The Association of Human Parvovirus B19 Infection on the Course of Vietnamese Patients with Rheumatoid Arthritis
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Consent and Ethical Approval
2.2. Data Collection and Study Design
2.3. RA Diagnosis and Clinical Evaluation Indices
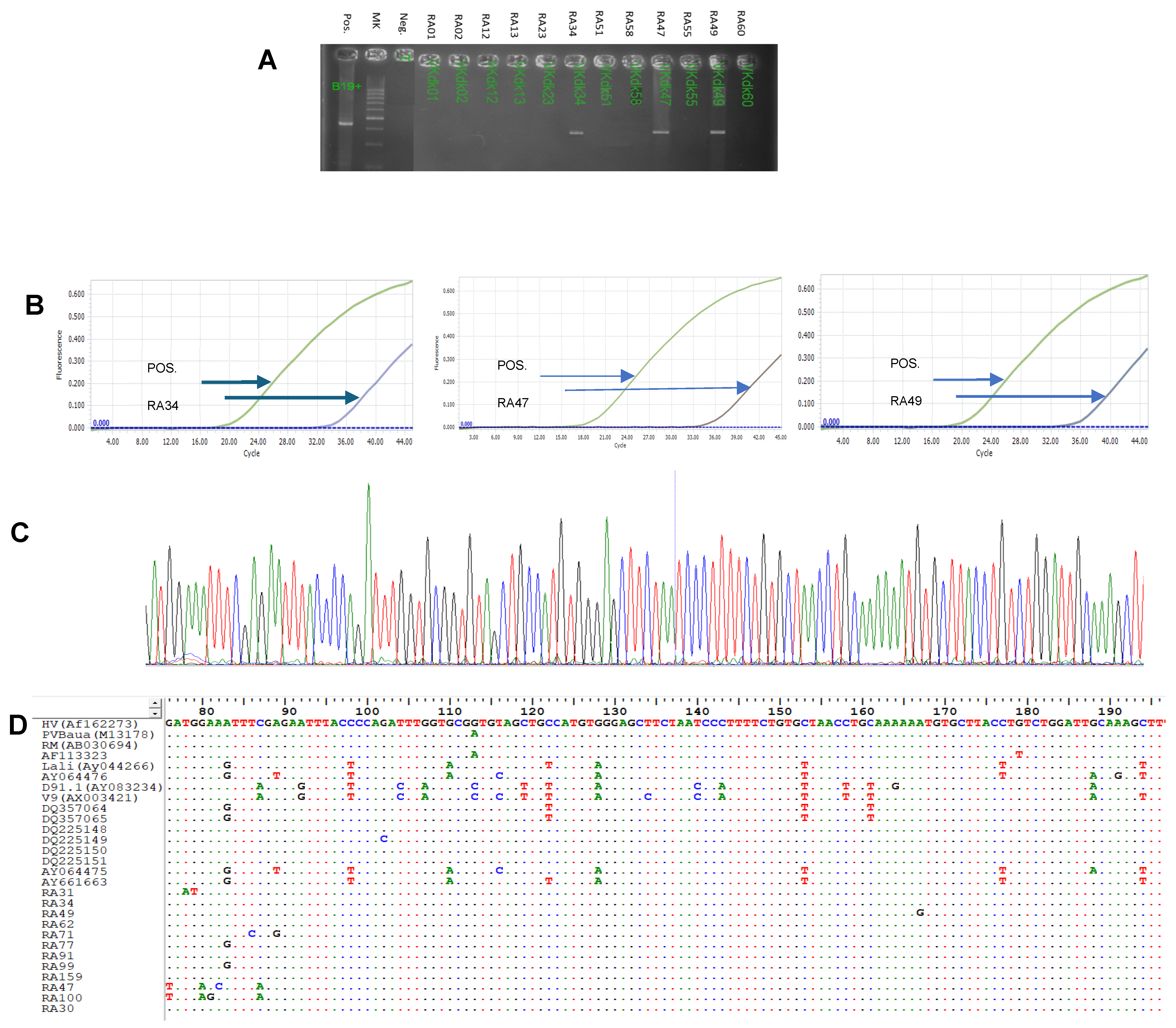
2.4. Detection of B19V DNA and DNA Sequence Analysis
2.5. Detect Anti-B19V Antibodies in Serum
2.6. B19V Genotype Analysis
2.7. Statistical Analysis
3. Results
3.1. Clinical Manifestations of RA Patients
3.2. The Prevalence of B19V Genome in RA Patients and Healthy Controls
3.3. The Seropositive Rate of Anti-B19V IgM and IgG
3.4. The Genetic Distribution of B19V Among Vietnamese RA Patients
3.5. B19V Infection Associated with RA Progression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Di Matteo, A.; Bathon, J.M.; Emery, P. Rheumatoid arthritis. Lancet 2023, 402, 2019–2033. [Google Scholar] [CrossRef]
- Shi, G.; Liao, X.; Lin, Z.; Liu, W.; Luo, X.; Zhan, H.; Cai, X. Estimation of the global prevalence, incidence, years lived with disability of rheumatoid arthritis in 2019 and forecasted incidence in 2040: Results from the Global Burden of Disease Study 2019. Clin. Rheumatol. 2023, 42, 2297–2309. [Google Scholar] [CrossRef] [PubMed]
- Venetsanopoulou, A.I.; Alamanos, Y.; Voulgari, P.V.; Drosos, A.A. Epidemiology and Risk Factors for Rheumatoid Arthritis Development. Mediterr. J. Rheumatol. 2023, 34, 404–413. [Google Scholar] [CrossRef] [PubMed]
- Videm, V.; Liff, M.H.; Hoff, M. Relative importance of inflammation and cardiorespiratory fitness for all-cause mortality risk in persons with rheumatoid arthritis: The population-based Trondelag Health Study. RMD Open 2023, 9, e003194. [Google Scholar] [CrossRef] [PubMed]
- Van den Hoek, J.; Boshuizen, H.C.; Roorda, L.D.; Tijhuis, G.J.; Nurmohamed, M.T.; van den Bos, G.A.; Dekker, J. Mortality in patients with rheumatoid arthritis: A 15-year prospective cohort study. Rheumatol. Int. 2017, 37, 487–493. [Google Scholar] [CrossRef]
- Symmons, D.P. Epidemiology of rheumatoid arthritis: Determinants of onset, persistence and outcome. Best Pract. Res. Clin. Rheumatol. 2002, 16, 707–722. [Google Scholar] [CrossRef]
- Munir, A.; Khan, S.; Saleem, A.; Nusrat, H.; Khan, S.A.; Sayyed, H.; Khalid, A.; Javed, B.; Hidayat, F. The Role of Epstein-Barr Virus Molecular Mimicry in Various Autoimmune Diseases. Scand. J. Immunol. 2025, 101, e70016. [Google Scholar] [CrossRef]
- Ishii, K.K.; Takahashi, Y.; Kaku, M.; Sasaki, T. Role of human parvovirus B19 in the pathogenesis of rheumatoid arthritis. Jpn. J. Infect. Dis. 1999, 52, 201–207. [Google Scholar] [CrossRef]
- Kishore, J.; Kishore, D. Clinical impact & pathogenic mechanisms of human parvovirus B19: A multiorgan disease inflictor incognito. Indian J. Med. Res. 2018, 148, 373–384. [Google Scholar] [CrossRef]
