Expanding Robotic-Assisted Surgery in Gynecology Using the Potential of an Advanced Robotic System
Abstract
:1. Introduction—Trends in Minimally Invasive Surgery in Gynecology
2. Advantages of RAS for Surgeons
3. Equivalence of RAS and LAP in Patient Outcomes
4. Equivalence of RAS and LAP in Technology Adoption Challenges (Training and Learning Curve)
5. Limitations of RAS and Advantages of LAP
6. On-Demand Robotic Assistance
7. The Future of Gynecological Surgery
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kelley, W.E. The Evolution of Laparoscopy and the Revolution in Surgery in the Decade of the 1990s. J. Soc. Laparoendosc. Surg. 2008, 12, 351–357. [Google Scholar]
- Alkatout, I.; Mechler, U.; Mettler, L.; Pape, J.; Maass, N.; Biebl, M.; Gitas, G.; Laganà, A.S.; Freytag, D. The Development of Laparoscopy—A Historical Overview. Front. Surg. 2021, 8, 799442. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.M.; Neubauer, N.L. Minimally Invasive Surgery in Gynecologic Oncology. ISRN Obstet. Gynecol. 2013, 2013, 312982. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Tang, H.; Xie, Z.; Deng, S. Robotic-assisted vs. laparoscopic and abdominal myomectomy for treatment of uterine fibroids: A meta-analysis. Minim. Invasive Ther. Allied Technol. 2018, 27, 249–264. [Google Scholar] [CrossRef] [PubMed]
- Kampers, J.; Gerhardt, E.; Sibbertsen, P.; Flock, T.; Klapdor, R.; Hertel, H.; Jentschke, M.; Hillemanns, P. Protective operative techniques in radical hysterectomy in early cervical carcinoma and their influence on disease-free and overall survival: A systematic review and meta-analysis of risk groups. Arch. Gynecol. Obstet. 2021, 304, 577–587. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.A.; Bandari, J.; Pelzman, D.; Davies, B.J.; Jacobs, B.L. Diffusion and adoption of the surgical robot in urology. Transl. Androl. Urol. 2021, 10, 2151–2157. [Google Scholar] [CrossRef] [PubMed]
- Ahlering, T.E.; Skarecky, D.; Lee, D.; Clayman, R.V. Successful transfer of open surgical skills to a laparoscopic environment using a robotic interface: Initial experience with laparoscopic radical prostatectomy. J. Urol. 2003, 170, 1738–1741. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Kuchta, K.; Rojas, A.; Mehdi, S.A.; Ramirez Barriga, M.; Hays, S.; Talamonti, M.S.; Hogg, M.E. Residents perform better technically, have less stress and workload, and prefer robotic to laparoscopic technique during inanimate simulation. Surg. Endosc. 2023, 37, 7230–7237. [Google Scholar] [CrossRef]
- Capozzi, V.A.; Scarpelli, E.; Armano, G.; Monfardini, L.; Celardo, A.; Munno, G.M.; Fortunato, N.; Vagnetti, P.; Schettino, M.T.; Grassini, G. Update of Robotic Surgery in Benign Gynecological Pathology: Systematic Review. Medicina 2022, 58, 552. [Google Scholar] [CrossRef]
- Truong, M.D.; Tholemeier, L.N. Role of Robotic Surgery in Benign Gynecology. Obstet. Gynecol. Clin. N. Am. 2022, 49, 273–286. [Google Scholar] [CrossRef]
