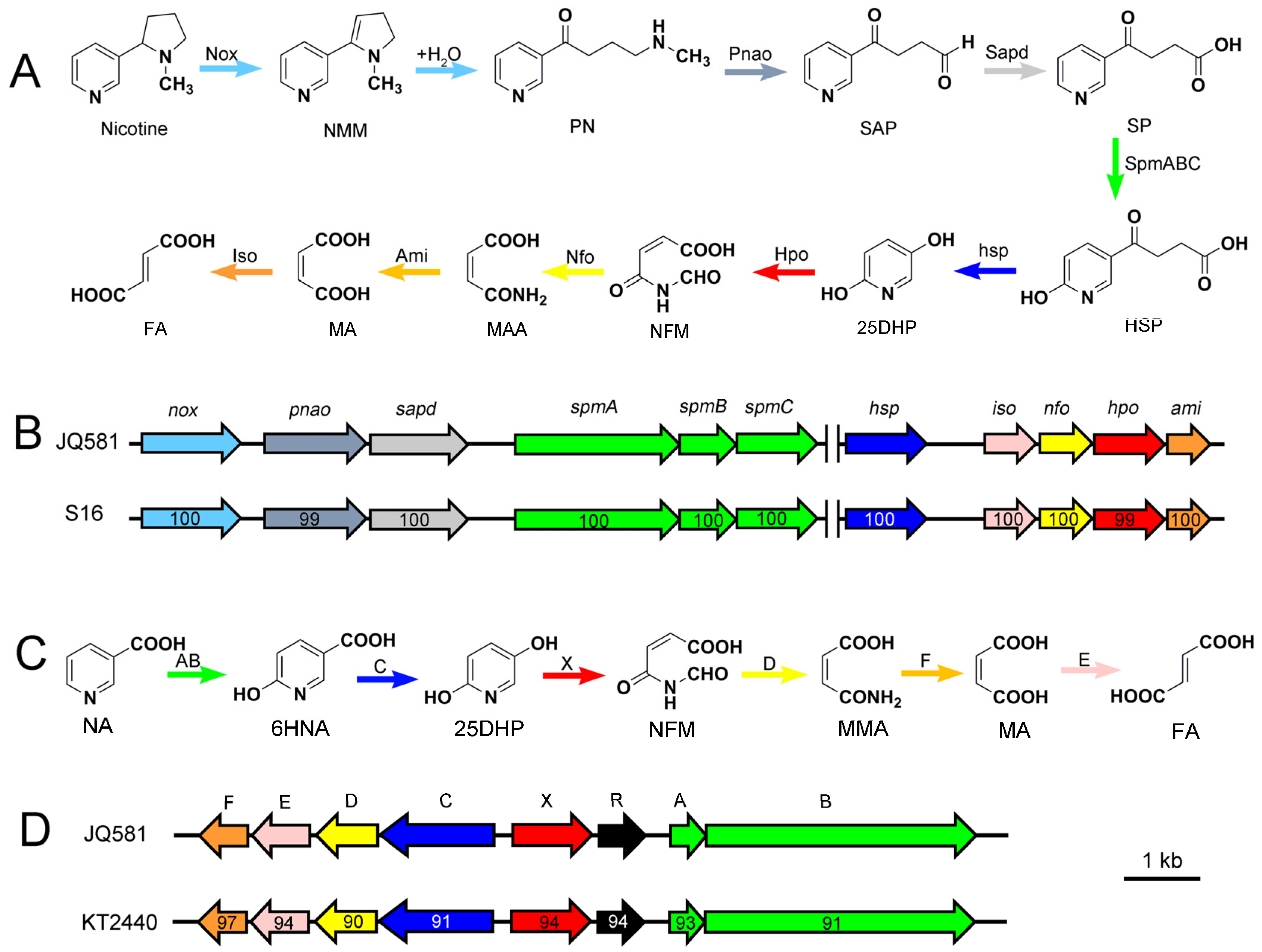
2.3. Metabolic Pathway of Nicotine and NA Biodegradation
2.4. Genome Features of Strain JQ581
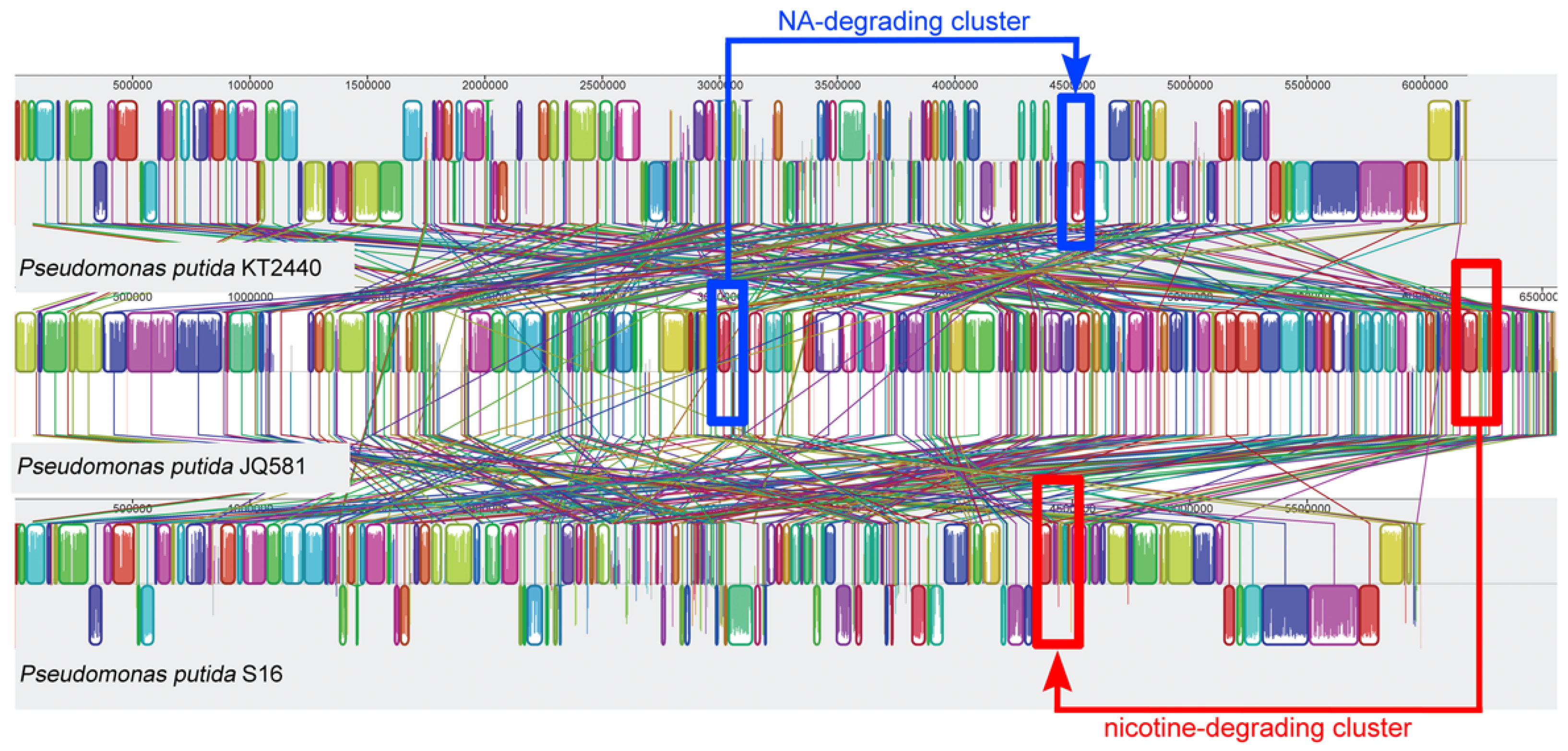
2.5. Genome Sequence Analysis and the Identification of Gene Clusters for Nicotine and NA Degradation
3. Materials and Methods
3.1. Chemicals and Media
3.2. Screening and Isolation of Bacteria of Degrading Nicotine and NA
3.3. Identification of Strain that Degrades Nicotine and NA
3.4. Biodegradation Assay
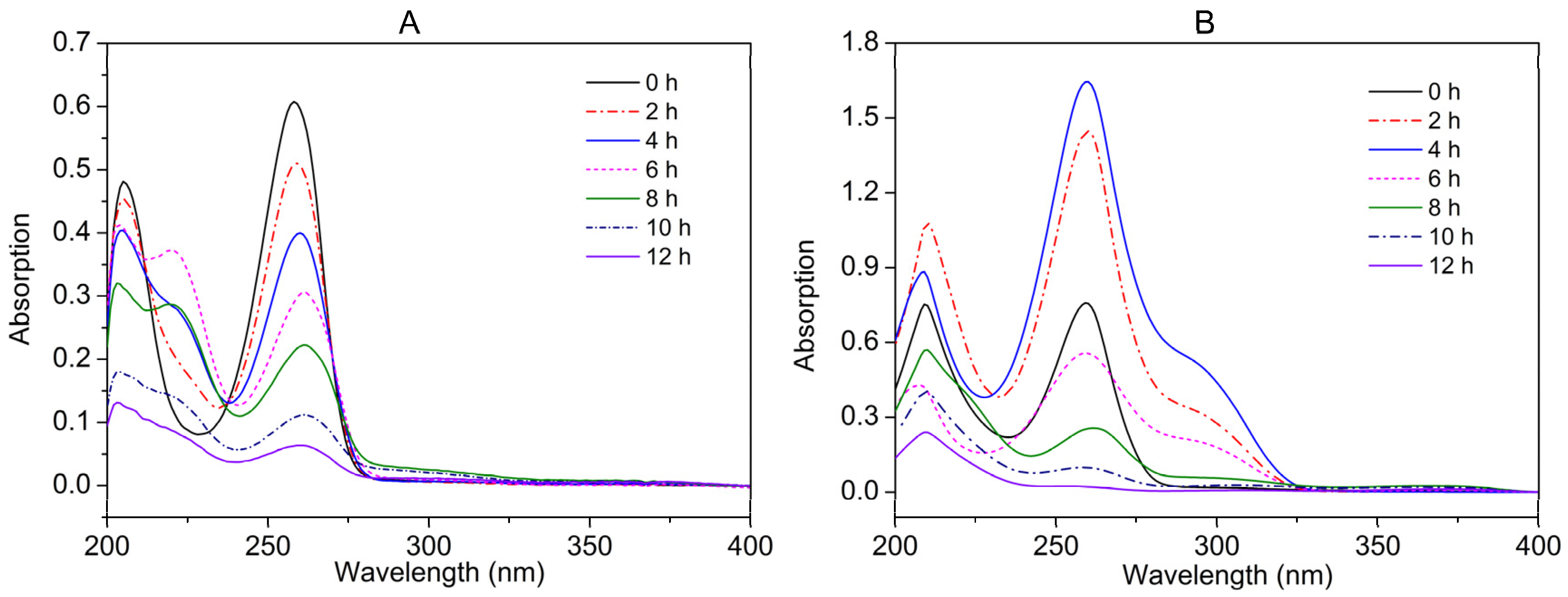
3.5. Identification of Nicotine/NA Biodegradation Metabolites
3.6. Analytical Methods
3.7. Draft Genome Sequencing and Bioinformatic Analysis of Strain JQ581
3.8. Deposition of the Nucleotide Sequence Accession Number
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Benowitz, N.L. Nicotine Addiction. N. Engl. J. Med. 2010, 362, 2295–2303. [Google Scholar] [CrossRef]
- Harris, A.C.; Tally, L.; Muelken, P.; Banal, A.; Schmidt, C.E.; Cao, Q.; LeSage, M.G. Effects of nicotine and minor tobacco alkaloids on intracranial-self-stimulation in rats. Drug Alcohol Depend. 2015, 153, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Civilini, M.; Domenis, C.; Sebastianutto, N.; deBertoldi, M. Nicotine decontamination of tobacco agro-industrial waste and its degradation by micro-organisms. Waste Manag. Res. 1997, 15, 349–358. [Google Scholar] [CrossRef]
- Wang, S.N.; Xu, P.; Tang, H.Z.; Meng, J.; Liu, X.L.; Ma, C.Q. “Green” route to 6-hydroxy-3-succinoyl-pyridine from (S)-nicotine of tobacco waste by whole cells of a Pseudomonas sp. Environ. Sci. Technol. 2005, 39, 6877–6880. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ma, G.; Chen, T.; Hou, Y.; Yang, S.; Zhang, K.-Q.; Yang, J. Nicotine-degrading microorganisms and their potential applications. Appl. Microbiol. Biotechnol. 2015, 99, 3775–3785. [Google Scholar] [CrossRef] [PubMed]
