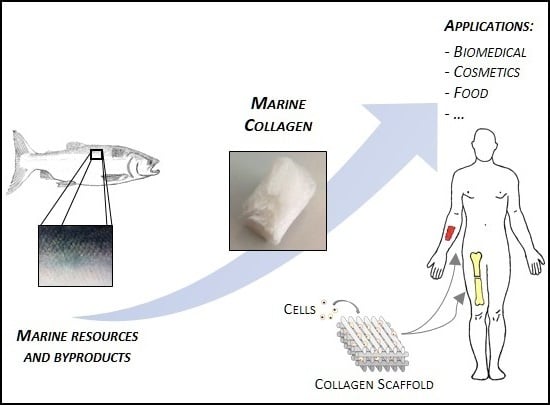
Marine Origin Collagens and Its Potential Applications
Abstract
:1. Introduction
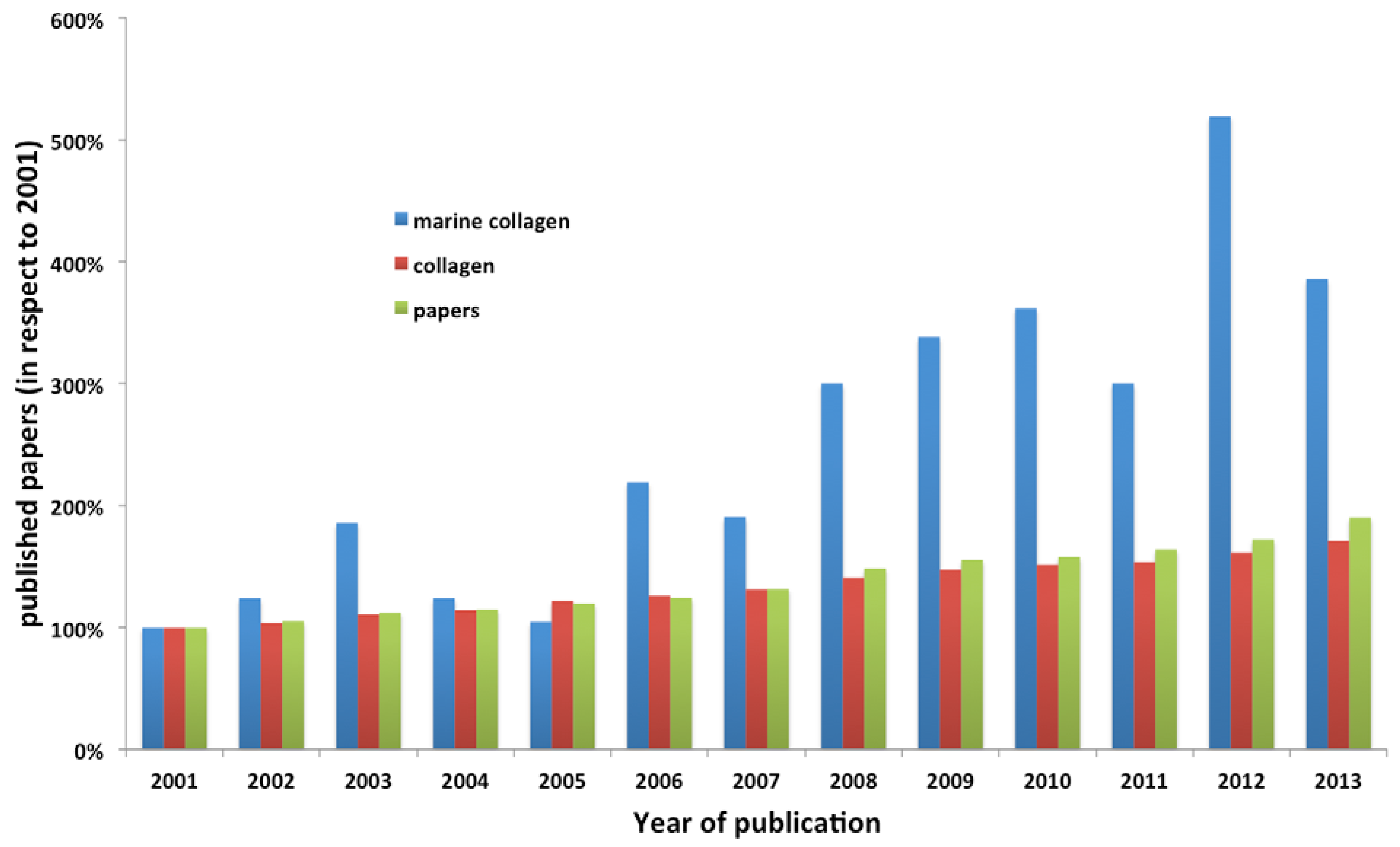
2. Marine Collagens
2.1. Sources and Isolation
| Collagen Type | Source of Collagen | Source Tissue | Yield | References |
|---|---|---|---|---|
| Type I | Bigeye snapper | Bone | ASC: 1.59% | [28] |
| Skin | ASC: 10.94% | |||
| Largefin longbarbel catfish | ASC: 16.8%; PSC: 28.0% | [23] | ||
| Seaweed pipefish | ASC: 5.5%; PSC: 33.2% | [32] | ||
| Brown backed toadfish | PSC: 54.3% | [19] | ||
| Ocellate pufferfish | ASC: 10.7%; PSC: 44.7% | [24] | ||
| Lizard fish | Scales | ASC: 0.79% | [33] | |
| Horse mackerel | ASC: 1.51% | |||
| Grey mullet | ASC: 0.43% | |||
| Flying fish | ASC: 0.72% | |||
| Yellowback seabream | ASC: 0.90% | |||
| Bigeye tuna | Bone | - | [34] | |
| Squid | Skin | 53% | [7] | |
| Cuttlefish | Skin | ASC: 0.58%; PSC: 16.23% | [35] | |
| Edible Jellyfish | Umbrella | 46.4% | [36] | |
| Type II | Brownbanded bamboo shark | Cartilage | ASC: 1.27%; PSC: 9.59% | [29] |
| Blacktip shark | Cartilage | ASC: 1.04%; PSC: 10.30% | ||
| Ribbon jellyfish | Umbrella | PSC: 9%–19% | [21] | |
| Type IV | Marine Sponge | 30% | [37,38,39] |
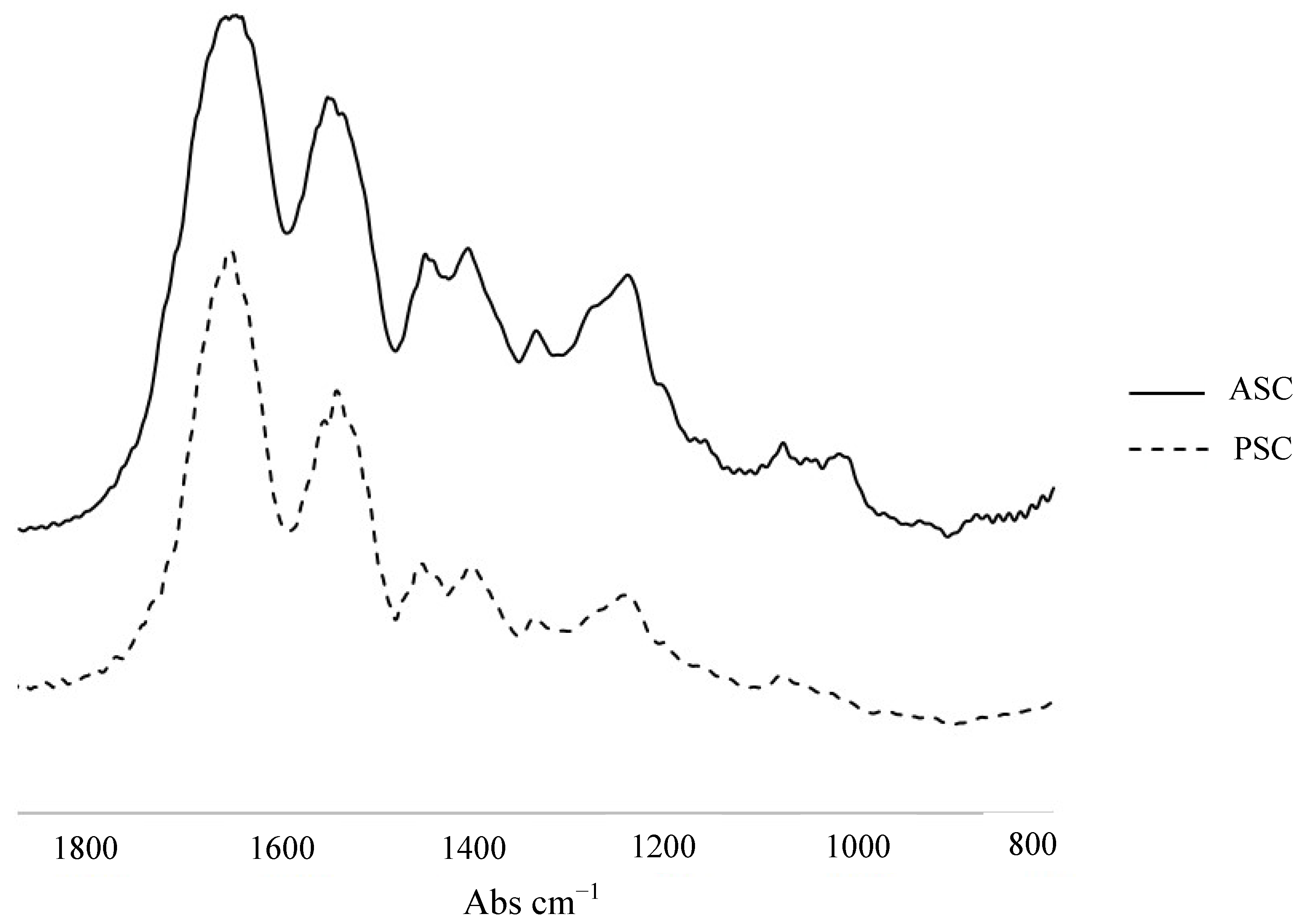
2.2. Characterization Methods
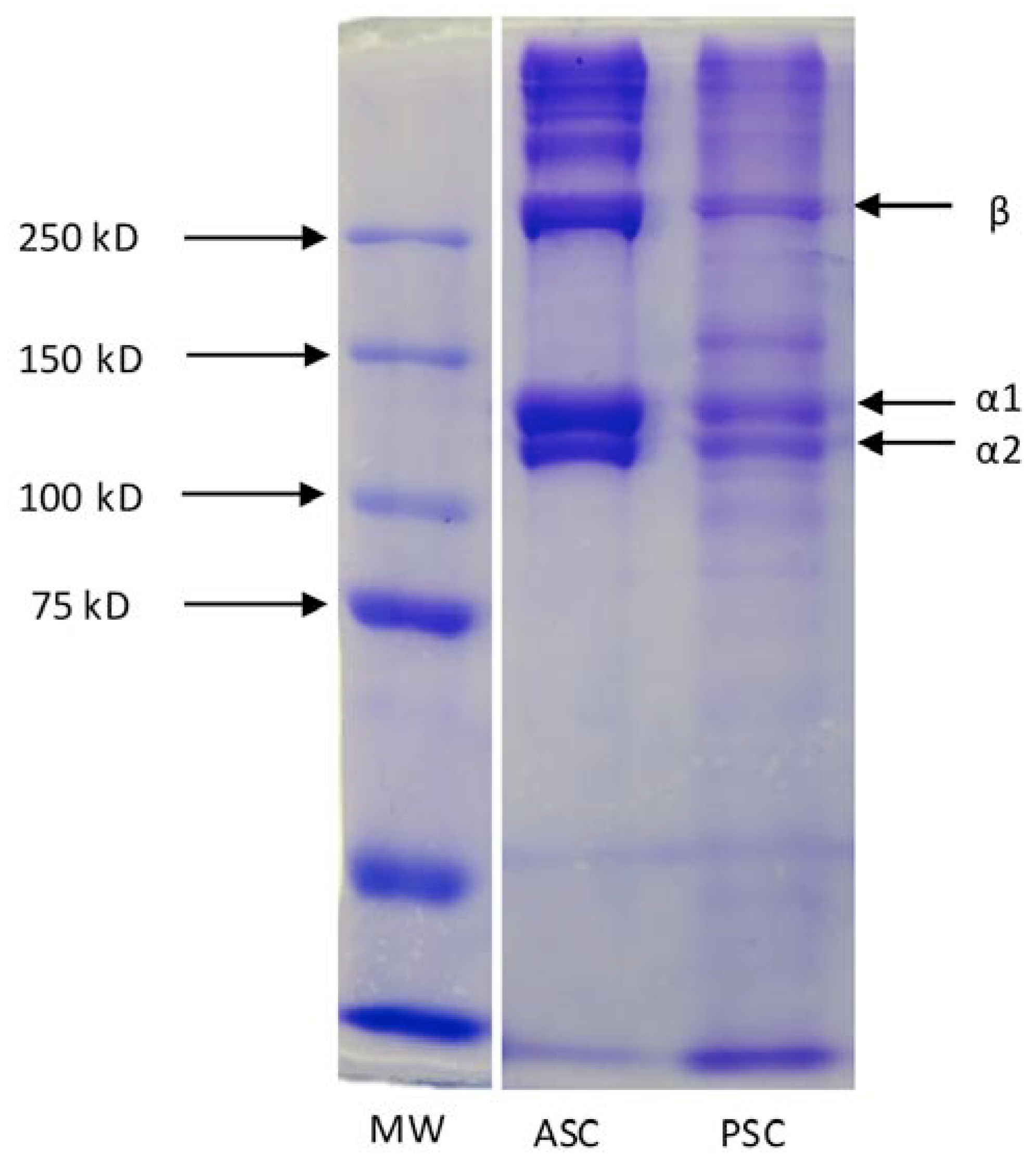
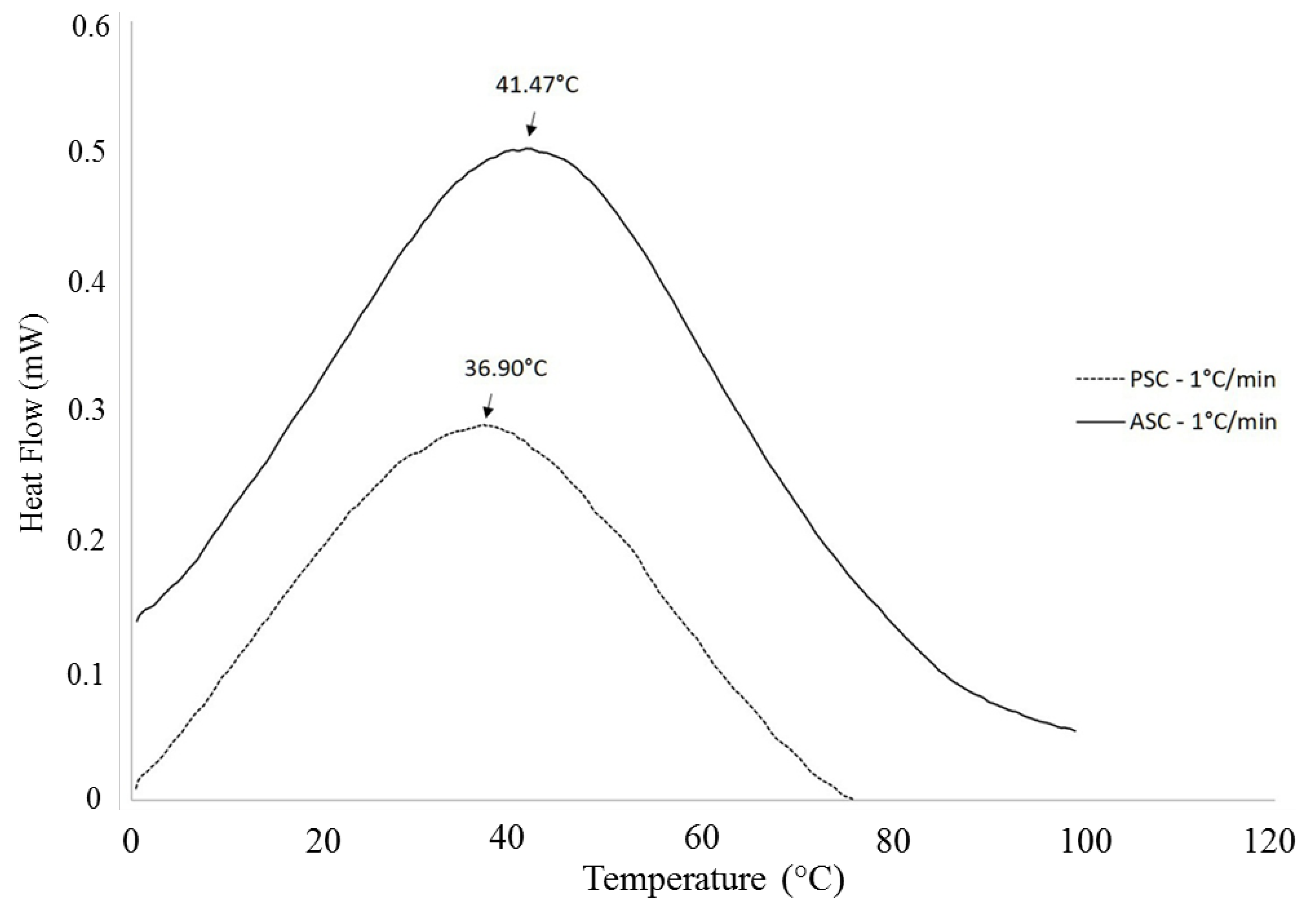
3. Applications
3.1. Tissue Engineering and Regeneration
3.2. Cosmetic, Skin Care and Other Medical Applications
4. Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vanderrest, M.; Garrone, R. Collagen family of proteins. FASEB J. 1991, 5, 2814–2823. [Google Scholar] [PubMed]
- Gelse, K.; Poschl, E.; Aigner, T. Collagens—Structure, function, and biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1531–1546. [Google Scholar] [CrossRef] [PubMed]
- Prockop, D.J.; Kivirikko, K.I. Collagens—Molecular-biology, diseases, and potentials for therapy. Ann. Rev. Biochem. 1995, 64, 403–434. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.M.; Gentile, P.; Chiono, V.; Ciardelli, G. Collagen for bone tissue regeneration. Acta Biomater. 2012, 8, 3191–3200. [Google Scholar] [CrossRef] [PubMed]
- Deyl, Z.; Miksik, I.; Eckhardt, A. Preparative procedures and purity assessment of collagen proteins. J. Chromatogr. B 2003, 790, 245–275. [Google Scholar] [CrossRef]
- Gomez-Guillen, M.C.; Turnay, J.; Fernandez-Diaz, M.D.; Ulmo, N.; Lizarbe, M.A.; Montero, P. Structural and physical properties of gelatin extracted from different marine species: A comparative study. Food Hydrocoll. 2002, 16, 25–34. [Google Scholar] [CrossRef]
- Kolodziejska, I.; Sikorski, Z.E.; Niecikowska, C. Parameters affecting the isolation of collagen from squid (Illex argentinus) skins. Food Chem. 1999, 66, 153–157. [Google Scholar] [CrossRef]
- Mendis, E.; Rajapakse, N.; Byun, H.-G.; Kim, S.-K. Investigation of jumbo squid (Dosidicus gigas) skin gelatin peptides for their in vitro antioxidant effects. Life Sci. 2005, 77, 2166–2178. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, H. Chitin and collagen as universal and alternative templates in biomineralization. Int. Geol. Rev. 2010, 52, 661–699. [Google Scholar] [CrossRef]
