Two Novel Antioxidant Nonapeptides from Protein Hydrolysate of Skate (Raja porosa) Muscle
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of Protein Hydrolysates from Skate Muscle
| Enzymes | Enzymolysis Conditions | DH (%) | EC50 (mg/mL) | ||
|---|---|---|---|---|---|
| pH | Temp. (°C) | Buffer Solution | |||
| Papain | 6.5 | 50 | 0.05 M phosphate | 12.57 ± 0.65 a | 4.66 ± 0.21 a |
| Flavourzyme | 7.0 | 55 | 0.05 M phosphate | 13.36 ± 0.54 b | 3.79 ± 0.16 b |
| Neutrase | 7.0 | 60 | 0.05 M phosphate | 14.07 ± 0.86 c | 2.57 ± 0.23 c |
| Trypsin | 8.0 | 37 | 0.05 M Tris-HCl | 11.57 ± 0.75 d | 5.47 ± 0.31 d |
| Alcalase | 9.5 | 50 | 0.05 M Gly-NaOH | 13.69 ± 0.93 e | 3.06 ± 0.19 e |
2.2. Optimization of Hydrolysis Conditions for Neutrase
| No. | Factors | EC50 (mg/mL) | |||
|---|---|---|---|---|---|
| (A) pH | (B) E/S (%) | (C) Time (h) | (D) Temp. (°C) | ||
| 1 | 1 (6.5) | 1(1.0) | 1 (3) | 1 (55) | 3.05 ± 0.34 a,b |
| 2 | 1 | 2 (1.5) | 2 (4) | 2 (60) | 2.97 ± 0.12 b |
| 3 | 1 | 3 (2.0) | 3 (5) | 3 (65) | 2.95 ± 0.29 b,c |
| 4 | 2 (7.0) | 1 | 2 | 3 | 2.86 ± 0.18 b,c |
| 5 | 2 | 2 | 3 | 1 | 2.65 ± 0.18 d |
| 6 | 2 | 3 | 1 | 2 | 2.39 ± 0.24 e |
| 7 | (7.5) | 1 | 3 | 2 | 2.78 ± 0.15 c,d |
| 8 | 3 | 2 | 1 | 3 | 3.17 ± 0.16 a |
| 9 | 3 | 3 | 2 | 1 | 2.87 ± 0.29 b,c |
| k1 | 8.97 | 8.69 | 8.61 | 8.57 | |
| k2 | 7.90 | 8.79 | 8.70 | 8.14 | |
| k3 | 8.82 | 8.21 | 8.38 | 8.98 | |
| Best level | A2 | B3 | C3 | D2 | |
| K1 | 2.99 | 2.90 | 2.87 | 2.86 | |
| K2 | 2.63 | 2.93 | 2.90 | 2.71 | |
| K3 | 2.94 | 2.74 | 2.79 | 2.99 | |
| R | 0.36 | 0.19 | 0.11 | 0.28 | |
| R order | A >D > B >C | ||||
2.3. Purification of Antioxidant Peptides from SMH
2.3.1. Fractionation of SMH by Ultrafiltration
2.3.2. Anion-Exchange Chromatography of SMH-III
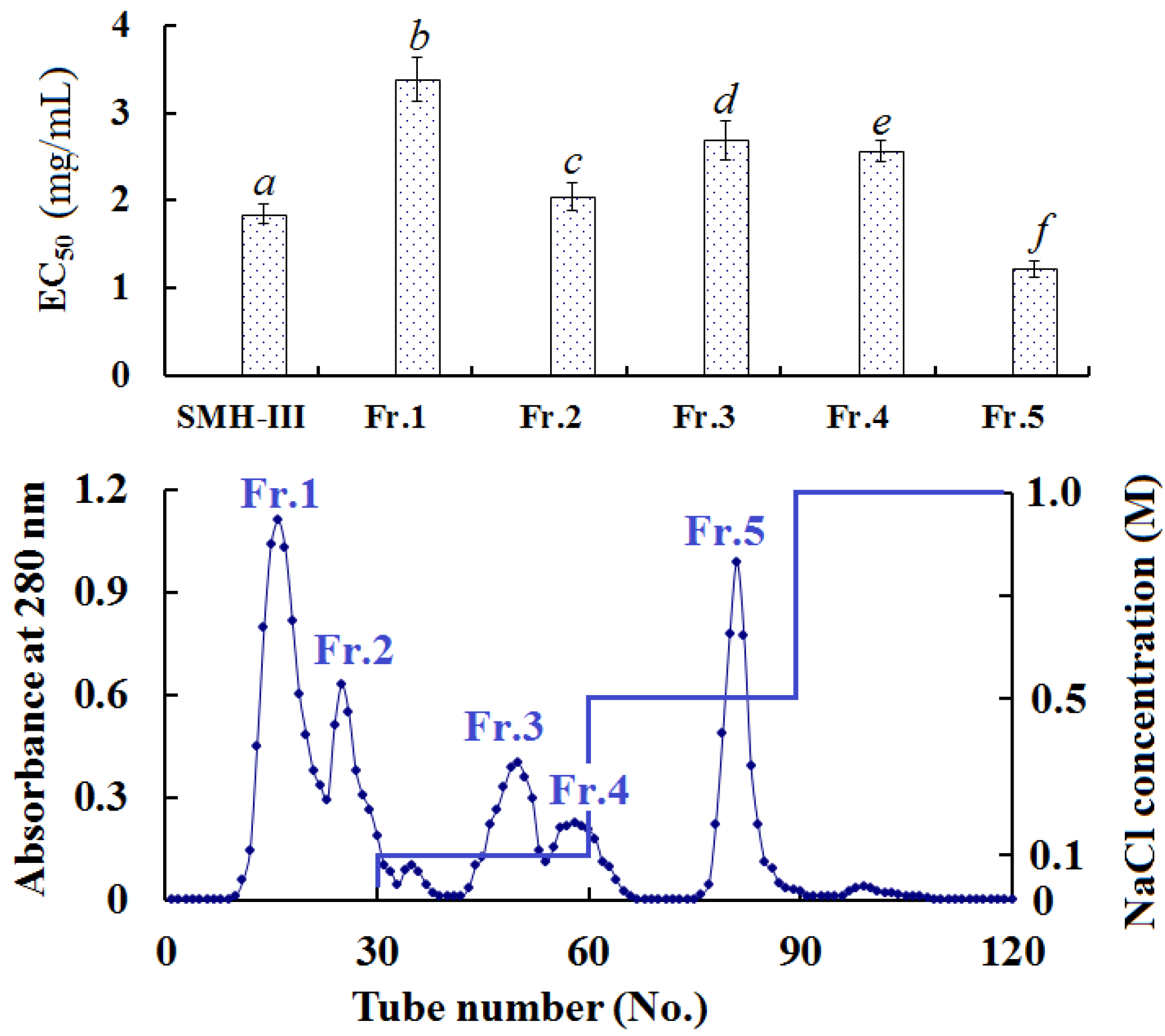
