CGRP Antagonism and Ketogenic Diet in the Treatment of Migraine
Abstract
1. Introduction
2. Classification of Headaches
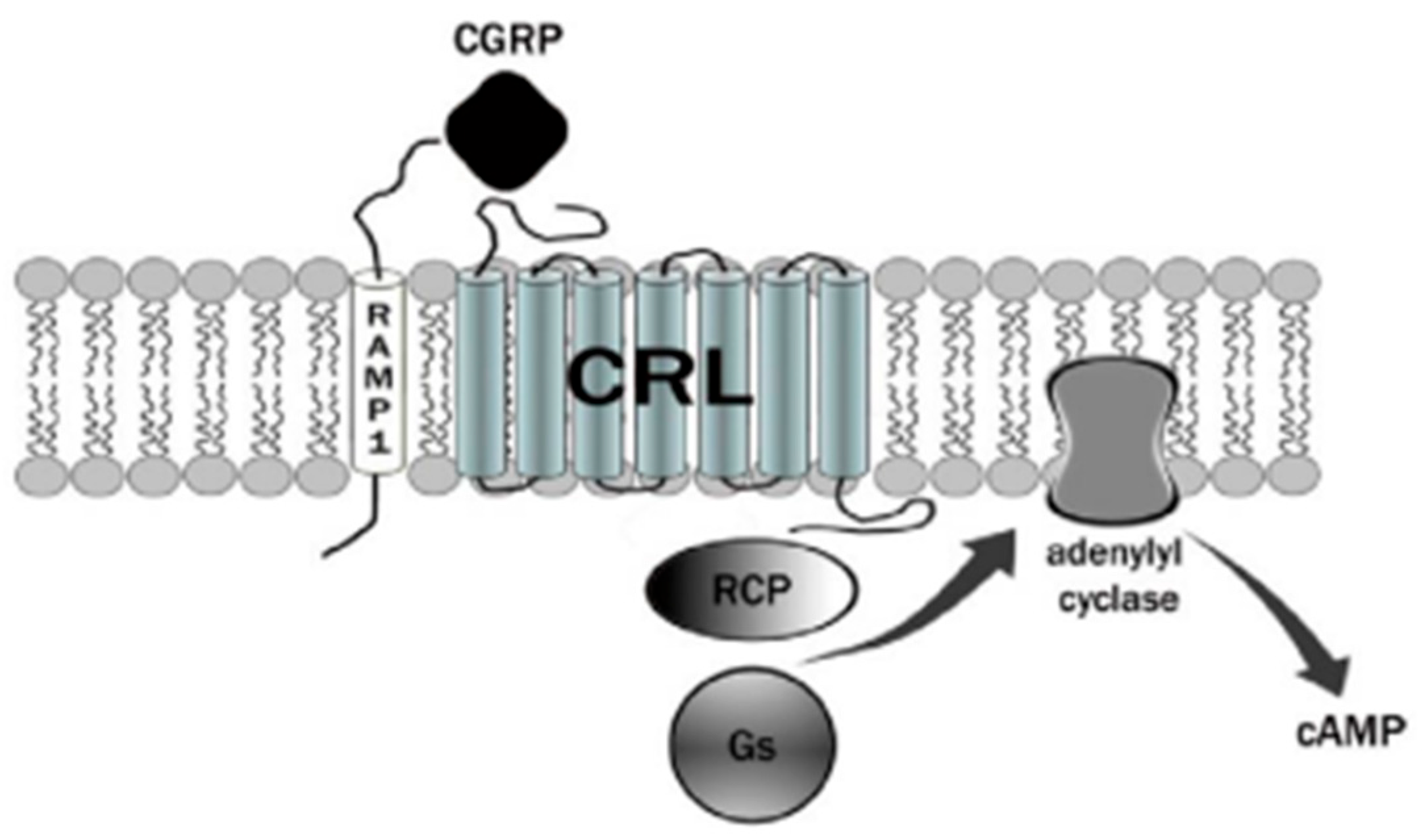
3. Calcitonin Gene-Related Peptide (CGRP) Receptor
4. Therapies Based on the Different Forms of Migraine
4.1. CGRP Antagonists
4.2. Monoclonal Antibodies
4.3. Innovative Medicines
5. Recent Preclinical and Clinical Studies on Gepans and Monoclonal Antibodies
6. Ketogenic Diet
7. Influence of Migraine on Lifestyle
8. Migraine and Food
9. Conclusions
10. Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Steiner, T.J.; Stovner, L.J. Global epidemiology of migraine and its implications for public health and health policy. Nat. Rev. Neurol. 2023, 19, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, R.; Tiwari, G.; Mishra, S.; Ramachandran, V. Preventive and therapeutic aspects of migraine for patient care: An insight. Curr. Mol. Pharmacol. 2023, 16, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Mangrum, R.; Gerstein, M.T.; Hall, C.J., III; Buse, D.C.; Houts, C.R.; McGinley, J.S.; McCarrier, K.P.; Lipton, R.B.; Wirth, R.J. Priority acute and preventive migraine treatment benefits: Results of the Migraine Clinical Outcome Assessment System (MiCOAS) qualitative study of people living with migraine. Headache, 2023; in press. [Google Scholar] [CrossRef]
- Silvestro, M.; Iannone, L.F.; Orologio, I.; Tessitore, A.; Tedeschi, G.; Geppetti, P.; Russo, A. Migraine treatment: Towards new pharmacological targets. Int. J. Mol. Sci. 2023, 24, 12268. [Google Scholar] [CrossRef] [PubMed]
- Ursitti, F.; Valeriani, M. Migraine in childhood: Gender differences. Eur. J. Paediatr. Neurol. 2023, 42, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Marti-Marca, A.; Vilà-Balló, A.; Cerda-Company, X.; Ikumi, N.; Torres-Ferrus, M.; Caronna, E.; Gallardo, V.J.; Alpuente, A.; Torralba Cuello, M.; Soto-Faraco, S.; et al. Exploring sensory sensitivity, cortical excitability, and habituation in episodic migraine, as a function of age and disease severity, using the pattern-reversal task. J. Headache Pain 2023, 24, 104. [Google Scholar] [CrossRef] [PubMed]
- Freddi, T.A.L.; Ottaiano, A.C.; Lucio, L.L.; Corrêa, D.G.; Hygino da Cruz, L.C., Jr. The trigeminal nerve: Anatomy and pathology. Semin. Ultrasound CT MR 2022, 43, 403–413. [Google Scholar] [CrossRef]
- Bigal, M.E.; Lipton, R.B. Modifiable risk factors for migraine progression (or for chronic daily headaches)—Clinical lessons. Headache 2006, 46 (Suppl. S3), S144–S146. [Google Scholar] [CrossRef]
- Pezzella, P. The ICD-11 is now officially in effect. World Psych. 2022, 21, 331. [Google Scholar] [CrossRef]
- Puledda, F.; Wang, S.J.; Diener, H.C.; Schytz, H.W. A history of International Headache Society grants and their impact on headache careers. Cephalalgia 2022, 42, 1288–1293. [Google Scholar] [CrossRef]
- Silva-Néto, R.; Holle-Lee, D. Headaches Classification. In Hypnic Headache; Springer: Cham, Switzerland, 2023. [Google Scholar] [CrossRef]
- Olesen, J. Classification of migraine and tension-type headache. Cephalalgia 2023, 43, 03331024221139238. [Google Scholar] [CrossRef]
- de Dhaem, B.O.; Rizzoli, P. Refractory Headaches. In Seminars in Neurology; Thieme Medical Publishers, Inc.: New York, NY, USA, 2022; Volume 42, pp. 512–522. [Google Scholar] [CrossRef]
- Iacopetta, D.; Ceramella, J.; Catalano, A.; Saturnino, C.; Pellegrino, M.; Mariconda, A.; Longo, P.; Sinicropi, M.S.; Aquaro, S. COVID-19 at a glance: An up-to-date overview on variants, drug design and therapies. Viruses 2022, 14, 573. [Google Scholar] [CrossRef] [PubMed]
- Tana, C.; Bentivegna, E.; Cho, S.J.; Harriott, A.M.; García-Azorín, D.; Labastida-Ramirez, A.; Ornello, R.; Raffaelli, B.; Beltrán, E.R.; Ruscheweyh, R.; et al. Long COVID headache. J. Headache Pain 2022, 23, 93. [Google Scholar] [CrossRef] [PubMed]
- Catalano, A.; Iacopetta, D.; Ceramella, J.; Maio, A.C.; Basile, G.; Giuzio, F.; Bonomo, M.G.; Aquaro, S.; Walsh, T.J.; Sinicropi, M.S.; et al. Are Nutraceuticals Effective in COVID-19 and Post-COVID Prevention and Treatment? Foods 2022, 11, 2884. [Google Scholar] [CrossRef] [PubMed]