- Ehrlich, H.; Deutzmann, R.; Brunner, E.; Cappellini, E.; Koon, H.; Solazzo, C.; Yang, Y.; Ashford, D.; Thomas-Oates, J.; Lubeck, M.; et al. Mineralization of the metre-long biosilica structures of glass sponges is templated on hydroxylated collagen. Nat. Chem. 2010, 2, 1084–1088. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, H. Biological Materials of Marine Origin: Invertebrates; Springer: New York, NY, USA, 2010; Volume 1. [Google Scholar]
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002. [Google Scholar]
- Wright, N.T.; Humphrey, J.D. Denaturation of collagen via heating: An irreversible rate process. Ann. Rev. Biomed. Eng. 2002, 4, 109–128. [Google Scholar] [CrossRef]
- Walker, A.A. Oldest glue discovered. Archaeology Online News, 21 May 1998. [Google Scholar]
- Meena, C.; Mengi, S.; Deshpande, S. Biomedical and industrial applications of collagen. J. Chem. Sci. 1999, 111, 319–329. [Google Scholar]
- Venugopal, V. Marine Products for Healthcare: Functional and Bioactive Nutraceutical Compounds from the Ocean; CRC Press: Boca Raton, FI, USA, 2009. [Google Scholar]
- Arvanitoyannis, I.S.; Kassaveti, A. Fish industry waste: Treatments, environmental impacts, current and potential uses. Int. J. Food Sci. Technol. 2008, 43, 726–745. [Google Scholar] [CrossRef]
- Leary, D.; Vierros, M.; Hamon, G.; Arico, S.; Monagle, C. Marine genetic resources: A review of scientific and commercial interest. Mar. Policy 2009, 33, 183–194. [Google Scholar] [CrossRef]
- Senaratne, L.S.; Park, P.J.; Kim, S.K. Isolation and characterization of collagen from brown backed toadfish (Lagocephalus gloveri) skin. Bioresour. Technol. 2006, 97, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.T.; Karmas, E.; Lu, M.F. Age-related-changes in the collagen of bovine corium—studies on extractability, solubility and molecular-size distribution. J. Food Sci. 1983, 48, 681–685. [Google Scholar] [CrossRef]
- Barzideh, Z.; Latiff, A.; Gan, C.Y.; Benjakul, S.; Karim, A. Isolation and characterisation of collagen from the ribbon jellyfish (Chrysaora sp.). Int. J. Food Sci. Technol. 2014, 49, 1490–1499. [Google Scholar] [CrossRef]
- Sadowska, M.; Kolodziejska, I.; Niecikowska, C. Isolation of collagen from the skins of baltic cod (Gadus morhua). Food Chem. 2003, 81, 257–262. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, W.T.; Li, G.Y. Isolation and characterisation of collagens from the skin of largefin longbarbel catfish (Mystus macropterus). Food Chem. 2009, 115, 826–831. [Google Scholar] [CrossRef]
- Nagai, T.; Araki, Y.; Suzuki, N. Collagen of the skin of ocellate puffer fish (Takifugu rubripes). Food Chem. 2002, 78, 173–177. [Google Scholar] [CrossRef]
- Jonglareonrak, A.; Benjakul, S.; Visessanguan, W.; Nagai, T.; Tanaka, M. Isolation and characterisation of acid and pepsin-solubilised collagens from the skin of brownstripe red snapper (Lutjanus vitta). Food Chem. 2005, 93, 475–484. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, N. Isolation of collagen from fish waste material—skin, bone and fins. Food Chem. 2000, 68, 277–281. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, N.; Tanoue, Y.; Kai, N.; Nagashima, T. Characterization of acid-soluble collagen from skins of surf smelt (Hypomesus pretiosus japonicus Brevoort). Food Nutr. Sci. 2010, 1, 59–66. [Google Scholar] [CrossRef]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Nagai, T.; Tanaka, M. Characterisation of acid-soluble collagen from skin and bone of bigeye snapper (Priacanthus tayenus). Food Chem. 2005, 89, 363–372. [Google Scholar] [CrossRef]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Shahidi, F. Isolation and characterization of collagen from the cartilages of brownbanded bamboo shark (Chiloscyllium punctatum) and blacktip shark (Carcharhinus limbatus). LWT Food Sci. Technol. 2010, 43, 792–800. [Google Scholar] [CrossRef]
