Collagen from the Marine Sponges Axinella cannabina and Suberites carnosus: Isolation and Morphological, Biochemical, and Biophysical Characterization
Abstract
:1. Introduction
2. Results
2.1. Isolation of Sponge Collagen
2.2. Examination of Surface Morphology
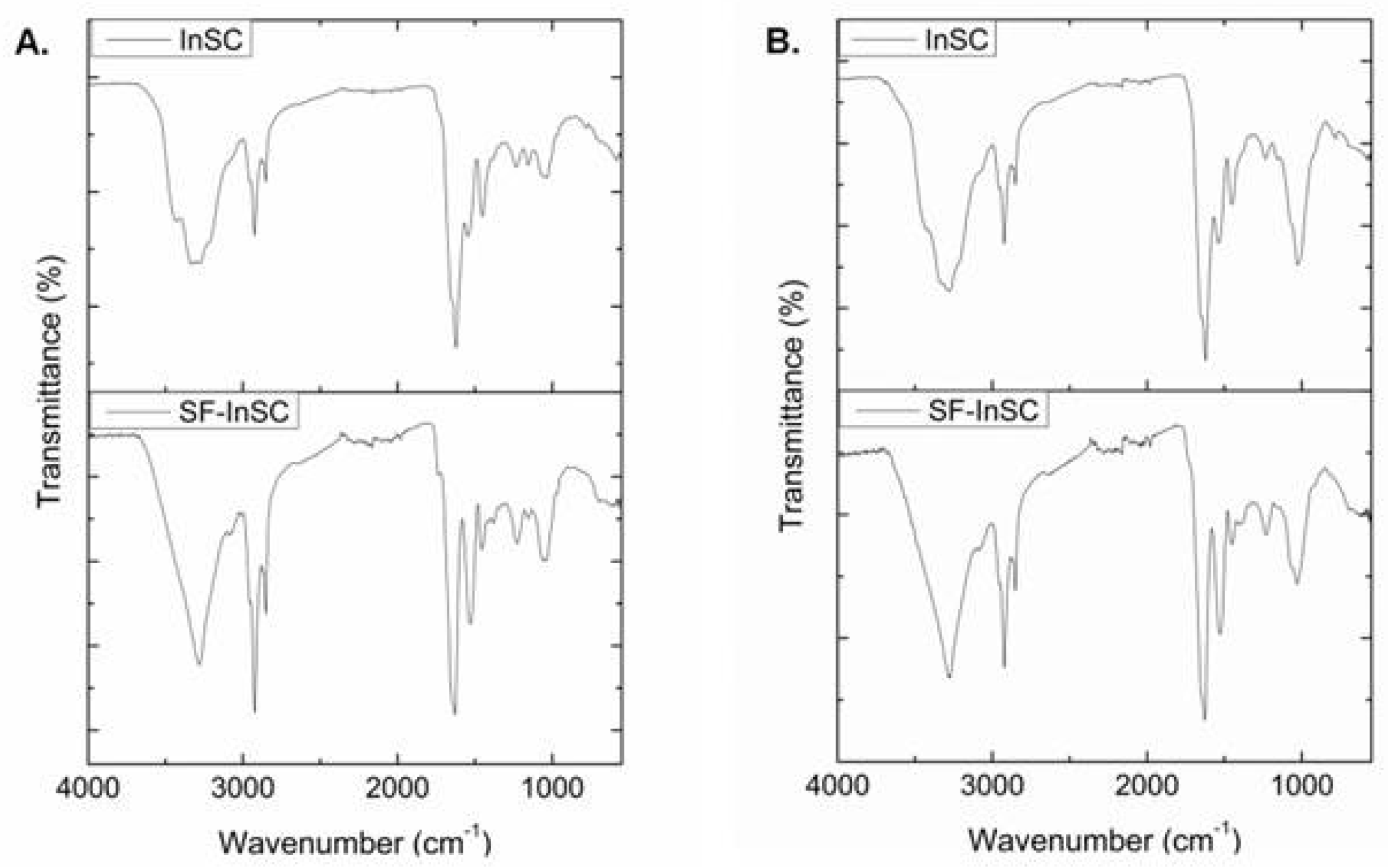
2.3. Infrared Spectroscopic Analysis
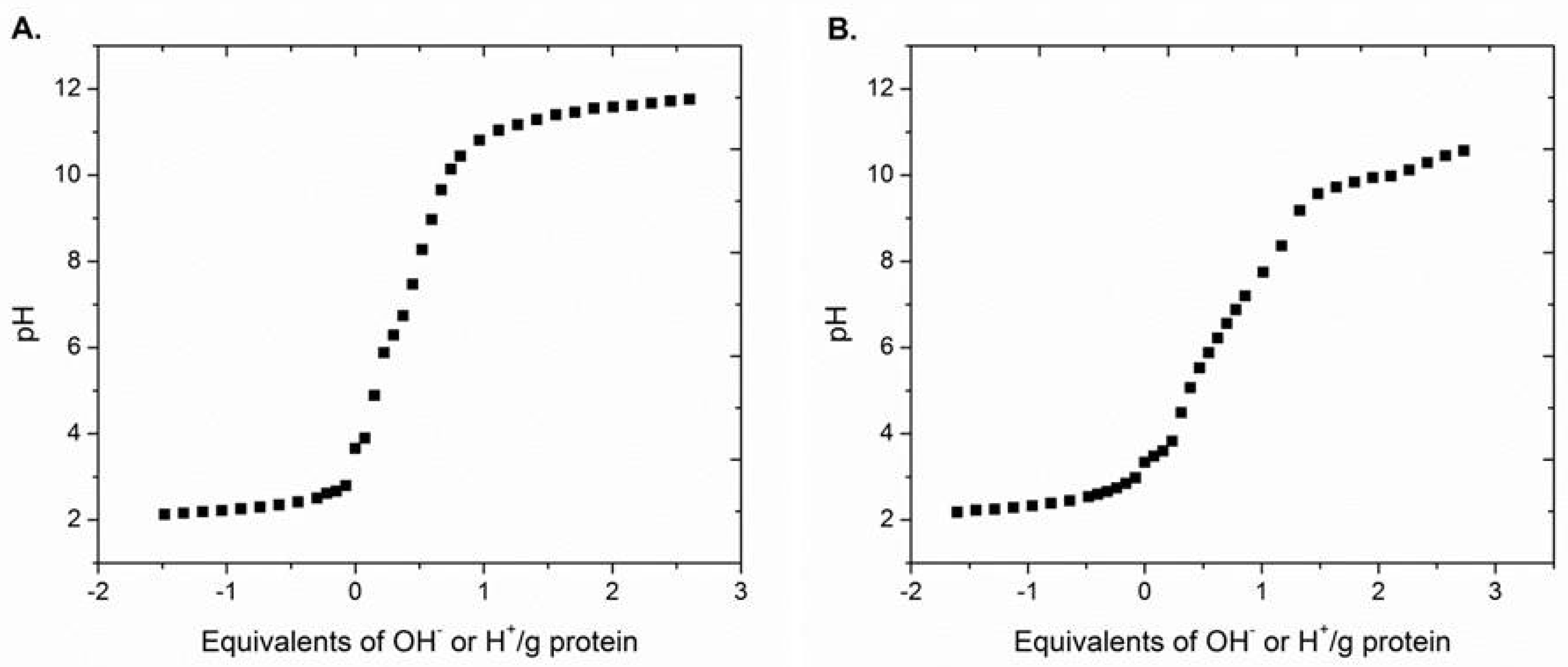
2.4. Isoelectric Point Determination
2.5. Amino Acid Profile
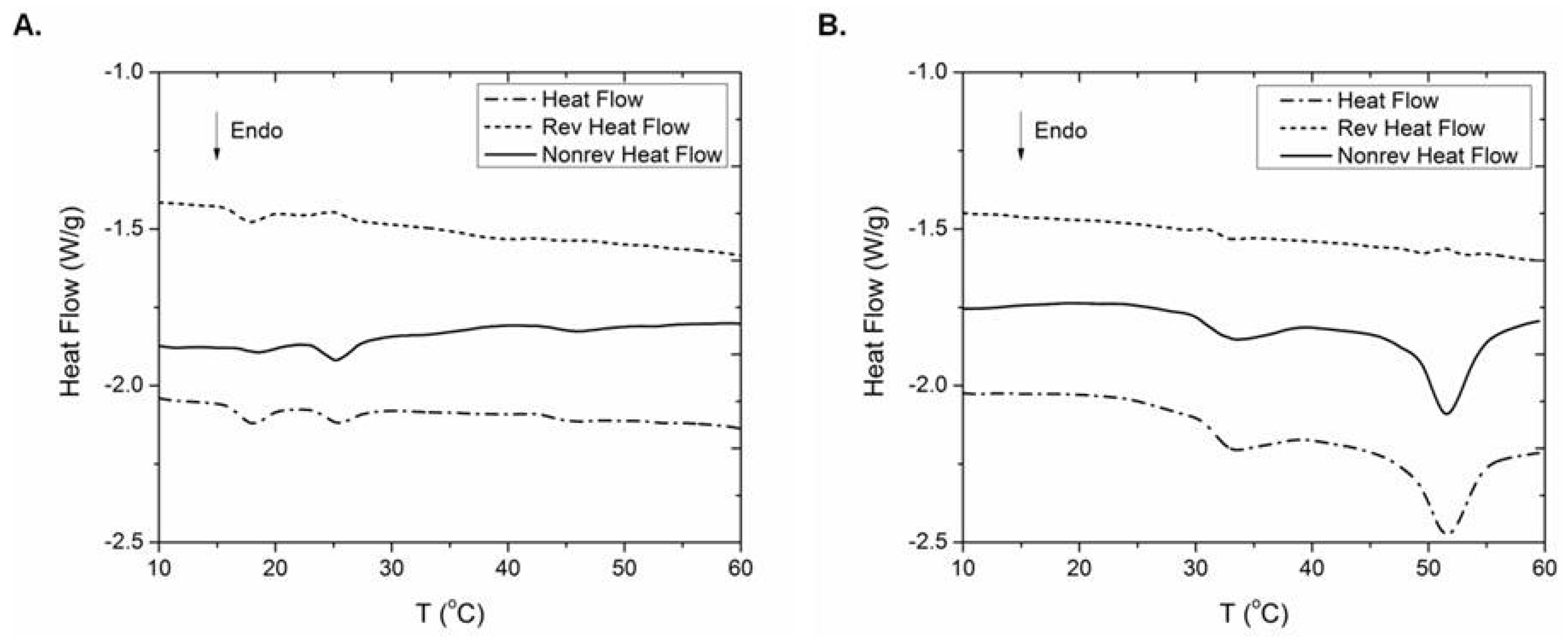
2.6. Thermal Behaviour
3. Discussion
4. Materials and Methods
4.1. Animal Material
4.2. Chemicals
4.3. Isolation of Insoluble Collagen, Intercellular Collagen, and Spongin-Like Collagen
4.4. Removal of Spicules from Insoluble Collagen
4.5. Electron Microscopy
4.6. Infrared Spectroscopy
4.7. Amino Acid Analysis
4.8. Titration
4.9. Differential Scanning Calorimetry
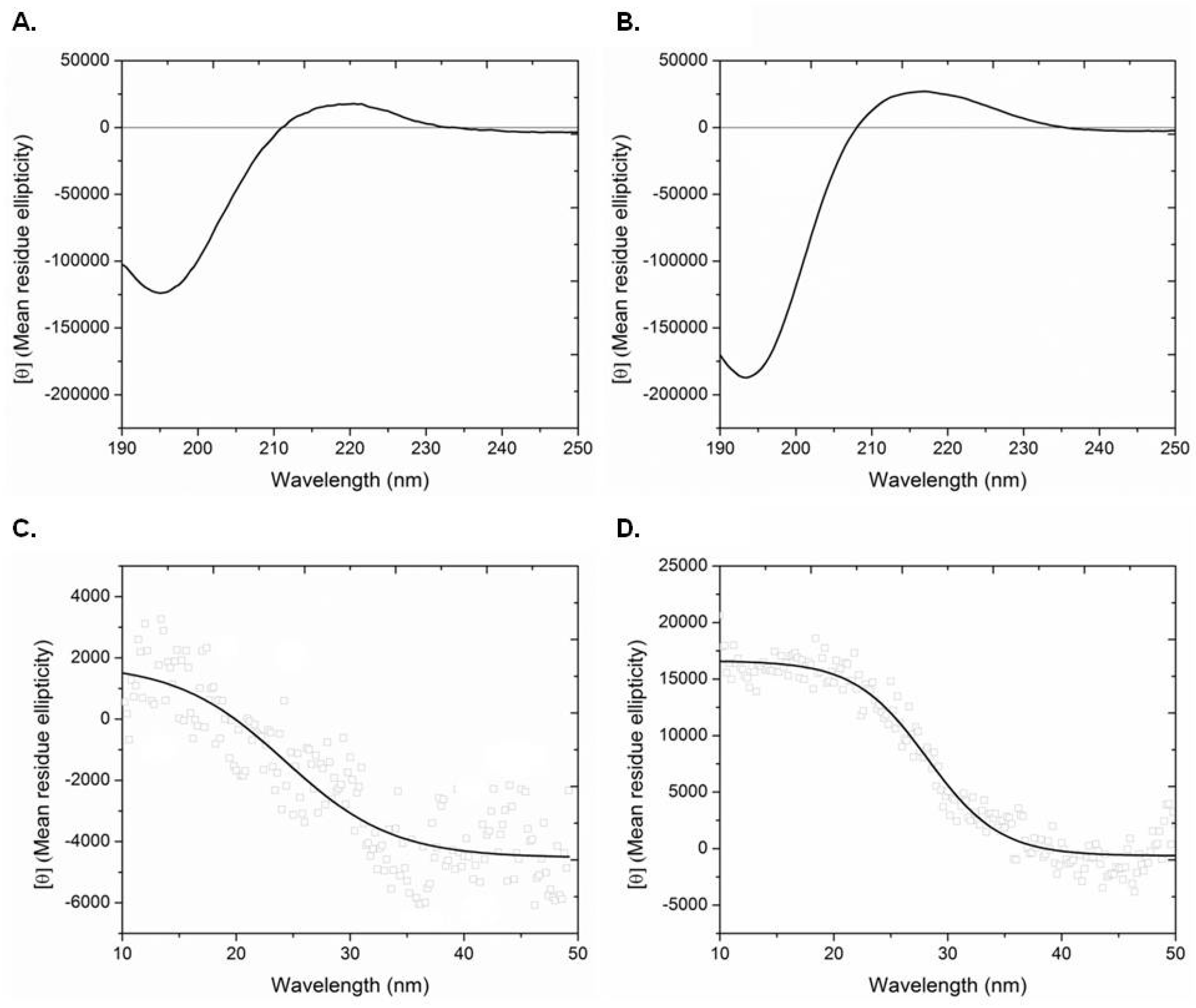
4.10. Circular Dichroism Spectroscopy
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gelse, K.; Pöschl, E.; Aigner, T. Collagens-structure, function, and biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1531–1546. [Google Scholar] [CrossRef] [PubMed]
- Myllyharju, J.; Kivirikko, K.I. Collagens, modifying enzymes and their mutations in humans, flies and worms. Trends Genet. 2004, 20, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.H.; Moreira-Silva, J.; Marques, A.L.P.; Domingues, A.; Bayon, Y.; Reis, R.L. Marine origin collagens and its potential applications. Mar. Drugs 2014, 12, 5881–5901. [Google Scholar] [CrossRef] [PubMed]
- Meena, C.; Mengi, S.A.; Deshpande, S.G. Biomedical and industrial applications of collagen. J. Chem. Sci. 1999, 111, 319–329. [Google Scholar] [CrossRef]
- Berillis, P. Marine collagen: Extraction and applications. In Research Trends in Biochemistry, Molecular Biology and Microbiology; Madhukar, S., Ed.; SM Group: Dover, DE, USA, 2015; pp. 1–13. [Google Scholar]
- Venugopal, V. Marine Products for Healthcare: Functional and Bioactive Nutraceutical Compounds from the Ocean; CRC Press: Boca Raton, FI, USA, 2009. [Google Scholar]
- Patino, M.G.; Neiders, M.E.; Andreana, S.; Noble, B.; Cohen, R.E. Collagen: An overview. Implant Dent. 2002, 11, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Friess, W. Collagen-biomaterial for drug delivery. Eur. J. Pharm. Biopharm. 1998, 45, 113–136. [Google Scholar] [CrossRef]
