6-Bromohypaphorine from Marine Nudibranch Mollusk Hermissenda crassicornis is an Agonist of Human α7 Nicotinic Acetylcholine Receptor
Abstract
:1. Introduction
2. Results and Discussion
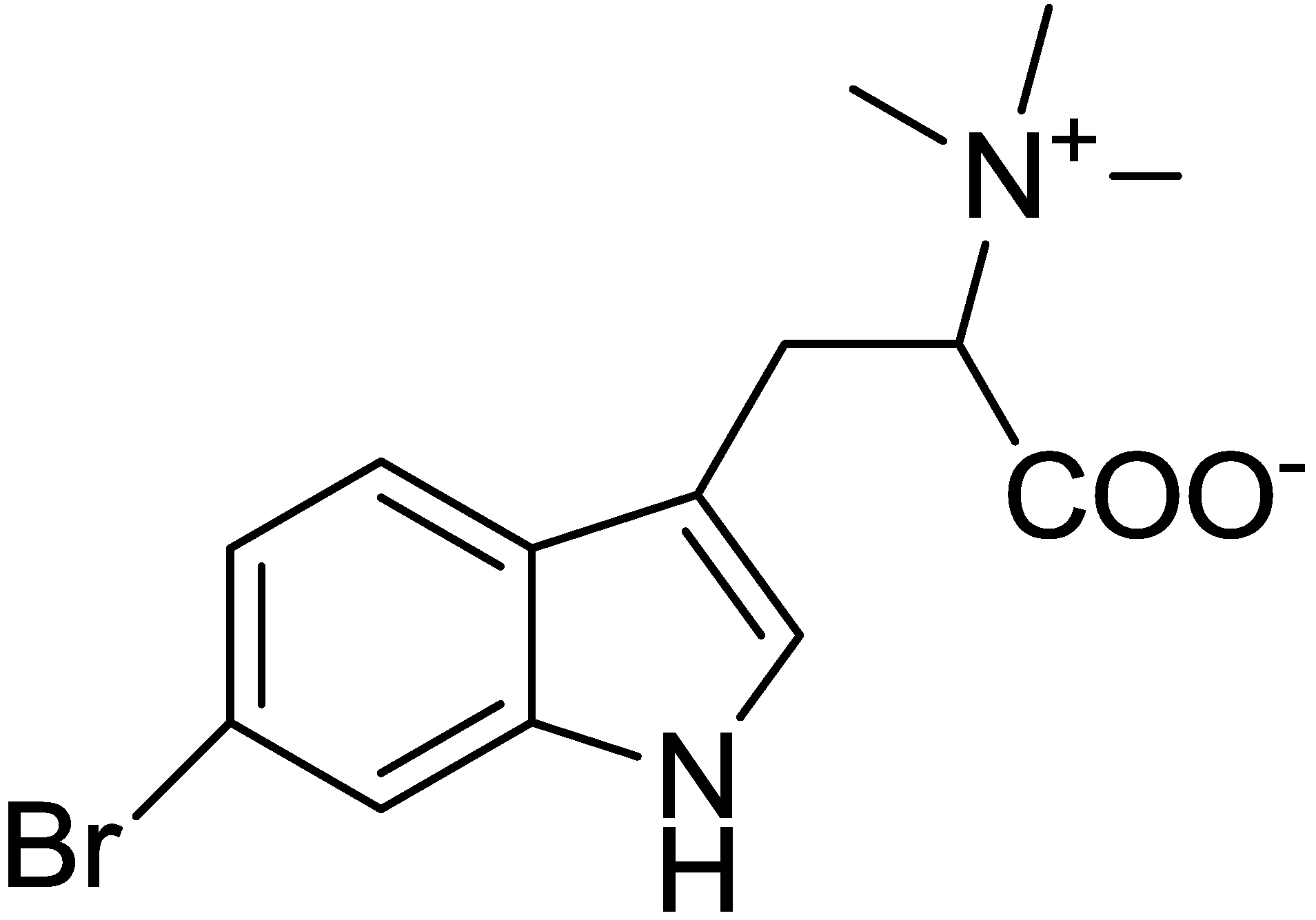
2.1. Isolation and Structure Determination for 6-Bromohypaphorine from Hermissenda crassicornis
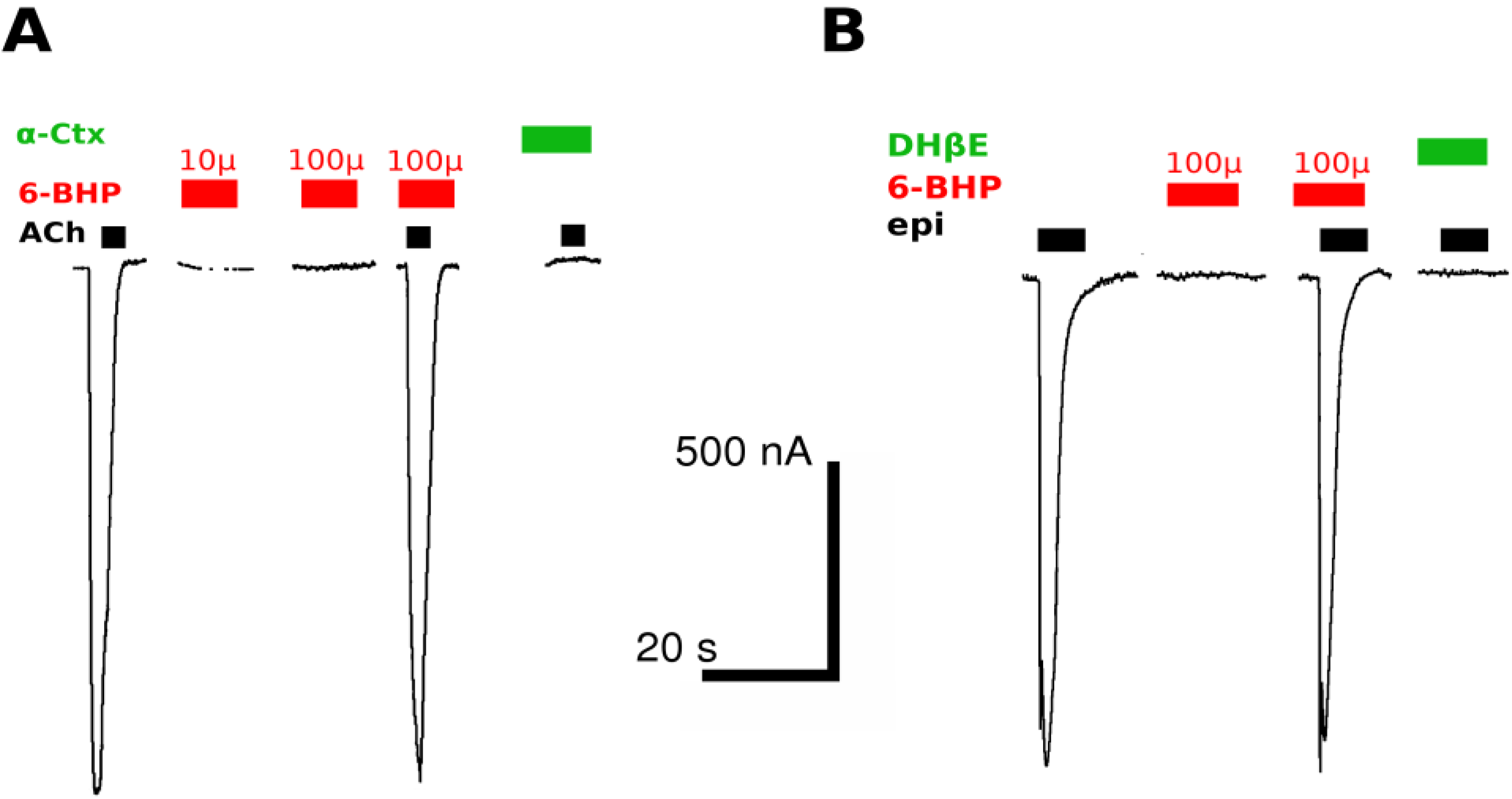
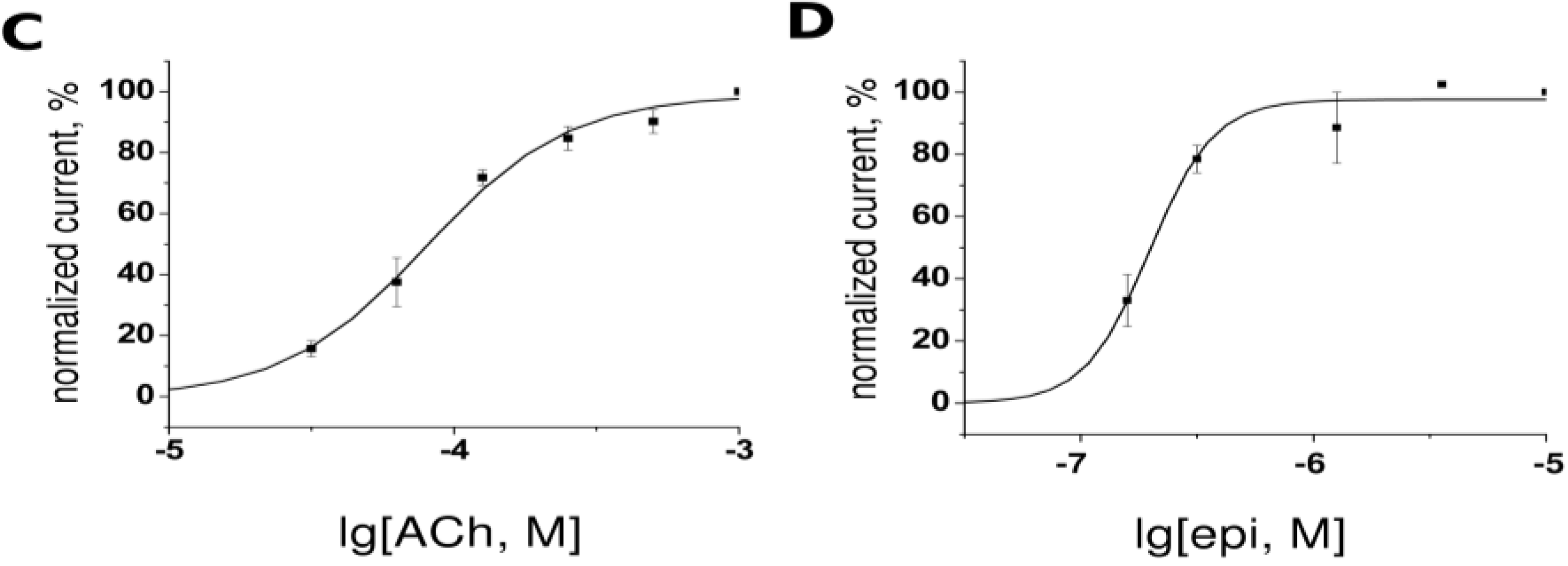
2.2. Two-Electrode Voltage-Clamp
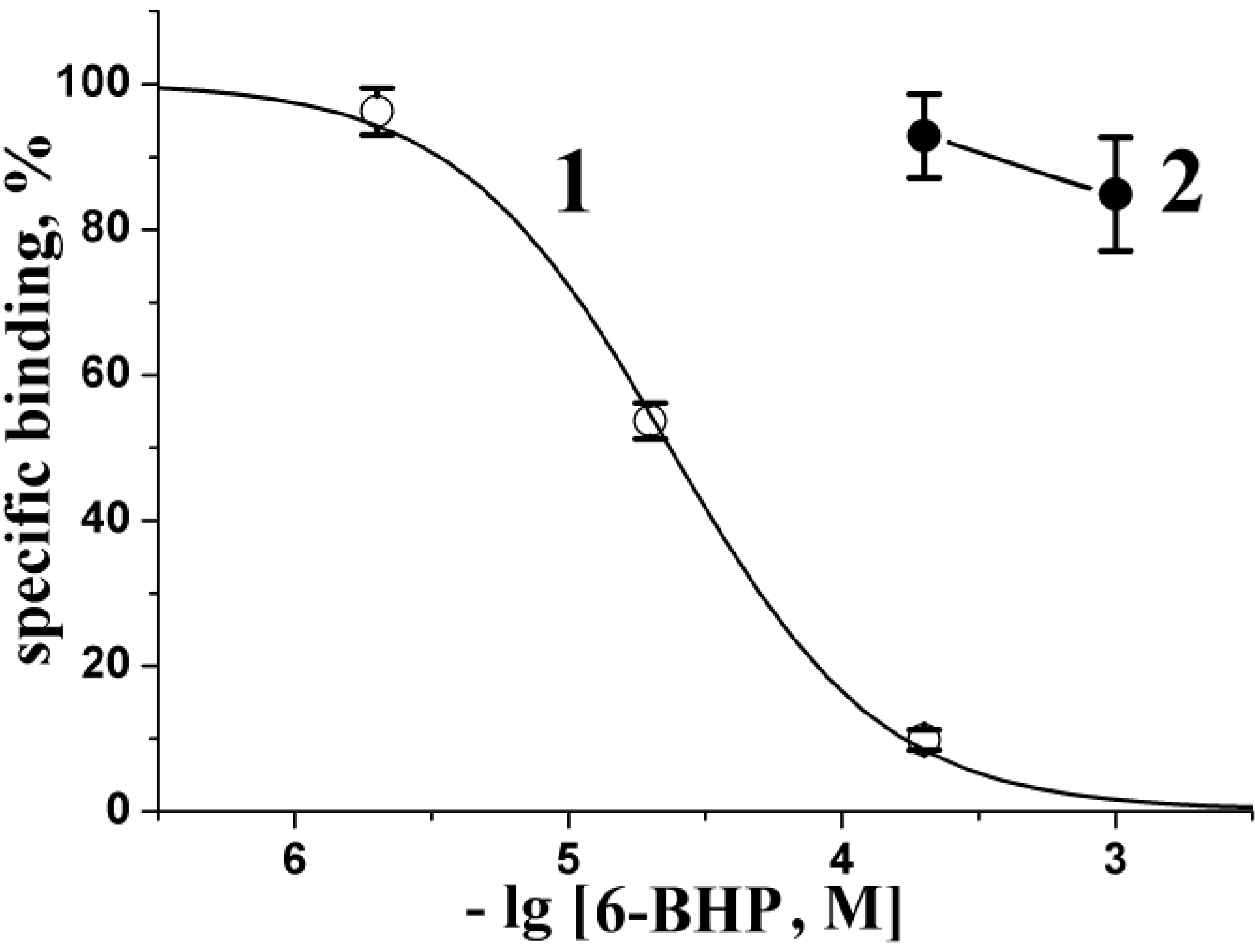
2.3. Radioligand Analysis
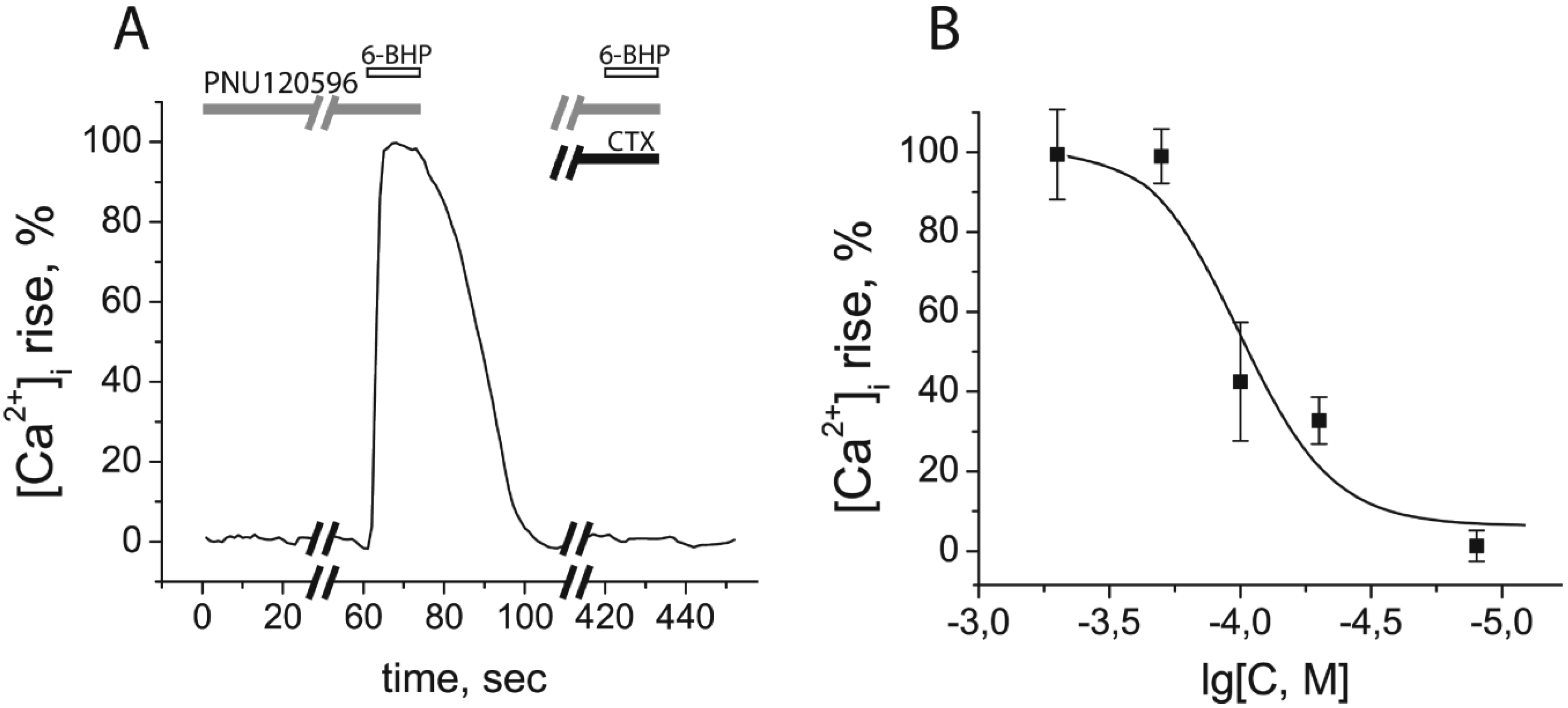
2.4. Calcium Imaging
2.5. Discussion
3. Experimental Section
3.1. Isolation and Structure Determination of l-6-Bromohypaphorine
3.1.1. Animal Material and Isolation of 6-BHP
3.1.2. Physical Characteristics and Spectral Data fo 6-BHP
3.2. Heterologous Expression of the nAChR Subtypes in Xenopus Oocytes and Electrophysiology Measurements
3.3. Heterlogous Expression of the α7 nAChR in Neuroblastoma Neuro2a Cell Line and Ca2+ Measurements in Response to Agonists and Antagonists
3.4. Radioligand Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug Discov. 2009, 8, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Mollica, A.; Locatelli, M.; Stefanucci, A.; Pinnen, F. Synthesis and bioactivity of secondary metabolites from marine sponges containing dibrominated indolic systems. Molecules 2012, 17, 6083–6099. [Google Scholar] [CrossRef] [PubMed]
