Immunostimulative Activity of Low Molecular Weight Chitosans in RAW264.7 Macrophages
Abstract
:1. Introduction
2. Results
2.1. Effects of LMWCs on the Cell Viability of RAW264.7 Macrophage
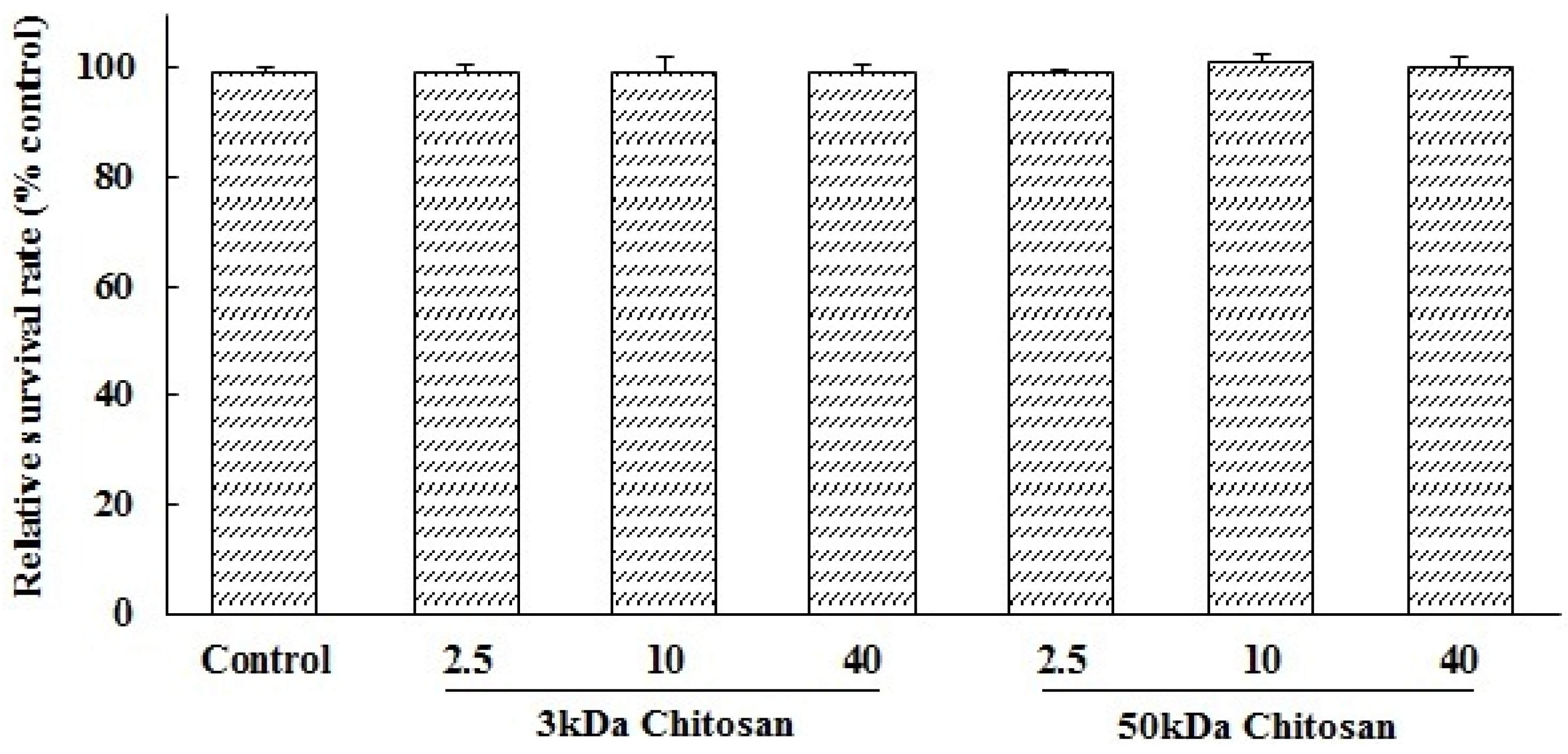
2.2. Effects of LMWCs on Pinocytic Activity
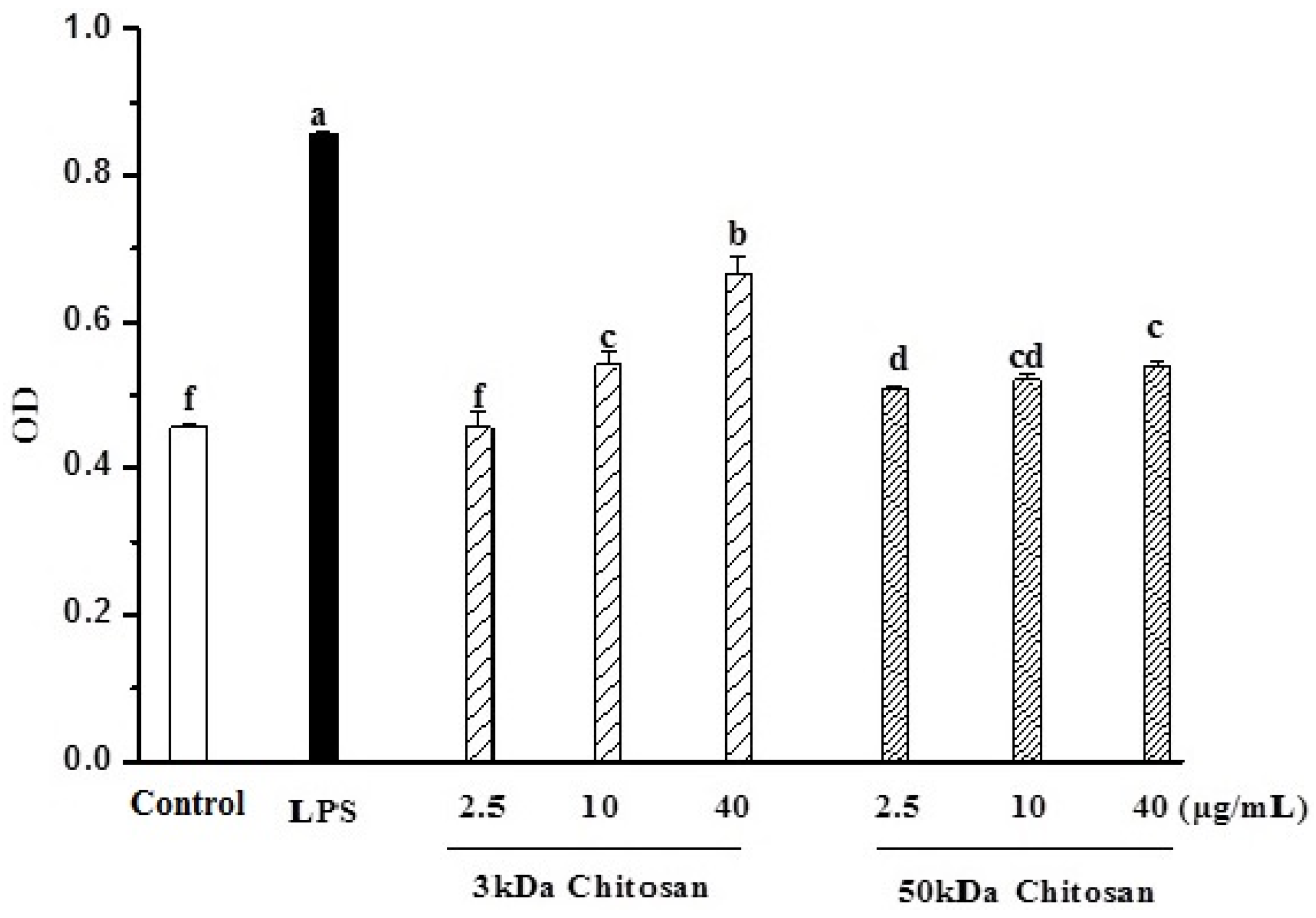
2.3. Effect of LMWCs on Macrophage Cytokines Production
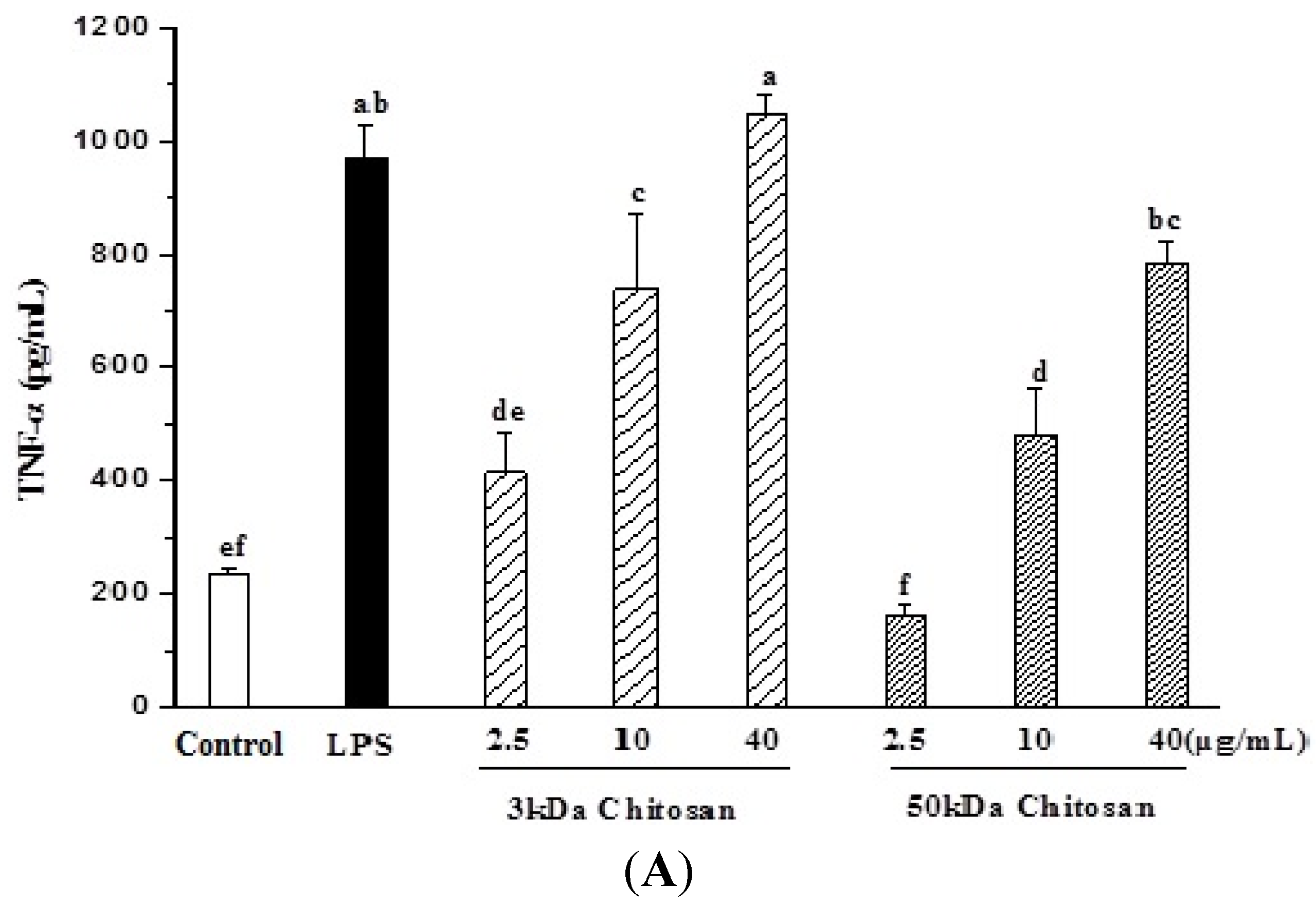
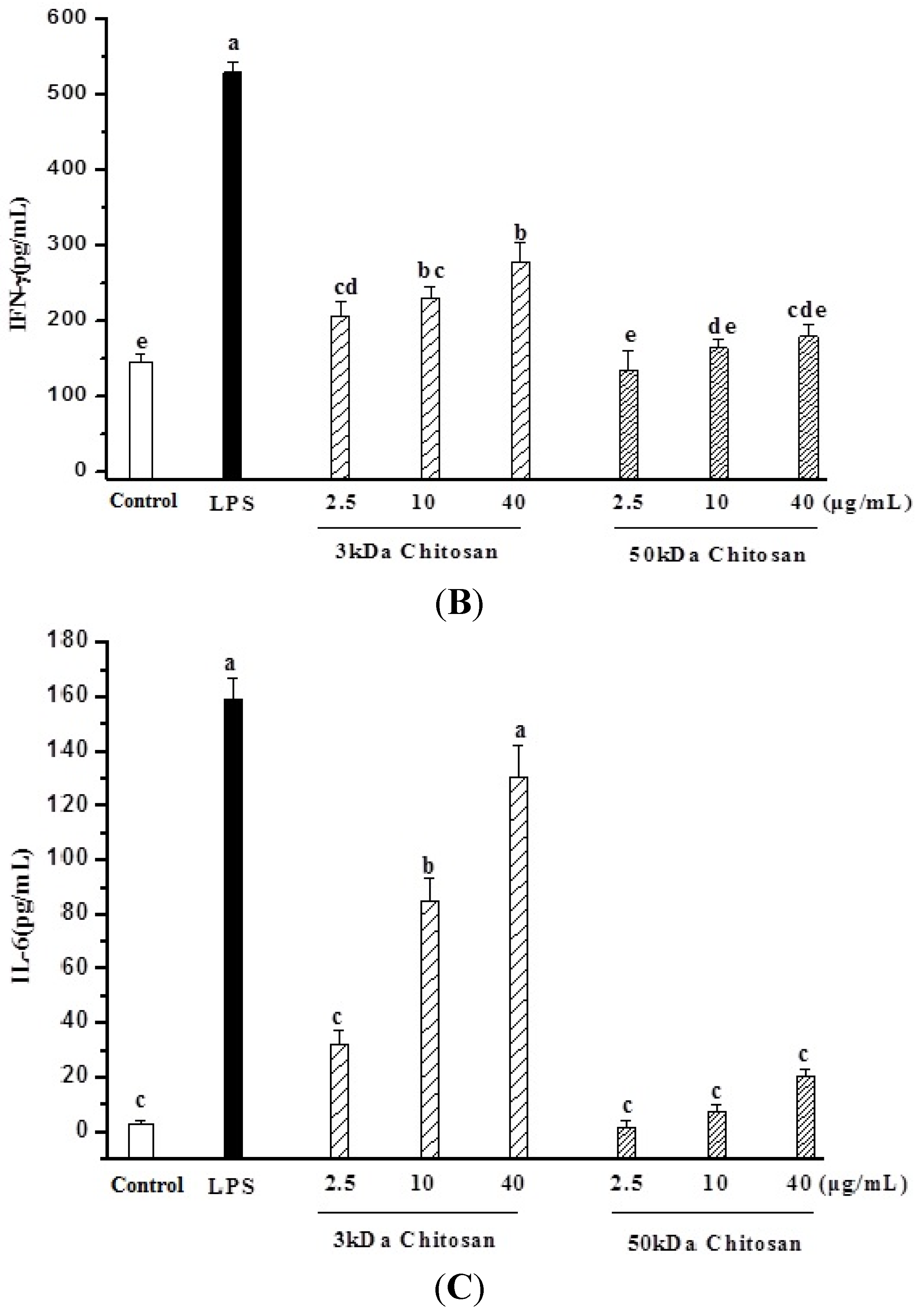
2.4. Effect of LMWCs on Nitric Oxide (NO) Production and Activities of Inducible Nitric Oxide Synthase (iNOS) in RAW264.7 Macrophage
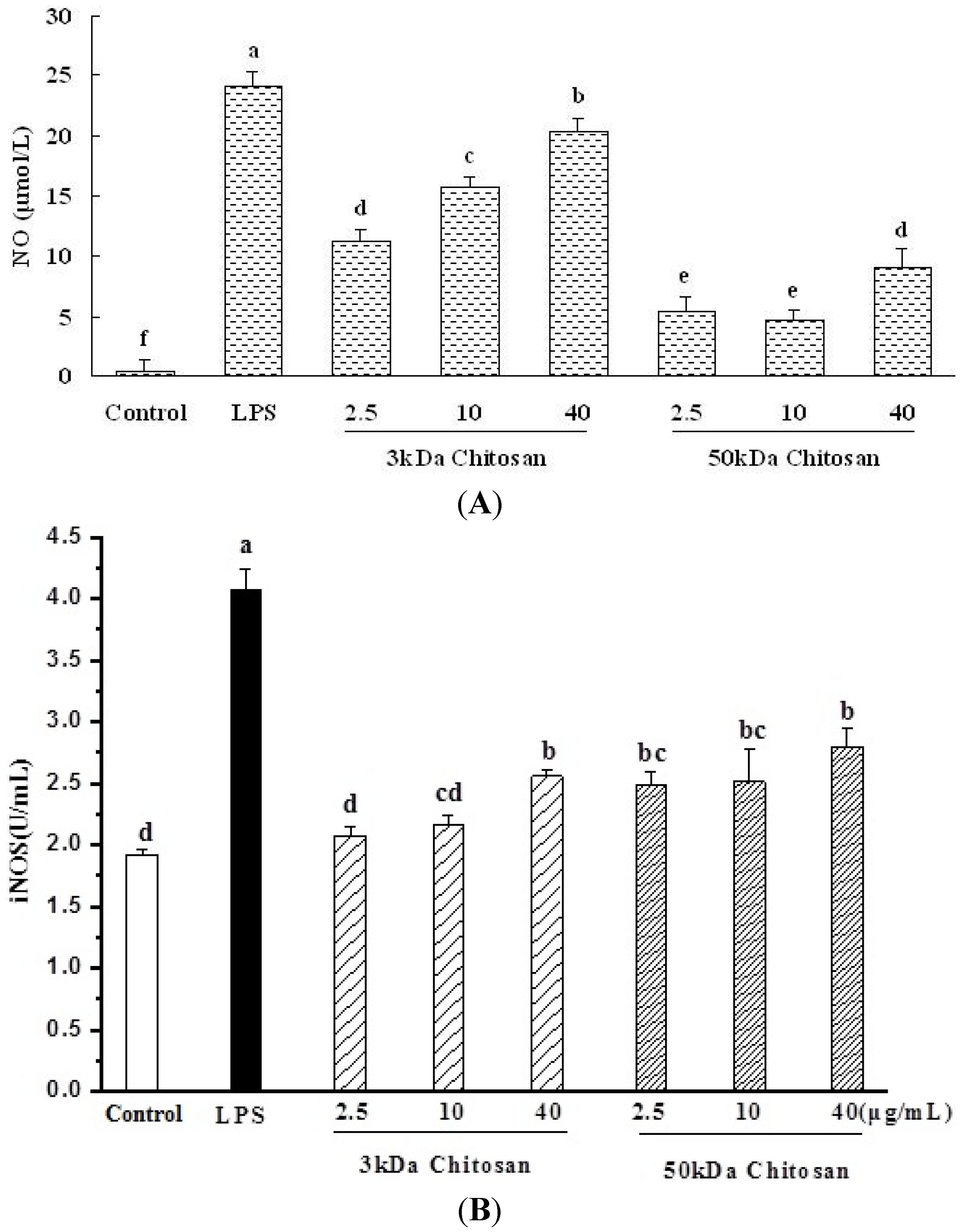
2.5. Effect of LMWCs on the mRNA Expression Levels of TNF-α and iNOS in RAW264.7 Macrophage
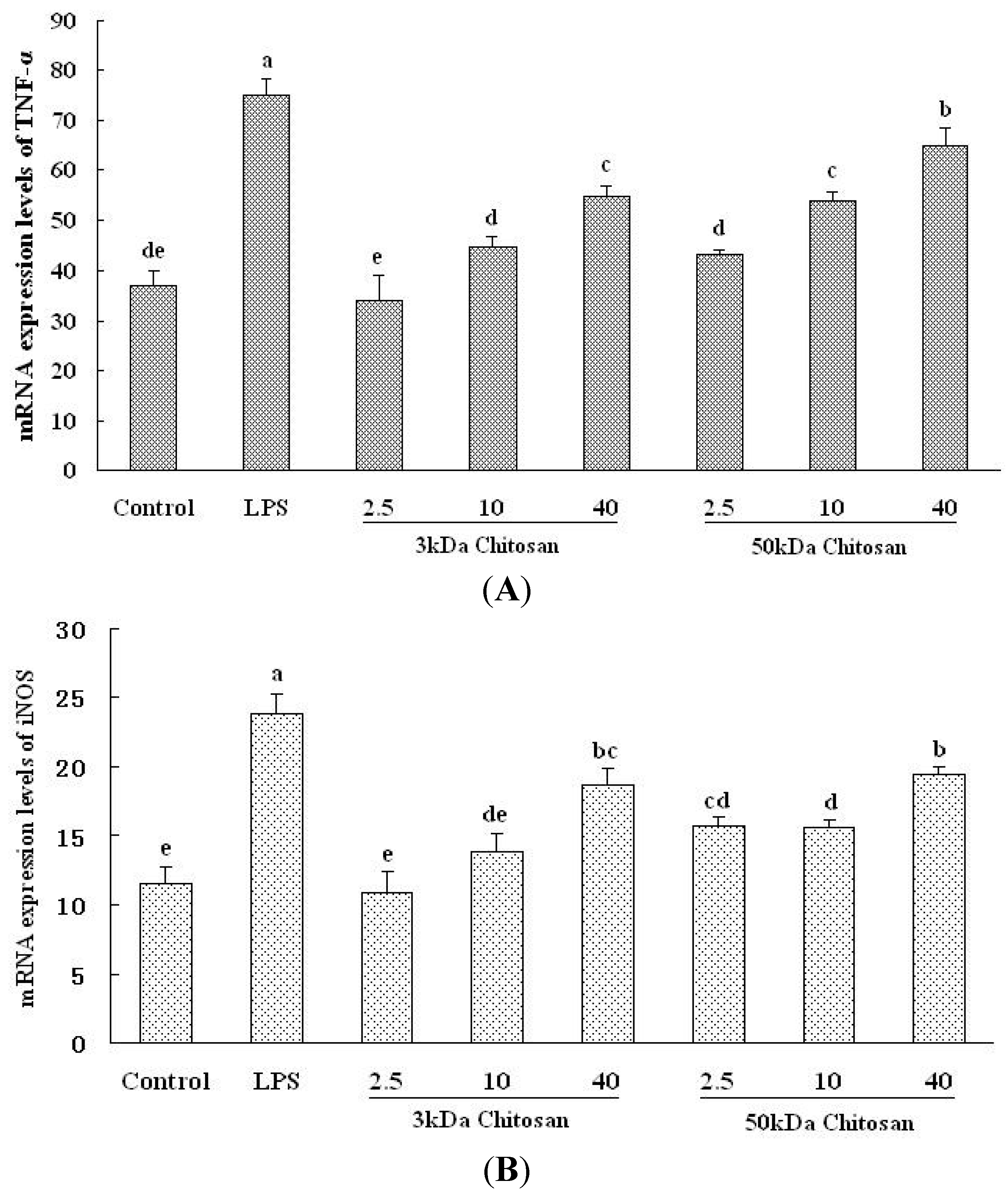
3. Discussion
4. Experimental Section
4.1. Chemicals and Reagents
4.2. Cell Culture and Treatment
4.3. Cell Viability Assay of RAW264.7 Macrophages
4.4. Pinocytic Activity Assay
4.5. Preparation of Cell Lysates
4.6. Measurement of Cytokine Levels in RAW264.7 Macrophage Cultures Using an Enzyme Linked Immunosorbent Assay (ELISA)
4.7. Measurement of Nitric Oxide (NO) Release and Intracellular Contents of iNOS
4.8. Measurement of the mRNA Expression Levels of iNOS and TNF-α by Real-Time PCR
| Gene | Genbank Accession | Primer Sequence | Product Size (bp) | Annealing (°C) |
|---|---|---|---|---|
| TNF-α | NM_013693 | CGGTGCCTATGTCTCAGCCTCTT | 88 | 64 |
| GACCGATCACCCCGAAGTTCAGTA | ||||
| iNOS | NM_010927.3 | TGCCACGGACGAGACGGATA | 141 | 64 |
| AGGAAGGCAGCGGGCACAT | ||||
| 18s | NR_003278 | CGGACACGGACAGGATTGACA | 94 | 64 |
| CCAGACAAATCGCTCCACCAACTA |