2.3.3. Gel Filtration Chromatography of Fr. 5

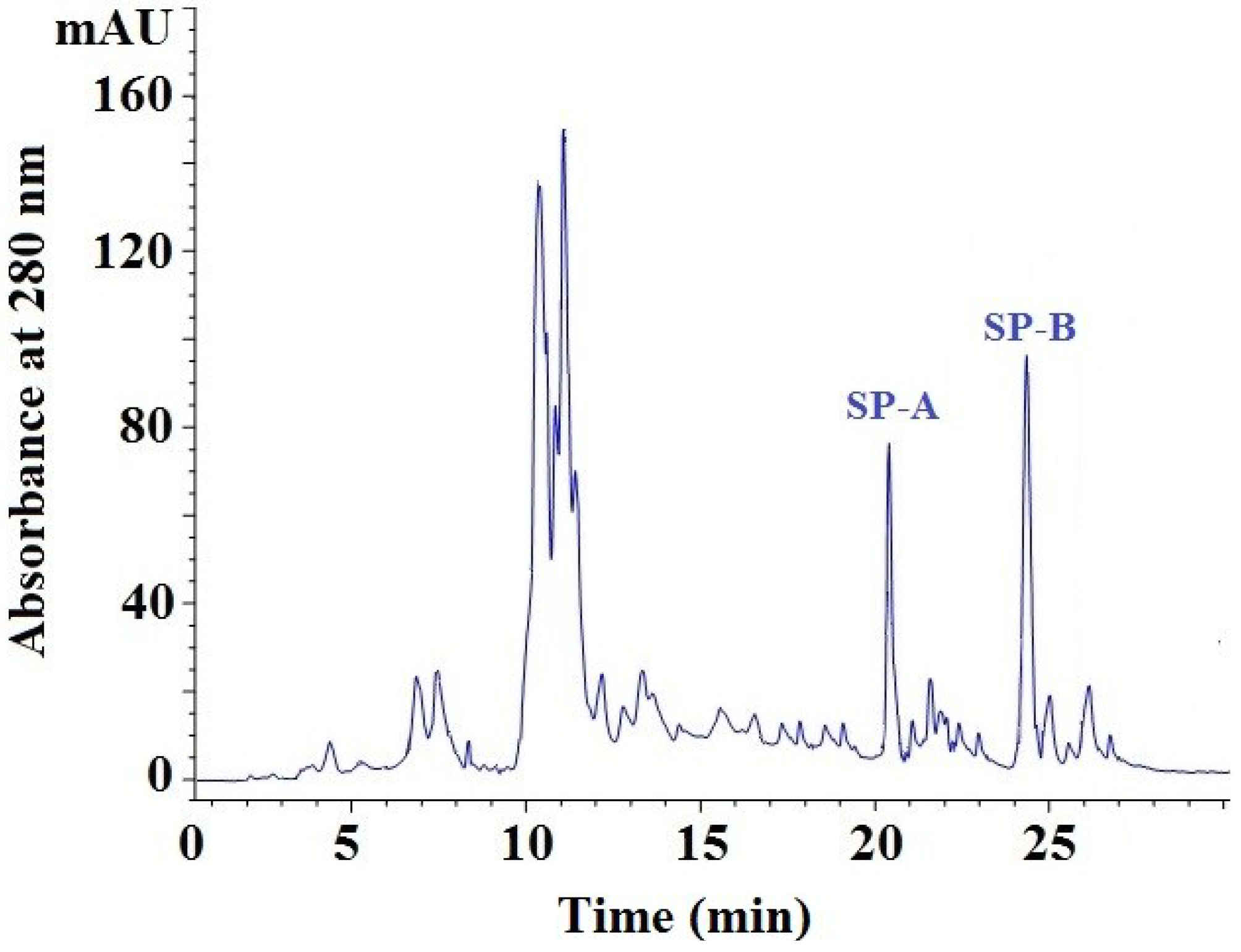
2.3.4. Isolation of Peptides from Fr. 5-3 by Reversed-Phase High Performance Liquid Chromatography (RP-HPLC)
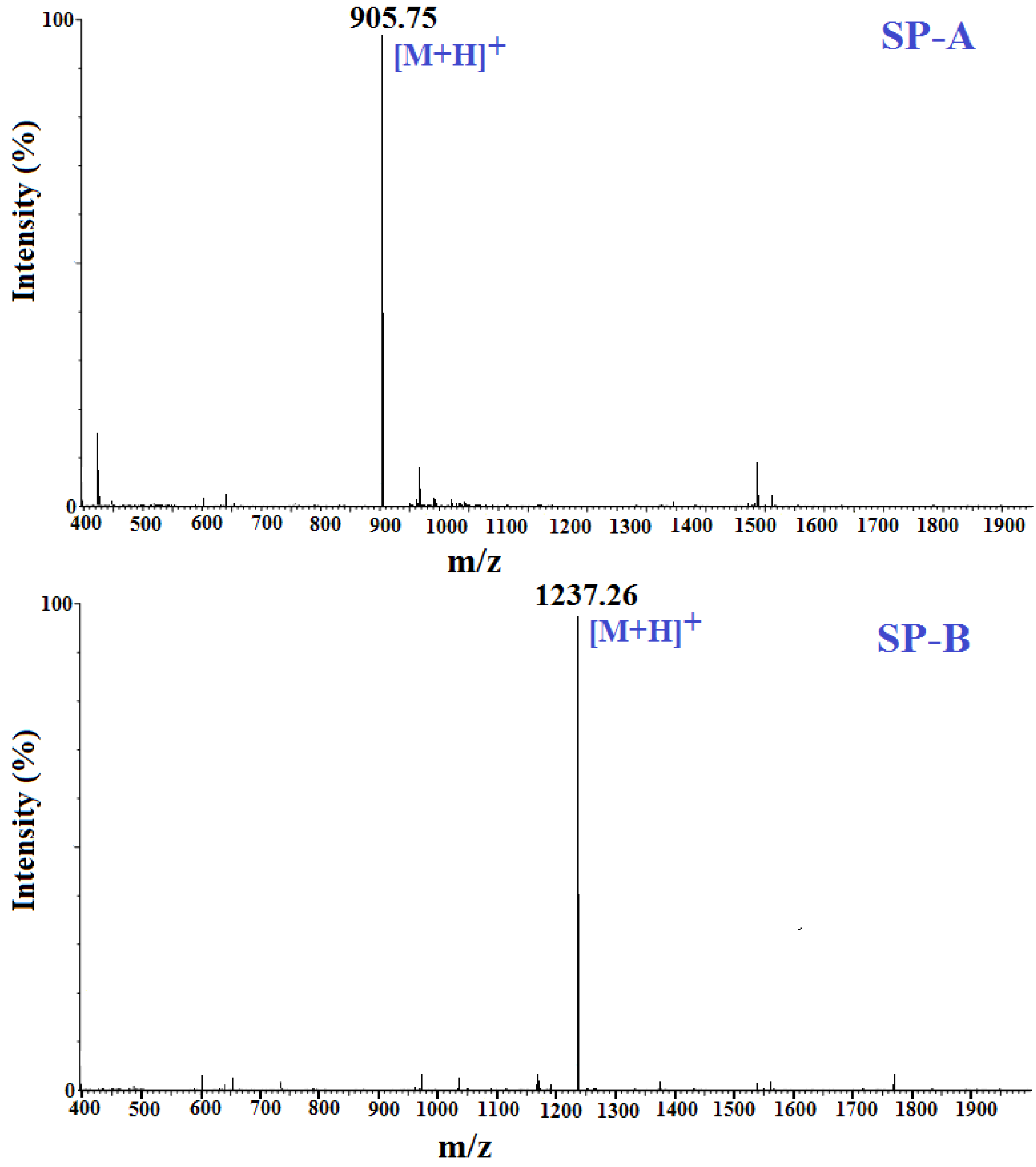
2.4. Amino Acid Sequence Analysis and Molecular Mass Determination
2.5. Antioxidant Activity of SP-A and SP-B
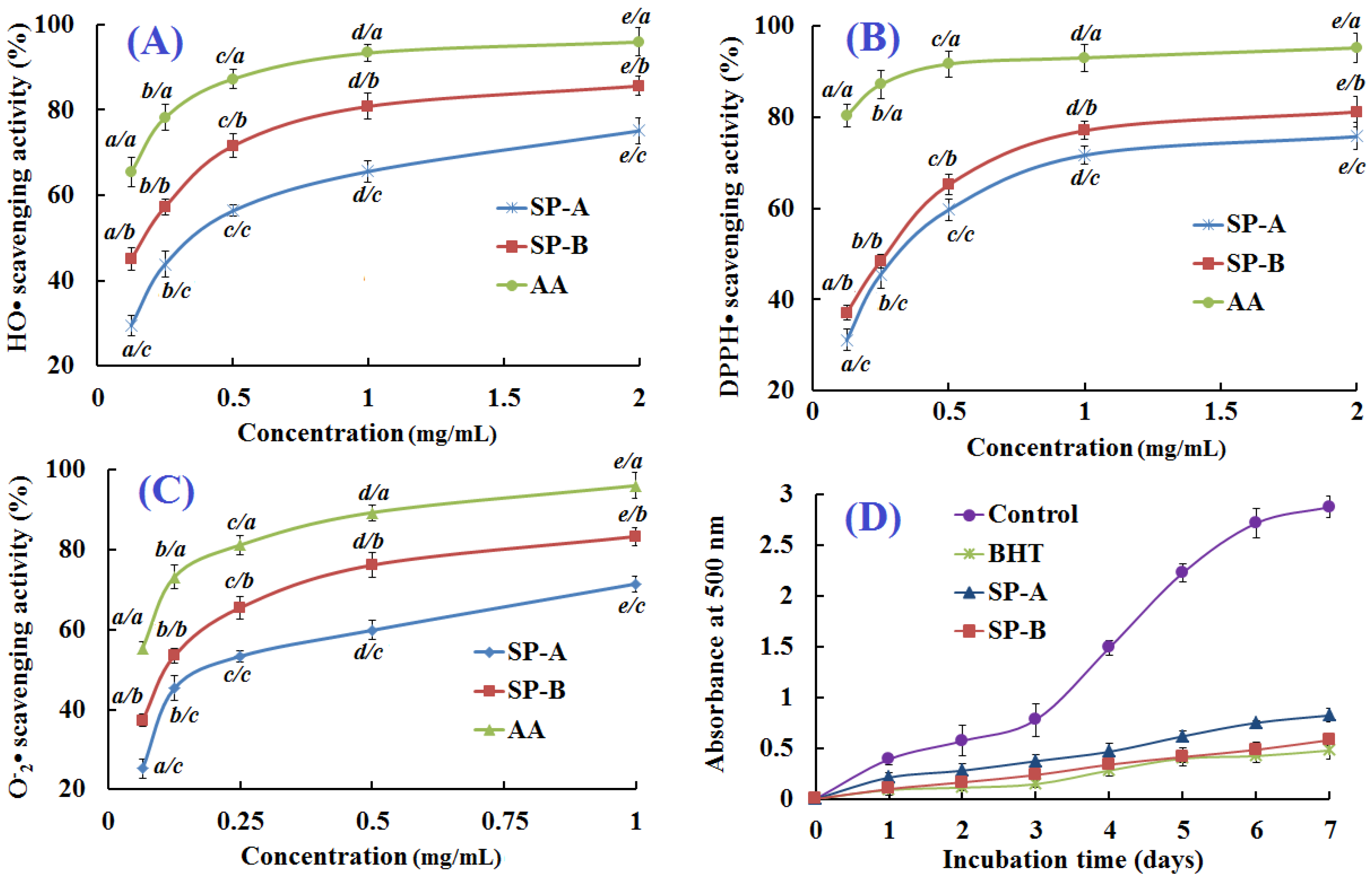
2.5.1. Hydroxyl Radical Scavenging Activity
2.5.2. 1,1-Diphenyl-2-picrylhydrazyl (DPPH) Radical Scavenging Activity
2.5.3. Superoxide Anion Radical Scavenging Activity
2.5.4. Lipid Peroxidation Inhibition Assay
2.6. Relationship between Antioxidant Activities and Amino Acid Compositions of Peptides
3. Experimental Section
3.1. Materials
3.2. Protein Hydrolysates of Skate Muscle (SMHs) Using Proteases
3.2.1. Preparation of Protein Hydrolysates Using Five Proteases
3.2.2. Optimum Conditions for the Active Enzymatic Hydrolysate
3.3. Isolation of Antioxidant Peptides from SMH
3.3.1. Fractionation of SMH by Ultrafiltration
3.3.2. Anion-Exchange Chromatography
3.3.3. Gel Filtration Chromatography of SMH-III
3.3.4. Isolation of Peptides from Fr. 5-3 by RP-HPLC
3.4. Degree of Hydrolysis (DH)
3.5. Amino Acid Sequence Analysis and Molecular Mass Determination
3.6. Antioxidative Activity
3.6.1. HO· Scavenging Activity
3.6.2. DPPH· Scavenging Activity
3.6.3. O2−· Scavenging Activity
3.6.4. Lipid Peroxidation Inhibition Assay
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Girgih, A.T.; He, R.; Hasan, F.M.; Udenigwe, C.C.; Gill, T.A.; Aluko, R.E. Evaluation of the in vitro antioxidant properties of a cod (Gadus morhua) protein hydrolysate and peptide fractions. Food Chem. 2015, 173, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wang, Y.; Chi, C.; Hu, F.; Deng, S.; Ma, J.Y. Isolation and characterization of collagen and antioxidant collagen peptides from scales of croceine croaker (Pseudosciaena crocea). Mar. Drugs 2013, 11, 4641–4661. [Google Scholar] [CrossRef] [PubMed]