- Hukkanen, J.; Jacob, P.; Benowitz, N. Metabolism and disposition kinetics of nicotine. Pharmacol. Rev. 2005, 57, 79–115. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Wang, L.; Wang, W.; Yu, H.; Zhang, K.; Yao, Y.; Xu, P. Systematic unraveling of the unsolved pathway of nicotine degradation in Pseudomonas. PLoS Genet. 2013, 9, e1003923. [Google Scholar] [CrossRef] [PubMed]
- Alhapel, A.; Darley, D.J.; Wagener, N.; Eckel, E.; Elsner, N.; Pierik, A.J. Molecular and functional analysis of nicotinate catabolism in Eubacterium barkeri. Proc. Natl. Acad. Sci. USA 2006, 103, 12341–12346. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, J.I.; Canales, A.; Jiménezbarbero, J.; Ginalski, K.; Rychlewski, L.; García, J.L.; Díaz, E. Deciphering the genetic determinants for aerobic nicotinic acid degradation: The nic cluster from Pseudomonas putida KT2440. Proc. Natl. Acad. Sci. USA 2008, 105, 11329–11334. [Google Scholar] [CrossRef] [PubMed]
- Brandsch, R. Microbiology and biochemistry of nicotine degradation. Appl. Microbiol. Biotechnol. 2006, 69, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, X.; Duan, Y.; Zhang, K.-Q.; Yang, J. Biotransformation of nicotine by microorganism: The case of Pseudomonas spp. Appl. Microbiol. Biotechnol. 2010, 86, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Wei, Y.; Qiu, J.; Wen, R.; Hong, J.; Liu, W. Isolation, transposon mutagenesis, and characterization of the novel nicotine-degrading strain Shinella sp. HZN7. Appl. Microbiol. Biotechnol. 2013, 98, 2625–2636. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Wei, Y.; Ma, Y.; Wen, R.; Wen, Y.; Liu, W. A novel (S)-6-hydroxynicotine oxidase gene from Shinella sp. strain HZN7. Appl. Environ. Microbiol. 2014, 80, 5552–5560. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Liu, M.; Wen, R.; Zhang, D.; Hong, J. Regulators essential for nicotine degradation in Shinella sp. HZN7. Proc. Biochem. 2015, 50, 1947–1950. [Google Scholar] [CrossRef]
