New Prenylxanthones from the Deep-Sea Derived Fungus Emericella sp. SCSIO 05240
Abstract
:1. Introduction
2. Results and Discussion
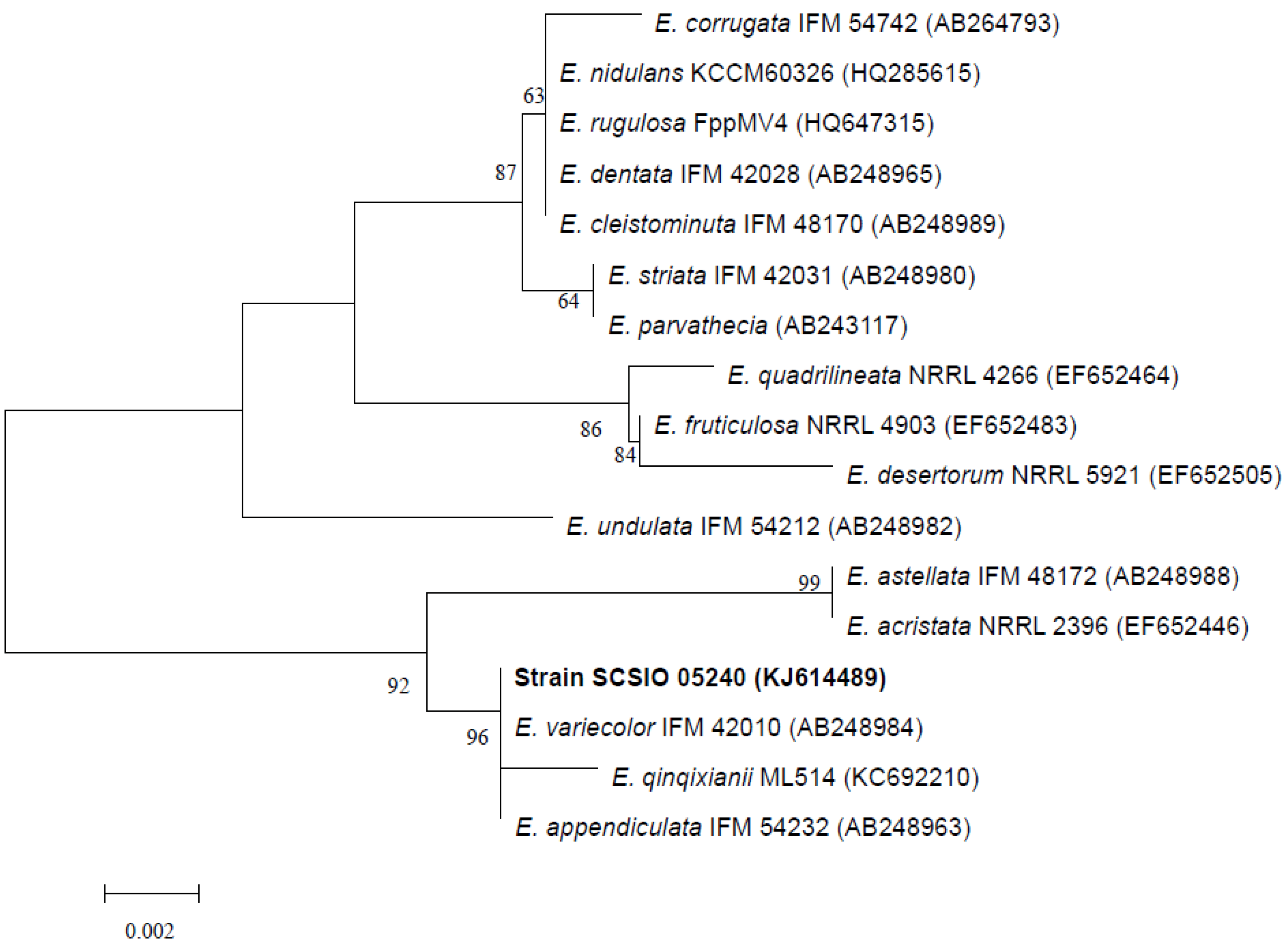
2.1. Identification of the Fungus Strain

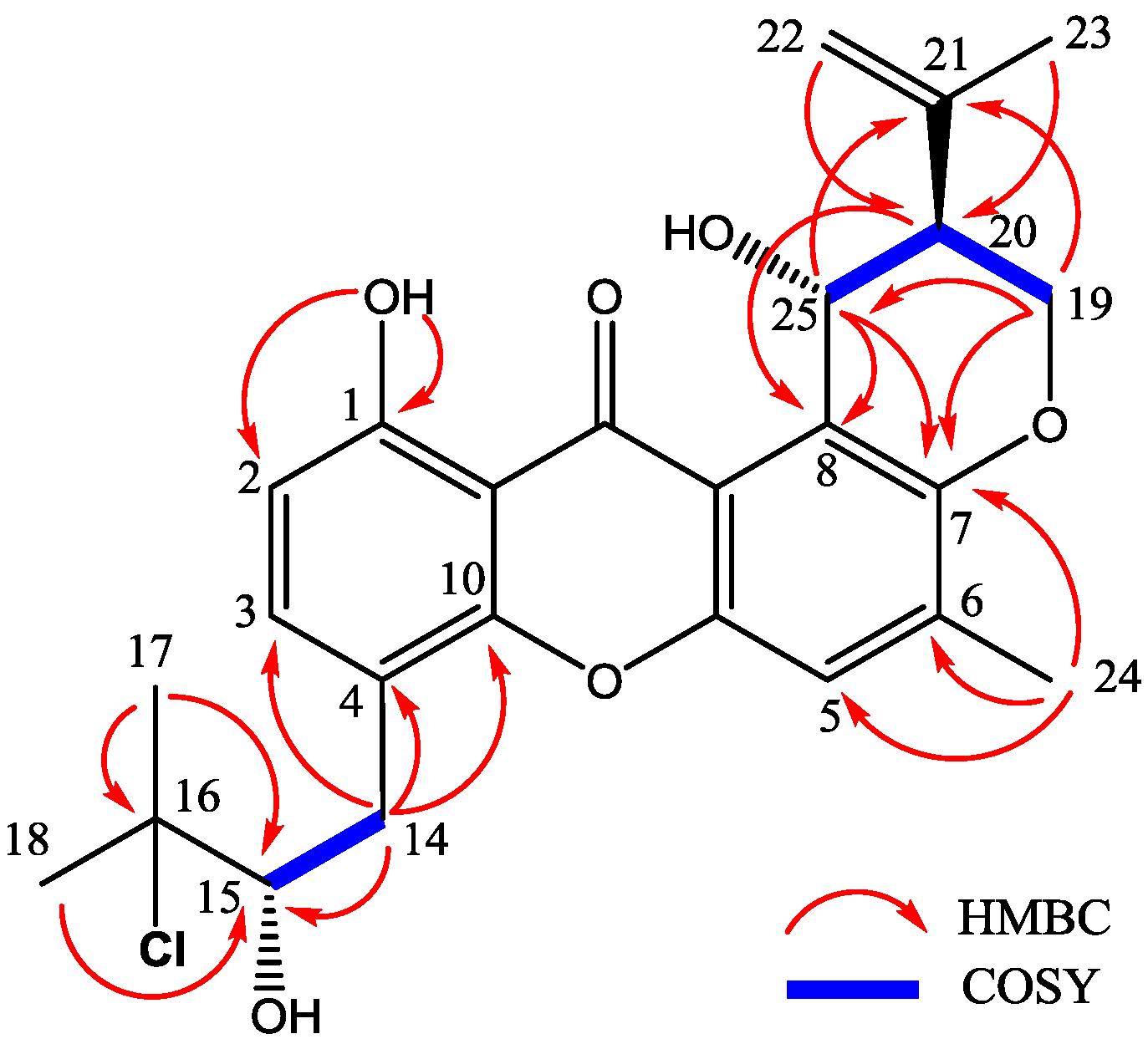
2.2. Structure Elucidation
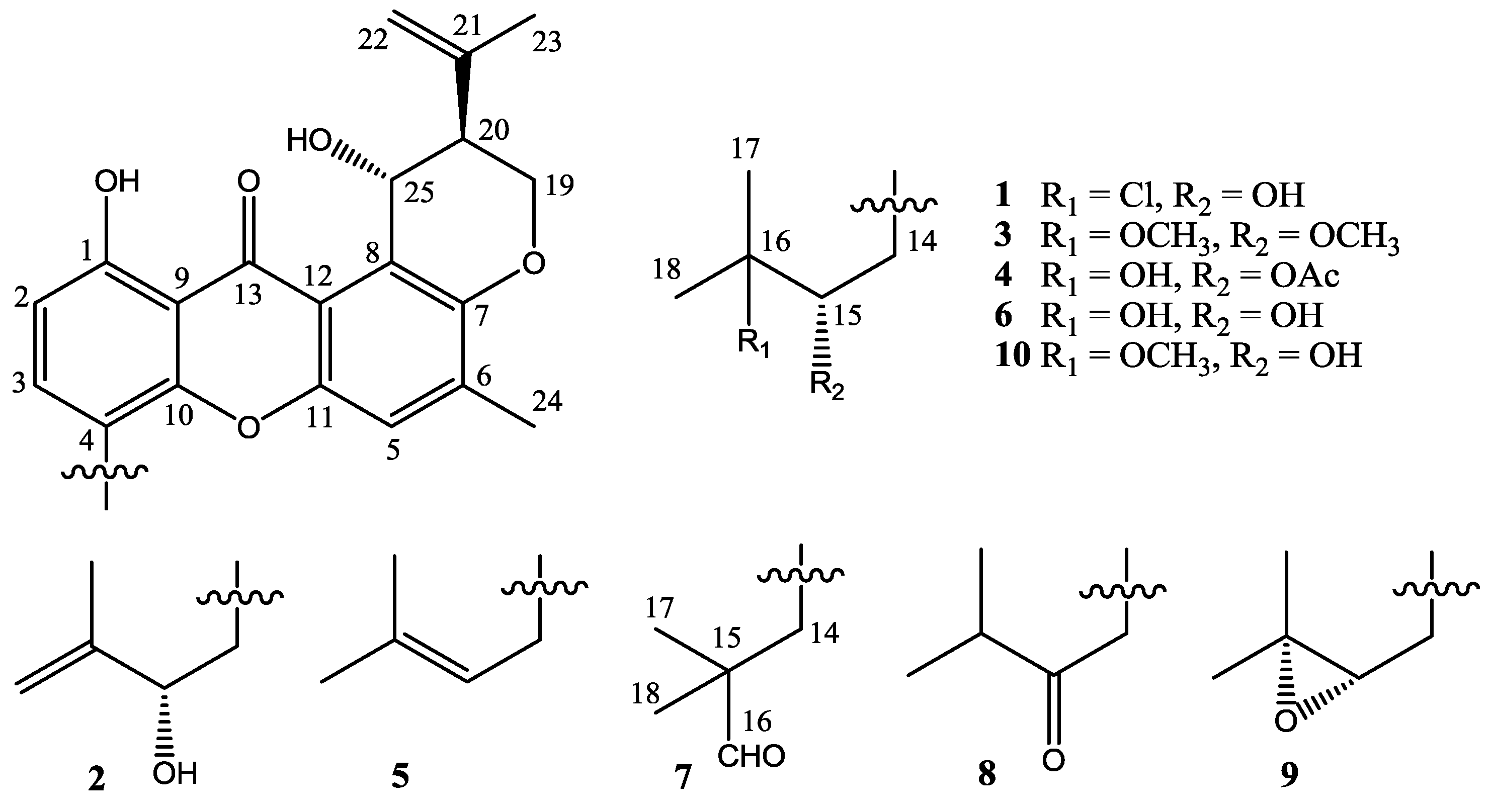
| No. | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 2 | 6.78, d, 8.5 | 6.76, d, 8.5 | 6.78, d, 8.5 | 6.71, d, 8.5 |
| 3 | 7.55, d, 8.5 | 7.51, d, 8.5 | 7.59, d, 8.5 | 7.41, d, 8.5 |
| 5 | 7.22, s | 7.26, s | 7.22, s | 7.25, s |
| 14a | 3.35, brd, 14.0 | 3.18, dd, 14.0, 5.0 | 3.14, brd, 14.0 | 3.34, dd, 14.0, 2.5 |
| 14b | 2.76, dd, 14.0, 10.5 | 2.97, dd, 14.0, 8.5 | 2.72, dd, 14.0,10.0 | 2.91, dd,14.0, 10.5 |