- Gitas, G.; Hanker, L.; Rody, A.; Ackermann, J.; Alkatout, I. Robotic surgery in gynecology: Is the future already here? Minim. Invasive Ther. Allied. Technol. 2022, 31, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Alkatout, I.; Mettler, L.; Maass, N.; Ackermann, J. Robotic surgery in gynecology. J. Turk. Ger. Gynecol. Assoc. 2016, 17, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Li, X.; Tian, S.; Zhu, T.; Yao, Y.; Tao, Y. Superiority of robotic surgery for cervical cancer in comparison with traditional approaches: A systematic review and meta-analysis. Inter. J. Surg. 2017, 40, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Swenson, C.W.; Kamdar, N.S.; Harris, J.A.; Uppal, S.; Campbell, D.A.; Morgan, D.M. Comparison of robotic and other minimally invasive routes of hysterectomy for benign indications. Am. J. Obstet. Gynecol. 2016, 215, e1–e650. [Google Scholar] [CrossRef] [PubMed]
- Aarts, J.W.M.; Nieboer, T.E.; Johnson, N.; Tavender, E.; Garry, R.; Mol, B.W.; Kluivers, K.B. Surgical approach to hysterectomy for benign gynaecological disease. Cochrane Database Syst. Rev. 2015, 8, CD003677. [Google Scholar] [CrossRef] [PubMed]
- Anger, J.T.; Mueller, E.R.; Tarnay, C.; Smith, B.; Stroupe, K.; Rosenman, A.; Brubaker, L.; Bresee, C.; Kenton, K. Robotic compared with laparoscopic sacrocolpopexy: A randomized controlled trial. Obstet. Gynecol. 2014, 123, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Restaino, S.; Mereu, L.; Finelli, A.; Spina, M.R.; Marini, G.; Catena, U.; Turco, L.C.; Moroni, R.; Milani, M.; Cela, V.; et al. Robotic surgery vs laparoscopic surgery in patients with diagnosis of endometriosis: A systematic review and meta-analysis. J. Robot. Surg. 2020, 14, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Soto, E.; Luu, T.H.; Liu, X.; Magrina, J.F.; Wasson, M.N.; Einarsson, J.I.; Cohen, S.L.; Falcone, T. Laparoscopy vs. Robotic Surgery for Endometriosis (LAROSE): A multicenter, randomized, controlled trial. Fertil. Steril. 2017, 107, 996–1002. [Google Scholar] [CrossRef]
- Mäenpää, M.M.; Nieminen, K.; Tomás, E.I.; Laurila, M.; Luukkaala, T.H.; Mäenpää, J.U. Robotic-assisted vs traditional laparoscopic surgery for endometrial cancer: A randomized controlled trial. Am. J. Obstet. Gynecol. 2016, 215, 588.e1–588.e7. [Google Scholar] [CrossRef]
- Baracy, M.G.; Kerl, A.; Hagglund, K.; Fennell, B.; Corey, L.; Aslam, M.F. Trends in surgical approach to hysterectomy and perioperative outcomes in Michigan hospitals from 2010 through 2020. J. Robot. Surg. 2023, 17, 2211–2220. [Google Scholar] [CrossRef]
- Buderath, P.; Kimmig, R.; Dominowski, L.; Mach, P. Hysterectomy in benign conditions: A 20-year single-center retrospective on the development of surgical techniques. Arch. Gynecol. Obstet. 2023, 307, 807–812. [Google Scholar] [CrossRef]
- Carbonnel, M.; Moawad, G.N.; Tarazi, M.M.; Revaux, A.; Kennel, T.; Favre-Inhofer, A.; Ayoubi, J.M. Robotic hysterectomy for benign indications: What have we learned from a decade? J. Soc. Laparoendosc. Surg. 2021, 25, e2020.00091. [Google Scholar] [CrossRef]
