Anti-Fatigue Effect by Peptide Fraction from Protein Hydrolysate of Croceine Croaker (Pseudosciaena crocea) Swim Bladder through Inhibiting the Oxidative Reactions including DNA Damage
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of Protein Hydrolysates of Swim Bladder and Their HO• Scavenging Activities
2.2. Preparation of Antioxidant Peptides from SBP
2.2.1. Fractionation of SBP by Ultrafiltration
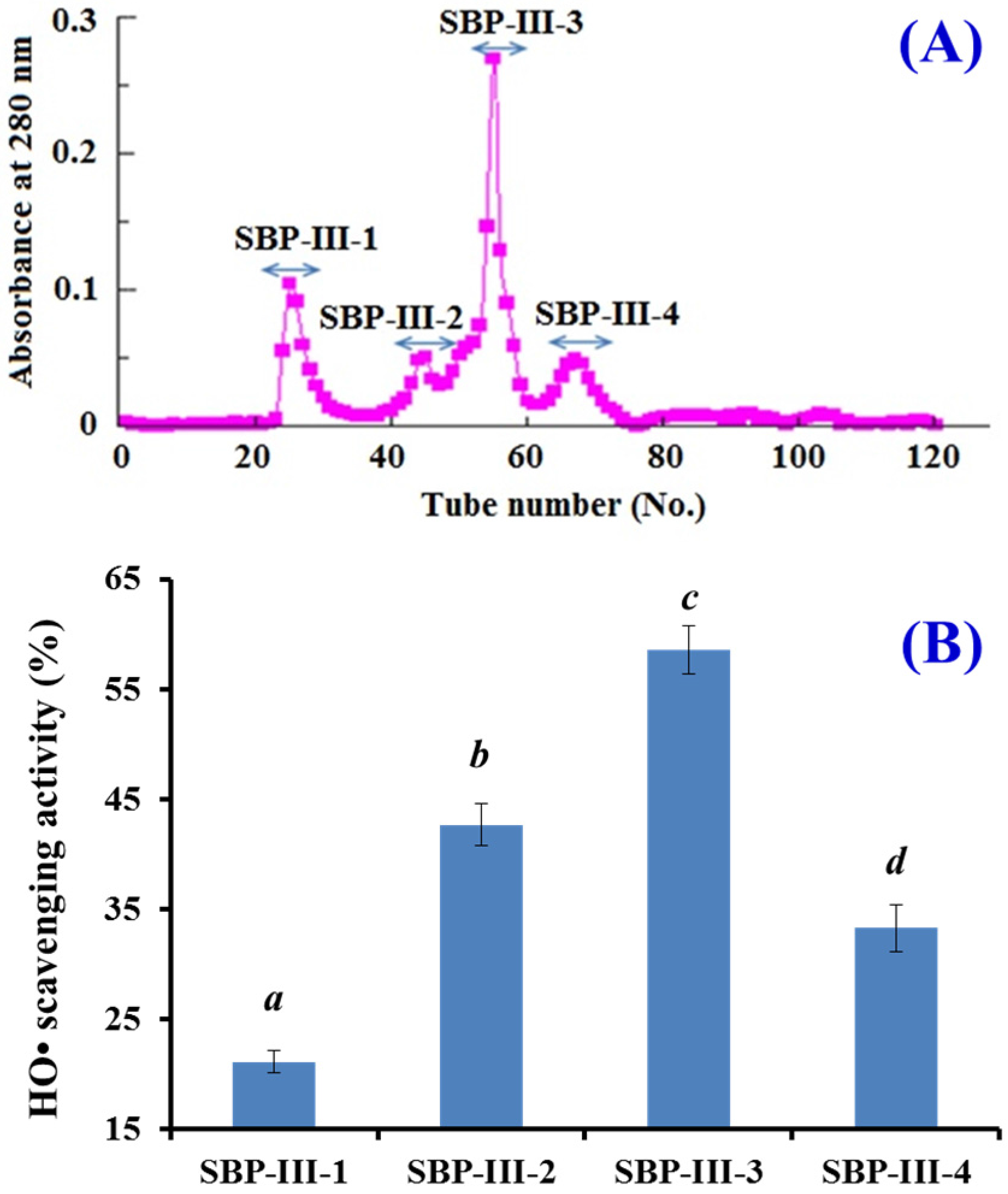
2.2.2. Gel Filtration Chromatography of SBP-III
2.3. In Vitro Antioxidant Activity of SBP-III-3
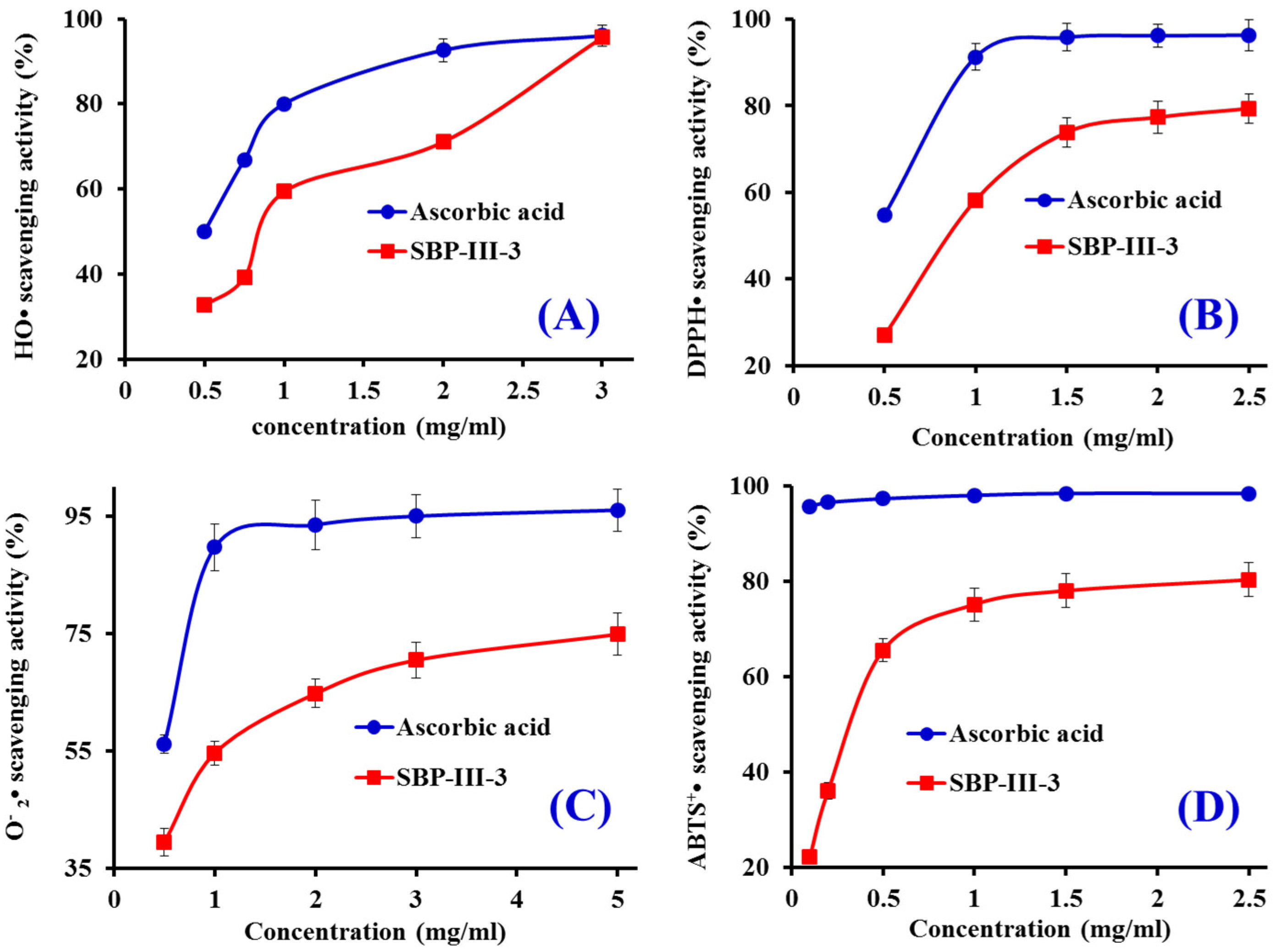
2.3.1. HO• Scavenging Activity
2.3.2. DPPH• Scavenging Activity
2.3.3. • Scavenging Activity
2.3.4. ABTS+• Scavenging Activity
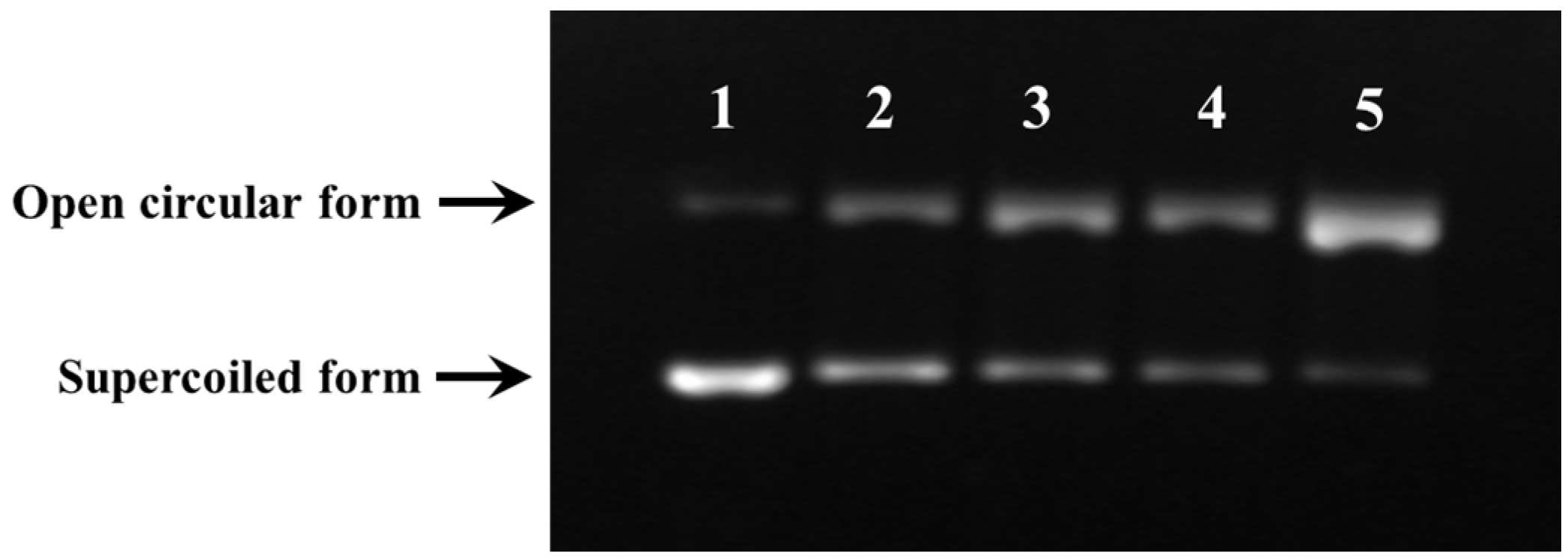
2.3.5. Protective Activity against Free Radical-Induced DNA Damage
2.4. In Vivo Anti-Fatigue Effects of SBP-III-3
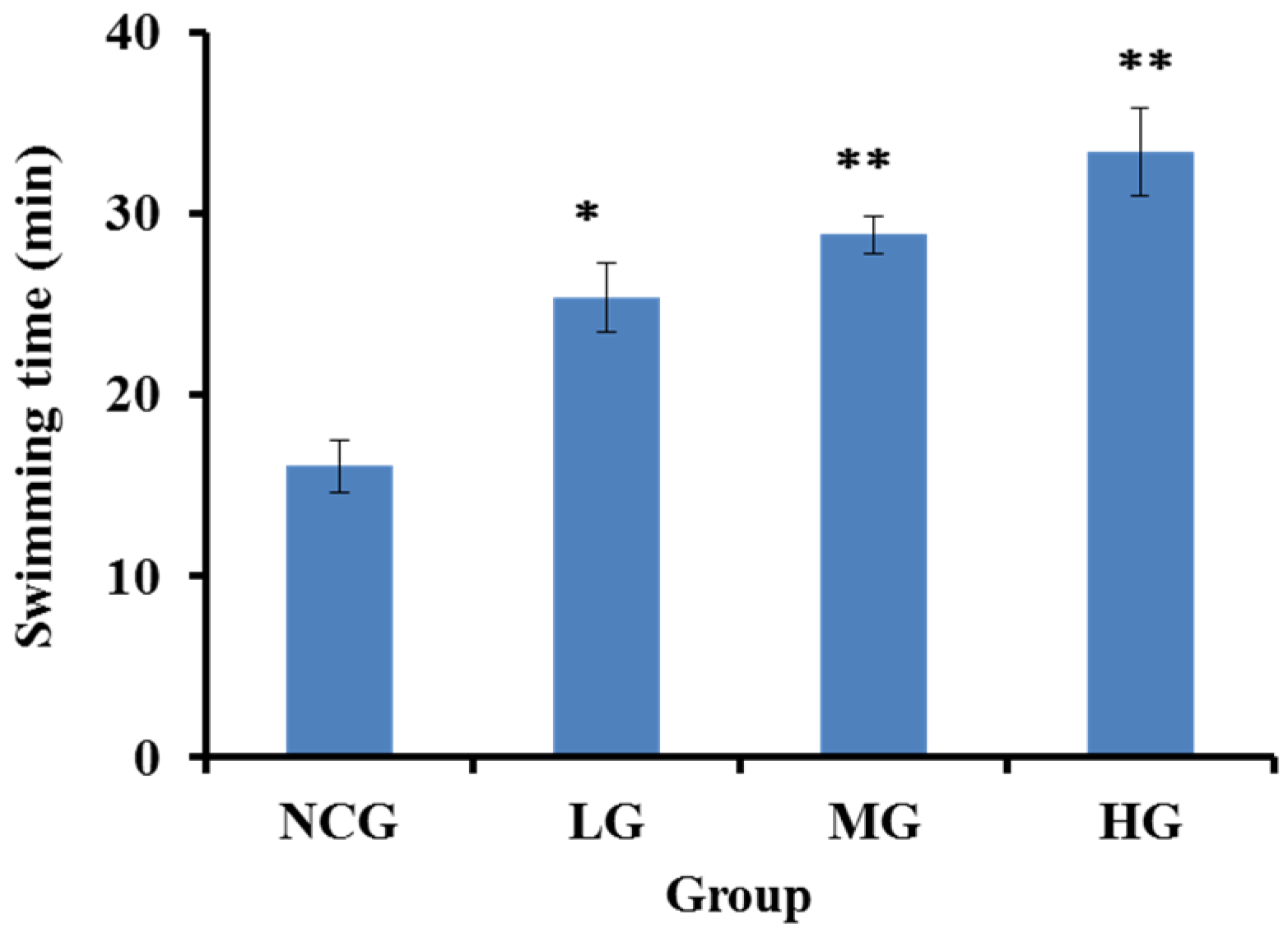
2.4.1. SBP-III-3 Prolonged Exhaustive Swimming Time
2.4.2. Biologic Parameters Determination
SBP-III-3 Decreased Blood Urea Nitrogen (BUN)
SBP-III-3 Decreased Lactic Acid (LA)
SBP-III-3 Increased the Activity of Lactic Dehydrogenase (LDH)
SBP-III-3 Increased Liver and Muscle Glycogens
2.4.3. SBP-III-3 Enhanced the Antioxidant Enzymes and Decreased the Malondialdehyde (MDA)
2.5. Amino Acid Composition of SBP-III-3
3. Experimental Section
3.1. Chemicals and Reagents
3.2. Preparation Protein Hydrolysate of Swim Bladders
3.3. Isolation and Purification of Antioxidant Peptide from SBP
3.4. Degree of Hydrolysis (DH)
3.5. MW Distribution
3.6. Amino Acid Composition Analysis
3.7. Antioxidant Activity
3.7.1. Radical Scavenging Activities
HO• Scavenging Activity
DPPH• Scavenging Activity
• Scavenging Activity
ABTS+• Scavenging Activity
3.7.2. DNA Damage Protective Effect
3.8. Animals and Experimental Diets
3.9. In Vivo Anti-Fatigue Effect of SBP-III-3
3.9.1. Weight-Loaded Swimming Test in ICR Mice
3.9.2. Biochemical Parameter Determination on Anti-Fatigue
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- You, L.; Zhao, M.; Regenstein, J.M.; Ren, J. In vitro antioxidant activity and in vivo anti-fatigue effect of loach (Misgurnus anguillicaudatus) peptides prepared by papain digestion. Food Chem. 2011, 124, 188–194. [Google Scholar] [CrossRef]