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Guan, D.; Zhang, Z.; Yang, Y.; Xing, G.; Liu, J. Immunomodulatory Activity of Polysaccharide from the Roots of Actinidia kolomikta on Macrophages. Int. J. Biol. 2011, 3. [Google Scholar] [CrossRef]
- De Oliveira, C.C.; De Oliveira, S.M.; Godoy, L.M.; Gabardo, J.; Buchi, D.D.F. Canova, Brazilian medical formulation, alters oxidative metabolism of mice macrophages. J. Infect. 2006, 52, 420–432. [Google Scholar] [CrossRef] [PubMed]
- Schepetkin, I.A.; Xie, G.; Kirpotina, L.N.; Klein, R.A.; Jutila, M.A.; Quinn, M.T. Macrophage immunomodulatory activity of polysaccharides isolated from Opuntia polyacantha. Int. Immunopharmacol. 2008, 8, 1455–1466. [Google Scholar] [CrossRef] [PubMed]
- Schepetkin, I.A.; Quinn, M.T. Botanical polysaccharides: Macrophage immunomodulation and therapeutic potential. Int. Immunopharmacol. 2006, 6, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.W.; Wan, F.C.; Wang, J.Q.; Jin, Z.Y.; Xu, X.M. Macrophage immunomodulatory activity of polysaccharides isolated from Glycyrrhiza uralensis Fish. Int. Immunopharmacol. 2008, 8, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Zaharoff, D.A.; Rogers, C.J.; Hance, K.W.; Schlom, J.; Greiner, J.W. Chitosan solution enhances both humoral and cell-mediated immune responses to subcutaneous vaccination. Vaccine 2007, 25, 2085–2094. [Google Scholar] [CrossRef] [PubMed]
