Matrix Metalloproteinases and Heart Transplantation—A Pathophysiological and Clinical View
Abstract
1. Introduction
2. Aim
3. MMPs: A General Introduction
4. MMP Classification
5. MMP Role and Localization
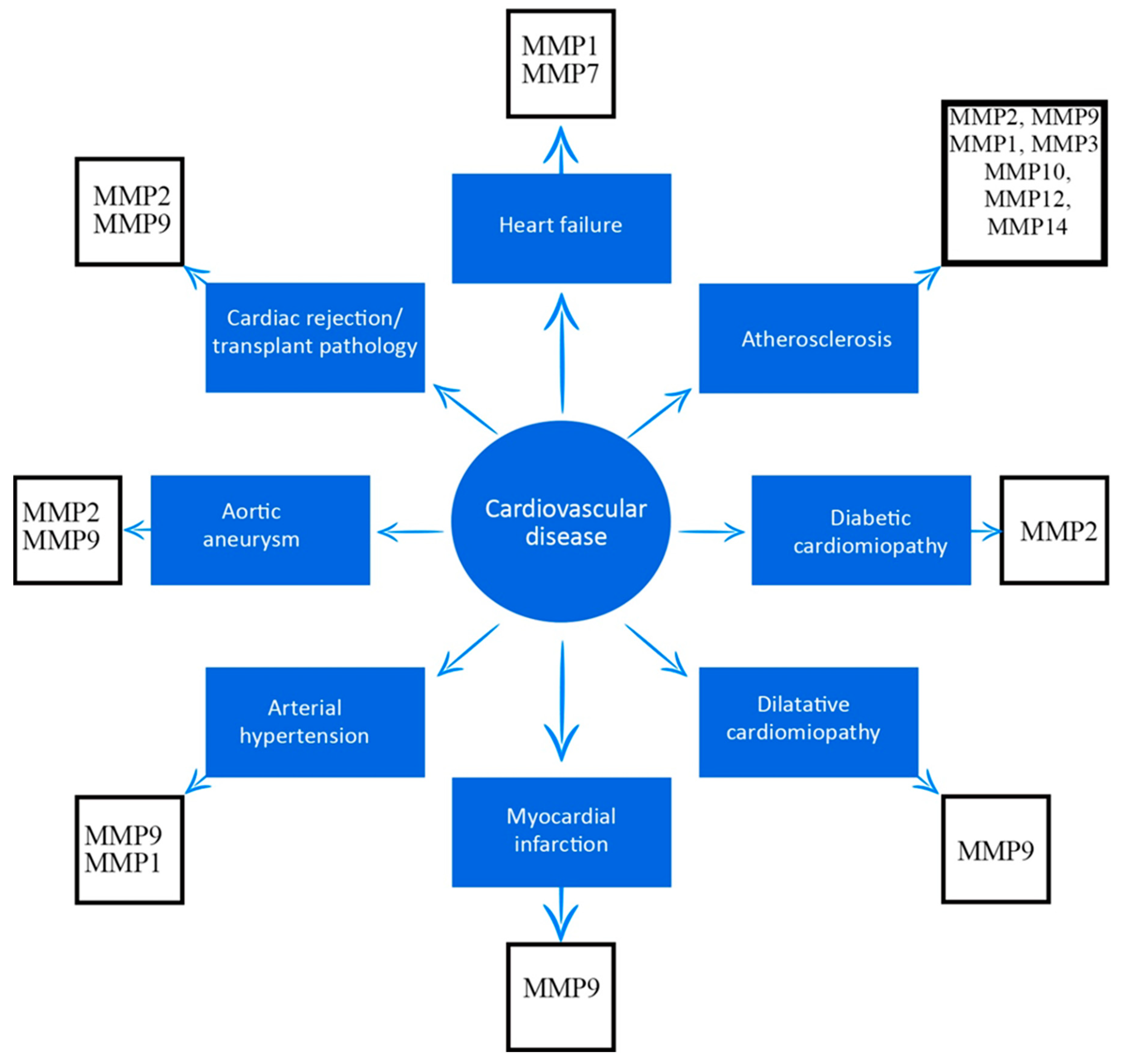
6. MMPs and Cardiovascular Disease
6.1. Atherosclerosis
6.2. Ischemia and Reperfusion Injuries
6.3. Hypertension
6.4. Myocardial Infarction
6.5. Aortic Aneurysm
6.6. Cardiac Rejection Pathology
7. Discussion
8. Future Directions
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Koomalsingh, K.; Kobashigawa, J.A. The future of cardiac transplantation. Ann. Cardiothorac. Surg. 2018, 7, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Kobashigawa, J.A. The future of heart transplantation. Am. J. Transpl. 2012, 12, 2875–2891. [Google Scholar] [CrossRef] [PubMed]
- Kobashigawa, J.A.; Lindenfeld, J.; Masri, S.C.; Miller, D.; O’Connell, J.; Rodriguez, E.R.; Rosengard, B.; Self, S.; White-Williams, C.; Zeevi, A. Antibody-mediated rejection in cardiac transplantation: Emerging knowledge in diagnosis and management. Circulation 2015, 131, 1608–1639. [Google Scholar]
- Suzuki, J.; Isobe, M.; Kawauki, M.; Amano, J. Altered expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases in acutely rejected myocardium and coronary arteriosclerosis in cardiac allografts of nonhuman primates. Transpl. Int. 2000, 13, 106–113. [Google Scholar] [CrossRef]
- Vanhoutte, D.; van Almen, G.C.; Van Aelst, L.N.; Van Cleemput, J.; Droogne, W.; Jin, Y.; Van de Werf, F.; Carmeliet, P.; Vanhaecke, J. Matricellular proteins and matrix metalloproteinases mark the inflammatory and fibrotic response in human cardiac allograft rejection. Eur. Heart J. 2013, 34, 1930–1941. [Google Scholar] [CrossRef]
- Liu, P.; Sun, M.; Sader, S. Matrix metalloproteinases in cardiovascular disease. Can. J. Cardiol. 2006, 22 (Suppl. B), 25B–30B. [Google Scholar] [CrossRef]
- Spinale, F.G.; Coker, M.L.; Heung, L.J.; Bond, B.R.; Gunasinghe, H.R.; Etoh, T.; Goldberg, A.T.; Zellner, J.L.; Crumbley, A.J. A matrix metalloproteinase induction/activation system exists in the human left ventricular myocardium and is upregulated in heart failure. Circulation 2000, 102, 1944–1949. [Google Scholar] [CrossRef]
- Doxakis, A.; Polyanthi, K.; Androniki, T.; Savvas, P.; Eleni, Z.; Roubini, L.; Nikolaos, R. Targeting metalloproteinases in cardiac remodelling. J. Cardiovasc. Med. Cardiol. 2019, 6, 51–60. [Google Scholar] [CrossRef]
- Campbell, L.G.; Ramachandran, S.; Liu, W.; Shipley, J.M.; Itohara, S.; Rogers, J.G.; Moazami, N.; Senior, R.M.; Jaramillo, A. Different roles for matrix metalloproteinase-2 and matrix metalloproteinase-9 in the pathogenesis of cardiac allograft rejection. Am. J. Transpl. 2005, 5, 517–528. [Google Scholar] [CrossRef]
- Raeeszadeh-Sarmazdeh, M.; Do, L.D.; Hritz, B.G. Metalloproteinases and Their Inhibitors: Potential for the Development of New Therapeutics. Cells 2020, 9, 1313. [Google Scholar] [CrossRef]