- Ding, J.F.; Li, Y.Y.; Xu, J.J.; Su, X.Y.; Gao, X.; Yue, F.P. Study on effect of jellyfish collagen hydrolysate on anti-fatigue and anti-oxidation. Food Hydrocoll. 2011, 25, 1350–1353. [Google Scholar] [CrossRef]
- Xu, C.; Lv, J.; Lo, Y.M.; Cui, S.W.; Hu, X.; Fan, M. Effects of oat β-glucan on endurance exercise and its anti-fatigue properties in trained rats. Carbohyd. Polym. 2013, 92, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- Agyei, D.; Ongkudon, C.M.; Wei, C.Y.; Chan, A.S.; Danquah, M.K. Bioprocess challenges to the isolation and purification of bioactive peptides. Food Bioprod. Process 2016, 98, 244–256. [Google Scholar] [CrossRef]
- Sila, A.; Bougatef, A. Antioxidant peptides from marine by-products: Isolation, identification and application in food systems. A review. J. Funct. Foods 2016, 21, 10–26. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, H.L.; Lu, R.; Zhou, Y.J.; Ma, R.; Lv, J.Q.; Li, X.L.; Chen, L.J.; Yao, Z. The decapeptide CMS001 enhances swimming endurance in mice. Peptides 2008, 29, 1176–1182. [Google Scholar] [CrossRef] [PubMed]
- Chi, C.F.; Hu, F.Y.; Wang, B.; Li, Z.R.; Luo, H.Y. Influence of amino acid compositions and peptide profiles on antioxidant capacities of two protein hydrolysates from skipjack tuna (Katsuwonus pelamis) dark muscle. Mar. Drugs 2015, 13, 2580–2601. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Huang, C.; Luo, H.; Song, R. Progress in preparation and application of high Fischer ratio oligopeptides derived from food protein source. Food Sci. 2014, 35, 289–294. [Google Scholar]
- Li, Z.R.; Wang, B.; Chi, C.F.; Zhang, Q.H.; Gong, Y.D.; Tang, J.J.; Luo, H.Y.; Ding, G.F. Isolation and characterization of acid soluble collagens and pepsin soluble collagens from the skin and bone of spanish mackerel (Scomberomorous niphonius). Food Hydrocoll. 2013, 31, 103–113. [Google Scholar] [CrossRef]
- Pan, X.; Zhao, Y.Q.; Hu, F.Y.; Wang, B. Preparation and identification of antioxidant peptides from protein hydrolysate of skate (Raja porosa) cartilage. J. Funct. Foods 2016, 25, 220–230. [Google Scholar] [CrossRef]
