An Insight into Sargassum muticum Cytoprotective Mechanisms against Oxidative Stress on a Human Cell In Vitro Model
Abstract
:1. Introduction
2. Results
2.1. Antioxidant Activity
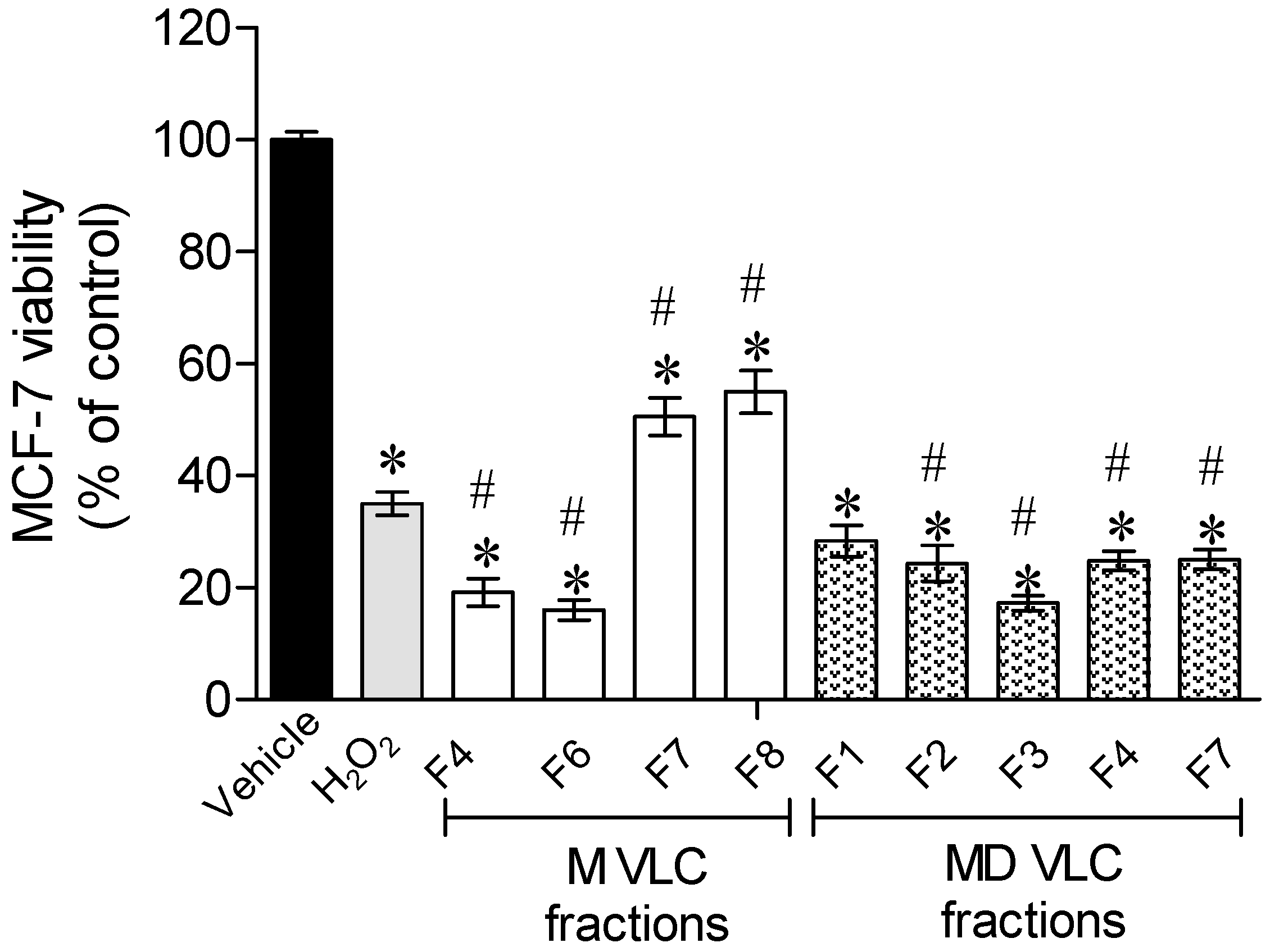
2.2. Protective Effect of Sargassum muticum Fractions on MCF-7 Cells Exposed to H2O2
2.3. Cellular Mechanisms Involved in the Cytotoxicity Induced by H2O2 on MCF-7 Cells in the Presence or Absence of Sargassum muticum Fractions
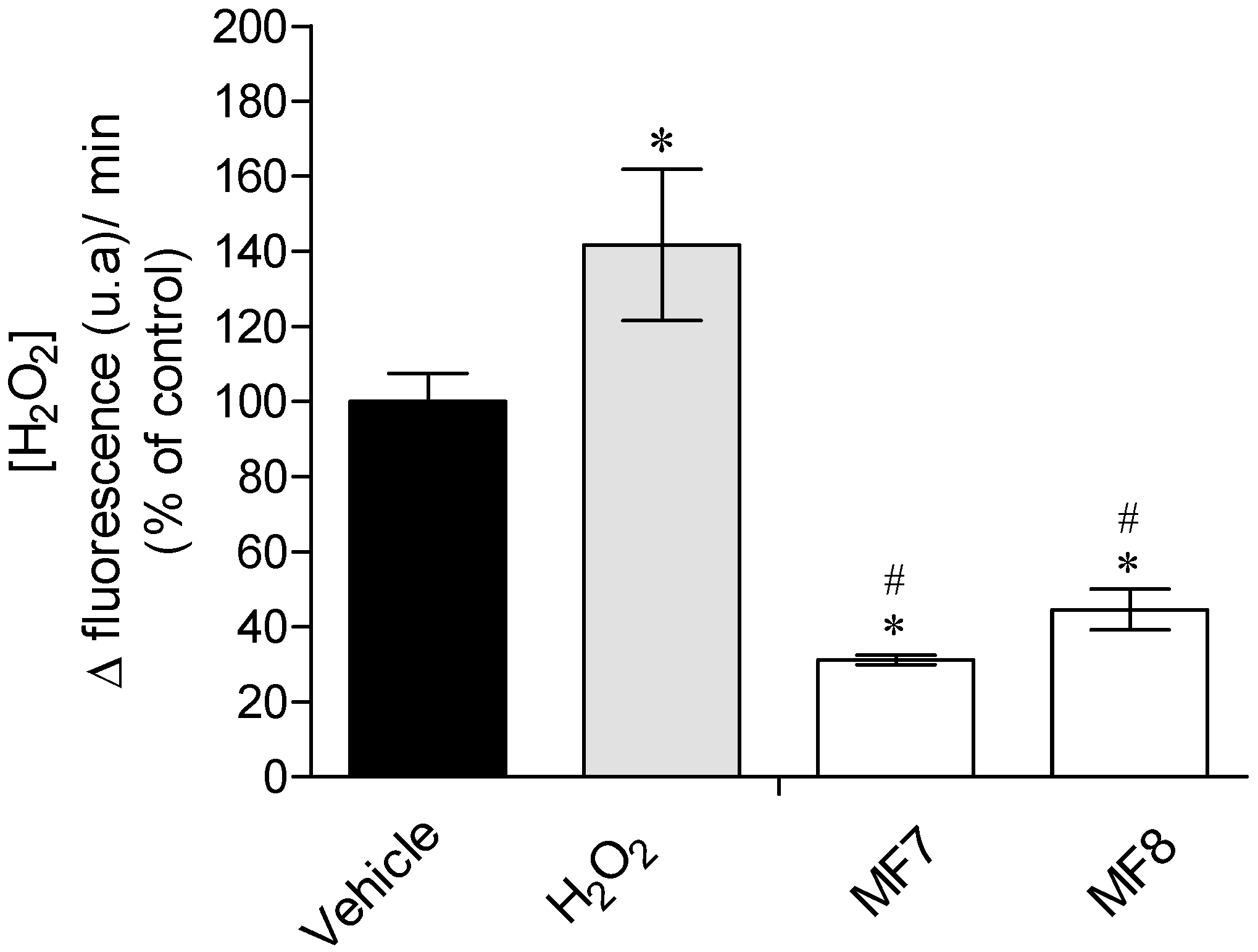
2.3.1. Real-Time Quantification of H2O2 Production
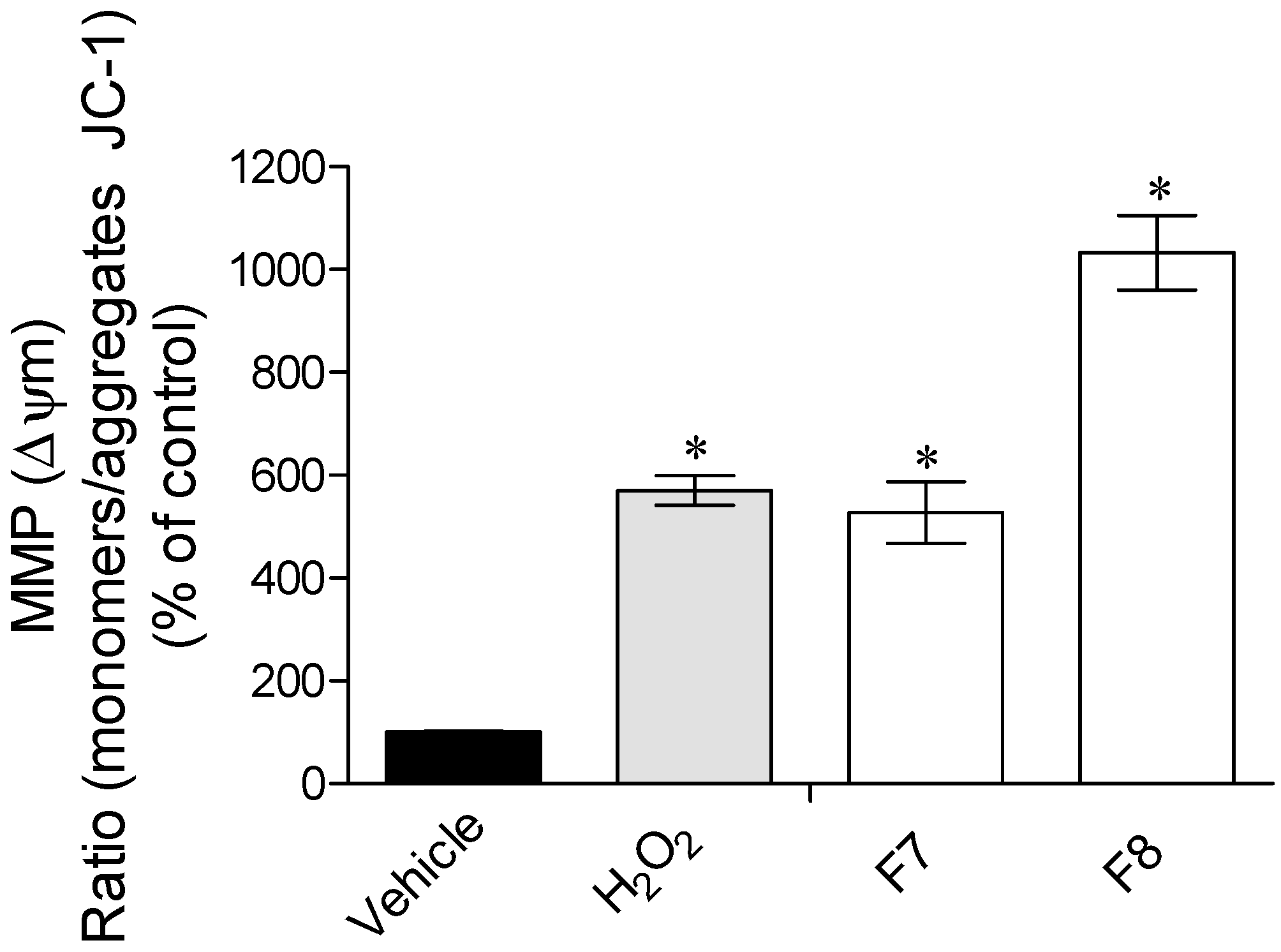
2.3.2. Mitochondrial Membrane Potential (ΔΨm)
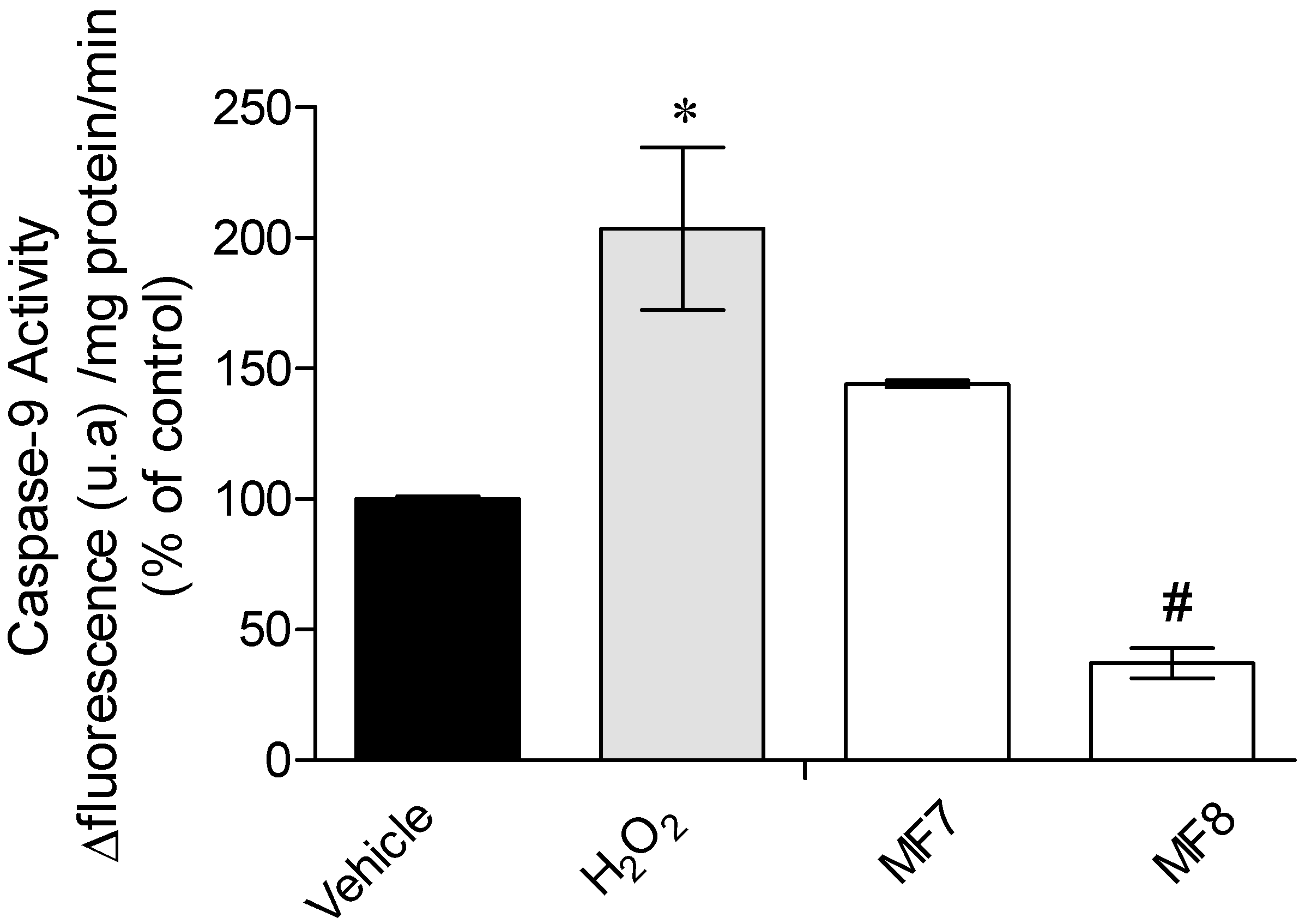
2.3.3. Caspase-9 Activity
3. Discussion
3.1. Vacuum Liquid Chromatography for Bioactive Compounds Concentration
3.2. Protective Effects of Sargassum muticum Enriched Fractions on an Oxidative Stress Condition Induced by H2O2
3.3. Mechanisms of Action Insight: Mitochondrial Membrane Potential and Caspase-9 Activity Evaluation
4. Material and Methods
4.1. Chemicals and Reagents
4.2. Collection, Preparation and Extraction of Sargassum muticum
4.3. Fractionation of Sargassum muticum by Vacuum Liquid Chromatography (VLC)
4.4. Analysis of Total Phenolic Content (TPC)
4.5. Evaluation of Antioxidant Activity
4.5.1. DPPH (1,1-Diphenyl-2-picryl-hydrazyl) Radical Scavenging Activity
4.5.2. Oxygen Radical Absorbance Capacity (ORAC)
4.6. In Vitro Assay of Oxidative Stress Prevention
4.6.1. Cell Maintenance Culture Conditions
4.6.2. Sargassum muticum Fractions Cytotoxicity Evaluation
4.6.3. Evaluation of the Protective Effect of Sargassum muticum Fractions on an Oxidative Stress Condition Induced by H2O2 on MCF-7 Cells
4.6.4. Real-Time Quantification of H2O2 Production
4.6.5. Mitochondrial Membrane Potential (ΔΨm)
4.6.6. Caspase-9 Activity
