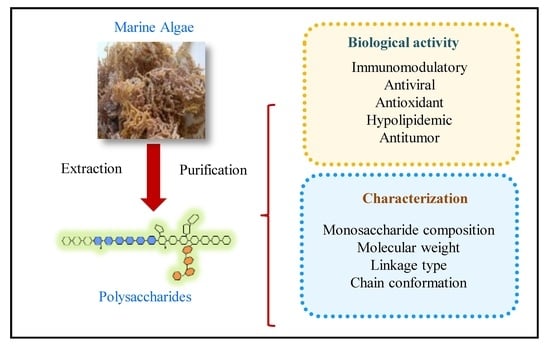
Recent Advances in Marine Algae Polysaccharides: Isolation, Structure, and Activities
Abstract
1. Introduction
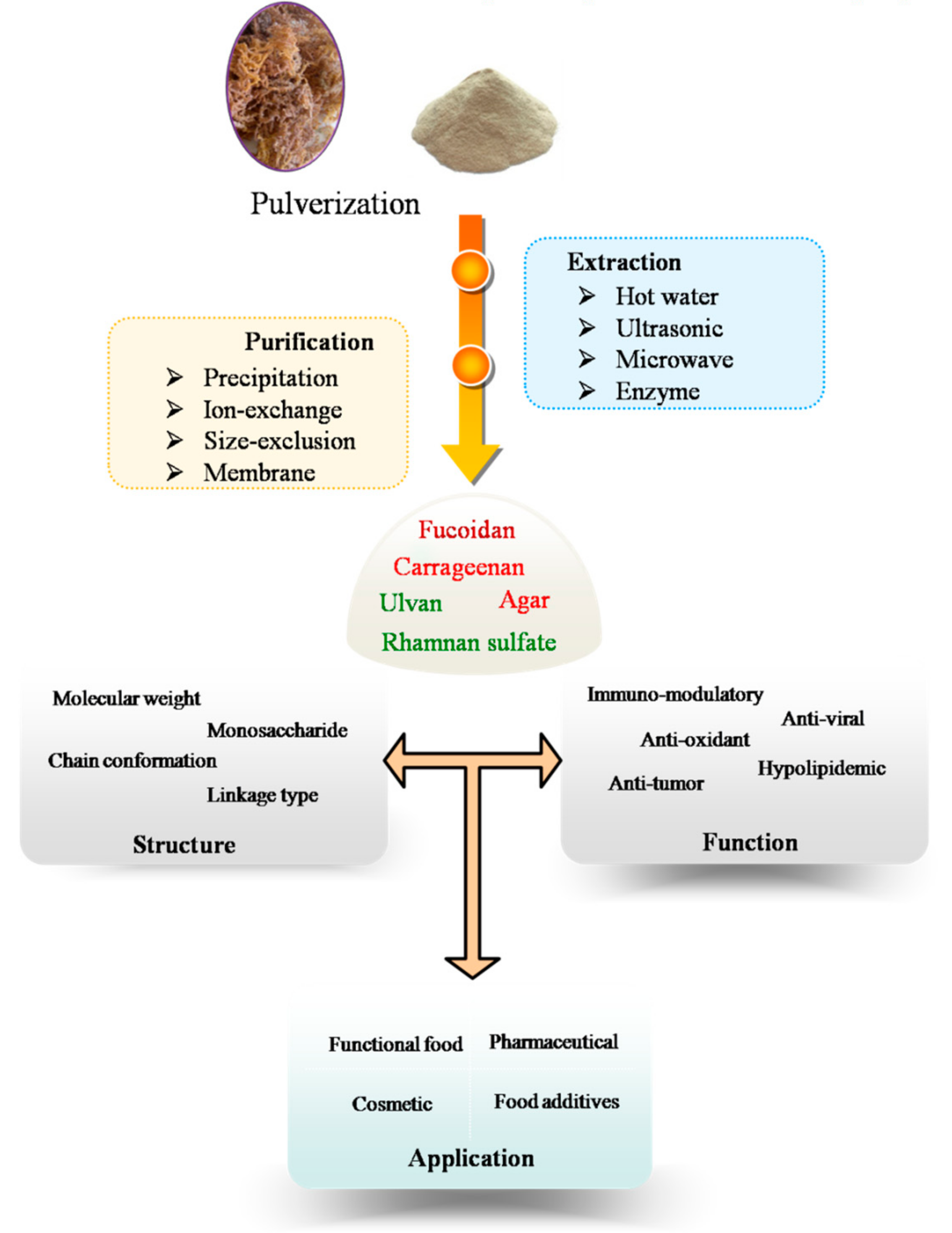
2. Extraction of Polysaccharides from Marine Algae
3. Purification Procedure
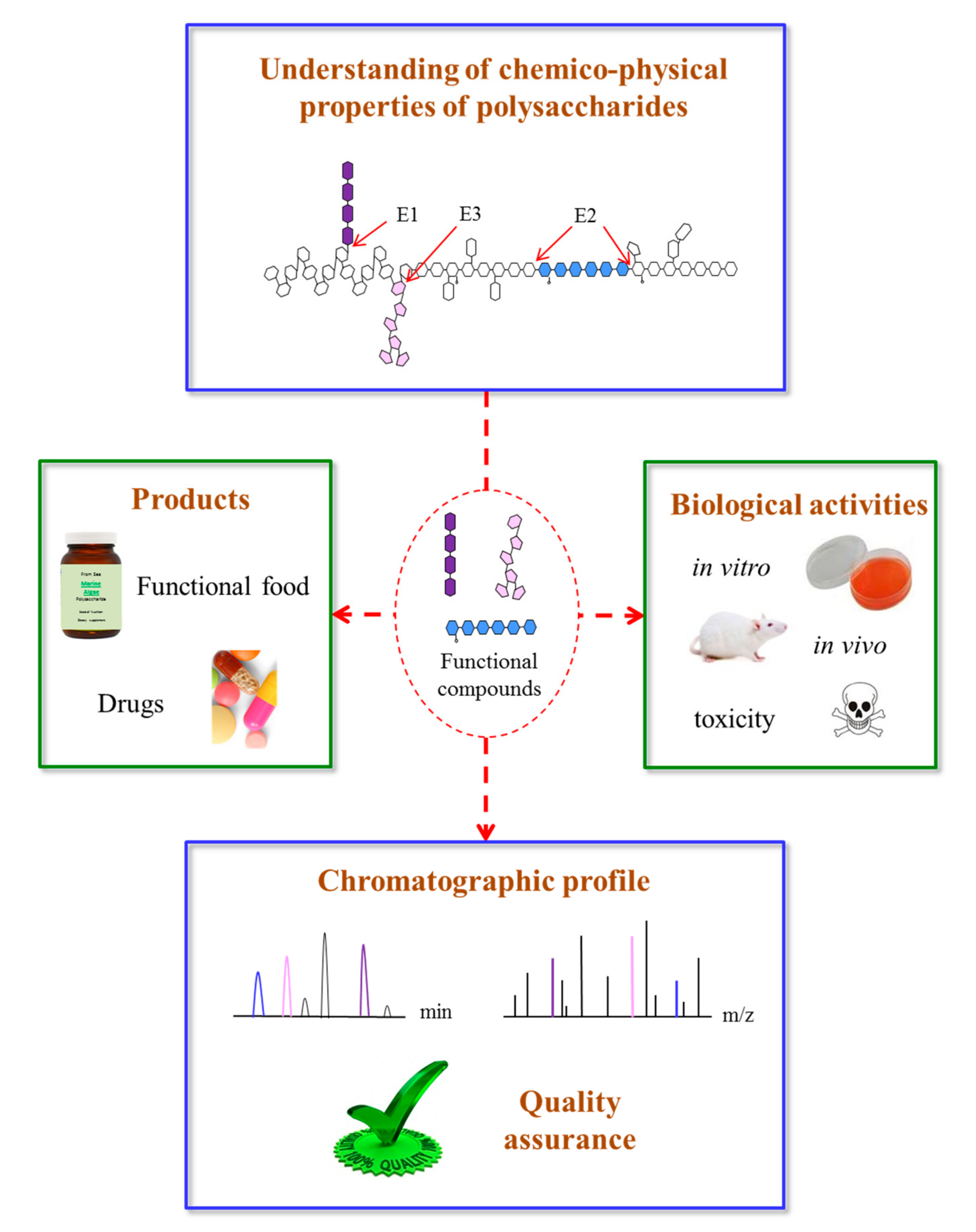
4. Structural and Physical Properties of Marine Algae Polysaccharides
5. Quality Control
6. Biological Activity of Polysaccharides from Marine Algae
6.1. Immunomodulatory Activity
6.2. Antitumor Activity
6.3. Antiviral Activity
6.4. Antioxidant Activity
6.5. Hypolipidemic Activity
7. Future Perspective
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brown, E.S.; Allsopp, P.J.; Magee, P.J.; Gill, C.I.R.; Nitecki, S.; Strain, C.R.; McSorley, E.M. Seaweed and human health. Nutr. Rev. 2014, 72, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Rioux, L.-E.; Beaulieu, L.; Turgeon, S.L. Seaweeds: A traditional ingredients for new gastronomic sensation. Food Hydrocoll. 2017, 68, 255–265. [Google Scholar] [CrossRef]
- Mohamed, S.; Hashim, S.N.; Rahman, H.A. Seaweeds: A sustainable functional food for complementary and alternative therapy. Trends Food Sci. Technol. 2012, 23, 83–96. [Google Scholar] [CrossRef]
- de Jesus Raposo, M.F.; de Morais, A.M.; de Morais, R.M.S.C. Marine polysaccharides from algae with potential biomedical applications. Mar. Drugs 2015, 13, 2967–3028. [Google Scholar] [CrossRef] [PubMed]
- Hamed, I.; Özogul, F.; Özogul, Y.; Regenstein, J.M. Marine bioactive compounds and their health benefits: A review. Compr. Rev. Food Sci. Food Saf. 2015, 14, 446–465. [Google Scholar] [CrossRef]
- Hu, D.J.; Cheong, K.I.; Zhao, J.; Li, S.P. Chromatography in characterization of polysaccharides from medicinal plants and fungi. J. Sep. Sci. 2013, 36, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A. An overview of the medical applications of marine skeletal matrix proteins. Mar. Drugs 2016, 14, 167. [Google Scholar] [CrossRef] [PubMed]
- Davis, T.A.; Volesky, B.; Mucci, A. A review of the biochemistry of heavy metal biosorption by brown algae. Water Res. 2003, 37, 4311–4330. [Google Scholar] [CrossRef]
- Zhang, Z.S.; Wang, F.; Wang, X.M.; Liu, X.L.; Hou, Y.; Zhang, Q.B. Extraction of the polysaccharides from five algae and their potential antioxidant activity in vitro. Carbohydr. Polym. 2010, 82, 118–121. [Google Scholar] [CrossRef]
- Savage, P.E. Algae under pressure and in hot water. Science 2012, 338, 1039–1040. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.J.; Aida, W.M.W.; Maskat, M.T.; Mamot, S.; Ropien, J.; Mohd, D.M. Isolation and antioxidant capacity of fucoidan from selected Malaysian seaweeds. Food Hydrocoll. 2014, 42, 280–288. [Google Scholar] [CrossRef]
- Sousa, A.M.M.; Alves, V.D.; Morais, S.; Delerue-Matos, C.; Gonçalves, M.P. Agar extraction from integrated multitrophic aquacultured Gracilaria vermiculophylla: Evaluation of a microwave-assisted process using response surface methodology. Bioresour. Technol. 2010, 101, 3258–3267. [Google Scholar] [CrossRef] [PubMed]
- Hahn, T.; Lang, S.; Uiber, R.; Muffler, K. Novel procedures for the extraction of fucoidan from brown algae. Process Biochem. 2012, 47, 1691–1698. [Google Scholar] [CrossRef]
- Yuan, Y.; Macquarrie, D. Microwave assisted extraction of sulfated polysaccharides (fucoidan) from Ascophyllum nodosum and its antioxidant activity. Carbohydr. Polym. 2015, 129, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Tsubaki, S.; Oono, K.; Hiraoka, M.; Onda, A.; Mitani, T. Microwave-assisted hydrothermal extraction of sulfated polysaccharides from Ulva spp. and Monostroma latissimum. Food Chem. 2016, 210, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Jasso, R.M.; Mussatto, S.I.; Pastrana, L.; Aguilar, C.N.; Teixeira, J.A. Microwave-assisted extraction of sulfated polysaccharides (fucoidan) from brown seaweed. Carbohydr. Polym. 2011, 86, 137–1144. [Google Scholar] [CrossRef]