- Francis, G.; Thomas, J. Isolation and chemical characterization of collagen in bovine pulmonary tissues. Biochem. J. 1975, 145, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Lynn, A.K.; Yannas, I.V.; Bonfield, W. Antigenicity and immunogenicity of collagen. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 71, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Arvanitoyannis, I.S.; Kassaveti, A. Fish industry waste: Treatments, environmental impacts, current and potential uses. Int. J. Food Sci. Technol. 2008, 43, 726–745. [Google Scholar] [CrossRef]
- Leary, D.; Vierros, M.; Hamon, G.; Arico, S.; Monagle, C. Marine genetic resources: A review of scientific and commercial interest. Mar. Policy 2009, 33, 183–194. [Google Scholar] [CrossRef]
- Garrone, R. The evolution of connective tissue. Phylogenetic distribution and modifications during development. Prog. Clin. Biol. Res. 1981, 54, 141–149. [Google Scholar] [PubMed]
- Boot-Handford, R.P.; Tuckwell, D.S. Fibrillar collagen: The key to vertebrate evolution? A tale of molecular incest. BioEssays 2003, 25, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Simon, P.; Lichte, H.; Formanek, P.; Lehmann, M.; Huhle, R.; Carrillo-Cabrera, W.; Harscher, A.; Ehrlich, H. Electron holography of biological samples. Micron 2008, 39, 229–256. [Google Scholar] [CrossRef] [PubMed]
- Garrone, R. The Collagen of the Porifera. In Biology of Invertebrate and Lower Vertebrate Collagens; Bairati, A., Garrone, R., Eds.; NATO ASI Series; Plenum Press: New York, NY, USA, 1985; pp. 157–175. [Google Scholar]
- Exposito, J.-Y.; Cluzel, C.; Garrone, R.; Lethias, C. Evolution of collagens. Anat. Rec. 2002, 268, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Mehbub, M.F.; Lei, J.; Franco, C.; Zhang, W. Marine sponge derived natural products between 2001 and 2010: Trends and opportunities for discovery of bioactives. Mar. Drugs 2014, 12, 4539–4577. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2016, 33, 382–431. [Google Scholar] [CrossRef] [PubMed]
- Costantino, V.; Fattorusso, E.; Imperatore, C.; Mangoni, A.; Freigang, S.; Teyton, L. Glycolipids from sponges. 18. Corrugoside, a new immunostimulatory alpha-galactoglycosphingolipid from the marine sponge Axinella corrugata. Bioorg. Med. Chem. 2008, 16, 2077–2085. [Google Scholar] [CrossRef] [PubMed]