- Read, R.C.; Naylor, S.C.; Potter, C.W.; Bond, J.; Jabbal-Gill, I.; Fisher, A.; Illum, L.; Jennings, R. Effective nasal influenza vaccine delivery using chitosan. Vaccine 2005, 23, 4367–4374. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.H.; Cosgrove, C.; McNeela, E.A.; Sexton, A.; Giemza, R.; Jabbal-Gill, I.; Church, A.; Lin, W.; Illum, L.; Podda, A.; et al. Protective levels of diphtheria-neutralizing antibody induced in healthy volunteers by unilateral priming-boosting intranasal immunization associated with restricted ipsilateral mucosal secretory immunoglobulin A. Infect. Immun. 2003, 71, 726–732. [Google Scholar] [CrossRef] [PubMed]
- McNeela, E.A.; Jabbal-Gill, I.; Illum, L.; Pizza, M.; Rappuoli, R.; Podda, A.; Lewis, D.J.; Mills, K.H. Intranasal immunization with genetically detoxified diphtheria toxin induces T cell responses in humans: Enhancement of Th2 responses and toxin-neutralizing antibodies by formulation with chitosan. Vaccine 2004, 22, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.S.; Xu, Y.L.; Zou, X.T.; Xu, Z.R. Chitosan Nanoparticles Act as an Adjuvant to Promote both Th1 and Th2 Immune Responses Induced by Ovalbumin in Mice. Mar. Drugs 2011, 9, 1038–1055. [Google Scholar] [CrossRef] [PubMed]
- Ngo, D.H.; Vo, T.S.; Ngo, D.N.; Kang, K.H.; Je, J.Y.; Pham, H.N.D.; Byun, H.G.; Kim, S.K. Biological effects of chitosan and its derivatives. Food Hydrocoll. 2015, 51, 200–216. [Google Scholar] [CrossRef]
- Park, B.K.; Kim, M.M. Applications of Chitin and Its Derivatives in Biological Medicine. Int. J. Mol. Sci. 2010, 11, 5152–5164. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.C.; Spindola, H.; de Sousa, V.; Santos-Silva, A.; Pintado, M.E.; Malcata, F.X.; Carvalho, J.E. Anti-inflammatory activity of chitooligosaccharides in vivo. Mar. Drugs 2010, 8, 1763–1768. [Google Scholar] [CrossRef] [PubMed]