- Kadam, S.U.; Tiwari, B.K.; O’Donnell, C.P. Application of novel extraction technologies for bioactives from marine algae. J. Agric. Food Chem. 2013, 61, 4667–4675. [Google Scholar] [CrossRef] [PubMed]
- Kadam, S.U.; O’Donnel, C.P.; Rai, D.K.; Hossain, M.B.; Burgess, C.M.; Walsh, D.; Tiwari, B.K. Laminarin from Irish brown seaweeds Ascophyllum nodosum and Laminaria hyperborea: Ultrasound assisted extraction, characterization and bioactivity. Mar. Drugs 2015, 13, 4270–4280. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, F.; Tabarsa, M.; Rezaei, M. Ulvan from green algae Ulva intestinalis: Optimization of ultrasound-assisted extraction and antioxidant activity. J. Appl. Phycol. 2016, 28, 2979–2990. [Google Scholar] [CrossRef]
- Tang, W.; Lin, L.H.; Xie, J.H.; Wang, Z.; Wang, H.J.; Dong, Y.J.; Shen, M.Y.; Xie, M.Y. Effect of ultrasonic treatment on the physicochemical properties and antioxidant activities of polysaccharide from Cyclocarya paliurus. Carbohydr. Polym. 2016, 151, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.; Sousa, S.; Silva, A.; Amorim, M.; Pereira, L.; Rocha-Santos, T.A.P.; Gomes, A.M.P.; Duarte, A.C.; Freitas, A.C. Impact of enzyme- and ultrasound-assisted extraction methods on biological properties of red, brown, and green seaweeds from the central west coast of Portugal. J. Agric. Food Chem. 2015, 63, 3177–3188. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghe, W.A.J.P.; Jeon, Y.-J. Enzyme-assistant extraction (EAE) of bioactive components: A useful approach for recovery of industrially important metabolites from seaweeds: A review. Fitoterapia 2012, 83, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.-M.; Kim, K.-N.; Lee, S.-H.; Ahn, G.; Cha, S.-H.; Kim, A.-D.; Yang, X.-D.; Kang, M.-C.; Jeon, Y.-J. Anti-inflammatory activity of polysaccharide purified from AMG-assistant extract of Ecklonia cava in LPS-stimulated RAW 264.7 macrophages. Carbohydr. Polym. 2011, 85, 80–85. [Google Scholar] [CrossRef]
- Heo, S.-J.; Park, E.-J.; Lee, K.-W.; Jeon, Y.-J. Antioxidant activities of enzymatic extracts from brown seaweeds. Bioresour. Technol. 2005, 96, 1613–1623. [Google Scholar] [CrossRef] [PubMed]
- Hardouin, K.; Bedoux, G.; Burlot, A.-S.; Donnay-Moreno, C.; Bergé, J.P.; Nyvall-Collén, P.; Bourgougnon, N. Enzyme-assisted extraction (EAE) for the production of antiviral and antioxidant extracts from the green seaweed Ulva armoricana (Ulvales, Ulvophyceae). Algal Res. 2016, 16, 233–239. [Google Scholar] [CrossRef]
- Herrero, M.; Ibáñez, E. Green processes and sustainability: An overview on the extraction of high added-value products from seaweeds and microalgae. J. Supercrit. Fluids 2015, 96, 211–216. [Google Scholar] [CrossRef]
- Martins, M.; Vieira, F.A.; Correia, I.; Ferreira, R.A.S.; Abreu, H.; Coutinho, J.A.P.; Ventura, S.P.M. Recovery of phycobiliproteins from the red macroalga Gracilaria sp. using ionic liquid aqueous solutions. Green Chem. 2016, 18, 4287–4296. [Google Scholar] [CrossRef]
- Chen, X.M.; Nie, W.J.; Yu, G.Q.; Li, Y.; Hu, Y.L.; Lu, J.S.; Lu, J.X.; Jin, L.Q. Antitumor and immunomodulatory activity of polysaccharides from Sargassum fusiforme. Food Chem. Toxicol. 2012, 50, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, M.; Miao, C.; McDonald, A.; Chen, S. Concomitant extraction of bio-oil and value added polysaccharides from Chlorella sorokiniana using a unique sequential hydrothermal extraction technology. Fuel 2012, 95, 63–70. [Google Scholar] [CrossRef]