- Bassiouni, W.; Ali, M.A.M.; Schulz, R. Multifunctional intracellular matrix metalloproteinases: Implications in disease. FEBS J. 2021, 288, 7162–7182. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.P.; Hansch, C. Matrix metalloproteinases (MMPs): Chemical-biological functions and (Q)SARs. Bioorg. Med. Chem. 2007, 15, 2223–2268. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.H.; Edwards, D.R.; Murphy, G. Metalloproteinase inhibitors: Biological actions and therapeutic opportunities. J. Cell Sci. 2002, 115, 3719–3727. [Google Scholar] [CrossRef]
- Berg, G.; Barchuk, M.; Miksztowicz, V. Behavior of Metalloproteinases in Adipose Tissue, Liver and Arterial Wall: An Update of Extracellular Matrix Remodeling. Cells 2019, 8, 158. [Google Scholar] [CrossRef]
- Cui, N.; Hu, M.; Khalil, R.A. Biochemical and Biological Attributes of Matrix Metalloproteinases. Prog. Mol. Biol. Transl. Sci. 2017, 147, 1–73. [Google Scholar]
- Laronha, H.; Caldeira, J. Structure and Function of Human Matrix Metalloproteinases. Cells 2020, 9, 1076. [Google Scholar] [CrossRef] [PubMed]
- Berg, G.; Miksztowicz, V.; Schreier, L. Metalloproteinases in metabolic syndrome. Clin. Chim. Acta 2011, 412, 1731–1739. [Google Scholar] [CrossRef]
- Spinale, F.G. Matrix metalloproteinases: Regulation and dysregulation in the failing heart. Circ. Res. 2002, 90, 520–530. [Google Scholar] [CrossRef]
- Cabral-Pacheco, G.A.; Garza-Veloz, I.; Castruita-De la Rosa, C.; Ramirez-Acuña, J.M.; Perez-Romero, B.A.; Guerrero-Rodriguez, J.F.; Martinez-Avila, N.; Martinez-Fierro, M.L. The Roles of Matrix Metalloproteinases and Their Inhibitors in Human Diseases. Int. J. Mol. Sci. 2020, 21, 9739. [Google Scholar] [CrossRef]
- Wang, X.; Khalil, R.A. Matrix Metalloproteinases, Vascular Remodeling, and Vascular Disease. Adv. Pharmacol. 2018, 81, 241–330. [Google Scholar] [CrossRef]
- Kapoor, C.; Vaidya, S.; Wadhwan, V.; Kaur, G.; Pathak, A. Seesaw of matrix metalloproteinases (MMPs). J. Cancer Res. Ther. 2016, 12, 28–35. [Google Scholar] [CrossRef] [PubMed]
- DeLeon-Pennell, K.Y.; Meschiari, C.A.; Jung, M.; Lindsey, M.L. Matrix Metalloproteinases in Myocardial Infarction and Heart Failure. Prog. Mol. Biol. Transl. Sci. 2017, 147, 75–100. [Google Scholar] [CrossRef] [PubMed]
- Shadrina, A.S.; Plieva, Y.Z.; Kushlinskiy, D.N.; Morozov, A.A.; Filipenko, M.L.; Chang, V.L.; Kushlinskii, N.E. Classification, regulation of activity, and genetic polymorphism of matrix metalloproteinases in health and disease. Alm. Clin. Med. 2017, 45, 266–279. [Google Scholar] [CrossRef]
- Baghirova, S.; Hughes, B.G.; Poirier, M.; Kondo, M.Y.; Schulz, R. Nuclear matrix metalloproteinase-2 in the cardiomyocyte and the ischemic-reperfused heart. J. Mol. Cell Cardiol. 2016, 94, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Lovett, D.H.; Mahimkar, R.; Raffai, R.L.; Cape, L.; Maklashina, E.; Cecchini, G.; Karliner, J.S. A novel intracellular isoform of matrix metalloproteinase-2 induced by oxidative stress activates innate immunity. PLoS ONE 2012, 7, e34177. [Google Scholar] [CrossRef]
