Metabolites with Insecticidal Activity from Aspergillus fumigatus JRJ111048 Isolated from Mangrove Plant Acrostichum specioum Endemic to Hainan Island
Abstract
:1. Introduction
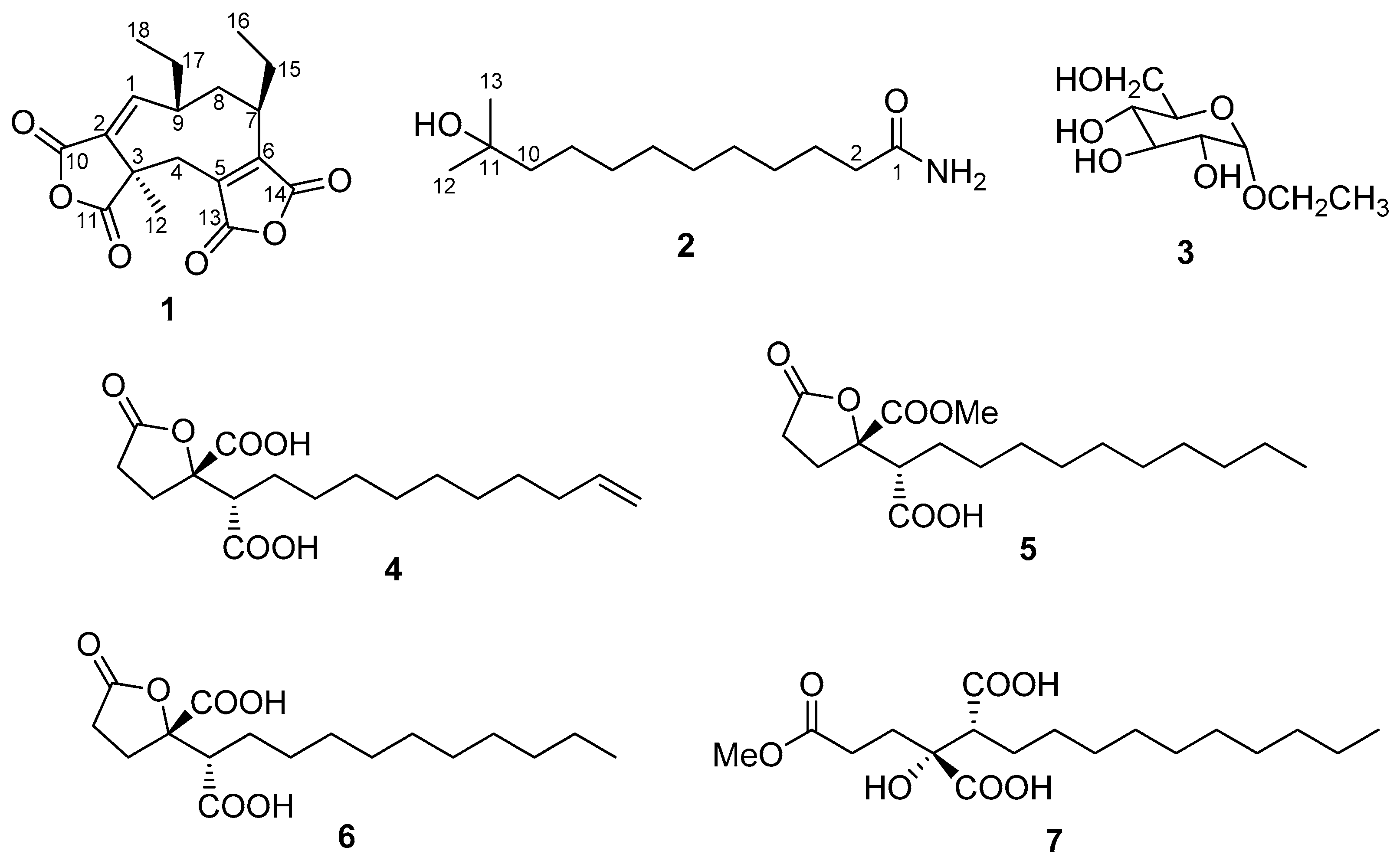
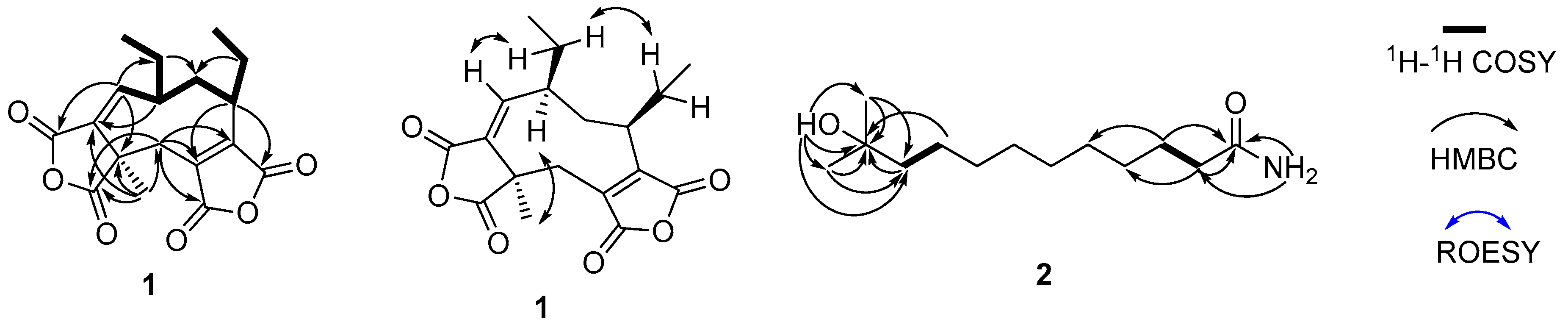
2. Results
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Fungal Material and Cultivation
3.3. Extraction and Isolation
3.4. Antifungal Activity Assay
3.5. Insecticidal Activity Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Elissawy, A.M.; EI-Shazly, M.; Ebada, S.S.; Singab, A.B.; Proksch, P. Bioactive terpenes from marine-derived fungi. Mar. Drugs 2015, 13, 1966–1992. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Xu, G.M.; Li, X.M.; Li, C.S.; Wang, B.G. Characterization of a newly isolated marine fungus Aspergillus dimorphicus for optimized production of the anti-tumor agent Wentilactones. Mar. Drugs 2015, 13, 7040–7054. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.J.; Shao, C.L.; Wu, L.Y.; Chen, M.; Wang, K.L.; Zhao, D.L.; Sun, X.P.; Chen, G.Y.; Wang, C.Y. Bioactive phenylalanine derivatives and cytochalasins from the soft coral-derived fungus, Aspergillus elegans. Mar. Drugs 2013, 11, 2054–2068. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xu, Y.; Shao, C.L.; Yang, R.Y.; Zheng, C.J.; Chen, Y.Y.; Fu, X.M.; Qian, P.Y.; She, Z.G.; de Voogd, N.J.; et al. Antibacterial bisabolane-type sesquiterpenoids from the sponge-derived fungus Aspergillus sp. Mar. Drugs 2012, 10, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Li, W.S.; Xiong, P.; Zheng, W.X.; Zhu, X.W.; She, Z.G.; Ding, W.J.; Li, C.Y. Identification and antifungal activity of compounds from the mangrove endophytic fungus Aspergillus clavatus R7. Mar. Drugs 2017, 15, 259. [Google Scholar] [CrossRef] [PubMed]