- Pattison, J.R. B19 virus—A pathogenic human parvovirus. Blood Rev. 1987, 1, 58–64. [Google Scholar] [CrossRef]
- Young, N.S.; Brown, K.E. Parvovirus B19. N. Engl. J. Med. 2004, 350, 586–597. [Google Scholar] [CrossRef]
- Jacquot, R.; Gerfaud-Valentin, M.; Mekki, Y.; Billaud, G.; Jamilloux, Y.; Seve, P. [Parvovirus B19 infections in adults]. Rev. Med. Interne 2022, 43, 713–726. [Google Scholar] [CrossRef]
- Marks, M.; Marks, J.L. Viral arthritis. Clin. Med. 2016, 16, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J.R. Pathogenesis of parvovirus B19 infection: Host gene variability, and possible means and effects of virus persistence. J. Vet. Med. B Infect. Dis. Vet. Public Health 2005, 52, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Soderlund, M.; von Essen, R.; Haapasaari, J.; Kiistala, U.; Kiviluoto, O.; Hedman, K. Persistence of parvovirus B19 DNA in synovial membranes of young patients with and without chronic arthropathy. Lancet 1997, 349, 1063–1065. [Google Scholar] [CrossRef] [PubMed]
- Kvien, T.K.; Uhlig, T.; Odegard, S.; Heiberg, M.S. Epidemiological aspects of rheumatoid arthritis: The sex ratio. Ann. N. Y. Acad. Sci. 2006, 1069, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Sharma, A. Infectious mimics of rheumatoid arthritis. Best Pract. Res. Clin. Rheumatol. 2022, 36, 101736. [Google Scholar] [CrossRef]
- Banal, F.; Dougados, M.; Combescure, C.; Gossec, L. Sensitivity and specificity of the American College of Rheumatology 1987 criteria for the diagnosis of rheumatoid arthritis according to disease duration: A systematic literature review and meta-analysis. Ann. Rheum. Dis. 2009, 68, 1184–1191. [Google Scholar] [CrossRef]
- Wewers, M.E.; Lowe, N.K. A critical review of visual analogue scales in the measurement of clinical phenomena. Res. Nurs. Health 1990, 13, 227–236. [Google Scholar] [CrossRef]
- Arya, V.; Malaviya, A.N.; Raja, R.R. CDAI (clinical disease activity index) in rheumatoid arthritis: Cut-off values for classification into different grades of disease activity. Indian J. Rheumatol. 2007, 2, 91–94. [Google Scholar] [CrossRef]
- Aletaha, D.; Smolen, J. The Simplified Disease Activity Index (SDAI) and the Clinical Disease Activity Index (CDAI): A review of their usefulness and validity in rheumatoid arthritis. Clin. Exp. Rheumatol. 2005, 23 (Suppl. S39), S100–S108. [Google Scholar] [PubMed]
- van Riel, P.L.; Renskers, L. The Disease Activity Score (DAS) and the Disease Activity Score using 28 joint counts (DAS28) in the management of rheumatoid arthritis. Clin. Exp. Rheumatol. 2016, 34 (Suppl. S101), S40–S44. [Google Scholar] [PubMed]
- van Riel, P.L. The development of the disease activity score (DAS) and the disease activity score using 28 joint counts (DAS28). Clin. Exp. Rheumatol. 2014, 32 (Suppl. S85), S65–S74. [Google Scholar]