- Ju, C.X.; Yue, W.; Yang, Z.H.; Zhang, Q.; Yang, X.; Liu, Z.; Zhang, F. Antidiabetic effect and mechanism of chitooligosaccharides. Biol. Pharm. Bull. 2010, 33, 1511–1516. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.J.; Youn, D.K.; Lee, S.H.; No, H.K.; Ha, J.G.; Prinyawiwatkul, W. Antibacterial activity of chitosans with different degrees of deacetylation and viscosities. Int. J. Food Sci. Technol. 2010, 45, 676–682. [Google Scholar] [CrossRef]
- Artan, M.; Karadeniz, F.; Karagozlu, M.Z.; Kim, M.M.; Kim, S.K. Anti-HIV-1 activity of low molecular weight sulfated chitooligosaccharides. Carbohydr. Res. 2010, 345, 656–662. [Google Scholar] [CrossRef] [PubMed]
- Ngo, D.N.; Lee, S.H.; Kim, M.; Kim, S.K. Production of chitin-oligosaccharides with different molecular weights and their antioxidant effect in RAW264.7 cells. J. Funct. Foods 2009, 1, 188–198. [Google Scholar] [CrossRef]
- Quan, H.; Zhu, F.; Han, X.; Xu, Z.; Zhao, Y.; Miao, Z. Mechanism of anti-angiogenic activities of chitooligosaccharides may be through inhibiting heparanase activity. Med. Hypotheses 2009, 73, 205–206. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Gong, L.; Gu, X.; Ding, F. Chitooligosaccharides promote peripheral nerve regeneration in a rabbit common peroneal nerve crush injury model. Microsurgery 2009, 29, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Da Silva, C.A.; Lee, J.Y.; Hartl, D.; Elias, J.A. Chitin regulation of immune responses: An old molecule with new roles. Curr. Opin. Immunol. 2008, 20, 684–689. [Google Scholar] [CrossRef]
