Effects of n-3 Polyunsaturated Fatty Acids (ω-3) Supplementation on Some Cardiovascular Risk Factors with a Ketogenic Mediterranean Diet
Abstract
:1. Introduction
2. Results
| KD Group | KDO3 Group | |
|---|---|---|
| Age (years) | 56.3 ± 5.1 | 58.1 ± 6.0 |
| Height (cm) | 177.2 ± 4.7 | 180 ± 7.9 |
| Weight (kg) | 92.25 ± 12.53 | 94.5 ± 15.20 |
| BMI | 29.34 ± 2.40 | 29.17 ± 2.37 |
| LBM (kg) | 58.06 ± 14.68 | 58.87 ± 18.19 |
| FM (kg) | 34.19 ± 3.93 | 35.93 ± 6.76 |
| KD Pre | KD Post | Δ If Pre-Post Tukey’s t | KDO3 Pre | KDO3 Post | Δ CTR Pre-Post Tukey’s t | Between Treatment ANOVA t | |
|---|---|---|---|---|---|---|---|
| Weight (kg) | 92.25 ± 12.53 | 87.55 ± 10.03 | p < 0.05 | 94.5 ± 15.2 | 90.48 ± 12.71 | p < 0.05 | n.s. |
| BMI | 29.34 ± 2.41 | 27.87±1.70 | p < 0.05 | 29.17 ± 2.38 | 27.96 ± 1.80 | p < 0.05 | n.s. |
| LBM (kg) | 58.06 ± 14.68 | 58.77±12.02 | n.s. | 58.87 ± 18.19 | 60.41 ± 15.09 | n.s. | n.s. |
| FM (kg) | 34.19 ± 3.93 | 28.78 ± 4.37 | p < 0.005 | 35.93 ± 6.76 | 30.06 ± 5.09 | p < 0.005 | n.s. |
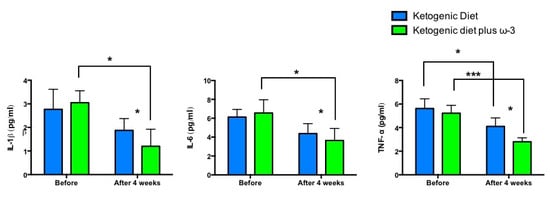
| IL-6 (pg/mL) | 6.13 ± 0.81 | 4.38 ± 1.05 | n.s. | 6.55 ± 1.41 | 3.65 ± 1.27 | p < 0.05 | p < 0.05 |
| IL-1β (pg/mL) | 2.77 ± 0.85 | 1.88 ± 0.5 | n.s. | 3.05 ± 0.51 | 1.2 ± 0.73 | p < 0.05 | p < 0.05 |
| TNF-α (pg/mL) | 5.63 ± 0.82 | 4.1 ± 0.73 | p < 0.05 | 5.23 ± 0.67 | 2.8 ± 0.34 | p < 0.001 | p < 0.05 |
| IL-1Ra | 287 ± 101.54 | 307 ± 89.57 | n.s. | 299 ± 72.36 | 301 ± 88.95 | n.s. | n.s. |
| IL-10 | 5.10 ± 3.8 | 6.25 ± 2.25 | n.s. | 4.7 ± 4.3 | 5.15 ± 3.37 | n.s. | n.s. |
| Insulin (μIU/mL) | 9.3 ± 2.00 | 7.4 ± 0.90 | p < 0.05 | 11.1 ± 1.90 | 6.3 ± 1.10 | p < 0.05 | p < 0.05 |
| Glucose (mg/dL) | 103.5 ± 5.30 | 85.3 ± 4.70 | p < 0.05 | 107.8 ± 6.30 | 82.1 ± 7.20 | p < 0.05 | n.s. |
| Adiponectin (μg/mL) | 7.18 ± 0.67 | 7.83 ± 0.39 | n.s. | 6.55 ± 1.02 | 7.85 ± 0.37 | p < 0.05 | p < 0.05 |
| Tot Chol (mg/dL) | 217.25 ± 15.84 | 201.28 ± 6.79 | p < 0.05 | 222.39 ± 6.10 | 204.52 ± 9.78 | p < 0.05 | n.s. |
| HDL-c (mg/dL) | 36.28 ± 2.23 | 39.25 ± 1.37 | n.s. | 39.55 ± 2.99 | 40.25 ± 2.63 | n.s. | n.s. |
| LDL-c (mg/dL) | 133.41 ± 15.86 | 123.60 ± 7.99 | p < 0.05 | 136.98 ± 7.06 | 127.56 ± 7.19 | P < 0.05 | n.s. |
| TG (mg/dL) | 237.81 ± 20.26 | 197.27 ± 6.1 | p < 0.05 | 230.79 ± 25.66 | 185.54 ± 9.64 | P < 0.05 | p < 0.05 |
| Urea (mg/dL) | 33.91 ± 7.18 | 35.22 ± 6.35 | n.s. | 35.18 ± 6.58 | 36.33 ± 7.35 | n.s. | n.s. |
| Uric acid (mg/dL) | 4.78 ± 1.03 | 4.89 ± 0.90 | n.s. | 5.05 ± 1.22 | 4.93 ± 0.78 | n.s. | n.s. |
| Creatinine (mg/dL) | 0.78 ± 0.15 | 0.76 ± 0.13 | n.s. | 0.75 ± 0.17 | 0.73 ± 0.15 | n.s. | n.s. |
3. Discussion
4. Materials and Methods
4.1. Subjects
4.2. Experimental Design and Randomization
4.3. Diet Protocol
| KD Group | KDO3 Group | |
|---|---|---|
| Kcal/day | 1187 ± 89 | 1218 ± 105 |
| Protein (% total daily Kcal) | 43.4 ± 3.2 | 43.2 ± 4 |
| Fat (% total daily Kcal) | 45.8 ± 4 | 53.7 ± 5.2 |
| Carbohydrate (% total daily Kcal) | 10.8 ± 2.1 | 10.5 ± 1.8 |
| Protein(g/day) | 128.8 ± 10 | 131.6 ± 8.2 |
| Fat(g/day) | 60.5 ± 8.9 | 62.6 ± 7.6 |
| Carbohydrates(g/day) | 32 ± 1.8 | 32 ± 2.1 |
| Plant Extracts | mL/day | Composition |
|---|---|---|