- Patel, A.K.; Laroche, C.; Marcati, A.; Ursu, A.V.; Jubeau, S.; Marchal, L.; Petit, E.; Djelveh, G.; Michaud, P. Separation and fractionation of exopolysaccharides from Porphyridium cruentum. Bioresour. Technol. 2013, 145, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.H.; Shen, M.Y.; Nie, S.P.; Zhao, Q.; Li, C.; Xie, M.Y. Separation of water-soluble polysaccharides from Cyclocarya paliurus by ultrafiltration process. Carbohydr. Polym. 2014, 101, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Usoltseva, R.V.; Anastyuk, S.D.; Shevchenko, N.M.; Zvyagintseva, T.N.; Ermakova, S.P. The comparison of structure and anticancer activity in vitro of polysaccharides from brown algae Alaria marginata and A. angusta. Carbohydr. Polym. 2016, 153, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Ermakova, S.; Men’shova, R.; Vishchuk, O.; Kim, S.-M.; Um, B.-H.; Isakov, V.; Zvyagintseva, T. Water-soluble polysaccharides from the brown alga Eisenia bicyclis: Structural characteristics and antitumor activity. Algal Res. 2013, 2, 51–58. [Google Scholar] [CrossRef]
- Anastyuk, S.D.; Shevchenko, N.M.; Usoltseva, R.V.; Silchenko, A.S.; Zadorozhny, P.A.; Dmitrenok, P.S.; Ermakova, S.P. Structural features and anticancer activity in vitro of fucoidan derivatives from brown alga Saccharina cichorioides. Carbohydr. Polym. 2017, 157, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.F.; Liu, M.; Fang, Z.X.; Wu, J.L.; Zhang, Q.Q. Composition and cytotoxicity of a novel polysaccharide from brown alga (Laminaria japonica). Carbohydr. Polym. 2012, 89, 1022–1026. [Google Scholar] [CrossRef] [PubMed]
- Di, T.; Chen, G.J.; Sun, Y.; Ou, S.Y.; Zeng, X.X.; Ye, H. Antioxidant and immunostimulating activities in vitro of sulfated polysaccharides isolated from Gracilaria rubra. J. Funct. Foods 2017, 28, 64–75. [Google Scholar] [CrossRef]
- Arman, M.; Qader, S.A.U. Structural analysis of kappa-carrageenan isolated from Hypnea musciformis (red algae) and evaluation as an elicitor of plant defense mechanism. Carbohydr. Polym. 2012, 88, 1264–1271. [Google Scholar] [CrossRef]
- Wu, G.J.; Shiu, S.M.; Hsieh, M.C.; Tsai, G.J. Anti-inflammatory activity of a sulfated polysaccharide from the brown alga Sargassum cristaefolium. Food Hydrocoll. 2016, 53, 16–23. [Google Scholar] [CrossRef]
- Yu, Y.; Li, Y.P.; Du, C.Y.; Mou, H.J.; Wang, P. Compositional and structural characteristics of sulfated polysaccharide from Enteromorpha prolifera. Carbohydr. Polym. 2017, 165, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Zayed, A.; Muffler, K.; Hahn, T.; Rupp, S.; Finkelmeier, D.; Burger-Kentischer, A.; Ulber, R. Physicochemical and biological characterization of fucoidan from Fucus vesiculosus purified by dye affinity chromatography. Mar. Drugs 2016, 14, 79. [Google Scholar] [CrossRef] [PubMed]
- Hahn, T.; Zayed, A.; Kovacheva, M.; Stadtmüller, R.; Lang, S.; Muffler, K.; Ulber, R. Dye affinity chromatography for fast and simple purification of fucoidan from marine brown algae. Eng. Life Sci. 2016, 16, 78–87. [Google Scholar] [CrossRef]
- Marcati, A.; Ursu, A.V.; Laroche, C.; Soanen, N.; Marchal, L.; Jubeau, S.; Djelveh, G.; Michaud, P. Extraction and fractionation of polysaccharides and B-phycoerythrin from the microalga Porphyridium cruentum by membrane technology. Algal Res. 2014, 5, 258–263. [Google Scholar] [CrossRef]
- Yu, P.; Sun, H.S. Purification of a fucoidan from kelp polysaccharide and its inhibitory kinetics for tyrosinase. Carbohydr. Polym. 2014, 99, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Yeon, J.H.; Lee, S.E.; Choi, W.Y.; Kang, D.H.; Lee, H.Y.; Jung, K.H. Repeated-batch operation of surface-aerated fermentor for bioethanol production from the hydrolysate of seaweed Sargassum sagamianum. J. Microbiol. Biotechnol. 2011, 21, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Foley, S.A.; Szegezdi, E.; Mulloy, B.; Samali, A.; Tuohy, M.G. An unfractionated fucoidan from Ascophyllum nodosum: Extraction, characterization, and apoptotic effects in vitro. J. Nat. Prod. 2011, 74, 1851–1861. [Google Scholar] [CrossRef] [PubMed]
- Fleita, D.; El-Sayed, M.; Rifaat, D. Evaluation of the antioxidant activity of enzymatically-hydrolyzed sulfated polysaccharides extracted from red algae; Pterocladia capillacea. LWT-Food Sci. Technol. 2015, 63, 1236–1244. [Google Scholar] [CrossRef]
