1. Introduction
Since the 1980s, numerous clinical studies have shown that both the oil of
Perna canaliculus (PCSO-524™) and 18/12 fish oil, standardised to contain 18% eicosapentaenoic (EPA) and 12% docosahexaenoic acids (DHA), have anti-inflammatory activity that can contribute to reduced pain and improved joint mobility for patients who suffer osteoarthritis (OA) [
1,
2,
3]. PCSO-524™ has unique polyunsaturated fatty acids (PUFAs) [
4,
5]; including 5,9,12,15-octodecatetraenoic acid (OTA), 5,9,12,16-nonadecatertraenoic acid, 7,11,14,17-eicosatetraenoic acid (ETA), and 5,9,12,15,18-heneicosapentaenoic acid [
4]. These molecules (including EPA and DHA) are similar to arachidonic acid (AA) (5,8,11,14-eicosatetraenoic acid), the precursor to the inflammatory agents, prostaglandins and leukotrienes. In case of ETA the first double bond is located at the seventh position, and the second double bond is interrupted from the first by two methylene groups, resulting in the double bonds at positions 7, 11, 14 and 17. A similar pattern is shown for the three other novel compounds, whereby the second double bond is separated from the first by more than one methylene group. The interrupted bond positioning of these structural analogues of AA may account for their anti-inflammatory (AI) behaviour, by competitively inhibiting the active site of enzymes that use AA as a substrate [
5,
6]. This combination of omega-3 fatty acids is not found in any other known marine oils [
4]. Furthermore, fish oil studies typically use large dosages of standardised EPA and DHA fish oil [
7,
8] compared with similar studies [
9,
10] that used the oil of
P. canaliculus to achieve reductions in inflammatory markers.
Former studies [
11,
12] had compared the lipid fraction of
P. canaliculus with a freeze-dried preparation and demonstrated significant improvements in the AI effect of the extracts of
P. canaliculus due to a new stabilisation production process. Further research compared both the stabilised extract powder and the stabilised oil (now trademarked as PCSO-524™ and available commercially as Lyprinol
® and OmegaXL
®) and confirmed the effectiveness of the stabilised extract [
13,
