The Effects of Environmental Exposure on Epigenetic Modifications in Allergic Diseases
Abstract
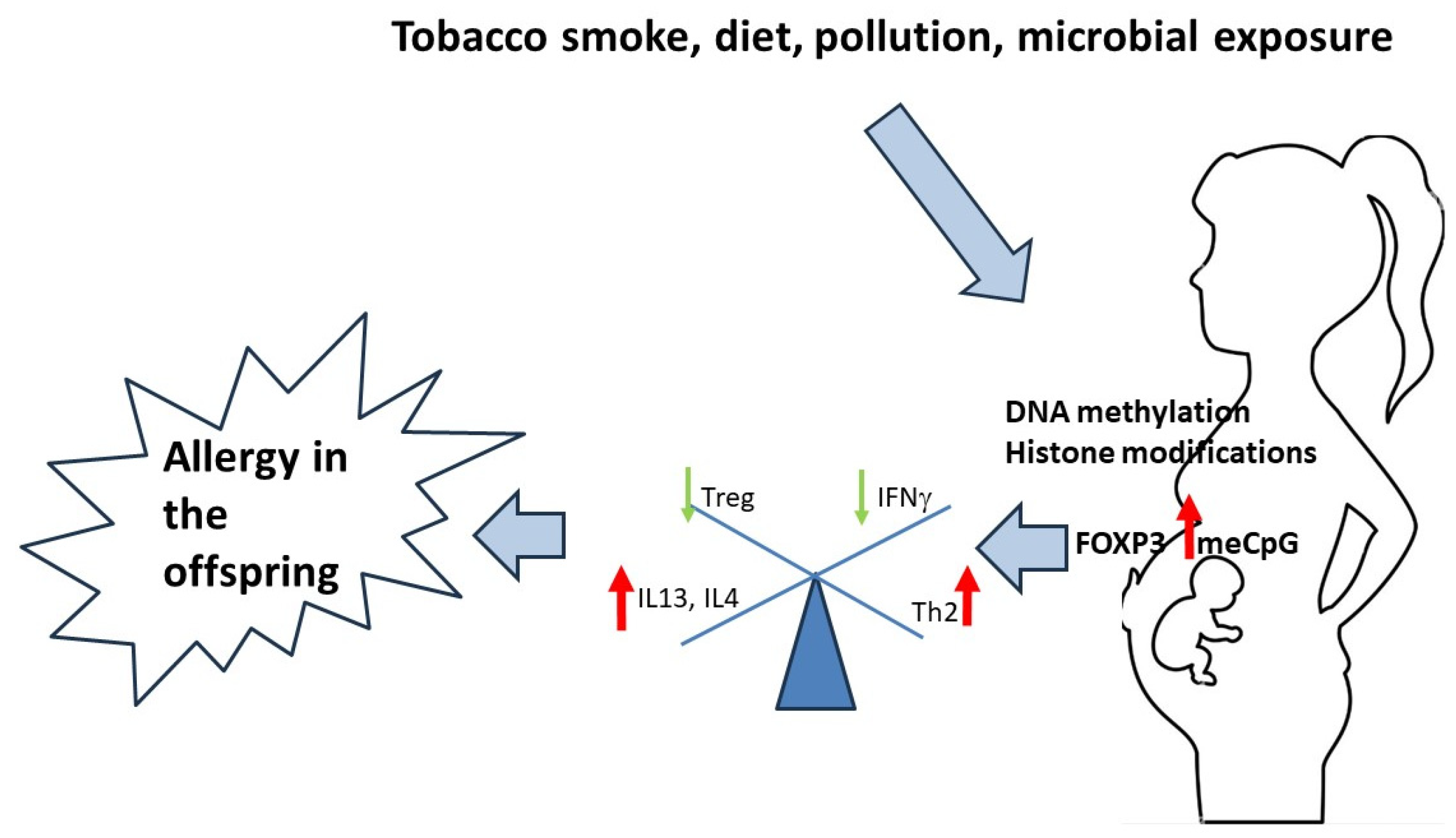
1. Introduction
- The effects of maternal and in utero exposure
- The effects of exposure to tobacco smoke
- The effects of diet
- The effects of microbial exposure
- The effects of exposure to air pollution
- The effects of exposure to persistent organic pollutants, such as pesticides
- The effects of other (intrinsic) in utero exposure
- The effects of early life events, such as viral infections in infancy
- Differential expression of small non-coding RNAs in allergy
- Epigenetic changes in treatment outcomes in allergic diseases (primarily asthma).