- An, C.Y.; Li, X.M.; Li, C.S.; Wang, M.H.; Xu, G.M.; Wang, B.G. Aniquinazolines A–D, four new quinazolinone alkaloids from marine-derived endophytic fungus Aspergillus nidulans. Mar. Drugs 2013, 11, 2682–2694. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Li, X.M.; Meng, L.; Li, C.S.; Gao, S.S.; Shang, Z.; Proksch, P.; Huang, C.G.; Wang, B.G. Nigerapyrones A–H, α-pyrone derivatives from the marine mangrove-derived endophytic fungus Aspergillus niger MA-132. J. Nat. Prod. 2011, 74, 1787–1791. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.M.; Peng, H.; Guo, Z.K.; Cui, J.T.; Song, Y.C.; Tan, R.X. Bioactive alkaloids from the plant endophytic fungus Aspergillus terreus. Planta Med. 2010, 76, 822–824. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.K.; Yan, T.; Guo, Y.; Song, Y.C.; Tan, R.X.; Ge, H.M. p-Terphenyl and diterpenoid metabolites from endophytic Aspergillus sp.YXf3. J. Nat. Prod. 2012, 75, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Yan, T.; Guo, Z.K.; Jiang, R.; Wei, W.; Wang, T.; Guo, Y.; Song, Y.C.; Jiao, R.H.; Tan, R.X.; Ge, H.M. New flavonol and diterpenoids from the endophytic fungus Aspergillus sp. YXf3. Planta Med. 2013, 79, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.K.; Wang, R.; Huang, W.; Li, X.N.; Jiang, R.; Tan, R.X.; Ge, H.M. Aspergiloid I, an unprecedented spirolactone norditerpenoid from the plant-derived endophytic fungus Aspergillus sp. YXf3. Beilstein J. Org. Chem. 2014, 10, 2677–2682. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Li, S.J.; Guo, Z.K.; Zhang, W.J.; Wei, W.; Tan, R.X.; Jiao, R.H. New p-terphenyls from the endophytic fungus Aspergillus sp. YXf3. Bioorg. Med. Chem. Lett. 2017, 27, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Mei, W.L.; Zheng, B.; Zhao, Y.X.; Zhong, H.M.; Wu, X.L.; Zeng, Y.B.; Dong, W.H.; Huang, J.L.; Proksch, P.; Dai, H.F. Meroterpenes from endophytic fungus A1 of mangrove plant Scyphiphora hydrophyllacea. Mar. Drugs 2012, 10, 1993–2001. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.B.; Wang, H.; Zuo, W.J.; Zheng, B.; Yang, T.; Dai, H.F.; Mei, W.L. A fatty acid glycoside from a marine-derived fungus isolated from mangrove plant Scyphiphora hydrophyllacea. Mar. Drugs 2012, 10, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Wang, P.; Chen, H.Q.; Guo, Z.K.; Wang, H.; Dai, H.F.; Mei, W.L. New furan derivatives from a mangrove-derived endophytic fungus Coriolopsis sp. J5. Molecules 2017, 22, 261. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Kong, F.D.; Wang, P.; Yuan, J.Z.; Guo, Z.K.; Wang, H.; Dai, H.F.; Mei, W.L. Two new tremulane sesquiterpenes from a mangrove endophytic fungus, Coriolopsis sp. J5. Chin. Chem. Lett. 2017, 28, 222–225. [Google Scholar] [CrossRef]
- Li, Y.Q.; Wang, B.; Yang, B.; Huang, R.M.; Liu, Y.H.; Zou, X.L. Study on the compounds of marine sponge Haliclona sp. J. Trop. Oceanogr. 2011, 30, 124–126. [Google Scholar]
- Wang, R.; Liu, T.M.; Shen, M.H.; Yang, M.Q.; Feng, Q.Y.; Tang, X.M.; Li, X.M. Spiculisporic acids B–D, three new γ-butenolide derivatives from a sea urchin-derived fungus Aspergillus sp. HDf2. Molecules 2012, 17, 13175–13182. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Guo, Z.K.; Li, X.M.; Chen, F.X.; Zhan, X.F.; Shen, M.H. Spiculisporic acid analogues of the marine-derived fungus, Aspergillus candidus strain HDf2, and their antibacterial activity. Antonie Van Leeuwenhoek 2015, 108, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Fu, Z.D.; He, X.N.; Tang, X.M.; Li, X.M.; Guo, Z.K. New secondary metabolites produced by the marine-derived fungus Aspergillus sp. HDf2. Chin. J. Trop. Crops 2014, 35, 1658–1662. [Google Scholar]
| No. | 1 a | 2 b | ||
|---|---|---|---|---|
| δH, mult. (J in Hz) | δC | δH, mult. (J in Hz) | δC | |
| 1 | 7.00, d (12.5) | 150.1, CH | 174.8, C | |
| 1-NH2 | 7.20, br s, 6.66, br s | |||
| 2 | 131.9, C | 2.02, t (7.7) | 35.6, CH2 | |
| 3 | 48.6, C | 1.47, m | 25.5, CH2 | |
| 4 | 3.28, d (13.0), 2.66, d (13.0) | 31.7, CH2 | 1.20–1.27, m c | 29.2 d, CH2 |
| 5 | 140.5, C | 1.20–1.27, m c | 29.3 d, CH2 | |
| 6 | 148.4, C | 1.20–1.27, m c | 29.4 d, CH2 | |
| 7 | 2.12, m, overlap | 48.1, CH | 1.20–1.27, m c | 29.6 d, CH2 |
| 8 | 2.87, m, 1.98, m | 28.4, CH2 | 1.20–1.27, m c | 30.3 d, CH2 |
| 9 | 2.10, m, overlap | 43.9, CH | 1.28, m | 24.3, CH2 |