4.7. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. BioMed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef] [PubMed]
- Anand, S.S.; Hawkes, C.; de Souza, R.J.; Mente, A.; Dehghan, M.; Nugent, R.; Zulyniak, M.A.; Weis, T.; Bernstein, A.M.; Krauss, R.M.; et al. Food consumption and its impact on cardiovascular disease: Importance of solutions focused on the globalized food system: A report from the workshop convened by the world heart federation. J. Am. Coll. Cardiol. 2015, 66, 1590–1614. [Google Scholar] [CrossRef] [PubMed]
- McHugh, D.J. A Guide to the Seaweed Industry; Food and Agriculture Organization of the United Nations Rome: Rome, Italy, 2003. [Google Scholar]
- Teas, J.; Vena, S.; Cone, D.L.; Irhimeh, M. The consumption of seaweed as a protective factor in the etiology of breast cancer: Proof of principle. J. Appl. Phycol. 2013, 25, 771–779. [Google Scholar] [CrossRef] [PubMed]
- Sharifuddin, Y.; Chin, Y.-X.; Lim, P.-E.; Phang, S.-M. Potential bioactive compounds from seaweed for diabetes management. Mar. Drugs 2015, 13, 5447–5491. [Google Scholar] [CrossRef] [PubMed]
- Dore, C.M.P.G.; Faustino Alves, M.G.D.C.; Pofírio Will, L.S.E.; Costa, T.G.; Sabry, D.A.; de Souza Rêgo, L.A.R.; Accardo, C.M.; Rocha, H.A.O.; Filgueira, L.G.A.; Leite, E.L. A sulfated polysaccharide, fucans, isolated from brown algae Sargassum vulgare with anticoagulant, antithrombotic, antioxidant and anti-inflammatory effects. Carbohydr. Polym. 2013, 91, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Mayer, A.M.S.; Rodríguez, A.D.; Berlinck, R.G.S.; Fusetani, N. Marine pharmacology in 2007–2008: Marine compounds with antibacterial, anticoagulant, antifungal, anti-inflammatory, antimalarial, antiprotozoal, antituberculosis, and antiviral activities; affecting the immune and nervous system, and other miscellaneous mechanisms of action. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2011, 153, 191–222. [Google Scholar]