- Chi, C.F.; Hu, F.Y.; Wang, B.; Ren, X.J.; Deng, S.G.; Wu, C.W. Purification and characterization of three antioxidant peptides from protein hydrolyzate of croceine croaker (Pseudosciaena crocea) muscle. Food Chem. 2015, 168, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.Q.; Li, T.; Wang, B.; Ding, G.F. Preparation and characterization of acid and pepsin-soluble collagens from scales of croceine and redlip croakers. Food Sci. Biotechnol. 2015, 24, 2003–2010. [Google Scholar] [CrossRef]
- Wang, B.; Wang, Y.M.; Chi, C.F.; Luo, H.Y.; Deng, S.G.; Ma, J.Y. Isolation and characterization of collagen and antioxidant collagen peptides from scales of croceine croaker (Pseudosciaena crocea). Mar. Drugs 2013, 11, 4641–4661. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Qian, Y.; Li, G.J.; Tan, J. Preventive effects of the polysaccharide of Larimichthys crocea swim bladder on carbon tetrachloride (CCl4)-induced hepatic damage. Chin. J. Nat. Med. 2015, 13, 521–528. [Google Scholar] [CrossRef]
- Abeyrathne, E.D.; Lee, H.Y.; Jo, C.; Suh, J.W.; Ahn, D.U. Enzymatic hydrolysis of ovomucin and the functional and structural characteristics of peptides in the hydrolysates. Food Chem. 2016, 192, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Chi, C.; Hu, F.; Li, Z.; Wang, B.; Luo, H. Influence of different hydrolysis processes by trypsin on the physicochemical, antioxidant, and functional properties of collagen hydrolysates from Sphyrna lewini, Dasyatis akjei, and Raja porosa. J. Aquat. Food Prod. Technol. 2016, 25, 616–632. [Google Scholar] [CrossRef]
- Chi, C.F.; Wang, B.; Wang, Y.M.; Zhang, B.; Deng, S.G. Isolation and characterization of three antioxidant peptides from protein hydrolysate of bluefin leatherjacket (Navodon septentrionalis) heads. J. Funct. Foods 2015, 12, 1–10. [Google Scholar] [CrossRef]
- You, L.; Zhao, M.; Regenstein, J.M.; Ren, J. Purification and identification of antioxidative peptides from loach (Misgurnus anguillicaudatus) protein hydrolysate by consecutive chromatography and electrospray ionization-mass spectrometry. Food Res. Int. 2010, 43, 1167–1173. [Google Scholar] [CrossRef]