- Duan, R.; Zhang, J.J.; Du, X.Q.; Yao, X.C.; Konno, K. Properties of collagen from skin, scale and bone of carp (Cyprinus carpio). Food Chem. 2009, 112, 702–706. [Google Scholar] [CrossRef]
- Zelechowska, E.; Sadowska, M.; Turk, M. Isolation and some properties of collagen from the backbone of baltic cod (Gadus morhua). Food Hydrocoll. 2010, 24, 325–329. [Google Scholar] [CrossRef]
- Khan, S.B.; Qian, Z.J.; Ryu, B.; Kim, S.K. Isolation and biochemical characterization of collagens from seaweed pipefish, Syngnathus schlegeli. Biotechnol. Bioproc. E 2009, 14, 436–442. [Google Scholar] [CrossRef]
- Thuy, L.T.M.; Okazaki, E.; Osako, K. Isolation and characterization of acid-soluble collagen from the scales of marine fishes from Japan and Vietnam. Food Chem. 2014, 149, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.S.; Venkatesan, J.; Kim, S.K. Isolation and characterization of collagen from marine fish (Thunnus obesus). Biotechnol. Bioproc. E 2013, 18, 1185–1191. [Google Scholar] [CrossRef]
- Shanmugam, V.; Ramasamy, P.; Subhapradha, N.; Sudharsan, S.; Seedevi, P.; Moovendhan, M.; Krishnamoorthy, J.; Shanmugam, A.; Srinivasan, A. Extraction, structural and physical characterization of type I collagen from the outer skin of Sepiella inermis (orbigny, 1848). Afr. J. Biotechnol. 2012, 11, 14326–14337. [Google Scholar] [CrossRef]
- Nagai, T.; Ogawa, T.; Nakamura, T.; Ito, T.; Nakagawa, H.; Fujiki, K.; Nakao, M.; Yano, T. Collagen of edible jellyfish exumbrella. J. Sci. Food Agric. 1999, 79, 855–858. [Google Scholar] [CrossRef]
- Lin, Z.; Solomon, K.L.; Zhang, X.L.; Pavlos, N.J.; Abel, T.; Willers, C.; Dai, K.R.; Xu, J.K.; Zheng, Q.J.; Zheng, M.H. In vitro evaluation of natural marine sponge collagen as a scaffold for bone tissue engineering. Int. J. Biol. Sci. 2011, 7, 968–977. [Google Scholar] [CrossRef] [PubMed]
- Boute, N.; Exposito, J.Y.; BouryEsnault, N.; Vacelet, J.; Noro, N.; Miyazaki, K.; Yoshizato, K.; Garrone, R. Type IV collagen in sponges, the missing link in basement membrane ubiquity. Biol. Cell 1996, 88, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Swatschek, D.; Schatton, W.; Kellermann, J.; Muller, W.E.G.; Kreuter, J. Marine sponge collagen: Isolation, characterization and effects on the skin parameters surface-pH, moisture and sebum. Eur. J. Pharm. Biopharm. 2002, 53, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Skierka, E.; Sadowska, M.; Karwowska, A. Optimization of condition for demineralization baltic cod (Gadus morhua) backbone. Food Chem. 2007, 105, 215–218. [Google Scholar] [CrossRef]
- Skierka, E.; Sadowska, M. The influence of different acids and pepsin on the extractability of collagen from the skin of baltic cod (Gadus morhua). Food Chem. 2007, 105, 1302–1306. [Google Scholar] [CrossRef]
- Nagai, T.; Yamashita, E.; Taniguchi, K.; Kanamori, N.; Suzuki, N. Isolation and characterisation of collagen from the outer skin waste material of cuttlefish (Sepia lycidas). Food Chem. 2001, 72, 425–429. [Google Scholar] [CrossRef]