- Pinteus, S.; Silva, J.; Alves, C.; Horta, A.; Fino, N.; Rodrigues, A.I.; Mendes, S.; Pedrosa, R. Cytoprotective effect of seaweeds with high antioxidant activity from the Peniche coast (portugal). Food Chem. 2017, 218, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.; Alves, C.; Horta, A.; Pinteus, S.; Silva, J.; Culioli, G.; Thomas, O.P.; Pedrosa, R. Antitumor and antimicrobial potential of bromoditerpenes isolated from the red alga, Sphaerococcus coronopifolius. Mar. Drugs 2015, 13, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Yende, S.R.; Harle, U.N.; Chaugule, B.B. Therapeutic potential and health benefits of Sargassum species. Pharmacogn. Rev. 2014, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Pádua, D.; Rocha, E.; Gargiulo, D.; Ramos, A.A. Bioactive compounds from brown seaweeds: Phloroglucinol, fucoxanthin and fucoidan as promising therapeutic agents against breast cancer. Phytochem. Lett. 2015, 14, 91–98. [Google Scholar] [CrossRef]
- Hamed, I.; Özogul, F.; Özogul, Y.; Regenstein, J.M. Marine bioactive compounds and their health benefits: A review. Comp. Rev. Food Sci. Food Saf. 2015, 14, 446–465. [Google Scholar] [CrossRef]
- Peng, J.; Yuan, J.-P.; Wu, C.-F.; Wang, J.-H. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar] [CrossRef] [PubMed]
- El Shoubaky, G.A.; Salem, E.A. Terpenes and sterols composition of marine brown algae Padina pavonica (Dictyotales) and Hormophysa triquetra (fucales). Int. J. Pharm. Phytochem. Res 2014, 6, 894–900. [Google Scholar]
- Critchley, A.; Farnham, W.; Yoshida, T.; Norton, T. A bibliography of the invasive alga Sargassum muticum (yendo) fensholt (Fucales; Sargassaceae). Bot. Mar. 1990, 33, 551–562. [Google Scholar] [CrossRef]
- Farnham, W.; Fletcher, R.; Irvine, L.M. Attached Sargassum found in Britain. Nature 1973, 243, 231–232. [Google Scholar] [CrossRef]