- Ren, J.; Zhao, M.; Shi, J.; Wang, J.; Jiang, Y.; Cui, C.; Kakuda, Y.; Xue, S.J. Purification and identification of antioxidant peptides from grass carp muscle hydrolysates by consecutive chromatography and electrospray ionization-mass spectrometry. Food Chem. 2008, 108, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Wu, X.; Zhang, Y.; Li, X.; Ma, S.; Li, J. Purification and characterization of three antioxidant peptides from protein hydrolysate of grass carp (Ctenopharyngodon idella) skin. J. Funct. Foods 2015, 16, 234–242. [Google Scholar] [CrossRef]
- Wang, B.; Li, L.; Chi, C.F.; Ma, J.H.; Luo, H.Y.; Xu, Y.F. Purification and characterisation of a novel antioxidant peptide derived from blue mussel (Mytilus edulis) protein hydrolysate. Food Chem. 2013, 138, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Ahn, C.B.; Kim, J.G.; Je, J.Y. Purification and antioxidant properties of octapeptide from salmon byproduct protein hydrolysate by gastrointestinal digestion. Food Chem. 2014, 147, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Kim, Y.S.; Ahn, C.B.; Je, J.Y. Partial purification and identification of three antioxidant peptides with hepatoprotective effects from blue mussel (Mytilus edulis) hydrolysate by peptic hydrolysis. J. Funct. Foods 2016, 20, 88–95. [Google Scholar] [CrossRef]
- Zhuang, H.; Tang, N.; Yuan, Y. Purification and identification of antioxidant peptides from corn gluten meal. J. Funct. Foods 2013, 5, 1810–1821. [Google Scholar] [CrossRef]
- Hong, J.; Chen, T.T.; Hu, P.; Yang, J.; Wang, S.Y. Purification and characterization of an antioxidant peptide (GSQ) from Chinese leek (Allium tuberosum Rottler) seeds. J. Funct. Foods 2014, 10, 1–10. [Google Scholar] [CrossRef]
- Rajapakse, N.; Mendis, E.; Jung, W.K.; Je, J.Y.; Kim, S.K. Purification of a radical scavenging peptide from fermented mussel sauce and its antioxidant properties. Food Res. Int. 2005, 38, 175–182. [Google Scholar] [CrossRef]
- Apostolou, A.; Stagos, D.; Galitsiou, E.; Spyrou, A.; Haroutounian, S.; Portesis, N.; Trizoglou, I.; Wallace Hayes, A.; Tsatsakis, A.M.; Kouretas, D. Assessment of polyphenolic content, antioxidant activity, protection against ROS-induced DNA damage and anticancer activity of Vitisvinifera stem extracts. Food Chem. Toxicol. 