2. Environmental Exposure and Epigenetic Regulation in Allergic Diseases
2.1. The Effect of Maternal and In Utero Exposure on Epigenetic Modifications
2.2. Exposure to Tobacco Smoke
2.3. Epigenetic Modifications and the Effects of Diet
2.4. Effects of Microbial Exposure to Epigenetic Regulation in Allergic Diseases
2.5. Exposure to Air Pollution and Epigenetic Effects in Allergic Diseases
2.6. Exposure to Persistent Organic Pollutants and Epigenetic Effects
2.7. Other Intrauterine Factors That Affect Epigenetic Modifications and Allergic Disease Risk
2.8. Early Life Events and Epigenetic Changes Affecting Allergic Diseases
2.9. Effects of Exposure to Heavy Metals on Epigenetic Changes in Allergy
2.10. The Changing Environment and Epigenetic Changes in Allergic Diseases
2.11. Expression of Small Non-Coding RNAs and Allergic Diseases
3. Epigenetic Changes and Treatment Outcomes in Allergic Diseases
4. Environmental Exposure, Epigenetic Changes and Protection from Allergy
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Potaczek, D.P.; Harb, H.; Michel, S.; Alhamwe, B.A.; Renz, H.; Tost, J. Epigenetics and allergy: From basic mechanisms to clinical applications. Epigenomics 2017, 9, 539–571. [Google Scholar] [CrossRef]
- Galli, S.J.; Tsai, M.; Piliponsky, A.M. The Development of Allergic Inflammation. Nature 2008, 454, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Shamji, M.H.; Valenta, R.; Jardetzky, T.; Verhasselt, V.; Durham, S.R.; Würtzen, P.A.; van Neerven, R.J. The role of allergen-specific IgE, IgG and IgA in allergic disease. Allergy 2021, 76, 3627–3641. [Google Scholar] [CrossRef] [PubMed]
- Rothbauer, M.; Charwat, V.; Bachmann, B.; Sticker, D.; Novak, R.; Wanzenböck, H.; Mathies, R.A.; Ertl, P. Monitoring transient cell-to-cell interactions in a multi-layered and multi-functional allergy-on-a-chip system. Lab Chip 2019, 19, 1916–1921. [Google Scholar] [CrossRef] [PubMed]
- Dierick, B.J.H.; van der Molen, T.; Blok, B.M.J.F.-D.; Muraro, A.; Postma, M.J.; Kocks, J.W.; van Boven, J.F. Burden and socioeconomics of asthma, allergic rhinitis, atopic dermatitis and food allergy. Expert Rev. Pharmacoeconomics Outcomes Res. 2020, 20, 437–453. [Google Scholar] [CrossRef] [PubMed]
- Akdis, C.A.; Akdis, M. Mechanisms of allergen-specific immunotherapy. J. Allergy Clin. Immunol. 2011, 127, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.H.; Malone, D.C.; Lawson, K.A.; Okamoto, L.J.; Battista, C.; Saunders, W.B. A National Estimate of the Economic Costs of Asthma. Am. J. Respir. Crit. Care Med. 1997, 156, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Dupont, C.; Armant, D.R.; Brenner, C.A. Epigenetics: Definition, Mechanisms and Clinical Perspective. Semin. Reprod. Med. 2009, 27, 351–357. [Google Scholar] [CrossRef]
- Alhamwe, B.A.; Alhamdan, F.; Ruhl, A.; Potaczek, D.P.; Renz, H. The role of epigenetics in allergy and asthma development. Curr. Opin. Allergy Clin. Immunol. 2020, 20, 48–55. [Google Scholar] [CrossRef]
- Arshad, S.H.; Karmaus, W.; Raza, A.; Kurukulaaratchy, R.J.; Matthews, S.M.; Holloway, J.W.; Sadeghnejad, A.; Zhang, H.; Roberts, G.; Ewart, S.L. The effect of parental allergy on childhood allergic diseases depends on the sex of the child. J. Allergy Clin. Immunol. 2012, 130, 427–434. [Google Scholar] [CrossRef]
- Martino, D.; Prescott, S. Epigenetics and Prenatal Influences on Asthma and Allergic Airways Disease. Chest 2011, 139, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Fiuza, B.S.D.; Fonseca, H.F.; Meirelles, P.M.; Marques, C.R.; da Silva, T.M.; Figueiredo, C.A. Understanding Asthma and Allergies by the Lens of Biodiversity and Epigenetic Changes. Front. Immunol. 2021, 12, 623737. [Google Scholar] [CrossRef] [PubMed]
- Kabesch, M.; Tost, J. Recent findings in the genetics and epigenetics of asthma and allergy. Semin. Immunopathol. 2020, 42, 43–60. [Google Scholar] [CrossRef] [PubMed]
- Breton, C.V.; Byun, H.-M.; Wenten, M.; Pan, F.; Yang, A.; Gilliland, F.D. Prenatal Tobacco Smoke Exposure Affects Global and Gene-specific DNA Methylation. Am. J. Respir. Crit. Care Med. 2009, 180, 462–467. [Google Scholar] [CrossRef]
- Liang, L.; Willis-Owen, S.A.G.; Laprise, C.; Wong, K.C.C.; Davies, G.A.; Hudson, T.J.; Binia, A.; Hopkin, J.M.; Yang, I.V.; Grundberg, E.; et al. An epigenome-wide association study of total serum immunoglobulin E concentration. Nature 2015, 520, 670–674. [Google Scholar] [CrossRef]