- Streftaris, N.; Zenetos, A. Alien marine species in the mediterranean-the 100 ‘worst invasives’ and their impact. Mediterr. Mar. Sci. 2006, 7, 87–118. [Google Scholar] [CrossRef]
- Boudouresque, C.F.; Verlaque, M. Biological pollution in the mediterranean sea: Invasive versus introduced macrophytes. Mar. Pollut. Bull. 2002, 44, 32–38. [Google Scholar] [CrossRef]
- Liu, L.; Heinrich, M.; Myers, S.; Dworjanyn, S.A. Towards a better understanding of medicinal uses of the brown seaweed Sargassum in traditional chinese medicine: A phytochemical and pharmacological review. J. Ethnopharmacol. 2012, 142, 591–619. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, S.; Shanmugiahthevar, K.P.; Kasi, P.D. Cholinesterase inhibitors from Sargassum and Gracilaria gracilis: Seaweeds inhabiting south indian coastal areas (hare island, gulf of mannar). Nat. Prod. Res. 2009, 23, 355–369. [Google Scholar] [CrossRef] [PubMed]
- Ina, A.; Hayashi, K.-I.; Nozaki, H.; Kamei, Y. Pheophytin a, a low molecular weight compound found in the marine brown alga Sargassum fulvellum, promotes the differentiation of pc12 cells. Int. J. Dev. Neurosci. 2007, 25, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Tsang, C.; Ina, A.; Goto, T.; Kamei, Y. Sargachromenol, a novel nerve growth factor-potentiating substance isolated from Sargassum macrocarpum, promotes neurite outgrowth and survival via distinct signaling pathways in pc12d cells. Neuroscience 2005, 132, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Zandi, K.; Ahmadzadeh, S.; Tajbakhsh, S.; Rastian, Z.; Yousefi, F.; Farshadpour, F.; Sartavi, K. Anticancer activity of Sargassum oligocystum water extract against human cancer cell lines. Eur. Rev. Med. Pharmacol. Sci. 2010, 14, 669–673. [Google Scholar] [PubMed]