- Lamoral-Theys, D.; Fattorusso, E.; Mangoni, A.; Perinu, C.; Kiss, R.; Costantino, V. An in vitro valuation of the anticancer activity of diterpene isonitriles from the sponge Pseudoaxinella flava in apoptosis-sensitive and apoptosis-resistant cancer cell lines. J. Nat. Prod. 2011, 74, 2299–2303. [Google Scholar] [CrossRef] [PubMed]
- Gross, J.; Sokal, Z.; Rougvie, M. Structural and chemical studies on the connective tissue of marine sponges. J. Histochem. Cytochem. 1956, 4, 227–246. [Google Scholar] [CrossRef] [PubMed]
- Junqua, S.; Robert, L.; Garrone, R.; Pavans de Ceccatty, M.; Vacelet, J. Biochemical and morphological studies on collagens of horny sponges. Ircinia filaments compared to sponginess. Connect. Tissue Res. 1974, 2, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Diehl-Seifert, B.; Kurelec, B.; Zahn, R.K.; Dorn, A.; Jeričevic, B.; Uhlenbruck, G.; Müller, W.E.G. Attachment of sponge cells to collagen substrata: Effect of a collagen assembly factor. J. Cell Sci. 1985, 79, 271–285. [Google Scholar] [PubMed]
- Garrone, R.; Huc, A.; Junqua, S. Fine structure and physicochemical studies on the collagen of the marine sponge Chondrosia reniformis Nardo. J. Ultrastruct. Res. 1975, 52, 261–275. [Google Scholar] [CrossRef]
- Swatschek, D.; Schatton, W.; Kellermann, J.; Müller, W.E.G.; Kreuter, J. Marine sponge collagen: Isolation, characterization and effects on the skin parameters surface-pH, moisture and sebum. Eur. J. Pharm. Biopharm. 2002, 53, 107–113. [Google Scholar] [CrossRef]
- Pallela, R.; Bojja, S.; Janapala, V.R. Biochemical and biophysical characterization of collagens of marine sponge, Ircinia fusca (Porifera: Demospongiae: Irciniidae). Int. J. Biol. Macromol. 2011, 49, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.M.; Gentile, P.; Chiono, V.; Ciardelli, G. Collagen for bone tissue regeneration. Acta. Biomater. 2012, 8, 3191–3200. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, S.; Ehrlich, H.; Douglas, T.; Heinemann, C.; Worch, H.; Schatton, W.; Hanke, T. Ultrastructural studies on the collagen of the marine sponge Chondrosia reniformis Nardo. Biomacromolecules 2007, 8, 3452–3457. [Google Scholar] [CrossRef] [PubMed]
- Imhoff, J.M.; Garrone, R. Solubilization and characterization of Chondrosia reniformis sponge collagen. Connect. Tissue Res. 1983, 11, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, H.; Hanke, T.; Simon, P.; Goebel, C.; Heinemann, S.; Born, R.; Worch, H. Demineralisation von natürlichen Silikat-basierten Biomaterialien: Neue Strategie zur Isolation organischer Gerüststrukturen. BIOmaterialien 2005, 6, 297–302. [Google Scholar] [CrossRef]
