Evaluating the Diagnostic Value of Electrovestibulography (EVestG) in Alzheimer’s Patients with Mixed Pathology: A Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
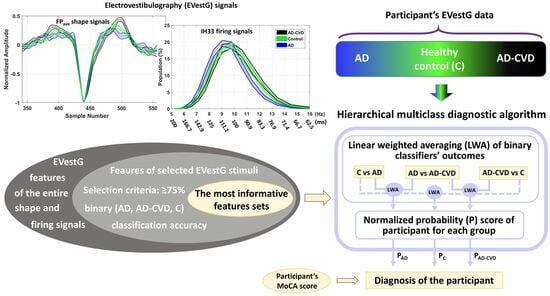
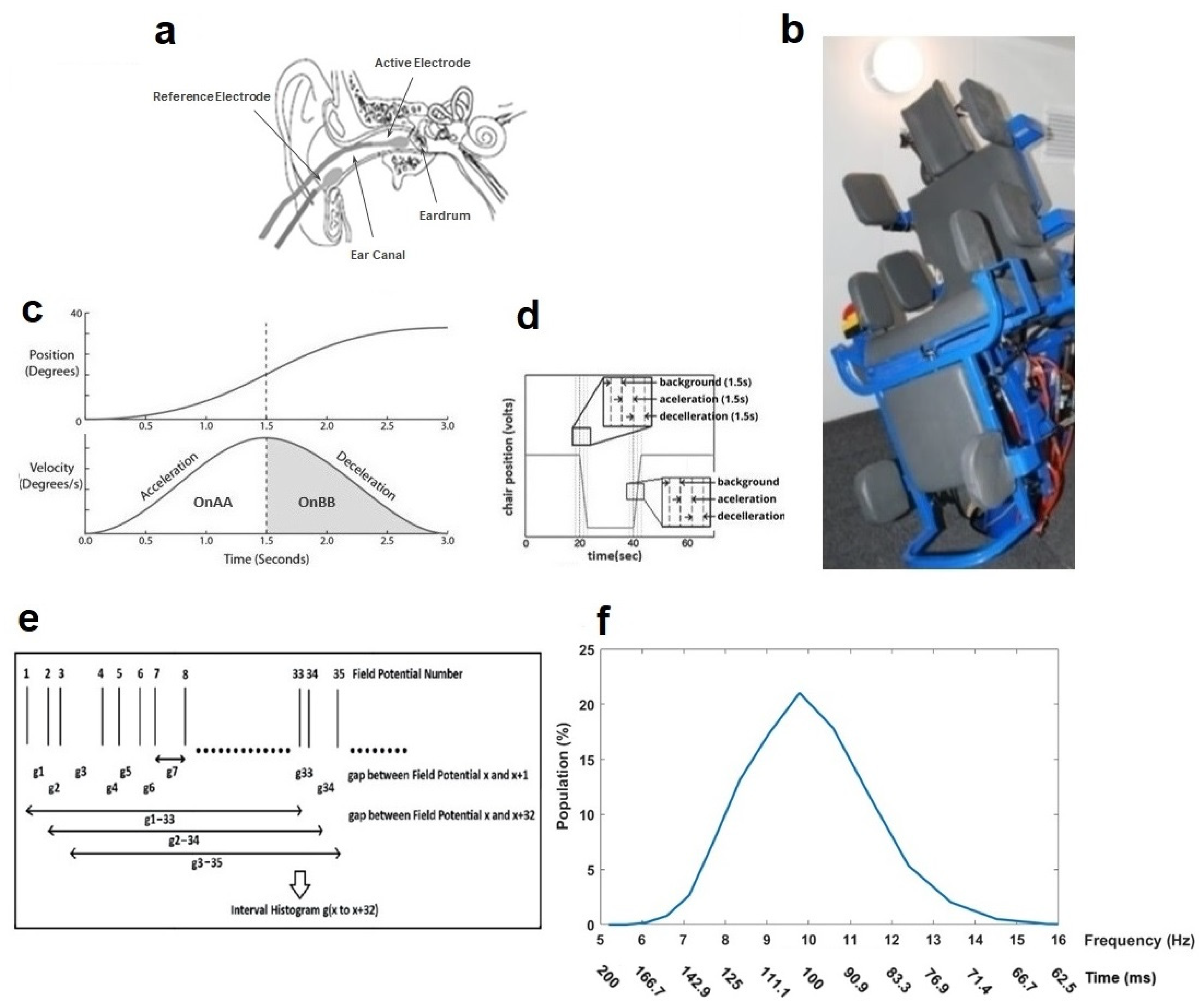
2.2. Electrovestibulography (EVestG)
2.3. Signal Analysis
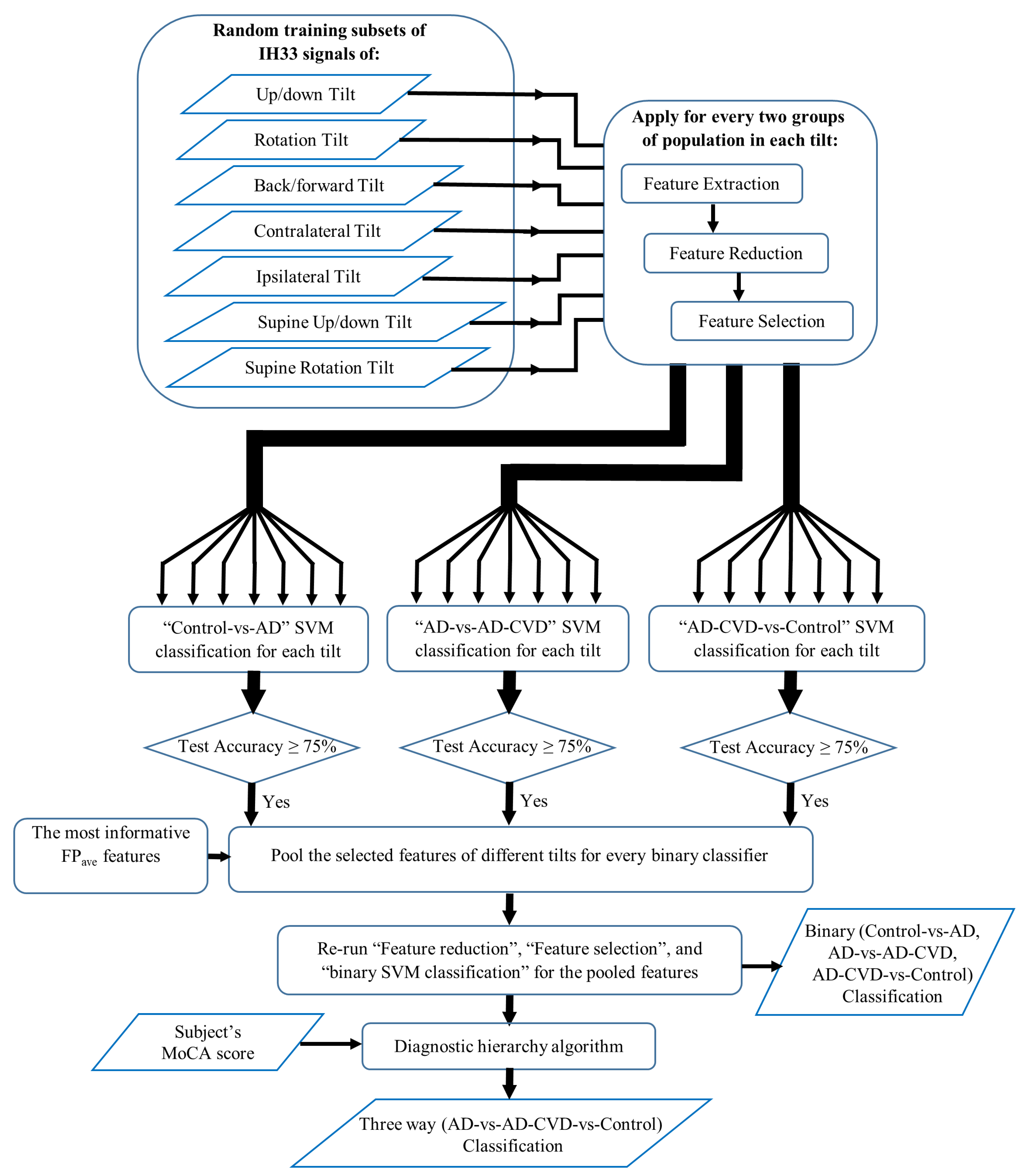
2.4. Feature Extraction
2.5. Feature Reduction and Selection
2.6. Binary Classification
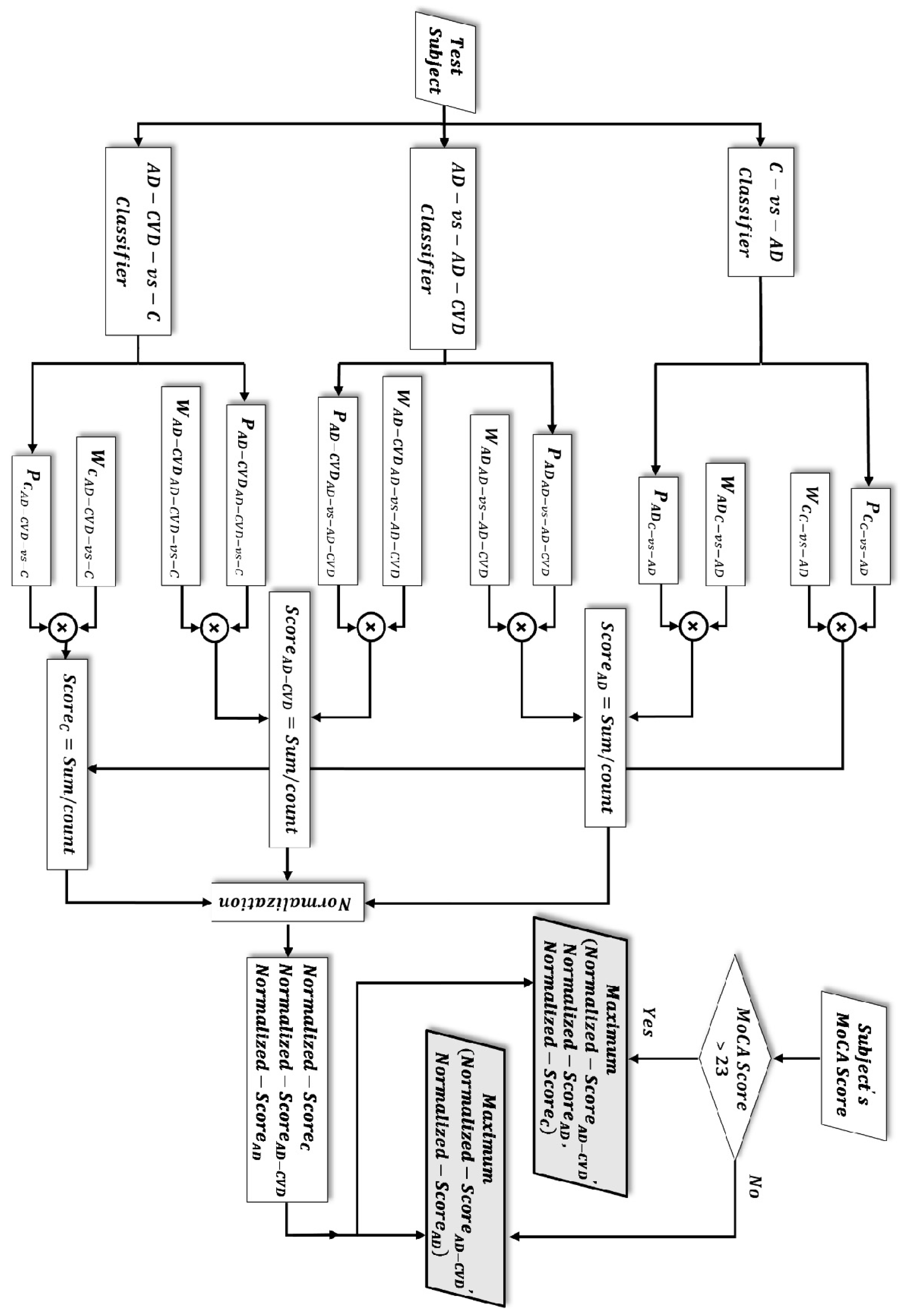
2.7. Diagnostic Hierarchy Algorithm
2.8. Statistical Analysis
3. Results
Statistical Analysis
4. Discussion
5. Conclusions
6. Limitations and the Future of the Study
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Aβ | Amyloid-β |
| AβO | Amyloid-β oligomers |
| Acc | Accuracy |
| AD | Alzheimer’s disease |
| AD-CVD | AD mixed with levels of cerebrovascular disease symptomology |
| AUC | Area under the curve |
| BGi | Background segment |
| C | Control |
| CT | Contralateral tilt |
| CVD | Cerebrovascular disease |
| EVestG | Electrovestibulography |
| FP | Field potential |
| FPave | Average of spontaneous and driven vestibular field potentials |
| GABA | Gamma-aminobutyric acid |
| HIS | Hachinski ischemic score |
| IH | Interval histogram |
| IH33 | 33-Interval histogram |
| IT | Ipsilateral tilt |
| L | left |
| LC | Locus Coeruleus |
| LR | Summation of left and right signals |
| L-R | Subtraction of left and right signals |
| µ | Mean |
| MADRS | Montgomery–Asberg Depression Rating Scale |
| MANCOVA | Multivariate analysis of covariance |
| MoCA | Montreal Cognitive Assessment |