2013, 61, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.Y.; Lu, Y.H.; Tian, C.R.; Xu, J.G.; Guo, X.P.; Zhou, R.; Hao, G. Main nutrients, phenolics, antioxidant activity, DNA damage protective effect and microstructure of Sphallerocarpus gracilis root at different harvest time. Food Chem. 2011, 127, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D.Q.; Guo, Y.; Xu, D.H.; Huang, Y.S.; Yuan, K.; Lv, Z.Q. Antioxidant and anti-fatigue effects of anthocyanins of mulberry juice purification (MJP) and mulberry marc purification (MMP) from different varieties mulberry fruit in China. Food Chem. Toxicol. 2013, 59, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Koo, H.N.; Lee, J.K.; Hong, S.H.; Kim, H.M. Herbkines increases physical stamina in mice. Biol. Pharm. Bull. 2004, 27, 117–119. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.A.; Subramoniam, A.; Rajasekharan, S.; Pushpangadan, P. Effect of Trichopus zeylanicus leaf extract on the energy metabolism in mice during exercise and at rest. Indian J. Pharmacol. 2002, 34, 32–37. [Google Scholar]
- Jia, J.M.; Wu, C.F. Antifatigue activity of tissue culture extracts of Saussurea involucrate. Pharm. Biol. 2008, 46, 433–436. [Google Scholar] [CrossRef]
- Van-Loon, L.J.; Saris, W.H.; Kruijshoop, M.; Wagenmakers, A.J. Maximizing postexercise muscle glycogen synthesis: carbohydrate supplementation and the application of amino acid or protein hydrolysate mixtures. Am. J. Clin. Nutr. 2000, 72, 106–111. [Google Scholar] [PubMed]
- Wang, J.J.; Shieh, M.J.; Kuo, S.L.; Lee, C.L.; Pan, T.M. Effect of red mold rice on antifatigue and exercise-related changes in lipid peroxidation in endurance exercise. Appl. Microbiol. Biotechnol. 2006, 70, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; DeRuisseau, K.C.; Quindry, J.; Hamilton, K.L. Dietary antioxidants and exercise. J. Sport Sci. 2004, 22, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.C.; Kim, D.; Jeon, Y.J. Protective effect of a novel antioxidative peptide purified from a marine Chlorella ellipsoidea protein against free radical-induced oxidative stress. Food Chem. Toxicol. 2012, 50, 2294–2302. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, S.; Fan, Y.; Chen, Y.; Liu, D.; Cheng, H.; Gao, X.; Zhou, Y. Anti-fatigue activity of the water-soluble polysaccharides isolated from Panax ginseng C. A. Meyer. J. Ethnopharmacol. 2010, 130, 421–423. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chi, Y.J.; Zhao, M.Y.; Lv, L. Purification and identification of antioxidant peptides from egg white protein hydrolysate. Amino Acids 2012, 43, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Nimalaratne, C.; Bandara, N.; Wu, J. Purification and characterization of antioxidant peptides from enzymatically hydrolyzed chicken egg white. Food Chem. 2015, 188, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Vineyard, K.R.; Gordonm, M.E.; Graham-Thiersm, P.; Jerina, M. Effects of daily administration of an amino acid-based supplement on muscle and exercise metabolism in working horses. J. Equine Vet. Sci. 2013, 33, 321–399. [Google Scholar] [CrossRef]
- Bird, S.P.; Tarpenning, K.M.; Marino, F.E. Liquid carbohydrate/essential amino acid ingestion during a short-termbout of resistance exercise suppresses myofibrillar protein degradation. Metabolism 2006, 55, 570–577. [Google Scholar] [CrossRef] [PubMed]
- Guezennec, C.Y.; Abdelmalki, A.; Serrurier, B.; Merino, D.; Bigard, X.; Berthelot, M.; Pierard, C.; Peres, M. Effect of prolonged exercise on brain ammonia and amino acids. Int. J. Sports Med. 1998, 19, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Marquezi, M.L.; Roschel, H.A.; Costa, A.D.S.; Sawada, L.A.; Lancha, A.H., Jr. Effect of aspartate and asparagine supplementation on fatigue determinants in intense exercise. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Bazzarre, T.L.; Murdoch, S.D.; Wu, S.M.; Herr, D.G.; Snider, I.P. Plasma amino acid responses of trained athletes to two successive exhaustion trials with and without interim carbohydrate feeding. J. Am. Coll. Nutr. 1992, 11, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, B.; Chi, C.; Gong, Y.; Luo, H.; Ding, G. Influence of average molecular weight on antioxidant and functional properties of cartilage collagen hydrolysates from Sphyrna lewini, Dasyatis akjei and Raja porosa. Food Res. Int. 2013, 51, 283–293. [Google Scholar] [CrossRef]
- Chi, C.F.; Wang, B.; Li, Z.R.; Luo, H.Y.; Ding, G.F.; Wu, C.W. Characterization of acid-soluble collagen from the skin of hammerhead shark (Sphyrna lewini). J. Food Biochem. 2014, 38, 236–247. [Google Scholar] [CrossRef]
- Yust, M.M.; Pedroche, J.; Girón-Calle, J.; Vioque, J.; Millán, F.; Alaiz, M. Determination of tryptophan by high-performance liquid chromatography of alkaline hydrolysates with spectrophotometric detection. Food Chem. 2004, 85, 317–320. [Google Scholar] [CrossRef]