- Grangeon, L.; Lange, K.S.; Waliszewska-Prosół, M.; Onan, D.; Marschollek, K.; Wiels, W.; Mikulenka, P.; Farham, F.; Gollion, C.; Ducros, A. Genetics of migraine: Where are we now? J. Headache Pain 2023, 24, 12. [Google Scholar] [CrossRef] [PubMed]
- Greenbaum, T.; Emodi-Perlman, A. Headache and orofacial pain: A traffic-light prognosis-based management approach for the musculoskeletal practice. Front. Neurol. 2023, 14, 1146427. [Google Scholar] [CrossRef]
- Fan, X.; Fu, G.; Wang, L.; Shen, W.; Zhang, Y. A bibliometric analysis and visualization of tension-type headache. Front. Neurol. 2022, 13, 980096. [Google Scholar] [CrossRef]
- May, A.; Evers, S.; Goadsby, P.J.; Leone, M.; Manzoni, G.C.; Pascual, J.; Carvalho, V.; Romoli, M.; Aleksovska, K.; Pozo-Rosich, P. European Academy of Neurology guidelines on the treatment of cluster headache. Eur. J. Neurol. 2023; in press. [Google Scholar] [CrossRef]
- Martelletti, P. Non-migraine primary headaches in medicine: A machine-generated overview of current research. In A Machine-Generated Overview of Current Research; Springer: Berlin/Heidelberg, Germany, 2023; ISBN 978-3031208935. [Google Scholar]
- Russo, A.F.; Hay, D.L. CGRP physiology, pharmacology, and therapeutic targets: Migraine and beyond. Physiol. Rev. 2023, 103, 1565–1644. [Google Scholar] [CrossRef]
- Kamm, K. CGRP and migraine: What have we learned from measuring CGRP in migraine patients so far? Front. Neurol. 2022, 13, 930383. [Google Scholar] [CrossRef]
- Edvinsson, L.; Haanes, K.A.; Warfvinge, K.; Krause, D.N. CGRP as the target of new migraine therapies—Successful translation from bench to clinic. Nat. Rev. Neurol. 2018, 14, 338–350. [Google Scholar] [CrossRef]
- Pellesi, L.; Guerzoni, S.; Pini, L.A. Spotlight on anti-CGRP monoclonal antibodies in migraine: The clinical evidence to date. Clin. Pharmacol. Drug Dev. 2017, 6, 534–547. [Google Scholar] [CrossRef]
- Hoare, S.R. Mechanisms of peptide and nonpeptide ligand binding to class B G-protein-coupled receptors. Drug Discov. Today 2005, 10, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, D.; Tonin, P.; Nicotera, P.; Bagetta, G.; Corasaniti, M.T. Real world considerations for newly approved CGRP receptor antagonists in migraine care. Exp. Rev. Neurother. 2022, 22, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Yao, G.; Man, Y.-H.; Li, A.-R.; Guo, Y.; Dai, Y.; Wang, P.; Zhou, Y.-F. NO up-regulates migraine-related CGRP via activation of an Akt/GSK-3β/NF-κB signaling cascade in trigeminal ganglion neurons. Aging 2020, 12, 6370–6384. [Google Scholar] [CrossRef] [PubMed]
- Karsan, N.; Gosalia, H.; Goadsby, P.J. Molecular mechanisms of migraine: Nitric oxide synthase and neuropeptides. Int. J. Mol. Sci. 2023, 24, 11993. [Google Scholar] [CrossRef]
- Gaete, P.S.; Lillo, M.A.; Puebla, M.; Poblete, I.; Figueroa, X.F. CGRP signalling inhibits NO production through pannexin-1 channel activation in endothelial cells. Sci. Rep. 2019, 9, 7932. [Google Scholar] [CrossRef]
- Blumenfeld, A.M.; Kaur, G.; Mahajan, A.; Shukla, H.; Sommer, K.; Tung, A.; Knievel, K.L. Effectiveness and safety of chronic migraine preventive treatments: A systematic literature review. Pain Ther. 2022, 12, 251–274. [Google Scholar] [CrossRef] [PubMed]
- Lampl, C.; Versijpt, J.; Amin, F.M.; Deligianni, C.I.; Gil-Gouveia, R.; Jassal, T.; MaassenVanDenBrink, A.; Ornello, R.; Paungarttner, J.; Sanchez-Del-Rio, M.; et al. European Headache Federation (EHF) Critical Re-Appraisal and Meta-Analysis of Oral Drugs in Migraine Prevention-Part 1: Amitriptyline. J. Headache Pain 2023, 24, 39. [Google Scholar] [CrossRef]
- McLean, G.; Mercer, S.W. Chronic migraine, comorbidity, and socioeconomic deprivation: Cross-sectional analysis of a large nationally representative primary care database. J. Comorb. 2017, 7, 89–95. [Google Scholar] [CrossRef]
- Krymchantowski, A.V.; Silva-Néto, R.P.; Jevoux, C.; Krymchantowski, A.G. Indomethacin for refractory COVID or post-COVID headache: A retrospective study. Acta Neurol. Belg. 2022, 122, 465–469. [Google Scholar] [CrossRef]