| 15 | 3.84, brd, 10.5 | 4.39, m | 3.78, d, 10.0 | 5.17, dd, 10.5, 7.5 |
| 17 | 1.76, s | 4.84, s,4.91,s | 1.32, s | 1.37, s |
| 18 | 1.73, s | 1.88, s | 1.28, s | 1.33, s |
| 19a | 4.43, dd, 10.5, 2.5 | 4.43, dd, 11.0, 2.5 | 4.43, dd, 11.0, 3.0 | 4.43, dd, 10.5, 3.0 |
| 19b | 4.35, dd, 10.5,2.5 | 4.35, dd, 11.0, 2.5 | 4.36, dd, 11.0, 3.0 | 4.35, dd, 10.5, 3.0 |
| 20 | 2.73, brs | 2.73, brd, 3.0 | 2.73, brd, 2.5 | 2.73, brd, 2.5 |
| 22a | 4.79, s | 4.80, s | 4.80, s | 4.79, s |
| 22b | 4.56, s | 4.58, s | 4.57, s | 4.56, s |
| 23 | 1.84, s | 1.85, s | 1.84, s | 1.86, s |
| 24 | 2.35, s | 2.35, s | 2.35, s | 2.35, s |
| 25 | 5.40, brs | 5.41, brd 2.5 | 5.41, brs | 5.40, brd, 2.5 |
