Violapyrones H and I, New Cytotoxic Compounds Isolated from Streptomyces sp. Associated with the Marine Starfish Acanthaster planci
Abstract
:1. Introduction

2. Results and Discussion
2.1. Isolation of Compounds
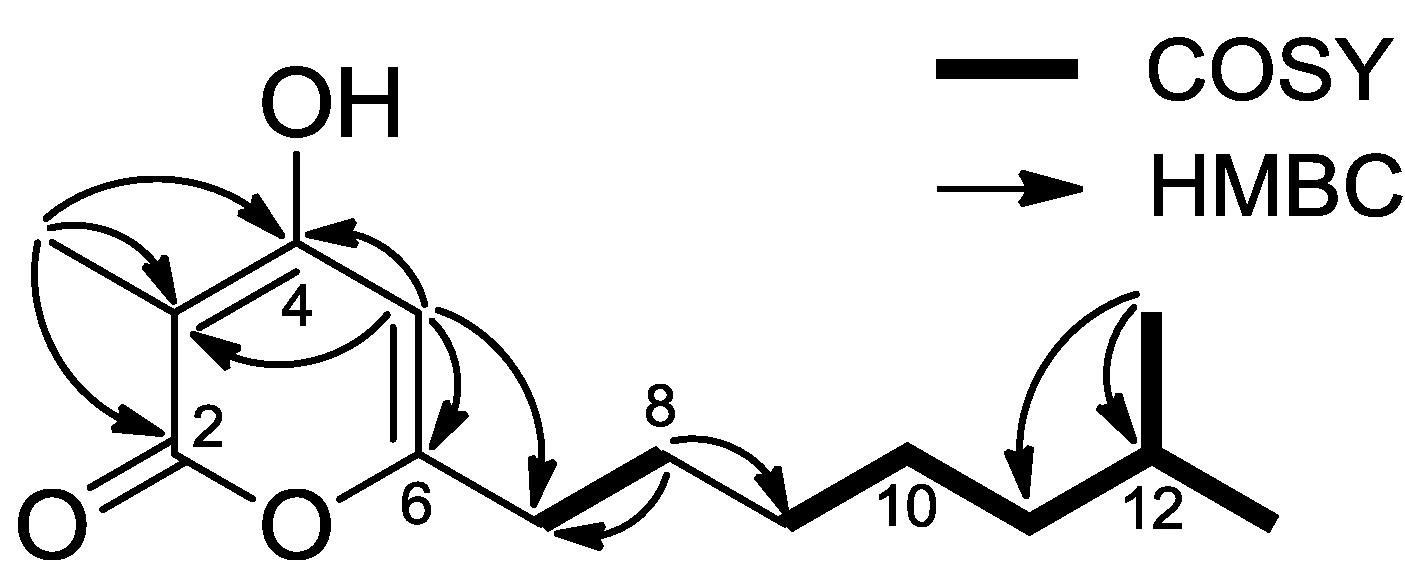
2.2. Structure Determination
| Position | 1 | 2 | ||
|---|---|---|---|---|
| δC, Type | δH, Mult. (J in Hz) | δC, Type | δH, Mult. (J in Hz) | |
| 2 | 169.9, C | 169.4, C | ||
| 3 | 98.7, C | 98.9, C | ||
| 4 | 169.5, C | 168.9, C | ||
| 5 | 102.0, CH | 5.96, s | 101.6, CH | 5.97, s |
| 6 | 164.8, C | 164.9, C | ||
| 7 | 34.4, CH2 | 2.46, t (7.5) | 34.4, CH2 | 2.46, t (7.5) |
| 8 | 28.1, CH2 | 1.64, m | 28.1, CH2 | 1.64, m |
| 9 | 30.4, CH2 | 1.34 | 30.3, CH2 | 1.33 |
| 10 | 28.3, CH2 | 1.34 | 30.2, CH2 | 1.35 |
| 11 | 40.1, CH2 | 1.19, m | 33.0, CH2 | 1.30, m |
| 12 | 29.7, CH | 1.53, m | 23.8, CH2 | 1.31, m |
| 13 | 23.1, CH3 (× 2) | 0.88, d (6.5) | 14.5, CH3 | 0.90, t (6.5) |
| 3-Me | 8.4, CH3 | 1.84, s | 8.4, CH3 | 1.85, s |
2.3. Cytotoxic Properties
| Cell Lines | GI50a (μg/mL) | ||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ADR b | |
| Cervical cancer: HeLa | 25.05 | 5.54 | 18.12 | 9.91 | 0.09 |
| Renal cancer: ACHN | 1.79 | 5.42 | 1.18 | 1.55 | 0.04 |
| Colon cancer: HCT-15 | 1.10 | 3.38 | 2.01 | 5.22 | 0.08 |
| Colon cancer: HCT-116 | 8.99 | 18.08 | 15.83 | 26.12 | 0.09 |
| Breast cancer: MDA-MB-231 | 1.51 | 6.29 | 1.80 | 4.94 | 0.99 |
| Lung cancer: NCI-H23 | 1.24 | 3.47 | 1.90 | 3.24 | 0.04 |
| Lung cancer: NCI-H460 | 4.45 | 21.04 | 6.37 | 10.80 | 0.07 |
| Stomach cancer: NUGC-3 | 1.27 | 3.36 | 2.24 | 4.02 | 0.12 |
| Liver cancer: Hep-G2 | 2.30 | 14.60 | 2.04 | 3.96 | 0.08 |
| Prostate cancer: PC-3 | 1.37 | 5.44 | 1.40 | 2.06 | 0.06 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Isolation and Identification of the Strain 112CH148
3.3. Seed and Mass Cultures of the Strain
3.4. Extraction and Isolation of Compounds
3.5. Cytotoxicity Test by SRB Assay
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Burgess, J.G.; Jordan, E.M.; Bregu, M.; Mearns-Spragg, A.; Boyd, K.G. Microbial antagonism: A neglected avenue of natural products research. J. Biotehcnol. 1999, 70, 27–32. [Google Scholar]
- Fenical, W. Chemical studies of marine bacteria: Developing a new resource. Chem. Rev. 1993, 93, 1673–1683. [Google Scholar]
- Lan, W.J.; Zhao, Y.; Xie, Z.L.; Liang, L.Z.; Shao, W.Y.; Zhu, L.P.; Yang, D.P.; Zhu, X.F.; Li, H.J. Novel sorbicillin analogues from the Marine fungus Trichoderma sp. associated with the seastar Acanthaster planci. Nat. Prod. Commun. 2012, 7, 1337–1340. [Google Scholar]