- Kudryavtsev, D.; Makarieva, T.; Utkina, N.; Santalova, E.; Kryukova, E.; Methfessel, C.; Tsetlin, V.; Stonik, V.; Kasheverov, I. Marine natural products acting on the acetylcholine-binding protein and nicotinic receptors: From computer modeling to binding studies and electrophysiology. Mar. Drugs 2014, 12, 1859–1875. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, J.; Hamilton, B.; Farley, J. Behavioral and neural bases of extinction learning in Hermissenda. Front. Behav. Neurosci. 2014, 19, 9126–9132. [Google Scholar]
- Raverty, W.; Thomson, R.; King, T. Metabolites from the sponge Pachymatisma johnstoni; l-6-bromohypaphorine, a new amino-acid (and its crystal structure). J. Chem. Soc. 1977, 10, 1204–1211. [Google Scholar]
- Longeon, A.; Copp, B.; Quévrain, E.; Roué, M. Bioactive indole derivatives from the South Pacific marine sponges Rhopaloeides odorabile and Hyrtios sp. Mar. Drugs 2011, 9, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Hurst, R.S.; Hajós, M.; Raggenbass, M.; Wall, T.M.; Higdon, N.R.; Lawson, J.A.; Rutherford-Root, K.L.; Berkenpas, M.B.; Hoffmann, W.E.; Piotrowski, D.W.; et al. A novel positive allosteric modulator of the alpha7 neuronal nicotinic acetylcholine receptor: In vitro and in vivo characterization. J. Neurosci. 2005, 25, 4396–4405. [Google Scholar] [CrossRef] [PubMed]
