Total Body Irradiation in Haematopoietic Stem Cell Transplantation: A Comprehensive Literature Review and Institutional Experience from the Policlinico of Catania
Abstract
1. Introduction
2. TBI Indications and Integration with Systemic Therapy
3. Technological Advancements in TBI
3.1. From Isotopes to Linear Accelerators
3.2. Intensity Modulated Total Body Irradiation
4. Ongoing Challenges and Future Directions
4.1. Cellular Hallmarks: Key Considerations
4.2. Ongoing Clinical Trials
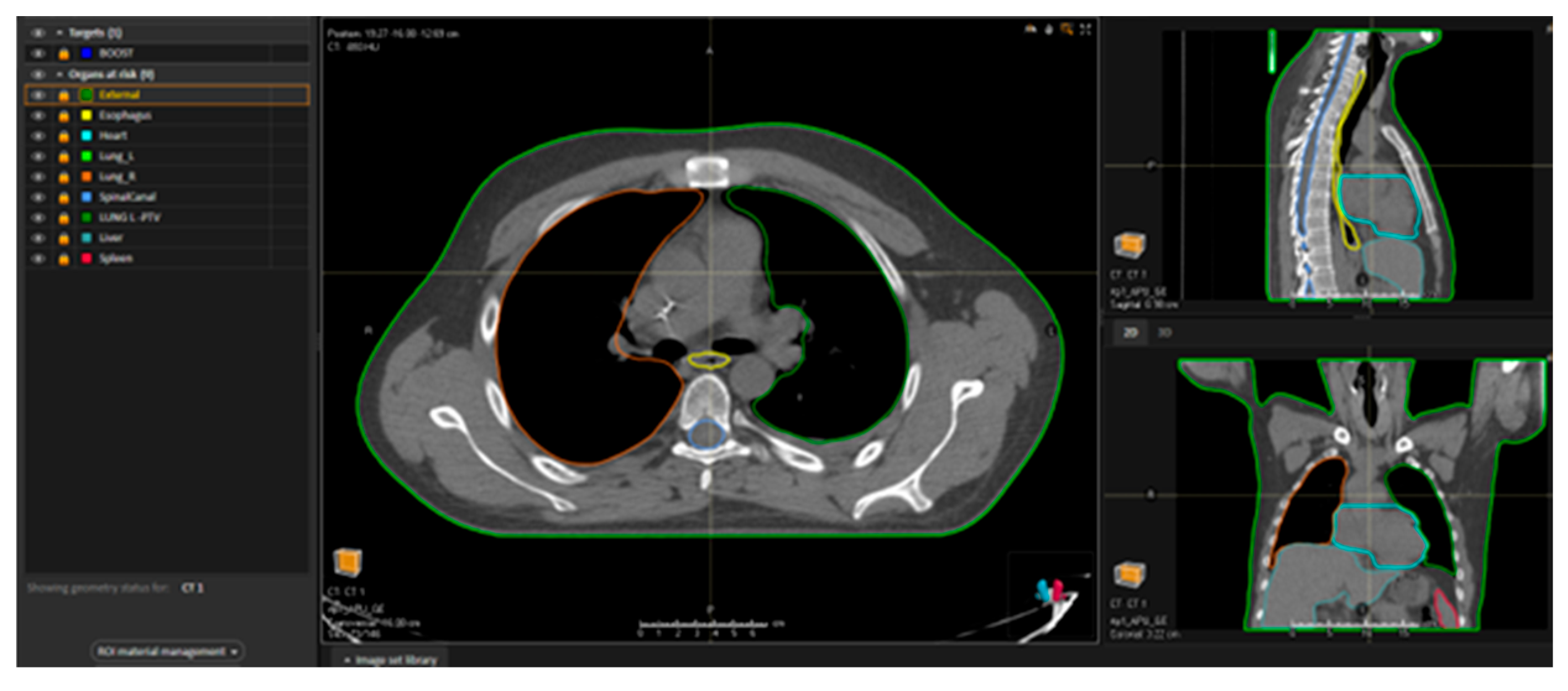
5. Experience at the University Hospital Policlinico G. Rodolico San Marco
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Paix, A.; Antoni, D.; Waissi, W.; Ledoux, M.P.; Bilger, K.; Fornecker, L.; Noel, G. Total body irradiation in allogeneic bone marrow transplantation conditioning regimens: A review. Crit. Rev. Oncol./Hematol. 2018, 123, 138–148. [Google Scholar] [CrossRef]
- Blume, K.G.; Weissman, I.L. E. Donnall Thomas (1920–2012). Proc. Natl. Acad. Sci. USA 2012, 109, 20777–20778. [Google Scholar] [CrossRef] [PubMed]
- Johns, H.E.; Bates, L.M.; Epp, E.R.; Cormack, D.V.; Fedorux, S.O.; Morrison, A.; Dixon, W.R.; Garrett, C. 1,000-Curie Cobalt 60 Units for Radiation Therapy. Nature 1951, 168, 1035–1036. [Google Scholar] [CrossRef]
- Sabloff, M.; Tisseverasinghe, S.; Babadagli, M.E.; Samant, R. Total Body Irradiation for Hematopoietic Stem Cell Transplantation: What Can We Agree on? Curr. Oncol. 2021, 28, 903–917. [Google Scholar] [CrossRef]
- Malicki, J.; Kosicka, G.; Stryczyńska, G.; Wachowiak, J. Cobalt 60 versus 15 MeV photons during total body irradiation: Doses in the critical organs and complexicity of the procedure. Ann. Transplant. 2001, 6, 18–22. [Google Scholar]
- Chiang, Y.; Tsai, C.H.; Kuo, S.H.; Liu, C.Y.; Yao, M.; Li, C.C.; Huang, S.Y.; Ko, B.S.; Lin, C.T.; Hou, H.A.; et al. Reduced incidence of interstitial pneumonitis after allogeneic hematopoietic stem cell transplantation using a modified technique of total body irradiation. Sci. Rep. 2016, 6, 36730. [Google Scholar] [CrossRef]
- Chilukuri, S.; Sundar, S.; Thiyagarajan, R.; Easow, J.; Sawant, M.; Krishanan, G.; Panda, P.K.; Sharma, D.; Jalali, R. Total marrow and lymphoid irradiation with helical tomotherapy: A practical implementation report. Radiat. Oncol. J. 2020, 38, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Hoeben, B.A.W.; Wong, J.Y.C.; Fog, L.S.; Losert, C.; Filippi, A.R.; Bentzen, S.M.; Balduzzi, A.; Specht, L. Total Body Irradiation in Haematopoietic Stem Cell Transplantation for Paediatric Acute Lymphoblastic Leukaemia: Review of the Literature and Future Directions. Front. Pediatr. 2021, 9, 774348. [Google Scholar] [CrossRef] [PubMed]