| Extracts A, mL/day | 20 | Durvillea antarctica, black radish, mint, liquorice, artichoke, horsetail, burdock, dandelion, rhubarb, gentian, lemon balm, chinaroot, juniper, spear grass, elder, fucus, anise, parsley, bearberry, horehound |
| Extracts B, mL/day | 20 | Serenoa, red clover, chervil, bean, elder, dandelion, uncaria, equisetum, horehound, rosemary |
| Extracts C, mL/day | 50 | Horsetail, asparagus, birch, cypress, couch grass, corn, dandelion, grape, fennel, elder, rosehip, anise |
| Extracts D, mL/day (only weeks 1 and 2) | 40 | Eleuthero, eurycoma longifolia, ginseng, corn, miura puama, grape, guaranà, arabic coffee, ginger |
4.4. Measurements
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Abbreviations
| IL-1β | interleukin 1 beta |
| IL-1Ra | interleukin-1 receptor antagonist |
| IL-10 | interleukin 10 |
| IL-6 | interleukin 6 |
| TNF-α | tumor necrosis factor alpha |
Conflicts of Interest
References
- Bueno, N.B.; de Melo, I.S.; de Oliveira, S.L.; da Rocha Ataide, T. Very-low-carbohydrate ketogenic diet v. Low-fat diet for long-term weight loss: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2013, 110, 1178–1187. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A. Ketogenic diet for obesity: Friend or foe? Int. J. Environ. Res. Public Health 2014, 11, 2092–2107. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Bianco, A.; Damiani, E.; Bosco, G. Ketogenic diet in neuromuscular and neurodegenerative diseases. BioMed Res. Int. 2014, 2014, 474296. [Google Scholar] [CrossRef] [PubMed]
- Hussain, T.A.; Mathew, T.C.; Dashti, A.A.; Asfar, S.; Al-Zaid, N.; Dashti, H.M. Effect of low-calorie versus low-carbohydrate ketogenic diet in type 2 diabetes. Nutrition (Burbank, Los Angeles County, Calif.) 2012, 28, 1016–1021. [Google Scholar] [CrossRef]
- Paoli, A.; Rubini, A.; Volek, J.S.; Grimaldi, K.A. Beyond weight loss: A review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur. J. Clin. Nutr. 2013, 67, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Cross, J.H.; McLellan, A.; Neal, E.G.; Philip, S.; Williams, E.; Williams, R.E. The ketogenic diet in childhood epilepsy: Where are we now? Arch. Dis. Child. 2010, 95, 550–553. [Google Scholar] [CrossRef] [PubMed]
- Veggiotti, P.; de Giorgis, V. Dietary treatments and new therapeutic perspective in glut1 deficiency syndrome. Curr. Treat. Options Neurol. 2014, 16, 291. [Google Scholar] [CrossRef] [PubMed]
- Kinzig, K.P.; Honors, M.A.; Hargrave, S.L. Insulin sensitivity and glucose tolerance are altered by maintenance on a ketogenic diet. Endocrinology 2010, 151, 3105–3114. [Google Scholar] [CrossRef] [PubMed]
- Krebs, H.A. The regulation of the release of ketone bodies by the liver. Adv. Enzyme Regul. 1966, 4, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.M.; Al-Zaid, N.S.; Mathew, T.C.; Al-Mousawi, M.; Talib, H.; Asfar, S.K.; Behbahani, A.I. Long term effects of ketogenic diet in obese subjects with high cholesterol level. Mol. Cell. Biochem. 2006, 286, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Dashti, H.M.; Mathew, T.C.; Hussein, T.; Asfar, S.K.; Behbahani, A.; Khoursheed, M.A.; Al-Sayer, H.M.; Bo-Abbas, Y.Y.; Al-Zaid, N.S. Long-term effects of a ketogenic diet in obese patients. Exp. Clin. Cardiol. 2004, 9, 200–205. [Google Scholar] [PubMed]