- Condezo-Hoyos, L.; Pérez-López, E.; Rupérez, P. Improved evaporative light scattering detection for carbohydrate analysis. Food Chem. 2015, 180, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Templeton, D.W.; Quinn, M.; Wychen, S.V.; Hyman, D.; Laurens, L.M.L. Separation and quantification of microalgal carbohydrates. J. Chromatogr. A 2012, 1270, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Synytsya, A.; Choi, D.J.; Pohl, R.; Na, Y.S.; Capek, P.; Lattová, E.; Taubner, T.; Choi, J.W.; Lee, C.W.; Park, J.K.; et al. Structural features and anti-coagulant activity of the sulphated polysaccharide SPS-CF from a green alga Capsosiphon fulvescens. Mar. Biotechnol. 2015, 17, 718–735. [Google Scholar] [CrossRef] [PubMed]
- Harazono, A.; Kobayashi, T.; Kawasaki, N.; Itoh, S.; Tada, M.; Hashii, N.; Ishii, A.; Arato, T.; Yanagihara, S.; Yagi, Y.; et al. A comparative study of monosaccharide composition analysis as a carbohydrate test for biopharmaceuticals. Biologicals 2011, 39, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Boucelkha, A.; Petit, E.; Elboutachfaiti, R.; Molinié, R.; Amari, S.; Zaidi-Yahaoui, R. Production of guluronate oligosaccharide of alginate from brown algae Stypocaulon scoparium using an alginate lyase. J. Appl. Phycol. 2017, 29, 509–519. [Google Scholar] [CrossRef]
- Striegel, A.; Yau, W.W.; Kirkland, J.J.; Bly, D.D. Modern Size-Exclusion Liquid Chromatography: Practice of Gel Permeation and Gel Filtration Chromatography; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; pp. 1–12. [Google Scholar]
- Li, N.; Mao, W.J.; Yan, M.X.; Liu, X.; Xia, Z.; Wang, S.Y.; Xiao, B.; Chen, C.L.; Zhang, L.F.; Cao, S.J. Structural characterization and anticoagulant activity of a sulfated polysaccharide from the green alga Codium divaricatum. Carbohydr. Polym. 2015, 121, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.; Kim, S.M. Characterization and immunomodulatory activities of polysaccharides extracted from green alga Chlorella ellipsoidea. Int. J. Biol. Macromol. 2017, 95, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Hammed, A.; Irwandi, J.; Senay, S.; Azura, A.; Zahangir, A. Chemical structure of sulfated polysaccharides from brown seaweed (Turbinaria turbinata). Int. J. Food Prop. 2017, 20, 1457–1469. [Google Scholar]
- Shevchenko, N.M.; Anastyuk, S.D.; Menshova, R.V.; Vishchuk, O.S.; Isakov, V.I.; Zadorozhny, P.A.; Sikorskaya, T.V.; Zvyagintseva, T.N. Further studies on structure of fucoidan from brown alga Saccharina gurjanovae. Carbohydr. Polym. 2015, 121, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, S.D.; Kim, H.; Lu, F.; Ralph, J. Whole plant cell wall characterization using solution-state 2D NMR. Nat. Protoc. 2012, 7, 1579–1589. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Liu, X.; He, X.X.; Wang, S.Y.; Cao, S.J.; Xia, Z.; Xian, H.; Qin, L.; Mao, W.J. Structure and anticoagulant property of a sulfated polysaccharide isolated from the green seaweed Monostroma angicava. Carbohydr. Polym. 2017, 159, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Youssouf, L.; Lallemand, L.; Giraud, P.; Soulé, F.; Bhaw-Luximon, A.; Meilhac, O.; D’Hellencourt, C.L.; Jhurry, D.; Couprie, J. Ultrasound-assisted extraction and structural characterization by NMR of alginates and carrageenans from seaweeds. Carbohydr. Polym. 2017, 166, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Ordóñez, E.; Jiménez-Escrig, A.; Rupérez, P. Bioactivity of sulfated polysaccharides from the edible red seaweed Mastocarpus stellatus. Bioact. Carbohydr. Diet. Fibre 2014, 3, 29–40. [Google Scholar] [CrossRef]
- Kalitnik, A.A.; Byankina Barabanova, A.O.; Nagorskaya, V.P.; Reunov, A.V.; Glazunov, V.P.; Solov’eva, T.F.; Yermak, I.M. Low molecular weight derivatives of different carrageenan types and their antiviral activity. J. Appl. Phycol. 2013, 25, 65–72. [Google Scholar] [CrossRef]
