Carotenoids from Haloarchaea and Their Potential in Biotechnology
Abstract
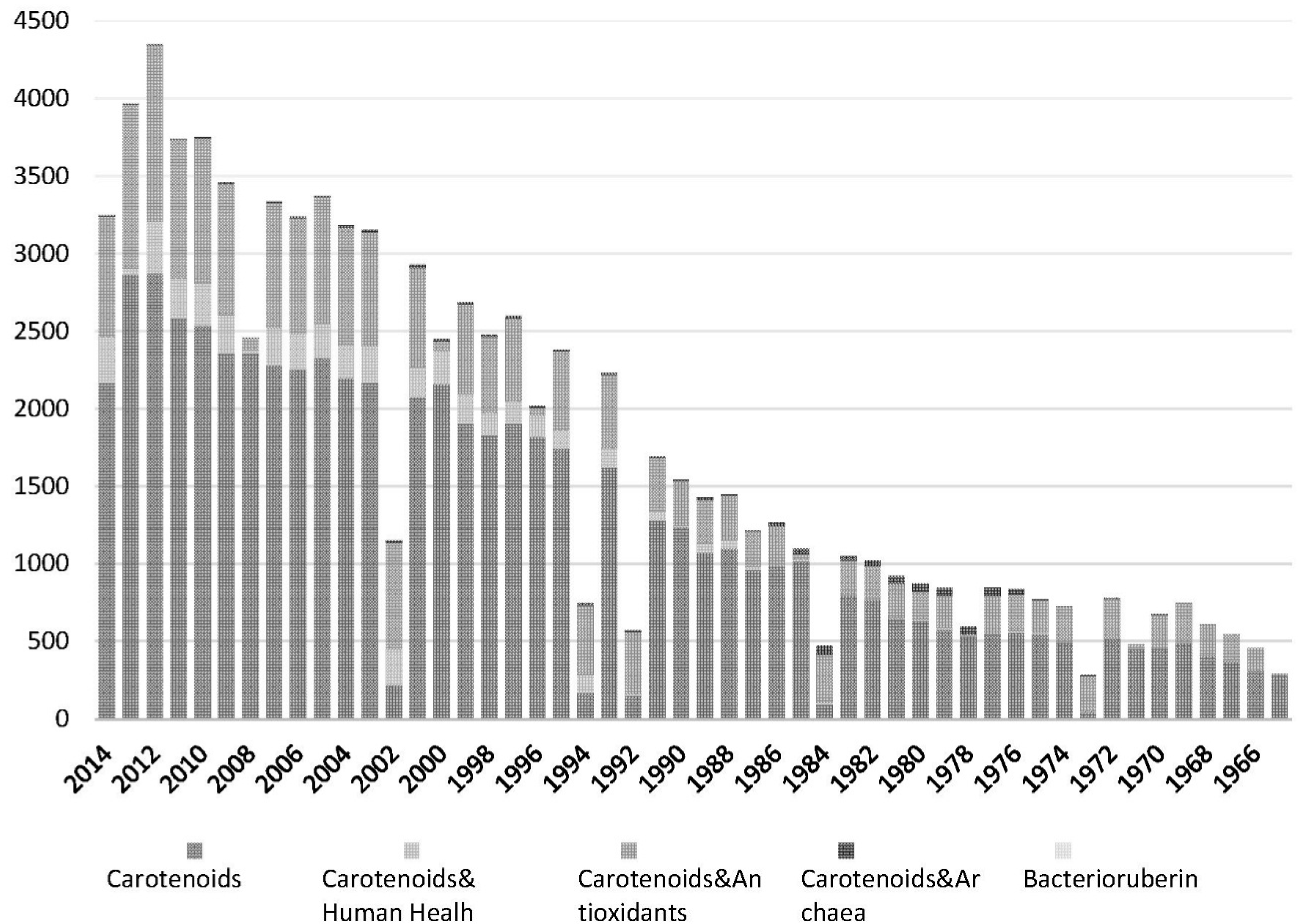
:1. Introduction
2. Carotenoids: Structure and Functionality
3. Carotenoids in the Context of Life
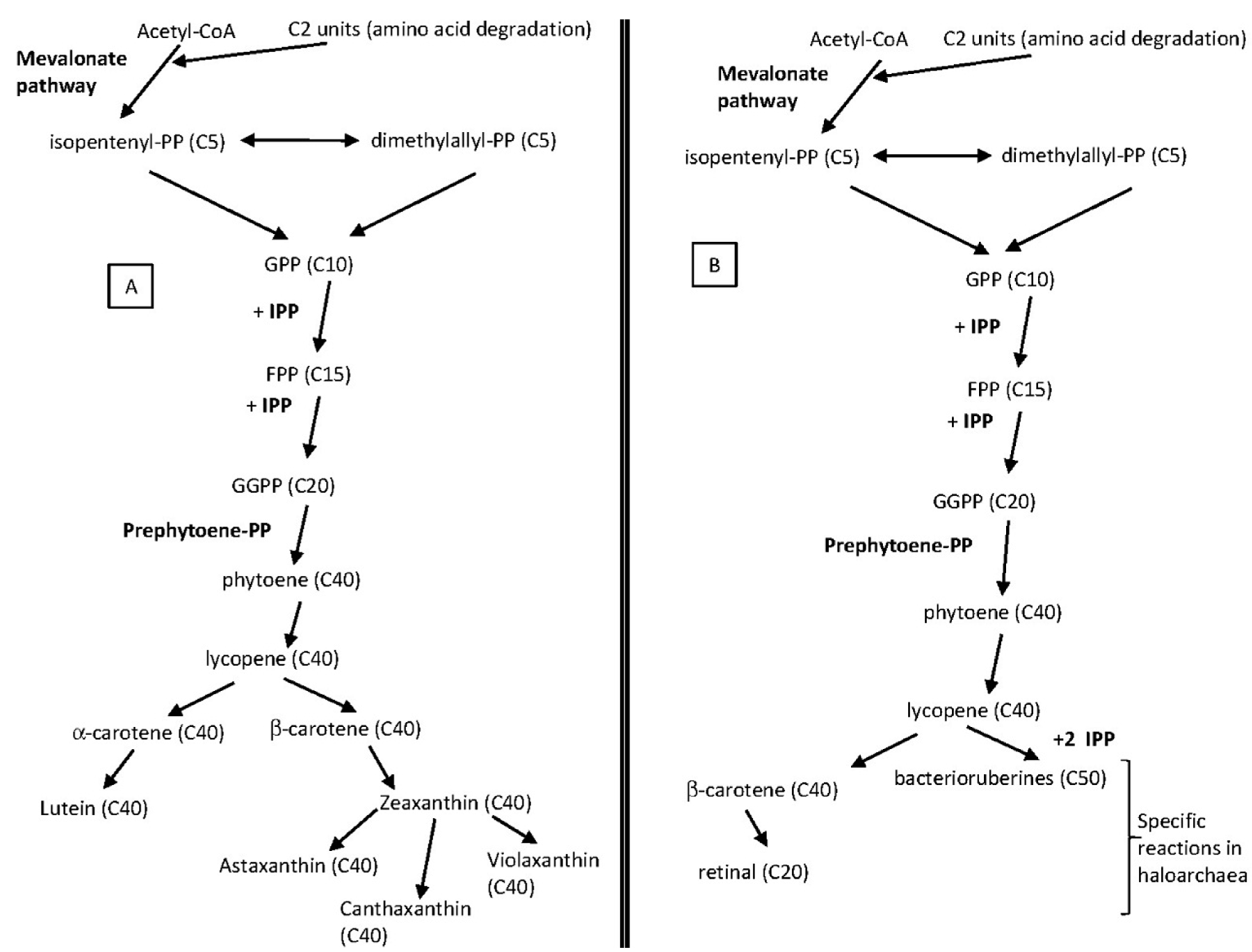
4. Carotenoids Metabolism
5. Production of Carotenoids by Haloarchaea
5.1. Type, Content and Biosynthesis of Haloarchaeal Carotenoids
5.2. Bacterioruberin Is One of the Major Carotenoids Produced by Haloarchaea
| Name | Chemical Structure |
|---|---|
| Bacterioruberin |  |
| Monoanhydrobacterioruberin |  |
| Bisanhydrobacterioruberin |  |
| Trisanhydrobacterioruberin |  |
| 2-isopentenyl-3,4-dehydrorhodopin |  |
| 5-cis-bacterioruberin |  |
| 9-cis-bacterioruberin |  |
| 13-cis-bacterioruberin |  |
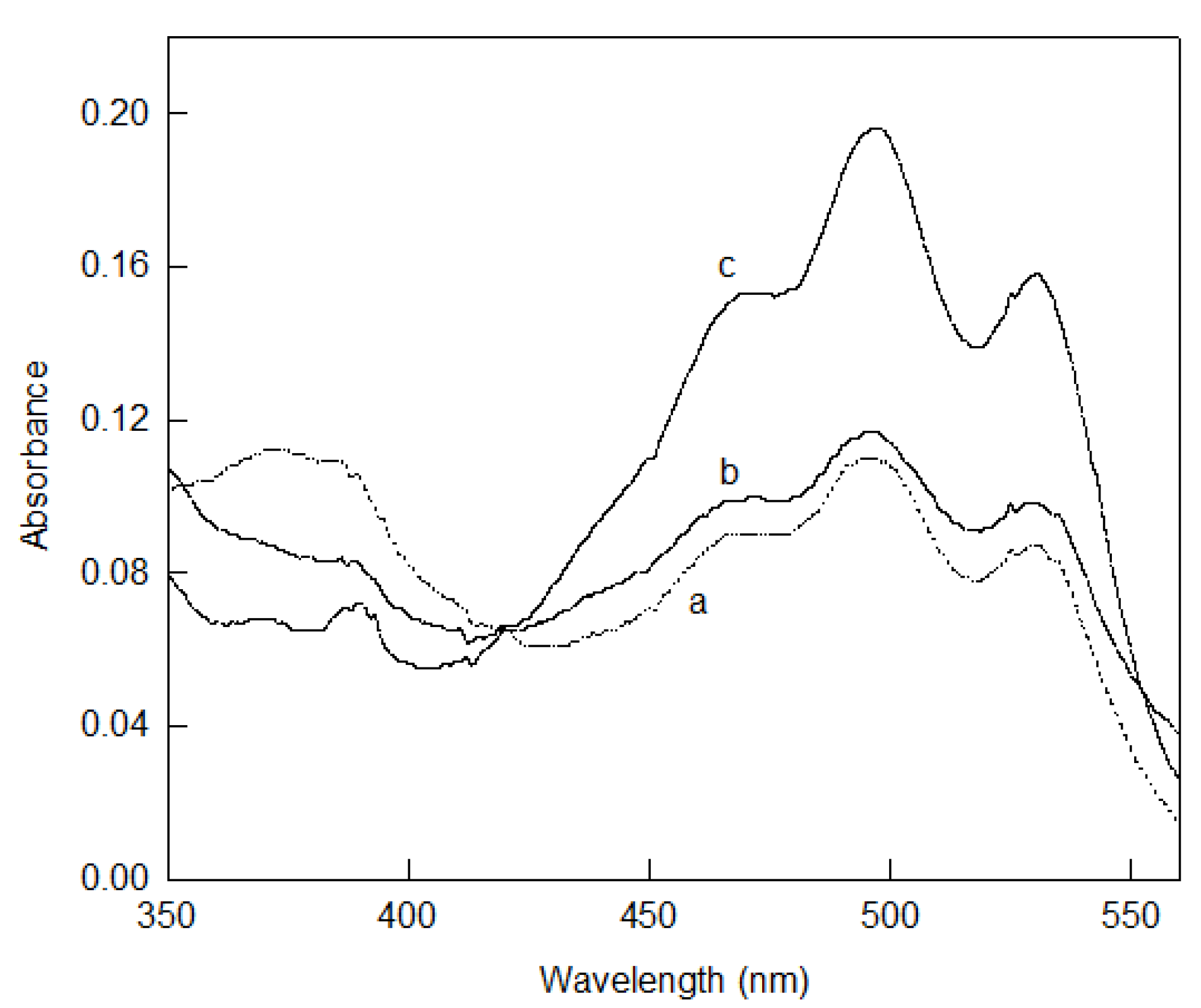
5.3. Bacterioruberin Biological Roles
6. Biotechnological Uses and Production Potentiality of Carotenoids from Haloarchaea
6.1. Biotechnological Uses of Carotenoids from Haloarchaea
| Publication Number | Publication Date | Title | International Application Number |
|---|---|---|---|
| WO/2009/042734 | 02.04.2009 | Radiation-resistant mutants of a halophilic archaeon and uses thereof | PCT/US2008/077596 |
| ES2324077 A1 | 29.07.2009 | Compuesto a base de membranas celulares liofilizadas | |
| US 7939220 B2 | 10.05.2011 | Proton-translocating retinal protein | PCT/EP2001/008715 |
| WO2011133907 A2 | 27.10.2011 | Methods to increase and harvest desired metabolite production in algae | PCT/US2011/033637 |
| WO2012169623 | 13.12.2012 | Method for producing carotenoid each having 50 carbon atoms | PCT/JP2012/064817 |
| WO2014045280 A1 | 27.03.2014 | Topical halobacteria extract composition for treating radiation skin tissue damage | PCT/IL2013/050786 |
| WO/2014/045279 | 27.03.2014 | Halobacteria extracts composition for tumour reduction | PCT/IL2013/050785 |
| US 20140356854 A1 | 4.10.2014 | Methods and compositions relating to mevalonate phosphate decarboxylase | |
| 07-132096 | 23.05.1995 | Production of C50 Carotenoid |
6.2. Production Potentiality of Carotenoids from Haloarchaea
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhang, J.; Sun, Z.; Sun, P.; Chen, T.; Chen, F. Microalgal carotenoids: Beneficial effects and potential in human health. Food Funct. 2014, 5, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef] [PubMed]
- Vílchez, C.; Forján, E.; Cuaresma, M.; Bédmar, F.; Garbayo, I.; Vega, J.M. Marine carotenoids: Biological functions and commercial applications. Mar. Drugs 2011, 9, 319–333. [Google Scholar] [CrossRef] [PubMed]
- Nisar, N.; Li, L.; Lu, S.; Khin, N.C.; Pogson, B.J. Carotenoid metabolism in plants. Mol. Plant 2015, 8, 68–82. [Google Scholar] [CrossRef] [PubMed]
- Yatsunami, R.; Ando, A.; Yang, Y.; Takaichi, S.; Kohno, M.; Matsumura, Y.; Ikeda, H.; Fukui, T.; Nakasone, K.; Fujita, N.; et al. Identification of carotenoids from the extremely halophilic archaeon Haloarcula japonica. Front. Microbiol. 2014, 5, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Mata-Gómez, L.C.; Montañez, J.C.; Méndez-Zavala, A.; Aguilar, C.N. Biotechnological production of carotenoids by yeasts: An overview. Microb. Cell Fact. 2014, 21, 12. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, T.W.; Britton, G. Distribution and analysis of carotenoids. In Plant Pigments; Goodwin, T.W., Ed.; Academic Press: London, UK, 1980; pp. 61–132. [Google Scholar]
- Cunningham, F.X.; Gantt, E. Genes and enzymes of carotenoid biosynthesis in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1998, 49, 557–583. [Google Scholar] [CrossRef] [PubMed]
- Blanco, A.M.; Moreno, J.; del Campo, J.A.; Rivas, J.; Guerrero, M.G. Outdoor cultivation of lutein-rich cells of Muriellopsis sp. in open ponds. Appl. Microbiol. Biotechnol. 2007, 73, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- Nelis, H.J.; de Leenheer, A.P. Microbial sources of carotenoid pigments used in foods and feeds. J. Appl. Bacteriol. 1991, 70, 181–191. [Google Scholar] [CrossRef]