| MRI | Magnetic resonance imaging |
| NEER | Neural Event Extraction Routine |
| NINDS-AIREN | National Institute of Neurological Disorders and Stroke–Association Internationale pour la Recherche et l’Enseignement en Neurosciences |
| OnAA | Acceleration segment |
| OnBB | Deceleration segment |
| R | right |
| ROC | Receiver operating characteristic |
| RTC | Return to center |
| SD | Standard deviation |
| Sens | Sensitivity |
| Spec | Specificity |
| SVM | Supervised support vector machine |
| VaD | Vascular dementia |
| VN | Vestibular nucleus |
References
- Elahi, F.M.; Miller, B.L. A Clinicopathological Approach to the Diagnosis of Dementia. Nat. Rev. Neurol. 2017, 13, 457–477. [Google Scholar] [CrossRef] [PubMed]
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Jack, C.R.; Kawas, C.H.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, R.; et al. The Diagnosis of Dementia Due to Alzheimer’s Disease: Recommendations from the National Institute on Aging-Alzheimer’s Association Workgroups on Diagnostic Guidelines for Alzheimer’s Disease. Alzheimers Dement. 2011, 7, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Rizzi, L.; Rosset, I.; Roriz-Cruz, M. Global Epidemiology of Dementia: Alzheimer’s and Vascular Types. Available online: https://www.hindawi.com/journals/bmri/2014/908915/ (accessed on 30 October 2020).