- Pérez-Recalde, M.; Matulewicz, M.C.; Pujol, C.A.; Carlucci, M.J. In vitro and in vivo immunomodulatory activity of sulfated polysaccharides from red seaweed Nemalion helminthoides. Int. J. Biol. Macromol. 2014, 63, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Kravchenko, A.O.; Anastyuk, S.D.; Isakov, V.V.; Sokolova, E.V.; Glazunov, V.P.; Yermak, I.M. Structural peculiarities of polysaccharide from sterile form of Far Eastern red alga Ahnfeltiopsis flabelliformis. Carbohydr. Polym. 2014, 111, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.M.; Zhang, Z.S. The antitumor activity of a red alga polysaccharide complexes carrying 5-fluorouracil. Int. J. Biol. Macromol. 2014, 69, 542–545. [Google Scholar] [CrossRef] [PubMed]
- Imjongjairak, S.; Ratanakhanokchai, K.; Laohakunjit, N.; Tachaapaikoon, C.; Pason, P.; Waeonukul, R. Biochemical characteristics and antioxidant activity of crude and purified sulfated polysaccharides from Gracilaria fisheri. Biosci. Biotechnol. Biochem. 2015, 80, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Mendes, G.S.; Duarte, M.E.R.; Colodi, F.G.; Noseda, M.D.; Ferreira, L.G.; Berté, S.D.; Cavalcanti, J.F.; Santos, N.; Romanos, M.T.V. Structure and anti-metapneumovirus activity of sulfated galactans from the red seaweed Cryptonemia seminervis. Carbohydr. Polym. 2014, 101, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Assreuy, A.M.S.; Amorim, R.M.F.; Brizeno, L.A.C.; de Paulo Pereira, L.; de Sousa, A.A.S.; Aragão, G.F.; Pereira, M.G. Edematogenic activity of a sulfated galactan from the red marine algae Gelidium crinale. Pharm. Biol. 2012, 50, 1194–1198. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.C.; Jeong, Y.T.; Lee, S.M.; Kim, J.H. Immune-modulating activities of polysaccharides extracted from brown algae Hizikia fusiforme. Biosci. Biotechnol. Biochem. 2015, 79, 1362–1365. [Google Scholar] [CrossRef] [PubMed]
- Hadj, A.H.; Lajili, S.; Ben, S.R.; Le, C.D.; Bouraoui, A.; Majdoub, H. Physico-chemical characterization and pharmacological evaluation of sulfated polysaccharides from three species of Mediterranean brown algae of the genus Cystoseira. DARU J. Pharm. Sci. 2015, 23, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Imbs, T.I.; Ermakova, S.P.; Malyarenko, O.S.; Isakov, V.V.; Zvyagintseva, T.N. Structural elucidation of polysaccharide fractions from the brown alga Coccophora langsdorfii and in vitro investigation of their anticancer activity. Carbohydr. Polym. 2016, 135, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Menshova, R.V.; Ermakova, S.P.; Anastyuk, S.D.; Isakov, V.V.; Dubrovskaya, Y.V.; Kusaykin, M.I.; Um, B.-H.; Zvyagintseva, T.N. Structure, enzymatic transformation and anticancer activity of branched high molecular weight laminaran from brown alga Eisenia bicyclis. Carbohydr. Polym. 2014, 99, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, M.; Bell, T.; Dénes, Á.; Falshaw, R.; Itzhaki, R. Anti-HSV1 activity of brown algal polysaccharides and possible relevance to the treatment of Alzheimer’s disease. Int. J. Biol. Macromol. 2015, 74, 530–540. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.H.; Zhang, W.J.; Wang, J.; Ren, S.M.; Song, N.; Duan, D.L.; Zhang, Q.B. Characterization of laminaran and a highly sulfated polysaccharide from Sargassum fusiforme. Carbohydr. Res. 2014, 385, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.Y.; Liang, B.; Li, M.X.; Jin, M.L. Influence of Laminarin polysaccahrides on oxidative damage. Int. J. Biol. Macromol. 2011, 48, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.M.; Zhang, Z.S.; Yao, Z.Y.; Zhao, M.X.; Qi, H.M. Sulfation, anticoagulant and antioxidant activities of polysaccharide from green algae Enteromorpha linza. Int. J. Biol. Macromol. 