- Heinzelmann, F.; Ottinger, H.; Müller, C.H.; Allgaier, S.; Faul, C.; Bamberg, M.; Belka, C. Total-body irradiation–Role and indications: Results from the German Registry for Stem Cell Transplantation (DRST). Strahlenther. Onkol. 2006, 182, 222–230. [Google Scholar] [CrossRef]
- Jung, J.; Lee, H.; Suh, Y.G.; Eom, H.S.; Lee, E. Current Use of Total Body Irradiation in Haploidentical Allogeneic Hematopoietic Stem Cell Transplantation. J. Korean Med. Sci. 2021, 36, e55. [Google Scholar] [CrossRef]
- Thomas, E.D.; Clift, R.A.; Hersman, J.; Sanders, J.E.; Stewart, P.; Buckner, C.D.; Fefer, A.; McGuffin, R.; Smith, J.W.; Storb, R. Marrow transplantation for acute nonlymphoblastic leukemic in first remission using fractionated or single-dose irradiation. Int. J. Radiat. Oncol. Biol. Phys. 1982, 8, 817–821. [Google Scholar] [CrossRef] [PubMed]
- Shank, B.; Chu, F.C.; Dinsmore, R.; Kapoor, N.; Kirkpatrick, D.; Teitelbaum, H.; Reid, A.; Bonfiglio, P.; Simpson, L.; O’Reilly, R.J. Hyperfractionated total body irradiation for bone marrow transplantation. Results in seventy leukemia patients with allogeneic transplants. Int. J. Radiat. Oncol. Biol. Phys. 1983, 9, 1607–1611. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.Y.C.; Filippi, A.R.; Dabaja, B.S.; Yahalom, J.; Specht, L. Total Body Irradiation: Guidelines from the International Lymphoma Radiation Oncology Group (ILROG). Int. J. Radiat. Oncol. Biol. Phys. 2018, 101, 521–529. [Google Scholar] [CrossRef]
- Blomain, E.S.; Jiang, A.; Donaldson, S.S.; Agarwal, R.; Bertaina, A.; Shyr, D.; Eisenberg, M.L.; Hoppe, R.T.; Hiniker, S.M.; Oh, J. Effect of Testicular Boost in Children with Leukemia Receiving Total Body Irradiation and Stem Cell Transplant: A Single-Institution Experience. Adv. Radiat. Oncol. 2022, 8, 101071. [Google Scholar] [CrossRef]
- Bruserud, O.; Reikvam, H.; Kittang, A.O.; Ahmed, A.B.; Tvedt, T.H.; Sjo, M.; Hatfield, K.J. High-dose etoposide in allogeneic stem cell transplantation. Cancer Chemother. Pharmacol. 2012, 70, 765–782. [Google Scholar] [CrossRef] [PubMed]
- Bunin, N.; Aplenc, R.; Kamani, N.; Shaw, K.; Cnaan, A.; Simms, S. Randomized trial of busulfan vs total body irradiation containing conditioning regimens for children with acute lymphoblastic leukemia: A Pediatric Blood and Marrow Transplant Consortium study. Bone Marrow Transplant. 2003, 32, 543–548. [Google Scholar] [CrossRef]
- Davies, S.M.; Ramsay, N.K.; Klein, J.P.; Weisdorf, D.J.; Bolwell, B.; Cahn, J.Y.; Camitta, B.M.; Gale, R.P.; Giralt, S.; Heilmann, C.; et al. Comparison of preparative regimens in transplants for children with acute lymphoblastic leukemia. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2000, 18, 340–347. [Google Scholar] [CrossRef]