- Sengul, I.; Akcay-Yalbuzdag, S.; Ince, B.; Goksel-Karatepe, A.; Kaya, T. Comparison of the DAS28-CRP and DAS28-ESR in patients with rheumatoid arthritis. Int. J. Rheum. Dis. 2015, 18, 640–645. [Google Scholar] [CrossRef]
- Toan, N.L.; Duechting, A.; Kremsner, P.G.; Song, L.H.; Ebinger, M.; Aberle, S.; Binh, V.Q.; Duy, D.N.; Torresi, J.; Kandolf, R.; et al. Phylogenetic analysis of human parvovirus B19, indicating two subgroups of genotype 1 in Vietnamese patients. J. Gen. Virol. 2006, 87 Pt 10, 2941–2949. [Google Scholar] [CrossRef]
- Bultmann, B.D.; Klingel, K.; Sotlar, K.; Bock, C.T.; Baba, H.A.; Sauter, M.; Kandolf, R. Fatal parvovirus B19-associated myocarditis clinically mimicking ischemic heart disease: An endothelial cell-mediated disease. Hum. Pathol. 2003, 34, 92–95. [Google Scholar] [CrossRef]
- Figus, F.A.; Piga, M.; Azzolin, I.; McConnell, R.; Iagnocco, A. Rheumatoid arthritis: Extra-articular manifestations and comorbidities. Autoimmun. Rev. 2021, 20, 102776. [Google Scholar] [CrossRef]
- Wu, D.; Luo, Y.; Li, T.; Zhao, X.; Lv, T.; Fang, G.; Ou, P.; Li, H.; Luo, X.; Huang, A.; et al. Systemic complications of rheumatoid arthritis: Focus on pathogenesis and treatment. Front. Immunol. 2022, 13, 1051082. [Google Scholar] [CrossRef]
- Taylor, P.C.; Atzeni, F.; Balsa, A.; Gossec, L.; Muller-Ladner, U.; Pope, J. The Key Comorbidities in Patients with Rheumatoid Arthritis: A Narrative Review. J. Clin. Med. 2021, 10, 509. [Google Scholar] [CrossRef]
- Pradeepkiran, J.A. Insights of rheumatoid arthritis risk factors and associations. J. Transl. Autoimmun. 2019, 2, 100012. [Google Scholar] [CrossRef]
- Alamanos, Y.; Drosos, A.A. Epidemiology of adult rheumatoid arthritis. Autoimmun. Rev. 2005, 4, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Yang, M.; Fu, H.; Ma, N.; Wei, T.; Tang, Q.; Hu, Z.; Liang, Y.; Yang, Z.; Zhong, R. Body mass index and the risk of rheumatoid arthritis: A systematic review and dose-response meta-analysis. Arthritis Res. Ther. 2015, 17, 86. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Xu, X.; Shi, Y.; Liu, X.; Liu, H.; Hou, H.; Ji, L.; Li, Y.; Wang, W.; Wang, Y.; et al. Body Mass Index and the Risk of Rheumatoid Arthritis: An Updated Dose-Response Meta-Analysis. Biomed. Res. Int. 2019, 2019, 3579081. [Google Scholar] [CrossRef]
- Vukovic, V.; Patic, A.; Ristic, M.; Kovacevic, G.; Hrnjakovic Cvjetkovic, I.; Petrovic, V. Seroepidemiology of Human Parvovirus B19 Infection among the Population of Vojvodina, Serbia, over a 16-Year Period (2008–2023). Viruses 2024, 16, 180. [Google Scholar] [CrossRef]