- Liu, J.; Jia, L.; Kan, J.; Jin, C.H. In vitro and in vivo antioxidant activity of ethanolic extract of white button mushroom (Agaricus bisporus). Food Chem. Toxicol. 2013, 51, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Yu, K.Q.; Liu, Y.Y.; Ouyang, M.Z.; Yan, M.H.; Luo, R.; Zhao, X.S. Anti-fatigue activity of polysaccharides extract from Radix Rehmanniae Preparata. Int. J. Biol. Macromol. 2012, 50, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Lu, Z.X.; Bie, X.M.; Lu, F.X.; Huang, X.Q. Scavenging and anti-fatigue activity of fermented defatted soybean peptides. Eur. Food Res. Technol. 2008, 226, 415–421. [Google Scholar] [CrossRef]
| Protease | Enzymolysis Condition | Degree of Hydrolysis (DH%) | HO• Scavenging Rate (%) |
|---|---|---|---|
| Papain | pH 7.0, 60 °C, 4 h, total enzyme dose 2.5% | 17.84 ± 0.71 a | 34.85 ± 1.05 a |
| Alcalase | pH 8.0, 50 °C, 4 h, total enzyme dose 2.5% | 22.32 ± 0.74 b | 54.76 ± 1.94 b |
| Trypsin | pH 8.0, 40 °C, 4 h, total enzyme dose 2.5% | 16.21 ± 0.37 c | 62.38 ± 1.67 c |
| Pepsin | pH 2.0, 37 °C, 4 h, total enzyme dose 2.5% | 19.52 ± 0.49 d | 55.47 ± 2.02 b |
| Neutrase | pH 6.0, 50 °C, 4 h, total enzyme dose 2.5% | 21.37 ± 0.67 b | 50.67 ± 1.85 d |
| NCG | SBP-III-3-LG | SBP-III-3-MG | SBP-III-3-HG | |
|---|---|---|---|---|
| BUN (mmol/L) | 9.34 ± 0.39 | 7.81 ± 0.61 ** | 7.79 ± 0.47 ** | 7.28 ± 0.43 **,a |
| LA (mmol/L) | 3.08 ± 0.21 | 2.65 ± 0.47 * | 2.56 ± 0.35 * | 2.46 ± 0.34 * |
| LDH (U/gprot) | 2784.95 ± 322.92 | 3397.10 ± 215.90 | 3605.87 ± 315.21 * | 3690.76 ± 337.18 * |
| Liver glycogen (mg/g) | 8.32 ± 0.47 | 11.29 ± 2.31 * | 17.36 ± 1.16 ** | 21.41 ± 5.23 **,b |
| Muscle glycogen (mg/g) | 2.23 ± 0.56 | 2.44 ± 0.36 | 3.39 ± 0.35 * | 4.80 ± 1.12 * |
| SOD (U/mg prot) | 68.82 ± 6.17 | 71.74 ± 2.52 | 79.63 ± 7.40 * | 99.24 ± 4.38 **,b |
| GSH-Px (IU) | 43.22 ± 4.09 | 71.89 ± 2.34 | 102.05 ± 5.78 ** | 147.16 ± 12.80 **,b |
| CAT (U/g prot) | 186.14 ± 2.26 | 325.27 ± 1.52 * | 349.75 ± 4.09 * | 483.00 ± 5.87 **,a |
| MDA in liver (mmol/L) | 2.39 ± 0.55 | 1.72 ± 0.25 ** | 1.23 ± 0.31 **,a | 1.11 ± 0.23 **,b |
| MDA in plasma (mmol/L) | 19.92 ± 2.87 | 11.75 ± 2.62 ** | 9.97 ± 1.31 ** | 9.50 ± 0.55 ** |