- Peters, C.; Dalle, J.H.; Locatelli, F.; Poetschger, U.; Sedlacek, P.; Buechner, J.; Shaw, P.J.; Staciuk, R.; Ifversen, M.; Pichler, H.; et al. Total Body Irradiation or Chemotherapy Conditioning in Childhood ALL: A Multinational, Randomized, Noninferiority Phase III Study. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2021, 39, 295–307. [Google Scholar] [CrossRef]
- Ballen, K.K.; Becker, P.S.; Emmons, R.V.; Fitzgerald, T.J.; Hsieh, C.C.; Liu, Q.; Heyes, C.; Clark, Y.; Levy, W.; Lambert, J.F.; et al. Low-dose total body irradiation followed by allogeneic lymphocyte infusion may induce remission in patients with refractory hematologic malignancy. Blood 2002, 100, 442–450. [Google Scholar] [CrossRef][Green Version]
- Ballen, K.K.; Colvin, G.; Porter, D.; Quesenberry, P.J. Low dose total body irradiation followed by allogeneic lymphocyte infusion for refractory hematologic malignancy—An updated review. Leuk. Lymphoma 2004, 45, 905–910. [Google Scholar] [CrossRef]
- McSweeney, P.A.; Niederwieser, D.; Shizuru, J.A.; Sandmaier, B.M.; Molina, A.J.; Maloney, D.G.; Chauncey, T.R.; Gooley, T.A.; Hegenbart, U.; Nash, R.A.; et al. Hematopoietic cell transplantation in older patients with hematologic malignancies: Replacing high-dose cytotoxic therapy with graft-versus-tumor effects. Blood 2001, 97, 3390–3400. [Google Scholar] [CrossRef] [PubMed]
- Kornblit, B.; Maloney, D.G.; Storb, R.; Storek, J.; Hari, P.; Vucinic, V.; Maziarz, R.T.; Chauncey, T.R.; Pulsipher, M.A.; Bruno, B.; et al. Fludarabine and 2-Gy TBI is superior to 2 Gy TBI as conditioning for HLA-matched related hematopoietic cell transplantation: A phase III randomized trial. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2013, 19, 1340–1347. [Google Scholar] [CrossRef]
- Modi, D.; Chi, J.; Kim, S.; Deol, A.; Ayash, L.; Ratanatharathorn, V.; Uberti, J.P. Outcomes of Fludarabine, Melphalan and Total Body Irradiation as a Reduced Intensity Conditioning Regimen in Matched Donor Allogeneic Peripheral Blood Stem Cell Transplantation. Transplant. Cell. Ther. 2021, 27, 665.e1–665.e7. [Google Scholar] [CrossRef]
- Nakamae, H.; Storer, B.E.; Storb, R.; Storek, J.; Chauncey, T.R.; Pulsipher, M.A.; Petersen, F.B.; Wade, J.C.; Maris, M.B.; Bruno, B.; et al. Low-dose total body irradiation and fludarabine conditioning for HLA class I-mismatched donor stem cell transplantation and immunologic recovery in patients with hematologic malignancies: A multicenter trial. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2010, 16, 384–394. [Google Scholar] [CrossRef]
- Jacobs, M.L.; Pape, L. Dosimetry for a total-body irradiation chamber. Radiology 1961, 77, 788–792. [Google Scholar] [CrossRef] [PubMed]
- Sahler, O.D. Development of a room specifically designed for total-body irradiation. Radiology 1959, 72, 266–267. [Google Scholar] [CrossRef]