- Cao, H.; Xu, S.Y. Purification and characterization of type II collagen from chick sternal cartilage. Food Chem. 2008, 108, 439–445. [Google Scholar] [CrossRef]
- Lin, Y.K.; Liu, D.C. Effects of pepsin digestion at different temperatures and times on properties of telopeptide-poor collagen from bird feet. Food Chem. 2006, 94, 621–625. [Google Scholar] [CrossRef]
- Lynn, A.K.; Yannas, I.V.; Bonfield, W. Antigenicity and immunogenicity of collagen. J. Biomed. Mater. Res. B 2004, 71B, 343–354. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, N. Preparation and partial characterization of collagen from paper nautilus (Argonauta argo, linnaeus) outer skin. Food Chem. 2002, 76, 149–153. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, N.; Nagashima, T. Collagen from common minke whale (Balaenoptera acutorostrata) unesu. Food Chem. 2008, 111, 296–301. [Google Scholar] [CrossRef]
- Sben, X.R.; Kurihara, H.; Takahashi, K. Characterization of molecular species of collagen in scallop mantle. Food Chem. 2007, 102, 1187–1191. [Google Scholar] [CrossRef]
- Song, E.; Kim, S.Y.; Chun, T.; Byun, H.J.; Lee, Y.M. Collagen scaffolds derived from a marine source and their biocompatibility. Biomaterials 2006, 27, 2951–2961. [Google Scholar] [CrossRef] [PubMed]
- Addad, S.; Exposito, J.Y.; Faye, C.; Ricard-Blum, S.; Lethias, C. Isolation, characterization and biological evaluation of jellyfish collagen for use in biomedical applications. Mar. Drugs 2011, 9, 967–983. [Google Scholar] [CrossRef] [PubMed]
- Abraham, L.C.; Zuena, E.; Perez-Ramirez, B.; Kaplan, D.L. Guide to collagen characterization for biomaterial studies. J. Biomed. Mater. Res. Part B 2008, 87B, 264–285. [Google Scholar] [CrossRef]
- Fernandes-Silva, S.; Moreira-Silva, J.; Silva, S.; Perez-Martin, R.; Sotelo, C.; Mano, J.; Marques, A.; Silva, T.; Reis, R. Marine collagen scaffolds crosslinked “in situ” with genipin for cartilage regeneration. J. Tissue Eng. Regener. Med. 2012, 6, 163–163. [Google Scholar] [CrossRef]
- Rosenblo, J.; Harsch, M.; Jimenez, S. Hydroxyproline content determines denaturation temperature of chick tendon collagen. Arch. Biochem. Biophys. 1973, 158, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, C.L.; Bretscher, L.E.; Guzei, I.A.; Raines, R.T. Effect of 3-hydroxyproline residues on collagen stability. J. Am. Chem. Soc. 2003, 125, 6422–6427. [Google Scholar] [CrossRef] [PubMed]
- Molnár-Perl, I. Quantitation of Amino Acids and Amines by Chromatography: Methods and Protocols; Elsevier: Amsterdam, The Netherlands, 2005. [Google Scholar]
- KomsaPenkova, R.; Koynova, R.; Kostov, G.; Tenchov, B.G. Thermal stability of calf skin collagen type I in salt solutions. BBA Protein Struct. Mol. Enzymol. 1996, 1297, 171–181. [Google Scholar] [CrossRef]
- Bruylants, G.; Wouters, J.; Michaux, C. Differential scanning calorimetry in life science: Thermodynamics, stability, molecular recognition and application in drug design. Curr. Med. Chem. 2005, 12, 2011–2020. [Google Scholar] [CrossRef] [PubMed]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Kishimura, H.; Shahidi, F. Isolation and characterisation of collagen from the skin of brownbanded bamboo shark (Chiloscyllium punctatum). Food Chem. 2010, 119, 1519–1526. [Google Scholar] [CrossRef]