- Katzman, R.L.; Lisowska, E.; Jeanloz, R.W. Invertebrate connective tissue. Isolation of D-arabinose from sponge acidic polysaccharide. Biochem. J. 1970, 119, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Barzideh, Z.; Abd Latiff, A.; Gan, C.-Y.; Benjakul, S.; Abd Karim, A. Isolation and characterisation of collagen from the ribbon jellyfish (Chrysaora sp.). Int. J. Food Sci. Technol. 2014, 49, 1490–1499. [Google Scholar] [CrossRef]
- Wang, L.; An, X.; Xin, Z.; Zhao, L.; Hu, Q. Isolation and characterization of collagen from the skin of deep-sea redfish (Sebastes mentella). J. Food Sci. 2007, 72, E450–E455. [Google Scholar] [CrossRef] [PubMed]
- Duan, R.; Zhang, J.; Du, X.; Yao, X.; Konno, K. Properties of collagen from skin, scale and bone of carp (Cyprinus carpio). Food Chem. 2009, 112, 702–706. [Google Scholar] [CrossRef]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Shahidi, F. Isolation and characterization of collagen from the cartilages of brownbanded bamboo shark (Chiloscyllium punctatum) and blacktip shark (Carcharhinus limbatus). LWT-Food Sci. Technol. 2010, 43, 792–800. [Google Scholar] [CrossRef]
- Ahmad, M.; Benjakul, S. Extraction and characterization of pepsin-solubilised collagen from the skin of unicorn leatherjacket (Aluterus monocerous). Food Chem. 2010, 120, 817–824. [Google Scholar] [CrossRef]
- Cao, H.; Xu, S.-Y. Purification and characterization of type II collagen from chick sternal cartilage. Food Chem. 2008, 108, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Kishimura, H.; Shahidi, F. Isolation and characterisation of collagen from the skin of brownbanded bamboo shark (Chiloscyllium punctatum). Food Chem. 2010, 119, 1519–1526. [Google Scholar] [CrossRef]
- Pati, F.; Adhikari, B.; Dhara, S. Isolation and characterization of fish scale collagen of higher thermal stability. Bioresour. Technol. 2010, 101, 3737–3742. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.; Choo, L.; Watson, P.H.; Halliday, W.C.; Mantsch, H.H. Beware of connective tissue proteins: Assignment and implications of collagen absorptions in infrared spectra of human tissues. Biochim. Biophys. Acta 1995, 1270, 1–6. [Google Scholar] [CrossRef]