- Leung, P.M.; Rider, W.D.; Webb, H.P.; Aget, H.; Johns, H.E. Cobalt-60 therapy unit for large field irradiation. Int. J. Radiat. Oncol. Biol. Phys. 1981, 7, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Peters, V.G.; Herer, A.S. Modification of a standard cobalt-60 unit for total body irradiation at 150 cm SSD. Int. J. Radiat. Oncol. Biol. Phys. 1984, 10, 927–932. [Google Scholar] [CrossRef]
- Quast, U. Physical treatment planning of total-body irradiation: Patient translation and beam-zone method. Med. Phys. 1985, 12, 567–574. [Google Scholar] [CrossRef]
- Pla, M.; Chenery, S.G.; Podgorsak, E.B. Total body irradiation with a sweeping beam. Int. J. Radiat. Oncol. Biol. Phys. 1983, 9, 83–89. [Google Scholar] [CrossRef]
- Litoborska, J.; Piotrowski, T.; Jodda, A.; Malicki, J. Evolution of treatment planning and dose delivery methods during radiotherapy for patients undergoing bone marrow transplantation: A review. Nukleonika 2020, 65, 19–30. [Google Scholar] [CrossRef]
- Girinsky, T.; Socie, G.; Ammarguellat, H.; Cosset, J.M.; Briot, E.; Bridier, A.; Gluckman, E. Consequences of two different doses to the lungs during a single dose of total body irradiation: Results of a randomized study on 85 patients. Int. J. Radiat. Oncol. Biol. Phys. 1994, 30, 821–824. [Google Scholar] [CrossRef]
- Mekdash, H.; Shahine, B.; Jalbout, W.; Chehab, C.; Abdel Khalek, H.; Youssef, B. A simple technique for an accurate shielding of the lungs during total body irradiation. Tech. Innov. Patient Support Radiat. Oncol. 2017, 3–4, 13–18. [Google Scholar] [CrossRef]
- Zeverino, M.; Agostinelli, S.; Taccini, G.; Cavagnetto, F.; Garelli, S.; Gusinu, M.; Vagge, S.; Barra, S.; Corvò, R. Advances in the implementation of helical tomotherapy-based total marrow irradiation with a novel field junction technique. Med. Dosim. Off. J. Am. Assoc. Med. Dosim. 2012, 37, 314–320. [Google Scholar] [CrossRef]
- Hui, S.K.; Kapatoes, J.; Fowler, J.; Henderson, D.; Olivera, G.; Manon, R.R.; Gerbi, B.; Mackie, T.R.; Welsh, J.S. Feasibility study of helical tomotherapy for total body or total marrow irradiation. Med. Phys. 2005, 32, 3214–3224. [Google Scholar] [CrossRef]
- Hui, C.; Simiele, E.; Lozko, Y.; Romero, I.; Skinner, L.; Binkley, M.S.; Hoppe, R.; Kovalchuk, N.; Hiniker, S.M. Volumetric modulated arc therapy total body irradiation improves toxicity outcomes compared to 2D total body irradiation. Front. Oncol. 2024, 14, 1459287. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.Y.C.; Filippi, A.R.; Scorsetti, M.; Hui, S.; Muren, L.P.; Mancosu, P. Total marrow and total lymphoid irradiation in bone marrow transplantation for acute leukaemia. Lancet. Oncol. 2020, 21, e477–e487. [Google Scholar] [CrossRef] [PubMed]