| 15-OCH3 | 3.48, s | |||
| 16-OCH3 | 3.29, s | |||
| OAc | 1.84, s | |||
| OH-1 | 12.63, s | 12.65, s | 12.62, s | 12.63, s |
| OH-25 | 5.0, brs | 5.03, d, 4.0 | 5.05, d, 3.5 | 4.97, d, 3.5 |
| No. | 1 | 2 | 3 | 4 | No. | 1 | 2 | 3 | 4 |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 160.5 s | 160.4 s | 160.2 s | 160.6 s | 16 | 74.9 s | 146.8 s | 76.5 d | 72.3 s |
| 2 | 110.0 d | 109.9 d | 109.9 d | 109.6 d | 17 | 28.9 q | 111.4 t | 22.5 q | 26.9 q |
| 3 | 138.3 d | 138.2 d | 138.2 d | 137.8 d | 18 | 27.9 q | 18.0 q | 20.9 q | 25.2 q |
| 4 | 115.9 s | 115.6 s | 116.8 s | 115.1 s | 19 | 64.6 t | 64.6 t | 64.6 t | 64.5 t |
| 5 | 119.1 d | 119.1 d | 119.1 d | 119.2 d | 20 | 44.9 d | 44.9 d | 44.9 d | 44.9 d |
| 6 | 138.5 s | 138.5 s | 138.3 s | 138.5 s | 21 | 142.5 s | 142.5 s | 142.6 s | 142.6 s |
| 7 | 149.6 s | 149.5 s | 149.5 s | 149.6 s | 22 | 112.3 t | 112.3 t | 112.3 t | 112.2 t |
| 8 | 121.1 s | 121.1 s | 121.1 s | 121.9 s | 23 | 22.5 q | 22.5 q | 19.3 q | 20.7 q |
| 9 | 109.2 s | 109.2 s | 109.2 s | 109.2 s | 24 | 17.4 q | 17.4 q | 17.4 q | 17.4 q |
| 10 | 153.1 s | 153.2 s | 153.0 s | 153.2 s | 25 | 63.2 d | 63.2 d | 63.2 d | 63.1 d |
| 11 | 152.0 s | 152.0 s | 152.1 s | 152.0 s | 15-OCH3 | 29.7 q | |||
| 12 | 116.9 s | 116.8 s | 116.9 s | 116.9 s | 16-OCH3 | 49.3 q | |||
| 13 | 184.4 s | 184.4 s | 184.5 s | 184.4 s | OAc(CO) | 170.3 s | |||
| 14 | 31.9 t | 35.3 t | 31.2 t | 29.6 t | OAc(CH3) | 20.7 q | |||