- Sharman, M.J.; Kraemer, W.J.; Love, D.M.; Avery, N.G.; Gomez, A.L.; Scheett, T.P.; Volek, J.S. A ketogenic diet favorably affects serum biomarkers for cardiovascular disease in normal-weight men. J. Nutr. 2002, 132, 1879–1885. [Google Scholar] [PubMed]
- Westman, E.C.; Yancy, W.S., Jr.; Olsen, M.K.; Dudley, T.; Guyton, J.R. Effect of a low-carbohydrate, ketogenic diet program compared to a low-fat diet on fasting lipoprotein subclasses. Int. J. Cardiol. 2006, 110, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Yancy, W.S., Jr.; Olsen, M.K.; Guyton, J.R.; Bakst, R.P.; Westman, E.C. A low-carbohydrate, ketogenic diet versus a low-fat diet to treat obesity and hyperlipidemia: A randomized, controlled trial. Ann. Intern. Med. 2004, 140, 769–777. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Cenci, L.; Grimaldi, K.A. Effect of ketogenic mediterranean diet with phytoextracts and low carbohydrates/high-protein meals on weight, cardiovascular risk factors, body composition and diet compliance in italian council employees. Nutr. J. 2011, 10, 112. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Cenci, L.; Fancelli, M.; Parmagnani, A.; Fratter, A.; Cucchi, A.; Bianco, A. Ketogenic diet and phytoextracts comparison of the efficacy of mediterranean, zone and tisanoreica diet on some health risk factors. Agro Food Ind. Hi Tech 2010, 21, 24–29. [Google Scholar]
- Volek, J.S.; Sharman, M.J.; Forsythe, C.E. Modification of lipoproteins by very low-carbohydrate diets. J. Nutr. 2005, 135, 1339–1342. [Google Scholar] [PubMed]
- Lofgren, I.; Zern, T.; Herron, K.; West, K.; Sharman, M.J.; Volek, J.S.; Shachter, N.S.; Koo, S.I.; Fernandez, M.L. Weight loss associated with reduced intake of carbohydrate reduces the atherogenicity of ldl in premenopausal women. Metabolism 2005, 54, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Kromhout, D.; Bosschieter, E.B.; de Lezenne Coulander, C. The inverse relation between fish consumption and 20-year mortality from coronary heart disease. N. Engl. J. Med. 1985, 312, 1205–1209. [Google Scholar] [CrossRef]
- Rizos, E.C.; Ntzani, E.E.; Bika, E.; Kostapanos, M.S.; Elisaf, M.S. Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: A systematic review and meta-analysis. JAMA 2012, 308, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Harris, W.S.; Appel, L.J.; American Heart Association; Nutrition, C. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation 2002, 106, 2747–2757. [Google Scholar] [CrossRef]
- Sun, R.; Wang, X.; Liu, Y.; Xia, M. Dietary supplementation with fish oil alters the expression levels of proteins governing mitochondrial dynamics and prevents high-fat diet-induced endothelial dysfunction. Br. J. Nutr. 2014, 112, 145–153. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wainwright, C.L.; Michel, L. Endocannabinoid system as a potential mechanism for n-3 long-chain polyunsaturated fatty acid mediated cardiovascular protection. Proc. Nutr. Soc. 2013, 72, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Abeywardena, M.Y.; Patten, G.S. Role of omega3 long-chain polyunsaturated fatty acids in reducing cardio-metabolic risk factors. Endocr. Metab. Immune Disord. Drug Targets 2011, 11, 232–246. [Google Scholar] [CrossRef] [PubMed]