- Qiu, J.; Li, N.; Lu, Z.; Yang, Y.; Ma, Y.; Niu, L.; He, J.; Liu, W. Conversion of nornicotine to 6-hydroxy-nornicotine and 6-hydroxy-myosmine by Shinella sp. strain HZN7. Appl. Microbiol. Biotechnol. 2016, 100, 10019–10029. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Yang, Y.; Zhang, J.; Wang, H.; Ma, Y.; He, J.; Lu, Z. The complete genome sequence of the nicotine-degrading bacterium Shinella sp. HZN7. Front. Microbiol. 2016, 7, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Gurusamy, R.; Natarajan, S. Current status on biochemistry and molecular biology of microbial degradation of nicotine. Sci. World J. 2013, 2013, 1653–1656. [Google Scholar] [CrossRef] [PubMed]
- Harvey, Z.H.; Snider, M.J. Draft genome sequence of the nicotinate-metabolizing soil bacterium Bacillus niacini DSM 2923. Genome Announc. 2014, 2, e01251-14. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Chen, T.; Ma, P.J.; Shang, G.D.; Dai, Y.J.; Yuan, S.N. Cloning, expression and functional analysis of nicotinate dehydrogenase gene cluster from Comamonas testosteroni JA1 that can hydroxylate 3-cyanopyridine. Biodegradation 2010, 21, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Yao, Y.; Zhang, D.; Meng, X.; Wang, L.; Yu, H.; Ma, L.; Xu, P. A novel NADH-dependent and FAD-containing hydroxylase is crucial for nicotine degradation by Pseudomonas putida. J. Biol. Chem. 2011, 286, 39179–39187. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.Z.; Wang, L.J.; Meng, X.Z.; Ma, L.Y.; Wang, S.N.; He, X.F.; Wu, G.; Xu, P. Novel nicotine oxidoreductase-encoding gene involved in nicotine degradation by Pseudomonas putida Strain S16. Appl. Environ. Microbiol. 2009, 75, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.Z.; Wang, S.N.; Ma, L.Y.; Meng, X.Z.; Deng, Z.X.; Zhang, D.; Ma, C.Q.; Xu, P. A novel gene, encoding 6-hydroxy-3-succinoylpyridine hydroxylase, involved in nicotine degradation by Pseudomonas putida strain S16. Appl. Environ. Microbiol. 