- Yousufuddin, M.; Young, N. Aging and Ischemic Stroke. Aging 2019, 11, 2542–2544. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, K.A. Pathobiological Subtypes of Alzheimer Disease. Dement. Geriatr. Cogn. Disord. 2020, 49, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Román, G.C.; Tatemichi, T.K.; Erkinjuntti, T.; Cummings, J.L.; Masdeu, J.C.; Garcia, J.H.; Amaducci, L.; Orgogozo, J.M.; Brun, A.; Hofman, A. Vascular Dementia: Diagnostic Criteria for Research Studies. Report of the NINDS-AIREN International Workshop. Neurology 1993, 43, 250–260. [Google Scholar] [CrossRef]
- Hachinski, V.C.; Iliff, L.D.; Zilhka, E.; Du Boulay, G.H.; McAllister, V.L.; Marshall, J.; Russell, R.W.; Symon, L. Cerebral Blood Flow in Dementia. Arch. Neurol. 1975, 32, 632–637. [Google Scholar] [CrossRef]
- Beach, T.G.; Monsell, S.E.; Phillips, L.E.; Kukull, W. Accuracy of the Clinical Diagnosis of Alzheimer Disease at National Institute on Aging Alzheimer Disease Centers, 2005–2010. J. Neuropathol. Exp. Neurol. 2012, 71, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Heyman, A.; Fillenbaum, G.G.; Welsh-Bohmer, K.A.; Gearing, M.; Mirra, S.S.; Mohs, R.C.; Peterson, B.L.; Pieper, C.F. Cerebral Infarcts in Patients with Autopsy-Proven Alzheimer’s Disease: CERAD, Part XVIII. Neurology 1998, 51, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Zea-Sevilla, M.A.; Fernández-Blázquez, M.A.; Calero, M.; Bermejo-Velasco, P.; Rábano, A. Combined Alzheimer’s Disease and Cerebrovascular Staging Explains Advanced Dementia Cognition. Alzheimer’s Dement. 2015, 11, 1358–1366. [Google Scholar] [CrossRef]