- Plepis, A.M.D.G.; Goissis, G.; Das-Gupta, D.K. Dielectric and pyroelectric characterization of anionic and native collagen. Polym. Eng. Sci. 1996, 36, 2932–2938. [Google Scholar] [CrossRef]
- Ikoma, T.; Kobayashi, H.; Tanaka, J.; Walsh, D.; Mann, S. Physical properties of type I collagen extracted from fish scales of Pagrus major and Oreochromis niloticas. Int. J. Biol. Macromol. 2003, 32, 199–204. [Google Scholar] [CrossRef]
- Wong, D.W.S. Mechanism and Theory in Food Chemistry; Van Nostrand Reinhold: New York, NY, USA, 1989. [Google Scholar]
- Na, G.C. UV spectroscopic characterization of type I collagen. Collagen Relat. Res. 1988, 8, 315–330. [Google Scholar] [CrossRef]
- Junqua, S.; Lemonnier, M.; Robert, L. Glycoconjugates from “Spongia officinalis” (phylum porifera). Isolation, fractionation by affinity chromatography on lectins and partial characterization. Comp. Biochem. Physiol. B 1981, 69, 445–453. [Google Scholar] [CrossRef]
- Bischof, J.C.; He, X. Thermal stability of proteins. Ann. N. Y. Acad. Sci. 2006, 1066, 12–33. [Google Scholar] [CrossRef] [PubMed]
- Mu, C.; Li, D.; Lin, W.; Ding, Y.; Zhang, G. Temperature induced denaturation of collagen in acidic solution. Biopolymers 2007, 86, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Li, G. Non-isothermal kinetic analysis of the thermal denaturation of type I collagen in solution using isoconversional and multivariate non-linear regression methods. Polym. Degrad. Stab. 2010, 95, 2233–2240. [Google Scholar] [CrossRef]
- Miles, C.A. Kinetics of collagen denaturation in mammalian lens capsules studied by differential scanning calorimetry. Int. J. Biol. Macromol. 1993, 15, 265–271. [Google Scholar] [CrossRef]
- Miles, C.A.; Burjanadze, T.V.; Bailey, A.J. The kinetics of the thermal denaturation of collagen in unrestrained rat tail tendon determined by differential scanning calorimetry. J. Mol. Biol. 1995, 245, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Vyazovkin, S.; Vincent, L.; Sbirrazzuoli, N. Thermal denaturation of collagen analyzed by isoconversional method. Macromol. Biosci. 2007, 7, 1181–1186. [Google Scholar] [CrossRef] [PubMed]
- Nagai, T.; Ogawa, T.; Nakamura, T.; Ito, T.; Nakagawa, H.; Fujiki, K.; Nakao, M.; Yano, T. Collagen of edible jellyfish exumbrella. J. Sci. Food Agric. 1999, 79, 855–858. [Google Scholar] [CrossRef]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Nagai, T.; Tanaka, M. Characterisation of acid-soluble collagen from skin and bone of bigeye snapper (Priacanthus tayenus). Food Chem. 2005, 89, 363–372. [Google Scholar] [CrossRef]