- Li, X.; Min, M.; Du, N.; Gu, Y.; Hode, T.; Naylor, M.; Chen, D.; Nordquist, R.E.; Chen, W.R. Chitin, Chitosan, and Glycated Chitosan Regulate Immune Responses: The Novel Adjuvants for Cancer Vaccine. Clin. Dev. Immunol. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Ueno, H.; Nakamura, F.; Murakami, M.; Okumura, M.; Kadosawa, T.; Fujinag, T. Evaluation effects of chitosan for the extracellular matrix production by fibroblasts and the growth factors production by macrophages. Biomaterials 2001, 22, 2125–2130. [Google Scholar] [CrossRef]
- Sun, H.; Zhang, J.; Chen, F.; Chen, X.; Zhou, Z.; Wang, H. Activation of RAW264.7 macrophages by the polysaccharide from the roots of Actinidia eriantha and its molecular mechanisms. Carbohydr. Polym. 2015, 121, 388–402. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, A.; Tsuji, S.; Yamashita, M.; Henriksen, R.A.; Myrvik, Q.N.; Shibata, Y. Phagocytosis of N-acetyl-d-glucosamine particles, a Th1 adjuvant, by RAW 264.7 cells results in MAPK activation and TNF-α, but not IL-10, production. Cell. Immunol. 2006, 239, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Zhao, L.; Ke, H. Potential role of nuclear factor-κB in the induction of nitric oxide and tumor necrosis factor-α by oligochitosan in macrophages. Int. Immunopharmacol. 2004, 4, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Reese, T.A.; Liang, H.R.; Tager, A.M.; Luster, A.D.; van Rooijen, N.; Voehringer, D.; Locksley, R.M. Chitin induces accumulation in tissue of innate immune cells associated with allergy. Nature 2007, 447, 92–96. [Google Scholar] [PubMed]