- Ceramella, J.; Iacopetta, D.; Sinicropi, M.S.; Andreu, I.; Mariconda, A.; Saturnino, C.; Giuzio, F.; Longo, P.; Aquaro, S.; Catalano, A. Drugs for COVID-19: An update. Molecules 2022, 27, 8562. [Google Scholar] [CrossRef]
- Rissardo, J.P.; Caprara, A.L.F. Gepants for acute and preventive migraine treatment: A narrative review. Brain Sci. 2022, 12, 1612. [Google Scholar] [CrossRef] [PubMed]
- Sacco, S.; Amin, F.M.; Ashina, M.; Bendsten, L.; Deligianni, C.I.; Gil-Gouveia, R.; Katsarava, Z.; MassenVanDenBrink, A.; Martelletti, P.; Mitsikostas, D.D.; et al. European Headache Federation guideline on the use of monoclonal antibodies targeting the calcitonin gene related peptide pathway for migraine prevention—2022 update. J. Headache Pain 2022, 23, 67. [Google Scholar] [CrossRef] [PubMed]
- Castrillo, A.; Mendoza, A.; Caballero, L.; Cerdán, D.; Rodríguez, M.F.; Guerrero, P.; Tabernero, C.; Ferrero, M.; Benito, I.; Marín, I.; et al. Effectiveness of anti-CGRP monoclonal antibodies in the preventive treatment of migraine: A prospective study of 63 patients. Med. Clínica 2023, 160, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Al-Karagholi, M.A.M.; Kalatharan, V.; Fagerberg, P.S.; Amin, F.M. The vascular role of CGRP: A systematic review of human studies. Front. Neurol. 2023, 14, 1204734. [Google Scholar] [CrossRef]
- Masood, W.; Annamaraju, P.; Uppaluri, K.R. Ketogenic Diet. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Han, X.; Yu, S. Non-Pharmacological Treatment for Chronic Migraine. Curr. Pain Headache Rep. 2023, 27, 663–672. [Google Scholar] [CrossRef]
- Hokenek, N.M.; Erdogan, M.O.; Hokenek, U.D.; Algin, A.; Tekyol, D.; Seyhan, A.U. Treatment of migraine attacks by transcutaneous electrical nerve stimulation in emergency department: A randomize controlled trial. Am. J. Emerg. Med. 2020, 39, 80–85. [Google Scholar] [CrossRef]
- De Luca, A.; Talon, S.; de Bellis, M.; Desaphy, J.-F.; Franchini, C.; Lentini, G.; Catalano, A.; Corbo, F.; Tortorella, V.; Conte-Camerino, D. Inhibition of skeletal muscle sodium currents by mexiletine analogues: Specific hydrophobic interactions rather than lipophilia per se account for drug therapeutic profile. Naunyn Schmiedeberg’s Arch. Pharmacol. 2003, 367, 318–327. [Google Scholar] [CrossRef]
- Edvinsson, L. CGRP receptor antagonists and antibodies against CGRP and its receptor in migraine treatment. Br. J. Clin. Pharmacol. 2015, 80, 193–199. [Google Scholar] [CrossRef]
- Hong, P.; Wu, X.; Liu, Y.; Information, P.E.K.F.C. Calcitonin gene-related peptide monoclonal antibody for preventive treatment of episodic migraine: A meta analysis. Clin. Neurol. Neurosurg. 2017, 154, 74–78. [Google Scholar] [CrossRef]
- Khan, S.; Olesen, A.; Ashina, M. CGRP, a target for preventive therapy in migraine and cluster headache: Systematic review of clinical data. Cephalalgia 2019, 39, 374–389. [Google Scholar] [CrossRef]
- Durham, P.L. Calcitonin Gene-Related Peptide (CGRP) and migraine. Headache 2006, 46 (Suppl. S1), S3–S8. [Google Scholar] [CrossRef] [PubMed]
- Deen, M.; Correnti, E.; Kamm, K.; Kelderman, T.; Papetti, L.; Rubio-Beltran, E.; Vigneri, S.; Edvinsson, L.; Maassen Van Den Brink, A. Blocking CGRP in migraine patients—A review of pros and cons. J. Headache Pain 2017, 18, 96. [Google Scholar] [CrossRef] [PubMed]
- Rees, T.A.; Hendrikse, E.R.; Hay, D.L.; Walker, C.S. Beyond CGRP: The calcitonin peptide family as targets for migraine and pain. Br. J. Pharmacol. 2021, 179, 381–399. [Google Scholar] [CrossRef] [PubMed]
- Al-Hassany, L.; MaassenVanDenBrink, A. Drug interactions and risks associated with the use of triptans, ditans and monoclonal antibodies in migraine. Curr. Opin. Neurol. 2021, 34, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Capi, M.; De Angelis, V.; De Bernardini, D.; De Luca, O.; Cipolla, F.; Lionetto, L.; Simmaco, M.; Martelletti, P. CGRP receptor antagonists and 5-HT1F receptor agonist in the treatment of migraine. J. Clin. Med. 2021, 10, 1429. [Google Scholar] [CrossRef] [PubMed]