- Bourgaud, F.; Gravot, A.; Milesi, S.; Gontier, E. Production of plant secondary metabolites: A historical perspective. Plant Sci. 2001, 161, 839–851. [Google Scholar] [CrossRef]
- Olaizola, M. Commercial development of microalgal biotechnology: From the test tube to the marketplace. Biomol. Eng. 2003, 20, 459–466. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Buschmann, C. Chlorophylls and Carotenoids: Measurement and Characterization by UV-VIS Spectroscopy. Curr. Protoc. Food Analyt.Chem. 2001, F:F4:F4.3. [Google Scholar] [CrossRef]
- Azevedo-Meleiro, C.H.; Rodriguez-Amaya, D.B. Confirmation of the identity of the carotenoids of tropical fruits by HPLC-DAD and HPLC-MS. J. Food Comp. Anal. 2004, 17, 385–396. [Google Scholar] [CrossRef]
- Jaime, L.; Mendiola, J.; Herrero, M.; Soler-Rivas, C.; Santoyo, S.; Señorans, F.J.; Cifuentes, A.; Ibañez, E. Separation and characterization of antioxidants from Spirulina platensis microalga combining pressurized liquid extraction, TLC, and HPLC-DAD. J. Sep. Sci. 2005, 28, 2111–2119. [Google Scholar] [CrossRef] [PubMed]
- Hengartner, U.; Bernhard, K.; Meyer, K.; Englert, G.; Glinz, E. Synthesis, isolation, and NMR-Spectroscopic characterization of Fourteen (Z)-Isomers of Lycopene and of come AcetylenicDidehydro- and Tetradehydrolycopenes. Helv. Chim. Acta 1992, 75, 1848–1865. [Google Scholar] [CrossRef]
- Britton, G. Structure and properties of carotenoids in relation to function. FASEB J. 1995, 9, 1551–1558. [Google Scholar] [PubMed]
- Meléndez-Martínez, A.J.; Britton, G.; Vicario, I.M.; Heredia, F.J. Relationship between the colour and the chemical structure of carotenoid pigments. Food Chem. 2007, 101, 1145–1150. [Google Scholar] [CrossRef]
- De Lourdes Moreno, M.; Sánchez-Porro, C.; García, M.T.; Mellado, E. Carotenoids’ production from halophilic bacteria. Methods Mol. Biol. 2012, 892, 207–217. [Google Scholar] [PubMed]
- Oren, A. A hundred years of Dunaliella research: 1905–2005. Saline Syst. 2005, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Oren, A. The ecology of Dunaliella in high-salt environments. J. Biol. Res. 2014, 21, 23. [Google Scholar] [CrossRef] [PubMed]
- Hosseini Tafreshi, A.; Shariati, M. Dunaliella biotechnology: Methods and applications. J. Appl. Microbiol. 2009, 107, 14–35. [Google Scholar] [CrossRef] [PubMed]
- Lamers, P.P.; Janssen, M.; de Vos, R.C.H.; Bino, R.J.; Wijffels, R.H. Exploring and exploiting carotenoid accumulation in Dunaliella salina for cell-factory applications. Trends Biotechnol. 2008, 26, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Asker, D.; Ohta, Y. Production of Canthaxanthin by Extremely Halophilic Bacteria. J. Biosci. Bioeng. 1999, 88, 617–621. [Google Scholar] [CrossRef]
- Asker, D.; Awad, T.; Ohta, T. Lipids of Haloferax alexandrinus Strain TMT. An Extremely Halophilic Canthaxanthin-Producing Archaeon. J. Biosci. Bioeng. 2002, 93, 37–43. [Google Scholar] [CrossRef]
- Ronnekleiv, M.; Liaaen-Jensen, S. Bacterial Carotenoids 53*, C50-Carotenoids 23; Carotenoids of Haloferax volcanii versus other Halophilic Bacteria. Biochem. Syst. Ecol. 1995, 23, 627–734. [Google Scholar] [CrossRef]
- Gupta, R.S.; Naushad, S.; Baker, S. Phylogenomic analyses and molecular signatures for the class Halobacteria and its two major clades: A proposal for division of the class Halobacteria into an emended order Halobacteriales and two new orders, Haloferacales ord nov and Natrialbales ord. nov., containing the novel families Haloferacaceae fam. nov. and Natrialbaceae fam. nov. Int. J. Syst. Evol. Microbiol. 2015, 65, 1050–1069. [Google Scholar] [PubMed]
- Oren, A. Life at high salt concentrations, intracellular KCl concentrations, and acidic proteomes. Front. Microbiol. 2013, 5, 315. [Google Scholar] [CrossRef] [PubMed]
- Oren, A. Industrial and environmental applications of halophilic microorganisms. Environ. Technol. 2010, 31, 825–834. [Google Scholar] [CrossRef] [PubMed]