- Davoodi, J.; Wakarchuk, W.W.; Surewicz, W.K.; Carey, P.R. Scan-rate dependence in protein calorimetry: The reversible transitions of Bacillus circulans xylanase and a disulfide-bridge mutant. Protein Sci. 1998, 7, 1538–1544. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.A. Kinetics of collagen denaturation in mammalian lens capsules studied by differential scanning calorimetry. Int. J. Biol. Macromol. 1993, 15, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Leikina, E.; Mertts, M.V.; Kuznetsova, N.; Leikin, S. Type I collagen is thermally unstable at body temperature. Proc. Natl. Acad. Sci. USA 2002, 99, 1314–1318. [Google Scholar] [CrossRef] [PubMed]
- Belbachir, K.; Noreen, R.; Gouspillou, G.; Petibois, C. Collagen types analysis and differentiation by FTIR spectroscopy. Anal. Bioanal. Chem. 2009, 395, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Plepis, A.M.D.; Goissis, G.; DasGupta, D.K. Dielectric and pyroelectric characterization of anionic and native collagen. Polym. Eng. Sci. 1996, 36, 2932–2938. [Google Scholar] [CrossRef]
- Raspanti, M.; Alessandrini, A.; Gobbi, P.; Ruggeri, A. Collagen fibril surface: TMAFM, FEG-SEM and freeze-etching observations. Microsc. Res. Tech. 1996, 35, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.P.; Aparicio, C. Discerning the subfibrillar structure of mineralized collagen fibrils: A model for the ultrastructure of bone. PLoS One 2013, 8, e76782. [Google Scholar] [CrossRef] [PubMed]
- Starborg, T.; Kalson, N.S.; Lu, Y.H.; Mironov, A.; Cootes, T.F.; Holmes, D.F.; Kadler, K.E. Using transmission electron microscopy and 3View to determine collagen fibril size and three-dimensional organization. Nat. Protoc. 2013, 8, 1433–1448. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.; Yamada, S.; Ikeda, T.; Yanagiguchi, K. Fish collagen and tissue repair. In Marine Cosmeceuticals: Trends and Prospects; Kim, S.K., Ed.; CRC Press—Taylor & Francis Group: Boca Raton, FL, USA, 2011; pp. 133–141. [Google Scholar]
- Hoyer, B.; Bernhardt, A.; Heinemann, S.; Stachel, I.; Meyer, M.; Gelinsky, M. Biomimetically mineralized salmon collagen scaffolds for application in bone tissue engineering. Biomacromolecules 2012, 13, 1059–1066. [Google Scholar] [CrossRef] [PubMed]
- Nagai, N.; Yunoki, S.; Suzuki, T.; Sakata, M.; Tajima, K.; Munekata, M. Application of cross-linked salmon atelocollagen to the scaffold of human periodontal ligament cells. J. Biosci. Bioeng. 2004, 97, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Terada, M.; Izumi, K.; Ohnuki, H.; Saito, T.; Kato, H.; Yamamoto, M.; Kawano, Y.; Nozawa-Inoue, K.; Kashiwazaki, H.; Ikoma, T.; et al. Construction and characterization of a tissue-engineered oral mucosa equivalent based on a chitosan-fish scale collagen composite. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100B, 1792–1802. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, S.; Ehrlich, H.; Douglas, T.; Heinemann, C.; Worch, H.; Schatton, W.; Hanke, T. Ultrastructura studies on the collagen of the marine sponge Chondrosia reniformis Nardo. Biomacromolecules 2007, 8, 3452–3457. [Google Scholar] [CrossRef] [PubMed]
- Swatschek, D.; Schatton, W.; Muller, W.E.G.; Kreuter, J. Microparticles derived from marine sponge collagen (SCMPs): Preparation, characterization and suitability for dermal delivery of all-trans retinol. Eur. J. Pharm. Biopharm. 2002, 54, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Pallela, R.; Venkatesan, J.; Janapala, V.R.; Kim, S.K. Biophysicochemical evaluation of chitosan-hydroxyapatite-marine sponge collagen composite for bone tissue engineering. J. Biomed. Mater. Res. Part A 2012, 100A, 486–495. [Google Scholar] [CrossRef]