- Xu, C.-J.; Söderhäll, C.; Bustamante, M.; Baïz, N.; Gruzieva, O.; Gehring, U.; Mason, D.; Chatzi, L.; Basterrechea, M.; Llop, S.; et al. DNA methylation in childhood asthma: An epigenome-wide meta-analysis. Lancet Respir. Med. 2018, 6, 379–388. [Google Scholar] [CrossRef]
- Rodríguez, E.; Baurecht, H.; Wahn, A.F.; Kretschmer, A.; Hotze, M.; Zeilinger, S.; Klopp, N.; Illig, T.; Schramm, K.; Prokisch, H.; et al. An Integrated Epigenetic and Transcriptomic Analysis Reveals Distinct Tissue-Specific Patterns of DNA Methylation Associated with Atopic Dermatitis. J. Investig. Dermatol. 2014, 134, 1873–1883. [Google Scholar] [CrossRef]
- Martino, D.; Neeland, M.; Dang, T.; Cobb, J.; Ellis, J.; Barnett, A.; Tang, M.; Vuillermin, P.; Allen, K.; Saffery, R. Epigenetic dysregulation of naive CD4+ T-cell activation genes in childhood food allergy. Nat. Commun. 2018, 9, 3308. [Google Scholar] [CrossRef]
- Nestor, C.E.; Barrenäs, F.; Wang, H.; Lentini, A.; Zhang, H.; Bruhn, S.; Jörnsten, R.; Langston, M.A.; Rogers, G.; Gustafsson, M.; et al. DNA Methylation Changes Separate Allergic Patients from Healthy Controls and May Reflect Altered CD4+ T-Cell Population Structure. PLoS Genet. 2014, 10, e1004059. [Google Scholar] [CrossRef]
- DeVries, A.; Wlasiuk, G.; Miller, S.J.; Bosco, A.; Stern, D.A.; Lohman, I.C.; Rothers, J.; Jones, A.C.; Nicodemus-Johnson, J.; Vasquez, M.M.; et al. Epigenome-wide analysis links SMAD3 methylation at birth to asthma in children of asthmatic mothers. J. Allergy Clin. Immunol. 2016, 140, 534–542. [Google Scholar] [CrossRef]
- Hylkema, M.N.; Blacquiere, M.J. Intrauterine Effects of Maternal Smoking on Sensitization, Asthma, and Chronic Obstructive Pulmonary Disease. Proc. Am. Thorac. Soc. 2009, 6, 660–662. [Google Scholar] [CrossRef] [PubMed]
- Noakes, P.S.; Holt, P.G.; Prescott, S.L. Maternal smoking in pregnancy alters neonatal cytokine responses. Allergy 2003, 58, 1053–1058. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, J.A.; Mori, T.A.; Barden, A.; Beilin, L.J.; Taylor, A.L.; Holt, P.G.; Prescott, S.L. Fish oil supplementation in pregnancy modifies neonatal allergen-specific immune responses and clinical outcomes in infants at high risk of atopy: A randomized, controlled trial. J. Allergy Clin. Immunol. 2003, 112, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Chatzi, L.; Torrent, M.; Romieu, I.; Garcia-Esteban, R.; Ferrer, C.; Vioque, J.; Kogevinas, M.; Sunyer, J. Mediterranean diet in pregnancy is protective for wheeze and atopy in childhood. Thorax 2008, 63, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Bobetsis, Y.A.; Barros, S.P.; Lin, D.M.; Weidman, J.R.; Dolinoy, D.C.; Jirtle, R.L.; Boggess, K.A.; Beck, J.D.; Offenbacher, S. Bacterial Infection Promotes DNA Hypermethylation. J. Dent. Res. 2007, 86, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Schaub, B.; Liu, J.; Höppler, S.; Schleich, I.; Huehn, J.; Olek, S.; Wieczorek, G.; Illi, S.; von Mutius, E. Maternal farm exposure modulates neonatal immune mechanisms through regulatory T cells. J. Allergy Clin. Immunol. 2009, 123, 774–782. [Google Scholar] [CrossRef]
- Liu, J.; Ballaney, M.; Al-Alem, U.; Quan, C.; Jin, X.; Perera, F.; Chen, L.-C.; Miller, R.L. Combined Inhaled Diesel Exhaust Particles and Allergen Exposure Alter Methylation of T Helper Genes and IgE Production In Vivo. Toxicol. Sci. 2008, 102, 76–81. [Google Scholar] [CrossRef]
- Webster, R.B.; Rodriguez, Y.; Klimecki, W.T.; Vercelli, D. The Human IL-13 Locus in Neonatal CD4+ T Cells Is Refractory to the Acquisition of a Repressive Chromatin Architecture. J. Biol. Chem. 2007, 282, 700–709. [Google Scholar] [CrossRef]
- Jones, B.; Chen, J. Inhibition of IFN-γ transcription by site-specific methylation during T helper cell development. EMBO J. 2006, 25, 2443–2452. [Google Scholar] [CrossRef]
- Schoenborn, J.R.; Dorschner, M.O.; Sekimata, M.; Santer, D.M.; Shnyreva, M.; Fitzpatrick, D.R.; A Stamatoyannopoulos, J.; Wilson, C.B. Comprehensive epigenetic profiling identifies multiple distal regulatory elements directing transcription of the gene encoding interferon-γ. Nat. Immunol. 2007, 8, 732–742. [Google Scholar] [CrossRef]