- Jridi, M.; Lassoued, I.; Nasri, R.; Ayedi Mohamed, A.; Nasri, M.; Souissi, N. Characterization and potential use of cuttlefish skin gelatin hydrolysates prepared by different microbial proteases. Bio. Med. Res. Int. 2014. [Google Scholar] [CrossRef]
- Ahn, C.B.; Kim, J.G.; Je, J.Y. Purification and antioxidant properties of octapeptide from salmon byproduct protein hydrolysate by gastrointestinal digestion. Food Chem. 2014, 147, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Ji, N.; Sun, C.; Zhao, Y.; Xiong, L.; Sun, Q. Purification and identification of antioxidant peptides from peanut protein isolate hydrolysates using UHR-Q-TOF mass spectrometer. Food Chem. 2014, 161, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zheng, X.; Yang, Q.; Liang, Z.; Li, D.; Yang, X.; Xu, J. Optimization of ultrasonic-assisted extraction and radical-scavenging capacity of phenols and flavonoids from Clerodendrum cyrtophyllum Turcz leaves. PLoS ONE 2013, 8, e68392. [Google Scholar] [CrossRef] [PubMed]
- Zarei, M.; Ebrahimpour, A.; Abdul-Hamid, A.; Anwar, F.; Bakar, F.A.; Philip, R.; Saari, N. Identification and characterization of papain-generated antioxidant peptides from palm kernel cake proteins. Food Res. Int. 2014, 62, 726–734. [Google Scholar] [CrossRef]
- Onuh, J.O.; Girgih, A.T.; Aluko, R.E.; Aliani, M. In vitro antioxidant properties of chicken skin enzymatic protein hydrolysates and membrane fractions. Food Chem. 2014, 150, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.; Ren, J.; Sun, W.; You, L.; Yang, B.; Zhao, M. Isolation and identification of antioxidative peptides from frog (Hylarana guentheri) protein hydrolysate by consecutive chromatography and electrospray ionization mass spectrometry. Appl. Biochem. Biotechnol. 