- Jack, C.R., Jr.; Wiste, H.J.; Vemuri, P.; Weigand, S.D.; Senjem, M.L.; Zeng, G.; Bernstein, M.A.; Gunter, J.L.; Pankratz, V.S.; Aisen, P.S.; et al. Brain Beta-Amyloid Measures and Magnetic Resonance Imaging Atrophy Both Predict Time-to-Progression from Mild Cognitive Impairment to Alzheimer’s Disease. Brain 2010, 133, 3336–3348. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, K.A. Understanding the Pathology of Vascular Cognitive Impairment. J. Neurol. Sci. 2005, 229–230, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Zekry, D.; Duyckaerts, C.; Moulias, R.; Belmin, J.; Geoffre, C.; Herrmann, F.; Hauw, J.-J. Degenerative and Vascular Lesions of the Brain Have Synergistic Effects in Dementia of the Elderly. Acta Neuropathol. 2002, 103, 481–487. [Google Scholar] [CrossRef]
- Vemuri, P.; Knopman, D.S. The Role of Cerebrovascular Disease When There Is Concomitant Alzheimer Disease. Biochim. Et. Biophys. Acta BBA—Mol. Basis Dis. 2016, 1862, 952–956. [Google Scholar] [CrossRef] [PubMed]
- Dubois, B.; Feldman, H.H.; Jacova, C.; DeKosky, S.T.; Barberger-Gateau, P.; Cummings, J.; Delacourte, A.; Galasko, D.; Gauthier, S.; Jicha, G.; et al. Research Criteria for the Diagnosis of Alzheimer’s Disease: Revising the NINCDS–ADRDA Criteria. Lancet Neurol. 2007, 6, 734–746. [Google Scholar] [CrossRef] [PubMed]
- Custodio, N.; Montesinos, R.; Lira, D.; Herrera-Pérez, E.; Bardales, Y.; Valeriano-Lorenzo, L. Mixed Dementia: A Review of the Evidence. Dement. Neuropsychol. 2017, 11, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Moroney, J.T.; Bagiella, E.; Desmond, D.W.; Hachinski, V.C.; Mölsä, P.K.; Gustafson, L.; Brun, A.; Fischer, P.; Erkinjuntti, T.; Rosen, W.; et al. Meta-Analysis of the Hachinski Ischemic Score in Pathologically Verified Dementias. Neurology 1997, 49, 1096–1105. [Google Scholar] [CrossRef]