- Huehn, J.; Polansky, J.K.; Hamann, A. Epigenetic control of FOXP3 expression: The key to a stable regulatory T-cell lineage? Nat. Rev. Immunol. 2009, 9, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Yerkovich, S.T.; Wikström, M.E.; Suriyaarachchi, D.; Prescott, S.L.; Upham, J.W.; Holt, P.G. Postnatal Development of Monocyte Cytokine Responses to Bacterial Lipopolysaccharide. Pediatr. Res. 2007, 62, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, Y.; Yasutomi, M.; Omata, N.; Yamada, A.; Fujisawa, K.; Kasuga, K.; Hiraoka, M.; Mayumi, M. Dysregulation of IL-13 Production by Cord Blood CD4+ T Cells Is Associated with the Subsequent Development of Atopic Disease in Infants. Pediatr. Res. 2002, 51, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Baron, U.; Floess, S.; Wieczorek, G.; Baumann, K.; Grützkau, A.; Dong, J.; Thiel, A.; Boeld, T.J.; Hoffmann, P.; Edinger, M.; et al. DNA demethylation in the human FOXP3 locus discriminates regulatory T cells from activated FOXP3+ conventional T cells. Eur. J. Immunol. 2007, 37, 2378–2389. [Google Scholar] [CrossRef] [PubMed]
- Martino, D.J.; Prescott, S.L. Silent mysteries: Epigenetic paradigms could hold the key to conquering the epidemic of allergy and immune disease. Allergy 2010, 65, 7–15. [Google Scholar] [CrossRef]
- Han, P.; Gu, J.-Q.; Li, L.-S.; Wang, X.-Y.; Wang, H.-T.; Wang, Y.; Chang, C.; Sun, J.-L. The Association Between Intestinal Bacteria and Allergic Diseases—Cause or Consequence? Front. Cell. Infect. Microbiol. 2021, 11, 650893. [Google Scholar] [CrossRef]
- Zakarya, R.; Adcock, I.; Oliver, B.G. Epigenetic impacts of maternal tobacco and e-vapour exposure on the offspring lung. Clin. Epigenetics 2019, 11, 32. [Google Scholar] [CrossRef]
- Neophytou, A.M.; Oh, S.S.; Hu, D.; Huntsman, S.; Eng, C.; Rodríguez-Santana, J.R.; Kumar, R.; Balmes, J.R.; Eisen, E.A.; Burchard, E.G. In utero tobacco smoke exposure, DNA methylation, and asthma in Latino children. Environ. Epidemiology 2019, 3, e048. [Google Scholar] [CrossRef]
- Wu, C.-C.; Hsu, T.-Y.; Chang, J.-C.; Ou, C.-Y.; Kuo, H.-C.; Liu, C.-A.; Wang, C.-L.; Chuang, H.; Chen, C.-P.; Yang, K.D. Paternal Tobacco Smoke Correlated to Offspring Asthma and Prenatal Epigenetic Programming. Front. Genet. 2019, 10, 471. [Google Scholar] [CrossRef]
- Liu, J.; Bowatte, G.; Pham, J.; Perret, J.L.; Holloway, J.W.; Lowe, A.J.; Burgess, J.A.; Svanes, C.; Thomas, P.; Russell, M.A.; et al. Pre-pubertal smoke exposure of fathers and increased risk of offspring asthma: A possible transgenerational effect. Eur. Respir. J. 2022, 60, 2200257. [Google Scholar] [CrossRef]
- Ulrey, C.L.; Liu, L.; Andrews, L.G.; Tollefsbol, T.O. The impact of metabolism on DNA methylation. Hum. Mol. Genet. 2005, 14, R139–R147. [Google Scholar] [CrossRef] [PubMed]
- Prescott, S.; Saffery, R. The role of epigenetic dysregulation in the epidemic of allergic disease. Clin. Epigenetics 2011, 2, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Håberg, S.E.; London, S.J.; Stigum, H.; Nafstad, P.; Nystad, W. Folic acid supplements in pregnancy and early childhood respiratory health. Arch. Dis. Child. 2009, 94, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Whitrow, M.J.; Moore, V.M.; Rumbold, A.R.; Davies, M.J. Effect of Supplemental Folic Acid in Pregnancy on Childhood Asthma: A Prospective Birth Cohort Study. Am. J. Epidemiology 2009, 170, 1486–1493. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, N.; Alashkar Alhamwe, B.; Caraballo, L.; Ding, M.; Ferrante, A.; Garn, H.; Garssen, J.; Hii, C.S.; Irvine, J.; Llinás-Caballero, K.; et al. Perinatal and Early-Life Nutrition, Epigenetics, and Allergy. Nutrients 2021, 13, 724. [Google Scholar] [CrossRef] [PubMed]
- Idris, F.S.; Shaikh, H.A.; Vahora, I.; Moparthi, K.P.; Al Rushaidi, M.T.; Muddam, M.; Obajeun, O.A.; Abaza, A.; Jaramillo, A.P.; Hamid, P. Maternal Diet and Infant Risk of Eczema and Food Allergy: A Systematic Review. Cureus 2023, 15, e45114. [Google Scholar] [CrossRef] [PubMed]
- Wesemann, D.R.; Nagler, C.R. The Microbiome, Timing, and Barrier Function in the Context of Allergic Disease. Immunity 2016, 44, 728–738. [Google Scholar] [CrossRef] [PubMed]