- Lindner, D.; Zietsch, C.; Becher, P.M.; Schulze, K.; Schultheiss, H.P.; Tschöpe, C.; Westermann, D. Differential expression of matrix metalloproteases in human fibroblasts with different origins. Biochem. Res. Int. 2012, 2012, 875742. [Google Scholar] [CrossRef]
- Kelly, D.; Khan, S.; Cockerill, G.; Ng, L.L.; Thompson, M.; Samani, N.J.; Squire, I.B. Circulating stromelysin-1 (MMP-3): A novel predictor of LV dysfunction, remodelling and all-cause mortality after acute myocardial infarction. Eur. J. Heart Fail. 2008, 10, 133–139. [Google Scholar] [CrossRef]
- Suzuki, K.; Enghild, J.J.; Morodomi, T.; Salvesen, G.; Nagase, H. Mechanisms of activation of tissue procollagenase by matrix metalloproteinase 3 (stromelysin). Biochemistry 1990, 29, 10261–10270. [Google Scholar] [CrossRef]
- von Bredow, D.C.; Cress, A.E.; Howard, E.W.; Bowden, G.T.; Nagle, R.B. Activation of gelatinase-tissue-inhibitors-of-metalloproteinase complexes by matrilysin. Biochem. J. 1998, 331, 965–972. [Google Scholar] [CrossRef]
- Chiao, Y.A.; Zamilpa, R.; Lopez, E.F.; Dai, Q.; Escobar, G.P.; Hakala, K.; Weintraub, S.T.; Lindsey, M.L. In vivo matrix metalloproteinase-7 substrates identified in the left ventricle post-myocardial infarction using proteomics. J. Proteome Res. 2010, 9, 2649–2657. [Google Scholar] [CrossRef]
- Lindsey, M.L.; Escobar, G.P.; Mukherjee, R.; Goshorn, D.K.; Sheats, N.J.; Bruce, J.A.; Mains, I.M.; Hendrick, J.K.; Hewett, K.W.; Gourdie, R.G.; et al. Matrix metalloproteinase-7 affects connexin-43 levels, electrical conduction, and survival after myocardial infarction. Circulation 2006, 113, 2919–2928. [Google Scholar] [CrossRef] [PubMed]
- Webb, C.S.; Bonnema, D.D.; Ahmed, S.H.; Leonardi, A.H.; McClure, C.D.; Clark, L.L.; Stroud, R.E.; Corn, W.C.; Finklea, L.; Zile, M.R.; et al. Specific temporal profile of matrix metalloproteinase release occurs in patients after myocardial infarction: Relation to left ventricular remodeling. Circulation 2006, 114, 1020–1027. [Google Scholar] [CrossRef]
- Fertin, M.; Lemesle, G.; Turkieh, A.; Beseme, O.; Chwastyniak, M.; Amouyel, P.; Bauters, C.; Pinet, F. Serum MMP-8: A novel indicator of left ventricular remodeling and cardiac outcome in patients after acute myocardial infarction. PLoS ONE 2013, 8, e71280. [Google Scholar] [CrossRef] [PubMed]
- Spinale, F.G.; Coker, M.L.; Bond, B.R.; Zellner, J.L. Myocardial matrix degradation and metalloproteinase activation in the failing heart: A potential therapeutic target. Cardiovasc. Res. 2000, 46, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Romanic, A.M.; Harrison, S.M.; Bao, W.; Burns-Kurtis, C.L.; Pickering, S.; Gu, J.; Grau, E.; Mao, J.; Sathe, G.M.; Ohlstein, E.H.; et al. Myocardial protection from ischemia/reperfusion injury by targeted deletion of matrix metalloproteinase-9. Cardiovasc. Res. 2002, 54, 549–558. [Google Scholar] [CrossRef]