- Roth, K.; Kaier, K.; Stachon, P.; von Zur Mühlen, C.; Jungmann, P.; Grimm, J.; Klar, M.; Juhasz-Böss, I.; Taran, F.A. Evolving trends in the surgical therapy of patients with endometrial cancer in Germany: Analysis of a nationwide registry with special emphasis on perioperative outcomes. Arch. Gynecol. Obstet. 2023, 308, 1635–1640. [Google Scholar] [CrossRef] [PubMed]
- Han, E.S.; Advincula, A.P. Robotic Surgery: Advancements and Inflection Points in the Field of Gynecology. Obstet. Gynecol. Clin. N. Am. 2021, 48, 759–776. [Google Scholar] [CrossRef] [PubMed]
- AAGL. AAGL Position Statement: Route of Hysterectomy to Treat Benign Uterine Disease. J. Minim. Invasive Gynecol. 2011, 18, 1–3. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists’ Committee on Gynecologic Practice, The Society of Gynecologic Surgeons. Robot-Assisted Surgery for Noncancerous Gynecologic Conditions: ACOG COMMITTEE OPINION, Number 810. Obstet. Gynecol. 2020, 136, e22–e30. [Google Scholar] [CrossRef]
- Reynolds, R.K.; Advincula, A.P. Robot-assisted laparoscopic hysterectomy: Technique and initial experience. Am. J. Surg. 2006, 191, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Liu, S.; Lommen, J.; Pudwell, J.; Pelland, L.; Bougie, O. Prevalence of musculoskeletal pain among gynecologic surgeons performing laparoscopic procedures: A systematic review and meta-analysis. Int. J. Gynaecol. Obstet. 2023, 161, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Catanzarite, T.; Tan-Kim, J.; Whitcomb, E.L.; Menefee, S. Ergonomics in Surgery: A Review. Female Pelvic. Med. Reconstr. Surg. 2018, 24, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.; Young, R.; Shields, J.; Smith, K.; Chao, L. Growing pains: Strategies for improving ergonomics in minimally invasive gynecologic surgery. Curr. Opin. Obstet. Gynecol. 2023, 35, 361–367. [Google Scholar] [CrossRef]
- Tuschy, B.; Berlit, S.; Brade, J.; Sütterlin, M.; Hornemann, A. Solo surgery—Early results of robot-assisted three-dimensional laparoscopic hysterectomy. Minim. Invasive Ther. Allied. Technol. 2014, 23, 230–234. [Google Scholar] [CrossRef]
- Albright, B.B.; Witte, T.; Tofte, A.N.; Chou, J.; Black, J.D.; Desai, V.B.; Erekson, E.A. Robotic Versus Laparoscopic Hysterectomy for Benign Disease: A Systematic Review and Meta-Analysis of Randomized Trials. J. Minim. Invasive Gynecol. 2016, 23, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Gitas, G.; Alkatout, I.; Proppe, L.; Hanker, L.; Allahqoli, L.; Grimbizis, G.; Rody, A.; Werner, N.; Sommer, S.; Baum, S. Long-term satisfaction of patients after laparoscopic and robotic-assisted hysterectomy. Arch. Gynecol. Obstet. 2022, 305, 1481–1490. [Google Scholar] [CrossRef] [PubMed]