2013, 58, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.M.; Cheng, C.L.; Zhao, H.T.; Jing, J.; Gong, N.; Lu, W.H. In vivo anti-radiation activities of the Ulva pertusa polysaccharides and polysaccharide–iron(III) complex. Int. J. Biol. Macromol. 2013, 60, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Ropellato, J.; Carvalho, M.M.; Ferreira, L.G.; Noseda, M.D.; Zuconelli, C.R.; Gonçalves, A.G.; Ducatti, D.R.B.; Kenski, J.C.N.; Nasato, P.L.; Winnischofer, S.M.B.; et al. Sulfated heterorhamnans from the green seaweed Gayralia oxysperma: Partial depolymerization, chemical structure and antitumor activity. Carbohydr. Polym. 2015, 117, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Cheong, K.-L.; Wu, D.-T.; Deng, Y.; Leong, F.; Zhao, J.; Zhang, W.-J.; Li, S.-P. Qualitation and quantification of specific polysaccharides from Panax species using GC–MS, saccharide mapping and HPSEC-RID-MALLS. Carbohydr. Polym. 2016, 153, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, A.; Medzhitov, R. Medzhitov, Regulation of adaptive immunity by the innate immune system. Science 2010, 327, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.M.; Xu, S.S.; Li, L.; Pan, T.M.; Shi, C.L.; Liu, H.; Cao, M.J.; Su, W.J.; Liu, G.M. In vitro and in vivo immunomodulatory activity of sulfated polysaccharide from Porphyra haitanensis. Carbohydr. Polym. 2017, 165, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Fang, Q.; Wang, J.F.; Zha, X.Q.; Cui, S.H.; Cao, L.; Luo, J.P. Immunomodulatory activity on macrophage of a purified polysaccharide extracted from Laminaria japonica. Carbohydr. Polym. 2015, 134, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Khil’chenko, S.R.; Zaporozhets, T.S.; Shevchenko, N.M.; Zvyagintseva, T.N.; Vogel, U.; Seeberger, P.; Lepenies, B. Immunostimulatory activity of fucoidan from the brown alga Fucus evanescens: Role of sulfates and acetates. J. Carbohydr. Chem. 2011, 30, 291–305. [Google Scholar] [CrossRef]
- Jeong, B.-E.; Ko, E.-J.; Joo, H.-G. Cytoprotective effects of fucoidan, an algae-derived polysaccharide on 5-fluorouracil-treated dendritic cells. Food Chem. Toxicol. 2012, 50, 1480–1484. [Google Scholar] [CrossRef] [PubMed]
- Bielekova, B.; Catalfamo, M.; Reichert-Scrivner, S.; Packer, A.; Cerna, M.; Waldmann, T.A.; McFarland, H.; Henkart, P.A.; Martin, R. Regulatory CD56bright natural killer cells mediate immunomodulatory effects of IL-2Rα-targeted therapy (daclizumab) in multiple sclerosis. Proc. Natl. Acad. Sci. USA 2006, 103, 5941–5946. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Oda, T.; Yu, Q.; Jin, J.-O. Fucoidan from Macrocystis pyrifera has powerful immune-modulatory effects compared to three other fucoidans. Mar. Drugs 2015, 13, 1084–1104. [Google Scholar] [CrossRef] [PubMed]
- Besednova, N.N.; Zaporozhets, T.S.; Somova, L.M.; Kuznetsova, T.A. Review: Prospects for the use of extracts and polysaccharides from marine algae to prevent and treat the diseases caused by Helicobacter pylori. Helicobacter 2015, 20, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.N.; Li, H.; Wu, J.; Xu, X.T.; Sun, X.; Zhao, X.D.; Xu, N.J. Transcriptome profiling reveals the antitumor mechanism of polysaccharide from marine algae Gracilariopsis lemaneiformis. PLoS ONE 2016, 11, e0158279. [Google Scholar] [CrossRef] [PubMed]
- Fan, S.R.; Zhang, J.F.; Nie, W.J.; Zhou, W.Y.; Jin, L.Q.; Chen, X.M.; Lu, J.X. Antitumor effects of polysaccharide from Sargassum fusiforme against human hepatocellular carcinoma HepG2 cells. Food Chem. Toxicol. 2017, 102, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Vishchuk, O.S.; Ermakova, S.P.; Zvyagintseva, T.N. The fucoidans from brown algae of Far-Eastern seas: Anti-tumor activity and structure–function relationship. Food Chem. 2013, 141, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Teas, J.; Vena, S.; Cone, D. L.; Irhimeh, M. The consumption of seaweed as a protective factor in the etiology of breast cancer: Proof of principle. J. Appl. Phycol. 2013, 25, 771–779. [Google Scholar] [CrossRef] [PubMed]