- Shearer, G.C.; Savinova, O.V.; Harris, W.S. Fish oil—How does it reduce plasma triglycerides? Biochim. Biophys. Acta 2012, 1821, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Bulchandani, D. Why do omega-3 fatty acids lower serum triglycerides? Curr. Opin. Lipidol. 2006, 17, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Taha, A.Y.; McIntyre Burnham, W. Commentary on the effects of a ketogenic diet enriched with omega-3 polyunsaturated fatty acids on plasma phospholipid fatty acid profile in children with drug-resistant epilepsy. Epilepsy Res. 2007, 76, 148–149. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, P.; Davidson, N.C.; Schmidt, E.B.; Calder, P.C. Cardiovascular effects of marine omega-3 fatty acids. Lancet 2010, 376, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Miyagawa, N.; Miura, K.; Okuda, N.; Kadowaki, T.; Takashima, N.; Nagasawa, S.Y.; Nakamura, Y.; Matsumura, Y.; Hozawa, A.; Fujiyoshi, A.; et al. Long-chain n-3 polyunsaturated fatty acids intake and cardiovascular disease mortality risk in japanese: A 24-year follow-up of nippon data 80. Atherosclerosis 2014, 232, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Aarsetoey, H.; Grundt, H.; Nygaard, O.; Nilsen, D.W. The role of long-chained marine n-3 polyunsaturated fatty acids in cardiovascular disease. Cardiol. Res. Pract. 2012, 2012. [Google Scholar] [CrossRef]
- Bernstein, A.M.; Ding, E.L.; Willett, W.C.; Rimm, E.B. A meta-analysis shows that docosahexaenoic acid from algal oil reduces serum triglycerides and increases HDL-cholesterol and ldl-cholesterol in persons without coronary heart disease. J. Nutr. 2012, 142, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Bianco, A.; Grimaldi, K.A.; Lodi, A.; Bosco, G. Long term successful weight loss with a combination biphasic ketogenic mediterranean diet and mediterranean diet maintenance protocol. Nutrients 2013, 5, 5205–5217. [Google Scholar] [CrossRef] [PubMed]
- Siliprandi, N.; Tettamanti, G. Biochimica Medica: Strutturale, Metabolica e Funzionale; Piccin: Padova, Italy, 2005. [Google Scholar]
- Qi, K.; Fan, C.; Jiang, J.; Zhu, H.; Jiao, H.; Meng, Q.; Deckelbaum, R.J. Omega-3 fatty acid containing diets decrease plasma triglyceride concentrations in mice by reducing endogenous triglyceride synthesis and enhancing the blood clearance of triglyceride-rich particles. Clin. Nutr. 2008, 27, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Cresci, S. The ppar genes, cardiovascular disease and the emergence of ppar pharmacogenetics. Expert Opin. Pharmacother. 2005, 6, 2577–2591. [Google Scholar] [CrossRef] [PubMed]
- Caron-Dorval, D.; Paquet, P.; Paradis, A.M.; Rudkowska, I.; Lemieux, S.; Couture, P.; Vohl, M.C. Effect of the ppar-alpha l162v polymorphism on the cardiovascular disease risk factor in response to n-3 polyunsaturated fatty acids. J. Nutr. Nutr. 2008, 1, 205–212. [Google Scholar] [CrossRef]
- Griffin, B.A. How relevant is the ratio of dietary n-6 to n-3 polyunsaturated fatty acids to cardiovascular disease risk? Evidence from the optilip study. Curr. Opin. Lipidol. 