- Nagai, T.; Araki, Y.; Suzuki, N. Collagen of the skin of ocellate puffer fish (Takifugu rubripes). Food Chem. 2002, 78, 173–177. [Google Scholar] [CrossRef]
- Muyonga, J.H.; Cole, C.G.B.; Duodu, K.G. Characterisation of acid soluble collagen from skins of young and adult Nile perch (Lates niloticus). Food Chem. 2004, 85, 81–89. [Google Scholar] [CrossRef]
- Bae, I.; Osatomi, K.; Yoshida, A.; Osako, K.; Yamaguchi, A.; Hara, K. Biochemical properties of acid-soluble collagens extracted from the skins of underutilised fishes. Food Chem. 2008, 108, 49–54. [Google Scholar] [CrossRef]
- Venugopal, M.G.; Ramshaw, J.A.; Braswell, E.; Zhu, D.; Brodsky, B. Electrostatic interactions in collagen-like triple-helical peptides. Biochemistry 1994, 33, 7948–7956. [Google Scholar] [CrossRef] [PubMed]
- Miki, A.; Inaba, S.; Baba, T.; Kihira, K.; Fukada, H.; Oda, M. Structural and physical properties of collagen extracted from moon jellyfish under neutral pH conditions. Biosci. Biotechnol. Biochem. 2015, 79, 1603–1607. [Google Scholar] [CrossRef] [PubMed]
- Blumlein, A.; McManus, J.J. Reversible and non-reversible thermal denaturation of lysozyme with varying pH at low ionic strength. Biochim. Biophys. Acta 2013, 1834, 2064–2070. [Google Scholar] [CrossRef] [PubMed]
- Harrington, W.F.; Josephs, R.; Segal, D.M. Physical chemical studies on proteins and polypeptides. Ann. Rev. Biochem. 1966, 35, 599–650. [Google Scholar] [CrossRef] [PubMed]
- Engel, J.; Bächinger, H.P. Structure, stability and folding of the collagen triple helix. Top. Curr. Chem. 2005, 247, 7–33. [Google Scholar] [CrossRef]
- Heidemann, E.; Roth, W. Synthesis and investigation of collagen model peptides. Adv. Polym. Sci. 1982, 42, 143–203. [Google Scholar] [CrossRef]
- Burjanadze, T.V. New analysis of the phylogenetic change of collagen thermostability. Biopolymers 2000, 53, 523–528. [Google Scholar] [CrossRef]
- Sadowska, M.; Kolodziejska, I.; Niecikowska, C. Isolation of collagen from the skins of Baltic cod (Gadus morhua). Food Chem. 2003, 81, 257–262. [Google Scholar] [CrossRef]
- Brønsted, H.V.; Carlsen, F.E. A cortical cytoskeleton in expanded epithelium cells of sponge gemmules. Exp. Cell Res. 1951, 2, 90–96. [Google Scholar] [CrossRef]
