Discovery of New Eunicellin-Based Diterpenoids from a Formosan Soft Coral Cladiella sp.
Abstract
:1. Introduction
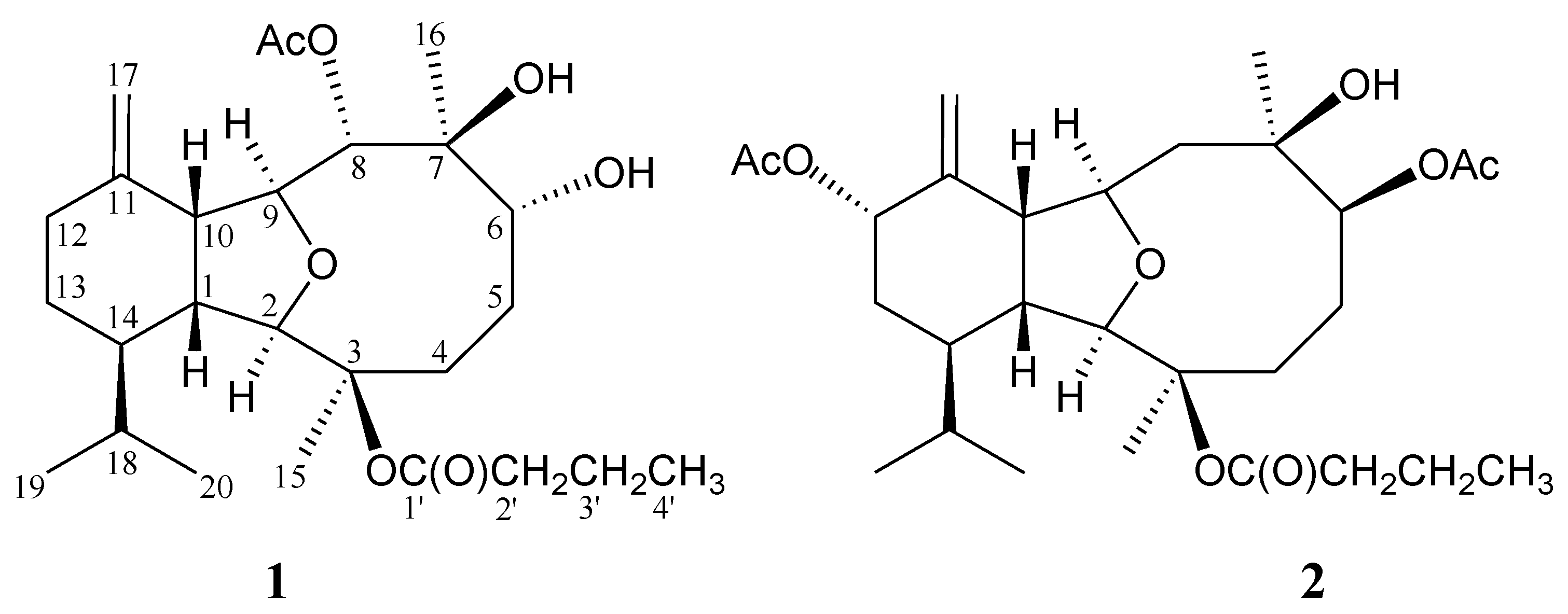
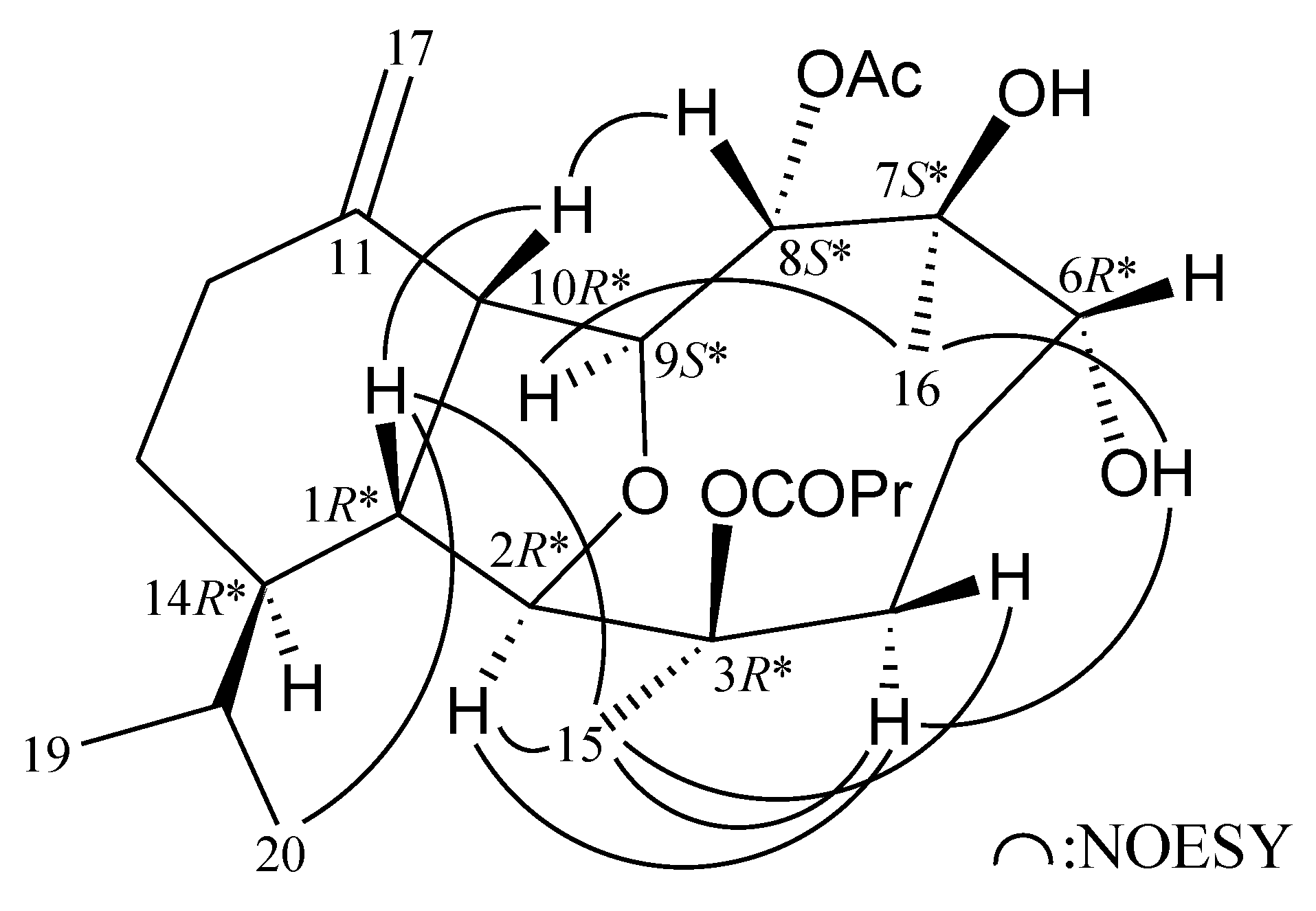
2. Results and Discussion
| Position | δH (J in Hz) | δC, Multiple | 1H–1H COSY | HMBC |
|---|---|---|---|---|
| 1 | 2.25 dd (11.5, 7.0) | 46.1, CH | H-10, H-14 | C-9, -10, -14, -18 |
| 2 | 3.61 s | 93.0, CH | n.o. | C-1, -3, -9, -10, -14, -15 |
| 3 | 85.2, C | |||
| 4 | 2.50 m; 1.97 m | 29.2, CH2 | H2-5 | C-2, -3, -6, -15 |
| 5 | 2.08 m; 1.70 m | 23.6, CH2 | H2-4, H-6 | C-3 |
| 6 | 3.69 dd (12.5, 11.0) | 76.3, CH | H2-5, OH-6 | C-4, -7, -16 |
| 7 | 78.6, C | |||
| 8 | 5.40 d (10.0) | 79.2, CH | H-9 | C-7, -9, -10, -16, acetate carbonyl |
| 9 | 3.77 dd (10.0, 7.0) | 78.8, CH | H-8, H-10 | C-2, -7, -8, -11 |
| 10 | 3.41 dd (7.0, 7.0) | 52.0, CH | H-1, H-9 | C-1, -8, -9, -11, -12, -14, -17 |
| 11 | 147.7, C | |||
| 12 | 2.30 br d (12.5); 2.01 m | 31.6, CH2 | H2-13 | n.o. |
| 13 | 1.78 m; 1.06 m | 25.2, CH2 | H2-12, H-14 | n.o. |
| 14 | 1.23 m | 44.3, CH | H-1, H2-13, H-18 | C-18 |
| 15 | 1.42 s | 23.3, CH3 | C-2, -3, -4 | |
| 16 | 1.31 s | 19.6, CH3 | C-6, -7, -8 | |
| 17 | 4.73 s; 4.58 s | 110.1, CH2 | C-10, -11, -12 | |
| 18 | 1.70 m | 29.0, CH | H-14, H3-19, H3-20 | C-1, -13, -14, -19, -20 |
| 19 | 0.99 d (7.0) | 21.9, CH3 | H-18 | C-14, -18, -20 |
| 20 | 0.79 d (6.5) | 15.3, CH3 | H-18 | C-14, -18, -19 |
| 3-OCOCH2CH2CH3 | ||||
| 1′ | 173.1, C | |||
| 2′ | 2.67 dt (16.0, 7.5); 2.51 dt (16.0, 7.5) | 36.7, CH2 | H2-3 | C-1′, -3′, -4′ |
| 3′ | 1.68 m | 18.5, CH2 | H2-2′, H3-4′ | C-1′, -2′, -4′ |
| 4′ | 1.01 t (7.5) | 13.5, CH3 | H2-3′ | C-2′, -3′ |
| 8-OAc | 171.5, C | |||
| 2.05 s | 21.4, CH3 | Acetate carbonyl | ||
| 6-OH | 4.41 br d (11.0) | H-6 | n.o. |
| Cell lines IC50 (µM) | |||||
|---|---|---|---|---|---|
| Compounds | HL-60 | K562 | DLD-1 | HCT-116 | T-47D |