- Oren, A. Halophilic archaea on Earth and in space: Growth and survival under extreme conditions. Philos. Trans. A Math. Phys. Eng. Sci. 2014, 13, 372. [Google Scholar] [CrossRef] [PubMed]
- Schwieter, U.; Rüegg, R.; Isler, O. Syntheses in the carotenoid series. 21. Synthesis of 2,2′-diketo-spirilloxanthin (P 518) and 2,2′-diketo-bacterioruberin. Helv. Chim. Acta 1966, 49, 992–996. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.; Jensen, S.L. Bacterial carotenoids. XXVI. C50-carotenoids. 2. Bacterioruberin. Acta Chem. Scand. 1967, 21, 2578–2580. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.V.; Rao, L.G. Carotenoids and human health. Pharmacol. Res. 2007, 55, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Del Campo, J.A.; García-González, M.; Guerrero, M.G. Outdoor cultivation of microalgae for carotenoid production: Current state and perspectives. Appl. Microbiol. Biotechnol. 2007, 74, 1163–1174. [Google Scholar] [CrossRef] [PubMed]
- Rivera, S.M.; Canela-Garayoa, R. Analytical tools for the analysis of carotenoids in diverse materials. J. Chromatogr. A 2012, 1224. [Google Scholar] [CrossRef] [PubMed]
- Fassett, R.G.; Coombes, J.S. Astaxanthin in cardiovascular health and disease. Molecules 2012, 17, 2030–2048. [Google Scholar] [CrossRef] [PubMed]
- Jehlička, J.; Oren, A. Raman spectroscopy in halophile research. Front. Microbiol. 2013, 10, 380. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Shnimizu, M.; Moriwaki, H. Cancer chemoprevention by carotenoids. Molecules 2012, 17, 3202–3242. [Google Scholar] [CrossRef] [PubMed]
- Higuera-Ciapara, I.; Félix-Valenzuela, L.; Goycoolea, F.M. Astaxanthin: A review of its chemistry and applications. Crit. Rev. Food Sci. Nutr. 2006, 46, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Ambati, R.R.; Phang, S.M.; Ravi, S.; Aswathanarayana, R.G. Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications—A review. Mar. Drugs 2014, 12, 128–152. [Google Scholar] [CrossRef] [PubMed]
- Stutz, H.; Bresgen, N.; Eckl, P.M. Analytical tools for the analysis of β-carotene and its degradation products. Free Radic. Res. 2015, 49, 650–680. [Google Scholar] [CrossRef] [PubMed]
- Englert, M.; Hammann, S.; Vetter, W. Isolation of β-carotene, α-carotene and lutein from carrots by countercurrent chromatography with the solvent system modifier benzotrifluoride. J. Chromatogr. A 2015, 1388, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, S.; Man, Y.; Li, N.; Zhou, Y.U. Effects of vitamins E and C combined with β-carotene on cognitive function in the elderly. Exp. Ther. Med. 2015, 9, 1489–1493. [Google Scholar] [CrossRef] [PubMed]
- Relevy, N.Z.; Harats, D.; Harari, A.; Ben-Amotz, A.; Bitzur, R.; Rühl, R.; Shaish, A. Vitamin A-Deficient Diet Accelerated Atherogenesis in Apolipoprotein E(−/−) Mice and Dietary β-Carotene Prevents This Consequence. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Makita, H.; Ohnishi, M.; Mori, H.; Satoh, K.; Hara, A. Chemoprevention of rat oral carcinogenesis by naturally occurring xanthophylls, astaxanthin and canthaxanthin. Cancer Res. 1995, 55, 4059–4064. [Google Scholar] [PubMed]
- Surai, P.F. The antioxidant properties of canthaxanthin and its potential effects in the poultry eggs and on embryonic development of the chick, Part 1. World Poult. Sci. J. 2012, 68, 465–476. [Google Scholar] [CrossRef]
- Rostami, F.; Razavi, S.H.; Sepahi, A.A.; Gharibzahedi, S.M. Canthaxanthin biosynthesis by Dietzia natronolimnaea HS-1: Effects of inoculation and aeration rate. Braz. J. Microbiol. 2014, 45, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Hojjati, M.; Razavi, S.H.; Rezaei, K.; Gilani, K. Stabilization of canthaxanthin produced by Dietzia natronolimnaea HS-1 with spray drying microencapsulation. J. Food Sci. Technol. 2014, 51, 2134–2140. [Google Scholar] [CrossRef] [PubMed]
- Heying, E.K.; Tanumihardjo, J.P.; Vasic, V.; Cook, M.; Palacios-Rojas, N.; Tanumihardjo, S.A. Biofortified orange maize enhances β-cryptoxanthin concentrations in egg yolks of laying hens better than tangerine peel fortificant. J. Agric. Food Chem. 2014, 62, 11892–11900. [Google Scholar] [CrossRef] [PubMed]