2014, 173, 1169–1182. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Li, L.; Chi, C.F.; Ma, JH.; Luo, H.Y.; Xu, Y.F. Purification and characterisation of a novel antioxidant peptide derived from blue mussel (Mytilus edulis) protein hydrolysate. Food Chem. 2013, 138, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Li, Z.R.; Chi, C.F.; Zhang, Q.H.; Luo, H.Y. Preparation and evaluation of antioxidant peptides from ethanol-soluble proteins hydrolysate of Sphyrna lewini muscle. Peptides 2012, 36, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Luo, HY.; Wang, B.; Li, Z.R.; Chi, C.F.; Zhang, Q.H.; He, G.Y. Preparation and evaluation of antioxidant peptide from papain hydrolysate of Sphyrna lewini muscle protein. LWT Food Sci. Technol. 2013, 51, 281–288. [Google Scholar] [CrossRef]
- Umayaparvathi, S.; Meenakshi, S.; Vimalraj, V.; Arumugam, M.; Balasubramanian, T. Isolation and structural elucidation of antioxidant peptides from Oyster (Saccostrea cucullata) protein hydrolysate. Protein Pept. Lett. 2014, 21, 1073–1083. [Google Scholar] [CrossRef] [PubMed]
- Taheri, A.; Farvin, K.H.S.; Jacobsen, C.; Baron, C.P. Antioxidant activities and functional properties of protein and peptide fractions isolated from salted herring brine. Food Chem. 2014, 142, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Nazeer, R.A.; Prabha, K.R.; Kumar, N.S.; Ganesh, R.J. Isolation of antioxidant peptides from clam, Meretrix casta (Chemnitz). J. Food Sci. Technol. 2013, 50, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, B.; Chi, C.; Gong, Y.; Luo, H.; Ding, G.F. Influence of average molecular weight on antioxidant and functional properties of cartilage collagen hydrolysates from Sphyrna lewini, Dasyatis akjei and Raja porosa. Food Res. Int. 2013, 51, 283–293. [Google Scholar] [CrossRef]