- Wiens, M.; Koziol, C.; Batel, R.; Müller, W.E.G. Prolidase in the marine sponge Suberites domuncula: Enzyme activity, molecular cloning, and phylogenetic relationship. Mar. Biotechnol. 1999, 1, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Sethmann, I.; Wörheide, G. Structure and composition of calcareous sponge spicules: A review and comparison to structurally related biominerals. Micron 2008, 39, 209–228. [Google Scholar] [CrossRef] [PubMed]
- Sudharsan, S.; Seedevi, P.; Saravanan, R.; Ramasamy, P.; Vasanth Kumar, S.; Vairamani, S.; Srinivasan, A.; Shanmugam, A. Isolation, characterization and molecular weight determination of collagen from marine sponge Spirastrella inconstans (Dendy). Afr. J. Biotechnol. 2013, 12, 504–511. [Google Scholar] [CrossRef]
- Wood, A.; Ogawa, M.; Portier, R.J.; Schexnayder, M.; Shirley, M.; Losso, J.N. Biochemical properties of alligator (Alligator mississippiensis) bone collagen. Comp. Biochem. Physiol. Part B 2008, 151, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Rigby, B.J. Amino acid composition and thermal stability of the skin collagen of the Antarctic ice fish. Nature 1968, 219, 166–167. [Google Scholar] [CrossRef] [PubMed]
- Minh Thuy, L.T.; Okazaki, E.; Osako, K. Isolation and characterization of acid-soluble collagen from the scales of marine fishes from Japan and Vietnam. Food Chem. 2014, 149, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Cha, J.; Stucky, G.D.; Morse, D.E. Silicatein a: Cathepsin L-like protein in sponge biosilica. Proc. Natl. Acad. Sci. USA 1998, 95, 6234–6238. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Douglas, E.P. Effects of various salts on structural polymorphism of reconstituted type I collagen fibrils. Colloids Surf. B Biointerfaces 2013, 112, 42–50. [Google Scholar] [CrossRef] [PubMed]

| Isolated Collagen | A. cannabina | S. carnosus |
|---|---|---|
| Insoluble collagen (InSC) | 12.6 | 5.0 |
| Intercellular collagen (ICC) | 3.0 | 1.9 |
| Spongin-like collagen (SlC) | 42.8 | 21.8 |
| Isolated Collagen | Period (nm) n = 10 | Fibril Width (nm) n = 10 |
|---|---|---|
| ICC from A. cannabina | 31.29 ± 1.14 1 | 18.74 ± 1.27 1 |
| ICC from S. carnosus | 28.81 ± 1.73 2 | 19.91 ± 1.65 1 |
| SlC from S. carnosus | 26.60 ± 0.95 3 | 24.10 ± 1.54 3 |
| InSC from S. carnosus | 17.62 ± 2.91 1,4 |
| Region | Peak Wavenumber (cm−1) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| A. cannabina | S. carnosus | BOC | |||||||