2008, 74, 1567–1574. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Li, X.; Yang, J.; Gong, X.; Li, B.; Zhang, K.-Q. Isolation of nicotine-degrading bacterium Pseudomonas sp. Nic22, and its potential application in tobacco processing. Int. Biodeterior. Biodegrad. 2008, 62, 226–231. [Google Scholar] [CrossRef]
- Sun, K.; Zhu, C.; Zhong, W.; Chen, J.; Ye, Z.; Liu, P.; Zhou, Q. Isolation and characterization of a high nicotine-degrading bacterium Pseudomonas sp. strain ZUTSKD. Acta Sci. Circumstantiae 2008, 28, 1294–1301. (In Chinese) [Google Scholar]
- Xia, Z.; Zhang, W.; Lei, L.; Liu, X.; Wei, H.L. Genome-wide investigation of the genes involved in nicotine metabolism in Pseudomonas putida J5 by Tn 5 transposon mutagenesis. Appl. Microbiol. Biotechnol. 2015, 99, 6503–6514. [Google Scholar] [CrossRef] [PubMed]
- Ruan, A.; Min, H.; Peng, X.; Huang, Z. Isolation and characterization of Pseudomonas sp. strain HF-1, capable of degrading nicotine. Res. Microbiol. 2005, 156, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Raman, G.; Mohan, K.; Manohar, V.; Sakthivel, N. Biodegradation of nicotine by a novel nicotine-degrading bacterium, Pseudomonas plecoglossicida TND35 and its new biotransformation intermediates. Biodegradation 2014, 25, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Ma, Y.; Chen, L.; Wu, L.; Wen, Y.; Liu, W. A sirA-like gene, sirA2, is essential for 3-succinoyl-pyridine metabolism in the newly isolated nicotine-degrading Pseudomonas sp. HZN6 strain. Appl. Microbiol. Biotechnol. 2011, 92, 1023–1032. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Ma, Y.; Wen, Y.; Chen, L.; Wu, L.; Liu, W. Functional identification of two novel genes from Pseudomonas sp. strain HZN6 involved in the catabolism of nicotine. Appl. Environ. Microbiol. 2012, 78, 2154–2160. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Ma, Y.; Zhang, J.; Wen, Y.; Liu, W. Cloning of a novel nicotine oxidase gene from Pseudomonas sp. strain HZN6 whose product nonenantioselectively degrades nicotine to pseudooxynicotine. Appl. Environ. Microbiol. 2013, 79, 2164–2171. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, L.; Huang, K.; Wang, W.; Nie, X.; Jiang, Y.; Li, P.; Liu, S.; Xu, P.; Tang, H. Physiological and biochemical characterization of a novel nicotine-degrading bacterium Pseudomonas geniculata N1. PLoS ONE 2014, 9, e84399. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Huang, H.; Xie, K.; Xu, P. Identification of nicotine biotransformation intermediates by Agrobacterium tumefaciens strain S33 suggests a novel nicotine degradation pathway. Appl. Microbiol. Biotechnol. 2012, 95, 1567–1578. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Tang, H.; Li, Y.; Xu, P. Novel genes coding for a molybdenum-containing nicotine hydroxylase in the VPP nicotine degradation pathway. Appl. Environ. Microbiol. 2015, 81, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yang, G.; Wang, X.; Yao, Y.; Min, H.; Lu, Z. Nicotine degradation by two novel bacterial isolates of Acinetobacter sp. TW and Sphingomonas sp. TY and their responses in the presence of neonicotinoid insecticides. World J. Microbiol. Biotechnol. 2011, 27, 1633–1640. [Google Scholar] [CrossRef]
- Zulema, U.; Lázaro, M.; Ana, S.; Estrella, D.; Ramos, J.L. Analysis of the core genome and pangenome of Pseudomonas putida. Environ. Microbiol. 2015, 18, 3268–3283. [Google Scholar]
- Yao, L.; Zhang, J.J.; Yu, L.L.; Chen, Q.; Zhu, J.C.; He, J.; Ding, D.R. Rhizorhabdus dicambivorans sp. nov., a dicamba-degrading bacterium isolated from compost. Int. J. Syst. Evol. Microbiol. 2016, 66, 3317–3323. [Google Scholar] [PubMed]
- Chen, K.; Xu, X.; Zhang, L.; Gou, Z.; Li, S.; Freilich, S.; Jiang, J. Comparison of four Comamonas catabolic plasmids reveals the evolution of pbhb to catabolize haloaromatics. Appl. Environ. Microbiol. 2015, 82, 1401–1411. [Google Scholar] [CrossRef] [PubMed]