| 1 | 32.15 | NA | 1.59 | NA | NA |
| 2 | 34.21 | NA | 37.95 | NA | NA |
| Doxorubicin a | 0.002 | 1.29 | 10.98 | 0.81 | 1.71 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Animal Material
3.3. Extraction and Isolation
 −8 (c 0.04, CHCl3); IR (neat) νmax 3457, 1736 cm−1; 1H (500 MHz, CDCl3) and 13C (125 MHz, CDCl3) NMR data, see Table 1; ESIMS: m/z 489 (M + Na)+; HRESIMS: m/z 489.2825 (calcd for C26H42O7Na, 489.2828).
−8 (c 0.04, CHCl3); IR (neat) νmax 3457, 1736 cm−1; 1H (500 MHz, CDCl3) and 13C (125 MHz, CDCl3) NMR data, see Table 1; ESIMS: m/z 489 (M + Na)+; HRESIMS: m/z 489.2825 (calcd for C26H42O7Na, 489.2828). +2 (c 0.08, CHCl3); IR (neat) νmax 3413, 1732 cm−1; 1H (400 MHz, CDCl3) δH 5.63 (1H, d, J = 5.6 Hz, H-6), 5.49 (1H, dd, J = 3.2, 3.2 Hz, H-12), 5.15 (1H, d, J = 1.2 Hz, H-17), 4.94 (1H, d, J = 1.2 Hz, H-17), 4.37 (1H, ddd, J = 8.4, 7.2, 7.2 Hz, H-9), 3.72 (1H, s, H-2), 3.03 (1H, dd, J = 7.2, 7.2 Hz, H-10), 2.61 (1H, dd, J = 14.4, 8.4 Hz, H-4), 2.39–2.27 (2H, m, H2-2′), 2.22 (1H, dd, J = 13.6, 7.2 Hz, H-1), 2.08 (3H, s, acetate methyl), 2.05 (3H, s, acetate methyl), 2.01 (1H, m, H-4), 1.94 (1H, ddd, J = 14.0, 3.6, 3.2 Hz, H-13), 1.85 (2H, m, H2-8), 1.81 (1H, m, H-18), 1.69 (1H, m, H-14), 1.67 (2H, m, H2-3′), 1.53–1.45 (2H, m, H2-5), 1.41 (3H, s, H3-15), 1.30 (1H, ddd, J = 14.0, 13.6, 3.2 Hz, H-13), 1.19 (3H, s, H3-16), 0.99 (3H, t, J = 7.2 Hz, H3-4′), 0.95 (3H, d, J = 6.8 Hz, H3-19), 0.79 (3H, d, J = 6.8 Hz, H3-20); 13C (100 MHz, CDCl3) δC 172.2 (C-1′, n-butyrate carbonyl), 171.8 (acetate carbonyl), 170.4 (acetate carbonyl), 142.8 (C-11), 116.8 (CH2-17), 91.3 (CH-2), 86.5 (C-3), 79.2 (CH-9), 84.4 (CH-6), 75.5 (C-7), 72.8 (CH-12), 51.9 (CH-10), 46.1 (CH2-8), 44.7 (CH-1), 37.4 (CH2-2′), 36.3 (CH-14), 35.6 (CH2-4), 29.1 (CH2-5), 28.5 (CH2-13), 28.5 (CH-18), 23.7 (CH3-16), 23.1 (CH3-15), 21.7 (CH3-19), 21.6 (acetate methyl), 21.4 (acetate methyl), 18.4 (CH2-3′), 15.3 (CH3-20), 13.7 (CH3-4′); ESIMS: m/z 531 (M + Na)+; HRESIMS: m/z 531.2931 (calcd for C28H44O8Na, 531.2934).