- Chen, X.; Nie, W.; Yu, G.; Li, Y.; Hu, Y.; Lu, J.; Jin, L. Antitumor and immunomodulatory activity of polysaccharides from Sargassum fusiforme. Food Chem. Toxicol. 2012, 50, 695–700. [Google Scholar] [CrossRef] [PubMed]
- De Zoysa, M.; Nikapitiya, C.; Jeon, Y.-J.; Jee, Y.; Lee, J. Anticoagulant activity of sulfated polysaccharide isolated from fermented brown seaweed Sargassum fulvellum. J. Appl. Phycol. 2008, 20, 67–74. [Google Scholar] [CrossRef]
- Silva, J.; Alves, C.; Pinteus, S.; Horta, A.; Pedrosa, R. High antioxidant activity of sargassum muticum and Padina pavonica collected from peniche coast (portugal). Curr. Opin. Biotechnol. 2013, 24, S116. [Google Scholar] [CrossRef]
- Namvar, F.; Mohamad, R.; Baharara, J.; Zafar-Balanejad, S.; Fargahi, F.; Rahman, H.S. Antioxidant, antiproliferative, and antiangiogenesis effects of polyphenol-rich seaweed (Sargassum muticum). BioMed Res. Int. 2013, 2013, 604787. [Google Scholar] [CrossRef] [PubMed]
- Cook, S.A.; Sugden, P.H.; Clerk, A. Regulation of bcl-2 family proteins during development and in response to oxidative stress in cardiac myocytes. Circ. Res. 1999, 85, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T.; Enokido, Y.; Aoshima, H.; Uchiyama, Y.; Hatanaka, H. Changes in mitochondrial membrane potential during oxidative stress-induced apoptosis in pc12 cells. J. Neurosci. Res. 1997, 50, 413–420. [Google Scholar] [CrossRef]
- Koivikko, R.; Loponen, J.; Pihlaja, K.; Jormalainen, V. High-performance liquid chromatographic analysis of phlorotannins from the brown alga Fucus vesiculosus. Phytochem. Anal. 2007, 18, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Gille, J.; Joenje, H. Cell culture models for oxidative stress: Superoxide and hydrogen peroxide versus normobaric hyperoxia. Mutat. Res./DNAging 1992, 275, 405–414. [Google Scholar] [CrossRef]