- Karsan, N.; Goadsby, P.J. New oral drugs for migraine. CNS Drugs 2022, 36, 933–949. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Mancilla, E.; MaassenVanDenBrink, A. Novel pharmacological targets of migraine: An overview. In Novel Synthetic Drugs in Migraine; Headache Series; Martelletti, P., Edvinsson, L., Eds.; Springer: Cham, Switzerland, 2022. [Google Scholar] [CrossRef]
- Moreno-Ajona, D.; Villar-Martínez, M.D.; Goadsby, P.J. New generation gepants: Migraine acute and preventive medications. J. Clin. Med. 2022, 11, 1656. [Google Scholar] [CrossRef]
- Pozo-Rosich, P.; Ailani, J.; Ashina, M.; Goadsby, P.J.; Lipton, R.B.; Reuter, U.; Guo, H.; Schwefel, B.; Lu, K.; Boinpally, R.; et al. Atogepant for the preventive treatment of chronic migraine (PROGRESS): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet, 2023; in press. [Google Scholar]
- Blair, H.A. Rimegepant: A review in the acute treatment and preventive treatment of migraine. CNS Drugs 2023, 37, 255–265. [Google Scholar] [CrossRef]
- Garelja, M.L.; Hay, D.L. A narrative review of the calcitonin peptide family and associated receptors as migraine targets: Calcitonin gene-related peptide and beyond. Headache 2022, 62, 1093–1104. [Google Scholar] [CrossRef]
- Bagherzadeh-Fard, M.; Yazdanifar, M.A.; Sadeghalvad, M.; Rezaei, N. Erenumab efficacy in migraine headache prophylaxis: A systematic review. Int. Immunopharmacol. 2023, 117, 109366. [Google Scholar] [CrossRef]
- Root, S.; Ahn, K.; Kirsch, J.; Hoskin, J.L. Review of tolerability of fremanezumab for episodic and chronic migraine. Neuropsych. Dis. Treat. 2023, 19, 391–401. [Google Scholar] [CrossRef]
- Lipton, R.B.; Buse, D.C.; Sandoe, C.H.; Ford, J.H.; Hand, A.L.; Jedynak, J.P.; Port, M.P.; Detke, H.C. Changes in migraine interictal burden following treatment with galcanezumab: Results from a phase III randomized, placebo-controlled study. Headache J. Head Face Pain, 2023; in press. [Google Scholar] [CrossRef]
- Igarashi, H.; Shibata, M.; Ozeki, A.; Matsumura, T. Galcanezumab effects on migraine severity and symptoms in Japanese patients with episodic migraine: Secondary analysis of a phase 2 randomized trial. Neurol. Ther. 2023, 12, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Skljarevski, V.; Matharu, M.; Millen, B.A.; Ossipov, M.H.; Kim, B.K.; Yang, J.Y. Efficacy and safety of galcanezumab for the prevention of episodic migraine: Results of the EVOLVE-2 phase 3 randomized controlled clinical trial. Cephalalgia 2018, 38, 1442–1454. [Google Scholar] [CrossRef] [PubMed]
- Goadsby, P.J.; Barbanti, P.; Lambru, G.; Ettrup, A.; Christoffersen, C.L.; Josiassen, M.K.; Phul, R.; Sperling, B. Eptinezumab improved patient-reported outcomes and quality of life in patients with migraine and prior preventive treatment failures. Eur. J. Neurol. 2023, 30, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Starling, A.J.; Cowan, R.P.; Buse, D.C.; Diener, H.C.; Marmura, M.J.; Hirman, J.; Brevig, T.; Cady, R. Eptinezumab improved patient-reported outcomes in patients with migraine and medication-overuse headache: Subgroup analysis of the randomized PROMISE-2 trial. Headache J. Head Face Pain 2023, 63, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Radis-Baptista, G.; Konno, K. Arthropod venom components and their potential usage. Toxins 2020, 12, 82. [Google Scholar] [CrossRef] [PubMed]
- Scieuzo, C.; Salvia, R.; Franco, A.; Pezzi, M.; Cozzolino, F.; Chicca, M.; Scapoli, C.; Vogel, H.; Monti, M.; Ferracini, C.; et al. An integrated transcriptomic and proteomic approach to identify the main Torymus sinensis venom components. Sci. Rep. 2021, 11, 5032. [Google Scholar] [CrossRef]
- Averin, A.S.; Utkin, Y.N. Cardiovascular effects of snake toxins: Cardiotoxicity and cardioprotection. Acta Nat. 2021, 13, 4–14. [Google Scholar] [CrossRef]
- Walker, A.A.; Weirauch, C.; Fry, B.G.; King, G.F. Venoms of heteropteran insects: A treasure trove of diverse pharmacological toolkits. Toxins 2016, 8, 43. [Google Scholar] [CrossRef]
- Nassar, M.I. Snake venom and therapeutic potential. In Snake Venom and Ecology; Intech Open: London, UK, 2022. [Google Scholar]