| Amino Acid | Concentration (μmol/L) | Composition (%) |
|---|---|---|
| Asp | 871.33 ± 18.56 | 4.60 ± 0.10 |
| Glu | 1317.30 ± 35.14 | 6.86 ± 0.18 |
| Ser | 540.61 ± 20.47 | 2.82 ± 0.11 |
| Gly | 6744.51 ± 143.47 | 35.10 ± 0.75 |
| His | 86.39 ± 2.34 | 0.45 ± 0.04 |
| Arg | 718.84 ± 12.38 | 3.74 ± 0.06 |
| Thr | 450.29 ± 13.54 | 2.34 ± 0.07 |
| Ala | 2343.97 ± 34.56 | 13.46 ± 0.19 |
| Pro | 2165.95 ± 40.68 | 11.28 ± 0.21 |
| Hyp | 1629.48 ± 26.87 | 8.49 ± 0.14 |
| Tyr | 60.96 ± 1.35 | 0.32 ± 0.01 |
| Val | 499.64 ± 12.58 | 2.60 ± 0.07 |
| Met | 244.28 ± 8.51 | 1.27 ± 0.05 |
| Ile | 207.49 ± 8.34 | 1.08 ± 0.04 |
| Leu | 388.61 ± 12.97 | 2.02 ± 0.07 |
| Phe | 233.10 ± 9.08 | 1.21 ± 0.05 |
| Lys | 453.21 ± 14.58 | 2.36 ± 0.08 |
| Essential amino acid (EAA) | 3017.23 ± 86.37 | 15.70 ± 0.45 |
| Total | 18,955.96 ± 315.20 | 100% |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Y.-Q.; Zeng, L.; Yang, Z.-S.; Huang, F.-F.; Ding, G.-F.; Wang, B. Anti-Fatigue Effect by Peptide Fraction from Protein Hydrolysate of Croceine Croaker (Pseudosciaena crocea) Swim Bladder through Inhibiting the Oxidative Reactions including DNA Damage. Mar. Drugs 2016, 14, 221. https://doi.org/10.3390/md14120221
Zhao Y-Q, Zeng L, Yang Z-S, Huang F-F, Ding G-F, Wang B. Anti-Fatigue Effect by Peptide Fraction from Protein Hydrolysate of Croceine Croaker (Pseudosciaena crocea) Swim Bladder through Inhibiting the Oxidative Reactions including DNA Damage. Marine Drugs. 2016; 14(12):221. https://doi.org/10.3390/md14120221
Chicago/Turabian StyleZhao, Yu-Qin, Li Zeng, Zui-Su Yang, Fang-Fang Huang, Guo-Fang Ding, and Bin Wang. 2016. "Anti-Fatigue Effect by Peptide Fraction from Protein Hydrolysate of Croceine Croaker (Pseudosciaena crocea) Swim Bladder through Inhibiting the Oxidative Reactions including DNA Damage" Marine Drugs 14, no. 12: 221. https://doi.org/10.3390/md14120221
APA StyleZhao, Y.-Q., Zeng, L., Yang, Z.-S., Huang, F.-F., Ding, G.-F., & Wang, B. (2016). Anti-Fatigue Effect by Peptide Fraction from Protein Hydrolysate of Croceine Croaker (Pseudosciaena crocea) Swim Bladder through Inhibiting the Oxidative Reactions including DNA Damage. Marine Drugs, 14(12), 221. https://doi.org/10.3390/md14120221