- Savage, J.H.; Lee-Sarwar, K.A.; Sordillo, J.; Bunyavanich, S.; Zhou, Y.; O’Connor, G.; Sandel, M.; Bacharier, L.B.; Zeiger, R.; Sodergren, E.; et al. A prospective microbiome-wide association study of food sensitization and food allergy in early childhood. Allergy 2018, 73, 145–152. [Google Scholar] [CrossRef]
- Huang, Y.J.; Marsland, B.J.; Bunyavanich, S.; O’Mahony, L.; Leung, D.Y.M.; Muraro, A.; Fleisher, T.A. The microbiome in allergic disease: Current understanding and future opportunities—2017 PRACTALL document of the American Academy of Allergy, Asthma & Immunology and the European Academy of Allergy and Clinical Immunology. J. Allergy Clin. Immunol. 2017, 139, 1099–1110. [Google Scholar] [CrossRef]
- Woo, V.; Alenghat, T. Host–microbiota interactions: Epigenomic regulation. Curr. Opin. Immunol. 2017, 44, 52–60. [Google Scholar] [CrossRef]
- Dai, Z.; Ramesh, V.; Locasale, J.W. The evolving metabolic landscape of chromatin biology and epigenetics. Nat. Rev. Genet. 2020, 21, 737–753. [Google Scholar] [CrossRef]
- Sandini, U.; Kukkonen, A.K.; Poussa, T.; Sandini, L.; Savilahti, E.; Kuitunen, M. Protective and Risk Factors for Allergic Diseases in High-Risk Children at the Ages of Two and Five Years. Int. Arch. Allergy Immunol. 2011, 156, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Vuillermin, P.J.; Ponsonby, A.L.; Saffery, R.; Tang, M.L.; Ellis, J.A.; Sly, P.; Holt, P. Microbial exposure, interferon gamma gene demethylation in naïve T-cells, and the risk of allergic disease. Allergy 2009, 64, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Omenetti, S.; Pizarro, T.T. The Treg/Th17 Axis: A Dynamic Balance Regulated by the Gut Microbiome. Front. Immunol. 2015, 6, 639. [Google Scholar] [CrossRef] [PubMed]
- Kamada, N.; Seo, S.-U.; Chen, G.Y.; Núñez, G. Role of the gut microbiota in immunity and inflammatory disease. Nat. Rev. Immunol. 2013, 13, 321–335. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I.; Adcock, I.M. Oxidative stress and redox regulation of lung inflammation in COPD. Eur. Respir. J. 2006, 28, 219–242. [Google Scholar] [CrossRef] [PubMed]
- Perera, F.; Tang, W.-Y.; Herbstman, J.; Tang, D.; Levin, L.; Miller, R.; Ho, S.-M. Relation of DNA Methylation of 5′-CpG Island of ACSL3 to Transplacental Exposure to Airborne Polycyclic Aromatic Hydrocarbons and Childhood Asthma. PLoS ONE 2009, 4, 4488. [Google Scholar] [CrossRef]
- Hew, K.M.; Walker, A.I.; Kohli, A.; Garcia, M.; Syed, A.; McDonald-Hyman, C.; Noth, E.M.; Mann, J.K.; Pratt, B.; Balmes, J.; et al. Childhood exposure to ambient polycyclic aromatic hydrocarbons is linked to epigenetic modifications and impaired systemic immunity in T cells. Clin. Exp. Allergy 2014, 45, 238–248. [Google Scholar] [CrossRef]
- Tsamou, M.; Vrijens, K.; Madhloum, N.; Lefebvre, W.; Vanpoucke, C.; Nawrot, T.S. Air pollution-induced placental epigenetic alterations in early life: A candidate miRNA approach. Epigenetics 2018, 13, 135–146. [Google Scholar] [CrossRef]
- Baskara-Yhuellou, I.; Tost, J. The Impact of MicroRNAs on Alterations of Gene Regulatory Networks in Allergic Diseases. Adv. Protein Chem. Struct. Biol. 2020, 120, 237–312. [Google Scholar] [CrossRef]
- Rodrigues, M.d.B.; de Carvalho, D.S.; Chong-Silva, D.C.; Urrutia-Pereira, M.; de Albuquerque, G.S.C.; Cieslak, F.; Chong-Neto, H.J. Association between exposure to pesticides and allergic diseases in children and adolescents: A systematic review with meta-analysis. J. Pediatr. 2022, 98, 551–564. [Google Scholar] [CrossRef]
- Daniel, V.; Huber, W.; Bauer, K.; Suesal, C.; Conradt, C.; Opelz, G. Associations of blood levels of PCB, HCHS, and HCB with numbers of lymphocyte subpopulations, in vitro lymphocyte response, plasma cytokine levels, and immunoglobulin autoantibodies. Environ. Health Perspect. 2001, 109, 173–178. [Google Scholar] [CrossRef]
- Baccarelli, A.; Bollati, V. Epigenetics and environmental chemicals. Curr. Opin. Pediatr. 2009, 21, 243–251. [Google Scholar] [CrossRef]
- Hoang, T.T.; Qi, C.; Paul, K.C.; Lee, M.; White, J.D.; Richards, M.; Auerbach, S.S.; Long, S.; Shrestha, S.; Wang, T.; et al. Epigenome-Wide DNA Methylation and Pesticide Use in the Agricultural Lung Health Study. Environ. Health Perspect. 2021, 129, 97008. [Google Scholar] [CrossRef]