- Wu, X.F.; Li, C.; Yang, G.; Wang, Y.Z.; Peng, Y.; Zhu, D.D.; Sui, A.R.; Wu, Q.; Li, Q.F.; Wang, B.; et al. Scorpion venom heat-resistant peptide attenuates microglia activation and neuroinflammation. Front. Pharmacol. 2021, 12, 704715. [Google Scholar] [CrossRef]
- dos Santos, A.T.; Cruz, G.S.; Baptista, G.R. Anti-inflammatory activities of arthropod peptides: A systematic review. J. Venom. Anim. Toxins Trop. Dis. 2021, 27, e20200152. [Google Scholar] [CrossRef] [PubMed]
- Killoran, P.M.; Capel, V.; D’Aloisio, V.; Schofield, A.; Aczél, T.; Bölcskei, K.; Helyes, Z.; von Mentzer, B.; Kendall, D.A.; Coxson, C.R.; et al. Novel peptide calcitonin gene-related pep-tide antagonists for migraine therapy. J. Pharm. Pharmacol. 2023, 75, 1581–1589. [Google Scholar] [CrossRef] [PubMed]
- Greco, R.; Demartini, C.; Francavilla, M.; Zanaboni, A.M.; Tassorelli, C. Antagonism of CGRP Receptor: Central and Peripheral Mechanisms and Mediators in an Animal Model of Chronic Migraine. Cells 2022, 11, 3092. [Google Scholar] [CrossRef] [PubMed]
- Haghdoost, F.; Puledda, F.; Garcia-Azorin, D.; Huessler, E.M.; Messina, R.; Pozo-Rosich, P. Evaluating the efficacy of CGRP mAbs and gepants for the preventive treatment of migraine: A systematic review and network meta-analysis of phase 3 randomised controlled trials. Cephalalgia 2023, 43, 03331024231159366. [Google Scholar] [CrossRef] [PubMed]
- Messina, R.; Huessler, E.M.; Puledda, F.; Haghdoost, F.; Lebedeva, E.R.; Diener, H.C. Safety and tolerability of monoclonal antibodies targeting the CGRP pathway and gepants in migraine prevention: A systematic review and network meta-analysis. Cephalalgia 2023, 43, 03331024231152169. [Google Scholar] [CrossRef] [PubMed]
- Valente, M.; Garbo, R.; Filippi, F.; Antonutti, A.; Ceccarini, V.; Tereshko, Y.; Di Lorenzo, C.; Gigli, G.L. Migraine prevention through ketogenic diet: More than body mass composition changes. J. Clin. Med. 2022, 11, 4946. [Google Scholar] [CrossRef] [PubMed]
- Lovati, C.; D’Alessandro, C.M.; Della Ventura, S.; Muzio, F.; Pantoni, L. Ketogenic diet in refractory migraine: Possible efficacy and role of ketone bodies—A pilot experience. Neurol. Sci. 2022, 43, 6479–6485. [Google Scholar] [CrossRef]
- Moskatel, L.S.; Zhang, N. Migraine and diet: Updates in understanding. Curr. Neurol. Neurosci. Rep. 2022, 22, 327–334. [Google Scholar] [CrossRef]
- Roehl, K.; Falco-Walter, J.; Ouyang, B.; Balabanov, A. Modified ketogenic diets in adults with refractory epilepsy: Efficacious improvements in seizure frequency, seizure severity, and quality of life. Epilepsy Behav. 2019, 93, 113–118. [Google Scholar] [CrossRef]
- Barbanti, P.; Fofi, L.; Aurilia, C.; Egeo, G.; Caprio, M. Ketogenic diet in migraine: Rationale, findings and perspectives. Neurol. Sci. 2017, 38 (Suppl. S1), 111–115. [Google Scholar] [CrossRef]
- Neri, L.D.C.L.; Ferraris, C.; Catalano, G.; Guglielmetti, M.; Pasca, L.; Pezzotti, E.; Carpani, A.; Tagliabue, A. Ketosis and migraine: A systematic review of the literature and meta-analysis. Front. Nutrit. 2023, 10, 1204700. [Google Scholar] [CrossRef]
- Murakami, M.; Tognini, P. Molecular mechanisms underlying the bioactive properties of a ketogenic diet. Nutrients 2022, 14, 782. [Google Scholar] [CrossRef] [PubMed]
- Newman, J.C.; Verdin, E. Ketone bodies as signaling metabolites. Trends Endocrinol. Metab. 2014, 25, 42–52. [Google Scholar] [CrossRef]
- Christensen, R.H.; Gollion, C.; Amin, F.M.; Moskowitz, M.A.; Hadjikhani, N.; Ashina, M. Imaging the inflammatory phenotype in migraine. J. Headache Pain 2022, 23, 60. [Google Scholar] [CrossRef] [PubMed]
- Reducha, P.V.; Edvinsson, L.; Haanes, K.A. Could experimental inflammation provide better understanding of migraines? Cells 2022, 11, 2444. [Google Scholar] [CrossRef]
- Lim, J.-M.; Letchumanan, V.; Tan, L.T.-H.; Hong, K.-W.; Wong, S.-H.; Ab Mutalib, N.-S.; Lee, L.-H.; Law, J.W.-F. Ketogenic diet: A dietary intervention via gut microbiome modulation for the treatment of neurological and nutritional disorders (a narrative review). Nutrients 2022, 14, 3566. [Google Scholar] [CrossRef] [PubMed]