- Arms, R.G.; Sun, C.C.; Burzawa, J.K.; Fleming, N.D.; Nick, A.M.; Rallapalli, V.; Westin, S.N.; Meyer, L.A.; Ramirez, P.T.; Soliman, P.T. Improvement in quality of life after robotic surgery results in patient satisfaction. Gynecol. Oncol. 2015, 138, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Pickett, C.M.; Seeratan, D.D.; Mol, B.W.J.; Nieboer, T.E.; Johnson, N.; Bonestroo, T.; Aarts, J.W. Surgical approach to hysterectomy for benign gynaecological disease. Cochrane Database Syst. Rev. 2023, 8, CD003677. [Google Scholar] [CrossRef] [PubMed]
- Lenfant, L.; Canlorbe, G.; Belghiti, J.; Kreaden, U.S.; Hebert, A.E.; Nikpayam, M.; Uzan, C.; Azaïs, H. Robotic-assisted benign hysterectomy compared with laparoscopic, vaginal, and open surgery: A systematic review and meta-analysis. J. Robot. Surg. 2023, 17, 2647–2662. [Google Scholar] [CrossRef] [PubMed]
- Tsakos, E.; Xydias, E.M.; Ziogas, A.C.; Sorrentino, F.; Nappi, L.; Vlachos, N.; Daniilidis, A. Multi-Port Robotic-Assisted Laparoscopic Myomectomy: A Systematic Review and Meta-Analysis of Comparative Clinical and Fertility Outcomes. J. Clin. Med. 2023, 12, 4134. [Google Scholar] [CrossRef] [PubMed]
- Narducci, F.; Bogart, E.; Hebert, T.; Gauthier, T.; Collinet, P.; Classe, J.M.; Lecuru, F.; Delest, A.; Motton, S.; Conri, V.; et al. Severe perioperative morbidity after robot-assisted versus conventional laparoscopy in gynecologic oncology: Results of the randomized ROBOGYN-1004 trial. Gynecol. Oncol. 2020, 158, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Lönnerfors, C.; Reynisson, P.; Persson, J. A randomized trial comparing vaginal and laparoscopic hysterectomy vs robot-assisted hysterectomy. J. Minim. Invasive Gynecol. 2015, 22, 78–86. [Google Scholar] [CrossRef]
- Kenton, K.; Mueller, E.R.; Tarney, C.; Bresee, C.; Anger, J.T. One-Year Outcomes after Minimally Invasive Sacrocolpopexy. Female Pelvic. Med. Reconstr. Surg. 2016, 22, 382–384. [Google Scholar] [CrossRef]
- Elessawy, M.; Schneekloth, S.; Günther, V.; Maass, N.; Mettler, L.; Alkatout, I. Postoperative telephone-based questionnaire on quality of life after robotic-assisted laparoscopic hysterectomy versus conventional total laparoscopic hysterectomy. J. Clin. Med. 2020, 9, 2849. [Google Scholar] [CrossRef]
- Rusch, P.; Ind, T.; Kimmig, R.; Maggioni, A.; Ponce, J.; Zanagnolo, V.; Coronado, P.J.; Verguts, J.; Lambaudie, E.; Falconer, H.; et al. Recommendations for a standardised educational program in robot assisted gynaecological surgery: Consensus from the Society of European Robotic Gynaecological Surgery (SERGS). Facts Views Vis. Obgyn. 2019, 11, 29–41. [Google Scholar]
- Alkatout, I.; Maass, N.; Günther, V. Der Stellenwert des Feedbacks beim Proctoring in der robotergestützten Chirurgie—Ein Grundstein des Erfolgskonzeptes? Zentralbl. Chir. 2023, 148, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Ballantyne, G.H. The Pitfalls of Laparoscopic Surgery: Challenges for Robotics and Telerobotic Surgery. Surg. Laparosc. Endosc. Percutaneous Tech. 2002, 12, 1–5. [Google Scholar] [CrossRef]
- Ackermann, J.; Baumann, J.; Pape, J.; Pahls, J.; Ruchay, Z.; Spüntrup, C.; Holthaus, B.; Noé, G.; Anapolski, M.; Meinhold-Heerlein, I.; et al. Factors influencing surgical performance and learning progress in minimally invasive surgery—Results of an interdisciplinary muticenter study. Int. J. Surg. 2023, 109, 2975–2986. [Google Scholar] [CrossRef] [PubMed]