| 15 | 78.5 d | 75.4 d | 77.6 s | 78.6 d |
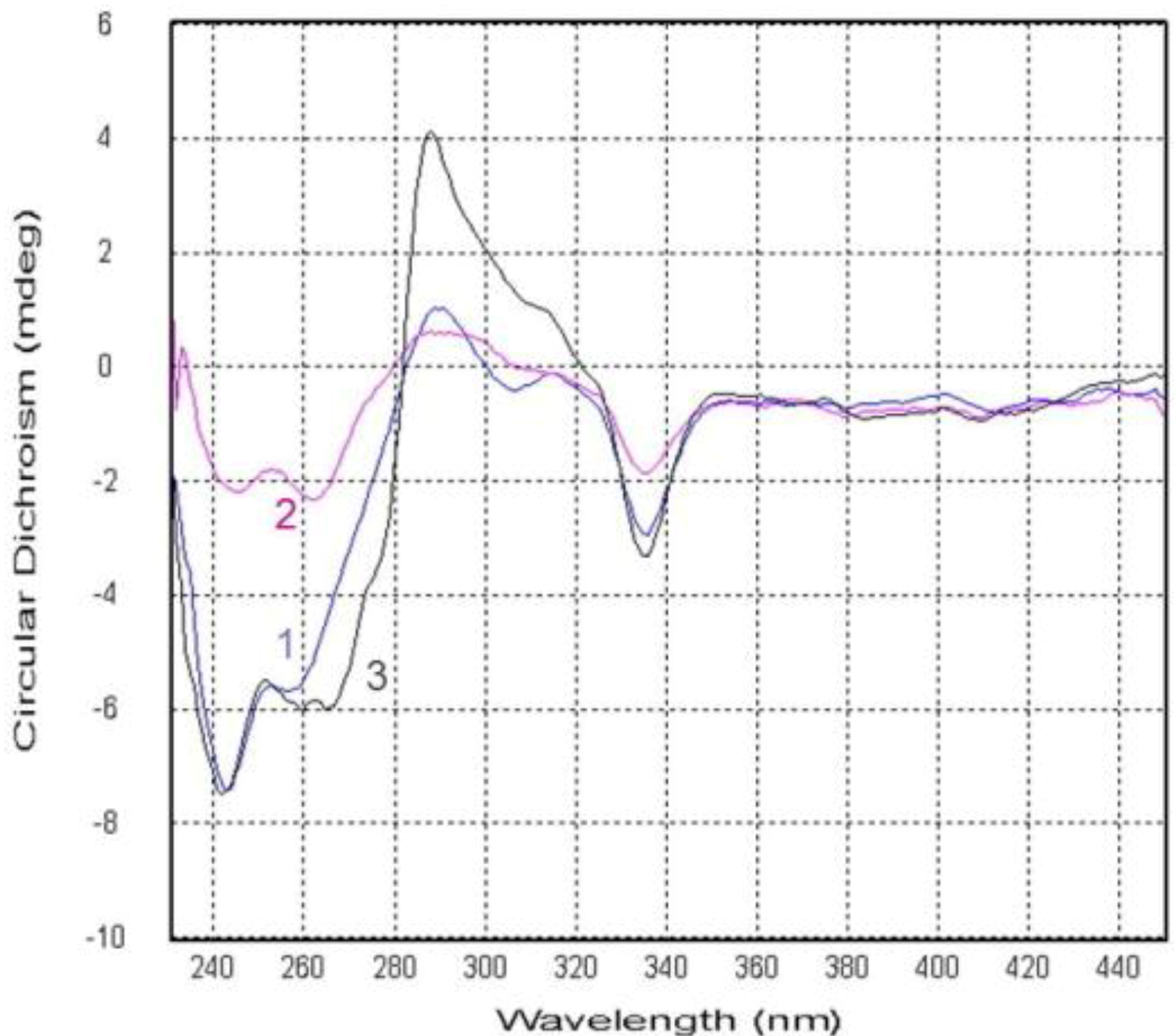
2.3. Biological Activities of the Isolated Compounds
2.4. Discussion
3. Experimental Section
3.1. General
3.2. Fungal Material and Fermentation
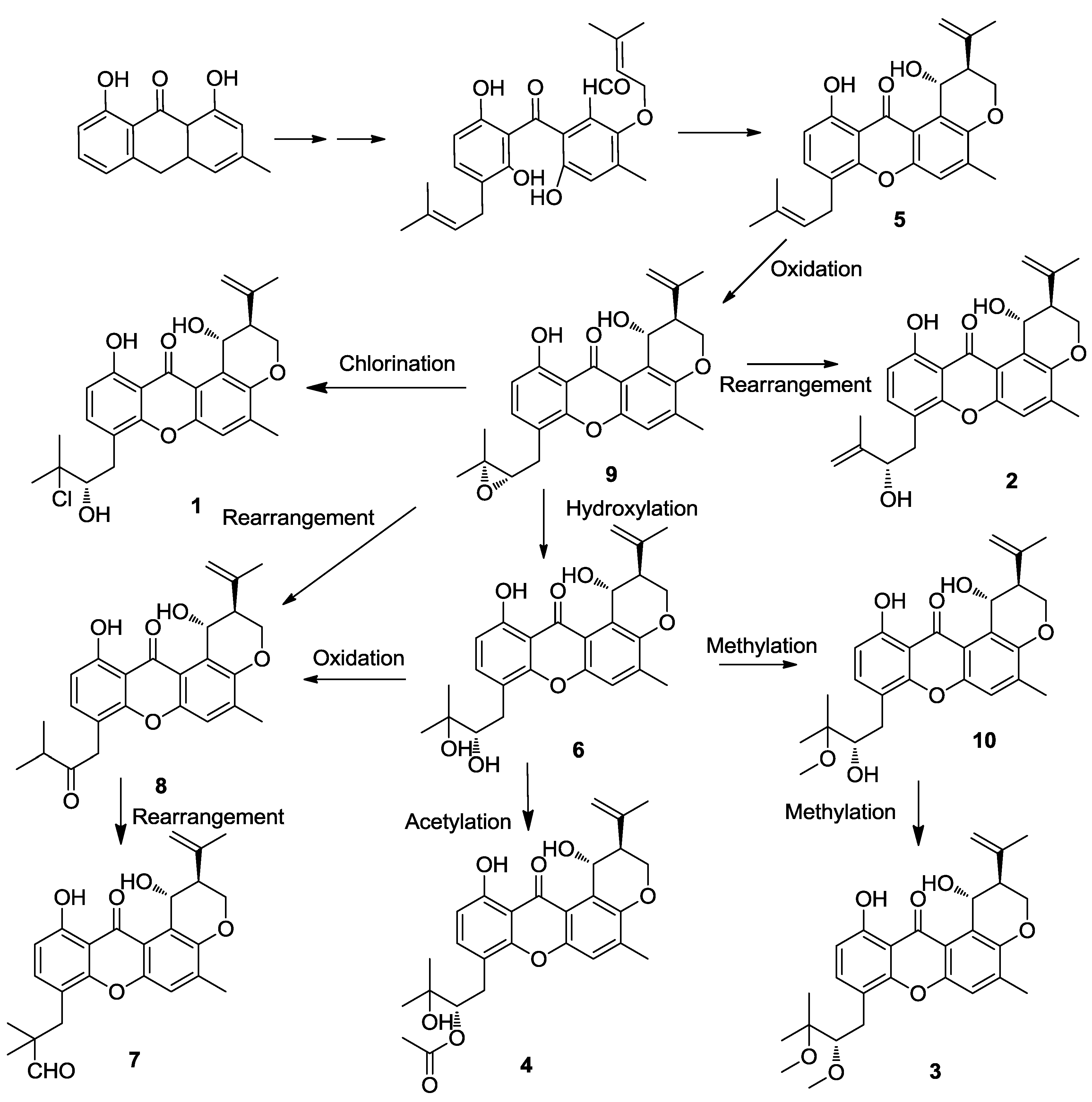
3.3. Extraction and Isolation
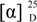 −78.1 (c 0.0031, CHCl3); UV(CHCl3) λmax (log ε) 395 (3.39), 298 (3.63), 294 (3.62), 277 (4.21), 248 (4.04), 228 (4.33) nm; IR νmax 3394, 2978, 2916, 1639, 1573, 1473, 1431, 1354, 1292, 1242, 1049 , 1018, 856 , 821 cm−1; 1H and 13C NMR data see Table 1 and Table 2; HRESIMS m/z 459.1569 [M + H]+ (calcd for C25H28ClO6, 459.1574) (Supplementary Figures S1–S9).