- Jeong, S.I.; Kim, S.Y.; Cho, S.K.; Chong, M.S.; Kim, K.S.; Kim, H.; Lee, S.B.; Lee, Y.M. Tissue-engineered vascular grafts composed of marine collagen and PLGA fibers using pulsatile perfusion bioreactors. Biomaterials 2007, 28, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, B.; Bernhardt, A.; Lode, A.; Heinemann, S.; Sewing, J.; Klinger, M.; Notbohm, H.; Gelinsky, M. Jellyfish collagen scaffolds for cartilage tissue engineering. Acta Biomater. 2014, 10, 883–892. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Duan, R.; Huang, L.; Song, Y.J.; Regenstein, J.M. Characterisation of acid-soluble and pepsin-solubilised collagen from jellyfish (Cyanea nozakii kishinouye). Food Chem. 2014, 150, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.J.; Liu, D.Y.; Keesing, J.K. Jellyfish blooms in china: Dominant species, causes and consequences. Mar. Pollut. Bull. 2010, 60, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Nazeer, R.A.; Suganya, U.S. Porous scaffolds of gelatin from the marine gastropod Ficus variegate with commercial cross linkers for biomedical applications. Food Sci. Biotechnol. 2014, 23, 327–335. [Google Scholar] [CrossRef]
- Yamamoto, K.; Igawa, K.; Sugimoto, K.; Yoshizawa, Y.; Yanagiguchi, K.; Ikeda, T.; Yamada, S.; Hayashi, Y. Biological safety of fish (tilapia) collagen. Biomed. Res. Int. 2014. [Google Scholar] [CrossRef]
- von Bomhard, A.; Veit, J.; Bermueller, C.; Rotter, N.; Staudenmaier, R.; Storck, K.; The, H.N. Prefabrication of 3D cartilage contructs: Towards a tissue engineered auricle—A model tested in rabbits. PLoS One 2013, 8, e71667. [Google Scholar] [CrossRef] [PubMed]
- Desimone, M.F.; Helary, C.; Quignard, S.; Rietveld, I.B.; Bataille, I.; Copello, G.J.; Mosser, G.; Giraud-Guille, M.M.; Livage, J.; Meddahi-Pelle, A.; et al. In vitro studies and preliminary in vivo evaluation of silicified concentrated collagen hydrogels. ACS Appl. Mater. Inter. 2011, 3, 3831–3838. [Google Scholar] [CrossRef]
- Pallela, R.; Venkatesan, J.; Bhatnagar, I.; Shim, Y.; Kim, S. Applications of marine collagen-based scaffolds in bone tissue engineering. In Marine Biomaterials: Characterization, Isolation and Applications; Kim, S.K., Ed.; CRC Press: Boca Raton, FL, USA, 2013; pp. 519–528. [Google Scholar]
- Kim, S.K.; Ngo, D.H.; Vo, T.S.; Ryu, B. Industry perspectives of marine-derived proteins as biomaterials. In Marine Biomaterials: Characterization, Isolation and Applications; Kim, S.K., Ed.; CRC Press: Boca Raton, FL, USA, 2013; pp. 737–746. [Google Scholar]
- Boran, G.; Regenstein, J.M. Fish gelatin. Adv. Food Nutr. Res. 2010, 60, 119–143. [Google Scholar] [PubMed]
- Xhauflaire-Uhoda, E.; Fontaine, K.; Pierard, G.E. Kinetics of moisturizing and firming effects of cosmetic formulations. Int. J. Cosmet. Sci. 2008, 30, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.H.; Chung, J.R.; Yoo, E.K.; Rha, S.J.; Lee, S.W.; Jeong, S.H.; Kim, H.W.; Han, K.H.; Kim, S.J.; Park, B.J.; et al. Cosmetic composition useful for moisturizing skin, comprises collagen separated from fish skin. KR2013066342-A; KR1339423-B1, 2013. [Google Scholar]