- Collins, T.; Young, G.; Millar, N. Competitive binding at a nicotinic receptor transmembrane site of two α7-selective positive allosteric modulators with differing effects on agonist-evoked. Neuropharmacology 2011, 68, 1306–1313. [Google Scholar] [CrossRef]
- Barron, S.C.; McLaughlin, J.T.; See, J.A.; Richards, V.L.; Rosenberg, R.L. An allosteric modulator of alpha7 nicotinic receptors, N-(5-Chloro-2,4-dimethoxyphenyl)-N′-(5-methyl-3-isoxazolyl)-urea (PNU-120596), causes conformational changes in the extracellular ligand binding domain similar to those caused by acetylcholine. Mol. Pharmacol. 2009, 76, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Young, G.T.; Zwart, R.; Walker, A.S.; Sher, E.; Millar, N.S. Potentiation of alpha7 nicotinic acetylcholine receptors via an allosteric transmembrane site. Proc. Natl. Acad. Sci. USA 2008, 105, 14686–14691. [Google Scholar] [CrossRef] [PubMed]
- Dauphin, A.; Gerard, J.; Lapeyrie, F.; Legue, V. Fungal hypaphorine reduces growth and induces cytosolic calcium increase in root hairs of Eucalyptus globulus. Protoplasma 2007, 231, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, M.; Honda, K.; Nakai, I. Hypaphorine, an indole alkaloid from Erythrina velutina, induced sleep on normal mice. Bioorg. Med. Chem. Lett. 2008, 18, 3992–3994. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, E.; Watkins, M.; Olivera, B. Multiple 6-bromotryptophan residues in a sleep-inducing peptide. Biochemistry 2004, 43, 12343–12348. [Google Scholar] [CrossRef] [PubMed]
- Kasheverov, I.E.; Utkin, Y.N.; Tsetlin, V.I. Naturally occurring and synthetic peptides acting on nicotinic acetylcholine receptors. Curr. Pharm. Des. 2009, 15, 2430–2452. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.D.; Norton, R.S. Conotoxin Gene Superfamilies. Mar. Drugs 2014, 12, 6058–6101. [Google Scholar] [CrossRef] [PubMed]
- Jambois, A.; Ditengou, F. The indole alkaloids brucine, yohimbine, and hypaphorine are indole-3-acetic acid-specific competitors which do not alter auxin transport. Physiol. Plant. 2004, 120, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Kawano, T. Possible use of indole-3-acetic acid and its antagonist tryptophan betaine in controlled killing of horseradish peroxidase-labeled human cells. Med. Hypotheses 2003, 60, 664–666. [Google Scholar] [CrossRef] [PubMed]
- Aiello, A.; Borrelli, F.; Capasso, R. Conicamin, a novel histamine antagonist from the mediterranean tunicate Aplidium conicum. Bioorg. Med. Chem. Lett. 2003, 13, 4481–4483. [Google Scholar] [CrossRef] [PubMed]