- Huatuco, E.M.; Durigon, E.L.; Lebrun, F.L.; Passos, S.D.; Gazeta, R.E.; Azevedo Neto, R.S.; Massad, E. Seroprevalence of human parvovirus B19 in a suburban population in Sao Paulo, Brazil. Rev. Saude Publica 2008, 42, 443–449. [Google Scholar] [CrossRef]
- Healy, K.; Aulin, L.B.S.; Freij, U.; Ellerstad, M.; Bruckle, L.; Hillmering, H.; Svae, T.E.; Broliden, K.; Gustafsson, R. Prevalence of Parvovirus B19 Viremia Among German Blood Donations and the Relationship to ABO and Rhesus Blood Group Antigens. J. Infect. Dis. 2023, 227, 1214–1218. [Google Scholar] [CrossRef]
- Russcher, A.; van Boven, M.; Beninca, E.; Verweij, E.; Molenaar-de Backer, M.W.A.; Zaaijer, H.L.; Vossen, A.; Kroes, A.C.M. Changing epidemiology of parvovirus B19 in the Netherlands since 1990, including its re-emergence after the COVID-19 pandemic. Sci. Rep. 2024, 14, 9630. [Google Scholar] [CrossRef]
- Toan, N.L.; Sy, B.T.; Song, L.H.; Luong, H.V.; Binh, N.T.; Binh, V.Q.; Kandolf, R.; Velavan, T.P.; Kremsner, P.G.; Bock, C.T. Co-infection of human parvovirus B19 with Plasmodium falciparum contributes to malaria disease severity in Gabonese patients. BMC Infect. Dis. 2013, 13, 375. [Google Scholar] [CrossRef]
- Toan, N.L.; Song, L.H.; Kremsner, P.G.; Duy, D.N.; Binh, V.Q.; Duechting, A.; Kaiser, H.; Torresi, J.; Kandolf, R.; Bock, C.T. Co-infection of human parvovirus B19 in Vietnamese patients with hepatitis B virus infection. J. Hepatol. 2006, 45, 361–369. [Google Scholar] [CrossRef]
- Zakrzewska, K.; Azzi, A.; De Biasi, E.; Radossi, P.; De Santis, R.; Davoli, P.G.; Tagariello, G. Persistence of parvovirus B19 DNA in synovium of patients with haemophilic arthritis. J. Med. Virol. 2001, 65, 402–407. [Google Scholar] [CrossRef]
- Ouranos, K.; Avila, D.V.; Mylona, E.K.; Vassilopoulos, A.; Vassilopoulos, S.; Shehadeh, F.; Mylonakis, E. Cumulative incidence and risk of infection in patients with rheumatoid arthritis treated with janus kinase inhibitors: A systematic review and meta-analysis. PLoS ONE 2024, 19, e0306548. [Google Scholar] [CrossRef]
- Kerr, J.R. Pathogenesis of human parvovirus B19 in rheumatic disease. Ann. Rheum. Dis. 2000, 59, 672–683. [Google Scholar] [CrossRef] [PubMed]
- Arvia, R.; Stincarelli, M.A.; Manaresi, E.; Gallinella, G.; Zakrzewska, K. Parvovirus B19 in Rheumatic Diseases. Microorganisms 2024, 12, 1708. [Google Scholar] [CrossRef]
- Jiang, H.; Qiu, Q.; Zhou, Y.; Zhang, Y.; Xu, W.; Cui, A.; Li, X. The epidemiological and genetic characteristics of human parvovirus B19 in patients with febrile rash illnesses in China. Sci. Rep. 2023, 13, 15913. [Google Scholar] [CrossRef]


| Clinical Symptoms | Signs | N | % |
|---|---|---|---|
| Joint pain level | No pain | 0 | 0 |
| Mild | 17 | 14.78 | |
| Medium | 62 | 53.91 | |
| Strong | 36 | 31.31 | |
| Knee effusion | Positive | 68 | 59.13 |
| Negative | 47 | 40.87 | |
| Morning stiffness | ≥1 h | 80 | 69.57 |
| <1 h | 35 | 30.43 | |