−78.1 (c 0.0031, CHCl3); UV(CHCl3) λmax (log ε) 395 (3.39), 298 (3.63), 294 (3.62), 277 (4.21), 248 (4.04), 228 (4.33) nm; IR νmax 3394, 2978, 2916, 1639, 1573, 1473, 1431, 1354, 1292, 1242, 1049 , 1018, 856 , 821 cm−1; 1H and 13C NMR data see Table 1 and Table 2; HRESIMS m/z 459.1569 [M + H]+ (calcd for C25H28ClO6, 459.1574) (Supplementary Figures S1–S9).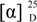 −54.3 (c 0.0065, CHCl3); UV(CHCl3) λmax (log ε) 397 (3.45), 300 (3.63), 292 (3.59), 277 (4.14), 242 (3.88), 228 (3.67) nm; IR νmax 3444, 2954, 1724, 1639, 1604, 1573, 1469, 1413, 1350, 1273, 1195, 1049, 1018, 902, 864 cm−1; 1H and 13C NMR data see Table 1 and Table 2; HRESIMS m/z 423.1804 [M + H]+ (calcd for C25H27O6, 423.1808) (Supplementary Figures S10–S15).
−54.3 (c 0.0065, CHCl3); UV(CHCl3) λmax (log ε) 397 (3.45), 300 (3.63), 292 (3.59), 277 (4.14), 242 (3.88), 228 (3.67) nm; IR νmax 3444, 2954, 1724, 1639, 1604, 1573, 1469, 1413, 1350, 1273, 1195, 1049, 1018, 902, 864 cm−1; 1H and 13C NMR data see Table 1 and Table 2; HRESIMS m/z 423.1804 [M + H]+ (calcd for C25H27O6, 423.1808) (Supplementary Figures S10–S15).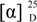 −95.5 (c 0.0018 CHCl3); UV(CHCl3) λmax (log ε) 398 (3.21), 300 (3.41), 291 (3.34), 277 (4.0), 249 (3.16), 228 (4.21) nm; IR νmax 3429, 2920, 1639, 1573, 1473, 1431, 1292, 1149, 1049, 1018, 856 cm−1; 1H and 13C NMR data see Table 1 and Table 2; HRESIMS m/z 469.2217 [M + H]+ (calcd for C27H33O7, 469.2226) (Supplementary Figures S16–S20).
−95.5 (c 0.0018 CHCl3); UV(CHCl3) λmax (log ε) 398 (3.21), 300 (3.41), 291 (3.34), 277 (4.0), 249 (3.16), 228 (4.21) nm; IR νmax 3429, 2920, 1639, 1573, 1473, 1431, 1292, 1149, 1049, 1018, 856 cm−1; 1H and 13C NMR data see Table 1 and Table 2; HRESIMS m/z 469.2217 [M + H]+ (calcd for C27H33O7, 469.2226) (Supplementary Figures S16–S20).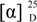 −93.3 (c 0.009 CHCl3); UV(CHCl3) λmax (log ε) 396 (3,61), 299 (3.79), 291 (3.74), 277 (4.32), 249 (3.84), 242 (3.96), 228 (3.29) nm; IR νmax 3479, 2924, 1708, 1643, 1573, 1469, 1242, 1114, 1033, 987, 856 cm−1; 1H and 13C NMR data see Table 1 and Table 2; HRESIMS m/z 483.2015 [M + H]+ (calcd for C27H31O8, 483.2014) (Supplementary Figures S21–S28).