- Kim, K.-Y.; Kim, D.-S.; Lee, S.-K.; Lee, I.-K.; Kang, J.-H.; Chang, Y.-S.; Jacobs, D.R.; Steffes, M.; Lee, D.-H. Association of Low-Dose Exposure to Persistent Organic Pollutants with Global DNA Hypomethylation in Healthy Koreans. Environ. Health Perspect. 2010, 118, 370–374. [Google Scholar] [CrossRef]
- Prescott, S.L.; Holt, P.G.; Jenmalm, M.; Björkstén, B. Effects of maternal allergen-specific IgG in cord blood on early postnatal development of allergen-specific T-cell immunity. Allergy 2000, 55, 469–474. [Google Scholar] [CrossRef]
- Prescott, S.L.; Breckler, L.A.; Witt, C.S.; Smith, L.; Dunstan, J.A.; Christiansen, F.T. Allergic women show reduced T helper type 1 alloresponses to fetal human leucocyte antigen mismatch during pregnancy. Clin. Exp. Immunol. 2010, 159, 65–72. [Google Scholar] [CrossRef]
- Gheorghe, C.P.; Goyal, R.; Mittal, A.; Longo, L.D. Gene expression in the placenta: Maternal stress and epigenetic responses. Int. J. Dev. Biol. 2010, 54, 507–523. [Google Scholar] [CrossRef]
- Prescott, S.L.; Clifton, V. Asthma and pregnancy: Emerging evidence of epigenetic interactions in utero. Curr. Opin. Allergy Clin. Immunol. 2009, 9, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Scott, N.M.; Hodyl, N.A.; Murphy, V.E.; Osei-Kumah, A.; Wyper, H.; Hodgson, D.M.; Smith, R.; Clifton, V.L. Placental Cytokine Expression Covaries with Maternal Asthma Severity and Fetal Sex. J. Immunol. 2009, 182, 1411–1420. [Google Scholar] [CrossRef] [PubMed]
- Yang, I.V.; Zhang, W.; Davidson, E.J.; Fingerlin, T.E.; Kechris, K.; Dabelea, D. Epigenetic marks of in utero exposure to gestational diabetes and childhood adiposity outcomes: The EPOCH study. Diabet. Med. 2018, 35, 612–620. [Google Scholar] [CrossRef]
- Hsu, M.-C.; Lin, C.-H.; Lin, M.-C. Maternal gestational diabetes mellitus and risk of allergic diseases in offspring. Pediatr. Neonatol. 2023; Epub ahead of print. [Google Scholar] [CrossRef]
- Tulic, M.K.; Hodder, M.; Forsberg, A.; McCarthy, S.; Richman, T.; D’Vaz, N.; Van Den Biggelaar, A.H.; Thornton, C.A.; Prescott, S.L. Differences in innate immune function between allergic and nonallergic children: New insights into immune ontogeny. J. Allergy Clin. Immunol. 2011, 127, 470–478.e1. [Google Scholar] [CrossRef]
- Kumar, R.K.; Hitchins, M.P.; Foster, P.S. Epigenetic changes in childhood asthma. Dis. Model. Mech. 2009, 2, 549–553. [Google Scholar] [CrossRef]
- Fonseca, W.; Lukacs, N.W.; Ptaschinski, C. Factors Affecting the Immunity to Respiratory Syncytial Virus: From Epigenetics to Microbiome. Front. Immunol. 2018, 9, 226. [Google Scholar] [CrossRef]
- Oddy, W.H.; de Klerk, N.; Sly, P.; Holt, P. The effects of respiratory infections, atopy, and breastfeeding on childhood asthma. Eur. Respir. J. 2002, 19, 899–905. [Google Scholar] [CrossRef]
- Higashimoto, Y.; Elliott, W.M.; Behzad, A.R.; Sedgwick, E.G.; Takei, T.; Hogg, J.C.; Hayashi, S. Inflammatory Mediator mRNA Expression by Adenovirus E1A-Transfected Bronchial Epithelial Cells. Am. J. Respir. Crit. Care Med. 2002, 166, 200–207. [Google Scholar] [CrossRef]
- Hogg, J.C. Role of Latent Viral Infections in Chronic Obstructive Pulmonary Disease and Asthma. Am. J. Respir. Crit. Care Med. 2001, 164, S71–S75. [Google Scholar] [CrossRef] [PubMed]
- Mikovits, J.A.; Young, H.A.; Vertino, P.; Issa, J.-P.J.; Pitha, P.M.; Turcoski-Corrales, S.; Taub, D.D.; Petrow, C.L.; Baylin, S.B.; Ruscetti, F.W. Infection with Human Immunodeficiency Virus Type 1 Upregulates DNA Methyltransferase, Resulting in De Novo Methylation of the Gamma Interferon (IFN-γ) Promoter and Subsequent Downregulation of IFN-γ Production. Mol. Cell. Biol. 1998, 18, 5166–5177. [Google Scholar] [CrossRef] [PubMed]
- Siegle, J.S.; Hansbro, N.; Herbert, C.; Rosenberg, H.F.; Domachowske, J.B.; Asquith, K.L.; Foster, P.S.; Kumar, R.K. Early-life viral infection and allergen exposure interact to induce an asthmatic phenotype in mice. Respir. Res. 2010, 11, 14. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.-M.; Johnson, A.; Tarapore, P.; Janakiram, V.; Zhang, X.; Leung, Y.-K. Environmental Epigenetics and Its Implication on Disease Risk and Health Outcomes. ILAR J. 2012, 53, 289–305. [Google Scholar] [CrossRef]