- Löffek, S.; Schilling, O.; Franzke, C.W. Series “matrix metalloproteinases in lung health and disease”: Biological role of matrix metalloproteinases: A critical balance. Eur. Respir. J. 2011, 38, 191–208. [Google Scholar] [CrossRef]
- Sivaraman, S.K.; Zachariah, G.; Annamala, P. Effect of Smoking on Metalloproteinases (MMPs) Activity in Patients with Acute Myocardial Infarction (AMI). J. Clin. Diagn. Res. 2014, 8, 27–30. [Google Scholar] [CrossRef]
- Vandenbroucke, R.E.; Libert, C. Is there new hope for therapeutic matrix metalloproteinase inhibition? Nat. Rev. Drug Discov. 2014, 13, 904–927. [Google Scholar] [CrossRef]
- Brew, K.; Nagase, H. The tissue inhibitors of metalloproteinases (TIMPs): An ancient family with structural and functional diversity. Biochim. Biophys. Acta 2010, 1803, 55–71. [Google Scholar] [CrossRef]
- Singh, D.; Srivastava, S.K.; Chaudhuri, T.K.; Upadhyay, G. Multifaceted role of matrix metalloproteinases (MMPs). Front. Mol. Biosci. 2015, 2, 19. [Google Scholar] [CrossRef]
- Trojanek, J.B. Role of matrix metalloproteinases and tissue inhibitors of metalloproteinases in hypertension. Pathogenesis of hypertension and obesity. Postepy Biochem. 2015, 61, 356–363. [Google Scholar] [PubMed]
- Amălinei, C.; Căruntu, I.D.; Giuşcă, S.E.; Bălan, R.A. Matrix metalloproteinases involvement in pathologic conditions. Rom. J. Morphol. Embryol. 2010, 51, 215–228. [Google Scholar]
- Lee, J.K.; Zaidi, S.H.; Liu, P.; Dawood, F.; Cheah, A.Y.; Wen, W.H.; Saiki, Y.; Rabinovitch, M. A serine elastase inhibitor reduces inflammation and fibrosis and preserves cardiac function after experimentally-induced murine myocarditis. Nat. Med. 1998, 4, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Provenzano, M.; Andreucci, M.; Garofalo, C.; Faga, T.; Michael, A.; Ielapi, N.; Grande, R.; Sapienza, P.; Franciscis, S.; Mastroroberto, P.; et al. The Association of Matrix Metalloproteinases with Chronic Kidney Disease and Peripheral Vascular Disease: A Light at the End of the Tunnel? Biomolecules 2020, 10, 154. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.; Pushpakumar, S.; Muradashvili, N.; Kundu, S.; Tyagi, S.C.; Sen, U. Regulation and involvement of matrix metalloproteinases in vascular diseases. Front. Biosci. Landmark 2016, 21, 89–118. [Google Scholar]
- Peeters, S.A.; Engelen, L.; Buijs, J.; Chaturvedi, N.; Fuller, J.H.; Jorsal, A.; Parving, H.H.; Tarnow, L.; Theilade, S.; Rossing, P.; et al. Circulating matrix metalloproteinases are associated with arterial stiffness in patients with type 1 diabetes: Pooled analysis of three cohort studies. Cardiovasc. Diabetol. 2017, 16, 139. [Google Scholar] [CrossRef]
- Hansson, J.; Lind, L.; Hulthe, J.; Sundström, J. Relations of serum MMP-9 and TIMP-1 levels to left ventricular measures and cardiovascular risk factors: A population-based study. Eur. J. Cardiovasc. Prev. Rehabil. 2009, 16, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Giannakos, E.; Vardali, E.; Bartekova, M.; Fogarassyova, M.; Barancik, M.; Radosinska, J. Changes in activities of circulating MMP-2 and MMP-9 in patients suffering from heart failure in relation to gender, hypertension and treatment: A cross-sectional study. Physiol. Res. 2016, 65 (Suppl. S1), S149–S152. [Google Scholar] [CrossRef] [PubMed]