- Wu, S.; Sun, J.; Tong, Z.; Lan, X.; Zhao, Z.; Liao, D. Optimization of hydrolysis conditions for the production of angiotensin-I converting enzyme-inhibitory peptides and isolation of a novel peptide from Lizard fish (Saurida elongata) muscle protein hydrolysate. Mar. Drugs 2012, 10, 1066–1080. [Google Scholar] [CrossRef] [PubMed]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Kishimura, H.; Shahidi, F. Isolation and characterisation of collagen from the skin of brownbanded bamboo shark (Chiloscyllium punctatum). Food Chem. 2010, 119, 1519–1526. [Google Scholar] [CrossRef]
- Chi, C.F.; Wang, B.; Li, Z.R.; Luo, H.Y.; Ding, G.F.; Wu, C.W. Characterization of acid-soluble collagen from the skin of hammerhead shark (Sphyrna lewini). J. Food Biochem. 2014, 38, 236–247. [Google Scholar] [CrossRef]
- Zheng, L.; Ling, P.; Wang, Z.; Niu, R.; Hu, C.; Zhang, T.; Lin, X. A novel polypeptide from shark cartilage with potent anti-angiogenic activity. Cancer Biol. Ther. 2007, 6, 775–780. [Google Scholar]
- Chi, C.F.; Wang, B.; Wang, Y.M.; Deng, S.G.; Ma, J.Y. Isolation and characterization of three antioxidant pentapeptides from protein hydrolysate of monkfish (Lophius litulon) muscle. Food Res. Int. 2014, 55, 222–228. [Google Scholar] [CrossRef]
- Ko, J.Y.; Lee, J.H.; Samarakoon, K.; Kim, J.S.; Jeon, Y.J. Purification and determination of two novel antioxidant peptides from flounder fish (Paralichthys olivaceus) using digestive proteases. Food Chem. Toxicol. 2013, 52, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Klompong, V.; Benjakul, S.; Kantachote, D.; Shahidi, F. Antioxidative activity and functional properties of protein hydrolysate of yellow stripe trevally (Selaroides leptolepis) as influenced by the degree of hydrolysis and enzyme type. Food Chem. 2007, 102, 1317–1327. [Google Scholar] [CrossRef]