| InSC | SF-InSC | ICC | SlC | InSC | SF-InSC | ICC | SlC | ||
| Amide A | 3288 | 3279 | 3294 | 3286 | 3282 | 3282 | 3292 | 3287 | 3295 |
| Amide B | 2924 | 2924 | 2922 | 2926 | 2924 | 2922 | 2922 | 2923 | 2933 |
| Amide I | 1622 | 1627 | 1654 | 1639 | 1622 | 1628 | 1652 | 1647 | 1635 |
| Amide II | 1543 | 1529 | 1547 | 1539 | 1535 | 1527 | 1543 | 1543 | 1545 |
| Amide III | 1232 | 1226 | 1238 | 1222 | 1234 | 1230 | 1238 | 1232 | 1235 |
| C-O stretch | 1055 | 1059 | 1078 | 1074 | 1066 | 1078 | |||
| 1035 | 1037 | 1028 | 1028 | 1028 | 1031 | 1035 | 1001 | ||
| Amino Acid | A. cannabina | S. carnosus | Amino Acid | A. cannabina | S. carnosus |
|---|---|---|---|---|---|
| Hyp | 38 | 47 | Met | 21 | 11 |
| Asx 1 | 100 | 94 | Ile | 37 | 24 |
| Thr | 56 | 56 | Leu | 62 | 48 |
| Ser | 63 | 57 | Tyr | 13 | 12 |
| Glx 2 | 82 | 81 | Phe | 33 | 22 |
| Pro | 58 | 56 | Hyl | 6 | 6 |
| Gly | 257 | 295 | Lys | 15 | 15 |
| Ala | 72 | 89 | His | 6 | 4 |
| Val | 44 | 43 | Arg | 37 | 43 |
| Total imino acids | 96 | 103 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tziveleka, L.-A.; Ioannou, E.; Tsiourvas, D.; Berillis, P.; Foufa, E.; Roussis, V. Collagen from the Marine Sponges Axinella cannabina and Suberites carnosus: Isolation and Morphological, Biochemical, and Biophysical Characterization. Mar. Drugs 2017, 15, 152. https://doi.org/10.3390/md15060152
Tziveleka L-A, Ioannou E, Tsiourvas D, Berillis P, Foufa E, Roussis V. Collagen from the Marine Sponges Axinella cannabina and Suberites carnosus: Isolation and Morphological, Biochemical, and Biophysical Characterization. Marine Drugs. 2017; 15(6):152. https://doi.org/10.3390/md15060152
Chicago/Turabian StyleTziveleka, Leto-Aikaterini, Efstathia Ioannou, Dimitris Tsiourvas, Panagiotis Berillis, Evangelia Foufa, and Vassilios Roussis. 2017. "Collagen from the Marine Sponges Axinella cannabina and Suberites carnosus: Isolation and Morphological, Biochemical, and Biophysical Characterization" Marine Drugs 15, no. 6: 152. https://doi.org/10.3390/md15060152
APA StyleTziveleka, L.-A., Ioannou, E., Tsiourvas, D., Berillis, P., Foufa, E., & Roussis, V. (2017). Collagen from the Marine Sponges Axinella cannabina and Suberites carnosus: Isolation and Morphological, Biochemical, and Biophysical Characterization. Marine Drugs, 15(6), 152. https://doi.org/10.3390/md15060152