- Bergman, M.R.; Teerlink, J.R.; Mahimkar, R.; Li, L.; Zhu, B.Q.; Nguyen, A.; Dahi, S.; Karliner, J.S.; Lovett, D.H. Cardiac matrix metalloproteinase-2 expression independently induces marked ventricular remodeling and systolic dysfunction. Am. J. Physiol. Heart Circ. Physiol. 2007, 292, H1847–H1860. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.Y.; Bergman, M.R.; Nguyen, A.P.; Turcato, S.; Swigart, P.M.; Rodrigo, M.C.; Simpson, P.C.; Karliner, J.S.; Lovett, D.H.; Baker, A.J. Cardiac transgenic matrix metalloproteinase-2 expression directly induces impaired contractility. Cardiovasc. Res. 2006, 69, 688–696. [Google Scholar] [CrossRef]
- Barhoumi, T.; Fraulob-Aquino, J.C.; Mian, M.O.R.; Ouerd, S.; Idris-Khodja, N.; Huo, K.G.; Rehman, A.; Caillon, A.; Dancose-Giambattisto, B.; Ebrahimian, T.; et al. Matrix metalloproteinase-2 knockout prevents angiotensin II-induced vascular injury. Cardiovasc. Res. 2017, 113, 1753–1762. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.W.; Lee, S.J.; Lee, M.Y.; Park, M.W.; Kim, S.S.; Shin, N.; Lovett, D.H.; Bae, S.S.; Ahn, J.; Park, J.S.; et al. Enhanced cardiac expression of two isoforms of matrix metalloproteinase-2 in experimental diabetes mellitus. PLoS ONE 2019, 14, e0221798. [Google Scholar] [CrossRef] [PubMed]
- Yaras, N.; Sariahmetoglu, M.; Bilginoglu, A.; Aydemir-Koksoy, A.; Onay-Besikci, A.; Turan, B.; Schulz, R. Protective action of doxycycline against diabetic cardiomyopathy in rats. Br. J. Pharmacol. 2008, 155, 1174–1184. [Google Scholar] [CrossRef]
- de Castro Brás, L.E.; Cates, C.A.; DeLeon-Pennell, K.Y.; Ma, Y.; Iyer, R.P.; Halade, G.V.; Yabluchanskiy, A.; Fields, G.B.; Weintraub, S.T.; Lindsey, M.L. Citrate synthase is a novel in vivo matrix metalloproteinase-9 substrate that regulates mitochondrial function in the postmyocardial infarction left ventricle. Antioxid. Redox Signal. 2014, 21, 1974–1985. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Zheng, Y.J.; Gu, S.S.; Tan, J.L.; Paul, C.; Wang, Y.G.; Yang, H.T. Degradation of cardiac myosin light chain kinase by matrix metalloproteinase-2 contributes to myocardial contractile dysfunction during ischemia/reperfusion. J. Mol. Cell Cardiol. 2014, 77, 102–112. [Google Scholar] [CrossRef]
- Fallata, A.M.; Wyatt, R.A.; Levesque, J.M.; Dufour, A.; Overall, C.M.; Crawford, B.D. Intracellular Localization in Zebrafish Muscle and Conserved Sequence Features Suggest Roles for Gelatinase A Moonlighting in Sarcomere Maintenance. Biomedicines 2019, 7, 93. [Google Scholar] [CrossRef]
- Tan, J.; Hua, Q.; Xing, X.; Wen, J.; Liu, R.; Yang, Z. Impact of the metalloproteinase-9/tissue inhibitor of metalloproteinase-1 system on large arterial stiffness in patients with essential hypertension. Hypertens. Res. 2007, 30, 959–963. [Google Scholar] [CrossRef]