- Chen, C.L.; Wang, Y.M.; Liu, C.F.; Wang, J.Y. The effect of water-soluble chitosan on macrophage activation and the attenuation of mite allergen-induced airway inflammation. Biomaterials 2008, 29, 2173–2182. [Google Scholar] [CrossRef] [PubMed]
- Shibata, Y.; Metzger, W.J.; Myrvik, Q.N. Chitin particle-induced cell-mediated immunity is inhibited by soluble mannan: Mannose receptor-mediated phagocytosis initiates IL-12 production. J. Immunol. 1997, 159, 2462–2467. [Google Scholar] [PubMed]
- Lechner, M.; Lirk, P.; Rieder, J. Inducible nitric oxide synthase (iNOS) in tumor biology: The two sides of the same coin. Semin. Cancer Biol. 2005, 15, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Druhan, L.J.; Zweier, J.L. Reactive oxygen and nitrogen species regulate inducible nitric oxide synthase function shifting the balance of nitric oxide and superoxide production. Arch. Bioch. Biophys. 2010, 494, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.J.; Tsai, G.J. Chitooligosaccharides in combination with interferon-gamma increase nitric oxide production via nuclear factor-kappaB activation in murine RAW264.7 macrophages. Food Chem. Toxicol. 2007, 45, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Peluso, G.; Petillo, O.; Ranieri, M.; Santin, M.; Ambrosio, L.; Calabro, D.; Avallone, B.; Balsamo, G. Chitosan-mediated stimulation of macrophage function. Biomaterials 1994, 15, 1215–1220. [Google Scholar] [CrossRef]
- Porporatto, C.; Bianco, I.D.; Riera, C.M.; Correa, S.G. Chitosan induces different l-arginine metabolic pathways in resting and inflammatory macrophages. Biochem. Biophys. Res. Commun. 2003, 304, 266–272. [Google Scholar] [CrossRef]