- Veech, R.L. The therapeutic implications of ketone bodies: The effects of ketone bodies in pathological conditions: Ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Prostaglandins Leukot. Essent. Fatty Acids 2004, 70, 309–319. [Google Scholar] [CrossRef]
- Caminha, M.C.; Moreira, A.B.; Matheus, F.C.; Rieger, D.K.; Moreira, J.D.; Dalmarco, E.M.; Demarchi, I.G.; Lin, K. Efficacy and tolerability of the ketogenic diet and its variations for preventing migraine in adolescents and adults: A systematic review. Nutr. Rev. 2021, 80, 1634–1647. [Google Scholar] [CrossRef]
- Bu, X.X.; Zhu, L.H.; Wang, Z.M.; Lu, C.; Chen, H.; Yu, D. Association of obesity with headache among US children and adolescents: Evidence from NHANES 1999–2004. Front. Endocrinol. 2023, 13, 1072419. [Google Scholar] [CrossRef]
- Jahromi, S.R.; Martami, F.; Morad Soltani, K.; Togha, M. Migraine and obesity: What is the real direction of their association? Exp. Rev. Neurother. 2023, 23, 75–84. [Google Scholar] [CrossRef]
- Katalinic, D.; Vcev, A.; Smolic, M.; Aleric, I. Serotonin receptor agonists in the treatment of migraine: A meta-analysis considering possible connection with paresthesia. Ann. Ind. Acad. Neurol. 2022, 25, 332. [Google Scholar] [CrossRef]
- Gollion, C.; De Icco, R.; Dodick, D.W.; Ashina, H. The premonitory phase of migraine is due to hypothalamic dysfunction: Revisiting the evidence. J. Headache Pain 2022, 23, 158. [Google Scholar] [CrossRef]
- Bic, Z.; Blix, G.G.; Hopp, H.P.; Leslie, F.M.; Schell, M.J. The influence of a low-fat diet on incidence and severity of migraine headaches. J. Women’s Health Gender-Based Med. 1999, 8, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, L.A.; Pacioni, D.; Di Fronzo, V.; Russo, B.F.; Speranza, E.; Carlino, V.; Gargiulo, F.; Ferrara, F. Low-lipid diet reduces frequency and severity of acute migraine attacks. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Nowaczewska, M.; Wiciński, M.; Kaźmierczak, W.; Kaźmierczak, H. To eat or not to eat: A review of the relationship between chocolate and migraines. Nutrients 2020, 12, 608. [Google Scholar] [CrossRef]
- Marcus, D.A.; Scharff, L.; Turk, D.C.; Gourley, L.M. A double-blind provocative study chocolate as a trigger of headache. Cephalalgia 1997, 17, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, M.D.; Goadsby, P.J.; Burstein, R.; Kurth, T.; Ayata, C.; Charles, A.; Ashina, M.; van den Maagdenberg, A.M.J.M.; Dodick, D.W. Migraine. Nat. Rev. Dis. Prim. 2022, 8, 2. [Google Scholar] [CrossRef]
- Casanova, A.; Vives-Mestres, M.; Donoghue, S.; Mian, A.; Wöber, C. The role of avoiding known triggers, embracing protectors, and adhering to healthy lifestyle recommendations in migraine prophylaxis: Insights from a prospective cohort of 1125 people with episodic migraine. Headache 2023, 63, 51–61. [Google Scholar] [CrossRef]
- D’Amico, D.; Tepper, S.J. Prophylaxis of migraine: General principles and patient acceptance. Neuropsychiatr. Dis. Treat. 2008, 4, 1155–1167. [Google Scholar] [CrossRef]
- Hammond, N.G.; Stinchcombe, A. Health behaviors and social determinants of migraine in a Canadian population-based sample of adults aged 45–85 years: Findings from the CLSA. Headache 2019, 59, 1547–1564. [Google Scholar] [CrossRef]
- Hagen, K.; Åsberg, A.N.; Stovner, L.; Linde, M.; Zwart, J.-A.; Winsvold, B.S.; Heuch, I. Lifestyle factors and risk of migraine and tension-type headache. Follow-up data from the Nord-Trøndelag Health Surveys 1995–1997 and 2006–2008. Cephalalgia 2018, 38, 1919–1926. [Google Scholar] [CrossRef]
- Oliveira, A.B.; Peres, M.F.P.; Mercante, J.P.P.; Molina, M.D.C.B.; Lotufo, P.A.; Benseñor, I.M.; Goulart, A.C. Physical activity pattern and migraine according to aura symptoms in the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil) cohort: A cross-sectional study. Headache 2022, 62, 977–988. [Google Scholar] [CrossRef] [PubMed]
- Granato, A.; Furlanis, G.; D’Acunto, L.; Olivo, S.; Buoite Stella, A.; Manganotti, P. Lifestyle impact on migraine during home confinement. Acta Neurol. Belg. 2022, 122, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Catalano, A. COVID-19: Could irisin become the handyman myokine of the 21st century? Coronaviruses 2020, 1, 32–41. [Google Scholar] [CrossRef]