- Ranathunga, S.; Rajapakse, N.; Kim, S.K. Purification and characterization of antioxidative peptide derived from muscle of conger eel (Conger myriaster). Eur. Food Res. Technol. 2006, 222, 310–315. [Google Scholar] [CrossRef]
- You, L.; Zhao, M.; Regenstein, J.M.; Ren, J. Purification and identification of antioxidative peptides from loach (Misgurnus anguillicaudatus) protein hydrolysate by consecutive chromatography and electrospray ionization-mass spectrometry. Food Res. Int. 2010, 43, 1167–1173. [Google Scholar] [CrossRef]
- Mendis, E.; Rajapakse, N.; Byun, H.G.; Kim, S.K. Investigation of jumbo squid (Dosidicus gigas) skin gelatin peptides for their in vitro antioxidant effects. Life Sci. 2005, 77, 2166–2178. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, N.; Mendis, E.; Byun, H.G.; Kim, S.K. Purification and in vitro antioxidative effects of giant squid muscle peptides on free radical-mediated oxidative systems. J. Nutr. Biochem. 2005, 9, 562–569. [Google Scholar] [CrossRef]
- Kim, E.K.; Lee, S.J.; Jeon, B.T.; Moon, S.H.; Kim, B.K.; Park, T.K.; Han, J.S.; Park, P.J. Purification and characterisation of antioxidative peptides from enzymatic hydrolysates of venison protein. Food Chem. 2009, 114, 1365–1370. [Google Scholar] [CrossRef]
- Qian, Z.J.; Jung, W.K.; Byun, H.G.; Kim, S.K. Protective effect of an antioxidative peptide purified from gastrointestinal digests of oyster, Crassostrea gigas against free radical induced DNA damage. Bioresour. Technol. 2008, 99, 3365–3371. [Google Scholar] [CrossRef] [PubMed]