| 10 | 163.7, C | 1.31, m | 44.2, CH2 | |
| 11 | 173.7, C | 69.2, C | ||
| 11-OH | 4.04, s | |||
| 12 | 1.49, s | 20.3, CH3 | 1.05, s | 29.8, CH3 |
| 13 | 165.3, C | 1.05, s | 29.8, CH3 | |
| 14 | 164.5, C | |||
| 15 | 1.86, m, 1.14, m | 21.4, CH2 | ||
| 16 | 1.07 t (7.0) | 12.4, CH3 | ||
| 17 | 1.67, m, 1.50, m | 26.0, CH2 | ||
| 18 | 0.81, t (7.5) | 12.8, CH3 | ||
| Samples | Mortality Rate (%) | |||
|---|---|---|---|---|
| 7 d | 10 d | 14 d | 20 d | |
| Compound 1 | 40.00 ± 10.00 | 46.67 ± 5.77 | 60.00 ± 0.00 | 76.67 ± 5.77 |
| Azadirachtin | 86.67 ± 5.77 | 93.33 ± 5.77 | 100.00 ± 0.00 | 100.00 ± 0.00 |
| Control | 0.00 ± 0.00 | 3.33 ± 5.77 | 3.33 ± 5.77 | 6.67 ± 5.77 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, Z.; Gai, C.; Cai, C.; Chen, L.; Liu, S.; Zeng, Y.; Yuan, J.; Mei, W.; Dai, H. Metabolites with Insecticidal Activity from Aspergillus fumigatus JRJ111048 Isolated from Mangrove Plant Acrostichum specioum Endemic to Hainan Island. Mar. Drugs 2017, 15, 381. https://doi.org/10.3390/md15120381
Guo Z, Gai C, Cai C, Chen L, Liu S, Zeng Y, Yuan J, Mei W, Dai H. Metabolites with Insecticidal Activity from Aspergillus fumigatus JRJ111048 Isolated from Mangrove Plant Acrostichum specioum Endemic to Hainan Island. Marine Drugs. 2017; 15(12):381. https://doi.org/10.3390/md15120381
Chicago/Turabian StyleGuo, Zhikai, Cuijuan Gai, Caihong Cai, Liangliang Chen, Shoubai Liu, Yanbo Zeng, Jingzhe Yuan, Wenli Mei, and Haofu Dai. 2017. "Metabolites with Insecticidal Activity from Aspergillus fumigatus JRJ111048 Isolated from Mangrove Plant Acrostichum specioum Endemic to Hainan Island" Marine Drugs 15, no. 12: 381. https://doi.org/10.3390/md15120381
APA StyleGuo, Z., Gai, C., Cai, C., Chen, L., Liu, S., Zeng, Y., Yuan, J., Mei, W., & Dai, H. (2017). Metabolites with Insecticidal Activity from Aspergillus fumigatus JRJ111048 Isolated from Mangrove Plant Acrostichum specioum Endemic to Hainan Island. Marine Drugs, 15(12), 381. https://doi.org/10.3390/md15120381