- Spille, J.; Wenners, A.; von Hehn, U.; Maass, N.; Pecks, U.; Mettler, L.; Alkatout, I. 2D Versus 3D in Laparoscopic Surgery by Beginners and Experts: A Randomized Controlled Trial on a Pelvitrainer in Objectively Graded Surgical Steps. J. Surg. Educ. 2017, 74, 867–877. [Google Scholar] [CrossRef] [PubMed]
- Degirmenci, Y.; Schepers, M.; Steetskamp, J.; Hasenburg, A.; Skala, C. Three-dimensional vs two-dimensional endoscopic approach in urogynecology: A retrospective cohort study of laparoscopic sacrocolpopexy. J. Obstet. Gynaecol. Res. 2023, 49, 1028–1035. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Kang, D.Y. A randomized, controlled trial comparing the clinical outcomes of 3D versus 2D laparoscopic hysterectomy. Videosurgery Other Miniinvasive Tech. 2022, 17, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Blavier, A.; Gaudissart, Q.; Cadière, G.B.; Nyssen, A. S Comparison of learning curves and skill transfer between classical and robotic laparoscopy according to the viewing conditions: Implications for training. Am. J. Surg. 2007, 194, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.C.H.; Wu, H.S.; Lin, M.S.; Chou, D.A.; Huang, M. Comparison of robot-assisted laparoscopic adrenalectomy with traditional laparoscopic adrenalectomy—1 Year follow-up. Surg. Endosc. 2008, 22, 463–466. [Google Scholar] [CrossRef]
- Ind, T. Providing a standardised educational programme in robot-assisted gynaecological surgery. Best Pract. Res. Clin. Obstet. Gynaecol. 2023, 91, 102399. [Google Scholar] [CrossRef]
- Angell, J.; Gomez, M.S.; Baig, M.M.; Abaza, R. Contribution of Laparoscopic Training to Robotic Proficiency. J. Endourol. 2013, 27, 1027–1031. [Google Scholar] [CrossRef]
- Kanji, F.; Cohen, T.; Alfred, M.; Caron, A.; Lawton, S.; Savage, S.; Shouhed, D.; Anger, J.T.; Catchpole, K. Room size influences flow in robotic-assisted surgery. Int. J. Environ. Res. Public Health 2021, 18, 7984. [Google Scholar] [CrossRef]
- Salkowski, M.; Checcucci, E.; Chow, A.K.; Rogers, C.C.; Adbollah, F.; Liatsikos, E.; Dasgupta, P.; Guimaraes, G.C.; Rassweiler, J.; Mottrie, A.; et al. New multiport robotic surgical systems: A comprehensive literature review of clinical outcomes in urology. Ther. Adv. Urol. 2023, 15, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Alkatout, I.; Salehiniya, H.; Allahqoli, L. Assessment of the Versius Robotic Surgical System in Minimal Access Surgery: A Systematic Review. J. Clin. Med. 2022, 11, 3754. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.R.; Lee, G.I. Does a robotic surgery approach offer optimal ergonomics to gynecologic surgeons? A comprehensive ergonomics survey study in gynecologic robotic surgery. J. Gynecol. Oncol. 2017, 28, e70. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, N.; Watanabe, G.; Inaki, N.; Moriyama, H.; Shimada, M.; Kawaguchi, M. The da Vinci Surgical System versus the Radius Surgical System. Surg. Sci. 2012, 3, 358–361. [Google Scholar] [CrossRef]