- Xie, Z.L.; Li, H.J.; Wang, L.Y.; Liang, W.L.; Liu, W.; Lan, W.J. Trichodermaerin, a new diterpenoid lactone from the marine fungus Trichoderma erinaceum associated with the sea star Acanthaster planci. Nat. Prod. Commun. 2013, 8, 67–68. [Google Scholar]
- Zhao, Y.; Li, S.-Q.; Li, H.-J.; Lan, W.-J. Lanostane triterpenoids from the fungus Ceriporia lacerate associated with Acanthaster planci. Chem. Nat. Compd. 2013, 49, 653–656. [Google Scholar] [CrossRef]
- Liu, D.; Li, X.M.; Meng, L.; Li, C.S.; Gao, S.S.; Shang, Z.; Proksch, P.; Huang, C.G.; Wang, B.G. Nigerapyrones A–H, alpha-pyrone derivatives from the marine mangrove-derived endophytic fungus Aspergillus niger MA-132. J. Nat. Prod. 2011, 74, 1787–1791. [Google Scholar] [CrossRef]
- Fairlamb, I.J.; Marrison, L.R.; Dickinson, J.M.; Lu, F.J.; Schmidt, J.P. 2-pyrones possessing antimicrobial and cytotoxic activities. Bioorg. Med. Chem. 2004, 12, 4285–4299. [Google Scholar] [CrossRef]
- Yao, T.; Larock, R.C. Synthesis of isocoumarins and alpha-pyrones via electrophilic cyclization. J. Org. Chem. 2003, 68, 5936–5942. [Google Scholar]
- Claydon, N.; Allan, M.; Hanson, J.R.; Avent, A.G. Antifungal alkyl pyrones of Trichoderma harzianum. Trans. Br. Mycol. Soc. 1987, 88, 503–513. [Google Scholar] [CrossRef]
- Simon, A.; Dunlop, R.W.; Ghisalberti, E.L.; Sivasithamparam, K. Trichoderma koningii produces a pyrone compound with antibiotic properties. Soil Biol. Biochem. 1988, 20, 263–264. [Google Scholar] [CrossRef]
- Hirota, A.; Nemoto, A.; Tsuchiya, Y.; Hojo, H.; Abe, N. Isolation of a 2-Pyrone Compound as an Antioxidant from a Fungus and Its New Reaction Product with 1,1-Diphenyl-2-picrylhydrazyl Radical. Biosci. Biotechnol. Biochem. 1999, 63, 418–420. [Google Scholar] [CrossRef]
- Sugiyama, Y.; Oya, A.; Kudo, T.; Hirota, A. Surugapyrone A from Streptomyces coelicoflavus strain USF-6280 as a new DPPH radical-scavenger. J. Antibiot. 2010, 63, 365–369. [Google Scholar] [CrossRef]
- Washida, K.; Abe, N.; Sugiyama, Y.; Hirota, A. Novel DPPH radical scavengers, demethylbisorbibutenolide and trichopyrone, from a fungus. Biosci. Biotechnol. Biochem. 2007, 71, 1052–1057. [Google Scholar] [CrossRef]
- Schlingmann, G.; Milne, L.; Carter, G.T. New α-pyrones produced by fungal culture LL-11G219 function as androgen receptor ligands. Tetrahedron 1998, 54, 13013–13022. [Google Scholar] [CrossRef]
- Hagen, S.E.; Prasad, J.V.N.V.; Boyer, F.E.; Domagala, J.M.; Ellsworth, E.L.; Gajda, C.; Hamilton, H.W.; Markoski, L.J.; Steinbaugh, B.A.; Tait, B.D.; et al. Synthesis of 5,6-dihydro-4-hydroxy-2-pyrones as HIV-1 protease inhibitors: The profound effect of polarity on antiviral activity. J. Med. Chem. 1997, 40, 3707–3711. [Google Scholar] [CrossRef]