- Lehoux, S.; Lemarié, C.A.; Esposito, B.; Lijnen, H.R.; Tedgui, A. Pressure-induced matrix metalloproteinase-9 contributes to early hypertensive remodeling. Circulation 2004, 109, 1041–1047. [Google Scholar] [CrossRef]
- Dale, M.A.; Suh, M.K.; Zhao, S.; Meisinger, T.; Gu, L.; Swier, V.J.; Agrawal, D.K.; Greiner, T.C.; Carson, J.S.; Baxter, B.T.; et al. Background differences in baseline and stimulated MMP levels influence abdominal aortic aneurysm susceptibility. Atherosclerosis 2015, 243, 621–629. [Google Scholar] [CrossRef]
- Zile, M.R.; Desantis, S.M.; Baicu, C.F.; Stroud, R.E.; Thompson, S.B.; McClure, C.D.; Mehurg, S.M.; Spinale, F.G. Plasma biomarkers that reflect determinants of matrix composition identify the presence of left ventricular hypertrophy and diastolic heart failure. Circ. Heart Fail. 2011, 4, 246–256. [Google Scholar] [CrossRef]
- Eaton, V.L.; Lerret, N.M.; Velásquez-Lopera, M.M.; John, R.; Caicedo, M.; DeCresce, R.P.; Jaramillo, A. Enhanced allograft survival and modulation of T-cell alloreactivity induced by inhibition of MMP/ADAM enzymatic activity. Am. J. Transplant. 2008, 8, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Page-McCaw, A.; Ewald, A.J.; Werb, Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat. Rev. Mol. Cell Biol. 2007, 8, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Chancey, A.L.; Brower, G.L.; Peterson, J.T.; Janicki, J.S. Effects of matrix metalloproteinase inhibition on ventricular remodeling due to volume overload. Circulation 2002, 105, 1983–1988. [Google Scholar] [CrossRef] [PubMed]
- Aharinejad, S.; Krenn, K.; Zuckermann, A.; Schäfer, R.; Gmeiner, M.; Thomas, A.; Aliabadi, A.; Schneider, B.; Grimm, M. Serum matrix metalloprotease-1 and vascular endothelial growth factor—A predict cardiac allograft rejection. Am. J. Transpl. 2009, 9, 149–159. [Google Scholar] [CrossRef]
- Vanhoutte, D.; Heymans, S. TIMPs and cardiac remodeling: ‘Embracing the MMP-independent-side of the family’. J. Mol. Cell Cardiol. 2010, 48, 445–453. [Google Scholar] [CrossRef]
- Booth, A.J.; Bishop, D.K. TGF-beta, IL-6, IL-17 and CTGF direct multiple pathologies of chronic cardiac allograft rejection. Immunotherapy 2010, 2, 511–520. [Google Scholar] [CrossRef]
- Fahmy, N.M.; Yamani, M.H.; Starling, R.C.; Ratliff, N.B.; Young, J.B.; McCarthy, P.M.; Feng, J.; Novick, A.C.; Fairchild, R.L. Chemokine and chemokine receptor gene expression indicates acute rejection of human cardiac transplants. Transplantation 2003, 75, 72–78. [Google Scholar] [CrossRef]
- Frangogiannis, N.G. Matricellular proteins in cardiac adaptation and disease. Physiol. Rev. 2012, 92, 635–688. [Google Scholar] [CrossRef]
- Cheung, P.Y.; Sawicki, G.; Wozniak, M.; Wang, W.; Radomski, M.W.; Schulz, R. Matrix metalloproteinase-2 contributes to ischemia-reperfusion injury in the heart. Circulation 2000, 101, 1833–1839. [Google Scholar] [CrossRef]