- Aydogan, B.; Mundt, A.J.; Roeske, J.C. Linac-based intensity modulated total marrow irradiation (IM-TMI). Technol. Cancer Res. Treat. 2006, 5, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Dogliotti, I.; Levis, M.; Martin, A.; Bartoncini, S.; Felicetti, F.; Cavallin, C.; Maffini, E.; Cerrano, M.; Bruno, B.; Ricardi, U.; et al. Maintain Efficacy and Spare Toxicity: Traditional and New Radiation-Based Conditioning Regimens in Hematopoietic Stem Cell Transplantation. Cancers 2024, 16, 865. [Google Scholar] [CrossRef] [PubMed]
- Loginova, A.A.; Tovmasian, D.A.; Lisovskaya, A.O.; Kobyzeva, D.A.; Maschan, M.A.; Chernyaev, A.P.; Egorov, O.B.; Nechesnyuk, A.V. Optimized Conformal Total Body Irradiation methods with Helical TomoTherapy and Elekta VMAT: Implementation, Imaging, Planning and Dose Delivery for Pediatric Patients. Front. Oncol. 2022, 12, 785917. [Google Scholar] [CrossRef]
- Salz, H.; Bohrisch, B.; Howitz, S.; Banz, N.; Weibert, K.; Wiezorek, T.; Wendt, T.G. Intensity-modulated Total Body Irradiation (TBI) with TomoDirect™. Radiat. Oncol. 2015, 10, 58. [Google Scholar] [CrossRef] [PubMed]
- Hoeben, B.A.W.; Saldi, S.; Aristei, C.; Engellau, J.; Ocanto, A.; Hiniker, S.M.; Misson-Yates, S.; Kobyzeva, D.A.; Pazos, M.; George Mikhaeel, N.; et al. Rationale, implementation considerations, delineation and planning target objective recommendations for volumetric modulated arc therapy and helical tomotherapy total body irradiation, total marrow irradiation, total marrow and lymphoid irradiation and total lymphoid irradiation. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 2025, 206, 110822. [Google Scholar] [CrossRef]
- Amin, A.E.; Wheldon, T.E.; O’Donoghue, J.A.; Barrett, A. Radiobiological modeling of combined targeted 131I therapy and total body irradiation for treatment of disseminated tumors of differing radiosensitivity. Int. J. Radiat. Oncol. Biol. Phys. 1993, 27, 323–330. [Google Scholar] [CrossRef]
- Wheldon, T.E. The radiobiological basis of total body irradiation. Br. J. Radiol. 1997, 70, 1204–1207. [Google Scholar] [CrossRef]
- Bosman, F.T.; Visser, B.C.; van Oeveren, J. Apoptosis: Pathophysiology of programmed cell death. Pathol. Res. Pract. 1996, 192, 676–683. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, T.; Su, W.; Dou, Z.; Zhao, D.; Jin, X.; Lei, H.; Wang, J.; Xie, X.; Cheng, B.; et al. Mutant p53 in cancer: From molecular mechanism to therapeutic modulation. Cell Death Dis. 2022, 13, 974. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, F.; Zhu, Q.; Ding, B.; Zhong, Q.; Huang, K.; Jiang, X.; Wang, Z.; Yin, C.; Zhu, Y.; et al. Gli-1/PI3K/AKT/NF-kB pathway mediates resistance to radiation and is a target for reversion of responses in refractory acute myeloid leukemia cells. Oncotarget 2016, 7, 33004–33015. [Google Scholar] [CrossRef] [PubMed]