2008, 19, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Rossmeisl, M.; Medrikova, D.; van Schothorst, E.M.; Pavlisova, J.; Kuda, O.; Hensler, M.; Bardova, K.; Flachs, P.; Stankova, B.; Vecka, M.; et al. Omega-3 phospholipids from fish suppress hepatic steatosis by integrated inhibition of biosynthetic pathways in dietary obese mice. Biochim. Biophys. Acta 2014, 1841, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Jeong, E.A.; Jeon, B.T.; Shin, H.J.; Kim, N.; Lee, D.H.; Kim, H.J.; Kang, S.S.; Cho, G.J.; Choi, W.S.; Roh, G.S. Ketogenic diet-induced peroxisome proliferator-activated receptor-gamma activation decreases neuroinflammation in the mouse hippocampus after kainic acid-induced seizures. Exp. Neurol. 2011, 232, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Ruskin, D.N.; Kawamura, M.; Masino, S.A. Reduced pain and inflammation in juvenile and adult rats fed a ketogenic diet. PLoS One 2009, 4, e8349. [Google Scholar] [CrossRef] [PubMed]
- Forsythe, C.E.; Phinney, S.D.; Fernandez, M.L.; Quann, E.E.; Wood, R.J.; Bibus, D.M.; Kraemer, W.J.; Feinman, R.D.; Volek, J.S. Comparison of low fat and low carbohydrate diets on circulating fatty acid composition and markers of inflammation. Lipids 2008, 43, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Longo, V.D.; Mattson, M.P. Fasting: Molecular mechanisms and clinical applications. Cell Metab. 2014, 19, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Teng, K.T.; Chang, C.Y.; Chang, L.F.; Nesaretnam, K. Modulation of obesity-induced inflammation by dietary fats: Mechanisms and clinical evidence. Nutr. J. 2014, 13. [Google Scholar] [CrossRef]
- Labonte, M.E.; Dewailly, E.; Lucas, M.; Couture, P.; Lamarche, B. Association of red blood cell n-3 polyunsaturated fatty acids with plasma inflammatory biomarkers among the quebec cree population. Eur. J. Clin. Nutr. 2014, 68, 1042–1047. [Google Scholar] [CrossRef] [PubMed]
- Flock, M.R.; Skulas-Ray, A.C.; Harris, W.S.; Gaugler, T.L.; Fleming, J.A.; Kris-Etherton, P.M. Effects of supplemental long-chain omega-3 fatty acids and erythrocyte membrane fatty acid content on circulating inflammatory markers in a randomized controlled trial of healthy adults. Prostaglandins Leukot. Essent. Fatty Acids 2014, 91, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Lund, A.S.; Hasselbalch, A.L.; Gamborg, M.; Skogstrand, K.; Hougaard, D.M.; Heitmann, B.L.; Kyvik, K.O.; Sorensen, T.I.; Jess, T. N-3 polyunsaturated fatty acids, body fat and inflammation. Obes. Facts 2013, 6, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Tishinsky, J.M. Modulation of adipokines by n-3 polyunsaturated fatty acids and ensuing changes in skeletal muscle metabolic response and inflammation. Appl. Physiol. Nutr. Metab. 2013, 38, 361. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Monk, J.M.; Hou, T.Y.; Turk, H.F.; Weeks, B.; Wu, C.; McMurray, D.N.; Chapkin, R.S. Dietary n-3 polyunsaturated fatty acids (PUFA) decrease obesity-associated th17 cell-mediated inflammation during colitis. PLoS One 2012, 7, e49739. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.Y.; Talukdar, S.; Bae, E.J.; Imamura, T.; Morinaga, H.; Fan, W.; Li, P.; Lu, W.J.; Watkins, S.M.; Olefsky, J.M. Gpr120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell 2010, 142, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Ye, J.; Gao, Z.; Youn, H.S.; Lee, W.H.; Zhao, L.; Sizemore, N.; Hwang, D.H. Reciprocal modulation of toll-like receptor-4 signaling pathways involving myd88 and phosphatidylinositol 3-kinase/akt by saturated and polyunsaturated fatty acids. J. Biol. Chem. 2003, 278, 37041–37051. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Zhang, Y.; Dusting, G.J. Nadph oxidase-mediated redox signaling: Roles in cellular stress response, stress tolerance, and tissue repair. Pharmacol. Rev. 2011, 63, 218–242. [Google Scholar] [CrossRef] [PubMed]