- Lalu, M.M.; Pasini, E.; Schulze, C.J.; Ferrari-Vivaldi, M.; Ferrari-Vivaldi, G.; Bachetti, T.; Schulz, R. Ischaemia-reperfusion injury activates matrix metalloproteinases in the human heart. Eur. Heart J. 2005, 26, 27–35. [Google Scholar] [CrossRef]
- Yamani, M.H.; Starling, R.C.; Young, J.B.; Cook, D.; Yu, Y.; Vince, D.G.; McCarthy, P.; Ratliff, N.B. Acute vascular rejection is associated with up-regulation of vitronectin receptor (alphavbeta3), increased expression of tissue factor, and activation of the extracellular matrix metalloproteinase induction system. J. Heart Lung Transpl. 2002, 21, 983–989. [Google Scholar] [CrossRef] [PubMed]
- Goetzl, E.J.; Banda, M.J.; Leppert, D. Matrix metalloproteinases in immunity. J. Immunol. 1996, 156, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Aharinejad, S.; Krenn, K.; Zuckermann, A.; Schäfer, R.; Paulus, P.; Seebacher, G.; Wolner, E.; Grimm, M. Matrix metalloproteases and their tissue inhibitor in cardiac transplantation. Eur. J. Cardiothorac. Surg. 2007, 32, 48–51. [Google Scholar] [CrossRef] [PubMed]
- Tsukioka, K.; Suzuki, J.; Kawauchi, M.; Wada, Y.; Zhang, T.; Nishio, A.; Koide, N.; Endoh, M.; Takayama, K.; Takamoto, S.; et al. Expression of membrane-type 1 matrix metalloproteinase in coronary vessels of allotransplanted primate hearts. J. Heart Lung Transpl. 2000, 19, 1193–1198. [Google Scholar] [CrossRef] [PubMed]
- Squire, I.B.; Evans, J.; Ng, L.L.; Loftus, I.M.; Thompson, M.M. Plasma mmp-9 and mmp-2 following acute myocardial infarction in man: Correlation with echocardiographic and neurohumoral parameters of left ventricular dysfunction. J. Card. Fail. 2004, 10, 328–333. [Google Scholar] [CrossRef]
- Peterson, J.T.; Hallak, H.; Johnson, L.; Li, H.; O’Brien, P.M.; Sliskovic, D.R.; Bocan, T.M.; Coker, M.L.; Etoh, T.; Spinale, F.G. Matrix metalloproteinase inhibition attenuates left ventricular remodeling and dysfunction in a rat model of progressive heart failure. Circulation 2001, 103, 2303–2309. [Google Scholar] [CrossRef]
- Wang, W.; Schulze, C.J.; Suarez-Pinzon, W.L.; Dyck, J.R.; Sawicki, G.; Schulz, R. Intracellular action of matrix metalloproteinase-2 accounts for acute myocardial ischemia and reperfusion injury. Circulation 2002, 106, 1543–1549. [Google Scholar] [CrossRef]
- Skrzypiec-Spring, M.; Urbaniak, J.; Sapa-Wojciechowska, A.; Pietkiewicz, J.; Orda, A.; Karolko, B.; Danielewicz, R.; Bil-Lula, I.; Woźniak, M.; Schulz, R.; et al. Matrix Metalloproteinase-2 Inhibition in Acute Ischemia-Reperfusion Heart Injury-Cardioprotective Properties of Carvedilol. Pharmaceuticals 2021, 14, 1276. [Google Scholar] [CrossRef]
- Falk, V.; Soccal, P.M.; Grünenfelder, J.; Hoyt, G.; Walther, T.; Robbins, R.C. Regulation of matrix metalloproteinases and effect of MMP-inhibition in heart transplant related reperfusion injury. Eur. J. Cardiothorac. Surg. 2002, 22, 53–58. [Google Scholar] [CrossRef]