- Ijomone, O.M.; Ijomone, O.K.; Iroegbu, J.D.; Ifenatuoha, C.W.; Olung, N.F.; Aschner, M. Epigenetic influence of environmentally neurotoxic metals. NeuroToxicology 2020, 81, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Stepanyan, A.; Petrackova, A.; Hakobyan, S.; Savara, J.; Davitavyan, S.; Kriegova, E.; Arakelyan, A. Long-term environmental metal exposure is associated with hypomethylation of CpG sites in NFKB1 and other genes related to oncogenesis. Clin. Epigenetics 2023, 15, 126. [Google Scholar] [CrossRef]
- Cardenas, A.; Fadadu, R.; Bunyavanich, S. Climate change and epigenetic biomarkers in allergic and airway diseases. J. Allergy Clin. Immunol. 2023, 152, 1060–1072. [Google Scholar] [CrossRef] [PubMed]
- Kasten-Jolly, J.; Heo, Y.; Lawrence, D.A. Impact of developmental lead exposure on splenic factors. Toxicol. Appl. Pharmacol. 2010, 247, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Mishra, K.P. Lead exposure and its impact on immune system: A review. Toxicol. In Vitro 2009, 23, 969–972. [Google Scholar] [CrossRef]
- Mitra, S.; Chakraborty, A.J.; Tareq, A.M.; Bin Emran, T.; Nainu, F.; Khusro, A.; Idris, A.M.; Khandaker, M.U.; Osman, H.; Alhumaydhi, F.A.; et al. Impact of heavy metals on the environment and human health: Novel therapeutic insights to counter the toxicity. J. King Saud Univ.-Sci. 2022, 34, 101865. [Google Scholar] [CrossRef]
- Pawankar, R.; Akdis, C.A. Climate change and the epithelial barrier theory in allergic diseases: A One Health approach to a green environment. Allergy 2023, 78, 2829–2834. [Google Scholar] [CrossRef]
- Luschkova, D.; Traidl-Hoffmann, C.; Ludwig, A. Climate change and allergies. Allergo J. Int. 2022, 31, 114–120. [Google Scholar] [CrossRef]
- Peng, C.; Cardenas, A.; Rifas-Shiman, S.L.; Hivert, M.-F.; Gold, D.R.; Platts-Mills, T.A.; Lin, X.; Oken, E.; Avila, L.; Celedón, J.C.; et al. Epigenetic age acceleration is associated with allergy and asthma in children in Project Viva. J. Allergy Clin. Immunol. 2019, 143, 2263–2270.e14. [Google Scholar] [CrossRef]
- Straight, B.; Qiao, X.; Ngo, D.; Hilton, C.E.; Olungah, C.O.; Naugle, A.; Lalancette, C.; Needham, B.L. Epigenetic mechanisms underlying the association between maternal climate stress and child growth: Characterizing severe drought and its impact on a Kenyan community engaging in a climate change-sensitive livelihood. Epigenetics 2022, 17, 2421–2433. [Google Scholar] [CrossRef]
- Prunicki, M.; Kelsey, R.; Lee, J.; Zhou, X.; Smith, E.; Haddad, F.; Wu, J.; Nadeau, K. The impact of prescribed fire versus wildfire on the immune and cardiovascular systems of children. Allergy 2019, 74, 1989–1991. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Targeting the Epigenome in the Treatment of Asthma and Chronic Obstructive Pulmonary Disease. Proc. Am. Thorac. Soc. 2009, 6, 693–696. [Google Scholar] [CrossRef] [PubMed]
- Heffler, E.; Allegra, A.; Pioggia, G.; Picardi, G.; Musolino, C.; Gangemi, S. MicroRNA Profiling in Asthma: Potential Biomarkers and Therapeutic Targets. Am. J. Respir. Cell Mol. Biol. 2017, 57, 642–650. [Google Scholar] [CrossRef] [PubMed]
- Pinkerton, M.; Chinchilli, V.; Banta, E.; Craig, T.; August, A.; Bascom, R.; Cantorna, M.; Harvill, E.; Ishmael, F.T. Differential expression of microRNAs in exhaled breath condensates of patients with asthma, patients with chronic obstructive pulmonary disease, and healthy adults. J. Allergy Clin. Immunol. 2013, 132, 217–219.e2. [Google Scholar] [CrossRef] [PubMed]
- Panganiban, R.P.L.; Pinkerton, M.H.; Maru, S.Y.; Jefferson, S.J.; Roff, A.N.; Ishmael, F.T. Differential microRNA epression in asthma and the role of miR-1248 in regulation of IL-5. Am. J. Clin. Exp. Immunol. 2012, 1, 154–165. [Google Scholar] [PubMed]
- Dong, X.; Xu, M.; Ren, Z.; Gu, J.; Lu, M.; Lu, Q.; Zhong, N. Regulation of CBL and ESR1 expression by microRNA-22-3p, 513a-5p and 625-5p may impact the pathogenesis of dust mite-induced pediatric asthma. Int. J. Mol. Med. 2016, 38, 446–456. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jardim, M.J.; Dailey, L.; Silbajoris, R.; Diaz-Sanchez, D. Distinct MicroRNA Expression in Human Airway Cells of Asthmatic Donors Identifies a Novel Asthma-Associated Gene. Am. J. Respir. Cell Mol. Biol. 2012, 47, 536–542. [Google Scholar] [CrossRef]
- Polikepahad, S.; Knight, J.M.; Naghavi, A.O.; Oplt, T.; Creighton, C.J.; Shaw, C.; Benham, A.L.; Kim, J.; Soibam, B.; Harris, R.A.; et al. Proinflammatory Role for let-7 MicroRNAS in Experimental Asthma. J. Biol. Chem. 2010, 285, 30139–30149. [Google Scholar] [CrossRef]