14] to potentially provide physicians with an additional complementary therapy for the treatment of symptoms associated with OA and rheumatoid arthritis (RA).
Numerous fish oil studies over the years have also shown reduced inflammatory responses [
15,
16]. These studies covered asthma, exercise-induced inflammation and arthritis. Positive results usually require large doses of up to 20 g of fish oil to achieve a therapeutic dose [
14,
16]. It is well known that people should use fish oil under medical supervision if they bruise easily, have a bleeding disorder, or are also using anticoagulants. Large doses of omega-3 fatty acids may increase the risk of bleeding and can cause stomach gas, bloating, belching and diarrhoea.
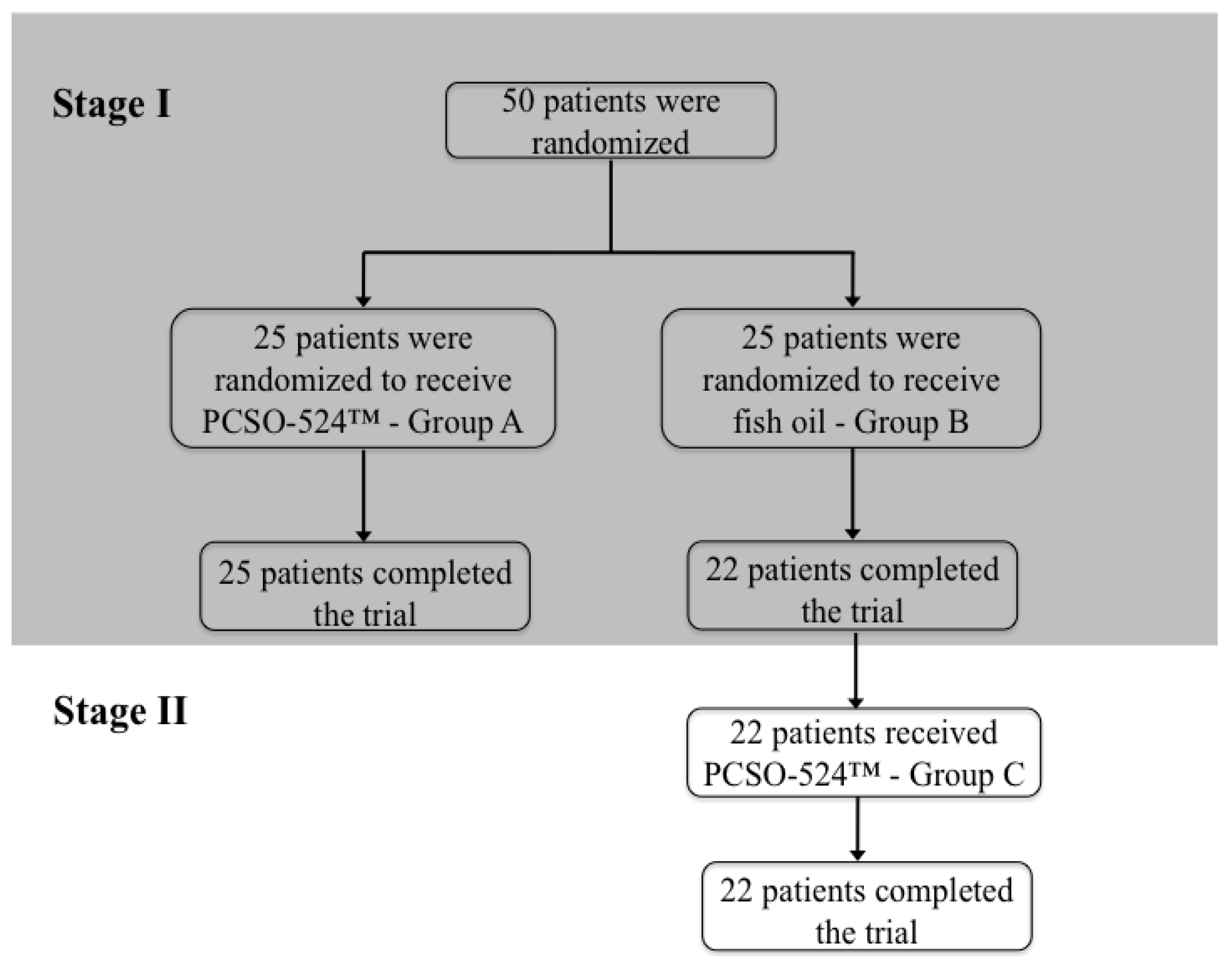
In the randomised double-blind, clinical trial performed by Szechinski and co-worker [
17], OA patients were administered with either PCSO-524™ (Group A) or standardised 18/12 fish oil capsules (Group B) for 12 weeks. A statistically significant improvement of both their pain symptoms related to OA and quality of life could be demonstrated (first stage of the study). The second stage provided an opportunity for the participants who had been administered 18/12 fish oil to also repeat the 12-week study taking PCSO-524™, the results of which are labelled under Group C. The results of the second stage are presented here. The significance of the study is comparative. Patients often use well-known analgesics such as paracetamol or ibuprofen to treat pain related to OA. Long-term intake of these analgesics may cause gastro-intestinal side effects and might also adversely affect the kidneys. PCSO-524™ has been shown to be an effective complementary or alternative active ingredient for the treatment of OA without any side effects [
3].
One of the objectives of the study was to ascertain if a similar dose of PCSO-524™ (400 mg) can achieve the same AI benefits as larger doses of standardised fish oil (1200 mg). If so this would provide practitioners with a safer first-line alternative that does not carry the risk of haemorrhage. It may also enable a reduction of intake of analgesics.
4. Discussion
The aim of this clinical trial was to evaluate the comparative effectiveness of PCSO-524™ relative to fish oil for pain relief, quality of life and safety.