| CRP (mg/L) | Increased | 104 | 90.43 |
| RF (UI/L) | Increased | 98 | 85.22 |
| anti-CCP (UI/L) | Increased | 102 | 88.70 |
| Steinbrocker stages | No lesion | 13 | 11.30 |
| Stage 1 | 45 | 39.13 | |
| Stage 2 | 27 | 23.48 | |
| Stage 3 | 19 | 16.52 | |
| Stage 4 | 11 | 9.57 | |
| DAS 28_CRP | <2.6 | 1 | 0.87 |
| 2.6–3.2 | 4 | 3.48 | |
| 3.2–5.1 | 55 | 47.83 | |
| >5.1 | 55 | 47.83 | |
| DAS 28_ESR | <2.6 | 0 | 0 |
| 2.6–3.2 | 1 | 0.87 | |
| 3.2–5.1 | 49 | 42.61 | |
| >5.1 | 65 | 56.52 | |
| CDAI | <2.8 | 0 | 0 |
| 2.8–10 | 7 | 6.1 | |
| 10–22 | 51 | 44.3 | |
| >22 | 57 | 49.6 | |
| SDAI | <3.3 | 0 | 0 |
| 3.3–11.0 | 1 | 0.87 | |
| 11.0–26 | 14 | 12.17 | |
| >26 | 100 | 86.96 | |
| Disease duration (years) | <5 | 68 | 59.13 |
| 5–10 | 26 | 22.61 | |
| >10 | 21 | 18.26 |
| Anti-B19V Status | HC (n = 86) | RA (n = 115) | Statistics |
|---|---|---|---|
| n (%) | n (%) | ||
| anti-IgG(+), −IgM(−) | 21 (24.42) | 49 (42.60) | OR = 2.31; 95%CI: 1.21–4.48; p < 0.01 |
| anti-IgM(+),−IgG(−) | 11 (12.79) | 3 (2.60) | OR = 0.18; 95%CI: 0.03–0.78; p < 0.01 |
| anti-IgG(+) and/or anti-IgM(+) | 22 (25.58) | 51 (44.34) | OR = 2.3; 95%CI: 1.19–4.49; p < 0.01 |
| Anti-B19V Status | OR | 95%CI | p |
|---|---|---|---|
| anti-IgG(+),−IgM(−) | 5.48 | 1.48–20.29 | 0.011 |
| anti-IgM(+), −IgG(−) | 0.44 | 0.24–0.81 | 0.008 |
| anti-IgM(+), and/or −IgG(+) | 3.385 | 1.18–22.55 | 0.03 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manh, T.V.; Nguyen, M.L.T.; Hang, N.T.; Giang, N.T.; Mao, C.V.; Binh, L.T.; Diep, N.T.; Sy, B.T.; Huyen, T.T.T.; Ha, V.N.; et al. The Association of Human Parvovirus B19 Infection on the Course of Vietnamese Patients with Rheumatoid Arthritis. Medicina 2025, 61, 1546. https://doi.org/10.3390/medicina61091546
Manh TV, Nguyen MLT, Hang NT, Giang NT, Mao CV, Binh LT, Diep NT, Sy BT, Huyen TTT, Ha VN, et al. The Association of Human Parvovirus B19 Infection on the Course of Vietnamese Patients with Rheumatoid Arthritis. Medicina. 2025; 61(9):1546. https://doi.org/10.3390/medicina61091546
Chicago/Turabian StyleManh, Trieu Van, Mai Ly Thi Nguyen, Ngo Thu Hang, Ngo Truong Giang, Can Van Mao, Luu Thi Binh, Nguy Thi Diep, Bui Tien Sy, Tran Thi Thanh Huyen, Vu Nhi Ha, and et al. 2025. "The Association of Human Parvovirus B19 Infection on the Course of Vietnamese Patients with Rheumatoid Arthritis" Medicina 61, no. 9: 1546. https://doi.org/10.3390/medicina61091546
APA StyleManh, T. V., Nguyen, M. L. T., Hang, N. T., Giang, N. T., Mao, C. V., Binh, L. T., Diep, N. T., Sy, B. T., Huyen, T. T. T., Ha, V. N., Cuong, L. D., Bui, K. C., Tong, H. V., & Toan, N. L. (2025). The Association of Human Parvovirus B19 Infection on the Course of Vietnamese Patients with Rheumatoid Arthritis. Medicina, 61(9), 1546. https://doi.org/10.3390/medicina61091546