- Hedner, E.; Sjögren, M. Brominated cyclodipeptides from the marine sponge Geodia barretti as selective 5-HT ligands. J. Nat. Prod. 2006, 69, 1421–1424. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Nishi, J. Two new tryptophan-derived alkaloids from the Okinawan marine sponge Aplysina sp. J. Nat. Prod. 1994, 57, 1008–1011. [Google Scholar] [CrossRef] [PubMed]
- Tsetlin, V.I.; Kuzmin, D.; Kasheverov, I.E. Assembly of nicotinic and other Cys-loop receptors. J. Neurochem. 2011, 116, 734–741. [Google Scholar] [CrossRef] [PubMed]
- Blanton, M.; McCardy, E.; Fryer, J. 5-Hydroxytryptamine interaction with the nicotinic acetylcholine receptor. Eur. J. Pharmacol. 2000, 389, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Colunga, J.; Miledi, R. Blockage of mouse muscle nicotinic receptors by serotonergic compounds. Exp. Physiol. 1999, 84, 847–864. [Google Scholar] [CrossRef] [PubMed]
- Rothlin, C.; Lioudyno, M.; Silbering, A. Direct interaction of serotonin type 3 receptor ligands with recombinant and native α9α10-containing nicotinic cholinergic receptors. Mol. Pharmacol. 