- Keil, M.; Viere, T.; Helms, K.; Rogowski, W. The impact of switching from single-use to reusable healthcare products: A transparency checklist and systematic review of life-cycle assessments. Eur. J. Public Health 2023, 33, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Polan, R.M.; Barber, E.L. Reimbursement for Female-Specific Compared With Male-Specific Procedures Over Time. Obstet. Gynecol. 2021, 138, 878–883. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.D.; Tergas, A.I.; Hou, J.Y.; Burke, W.M.; Chen, L.; Hu, J.C.; Neugut, A.I.; Ananth, C.V.; Hershman, D.L. Effect of regional hospital competition and hospital financial status on the use of robotic-assisted surgery. JAMA Surg. 2016, 151, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Gu, D.; Klein, R.; Zhou, S.; Shih, Y.T.; Tracy, T.; Soybel, D.; Dillon, P. Factors Associated With Hospital Decisions to Purchase Robotic Surgical Systems. MDM Policy Pract. 2020, 5, 2381468320904364. [Google Scholar] [CrossRef]
- El Hachem, L.; Andikyan, V.; Mathews, S.; Friedman, K.; Poeran, J.; Shieh, K.; Geoghegan, M.; Gretz, H.F. Robotic Single-Site and Conventional Laparoscopic Surgery in Gynecology: Clinical Outcomes and Cost Analysis of a Matched Case-Control Study. J. Minim. Invasive Gynecol. 2016, 23, 760–768. [Google Scholar] [CrossRef] [PubMed]
- De Nagy, J.; Youssef, Y.; Moawad, G. Strategies and factors to maximize cost-effectiveness of robotic surgery in benign gynecological disease. Best Pract. Res. Clin. Obstet. Gynaecol. 2023, 90, 102380. [Google Scholar] [CrossRef] [PubMed]
- Erskine, J.; Abrishami, P.; Charter, R.; Cicchetti, A.; Culbertson, R.; Faria, E.; Hiatt, J.C.; Khan, J.; Maddern, G.; Patel, A.; et al. Best practice considerations on the assessment of robotic assisted surgical systems: Results from an international consensus expert panel. Int. J. Technol. Assess. Health Care. 2023, 39, e39. [Google Scholar] [CrossRef] [PubMed]
- Böhlen, D.; Gerber, R. First Ever Radical Prostatectomy Performed with the New Dexter Robotic SystemTM. Eur. Urol. 2023, 83, 479–480. [Google Scholar] [CrossRef] [PubMed]
- Hahnloser, D.; Rrupa, D.; Grass, F. Feasibility of on-demand robotics in colorectal surgery: First cases. Surg. Endosc. 2023, 37, 8594–8600. [Google Scholar] [CrossRef] [PubMed]
- Thillou, D.; Robin, H.; Ricolleau, C.; Benali, N.A.; Forgues, A.; Emeriau, D.; Mignot, H.; Hugues, G. Robot-assisted Radical Prostatectomy with the Dexter Robotic System: Initial Experience and Insights into On-demand Robotics. Eur. Urol. 2023; in press. [Google Scholar] [CrossRef] [PubMed]
- Alkatout, I.; Mettler, L.; Maass, N.; Noé, G.K.; Elessawy, M. Abdominal anatomy in the context of port placement and trocars. J. Turk. Ger. Gynecol. Assoc. 2015, 16, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Troccaz, J.; Dagnino, G.; Yang, G.Z. Frontiers of Medical Robotics: From Concept to Systems to Clinical Translation. Annu. Rev. Biomed. Eng. 2019, 21, 193–218. [Google Scholar] [CrossRef]