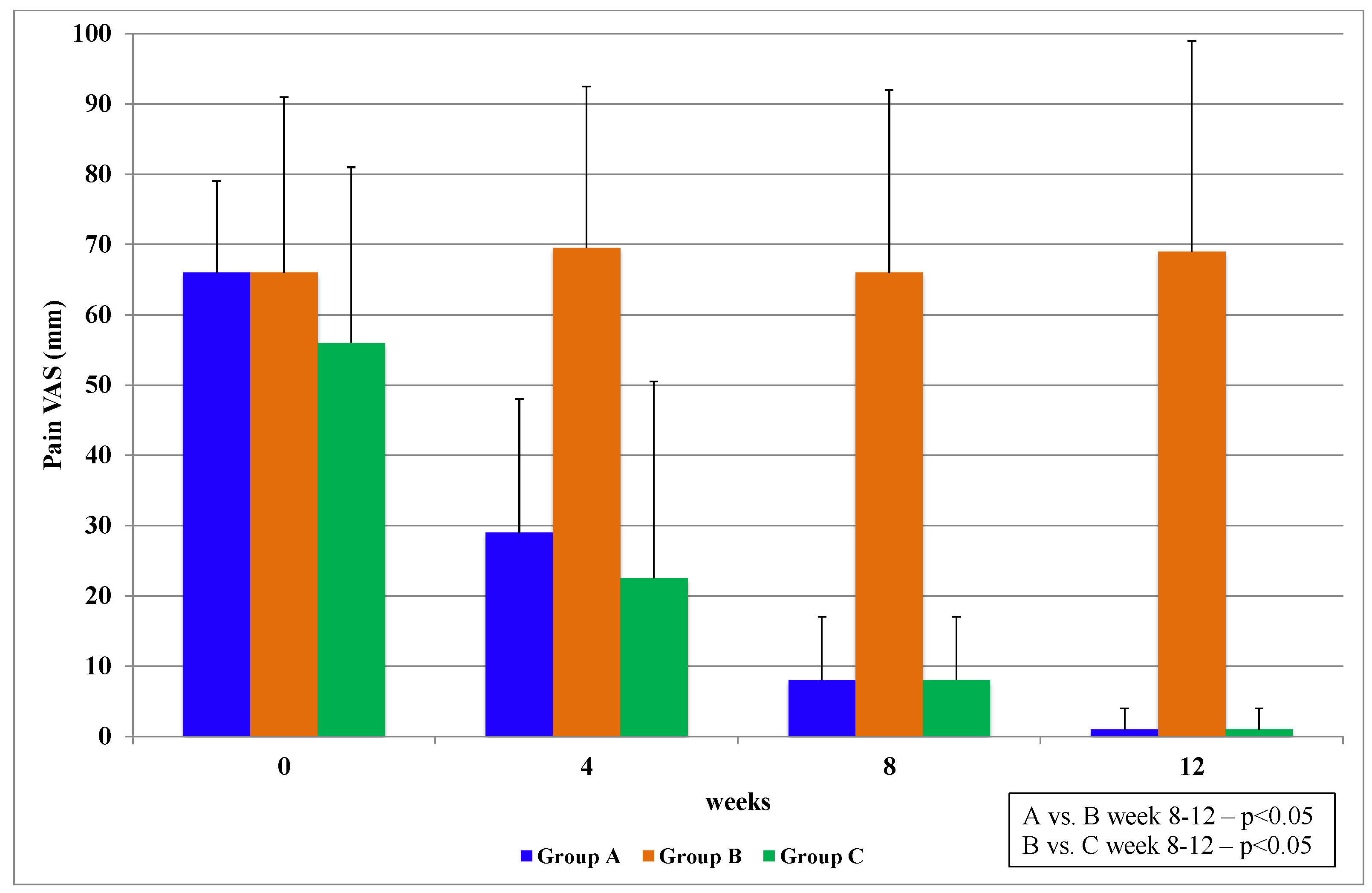
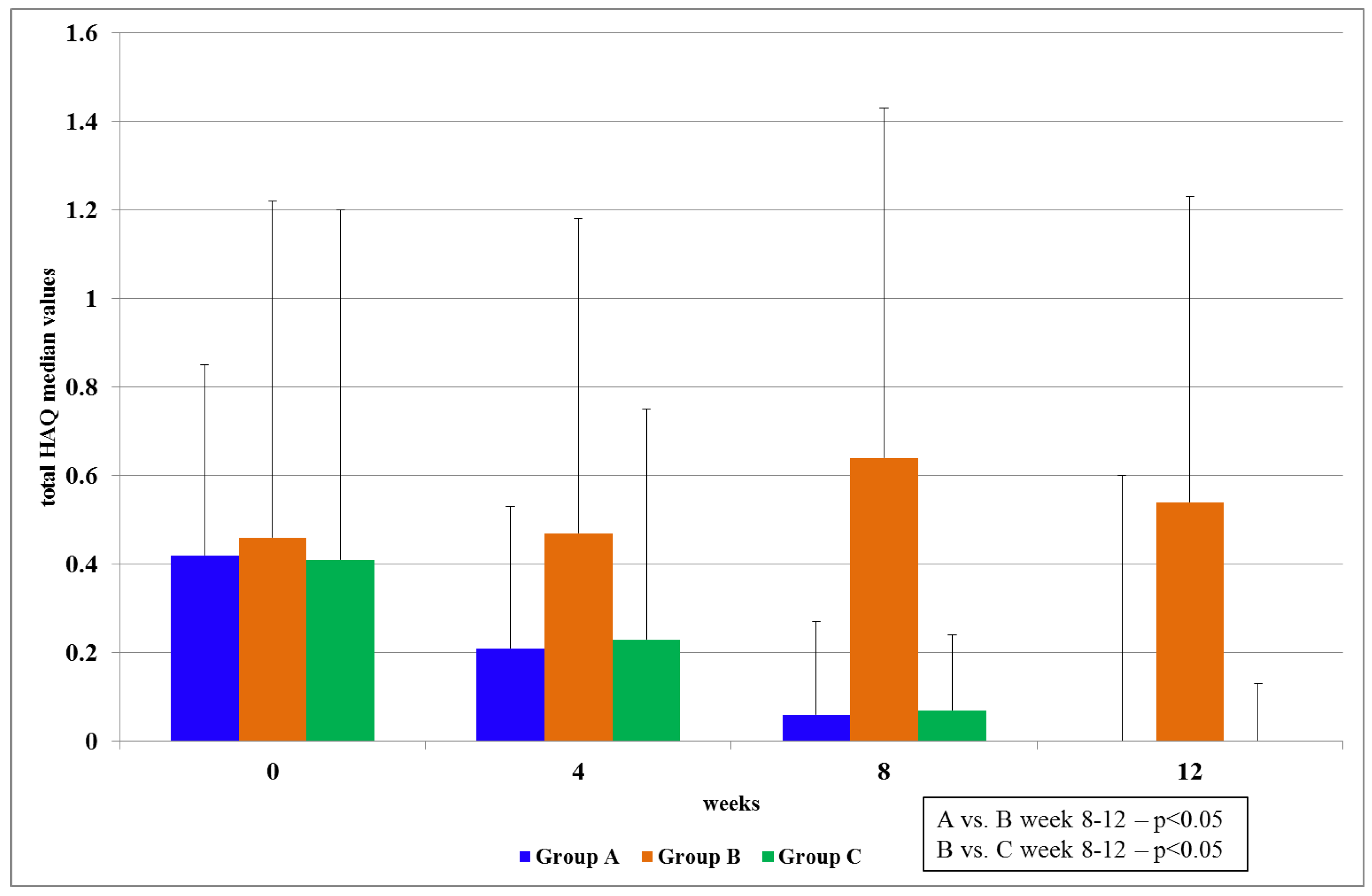
In stage I, the patients from Group A, who were treated with PCSO-524™, showed a statistically significant reduction of pain, improved levels of mobility and activity and 100% tolerance with no noted side effects. The efficacy results for PCSO-524™ oil are similar to those reported in other studies [
3,
11,
12,
13,
18,
19,
20]. In comparison, patients from Group B, who were treated with fish oil, did not show a notable reduction in pain, there was no significant improvement of mobility or activity and only 71% tolerance. In stage II, the patients knew that they had taken PCSO-524™ oil and those patients also showed a reduction of pain. However, due to the not-blinded part of the study, these results are consequently less valuable. Furthermore, neither stage was placebo-controlled (e.g., olive oil). Other PCSO-524™ studies have already documented the comparative results of placebo-controlled trials [
3].
PCSO-524™ is produced from the New Zealand green lipped mussel, a common species found in the protected waters around New Zealand. The non-polar lipids are removed from the mussel through the use of a patented CO
2 supercritical extraction process. PCSO-524™ is a very complex mixture of lipid classes which are high in free fatty acids. PCSO-524™ was compared with fish oil, which was found to be more effective in inhibition of the cyclooxygenase (COX) pathway [
6]. COXs are lipid metabolizing enzymes that catalyze the oxygenation of PUFA, usually AA, to form the prostanoids, which are potent cell-signaling molecules associated with the initiation, maintenance and resolution of inflammatory processes [
21].