- Joffs, C.; Gunasinghe, H.R.; Multani, M.M.; Dorman, B.H.; Kratz, J.M.; Crumbley, A.J., 3rd; Crawford, F.A., Jr.; Spinale, F.G. Cardiopulmonary bypass induces the synthesis and release of matrix metalloproteinases. Ann. Thorac. Surg. 2001, 71, 1518–1523. [Google Scholar] [CrossRef]
- Lin, T.C.; Li, C.Y.; Tsai, C.S.; Ku, C.H.; Wu, C.T.; Wong, C.S.; Ho, S.T. Neutrophil-mediated secretion and activation of matrix metalloproteinase-9 during cardiac surgery with cardiopulmonary bypass. Anesth. Analg. 2005, 100, 1554–1560. [Google Scholar] [CrossRef] [PubMed]
| Enzyme Name | MMP Classification (Number) | Common Description/Substrate | Cell | Cell Localization | Functional Role | Inhibition | References |
|---|---|---|---|---|---|---|---|
| Collagenase | MMP1, MMP8, MMP13, MMP18 | Collagen II, III, VIII, X, gelatins, aggrecan, entactin | Myocytes, macrophages, fibroblasts, megakaryocytes, cardiomyocytes, neutrophils | Cytosol (MMP18) Nucleus (MMP1) | Myocardial fibrosis Inflammation | TIMP-1 | [1] [11] [6] [18] |
| Gelatinase | MMP2 | Gelatins Collagen I, IV, V, VII Basal membrane component | Cardiomyocytes Smooth muscle Platelet Megakaryocytes Fibroblasts | Sarcoplasmic reticulum Nucleus cytosol Mitochondria Sarcomere | Cardiomyocytes death Heart failure Increase angiogenesis, inhibition of myocardial fibrosis | TIMP 1-4 | [18] [11] |
| MMP9 | Gelatins Collagen IV, V, XIV Basal membrane component | Leucocytes Megakaryocytes Cardiomyocytes | Cytosol Nucleus Sarcomere Mitochondria | Induce cardiac disfunction Increase migration rate of cardiac fibroblasts | TIMP1-3 | [11] [20] | |
| Stromelysines | MMP3, MMP10, MMP11 | Collagen III, IV, V, IX Fibronectin Laminin | Megakaryocytes Cardiomyocytes | Cytosol nucleus | Cardiomyocyte death Increase heart failure Increase migration rate of cardiac fibroblasts | TIMP1-3 | [6] [19] |
| Matrilysines | MMP7 MMP26 | Fibronectin Collagen IV gelatins | Cardiomyocytes Neuronal cells | Cytosol | Increase migration rate of cardiac fibroblasts Inflammation | TIMP4 TIMP1 | [11] [18] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patrichi, G.; Patrichi, A.; Satala, C.-B.; Sin, A.I. Matrix Metalloproteinases and Heart Transplantation—A Pathophysiological and Clinical View. Medicina 2023, 59, 1295. https://doi.org/10.3390/medicina59071295
Patrichi G, Patrichi A, Satala C-B, Sin AI. Matrix Metalloproteinases and Heart Transplantation—A Pathophysiological and Clinical View. Medicina. 2023; 59(7):1295. https://doi.org/10.3390/medicina59071295
Chicago/Turabian StylePatrichi, Gabriela, Andrei Patrichi, Catalin-Bogdan Satala, and Anca Ileana Sin. 2023. "Matrix Metalloproteinases and Heart Transplantation—A Pathophysiological and Clinical View" Medicina 59, no. 7: 1295. https://doi.org/10.3390/medicina59071295
APA StylePatrichi, G., Patrichi, A., Satala, C.-B., & Sin, A. I. (2023). Matrix Metalloproteinases and Heart Transplantation—A Pathophysiological and Clinical View. Medicina, 59(7), 1295. https://doi.org/10.3390/medicina59071295