2003, 63, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Pedersen, S.E.; White, M.M. Interaction of d-tubocurarine analogs with the 5HT3 receptor. Neuropharmacology 1998, 37, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Lummis, S.; Thompson, A. Varenicline is a potent agonist of the human 5-hydroxytryptamine3 receptor. J. Pharmacol. Exp. Ther. 2011, 339, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Azam, L.; McIntosh, J.M. Molecular basis for the differential sensitivity of rat and human α9α10 nAChRs to α-conotoxin RgIA. J. Neurochem. 2012, 122, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Lyukmanova, E.N.; Shulepko, M.A.; Buldakova, S.L.; Kasheverov, I.E.; Shenkarev, Z.O.; Reshetnikov, R.V.; Filkin, S.Y.; Kudryavtsev, D.S.; Ojomoko, L.O.; Kryukova, E.V.; et al. Water-soluble LYNX1 residues important for interaction with muscle-type and/or neuronal nicotinic receptors. J. Biol. Chem. 2013, 288, 15888–15899. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, J.; Mackessy, S.P.; Fry, B.G.; Bhatia, M.; Mourier, G.; Fruchart-Gaillard, C.; Servent, D.; Ménez, R.; Stura, E.; Ménez, A.; et al. Denmotoxin, a three-finger toxin from the colubrid snake Boiga dendrophila (Mangrove Catsnake) with bird-specific activity. J. Biol. Chem. 2006, 281, 29030–29041. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kasheverov, I.E.; Shelukhina, I.V.; Kudryavtsev, D.S.; Makarieva, T.N.; Spirova, E.N.; Guzii, A.G.; Stonik, V.A.; Tsetlin, V.I. 6-Bromohypaphorine from Marine Nudibranch Mollusk Hermissenda crassicornis is an Agonist of Human α7 Nicotinic Acetylcholine Receptor. Mar. Drugs 2015, 13, 1255-1266. https://doi.org/10.3390/md13031255
Kasheverov IE, Shelukhina IV, Kudryavtsev DS, Makarieva TN, Spirova EN, Guzii AG, Stonik VA, Tsetlin VI. 6-Bromohypaphorine from Marine Nudibranch Mollusk Hermissenda crassicornis is an Agonist of Human α7 Nicotinic Acetylcholine Receptor. Marine Drugs. 2015; 13(3):1255-1266. https://doi.org/10.3390/md13031255
Chicago/Turabian StyleKasheverov, Igor E., Irina V. Shelukhina, Denis S. Kudryavtsev, Tatyana N. Makarieva, Ekaterina N. Spirova, Alla G. Guzii, Valentin A. Stonik, and Victor I. Tsetlin. 2015. "6-Bromohypaphorine from Marine Nudibranch Mollusk Hermissenda crassicornis is an Agonist of Human α7 Nicotinic Acetylcholine Receptor" Marine Drugs 13, no. 3: 1255-1266. https://doi.org/10.3390/md13031255
APA StyleKasheverov, I. E., Shelukhina, I. V., Kudryavtsev, D. S., Makarieva, T. N., Spirova, E. N., Guzii, A. G., Stonik, V. A., & Tsetlin, V. I. (2015). 6-Bromohypaphorine from Marine Nudibranch Mollusk Hermissenda crassicornis is an Agonist of Human α7 Nicotinic Acetylcholine Receptor. Marine Drugs, 13(3), 1255-1266. https://doi.org/10.3390/md13031255