- Hyun, C.H.; Joo, H.H.; Hee, K.J.; Yoo, S.J.; Su, J.Y.; Dae, H.L.; Hye, M.P. Preparing collagen useful in cosmetic composition, involves hydrolyzing fish by-products using enzyme. KR2013094989-A, 2013. [Google Scholar]
- Zhang, J.J.; Duan, R.; Ye, C.; Konno, K. Isolation and characterization of collagens from scale of silver carp (Hypophthalmichthys molitrix). J. Food Biochem. 2010, 34, 1343–1354. [Google Scholar] [CrossRef]
- Calejo, M.T.; Almeida, A.J.; Fernandes, A.I. Exploring a new jellyfish collagen in the production of microparticles for protein delivery. J. Microencapsul. 2012, 29, 520–531. [Google Scholar] [CrossRef] [PubMed]
- Nicklas, M.; Schatton, W.; Heinemann, S.; Hanke, T.; Kreuter, J. Enteric coating derived from marine sponge collagen. Drug Dev. Ind. Pharm. 2009, 35, 1384–1388. [Google Scholar] [CrossRef] [PubMed]
- Nicklas, M.; Schatton, W.; Heinemann, S.; Hanke, T.; Kreuter, J. Preparation and characterization of marine sponge collagen nanoparticles and employment for the transdermal delivery of 17 beta-estradiol-hemihydrate. Drug Dev. Ind. Pharm. 2009, 35, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Simpson, D.A.; Masters, C.L.; Ohlrich, G.; Purdie, G.; Stuart, G.; Tannenberg, A.E.G. Iatrogenic Creutzfeldt-Jakob disease and its neurosurgical implications. J. Clin. Neurosci. 1996, 3, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.J.; Williams, M.A.; Khan, M.K.; Champion, H.C.; Nielsen, N.H.; Association, A.M. Risk of transmission of bovine spongiform encephalopathy to humans in the united states—Report of the council on scientific affairs. J. Am. Med. Assoc. 1999, 281, 2330–2339. [Google Scholar] [CrossRef]
- Cabrera-Crespo, J.; Goncalves, V.M.; Martins, E.A.L.; Grellet, S.; Lopes, A.P.Y.; Raw, I. Albumin purification from human placenta. Biotechnol. Appl. Biochem. 2000, 31, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.; Will, R.G.; Bradley, R.; Asher, D.M.; Detwiler, L. Bovine spongiform encephalopathy and variant Creutzfeldt-Jakob disease: Background, evolution, and current concerns. Emerg. Infect. Dis. 2001, 7, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Carruthers, J.; Carruthers, A. Mad cows, prions, and wrinkles. Arch. Dermatol. 2002, 138, 667–670. [Google Scholar] [PubMed]
- Easterbrook, C.; Maddern, G. Porcine and bovine surgical products: Jewish, muslim, and hindu perspectives. Arch. Surg. 2008, 143, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Goyal, D.; Goyal, A.; Brittberg, M. Consideration of religious sentiments while selecting a biological product for knee arthroscopy. Knee Surg. Sports Traumatol. Arthrosc. 2013, 21, 1577–1586. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, E.D.; Yip, M.; Melman, L.; Frisella, M.M.; Matthews, B.D. Informed consent: Cultural and religious issues associated with the use of allogeneic and xenogeneic mesh products. J. Am. Coll. Surg. 2010, 210, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.L.; Hillas, P.J.; Baez, J.A.; Nokelainen, M.; Balan, J.; Tang, J.; Spiro, R.; Polarek, J.W. The application of recombinant human collagen in tissue engineering. Biodrugs 2004, 18, 103–119. [Google Scholar] [CrossRef] [PubMed]
- Baez, J.; Olsen, D.; Polarek, J.W. Recombinant microbial systems for the production of human collagen and gelatin. Appl. Microbiol. Biotechnol. 2005, 69, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Myllyharju, J.; Nokelainen, M.; Vuorela, A.; Kivirikko, K.I. Expression of recombinant human type I-III collagens in the yeast Pichia pastoris. Biochem. Soc. Transact. 2000, 28, 353–357. [Google Scholar] [CrossRef]