- Burri, B.J. β-Cryptoxanthin as a source of vitamin A. J. Sci. Food Agric. 2015, 95, 1786–1794. [Google Scholar] [CrossRef] [PubMed]
- Granado-Lorencio, F.; de Las Heras, L.; Millán, C.S.; Garcia-López, F.J.; Blanco-Navarro, I.; Pérez-Sacristán, B.; Domínguez, G. β-Cryptoxanthin modulates the response to phytosterols in post-menopausal women carrying NPC1L1 L272L and ABCG8 A632 V polymorphisms: An exploratory study. Genes Nutr. 2014, 9, 428. [Google Scholar] [CrossRef] [PubMed]
- Chisté, R.C.; Freitas, M.; Mercadante, A.Z.; Fernandes, E. Carotenoids are effective inhibitors of in vitro hemolysis of human erythrocytes, as determined by a practical and optimized cellular antioxidant assay. J. Food Sci. 2014, 79, H1841–H1877. [Google Scholar] [CrossRef] [PubMed]
- Ghodratizadeh, S.; Kanbak, G.; Beyramzadeh, M.; Dikmen, Z.G.; Memarzadeh, S.; Habibian, R. Effect of carotenoid β-cryptoxanthin on cellular and humoral immune response in rabbit. Vet. Res. Commun. 2014, 38, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xiao, Y.; Zhang, Z.; Liu, C. Light-induced oxidation and isomerization of all-trans-β-cryptoxanthin in a model system. J. Photochem. Photobiol. B Biol. 2015, 142, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Riccioni, G.; D’Orazio, N.; Franceschelli, S.; Speranza, L. Marine carotenoids and cardiovascular risk markers. Mar. Drugs. 2011, 9, 1166–1175. [Google Scholar] [CrossRef] [PubMed]
- Igielska-Kalwat, J.; Gościańska, J.; Nowak, I. Carotenoids as natural antioxidants. Postepy Hig. Med. Dosw. 2015, 69, 418–428. [Google Scholar] [CrossRef]
- Pirayesh Islamian, J.; Mehrali, H. Lycopene as a carotenoid provides radioprotectant and antioxidant effects by quenching radiation-induced free radical singlet oxygen: An overview. Cell J. 2015, 16, 386–391. [Google Scholar] [PubMed]
- Naziri, D.; Hamidi, M.; Hassanzadeh, S.; Tarhriz, V.; Maleki Zanjani, B.; Nazemyieh, H.; Hejazi, M.A.; Hejazi, M.S. Analysis of Carotenoid Production by Halorubrum. sp. TBZ126: An Extremely Halophilic Archeon from Urmia Lake. Adv. Pharm. Bull. 2014, 4, 61–67. [Google Scholar] [PubMed]
- Flaks, B.; Bresloff, P. Some observations on the fine structure of the lutein cells of X-irradiated rat ovary. J. Cell Biol. 1966, 30, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Altemimi, A.; Lightfoot, D.A.; Kinsel, M.; Watson, D.G. Employing Response Surface Methodology for the Optimization of Ultrasound Assisted Extraction of Lutein and β-Carotene from Spinach. Molecules 2015, 20, 6611–6625. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.M.; Dou, H.L.; Huang, F.F.; Xu, X.R.; Zou, Z.Y.; Lin, X.M. Effect of supplemental lutein and zeaxanthin on serum, macular pigmentation, and visual performance in patients with early age-related macular degeneration. Biomed. Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Costa, S.; Giannantonio, C.; Romagnoli, C.; Barone, G.; Gervasoni, J.; Perri, A.; Zecca, E. Lutein and zeaxanthin concentrations in formula and human milk samples from Italian mothers. Eur. J. Clin. Nutr. 2015, 69, 531–532. [Google Scholar] [CrossRef] [PubMed]
- Li, X.R.; Tian, G.Q.; Shen, H.J.; Liu, J.Z. Metabolic engineering of Escherichia coli to produce zeaxanthin. J. Ind. Microbiol. Biotechnol. 2015, 42, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.Y.; Chang, J.L.; Aihara, M.S. Light-induced interconversion of violaxanthin and zeaxanthin in New Zealand spinach-leaf segments. Biochim. Biophys. Acta 1967, 141, 342–347. [Google Scholar] [CrossRef]
- Yamamoto, H.Y.; Kamite, L.; Wang, Y.Y. An Ascorbate-induced Absorbance Change in Chloroplasts from Violaxanthin De-epoxidation. Plant Physiol. 1972, 49, 224–228. [Google Scholar] [CrossRef] [PubMed]
- Sapozhnikov, D.I. Investigation on the violaxanthin cycle. Pure Appl. Chem. 1973, 35, 47–61. [Google Scholar] [CrossRef] [PubMed]