- Hamasaki, H. Effects of diaphragmatic breathing on health: A narrative review. Medicines 2020, 7, 65. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Sasaki, J.E.; Wei, G.X.; Huang, T.; Yeung, A.S.; Neto, O.B.; Chen, K.W.; Hui, S.S. Effects of mind⁻body exercises (tai chi/yoga) on heart rate variability parameters and perceived stress: A systematic review with meta-analysis of randomized controlled trials. J. Clin. Med. 2018, 7, 404. [Google Scholar] [CrossRef]
- Villemure, C.; Čeko, M.; Cotton, V.A.; Bushnell, M.C. Neuroprotective effects of yoga practice: Age-, experience-, and frequency-dependent plasticity. Front. Hum. Neurosci. 2015, 9, 281. [Google Scholar] [CrossRef]
- Terrin, A.; Mainardi, F.; Zanchin, G.; Maggioni, F. Sports, physical activity and headache in the classical age: Historical descriptions from the first sports textbook, “De arte gymnastica”, by Girolamo Mercuriale. Neurolog. Sci. 2019, 40, 1507–1517. [Google Scholar] [CrossRef]
- Kumaar, S.S. A critical analysis of chromotherapy (color therapy) and its impact on married life. Int. J. Jyotish Res. 2022, 7, 08–12. [Google Scholar] [CrossRef]
- Hall, S.; O’Brien, H. A review of the use of music therapy in the treatment of migraine. Music Med. 2022, 14, 62–68. [Google Scholar] [CrossRef]
- Derbarsegian, A.; Adams, S.M.; Phillips, K.M.; Sedaghat, A.R. The Burden of Migraine on Quality of Life in Chronic Rhinosinusitis. Laryngoscope, 2023; in press. [Google Scholar] [CrossRef]
- D’Onofrio, F.; Raimo, S.; Spitaleri, D.; Casucci, G.; Bussone, G. Usefulness of nutraceuticals in migraine prophylaxis. Neurol. Sci. 2017, 38, 117–120. [Google Scholar] [CrossRef]
- Bahrampour, N.; Mirzababaei, A.; Yarizadeh, H.; Barekzai, A.M.; Khorsha, F.; Clark, C.C.T.; Mirzaei, K. The relationship between dietary nutrients patterns and intensity and duration of migraine headaches. Br. J. Nutr. 2023, 129, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Helli, B.; Anjirizadeh, F.; Mehramiri, A.; Shalilahmadi, D.; Latifi, S.M. The effect of ginger (Zingiber officinale Rosc.) consumption in headache prophylaxis in patients with migraine: A randomized placebo-controlled clinical trial. Jundishapur J. Nat. Pharm. Prod. 2022, 17, e120449. [Google Scholar] [CrossRef]
- Chen, L.; Cai, Z. The efficacy of ginger for the treatment of migraine: A meta-analysis of randomized controlled studies. Am. J. Emerg. Med. 2021, 46, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Yarnell, E. Herbal medicine and migraine. Altern. Complement. Ther. 2017, 23, 192–201. [Google Scholar] [CrossRef]
- Egger, J.; Carter, C.M.; Wilson, J.; Turner, M.W.; Soothill, J.F. Is migraine food allergy? A double-blind controlled trial of oligoantigenic diet treatment. Lancet 1983, 2, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Arab, A.; Khorvash, F.; Karimi, E.; Heidari, Z.; Askari, G. The effects of the dietary approaches to stop hypertension (DASH) diet on oxidative stress and clinical indices of migraine patients: A randomized controlled trial. Nutrit. Neurosci. 2022, 25, 2259–2268. [Google Scholar] [CrossRef]
- Arab, A.; Khorvash, F.; Kazemi, M.; Heidari, Z.; Askari, G. Effects of the Dietary Approaches to Stop Hypertension (DASH) diet on clinical, quality of life and mental health outcomes in women with migraine: A randomised controlled trial. Br. J. Nutr. 2022, 128, 1535–1544. [Google Scholar] [CrossRef]
- Hajjarzadeh, S.; Bakhshimoghaddam, F.; Behrouz, M.; Nikniaz, Z.; Mahdavi, R.; Shalilahmadi, D.; Karandish, M. The relation of adherence to the DASH diet with migraine attack frequency and pain intensity in Iranian women: A cross-sectional study. Nutrit. Neurosci. 2023; in press. [Google Scholar] [CrossRef]
- Arab, A.; Khorvash, F.; Karimi, E.; Hadi, A.; Askari, G. Associations between adherence to Mediterranean dietary pattern and frequency, duration, and severity of migraine headache: A cross-sectional study. Nutr. Neurosci. 2021, 26, 1–10. [Google Scholar] [CrossRef]