+2 (c 0.08, CHCl3); IR (neat) νmax 3413, 1732 cm−1; 1H (400 MHz, CDCl3) δH 5.63 (1H, d, J = 5.6 Hz, H-6), 5.49 (1H, dd, J = 3.2, 3.2 Hz, H-12), 5.15 (1H, d, J = 1.2 Hz, H-17), 4.94 (1H, d, J = 1.2 Hz, H-17), 4.37 (1H, ddd, J = 8.4, 7.2, 7.2 Hz, H-9), 3.72 (1H, s, H-2), 3.03 (1H, dd, J = 7.2, 7.2 Hz, H-10), 2.61 (1H, dd, J = 14.4, 8.4 Hz, H-4), 2.39–2.27 (2H, m, H2-2′), 2.22 (1H, dd, J = 13.6, 7.2 Hz, H-1), 2.08 (3H, s, acetate methyl), 2.05 (3H, s, acetate methyl), 2.01 (1H, m, H-4), 1.94 (1H, ddd, J = 14.0, 3.6, 3.2 Hz, H-13), 1.85 (2H, m, H2-8), 1.81 (1H, m, H-18), 1.69 (1H, m, H-14), 1.67 (2H, m, H2-3′), 1.53–1.45 (2H, m, H2-5), 1.41 (3H, s, H3-15), 1.30 (1H, ddd, J = 14.0, 13.6, 3.2 Hz, H-13), 1.19 (3H, s, H3-16), 0.99 (3H, t, J = 7.2 Hz, H3-4′), 0.95 (3H, d, J = 6.8 Hz, H3-19), 0.79 (3H, d, J = 6.8 Hz, H3-20); 13C (100 MHz, CDCl3) δC 172.2 (C-1′, n-butyrate carbonyl), 171.8 (acetate carbonyl), 170.4 (acetate carbonyl), 142.8 (C-11), 116.8 (CH2-17), 91.3 (CH-2), 86.5 (C-3), 79.2 (CH-9), 84.4 (CH-6), 75.5 (C-7), 72.8 (CH-12), 51.9 (CH-10), 46.1 (CH2-8), 44.7 (CH-1), 37.4 (CH2-2′), 36.3 (CH-14), 35.6 (CH2-4), 29.1 (CH2-5), 28.5 (CH2-13), 28.5 (CH-18), 23.7 (CH3-16), 23.1 (CH3-15), 21.7 (CH3-19), 21.6 (acetate methyl), 21.4 (acetate methyl), 18.4 (CH2-3′), 15.3 (CH3-20), 13.7 (CH3-4′); ESIMS: m/z 531 (M + Na)+; HRESIMS: m/z 531.2931 (calcd for C28H44O8Na, 531.2934).3.4. Cytotoxicity Testing
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Chen, Y.-H.; Tai, C.-Y.; Hwang, T.-L.; Weng, C.-F.; Li, J.-J.; Fang, L.-S.; Wang, W.-H.; Wu, Y.-C.; Sung, P.-J. Cladielloides A and B: New eunicellin-type diterpenoids from an Indonesian octocoral Cladiella sp. Mar. Drugs 2010, 8, 2936–2945. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Tai, C.-Y.; Kuo, Y.-H.; Kao, C.-Y.; Li, J.-J.; Hwang, T.-L.; Fang, L.-S.; Wang, W.-H.; Sheu, J.-H.; Sung, P.-J. Cladieunicellins A–E, new eunicellins from an Indonesian soft coral Cladiella sp. Chem. Pharm. Bull. 2011, 59, 353–358. [Google Scholar] [CrossRef]
- Tai, C.-Y.; Chen, Y.-H.; Hwang, T.-L.; Fang, L.-S.; Wang, W.-H.; Liu, M.-C.; Su, J.-H.; Wu, Y.-C.; Sung, P.-J. Cladielloides C and D: Novel eunicellin-based diterpenoids from an Indonesian octocoral Cladiella sp. Bull. Chem. Soc. Jpn. 2011, 84, 531–536. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Tai, C.-Y.; Su, Y.-D.; Chang, Y.-C.; Lu, M.-C.; Weng, C.-F.; Su, J.-H.; Hwang, T.-L.; Wu, Y.-C.; Sung, P.-J. Discovery of new eunicellins from an Indonesian octocoral Cladiella sp. Mar. Drugs 2011, 9, 934–943. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Hwang, T.-L.; Su, Y.-D.; Chang, Y.-C.; Chen, Y.-H.; Hong, P.-H.; Hu, L.-C.; Yen, W.-H.; Hsu, H.-Y.; Huang, S.-J.; et al. New 6-hydroxyeunicellins from a soft coral Cladiella sp. Chem. Pharm. Bull. 2012, 60, 160–163. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Tai, C.-Y.; Hwang, T.-L.; Sung, P.-J. Cladieunicellin H, a new hemiketal eunicellin-based diterpenoid from the octocoral Cladiella sp. Nat. Prod. Commun. 2012, 7, 481–484. [Google Scholar]
- Wahlberg, I.; Eklund, A.-M. Cyclized cembranoids of natural occurrence. Fortschr. Chem. Org. Naturst. 1992, 60, 1–141. [Google Scholar] [CrossRef]
- Bernardelli, P.; Paquette, L.A. Survey of oxygenated 2,11-cyclized cembranoids of marine origin. Heterocycles 1998, 49, 531–556. [Google Scholar] [CrossRef]
- Welford, A.J.; Collins, I. The 2,11-cyclized cembranoids: Cladiellins, asbestinins, and briarellins (period 1998–2010). J. Nat. Prod. 2011, 74, 2318–2328. [Google Scholar] [CrossRef]