- Soontornchaiboon, W.; Joo, S.S.; Kim, S.M. Anti-inflammatory effects of violaxanthin isolated from microalga Chlorella ellipsoidea in RAW 264.7 macrophages. Biol. Pharm Bull. 2012, 35, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Hallin, E.I.; Guo, K.; Åkerlund, H.E. Violaxanthin de-epoxidase disulphides and their role in activity and thermal stability. Photosynth. Res. 2015, 124, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Burton, G.W.; Foster, D.O.; Perly, B.; Slater, T.F.; Smith, I.C.; Ingold, K.U. Biological antioxidants. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1985, 311, 565–578. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.A.; Riccioni, G.; D’Orazio, N. Carotenoids: Potential allies of cardiovascular health? Food Nutr. Res. 2015, 59. [Google Scholar] [CrossRef] [PubMed]
- LaFountain, A.M.; Prum, R.O.; Frank, H.A. Diversity, physiology, and evolution of avian plumage carotenoids and the role of carotenoid-protein interactions in plumage colour appearance. Arch. Biochem. Biophys. 2015, 572, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Namitha, K.K.; Negi, P.S. Chemistry and biotechnology of carotenoids. Crit. Rev. Food Sci. Nutr. 2010, 50, 728–760. [Google Scholar] [CrossRef] [PubMed]
- Zile, M.H. Vitamin A and embryonic development: An overview. J. Nutr. 1998, 128, 455S–458S. [Google Scholar] [PubMed]
- Kaulmann, A.; Bohn, T. Carotenoids, inflammation, and oxidative stress—Implications of cellular signaling pathways and relation to chronic disease prevention. Nutr. Res. 2014, 34, 907–929. [Google Scholar] [CrossRef] [PubMed]
- Gupta, C.; Prakash, D. Phytonutrients as therapeutic agents. J. Complement. Integr. Med. 2014, 11, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Ascenso, A.; Ribeiro, H.; Marques, H.C.; Oliveira, H.; Santos, C.; Simões, S. Chemoprevention of photocarcinogenesis by lycopene. Exp. Dermatol. 2014, 23, 874–878. [Google Scholar] [CrossRef] [PubMed]
- Amundsen, C.R.; Nordeide, J.T.; Gjøen, H.M.; Larsen, B.; Egeland, E.S. Conspicuous carotenoid-based pelvic spine ornament in three-spined stickleback populations—Occurrence and inheritance. Peer J. 2015, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jehlicka, J.; Edwards, H.G.; Oren, A. Bacterioruberin and salinixanthin carotenoids of extremely halophilic Archaea and Bacteria: A Raman spectroscopic study. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2013, 106, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Mandelli, F.; Miranda, V.S.; Rodrigues, E.; Mercadante, A.Z. Identification of carotenoids with high antioxidant capacity produced by extremophile microorganisms. World J. Microbiol. Biotechnol. 2012, 28, 1781–1790. [Google Scholar] [CrossRef] [PubMed]
- Othman, R.; Mohd Zaifuddin, F.A.; Hassan, N.M. Carotenoid biosynthesis regulatory mechanisms in plants. J. Oleo Sci. 2014, 63, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Palczewski, G.; Amengual, J.; Hoppel, C.L.; von Lintig, J. Evidence for compartmentalization of mammalian carotenoid metabolism. FASEB J. 2014, 28, 4457–4469. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, G. Plant carotenoids: Genomics meets multi-gene engineering. Curr. Opin. Plant Biol. 2014, 19, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Parker, R.S. Absorption, metabolism, and transport of carotenoids. FASEB J. 1996, 10, 542–551. [Google Scholar] [PubMed]
- Reboul, E.; Borel, P. Proteins involved in uptake, intracellular transport and basolateral secretion of fat-soluble vitamins and carotenoids by mammalian enterocytes. Prog. Lipid Res. 2011, 50, 388–402. [Google Scholar] [CrossRef] [PubMed]
- Desmarais, D.; Jablonski, P.E.; Fedarko, N.S.; Roberts, M.F. 2-Sulfotrehalose, a novel osmolyte in haloalkaliphilic archaea. J. Bacteriol. 1997, 179, 3146–3153. [Google Scholar] [PubMed]