- Rao, P.P. Robotic surgery: New robots and finally some real competition! World J. Urol. 2018, 36, 537–541. [Google Scholar] [CrossRef]
- Alkatout, I. Laparoscopic hysterectomy: Total or subtotal?—Functional and didactic aspects. Minim. Invasive Ther. Allied. Technol. 2022, 31, 13–23. [Google Scholar] [CrossRef]

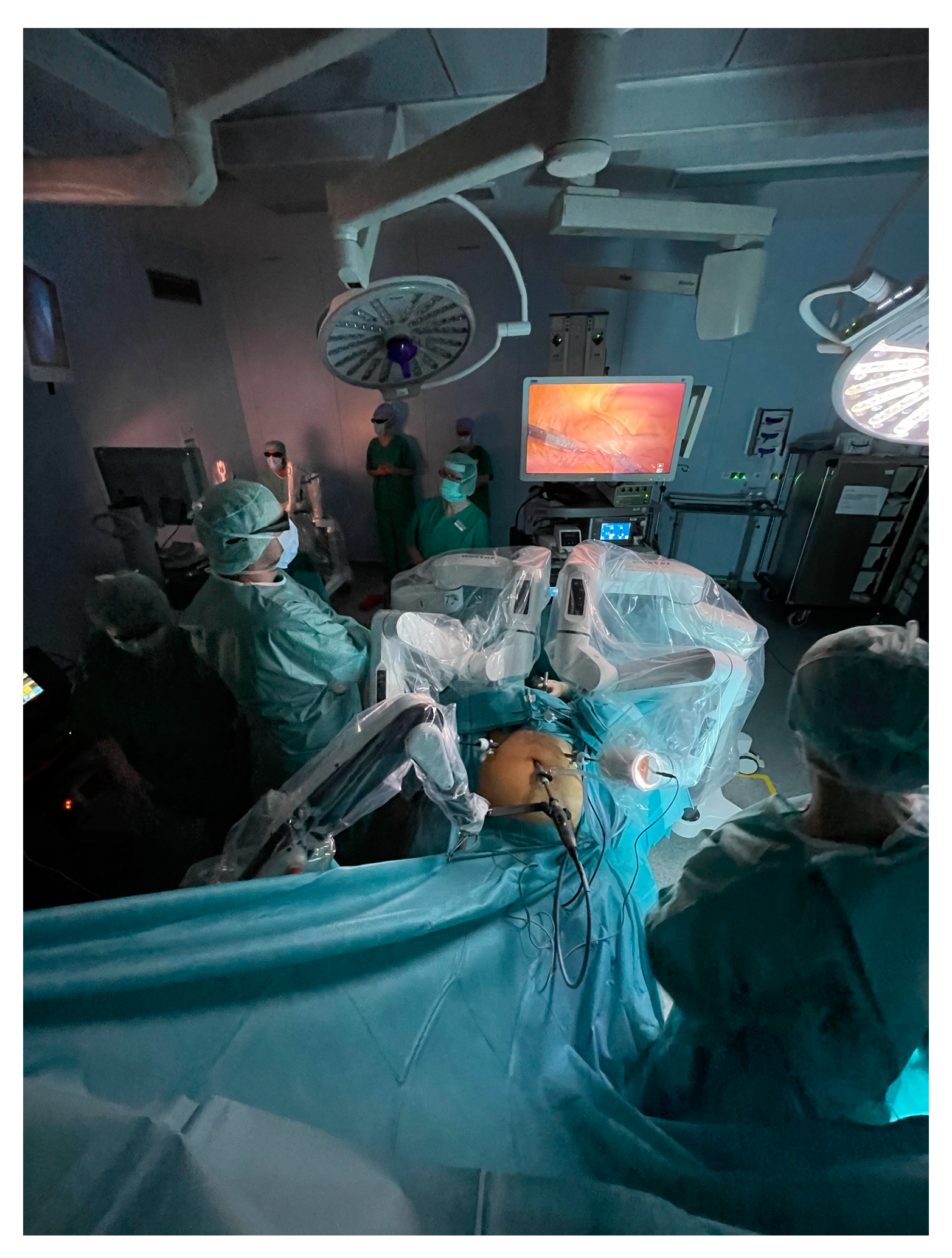
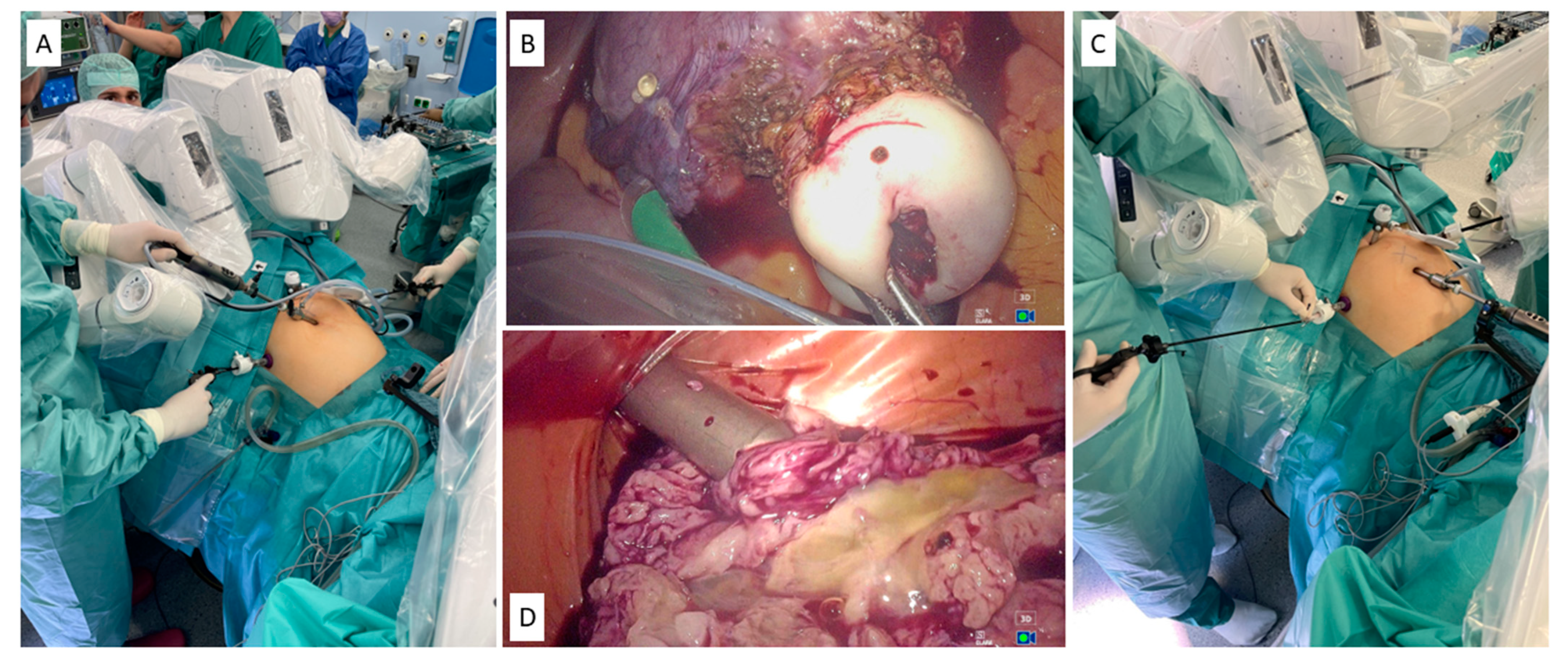
| Authors | Disease | Number of Patients (Number of Studies) | Study Design | Study Results |
|---|---|---|---|---|
| Lenfant et al. [36] | Benign hysterectomy | 24 studies | Meta-analysis RCT, prospective and retrospective databases | Overall shorter hospital stay and less blood loss in RAS vs. LAP, no difference in OT |
| Pickett et al. [35] | Benign Hysterectomy | 296 (three studies) | Meta-analysis RCT | Return to normal activities in RAS was lower (low grade evidence and inconclusive results) |
| Albright et al. [32] | Benign Hysterectomy | 326 (four studies) | Meta-analysis RCT | No difference in perioperative complication rates, LOS, OT, conversion, or blood loss |
| Liu et al. [13] * | Cervical cancer, hysterectomy | 19 studies | Meta-analysis, retrospective comparison of LAP, RAS, and abdominal approaches | Longer or equivalent OT, more blood loss, and shorter hospital stay with RAS |
| Wang et al. [4] | Uterine fibroids, myomectomy | 2852 (20 studies) | Meta-analysis, retrospective comparison of LAP, RAS, and abdominal approaches | Fewer intra-operative complications and laparotomy conversions, lower estimated blood loss, and less post-operative bleeding with RAS |
| Aarts et al. [15] ** | Variable benign gynecological diseases, hysterectomy | 5102 (47 studies) | Meta-analysis, retrospective comparison of LAP, RAS abdominal, and vaginal approaches | Longer OT and faster return to normal activities with RAS |
| Tsakos et al. [37] | Uterine fibroids, myomectomy | 53 studies | Meta-analysis, retrospective comparison of RAS, LAP, and abdominal approaches | Longer OT, lower blood loss with RAS; equivalent length of stay, transfusion, and complication rates |
| Narducci et al. [38] | Gynecologic cancer | 369 | RCT, prospective comparison of LAP and RAS | Longer OT and higher blood loss in RAS, similar conversion rates to open and perioperative morbidity |
| Lönnerfors et al. [39] | Variable benign gynecological diseases, hysterectomy | 122 | RCT, prospective comparison of LAP, RAS, and vaginal approaches | Shorter OT, lower blood loss, fewer intra- and post-operative complications |
| Mäenpää et al. [19] | Endometrial cancer, hysterectomy | 99 | RCT, prospective comparison of LAP and RAS | Shorter OT with RAS; laparotomy conversion rate higher with LAP |
| Soto et al. [18] | Endometriosis, endometriosis surgery | 73 | RCT, prospective comparison of LAP and RAS | Longer OT with RAS; equivalent blood loss, complication rates, and laparotomy conversion rates |