- Ryter, S.W.; Kim, H.P.; Hoetzel, A.; Park, J.W.; Nakahira, K.; Wang, X.; Choi, A.M. Mechanisms of cell death in oxidative stress. Antioxid. Redox Signal. 2007, 9, 49–89. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.G.; Chang, T.-S.; Jeong, W.; Kang, D. Methods for detection and measurement of hydrogen peroxide inside and outside of cells. Mol. Cells 2010, 29, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Reactive oxygen species and the central nervous system. J. Neurochem. 1992, 59, 1609–1623. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.-J.; Jeon, Y.-J. Evaluation of diphlorethohydroxycarmalol isolated from Ishige okamurae for radical scavenging activity and its protective effect against H2O2-induced cell damage. Process Biochem. 2009, 44, 412–418. [Google Scholar] [CrossRef]
- Kang, S.-M.; Cha, S.-H.; Ko, J.-Y.; Kang, M.-C.; Kim, D.; Heo, S.-J.; Kim, J.-S.; Heu, M.S.; Kim, Y.-T.; Jung, W.-K. Neuroprotective effects of phlorotannins isolated from a brown alga, Ecklonia cava, against H2O2-induced oxidative stress in murine hippocampal HT22 cells. Environ. Toxicol. Pharmacol. 2012, 34, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Pinteus, S.; Silva, J.; Alves, C.; Horta, A.; Thomas, O.P.; Pedrosa, R. Antioxidant and cytoprotective activities of Fucus spiralis seaweed on a human cell in vitro model. Int. J. Mol. Sci. 2017, 18, 292. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, A.M.; O’Callaghan, Y.C.; O’Grady, M.N.; Queguineur, B.; Hanniffy, D.; Troy, D.J.; Kerry, J.P.; O’Brien, N.M. In vitro and cellular antioxidant activities of seaweed extracts prepared from five brown seaweeds harvested in spring from the west coast of Ireland. Food Chem. 2011, 126, 1064–1070. [Google Scholar] [CrossRef]
- Blanc, C.; Deveraux, Q.L.; Krajewski, S.; Jänicke, R.U.; Porter, A.G.; Reed, J.C.; Jaggi, R.; Marti, A. Caspase-3 is essential for procaspase-9 processing and cisplatin-induced apoptosis of MCF-7 breast cancer cells. Cancer Res. 2000, 60, 4386–4390. [Google Scholar] [PubMed]