- Radhika, P. Chemical constituents and biological activities of the soft corals of genus Cladiella: A review. Biochem. Syst. Ecol. 2006, 34, 781–789. [Google Scholar] [CrossRef]
- Ahmed, A.F.; Wu, M.-H.; Wang, G.-H.; Wu, Y.-C.; Sheu, J.-H. Eunicellin-based diterpenoids, australins A–D, isolated from the soft coral Cladiella australis. J. Nat. Prod. 2005, 68, 1051–1055. [Google Scholar] [CrossRef]
- Liang, C.-H.; Wang, G.-H.; Liaw, C.-C.; Lee, M.-F.; Wang, S.-H.; Cheng, D.-L.; Chou, T.-H. Extracts from Cladiella australis, Clavularia viridis and Klyxum simplex (soft corals) are capable of inhibiting the growth of human oral squamous cell carcinoma cells. Mar. Drugs 2008, 6, 595–606. [Google Scholar] [CrossRef]
- Chen, B.-W.; Chang, S.-M.; Huang, C.-Y.; Chao, C.-H.; Su, J.-H.; Wen, Z.-H.; Hsu, C.-H.; Dai, C.-F.; Wu, Y.-C.; Sheu, J.-H. Hirsutalins A–H, eunicellin-based diterpenoids from the soft coral Cladiella hirsuta. J. Nat. Prod. 2010, 73, 1785–1791. [Google Scholar] [CrossRef]
- Tai, C.-J.; Su, J.-H.; Huang, M.-S.; Wen, Z.-H.; Dai, C.-F.; Sheu, J.-H. Bioactive eunicellin-based diterpenoids from the soft coral Cladiella krempfi. Mar. Drugs 2011, 9, 2036–2045. [Google Scholar] [CrossRef]
- Tai, C.-J.; Su, J.-H.; Huang, C.-Y.; Huang, M.-S.; Wen, Z.-H.; Dai, C.-F.; Sheu, J.-H. Cytotoxic and anti-inflammatory eunicellin-based diterpenoids from the soft coral Cladiella krempfi. Mar. Drugs 2013, 11, 788–799. [Google Scholar] [CrossRef]
- Chen, B.-W.; Wang, S.-Y.; Huang, C.-Y.; Chen, S.-L.; Wu, Y.-C.; Sheu, J.-H. Hirsutalins I–M, eunicellin-based diterpenoids from the soft coral Cladiella hirsuta. Tetrahedron 2013, 69, 2296–2301. [Google Scholar] [CrossRef]
- Lee, Y.-N.; Tai, C.-J.; Hwang, T.-L.; Sheu, J.-H. Krempfielins J–M, new eunicellin-based diterpenoids from the soft coral Cladiella krempfi. Mar. Drugs 2013, 11, 2741–2750. [Google Scholar] [CrossRef]