- Straube, A.; Müller, H.; Stiegelbauer, V.; Frauwallner, A. Migraine prophylaxis with a probiotic. Results of an uncontrolled observational study with 1020 patients. MMW Fortschr. Med. 2018, 160, 16–21. [Google Scholar] [CrossRef]
- Di Maio, A.C.; Basile, G.; Iacopetta, D.; Catalano, A.; Ceramella, J.; Cafaro, D.; Saturnino, C.; Sinicropi, M.S. The significant role of nutraceutical compounds in ulcerative colitis treatment. Curr. Med. Chem. 2022, 29, 4216–4234. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, Q.; Wang, A.; Lin, Z. Structural and functional characterization of the gut microbiota in elderly women with migraine. Front. Cell. Infect. Microbiol. 2020, 9, 470. [Google Scholar] [CrossRef] [PubMed]
- De Roos, N.M.; van Hemert, S.; Rovers, J.M.P.; Smits, M.G.; Witteman, B.J.M. The effects of a multispecies probiotic on migraine and markers of intestinal permeability—Results of a randomized placebo-controlled study. Eur. J. Clin. Nutr. 2017, 71, 1455–1462. [Google Scholar] [CrossRef]
- Gazerani, P. Diet and migraine: What is proven? Curr. Opin. Neurol. 2023, 36, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Nowaczewska, M.; Wiciński, M.; Kaźmierczak, W. The ambiguous role of caffeine in migraine headache: From trigger to treatment. Nutrients 2020, 12, 2259. [Google Scholar] [CrossRef]
- Yuan, S.; Daghlas, I.; Larsson, S.C. Alcohol, coffee consumption, and smoking in relation to migraine: A bidirectional Mendelian randomization study. Pain 2022, 163, e342–e348. [Google Scholar] [CrossRef]
- Nieswand, V.; Richter, M.; Gossrau, G. Epidemiology of Headache in Children and Adolescents-Another Type of Pandemia. Curr. Pain Headache Rep. 2020, 24, 62. [Google Scholar] [CrossRef]
- Błaszczyk, B.; Straburzyński, M.; Więckiewicz, M.; Budrewicz, S.; Niemiec, P.; Staszkiewicz, M.; Waliszewska-Prosół, M. Relationship between alcohol and primary headaches: A systematic review and meta-analysis. J. Headache Pain. 2023, 24, 116. [Google Scholar] [CrossRef]
| Structure | Name | Class |
|---|---|---|
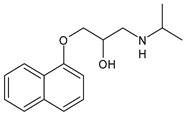 | Propranolol | β-blocker |
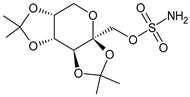 | Topiramate | β-blocker |
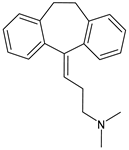 | Amitriptyline | Antidepressant |
 | Ibuprofen | FANS |
 | Acetylsalicylic acid | FANS |
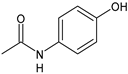 | Acetaminophen | FANS |
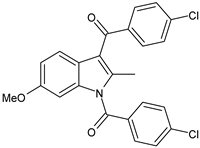 | Indomethacin | FANS |
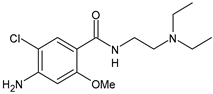 | Metoclopramide | Anti-emetic |
 | Capsaicin | Analgesic |
 | Olcegepant | CGRP inhibitor |
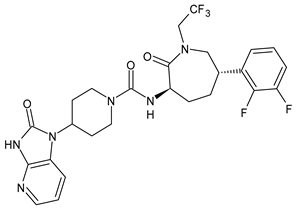 | Telcagepant | CGRP inhibitor |
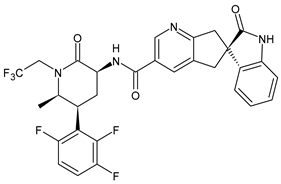 | Atogepant | CGRP inhibitor |
 | Rimegepant | CGRP inhibitor |
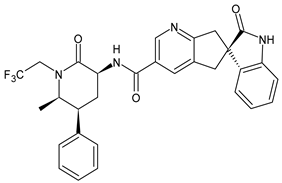 | Ubrogepant | CGRP inhibitor |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Finelli, F.; Catalano, A.; De Lisa, M.; Ferraro, G.A.; Genovese, S.; Giuzio, F.; Salvia, R.; Scieuzo, C.; Sinicropi, M.S.; Svolacchia, F.; et al. CGRP Antagonism and Ketogenic Diet in the Treatment of Migraine. Medicina 2024, 60, 163. https://doi.org/10.3390/medicina60010163
Finelli F, Catalano A, De Lisa M, Ferraro GA, Genovese S, Giuzio F, Salvia R, Scieuzo C, Sinicropi MS, Svolacchia F, et al. CGRP Antagonism and Ketogenic Diet in the Treatment of Migraine. Medicina. 2024; 60(1):163. https://doi.org/10.3390/medicina60010163
Chicago/Turabian StyleFinelli, Francesca, Alessia Catalano, Michele De Lisa, Giuseppe Andrea Ferraro, Sabino Genovese, Federica Giuzio, Rosanna Salvia, Carmen Scieuzo, Maria Stefania Sinicropi, Fabiano Svolacchia, and et al. 2024. "CGRP Antagonism and Ketogenic Diet in the Treatment of Migraine" Medicina 60, no. 1: 163. https://doi.org/10.3390/medicina60010163
APA StyleFinelli, F., Catalano, A., De Lisa, M., Ferraro, G. A., Genovese, S., Giuzio, F., Salvia, R., Scieuzo, C., Sinicropi, M. S., Svolacchia, F., Vassallo, A., Santarsiere, A., & Saturnino, C. (2024). CGRP Antagonism and Ketogenic Diet in the Treatment of Migraine. Medicina, 60(1), 163. https://doi.org/10.3390/medicina60010163