- Uckun, F.M.; Chandan-Langlie, M.; Jaszcz, W.; Obuz, V.; Waddick, K.; Song, C.W. Radiation damage repair capacity of primary clonogenic blasts in acute lymphoblastic leukemia. Cancer Res. 1993, 53, 1431–1436. [Google Scholar]
- Lo Greco, M.C.; Marletta, G.; Marano, G.; Fazio, A.; Buffettino, E.; Iudica, A.; Liardo, R.L.E.; Milazzotto, R.; Foti, P.V.; Palmucci, S.; et al. Hypofractionated Radiotherapy in Localized, Low-Intermediate-Risk Prostate Cancer: Current and Future Prospectives. Medicina 2023, 59, 1144. [Google Scholar] [CrossRef]
- Cosset, J.M.; Socie, G.; Dubray, B.; Girinsky, T.; Fourquet, A.; Gluckman, E. Single dose versus fractionated total body irradiation before bone marrow transplantation: Radiobiological and clinical considerations. Int. J. Radiat. Oncol. Biol. Phys. 1994, 30, 477–492. [Google Scholar] [CrossRef]
- Shaffer, B.C.; Kebriaei, P.; de Lima, M.; Jimenez Jimenez, A.M. Measurable residual disease testing and allogeneic hematopoietic cell transplantation for AML: Adapting Pre-MEASURE to clinical practice. Bone Marrow Transplant. 2025, 60, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Muffly, L.; Liang, E.C.; Dolan, J.G.; Pulsipher, M.A. How I use next-generation sequencing-MRD to plan approach and prevent relapse after HCT for children and adults with ALL. Blood 2024, 144, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Palmer, J.M.; Martin, P.; Tsai, N.; Kim, Y.; Chen, B.T.; Popplewell, L.; Siddiqi, T.; Thomas, S.H.; Mott, M.; et al. Results of a Multicenter Phase II Trial of Brentuximab Vedotin as Second-Line Therapy before Autologous Transplantation in Relapsed/Refractory Hodgkin Lymphoma. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2015, 21, 2136–2140. [Google Scholar] [CrossRef]
- Mawad, R.; Gooley, T.A.; Rajendran, J.G.; Fisher, D.R.; Gopal, A.K.; Shields, A.T.; Sandmaier, B.M.; Sorror, M.L.; Deeg, H.J.; Storb, R.; et al. Radiolabeled anti-CD45 antibody with reduced-intensity conditioning and allogeneic transplantation for younger patients with advanced acute myeloid leukemia or myelodysplastic syndrome. Biol. Blood Marrow Transplant. J. Am. Soc. Blood Marrow Transplant. 2014, 20, 1363–1368. [Google Scholar] [CrossRef]
- Orchard, K.; Langford, J.; Guy, M.; Lewis, G.; Michopoulou, S.; Cooper, M.; Zvavamwe, C.; Richardson, D.; Lewington, V. Efficient bone marrow irradiation and low uptake by non-haematological organs with an yttrium-90-anti-CD66 antibody prior to haematopoietic stem cell transplantation. Bone Marrow Transplant. 2024, 59, 1247–1257. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. Study Details|TBI Using IMRT (VMAT or Tomotherapy) for the Prevention of Pul Toxicities in Patients Undergoing Donor SCT. Available online: https://www.clinicaltrials.gov/study/NCT04281199 (accessed on 19 June 2025).