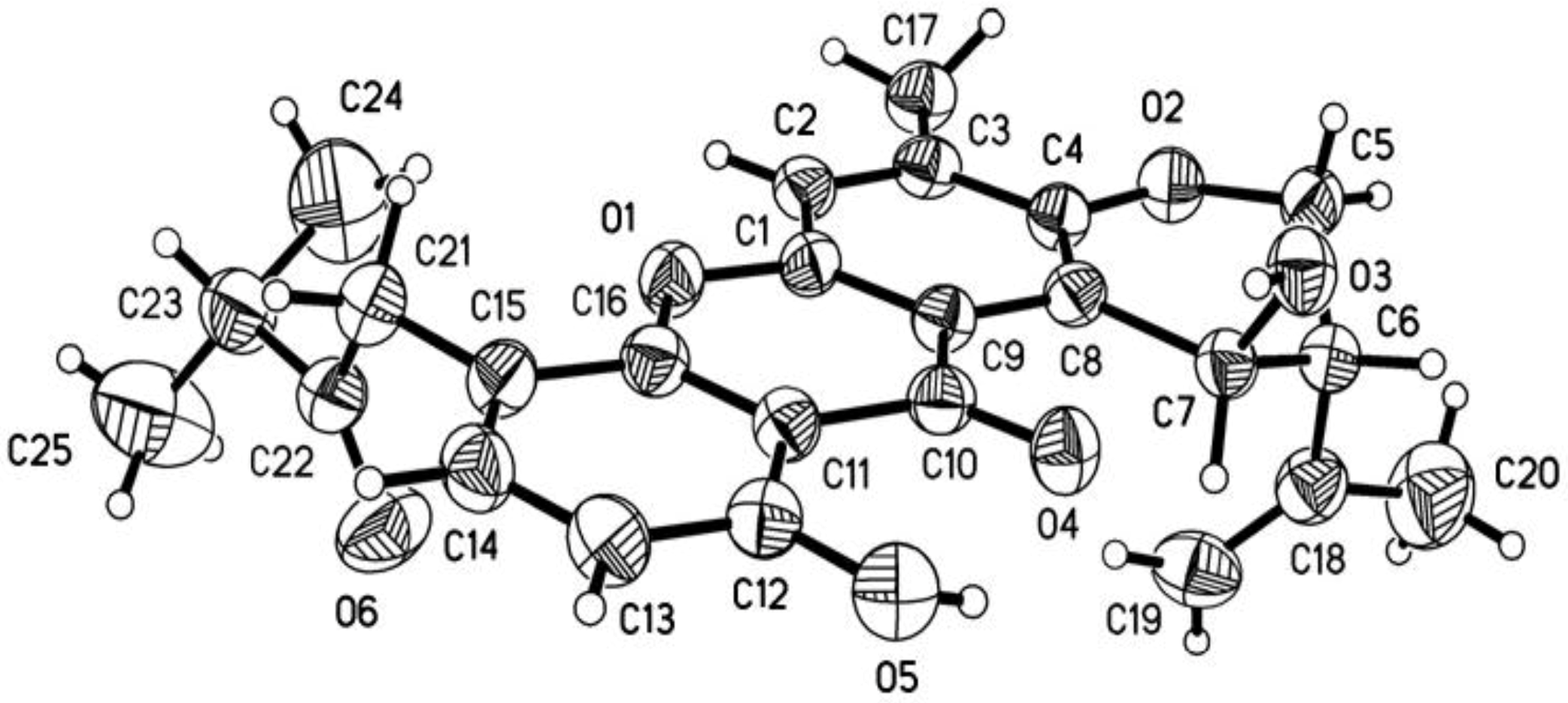
−93.3 (c 0.009 CHCl3); UV(CHCl3) λmax (log ε) 396 (3,61), 299 (3.79), 291 (3.74), 277 (4.32), 249 (3.84), 242 (3.96), 228 (3.29) nm; IR νmax 3479, 2924, 1708, 1643, 1573, 1469, 1242, 1114, 1033, 987, 856 cm−1; 1H and 13C NMR data see Table 1 and Table 2; HRESIMS m/z 483.2015 [M + H]+ (calcd for C27H31O8, 483.2014) (Supplementary Figures S21–S28).3.4. X-ray Crystallographic Analysis of Ruguloxanthone B (8)
3.5. Cytotoxic, Antibacterial, and Antifungal Assay
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pockrandt, D.; Ludwig, L.; Fan, A.; Konig, G.M.; Li, S.M. New insights into the biosynthesis of prenylated xanthones: Xptb from Aspergillus nidulans catalyses an O-prenylation of xanthones. ChemBioChem 2012, 13, 2764–2771. [Google Scholar] [CrossRef]
- Sousa, M.E.; Pinto, M.M.M. Synthesis of xanthones: An overview. Curr. Med. Chem. 2005, 12, 2447–2479. [Google Scholar] [CrossRef]
- El-Seedi, H.R.; El-Ghorab, D.M.H.; El-Barbary, M.A.; Zayed, M.F.; Goransson, U.; Larsson, S.; Verpoorte, R. Naturally occurring xanthones; latest investigations: Isolation, structure elucidation and chemosystematic significance. Curr. Med. Chem. 2009, 16, 2581–2626. [Google Scholar] [CrossRef]
- Pinto, M.M.M.; Castanheiro, R.A.P. Synthesis of prenylated xanthones: An overview. Curr. Org. Chem. 2009, 13, 1215–1240. [Google Scholar] [CrossRef]
- Pinto, M.M.M.; Sousa, M.E.; Nascimento, M.S.J. Xanthone derivatives: New insights in biological activities. Curr. Med. Chem. 2005, 12, 2517–2538. [Google Scholar] [CrossRef]
- Castanheiro, R.A.P.; Silva, A.M.S.; Campos, N.A.N.; Nascimento, M.S.J.; Pinto, M.M.M. Antitumor activity of some prenylated xanthones. Pharmaceuticals 2009, 2, 33–43. [Google Scholar]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2014, 31, 160–258. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2011, 28, 196–268. [Google Scholar] [CrossRef]
- Figueroa, M.; Gonzalez Mdel, C.; Rodriguez-Sotres, R.; Sosa-Peinado, A.; Gonzalez-Andrade, M.; Cerda-Garcia-Rojas, C.M.; Mata, R. Calmodulin inhibitors from the fungus Emericella sp. Bioorg. Med. Chem. 2009, 17, 2167–2174. [Google Scholar] [CrossRef]
- Kralj, A.; Kehraus, S.; Krick, A.; Eguereva, E.; Kelter, G.; Maurer, M.; Wortmann, A.; Fiebig, H.H.; Konig, G.M. Arugosins G and H: Prenylated polyketides from the marine-derived fungus Emericella nidulans var. acristata. J. Nat. Prod. 2006, 69, 995–1000. [Google Scholar] [CrossRef]
- Pornpakakul, S.; Liangsakul, J.; Ngamrojanavanich, N.; Roengsumran, S.; Sihanonth, P.; Piapukiew, J.; Sangvichien, E.; Puthong, S.; Petsom, A. Cytotoxic activity of four xanthones from Emericella variecolor, an endophytic fungus isolated from Croton oblongifolius. Arch. Pharm. Res. 2006, 29, 140–144. [Google Scholar] [CrossRef]