- Conway, V.; Gauthier, S.F.; Pouliot, Y. Antioxidant activities of buttermilk proteins, whey proteins, and their enzymatic hydrolysates. J. Agric. Food Chem. 2013, 61, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Chi, C.F.; Wang, B.; Wang, Y.M.; Zhang, B.; Deng, S.G. Isolation and characterization of three antioxidant peptides from protein hydrolysate of bluefin leatherjacket (Navodon septentrionalis) heads. J. Funct. Foods 2015, 12, 1–10. [Google Scholar] [CrossRef]
- Ren, J.Y.; Zhao, M.M.; Shi, J.; Wang, J.S.; Jiang, Y.M.; Cui, C.; Kakuda, Y.; Xue, S.J. Purification and identification of antioxidant peptides from grass carp muscle hydrolysates by consecutive chromatography and electrospray ionization-mass spectrometry. Food Chem. 2008, 108, 727–736. [Google Scholar] [CrossRef]
- Sarmadi, B.H.; Ismail, A. Antioxidative peptides from food proteins: A review. Peptides 2010, 31, 1949–1956. [Google Scholar] [CrossRef] [PubMed]
- Dávalos, A.; Miguel, M.; Bartolomé, B.; López-Fandiño, R. Antioxidant activity of peptides derived from egg white proteins by enzymatic hydrolysis. J. Food Protect. 2004, 67, 1939–1944. [Google Scholar]
- Zhang, M.; Mu, T.H.; Sun, M.J. Purification and identification of antioxidant peptides from sweet potato protein hydrolysates by Alcalase. J. Funct. Foods 2014, 7, 191–200. [Google Scholar] [CrossRef]
- Chang, O.K.; Ha, G.E.; Han, G.S.; Seol, K.H.; Kim, H.W.; Jeong, S.G.; Oh, M.H.; Park, B.Y.; Ham, J.S. Novel antioxidant peptide derived from the ultrafiltrate of ovomucin hydrolysate. J. Agric. Food Chem. 2013, 61, 7294–7300. [Google Scholar] [CrossRef] [PubMed]
- Kou, X.H.; Gao, J.; Xue, Z.H.; Zhang, Z.J.; Wang, H.; Wang, X. Purification and identification of antioxidant peptides from chickpea (Cicer arietinum L.) albumin hydrolysates. LWT Food Sci. Technol. 2013, 50, 591–598. [Google Scholar] [CrossRef]