- Shukla, D.; Liu, J.; Blaiklock, P.; Shworak, N.W.; Bai, X.; Esko, J.D.; Cohen, G.H.; Eisenberg, R.J.; Rosenberg, R.D.; Spear, P.G. A novel role for 3-O-sulfated heparan sulfate in herpes simplex virus 1 entry. Cell 1999, 99, 13–22. [Google Scholar] [CrossRef]
- Dinesh, S.; Menon, T.; Hanna, L.E.; Suresh, V.; Sathuvan, M.; Manikannan, M. In vitro anti-HIV-1 activity of fucoidan from Sargassum swartzii. Int. J. Biol. Macromol. 2016, 82, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Yasuhara-Bell, J.; Lu, Y. Marine compounds and their antiviral activities. Antivir. Res. 2010, 86, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, S.S.; Navid, M.H.; Ghosh, T.; Schnitzler, P.; Ray, B. Structural features and in vitro antiviral activities of sulfated polysaccharides from Sphacelaria indica. Phytochemistry 2011, 72, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Free radicals and antioxidants—Quo vadis? Trends Pharmacol. Sci. 2011, 32, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Sousa, W.M.; Silva, R.O.; Bezerra, F.F.; Bingana, R.D.; Barros, F.C.N.; Costa, L.E.C.; Sombra, V.G.; Soares, P.M.G.; Feitosa, J.P.A.; et al. Sulfated polysaccharide fraction from marine algae Solieria filiformis: Structural characterization, gastroprotective and antioxidant effects. Carbohydr. Polym. 2016, 152, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.Q.; Wang, L.; Li, J.; Liu, H.H. Characterization and antioxidant activities of degraded polysaccharides from two marine Chrysophyta. Food Chem. 2014, 160, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Escrig, A.; Gómez-Ordóñez, E.; Rupérez, P. Infrared characterisation, monosaccharide profile and antioxidant activity of chemical fractionated polysaccharides from the edible seaweed sugar Kombu (Saccharina latissima). Int. J. Food Sci. Technol. 2015, 50, 340–346. [Google Scholar] [CrossRef]
- Wang, W.; Lu, J.B.; Wang, C.; Wang, C.S.; Zhang, H.H.; Li, C.Y.; Qian, G.Y. Effects of Sargassum fusiforme polysaccharides on antioxidant activities and intestinal functions in mice. Int. J. Biol. Macromol. 2013, 58, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.J.; Wu, Y.; Qu, M.; Li, W.; Yan, X.F. Evaluation of antioxidant activities of water-soluble polysaccharides from brown alga Hizikia fusiformis. Int. J. Biol. Macromol. 2013, 56, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Halfar, J. First evidence of chitin in calcified coralline algae: New insights into the calcification process of Clathromorphum compactum. Sci. Rep. 2014, 4, 6162–6173. [Google Scholar] [CrossRef] [PubMed]
- Mayakrishnan, V.; Kannappan, P.; Abdullah, N.; Ahmed, A.B.A. Cardioprotective activity of polysaccharides derived from marine algae: An overview. Trends Food Sci. Technol. 2013, 30, 98–104. [Google Scholar] [CrossRef]
- Hoang, M.H.; Kim, J.-Y.; Lee, J.H.; You, S.; Lee, S.-J. Antioxidative, hypolipidemic, and anti-inflammatory activities of sulfated polysaccharides from Monostroma nitidum. Food Sci. Biotechnol. 2015, 24, 199–205. [Google Scholar] [CrossRef]
- Zha, X.Q.; Xiao, J.J.; Zhang, H.N.; Wang, J.H.; Pan, L.H.; Yang, X.F.; Luo, J.P. Polysaccharides in Laminaria japonica (LP): Extraction, physicochemical properties and their hypolipidemic activities in diet-induced mouse model of atherosclerosis. Food Chem. 2012, 134, 244–252. [Google Scholar] [CrossRef]
- Ben Gara, A.; Ben Abdallah Kolsi, R.; Chaaben, R.; Hammami, N.; Kammoun, M.; Paolo Patti, F.; El Feki, A.; Fki, L.; Belghith, H.; Belghith, K. Inhibition of key digestive enzymes related to hyperlipidemia and protection of liver-kidney functions by Cystoseira crinita sulphated polysaccharide in high-fat diet-fed rats. Biomed. Pharmacother. 2017, 85, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Wang, S.C.; Yao, C.W.; Xu, Z.; Xu, X.M. Hypolipidemic effect of porphyran extracted from Pyropia yezoensis in ICR mice with high fatty diet. J. Appl. Phycol. 2016, 28, 1315–1322. [Google Scholar] [CrossRef]