- Madern, D.; Camacho, M.; Rodríguez-Arnedo, A.; Bonete, M.J.; Zaccai, G. Salt-dependent studies of NADP-dependent isocitrate dehydrogenase from the halophilic archaeon Haloferax volcanii. Extremophiles 2004, 8, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Bonete, M.J.; Martínez-Espinosa, R.M. Enzymes from Halophilic Archaea: Open Questions. In Halophiles and Hypersaline Environments: Current Research and Future Trends; Ventosa, A., Oren, A., Eds.; Springer-Verlag GmbH: Berlin, Germany, 2011; pp. 358–370. [Google Scholar]
- Kushwaha, S.C.; Kramer, J.K.; Kates, M. Isolation and characterization of C50-carotenoid pigments and other polar isoprenoids from Halobacterium cutirubrum. Biochim. Biophys. Acta 1975, 398, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Bidle, K.A.; Hanson, T.E.; Howell, K.; Nannen, J. HMG-CoA reductase is regulated by salinity at the level of transcription in Haloferax volcanii. Extremophiles 2007, 11, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Oren, A.; Gurevich, P. Dynamics of a bloom of halophilic archaea in the Dead Sea. Hydrobiologia 1995, 315, 149–158. [Google Scholar] [CrossRef]
- Asker, D.; Awad, T.; Ohta, Y. Lipids of Haloferax. alexandrinus strain TMT: An extremely halophilic canthaxanthin-producing archaeon. J. Biosci. Bioeng. 2002, 93, 37–43. [Google Scholar] [CrossRef]
- Marshall, C.P.; Leuko, S.; Coyle, C.M.; Walter, M.R.; Burns, B.P.; Neilan, B.A. Carotenoid analysis of halophilic archaea by resonance Raman spectroscopy. Astrobiology 2007, 7, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Jehlička, J.; Edwards, H.G.; Oren, A. Raman spectroscopy of microbial pigments. Appl. Environ. Microbiol. 2014, 80, 3286–3295. [Google Scholar] [CrossRef] [PubMed]
- Lobasso, S.; Lopalco, P.; Mascolo, G.; Corcelli, A. Lipids of the ultra-thin square halophilic archaeon Haloquadratum walsbyi. Archaea 2008, 2, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, S.C.; Kates, M.; Porter, J.W. Enzymatic synthesis of C40 carotenes by cell-free preparation from Halobacterium cutirubrum. Can. J. Biochem. 1976, 54, 816–823. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, S.C.; Kates, M. Effect of nicotine on biosynthesis of C50 carotenoids in Halobacterium cutirubrum. Can. J. Biochem. 1976, 54, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, S.C.; Kates, M. Effect of glycerol on carotenogenesis in the extreme halophile, Halobacterium cutirubrum. Can. J. Microbiol. 1979, 25, 1288–1291. [Google Scholar] [CrossRef] [PubMed]
- Peck, R.F.; Echavarri-Erasun, C.; Johnson, E.A.; Ng, W.V.; Kennedy, S.P.; Hood, L.; DasSarma, S.; Krebs, M.P. brp and blh are required for synthesis of the retinal cofactor of bacteriorhodopsin in Halobacterium salinarum. J. Biol. Chem. 2001, 276, 5739–5744. [Google Scholar] [CrossRef] [PubMed]
- Dassarma, S.; Kennedy, S.P.; Berquist, B.; Victor, N.W.; Baliga, N.S.; Spudich, J.L.; Krebs, M.P.; Eisen, J.A.; Johnson, C.H.; Hood, L. Genomic perspective on the photobiology of Halobacterium species NRC-1, a phototrophic, phototactic, and UV-tolerant haloarchaeon. Photosynth. Res. 2001, 70, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Falb, M.; Müller, K.; Königsmaier, L.; Oberwinkler, T.; Horn, P.; von Gronau, S.; Gonzalez, O.; Pfeiffer, F.; Bornberg-Bauer, E.; Oesterhelt, D. Metabolism of halophilic archaea. Extremophiles 2008, 12, 177–196. [Google Scholar] [CrossRef] [PubMed]
- Oesterhelt, D. Bacteriorhodopsin as an example of a light-driven proton pump. Angew. Chem. Int. Ed. Engl. 1976, 15, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Sumper, M.; Reitmeier, H.; Oesterhelt, D. Biosynthesis of the purple membrane of halobacteria. Angew. Chem. Int. Ed. Engl. 1976, 15, 187–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.; Yatsunami, R.; Ando, A.; Miyoko, N.; Fukui, T.; Takaichi, S.; Nakamura, S. Complete Biosynthetic Pathway of the C50 Carotenoid Bacterioruberin from Lycopene in the extremely halophilic archaeon Haloarcula japonica. J. Bacteriol. 2015, 197, 1614–1623. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo-Baños, M.; Garbayo, I.; Vilchez, C.; Bonete, M.J.; Martínez.Espinosa, R.M. Genomic analysis of the biosynthesis of isoprenoids in Haloferax genus, to be submitted for publication.