- Chia, Y.Y.; Kanthimathi, M.; Khoo, K.S.; Rajarajeswaran, J.; Cheng, H.M.; Yap, W.S. Antioxidant and cytotoxic activities of three species of tropical seaweeds. BMC Complement. Altern. Med. 2015, 15, 339. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-S.; Shin, T.; Utsuki, T.; Choi, J.-S.; Byun, D.-S.; Kim, H.-R. Isolation and identification of phlorotannins from Ecklonia stolonifera with antioxidant and hepatoprotective properties in tacrine-treated HepG-2 cells. J. Agric. Food Chem. 2012, 60, 5340–5349. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Dávalos, A.; Gómez-Cordovés, C.; Bartolomé, B. Extending applicability of the oxygen radical absorbance capacity (ORAC−fluorescein) assay. J. Agric. Food Chem. 2004, 52, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.V.; Walsh, N.A. Antioxidant and antiproliferative activities of extracts from a variety of edible seaweeds. Food Chem. Toxicol. 2006, 44, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, J.; Jaffe, J.S.; Schulman, E.S.; Raible, D.G. A highly sensitive fluorescent micro-assay of H2O2 release from activated human leukocytes using a dihydroxyphenoxazine derivative. J. Immunol. Methods 1997, 202, 133–141. [Google Scholar] [CrossRef]
- Zar, J.H. Biostatistical Analysis; Prentice Hall: Upper Saddle River, NJ, USA, 2010. [Google Scholar]
| Fraction | TPC a | DPPH b | ORAC c |
|---|---|---|---|
| Crude MDE | 75.754 ± 0.026 | 28.42 (26.88–30.04) | 12,634.197 ± 312.511 |
| MDF1 | 25.786 ± 0.015 | >1000 | 1.456 ± 0.531 |
| MDF2 | 55.867 ± 0.025 | >1000 | 16.136 ± 0.330 |
| MDF3 | 38.854 ± 0.017 | >1000 | 29.088 ± 5.833 |
| MDF4 | 34.309 ± 0.006 | >1000 | 11.353 ± 3.432 |
| MDF5 | 60.056 ± 0.024 | 57.33 (51.63–63.66) | 280.989 ± 13.153 |
| MDF6 | 36.779 ± 0.008 | 81.13 (67.95–96.87) | 136.825 ± 14.404 |