- Jeong, H.J.; Koo, H.N.; Oh, E.Y.; Chae, H.J.; Kim, H.R.; Suh, S.B.; Kim, C.H.; Cho, K.H.; Park, B.R.; Park, S.T.; et al. Nitric oxide production by high molecular weight water-soluble chitosan via nuclear factor-κB activation. Int. J. Immunopharmacol. 2000, 22, 923–933. [Google Scholar] [CrossRef]
- Zhang, J.; Xia, W.; Liu, P.; Cheng, Q.; Tahirou, T.; Gu, W.; Li, B. Chitosan modification and pharmaceutical/biomedical applications. Mar. Drugs 2010, 8, 1962–1987. [Google Scholar] [PubMed]
- Beutler, B. Innate immunity: An overview. Mol. Immunol. 2004, 40, 845–859. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wang, L.; Zhou, Y. Immunomodulation and antitumor activities of different-molecular-weight polysaccharides from Porphyridium cruentum. Carbohydr. Polym. 2012, 87, 1206–1210. [Google Scholar] [CrossRef]
- Cho, M.; Yang, C.; Kim, S.M.; You, S. Molecular characterization and biological activities of water soluble sulfated polysaccharides from Enteromorpha prolifera. Food Sci. Biotechnol. 2010, 19, 525–533. [Google Scholar] [CrossRef]
- Zhang, L.; Li, X.L.; Xu, X.J.; Zeng, F.B. Correlation between antitumor activity, molecular weight, and conformation of lentinan. Carbohydr. Res. 2005, 340, 1515–1521. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.J.; Wu, C.H.; Tsai, G.J. Chitooligosaccharides from the shrimp chitosan hydrolysate induces differentiation of murine RAW264.7 macrophages into dendritic-like cells. J. Funct. Foods 2015, 12, 70–79. [Google Scholar] [CrossRef]