- Aziz, R.K.; Bartels, D.; Best, A.A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M. The RAST Server: Rapid annotations using subsystems technology. BMC Genom. 2008, 9, 75. [Google Scholar] [CrossRef] [PubMed]
| Tests | Result | Tests | Result |
|---|---|---|---|
| 1 Negative Control | − | 49 Methyl Pyruvate | w |
| 2 Dextrin | − | 50 d-Lactic Acid Methyl Ester | − |
| 3 d-Maltose | − | 51 α-Keto-Glutaric Acid | w |
| 4 d-Trehalose | − | 52 Nalidixic Acid | + |
| 5 d-Cellobiose | − | 53 Lithium Chloride | w |
| 6 Gentiobiose | − | 54 Potassium Tellurite | + |
| 7 Sucrose | − | 55 Tween 40 | − |
| 8 d-Turanose | − | 56 γ-Amino-Butryric Acid | + |
| 9 Stachyose | − | 57 α-Hydroxy-Butyric Acid | − |
| 10 d-Raffinose | − | 58 β-Hydroxy-d,l-Butyric Acid | − |
| 11 α-d-Lactose | − | 59 α-Keto-Butyric Acid | − |
| 12 d-Melibiose | − | 60 Acetoacetic Acid | w |
| 13 β-Methyl-d-Glucoside | − | 61 Gelatin | − |
| 14 d-Salicin | − | 62 Glycyl-l-Prolin | − |
| 15 N-Acetyl-d-Glucosamine | − | 63 l-Alanine | + |
| 16 N-Acetyl-β-DMannosamine | − | 64 l-Arginine | + |
| 17 N-Acetyl-d-Galactosamine | − | 65 l-Aspartic Acid | + |
| 18 N-AcetylNeuraminic Acid | − | 66 l-Glutamic | + |
| 19 α-d-Glucose | w | 67 l-Histidine | + |
| 20 d-Mannose | w | 68 l-Pyroglutamic | + |
| 21 d-Fructose | w | 69 l-Serine | w |
| 22 d-Galactose | w | 70 Lincomycin | + |
| 23 3-Methyl Glucose | − | 71 Guanidine HCl | w |
| 24 d-Fucose | w | 72 Niaproof 4 | + |
| 25 l-Fucose | w | 73 Pectin | − |
| 26 l-Rhamnose | − | 74 d-Galacturonic Acid | + |
| 27 Inosine | − | 75 l-Galactonic Acid Lactone | + |
| 28 1% Sodium Lactate | + | 76 d-Gluconic Acid | + |
| 29 Fusidic Acid | + | 77 d-Glucuronic Acid | + |
| 30 d-Serine | + | 78 Glucuronamide | + |
| 31 d-Sorbitol | − | 79 Mucic Acid | + |
| 32 d-Mannitol | − | 80 Quinic Acid | + |