- ClinicalTrials.gov. Total Marrow and Lymphoid Irradiation as Conditioning Regimen Before Hematopoietic Cell Transplantation in Patients with Myelodysplastic Syndrome or Acute Leukemia. Available online: https://clinicaltrials.gov/study/NCT04262843?cond=%22Myelodysplastic%20Syndrome%22&intr=%22Vidarabine%22&viewType=Table&rank=5 (accessed on 19 June 2025).
- ClinicalTrials.gov. Combination Chemotherapy, Total Body Irradiation, and Donor Blood Stem Cell Transplant in Treating Patients with Secondary Myelofibrosis. Available online: https://clinicaltrials.gov/study/NCT03118492 (accessed on 19 June 2025).
- Home|ClinicalTrials.gov. Chemotherapy, Total Body Irradiation, and Post-Transplant Cyclophosphamide in Reducing Rates of Graft Versus Host Disease in Patients with Hematologic Malignancies Undergoing Donor Stem Cell Transplant. Available online: https://www.clinicaltrials.gov/study/NCT03192397 (accessed on 19 June 2025).
- ClinicalTrials.gov. The EndRAD Trial: Eliminating Total Body Irradiation (TBI) for NGS-MRD Negative Children, Adolescents, and Young Adults with B-ALL. Available online: https://clinicaltrials.gov/study/NCT03509961 (accessed on 19 June 2025).
- ClinicalTrials.gov. Study Details|Intensity Modulated Total Marrow Irradiation in Fully Human Leukocyte Antigen (HLA)-Matched and Partially-HLA Mismatched Allogeneic Transplantation Patients with High-Risk Acute Myeloid Leukemia (AML), Chronic Myeloid Leukemia (CML), and Myelodysplastic Syndrome (MDS). Available online: https://clinicaltrials.gov/study/NCT06802315?cond=acute%20leukemia&aggFilters=status:,phase:2 (accessed on 19 June 2025).
- ClinicalTrials.gov. BMT-06: Study of Intensity Modulated Total Marrow Irradiation (IM-TMI). Available online: https://clinicaltrials.gov/study/NCT04187105 (accessed on 19 June 2025).
- ClinicalTrials.gov. Study Details|Total Body Irradiation and Astatine-211-Labeled BC8-B10 Monoclonal Antibody for the Treatment of Nonmalignant Diseases. Available online: https://clinicaltrials.gov/study/NCT04083183?term=astatine&rank=3 (accessed on 19 June 2025).




| Schedule | Administration | Conditioning | Clinical Indications | Key Notes |
|---|---|---|---|---|
| 10 Gy/1 fraction [11,12] | QD over 1 day | Historical myeloablative | Early AML/ALL transplants (pre-1980) | Excess toxicity; largely obsolete |
| 12 Gy/6 fractions [13] | BID over 3 days | Standard myeloablative | Front-line paediatric and adult allo-HSCT | Benchmark OS & GVHD profile |
| 12–13.5 Gy/8–9 fractions [13] | BID over 4–4.5 days | Standard myeloablative | Centres favouring smaller fraction sizes | Slightly longer course |
| 12–13.2 Gy/10–11 fractions [13] | TID over 4 days | Hyper-fractionated myeloablative | Centres favouring smaller fraction sizes | Lowest per-fraction dose |
| 12 Gy/4 fractions [13] | QD over 4 days | Condensed myeloablative | Logistical constraints or BID-intolerant patients | Requires meticulous lung shielding |
| 4 Gy testicular boost/2 fractions [14] | QD over 2 days | Sanctuary boost | Boys with ALL | Practice varies; omission under study |