| Anger et al. [16] | Pelvic organ prolapse, sacrocolpopexy | 78 | RCT, prospective comparison of LAP and RAS | More postoperative pain with RAS; equivalent complication rates and short-term outcomes |
| Restaino et al. [17] | Endometriosis, endometriosis surgery | 1527 | RCT, retrospective comparison of LAP and RAS | Longer OT with RAS; equivalent blood loss, complication rates, and length of hospital stay |
| Swenson et al. [14] | Variable benign gynecological diseases, hysterectomy | 1338 | RCT, retrospective comparison of RAS, LAP, and vaginal approaches | Longer OT, lower blood loss, and shorter hospital stays with RAS; equivalent intra- and major postoperative complication rates |
| Kenton et al. [40] | Pelvic organ prolapse, sacrocolpopexy | 78 | RCT, retrospective comparison of LAP and RAS | Equivalent one-year follow-up outcomes, return to normal activities, and recurrence rates |
| Property | Dexter Characteristics |
|---|---|
| Manufacturer | Distalmotion SA, Switzerland |
| Robotic system name | Dexter |
| Approach | Laparoscopic |
| Clinical approval | European CE Mark (2022) |
| Patient cart | Two carts with instrument arms, one optional endoscope cart |
| Arm configuration | Modular/LAP mode functionality |
| Surgeon console | Open/sterile/ergonomic |
| Endoscope | Compatible with any 3D endoscope/Indocyanine green florescence imaging system |
| Endoscope arm | Mountable on the patient bed or endoscope cart |
| Imaging platform | Compatible with any 3D imaging system installed in front of the surgeon console |
| Trocars | Compatible with any 10–12 mm trocars |
| Instruments | Five single-use instruments: needle holder, monopolar hook, monopolar scissors, bipolar Maryland dissector, and bipolar Johann grasper |
| Instrument diameter/Degree of Freedom | 8.3 mm/7° |
| Foot pedal control | Yes, clutch and endoscope |
| Simulator available | Yes |
| Fields of application | Gynecology, general surgery, and urology |
| Additional features |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alkatout, I.; O’Sullivan, O.; Peters, G.; Maass, N. Expanding Robotic-Assisted Surgery in Gynecology Using the Potential of an Advanced Robotic System. Medicina 2024, 60, 53. https://doi.org/10.3390/medicina60010053
Alkatout I, O’Sullivan O, Peters G, Maass N. Expanding Robotic-Assisted Surgery in Gynecology Using the Potential of an Advanced Robotic System. Medicina. 2024; 60(1):53. https://doi.org/10.3390/medicina60010053
Chicago/Turabian StyleAlkatout, Ibrahim, Odile O’Sullivan, Göntje Peters, and Nicolai Maass. 2024. "Expanding Robotic-Assisted Surgery in Gynecology Using the Potential of an Advanced Robotic System" Medicina 60, no. 1: 53. https://doi.org/10.3390/medicina60010053
APA StyleAlkatout, I., O’Sullivan, O., Peters, G., & Maass, N. (2024). Expanding Robotic-Assisted Surgery in Gynecology Using the Potential of an Advanced Robotic System. Medicina, 60(1), 53. https://doi.org/10.3390/medicina60010053