- Zhou, G.F.; Sun, Y.P.; Xin, H.; Zhang, Y.N.; Li, Z.E.; Xu, Z.H. In vivo antitumor and immunomodulation activities of different molecular weight λ-carrageenans from Chondrus ocellatus. Pharmacol. Res. 2004, 50, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Lai, S.L.; Pan, Z.Y.; Li, X.F. Study on the degradability of chitosan under microwave irradiation. J. Shanxi Univ. Sci. Technol. 2005, 23, 38–40. [Google Scholar]
- Zheng, T.S.; Wang, Y.N.; Zong, A.P. Spectrophotometry of chitosan with Bromocresol Green. J. China Pharm. Univ. 2005, 36, 543–545. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔCt Method. Methods 2001, 25, 402–408. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, N.; Wen, Z.-S.; Xiang, X.-W.; Huang, Y.-N.; Gao, Y.; Qu, Y.-L. Immunostimulative Activity of Low Molecular Weight Chitosans in RAW264.7 Macrophages. Mar. Drugs 2015, 13, 6210-6225. https://doi.org/10.3390/md13106210
Wu N, Wen Z-S, Xiang X-W, Huang Y-N, Gao Y, Qu Y-L. Immunostimulative Activity of Low Molecular Weight Chitosans in RAW264.7 Macrophages. Marine Drugs. 2015; 13(10):6210-6225. https://doi.org/10.3390/md13106210
Chicago/Turabian StyleWu, Ning, Zheng-Shun Wen, Xing-Wei Xiang, Yan-Na Huang, Yang Gao, and You-Le Qu. 2015. "Immunostimulative Activity of Low Molecular Weight Chitosans in RAW264.7 Macrophages" Marine Drugs 13, no. 10: 6210-6225. https://doi.org/10.3390/md13106210
APA StyleWu, N., Wen, Z.-S., Xiang, X.-W., Huang, Y.-N., Gao, Y., & Qu, Y.-L. (2015). Immunostimulative Activity of Low Molecular Weight Chitosans in RAW264.7 Macrophages. Marine Drugs, 13(10), 6210-6225. https://doi.org/10.3390/md13106210