- Nokelainen, M.; Tu, H.M.; Vuorela, A.; Notbohm, H.; Kivirikko, K.I.; Myllyharju, J. High-level production of human type I collagen in the yeast Pichia pastoris. Yeast 2001, 18, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Brodsky, B.; Kaplan, D.L. Shining light on collagen: Expressing collagen in plants. Tissue Eng. Part A 2013, 19, 1499–1501. [Google Scholar] [CrossRef] [PubMed]
- Shoseyov, O.; Posen, Y.; Grynspan, F. Human recombinant type I collagen produced in plants. Tissue Eng. Part A 2013, 19, 1527–1533. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, F.; Exposito, J.Y.; Bournat, P.; Gruber, V.; Perret, S.; Comte, J.; Olagnier, B.; Garrone, R.; Theisen, M. Triple helix assembly and processing of human collagen produced in transgenic tobacco plants. FEBS Lett. 2000, 469, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Suttle, C.A. Marine viruses—Major players in the global ecosystem. Nat. Rev. Microbiol. 2007, 5, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C.J.; Harrington, A.M.; Yates, T.L.; Simpson, G.L.; Baker, R.J. Global Disease Surveillance, Emergent Disease Preparedness, and National Security; Museum of Texas Tech University: Lubbock, TX, USA, 2009. [Google Scholar]
- Pati, F.; Datta, P.; Adhikari, B.; Dhara, S.; Ghosh, K.; Das Mohapatra, P.K. Collagen scaffolds derived from fresh water fish origin and their biocompatibility. J. Biomed. Mater. Res. A 2012, 100A, 1068–1079. [Google Scholar] [CrossRef] [PubMed]
- Collagen and HA-Based Biomaterials—Global Strategic Business Report, 338491; Global Industry Analysts, Inc.: San Jose, CA, USA, 2013.
- Biological Evaluation of Medical Devices—Part 1: Evaluation and Testing within a Risk Management Process; ISO 10993–1:2009; International Organization for Standardization: Geneva, Switzerland, 2009.
- Medical Devices—Application of Risk Management to Medical Devices; ISO 14971:2007; International Organization for Standardization: Geneva, Switzerland, 2007.
- Council Directive 93/42/EEC of 14 June 1993 Concerning Medical Devices; The Council of the European Communities: Luxembourg, Luxembourg, 1993.
- Fritz, B.E.M.A. Mitigating the Risks of Large-Scale Engineering Programs through Lean Management. Ph.D. Dissertation, ETH Zurich, Zurich, Switzerland, 2013. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, T.H.; Moreira-Silva, J.; Marques, A.L.P.; Domingues, A.; Bayon, Y.; Reis, R.L. Marine Origin Collagens and Its Potential Applications. Mar. Drugs 2014, 12, 5881-5901. https://doi.org/10.3390/md12125881
Silva TH, Moreira-Silva J, Marques ALP, Domingues A, Bayon Y, Reis RL. Marine Origin Collagens and Its Potential Applications. Marine Drugs. 2014; 12(12):5881-5901. https://doi.org/10.3390/md12125881
Chicago/Turabian StyleSilva, Tiago H., Joana Moreira-Silva, Ana L. P. Marques, Alberta Domingues, Yves Bayon, and Rui L. Reis. 2014. "Marine Origin Collagens and Its Potential Applications" Marine Drugs 12, no. 12: 5881-5901. https://doi.org/10.3390/md12125881
APA StyleSilva, T. H., Moreira-Silva, J., Marques, A. L. P., Domingues, A., Bayon, Y., & Reis, R. L. (2014). Marine Origin Collagens and Its Potential Applications. Marine Drugs, 12(12), 5881-5901. https://doi.org/10.3390/md12125881