| 1 Gy/1 fraction [19,20] | QD over 1 day | Minimal-intensity/experimental | Older/comorbid adults; | Very low toxicity |
| 2 Gy/1 fraction [21] | QD over 1 day | Standard RIC | Older/comorbid adults | Very low toxicity |
| 4 Gy/2 fractions [24] | BID or QD over 1–2 days | Augmented RIC | Mismatched donors or high-risk disease | Balances better engraftment vs. added toxicity |
| Trial | Aim | Investigational Arm | Status |
|---|---|---|---|
| NCT04281199 (Phase I) [56] | IMRT-TBI dosimetric optimisation | Conformal IMRT-TBI 12 Gy/6 fx | Active, not recruiting |
| NCT04262843 (Phase II) [57] | Escalated TMI/TMLI to 20 Gy | TMLI 20 Gy (lung < 8 Gy) | Recruiting at the main site, but several satellites are now “Active-not-recruiting” |
| NCT03118492 (Phase I) [58] | 4 Gy mini-TBI + Flu-Mel in haplo HSCT | 4 Gy (2 × 2 Gy) + Flu-Mel | Active, not-recruiting |
| NCT03192397 (Phase Ib/II) [59] | 4 Gy mini-TBI + PTCy | 4 Gy (2 × 2 Gy) + PTCy | Active, not recruiting |
| NCT03509961 (EndRAD, Phase II) [60] | MRD-guided omission of TBI | Chemo vs. 12 Gy TBI | Recruiting |
| NCT06802315 (BMT-13, Phase II) [61] | High-risk AML/CML/MDS | IM-TMI 9 Gy + Flu-Bu | Active, not recruiting |
| NCT04187105 (BMT-06, Phase II) [62] | Haplo-HSCT dose-escalated TMI | IM-TMI 6 Gy + Flu-Cy + PTCy | Recruiting |
| NCT04083183 (Phase I/II) [63] | 211At-anti-CD45 α-conditioning | ≤2 Gy TBI + 211At-BC8-B10 | Recruiting |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lo Greco, M.C.; Milazzotto, R.; Acquaviva, G.; Liardo, R.L.E.; Marano, G.; La Rocca, M.; Basile, A.; Foti, P.V.; Palmucci, S.; David, E.; et al. Total Body Irradiation in Haematopoietic Stem Cell Transplantation: A Comprehensive Literature Review and Institutional Experience from the Policlinico of Catania. Medicina 2025, 61, 1503. https://doi.org/10.3390/medicina61091503
Lo Greco MC, Milazzotto R, Acquaviva G, Liardo RLE, Marano G, La Rocca M, Basile A, Foti PV, Palmucci S, David E, et al. Total Body Irradiation in Haematopoietic Stem Cell Transplantation: A Comprehensive Literature Review and Institutional Experience from the Policlinico of Catania. Medicina. 2025; 61(9):1503. https://doi.org/10.3390/medicina61091503
Chicago/Turabian StyleLo Greco, Maria Chiara, Roberto Milazzotto, Grazia Acquaviva, Rocco Luca Emanuele Liardo, Giorgia Marano, Madalina La Rocca, Antonio Basile, Pietro Valerio Foti, Stefano Palmucci, Emanuele David, and et al. 2025. "Total Body Irradiation in Haematopoietic Stem Cell Transplantation: A Comprehensive Literature Review and Institutional Experience from the Policlinico of Catania" Medicina 61, no. 9: 1503. https://doi.org/10.3390/medicina61091503
APA StyleLo Greco, M. C., Milazzotto, R., Acquaviva, G., Liardo, R. L. E., Marano, G., La Rocca, M., Basile, A., Foti, P. V., Palmucci, S., David, E., Iní, C., Aliotta, L., Salamone, V., La Monaca, V. A., Pergolizzi, S., & Spatola, C. (2025). Total Body Irradiation in Haematopoietic Stem Cell Transplantation: A Comprehensive Literature Review and Institutional Experience from the Policlinico of Catania. Medicina, 61(9), 1503. https://doi.org/10.3390/medicina61091503