- Collison, A.; Mattes, J.; Plank, M.; Foster, P.S. Inhibition of house dust mite–induced allergic airways disease by antagonism of microRNA-145 is comparable to glucocorticoid treatment. J. Allergy Clin. Immunol. 2011, 128, 160–167.e4. [Google Scholar] [CrossRef]
- Lu, T.X.; Rothenberg, M.E. Diagnostic, functional, and therapeutic roles of microRNA in allergic diseases. J. Allergy Clin. Immunol. 2013, 132, 3–13. [Google Scholar] [CrossRef]
- Weidner, J.; Bartel, S.; Kılıç, A.; Zissler, U.M.; Renz, H.; Schwarze, J.; Schmidt-Weber, C.B.; Maes, T.; Rebane, A.; Krauss-Etschmann, S.; et al. Spotlight on microRNAs in allergy and asthma. Allergy 2020, 76, 1661–1678. [Google Scholar] [CrossRef] [PubMed]
- Herberth, G.; Bauer, M.; Gasch, M.; Hinz, D.; Röder, S.; Olek, S.; Kohajda, T.; Rolle-Kampczyk, U.; von Bergen, M.; Sack, U.; et al. Maternal and cord blood miR-223 expression associates with prenatal tobacco smoke exposure and low regulatory T-cell numbers. J. Allergy Clin. Immunol. 2014, 133, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Wang, W.; Jing, W. Indoor air pollution aggravates asthma in Chinese children and induces the changes in serum level of miR-155. Int. J. Environ. Health Res. 2019, 29, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Yang, I.V.; Pedersen, B.S.; Liu, A.H.; O’Connor, G.T.; Pillai, D.; Kattan, M.; Misiak, R.T.; Gruchalla, R.; Szefler, S.J.; Hershey, G.K.K.; et al. The nasal methylome and childhood atopic asthma. J. Allergy Clin. Immunol. 2016, 139, 1478–1488. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus-Johnson, J.; Myers, R.A.; Sakabe, N.J.; Sobreira, D.R.; Hogarth, D.K.; Naureckas, E.T.; Sperling, A.I.; Solway, J.; White, S.R.; Nobrega, M.A.; et al. DNA methylation in lung cells is associated with asthma endotypes and genetic risk. JCI Insight 2016, 1, e90151. [Google Scholar] [CrossRef] [PubMed]
- Tost, J. A translational perspective on epigenetics in allergic diseases. J. Allergy Clin. Immunol. 2018, 142, 715–726. [Google Scholar] [CrossRef]
- Lovinsky-Desir, S.; Miller, R.L. Epigenetics, Asthma, and Allergic Diseases: A Review of the Latest Advancements. Curr. Allergy Asthma Rep. 2012, 12, 211–220. [Google Scholar] [CrossRef]
- Li, J.; Panganiban, R.; Kho, A.T.; McGeachie, M.J.; Farnam, L.; Chase, R.P.; Weiss, S.T.; Lu, Q.; Tantisira, K.G. Circulating MicroRNAs and Treatment Response in Childhood Asthma. Am. J. Respir. Crit. Care Med. 2020, 202, 65–72. [Google Scholar] [CrossRef]
- Ito, K.; Lim, S.; Caramori, G.; Cosio, B.G.; Chung, K.F.; Adcock, I.M.; Barnes, P.J. A molecular mechanism of action of theophylline: Induction of histone deacetylase activity to decrease inflammatory gene expression. Proc. Natl. Acad. Sci. USA 2002, 99, 8921–8926. [Google Scholar] [CrossRef]
- Rebane, A.; Akdis, C.A. MicroRNAs: Essential players in the regulation of inflammation. J. Allergy Clin. Immunol. 2013, 132, 15–26. [Google Scholar] [CrossRef]
- Smits, H.H.; Akdis, C.A. In utero priming by worms protects against respiratory allergies. J. Allergy Clin. Immunol. 2014, 134, 1280–1281. [Google Scholar] [CrossRef] [PubMed]
- Arzola-Martínez, L.; Ptaschinski, C.; Lukacs, N.W. Trained innate immunity, epigenetics, and food allergy. Front. Allergy 2023, 4, 1105588. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mijač, S.; Banić, I.; Genc, A.-M.; Lipej, M.; Turkalj, M. The Effects of Environmental Exposure on Epigenetic Modifications in Allergic Diseases. Medicina 2024, 60, 110. https://doi.org/10.3390/medicina60010110
Mijač S, Banić I, Genc A-M, Lipej M, Turkalj M. The Effects of Environmental Exposure on Epigenetic Modifications in Allergic Diseases. Medicina. 2024; 60(1):110. https://doi.org/10.3390/medicina60010110
Chicago/Turabian StyleMijač, Sandra, Ivana Banić, Ana-Marija Genc, Marcel Lipej, and Mirjana Turkalj. 2024. "The Effects of Environmental Exposure on Epigenetic Modifications in Allergic Diseases" Medicina 60, no. 1: 110. https://doi.org/10.3390/medicina60010110
APA StyleMijač, S., Banić, I., Genc, A.-M., Lipej, M., & Turkalj, M. (2024). The Effects of Environmental Exposure on Epigenetic Modifications in Allergic Diseases. Medicina, 60(1), 110. https://doi.org/10.3390/medicina60010110