- Fang, C.J.; Ku, K.L.; Lee, M.H.; Su, N.W. Influence of nutritive factors on C50 carotenoids production by Haloferax mediterranei ATCC 33500 with two-stage cultivation. Bioresour. Technol. 2010, 101, 6487–6493. [Google Scholar] [CrossRef] [PubMed]
- Dundas, I.D.; Larsen, H. A study on the killing by light of photosensitized cells of Halobacterium salinarium. Arch. Mikrobiol. 1963, 46, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Shahmohammadi, H.R.; Asgarani, E.; Terato, H.; Saito, T.; Ohyama, Y.; Gekko, K.; Yamamoto, O.; Ide, H. Protective roles of bacterioruberin and intracellular KCl in the resistance of Halobacterium salinarium against DNA-damaging agents. J. Radiat. Res. 1998, 39, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.; Norgard, S.; Liaaen-Jensen, S. Bacterial carotenoids. 31. C50-carotenoids 5. Carotenoids of Halobacterium. salinarium, especially bacterioruberin. Acta Chem. Scand. 1970, 24, 2169–2182. [Google Scholar] [CrossRef] [PubMed]
- Becher, B.M.; Cassim, J.Y. Improved isolation procedures for the purple membrane of Halobacterium halobium. Prep. Biochem. 1975, 5, 161–178. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, S.C.; Kates, M. Studies of the biosynthesis of C50 carotenoids in Halobacterium cutirubrum. Can. J. Microbiol. 1979, 25, 1292–1297. [Google Scholar] [CrossRef] [PubMed]
- Shand, R.F.; Betlach, M.C. Expression of the bop gene cluster of Halobacterium halobium is induced by low oxygen tension and by light. J. Bacteriol. 1991, 173, 4692–4699. [Google Scholar] [PubMed]
- El-Sayed, W.S.; Takaichi, S.; Saida, H.; Kamekura, M.; Abu-Shady, M.; Seki, H.; Kuwabara, T. Effects of light and low oxygen tension on pigment biosynthesis in Halobacterium salinarum, revealed by a novel method to quantify both retinal and carotenoids. Plant Cell Physiol. 2002, 43, 379–383. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, S.E.; Altekar, W.; D’Souza, S.F. Adaptive response of Haloferax mediterranei to low concentrations of NaCl (<20%) in the growth medium. Arch. Microbiol. 1997, 168, 68–71. [Google Scholar]
- Raghavan, T.M.; Furtado, I. Expression of carotenoid pigments of haloarchaeal cultures exposed to aniline. Environ. Toxicol. 2005, 20, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, T.; Furtado, I. Occurrence of extremely halophilic Archaea in sediments from the continental shelf of west coast of India. Curr. Sci. 2004, 86, 1065–1067. [Google Scholar]
- Hamidi, M.; Abdin, M.Z.; Nazemyieh, H.; Hejazi, M.A.; Hejazi, M.S. Optimization of Total Carotenoid Production by Halorubrum sp. TBZ126 using response surface methodology. J. Microb. Biochem. Technol. 2014, 6, 286–294. [Google Scholar] [CrossRef]
- Fendrihan, S.; Musso, M.; Stan-Lotter, H. Raman spectroscopy as a potential method for the detection of extremely halophilic archaea embedded in halite in terrestrial and possibly extraterrestrial samples. J. Raman Spectrosc. 2009, 40, 1996–2003. [Google Scholar] [CrossRef] [PubMed]
- Miller, N.J.; Sampson, J.; Candeias, L.P.; Bramley, P.M.; Rice-Evans, C.A. Antioxidant activities of carotenes and xanthophylls. FEBS Lett. 1996, 384, 240–242. [Google Scholar] [CrossRef]
- Albrecht, M.; Takaichi, S.; Steiger, S.; Wang, Z.Y.; Sandmann, G. Novel hydroxycarotenoids with improved antioxidative properties produced by gene combination in Escherichia coli. Nat. Biotechnol. 2000, 18, 843–846. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Xu, Z.; Sun, Z.; Lin, J.; Hua, Y. Evaluation of the antioxidant effects of carotenoids from Deinococcus radiodurans through targeted mutagenesis, chemiluminescence, and DNA damage analyses. Biochim. Biophys. Acta 2007, 1770, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Miyabe, Y.; Ide, H.; Yamamoto, O. Hydroxyl radical scavenging ability of bacterioruberin. Radiat. Phys. Chem. 1997, 50, 267–269. [Google Scholar] [CrossRef]
- Kottemann, M.; Kish, A.; Iloanusi, C.; Bjork, S.; DiRuggiero, J. Physiological responses of the halophilic archaeon Halobacterium sp. strain NRC1 to desiccation and gamma irradiation. Extremophiles 2005, 9, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Lazrk, T.; Wolff, G.; Albrecht, A.M.; Nakatani, Y.; Ourisson, G.; Kates, M. Bacterioruberins reinforce reconstituted halobacterium lipid-membranes. Biochim. Biophys. Acta 1988, 939, 160–162. [Google Scholar] [CrossRef]