- Thaisrivongs, S.; Janakiraman, M.N.; Chong, K.T.; Tomich, P.K.; Dolak, L.A.; Turner, S.R.; Strohbach, J.W.; Lynn, J.C.; Horng, M.M.; Hinshaw, R.R.; et al. Structure-based design of novel HIV protease inhibitors: Sulfonamide-containing 4-hydroxycoumarins and 4-hydroxy-2-pyrones as potent non-peptidic inhibitors. J. Med. Chem. 1996, 39, 2400–2410. [Google Scholar] [CrossRef]
- Shi, X.; Leal, W.S.; Liu, Z.; Schrader, E.; Meinwald, J. A new synthesis of alkylated 2H-pyran-2-ones and its application to the determination of the relative and absolute configuration of supellapyrone, sex pheromone of the brownbanded cockroach, Supella longipalpa. Tetrahedron Lett. 1995, 36, 71–74. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, Y.; Cao, Y.; Liu, J.; Zheng, D.; Chen, X.; Han, L.; Jiang, C.; Huang, X. Violapyrones A–G, alpha-pyrone derivatives from Streptomyces violascens isolated from Hylobates hoolock feces. J. Nat. Prod. 2013, 76, 2126–2130. [Google Scholar] [CrossRef]
- Lee, J.S.; Shin, J.; Shin, H.J.; Lee, H.-S.; Lee, Y.-J.; Lee, H.-S.; Won, H. Total synthesis and configurational validation of (+)-violapyrone C. Eur. J. Org. Chem. 2014. submitted for publication. [Google Scholar]
- Skehan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New Colorimetric Cytotoxicity Assay for Anticancer-Drug Screening. J. Natl. Cancer Inst. 1990, 82, 1107–1112. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shin, H.J.; Lee, H.-S.; Lee, J.S.; Shin, J.; Lee, M.A.; Lee, H.-S.; Lee, Y.-J.; Yun, J.; Kang, J.S. Violapyrones H and I, New Cytotoxic Compounds Isolated from Streptomyces sp. Associated with the Marine Starfish Acanthaster planci. Mar. Drugs 2014, 12, 3283-3291. https://doi.org/10.3390/md12063283
Shin HJ, Lee H-S, Lee JS, Shin J, Lee MA, Lee H-S, Lee Y-J, Yun J, Kang JS. Violapyrones H and I, New Cytotoxic Compounds Isolated from Streptomyces sp. Associated with the Marine Starfish Acanthaster planci. Marine Drugs. 2014; 12(6):3283-3291. https://doi.org/10.3390/md12063283
Chicago/Turabian StyleShin, Hee Jae, Hwa-Sun Lee, Jong Seok Lee, Junho Shin, Min Ah Lee, Hyi-Seung Lee, Yeon-Ju Lee, Jieun Yun, and Jong Soon Kang. 2014. "Violapyrones H and I, New Cytotoxic Compounds Isolated from Streptomyces sp. Associated with the Marine Starfish Acanthaster planci" Marine Drugs 12, no. 6: 3283-3291. https://doi.org/10.3390/md12063283
APA StyleShin, H. J., Lee, H.-S., Lee, J. S., Shin, J., Lee, M. A., Lee, H.-S., Lee, Y.-J., Yun, J., & Kang, J. S. (2014). Violapyrones H and I, New Cytotoxic Compounds Isolated from Streptomyces sp. Associated with the Marine Starfish Acanthaster planci. Marine Drugs, 12(6), 3283-3291. https://doi.org/10.3390/md12063283