- Byun, H.G.; Lee, J.K.; Park, H.G.; Jeon, J.K.; Kim, S.K. Antioxidant peptides isolated from the marine rotifer, Brachionus rotundiformis. Proc. Biochem. 2009, 44, 842–846. [Google Scholar] [CrossRef]
- Gimenez, B.; Aleman, A.; Montero, P.; Gomez-Guillen, M.C. Antioxidant and functional properties of gelatin hydrolysates obtained from skin of sole and squid. Food Chem. 2009, 114, 976–983. [Google Scholar] [CrossRef]
- Memarpoor-Yazdia, M.; Mahakia, H.; Zare-Zardinib, H. Antioxidant activity of protein hydrolysates and purified peptides from Zizyphus jujuba fruits. J. Funct. Foods 2013, 5, 62–70. [Google Scholar] [CrossRef]
- Mitsuta, H.; Yasumoto, K.; Iwami, K. Antioxidative action of indole compounds during the autoxidation of linoleic acid. Eiyo Shokuryo 1996, 29, 238–244. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, F.-Y.; Chi, C.-F.; Wang, B.; Deng, S.-G. Two Novel Antioxidant Nonapeptides from Protein Hydrolysate of Skate (Raja porosa) Muscle. Mar. Drugs 2015, 13, 1993-2009. https://doi.org/10.3390/md13041993
Hu F-Y, Chi C-F, Wang B, Deng S-G. Two Novel Antioxidant Nonapeptides from Protein Hydrolysate of Skate (Raja porosa) Muscle. Marine Drugs. 2015; 13(4):1993-2009. https://doi.org/10.3390/md13041993
Chicago/Turabian StyleHu, Fa-Yuan, Chang-Feng Chi, Bin Wang, and Shang-Gui Deng. 2015. "Two Novel Antioxidant Nonapeptides from Protein Hydrolysate of Skate (Raja porosa) Muscle" Marine Drugs 13, no. 4: 1993-2009. https://doi.org/10.3390/md13041993
APA StyleHu, F.-Y., Chi, C.-F., Wang, B., & Deng, S.-G. (2015). Two Novel Antioxidant Nonapeptides from Protein Hydrolysate of Skate (Raja porosa) Muscle. Marine Drugs, 13(4), 1993-2009. https://doi.org/10.3390/md13041993