- Nakahira, K.; Kim, H.P.; Geng, X.H.; Nakao, A.; Wang, X.; Murase, N.; Drain, P.F.; Wang, X.; Sasidhar, M.; Nabel, E.G.; et al. Carbon monoxide differentially inhibits tlr signaling pathways by regulating ros-induced trafficking of tlrs to lipid rafts. J. Exp. Med. 2006, 203, 2377–2389. [Google Scholar] [CrossRef] [PubMed]
- Taouis, M.; Dagou, C.; Ster, C.; Durand, G.; Pinault, M.; Delarue, J. N-3 polyunsaturated fatty acids prevent the defect of insulin receptor signaling in muscle. Am. J. Physiol. Endocrinol. Metab. 2002, 282, E664–E671. [Google Scholar] [PubMed]
- Wu, J.H.; Cahill, L.E.; Mozaffarian, D. Effect of fish oil on circulating adiponectin: A systematic review and meta-analysis of randomized controlled trials. J. Clin. Endocrinol. Metab. 2013, 98, 2451–2459. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Kamon, J.; Minokoshi, Y.; Ito, Y.; Waki, H.; Uchida, S.; Yamashita, S.; Noda, M.; Kita, S.; Ueki, K.; et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating amp-activated protein kinase. Nat. Med. 2002, 8, 1288–1295. [Google Scholar] [CrossRef] [PubMed]
- Burri, L.; Berge, K.; Wibrand, K.; Berge, R.K.; Barger, J.L. Differential effects of krill oil and fish oil on the hepatic transcriptome in mice. Front. Genet. 2011, 2, 45. [Google Scholar] [CrossRef] [PubMed]
- Turconi, G.; Bazzano, R.; Roggi, C.; Cena, H. Reliability and relative validity of a quantitative food-frequency questionnaire for use among adults in italian population. Int. J. Food Sci. Nutr. 2010, 61, 846–862. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, A.; Pastori, G.; Codognotto, M.; Paoli, A. Equivalence of information from single frequency v. Bioimpedance spectroscopy in bodybuilders. Br. J. Nutr. 2007, 97, 182–192. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paoli, A.; Moro, T.; Bosco, G.; Bianco, A.; Grimaldi, K.A.; Camporesi, E.; Mangar, D. Effects of n-3 Polyunsaturated Fatty Acids (ω-3) Supplementation on Some Cardiovascular Risk Factors with a Ketogenic Mediterranean Diet. Mar. Drugs 2015, 13, 996-1009. https://doi.org/10.3390/md13020996
Paoli A, Moro T, Bosco G, Bianco A, Grimaldi KA, Camporesi E, Mangar D. Effects of n-3 Polyunsaturated Fatty Acids (ω-3) Supplementation on Some Cardiovascular Risk Factors with a Ketogenic Mediterranean Diet. Marine Drugs. 2015; 13(2):996-1009. https://doi.org/10.3390/md13020996
Chicago/Turabian StylePaoli, Antonio, Tatiana Moro, Gerardo Bosco, Antonino Bianco, Keith A. Grimaldi, Enrico Camporesi, and Devanand Mangar. 2015. "Effects of n-3 Polyunsaturated Fatty Acids (ω-3) Supplementation on Some Cardiovascular Risk Factors with a Ketogenic Mediterranean Diet" Marine Drugs 13, no. 2: 996-1009. https://doi.org/10.3390/md13020996
APA StylePaoli, A., Moro, T., Bosco, G., Bianco, A., Grimaldi, K. A., Camporesi, E., & Mangar, D. (2015). Effects of n-3 Polyunsaturated Fatty Acids (ω-3) Supplementation on Some Cardiovascular Risk Factors with a Ketogenic Mediterranean Diet. Marine Drugs, 13(2), 996-1009. https://doi.org/10.3390/md13020996