- Cao, Z.; Ding, X.; Peng, B.; Zhao, Y.; Ding, J.; Watts, A.; Zhao, X. Novel expression and characterization of a light driven proton pump archaerhodopsin-4 in a Halobacterium salinarum strain. Biochim. Biophys. Acta 2015, 1847, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Liu, H.C.; Chu, J.F.; Zhou, P.J.; Tang, J.A.; Liu, S.J. Genetic cloning and functional expression in Escherichia coli of an archaerhodopsin gene from Halorubrum xinjiangense. Extremophiles 2006, 10, 29–33. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Sun, Q.; Zhao, W.; Wang, H.; Xu, D. Newly isolated archaerhodopsin from a strain of Chinese halobacteria and its proton pumping behavior. Biochim. Biophys. Acta 2000, 1466, 260–266. [Google Scholar] [CrossRef]
- Yoshimura, K.; Kouyama, T. Structural role of bacterioruberin in the trimeric structure of archaerhodopsin-2. J. Mol. Biol. 2008, 375, 1267–1281. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Razak, N.W.; Kato, N.; Mukai, Y. Characteristics of halorhodopsin-bacterioruberin complex from Natronomonas pharaonis membrane in the solubilized system. Biochemistry 2012, 51, 2785–2794. [Google Scholar] [CrossRef] [PubMed]
- Google (Key words: caroten and haloarchaea). Available online: https://www.google.es/?tbm=pts&gws_rd=cr,ssl&ei=md8wVcesMYyCPeKdgdgC#tbm=pts&q=caroten+%26+haloarchaea+patents (accessed on 13 April 2015).
- Pantentscope (Key words: halobacteria, carotenoids and haloarchaea). Available online: https://patentscope.wipo.int/search/en/result.jsf (accessed on 14 April 2015).
- Oficina Española de patentes y marcas - Invenciones. Available online: http://www.oepm.es/es/invenciones/resultados.html?field=TITU_RESU&bases=0&keyword=carotenoid (accessed on 15 April 2015).
- World Intellectual Property Organization Global Brand Database. Available online: http://www.wipo.int/branddb/en (accessed on 21 April 2015).
- Japan Platform for Patent information website. Available online: https://www.j-platpat.inpit.go.jp/web/all/top/BTmTopEnglishPage (accessed on 22 April 2015).
- Espacenet Patent search. Available online: http://worldwide.espacenet.com/?locale=en_EP (accessed on 23 April 2015).
- European patent register. Available online: https://register.epo.org/regviewer (accessed on 23 April 2015).
- Markets and markets website—New market reports. Available online: http://www.marketsandmarkets.com/search.asp?Search=carotenoid&x=0&y=0 (accessed on 24 April 2015).
- Yachai, M. Carotenoid Production by Halophilic Archaea and Its Applications. Ph.D. Thesis, Prince of Songkla University, Songkhla, Thailand, 2009. [Google Scholar]
- Varela, J.C.; Pereira, H.; Vila, M.; León, R. Production of carotenoids by microalgae: Achievements and challenges. Photosynth. Res. 2015, 125. [Google Scholar] [CrossRef] [PubMed]
- Norsker, N.; Barbosa, M.; Vermue, M.; Wijffels, R. Microalgal production: A close look at the economics. Biotechnol. Adv. 2001, 29, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Wichuk, K.; Brynjolfsson, S.; Fu, W. Biotechnological production of value-added carotenoids from microalgae: Emerging technology and prospects. Bioengineered 2014, 5, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Zhu, J.C.; Liu, Y.; Yang, Y.; Zhu, J.Y.; Huang, Y.P.; Shen, P. Investigation of the influence of NaCl concentration on Halobacterium salinarum growth. J. Therm. Anal. Calorim. 2006, 84, 625–630. [Google Scholar] [CrossRef]
- Mata, T.; Martins, A.; Caetano, N. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef]
- Abbes, M.; Baati, H.; Guermazi, S.; Messina, C.; Santulli, A.; Gharsallah, N.; Ammar, E. Biological properties of carotenoids extracted from Halobacterium halobium isolated from a Tunisian solar saltern. BMC Complement. Altern. Med. 2013, 13, 255. [Google Scholar] [CrossRef] [PubMed]
- Sikkandar, S.; Murugan, K.; Al-Sohaibani, S.; Rayappan, F.; Nair, A.; Tilton, F. Halophilic bacteria-A potent source of carotenoids with antioxidant and anticancer potentials. J. Pure Appl. Microbiol. 2013, 7, 2825–2830. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigo-Baños, M.; Garbayo, I.; Vílchez, C.; Bonete, M.J.; Martínez-Espinosa, R.M. Carotenoids from Haloarchaea and Their Potential in Biotechnology. Mar. Drugs 2015, 13, 5508-5532. https://doi.org/10.3390/md13095508
Rodrigo-Baños M, Garbayo I, Vílchez C, Bonete MJ, Martínez-Espinosa RM. Carotenoids from Haloarchaea and Their Potential in Biotechnology. Marine Drugs. 2015; 13(9):5508-5532. https://doi.org/10.3390/md13095508
Chicago/Turabian StyleRodrigo-Baños, Montserrat, Inés Garbayo, Carlos Vílchez, María José Bonete, and Rosa María Martínez-Espinosa. 2015. "Carotenoids from Haloarchaea and Their Potential in Biotechnology" Marine Drugs 13, no. 9: 5508-5532. https://doi.org/10.3390/md13095508
APA StyleRodrigo-Baños, M., Garbayo, I., Vílchez, C., Bonete, M. J., & Martínez-Espinosa, R. M. (2015). Carotenoids from Haloarchaea and Their Potential in Biotechnology. Marine Drugs, 13(9), 5508-5532. https://doi.org/10.3390/md13095508