- Ochi, M.; Yamada, K.; Kataoka, K.; Kotsuki, H.; Shibata, K. Litophynins I and J, two new biologically active diterpenoids from the soft coral Litophyton sp. Chem. Lett. 1992, 21, 155–158. [Google Scholar]
- Fabricius, K.; Alderslade, P. Soft Corals and Sea Fans—A Comprehensive Guide to the Tropical Shallow-Water Genera of the Central-West Pacific, the Indian Ocean and the Red Sea, 1st ed.; Australian Institute of Marine Science: Queensland, Australia, 2001; pp. 84–85. [Google Scholar]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B.J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res. 1988, 48, 589–601. [Google Scholar]
- Scudiero, D.A.; Shoemaker, R.H.; Paull, K.D.; Monks, A.; Tierney, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, T.-H.; Lu, M.-C.; Chang, Y.-C.; Su, Y.-D.; Chen, Y.-H.; Lin, N.-C.; Fang, L.-S.; Wu, Y.-C.; Sung, P.-J. Discovery of New Eunicellin-Based Diterpenoids from a Formosan Soft Coral Cladiella sp. Mar. Drugs 2013, 11, 4585-4593. https://doi.org/10.3390/md11114585
Chen T-H, Lu M-C, Chang Y-C, Su Y-D, Chen Y-H, Lin N-C, Fang L-S, Wu Y-C, Sung P-J. Discovery of New Eunicellin-Based Diterpenoids from a Formosan Soft Coral Cladiella sp. Marine Drugs. 2013; 11(11):4585-4593. https://doi.org/10.3390/md11114585
Chicago/Turabian StyleChen, Tsung-Hung, Mei-Chin Lu, Yu-Chia Chang, Yin-Di Su, Yu-Hsin Chen, Nai-Cheng Lin, Lee-Shing Fang, Yang-Chang Wu, and Ping-Jyun Sung. 2013. "Discovery of New Eunicellin-Based Diterpenoids from a Formosan Soft Coral Cladiella sp." Marine Drugs 11, no. 11: 4585-4593. https://doi.org/10.3390/md11114585
APA StyleChen, T.-H., Lu, M.-C., Chang, Y.-C., Su, Y.-D., Chen, Y.-H., Lin, N.-C., Fang, L.-S., Wu, Y.-C., & Sung, P.-J. (2013). Discovery of New Eunicellin-Based Diterpenoids from a Formosan Soft Coral Cladiella sp. Marine Drugs, 11(11), 4585-4593. https://doi.org/10.3390/md11114585