| Species | Polysaccharide Type | Molecular Weight (Da) | Monosaccharide | Backbone | Biological Activities | Reference |
|---|---|---|---|---|---|---|
| Red algae | - | - | - | - | - | - |
| Mastocarpus stellatus | Carrageenan | 1248 k | Gal:Glc:Xyl:Man = 87.8:5.4:4.4:2.4 | β-1,3-Gal and α-1,4-Gal | Anticoagulant | [59,60] |
| Chondrus armatus | Carrageenan | 88 k | Gal | β-1,3-Gal and α-1,4-Gal | Antiviral | [61] |
| Nemalion helminthoides | Sulphated mannan | 43.8 k | Man:Xyl:Sulphate = 1:0.01:0.64 | α-1,3-Man | Immunomodulatory | [62] |
| Ahnfeltiopsis flabelliformis | Sulphated galactan | - | Gal:3,6-AnGal:Glc:Xyl:SO3Na = 34.9:15.0:2.0:2.1:18.7 | β-1,3-Gal and α-1,4-Gal | Anticoagulant | [63] |
| Porphyra haitanensis | porphyran | 277 k | Gal | β-1,3-Gal | Antitumor | [64] |
| Gracilaria fisheri | Sulphated galactan | - | Gal | β-1,3-Gal and α-1,4-Gal | Antioxidant | [65] |
| Cryptonemia seminervis | Sulphated galactan | 51.6 k | Gal, trace in Glc, Ara | β-1,3-Gal and α-1,4-Gal | Anti-metapneumovirus | [66] |
| Gelidium crinale | Sulphated galactan | 300–600 k | Gal | α-1,3-Gal and α-1,4-Gal | Antiinflammatory | [67] |
| Brown algae | - | - | - | - | - | - |
| Alaria marginata | Galactofucan | - | Fuc:Gal:Xyl = 47.5:47.3:5.2 | →3)-α-l-Fuc-(2,4-SO3−)-(1→ | Anticancer | [32] |
| Hizikia fusiforme | - | - | Fuc:Gal:Xyl:Glc = 1.00:0.50:0.24:0.21 | - | Immunomodulatory | [68] |
| Cystoseira sedoides | Fucoidan | 642 k | Fuc and Uronic acid | α-1,3 or α-1,4-Fuc | Antiinflammatory | [69] |
| Coccophora langsdorfii | Fucoidan | - | Fuc | α-1,3 and α-1,4-Fuc | Anticancer | [70] |
| Eisenia bicyclis | Laminaran | 19–27 k | Glc | β-1,3 and β-1,6-Glc | Anticancer | [71] |
| Scytothamnus australis | Sulphated fucan | - | Fuc:Xyl:Glc = 40.8:1.5:1 | α-1,3-Fuc | Anti-HSV1 | [72] |
| Sargassum fusiforme | Laminaran | 27.6 k | Glc:Gal = 1.13:0.38 | β-1,3-Glc, β-1,6-Glc | - | [73] |
| Laminaria japonica | Laminaran | - | Man:Ara:Glc:Gal:Fuc = 3.27:8.61:4.23:12.12:46.93 | - | Antioxidant | [74] |
| Green algae | - | - | - | - | - | - |
| Enteromorpha linza | Rhamnan sulphate | 108.4 k | Rha:Xyl:Man:Glc:Gal = 3.6:1.0:0.31:0.28:0.19 | 1,4-Rha | Antioxidant | [75] |
| Codium divaricatum | Sulphated galactan | 37.9 k | Gal:Glc = 97.8:2.16 | 1,3- β-Gal | Anti-coagulant | [53] |
| Capsosiphon fulvescens | Ulvan | - | Rha:Xyl:Man = 45.0:44.1:10.2 | 4)-β-Xyl-(1→4)-α-Rha-(1→ | Anticoagulant | [49] |
| Ulva armoricana | Ulvan | 140–500 k | Rha:Gal:Glc:Xyl = 40.0:6.7:26.2:4.4 | - | Antiviral | [25] |
| Ulva pertusa | Ulvan | 28.2 k | - | - | Antiradiation | [76] |
| Monostroma angicava | Rhamnan sulphate | 88.1 k | Rha | α-1,2-Rha, α-1,3-Rha | Anticoagulant | [58] |
| Gayralia oxysperma | Rhamnan sulphate | 109 k | Rha:Xyl:Glc = 76.0:17.3:4.4 | α-1,3-Rha | Antitumor | [77] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, S.-Y.; Huang, X.; Cheong, K.-L. Recent Advances in Marine Algae Polysaccharides: Isolation, Structure, and Activities. Mar. Drugs 2017, 15, 388. https://doi.org/10.3390/md15120388
Xu S-Y, Huang X, Cheong K-L. Recent Advances in Marine Algae Polysaccharides: Isolation, Structure, and Activities. Marine Drugs. 2017; 15(12):388. https://doi.org/10.3390/md15120388
Chicago/Turabian StyleXu, Shu-Ying, Xuesong Huang, and Kit-Leong Cheong. 2017. "Recent Advances in Marine Algae Polysaccharides: Isolation, Structure, and Activities" Marine Drugs 15, no. 12: 388. https://doi.org/10.3390/md15120388
APA StyleXu, S.-Y., Huang, X., & Cheong, K.-L. (2017). Recent Advances in Marine Algae Polysaccharides: Isolation, Structure, and Activities. Marine Drugs, 15(12), 388. https://doi.org/10.3390/md15120388