Fish oil is composed predominantly of triglyceride molecules that are rich in EPA and DHA, while PCSO-524™ is more complex with more than 60 lipid compounds. Seemingly, fish oil results in the release of inhibitory PUFAs that give it similar activity in the anti-inflammatory pathways, but with less efficacy than PCSO-524™ [
6]. In a preliminary toxicology assessment, it was shown that the CO
2 lipid extract and its free fatty acid (FFA) components caused biologically significant AI activity
in vivo, with no apparent adverse side effects [
6]. In a more detailed analysis, the FFA class was fractionated from the CO
2 lipid extract and the FFA components demonstrated inhibition of inflammatory markers [
5]. As already mentioned, the new structurally-related family of omega-3 PUFAs that was identified included C18:4, C19:4, C20:4, and C21:5 PUFA in the fractions with high anti-inflammatory effect. The C20:4 was the predominant PUFA in the
P. canaliculus lipids and was identified as a structural isomer that mimicked AA. The novel anti-inflammatory compounds are understood to compete much more efficiently than other pro-inflammatory fatty acids for the COX and lipoxygenase (LOX) pathways [
4]. The latter pathway is responsible for the transformation of AA in the cell membrane into leukotrienes and the mechanism of AI activity of PCSO-524™ has been analyzed before [
5]. It prevented the migration of neutrophils and alleviated the signs of OA, such as pain, swelling and stiffness. Taking PCSO-524™ to relieve pain is a process. Typically, the AI effect starts to be noted within four to eight weeks to reduce the symptoms of chronic inflammation.
As the fish oil capsules contain a standardized amount of EPA (18%) and DHA (12%), we did not expect to observe such a major difference between patients treated with PCSO-524™ (Lyprinol®: 5.2% EPA and 3.4% DHA) and patients treated with fish oil. This result requires further research regarding the active substances in the two intervention treatments and the dosage levels.
Interestingly, OA patients who took PCSO-524™ (Group A and Group C) had a notable reduction of clinical symptoms in the first four weeks and a continuous improvement during the 12 weeks of the study. This was in line with previous findings [
9] that the lipid fractions of
P. canaliculus could achieve significant clinical improvements in the first four weeks in some patients who had RA.
Other studies support the view that dietary omega-3 PUFA, particularly EPA and DHA, are important modulators of a host’s inflammatory/immune responses [
22,
23]. It has been reported that large dosages of omega-3 PUFA are probably needed during long-duration therapy for diseases characterized by immune dysfunction [
23]. Olveira and co-workers established results with fish oil after 12 months in patients who had cystic fibrosis [
22]. Our study demonstrated that EPA and DHA in PCSO-524™ alone are not responsible for the AI effects, which recognizes previous studies regarding PCSO-524™ and EPA [
13] in rats and COX inhibition analyses of PUFAs of PCSO-524™ [
19]. Indeed, in another study it could be demonstrated that furan fatty acids (F-acids) also play an important AI role in PCSO-524™ [
24]. However, this needs further investigations.
Additionally, the P. canaliculus oil certainly showed that a quicker and more observable reduction in pain could be achieved with smaller dosages. There were no statistically significant results from fish oil for the administered dosage during the 12 weeks. This could have been due to the dosage of fish oil and also to the short duration of the trial, but larger dosages of fish oil for elderly patients would generally not be recommended.
Furthermore, it was noteworthy that patients from the PCSO-524™ group had less demand for a supportive treatment with paracetamol during the study (data not shown). Only six patients (36%) from Group A and four patients (18%) from Group C decided to use additional treatment with paracetamol, while in Group B 64.64% of the patients used additional paracetamol treatment to alleviate pain.
5. Conclusions and Outlook
Stabilized P. canaliculus oil has become a well-known natural inhibitor of COX and LOX, the activity of which has been referenced in numerous clinical trials, with no adverse effects.
The patients from Groups A and C judged the efficacy of PCSO-524™ positively with regard to pain relief within the first four weeks of administration and considered it to be beneficial for their quality of life. In both Groups A and C, patients continued to show further reductions in pain during the 12-week period of the study. The benefits of fish oil were not evident during this 12-week trial. The reasons for this conclusion included the side effects, large dosages needed and the duration required for fish oil to have an effect. Practitioners, from these results could consider stabilized P. canaliculus oil as a safer and faster-acting alternative complementary therapy for patients who suffer from OA compared with the use of fish oil.
In the future, clinical trials should always be placebo-controlled and perform a deeper examination of the dosage of PCSO-524™. Could higher dosages lead to a quicker pain relief for OA patients without any side effect? In addition, biochemical analyses should have a closer investigation to the molecules, which leads to the AI effect of PCSO-524™, because EPA and DHA are not (alone) responsible for it.