- Moosophon, P.; Kanokmedhakul, S.; Kanokmedhakul, K.; Soytong, K. Prenylxanthones and a bicyclo[3.3.1]nona-2,6-diene derivative from the fungus Emericella rugulosa. J. Nat. Prod. 2009, 72, 1442–1446. [Google Scholar] [CrossRef]
- Chexal, K.K.; Fouweath, C; Holker, J.S.E.; Simpson, T.J.; Young, K. Biosynthesis of fungal metabolites. 3. Structure of shamixanthone and tajixanthone, metabolites of Aspergillus variecolor. J. Chem. Soc. Perkin Trans. 1974, 1, 1584–1593. [Google Scholar]
- Malmstrom, J.; Christophersen, C.; Barrero, A.F.; Oltra, J.E.; Justicia, J.; Rosales, A. Bioactive metabolites from a marine-derived strain of the fungus Emericella variecolor. J. Nat. Prod. 2002, 65, 364–367. [Google Scholar] [CrossRef]
- Ahmed, S.A.; Bardshiri, E.; Mcintyre, C.R.; Simpson, T.J. Biosynthetic studies on tajixanthone and shamixanthone, polyketide hemiterpenoid metabolites of Aspergillus variecolor. Aust. J. Chem. 1992, 45, 249–274. [Google Scholar] [CrossRef]
- Lin, X.; Zhou, X.; Wang, F.; Liu, K.; Yang, B.; Yang, X.; Peng, Y.; Liu, J.; Ren, Z.; Liu, Y. A new cytotoxic sesquiterpene quinone produced by Penicillium sp. F00120 isolated from a deep sea sediment sample. Mar. Drugs 2012, 10, 106–115. [Google Scholar] [CrossRef]
- Fang, W.; Lin, X.; Zhou, X.; Wan, J.; Lu, X.; Yang, B.; Ai, W.; Lin, J.; Zhang, T.; Tu, Z.; Liu, Y. Cytotoxic and antiviral nitrobenzoyl sesquiterpenoids from the marine-derived fungus Aspergillus ochraceus Jcma1F17. MedChemComm 2014. [Google Scholar] [CrossRef]
- Zeng, Y.-B.; Wang, H.; Zuo, W.-J.; Zheng, B.; Yang, T.; Dai, H.-F.; Mei, W.-L. A fatty acid glycoside from a marine-derived fungus isolated from mangrove plant Scyphiphora hydrophyllacea. Mar. Drugs 2012, 10, 598–603. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fredimoses, M.; Zhou, X.; Lin, X.; Tian, X.; Ai, W.; Wang, J.; Liao, S.; Liu, J.; Yang, B.; Yang, X.; et al. New Prenylxanthones from the Deep-Sea Derived Fungus Emericella sp. SCSIO 05240. Mar. Drugs 2014, 12, 3190-3202. https://doi.org/10.3390/md12063190
Fredimoses M, Zhou X, Lin X, Tian X, Ai W, Wang J, Liao S, Liu J, Yang B, Yang X, et al. New Prenylxanthones from the Deep-Sea Derived Fungus Emericella sp. SCSIO 05240. Marine Drugs. 2014; 12(6):3190-3202. https://doi.org/10.3390/md12063190
Chicago/Turabian StyleFredimoses, Mangaladoss, Xuefeng Zhou, Xiuping Lin, Xinpeng Tian, Wen Ai, Junfeng Wang, Shengrong Liao, Juan Liu, Bin Yang, Xianwen Yang, and et al. 2014. "New Prenylxanthones from the Deep-Sea Derived Fungus Emericella sp. SCSIO 05240" Marine Drugs 12, no. 6: 3190-3202. https://doi.org/10.3390/md12063190
APA StyleFredimoses, M., Zhou, X., Lin, X., Tian, X., Ai, W., Wang, J., Liao, S., Liu, J., Yang, B., Yang, X., & Liu, Y. (2014). New Prenylxanthones from the Deep-Sea Derived Fungus Emericella sp. SCSIO 05240. Marine Drugs, 12(6), 3190-3202. https://doi.org/10.3390/md12063190