| MDF7 | 21.668 ± 0.008 | 98.84 (84.62–115.5) | 770 ± 26.332 |
| MDF8 | 84.080 ± 0.026 | 32.29 (28.95–36.02) | 3040.143 ± 266.235 |
| Crude ME | 85.256 ± 1.158 | 53.1 (46.7–67.82) | 2672.80 ± 54.22 |
| MF2 | 20.447 ± 0.964 | 106.4 (92.9–121.9) | 14.662 ± 0.514 |
| MF3 | 16.860 ± 0.565 | 291.0 (261.9–323.3) | 17.458 ± 0.152 |
| MF4 | 25.619 ± 1.053 | >1000 | 42.489 ± 0.895 |
| MF5 | 26.653 ± 0.831 | >1000 | 77.877 ± 22.648 |
| MF6 | 11.663 ± 0.413 | >1000 | 42.892 ± 0.190 |
| MF7 | 36.723 ± 1.314 | >1000 | 44.404 ± 15.050 |
| MF8 | 61.668 ± 1.496 | 36.4 (32.4–41.0) | 51.975 ± 4.280 |
| BHT | - | 50.3 (36.0–54.3) | 330.69 ± 37.52 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinteus, S.; Lemos, M.F.L.; Silva, J.; Alves, C.; Neugebauer, A.; Freitas, R.; Duarte, A.; Pedrosa, R. An Insight into Sargassum muticum Cytoprotective Mechanisms against Oxidative Stress on a Human Cell In Vitro Model. Mar. Drugs 2017, 15, 353. https://doi.org/10.3390/md15110353
Pinteus S, Lemos MFL, Silva J, Alves C, Neugebauer A, Freitas R, Duarte A, Pedrosa R. An Insight into Sargassum muticum Cytoprotective Mechanisms against Oxidative Stress on a Human Cell In Vitro Model. Marine Drugs. 2017; 15(11):353. https://doi.org/10.3390/md15110353
Chicago/Turabian StylePinteus, Susete, Marco F. L. Lemos, Joana Silva, Celso Alves, Agnieszka Neugebauer, Rafaela Freitas, Adriana Duarte, and Rui Pedrosa. 2017. "An Insight into Sargassum muticum Cytoprotective Mechanisms against Oxidative Stress on a Human Cell In Vitro Model" Marine Drugs 15, no. 11: 353. https://doi.org/10.3390/md15110353
APA StylePinteus, S., Lemos, M. F. L., Silva, J., Alves, C., Neugebauer, A., Freitas, R., Duarte, A., & Pedrosa, R. (2017). An Insight into Sargassum muticum Cytoprotective Mechanisms against Oxidative Stress on a Human Cell In Vitro Model. Marine Drugs, 15(11), 353. https://doi.org/10.3390/md15110353