| 33 d-Arabitol | − | 81 d-Saccharic Acid | + |
| 34 myo-Inositol | − | 82 Vancomycin | + |
| 35 Glycerol | w | 83 Tetrazolium Violet | + |
| 36 d-Glucose-6-PO4 | − | 84 Tetrazolium Blue | + |
| 37 d-Fructose-6-PO4 | + | 85 l-Lactic Acid | w |
| 38 d-Aspartic Acid | + | 86 Citric Acid | + |
| 39 d-Serine | + | 87 d-Malic Acid | + |
| 40 Troleandomycin | + | 88 l-Malic Acid | + |
| 41 Rifamycin SV | + | 89 Bromo-Succinic Acid | w |
| 42 Minocycline | − | 90 Formic Acid | − |
| 43 Positive Control | + | 91 Propionic Acid | w |
| 44 pH 6 | + | 92 Acetic Acid | w |
| 45 pH 5 | + | 93 Aztreonam | w |
| 46 1% NaCl | + | 94 Sodium Butyrate | − |
| 47 4% NaCl | − | 95 Sodium Bromate | − |
| 48 7% NaCl | − | 96 p-Hydroxy-Phenylacetic Acid | w |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, A.; Qiu, J.; Chen, D.; Ye, J.; Wang, Y.; Tong, L.; Jiang, J.; Chen, J. Characterization and Genome Analysis of a Nicotine and Nicotinic Acid-Degrading Strain Pseudomonas putida JQ581 Isolated from Marine. Mar. Drugs 2017, 15, 156. https://doi.org/10.3390/md15060156
Li A, Qiu J, Chen D, Ye J, Wang Y, Tong L, Jiang J, Chen J. Characterization and Genome Analysis of a Nicotine and Nicotinic Acid-Degrading Strain Pseudomonas putida JQ581 Isolated from Marine. Marine Drugs. 2017; 15(6):156. https://doi.org/10.3390/md15060156
Chicago/Turabian StyleLi, Aiwen, Jiguo Qiu, Dongzhi Chen, Jiexu Ye, Yuhong Wang, Lu Tong, Jiandong Jiang, and Jianmeng Chen. 2017. "Characterization and Genome Analysis of a Nicotine and Nicotinic Acid-Degrading Strain Pseudomonas putida JQ581 Isolated from Marine" Marine Drugs 15, no. 6: 156. https://doi.org/10.3390/md15060156
APA StyleLi, A., Qiu, J., Chen, D., Ye, J., Wang, Y., Tong, L., Jiang, J., & Chen, J. (2017). Characterization and Genome Analysis of a Nicotine and Nicotinic Acid-Degrading Strain Pseudomonas putida JQ581 Isolated from Marine. Marine Drugs, 15(6), 156. https://doi.org/10.3390/md15060156