- Molsa, P.K.; Paljarvi, L.; Rinne, J.O.; Rinne, U.K.; Sako, E. Validity of Clinical Diagnosis in Dementia: A Prospective Clinicopathological Study. J. Neurol. Neurosurg. Psychiatry 1985, 48, 1085–1090. [Google Scholar] [CrossRef]
- Wilczyńska, K.; Waszkiewicz, N. Diagnostic Utility of Selected Serum Dementia Biomarkers: Amyloid β-40, Amyloid β-42, Tau Protein, and YKL-40: A Review. J. Clin. Med. 2020, 9, 3452. [Google Scholar] [CrossRef]
- Krishnan, S.; Rani, P. Evaluation of Selenium, Redox Status and Their Association with Plasma Amyloid/Tau in Alzheimer’s Disease. Biol. Trace Elem. Res. 2014, 158, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Bibl, M.; Esselmann, H.; Mollenhauer, B.; Weniger, G.; Welge, V.; Liess, M.; Lewczuk, P.; Otto, M.; Schulz, J.B.; Trenkwalder, C.; et al. Blood-Based Neurochemical Diagnosis of Vascular Dementia: A Pilot Study. J. Neurochem. 2007, 103, 467–474. [Google Scholar] [CrossRef]
- Schneider, J.A.; Arvanitakis, Z.; Bang, W.; Bennett, D.A. Mixed Brain Pathologies Account for Most Dementia Cases in Community-Dwelling Older Persons. Neurology 2007, 69, 2197–2204. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Balado, J.; Eich, T.S. GABAergic Dysfunction, Neural Network Hyperactivity and Memory Impairments in Human Aging and Alzheimer’s Disease. Semin. Cell Dev. Biol. 2021, 116, 146–159. [Google Scholar] [CrossRef]
- Lithgow, B. A Methodology for Detecting Field Potentials from the External Ear Canal: NEER and EVestG. Ann. Biomed. Eng. 2012, 40, 1835–1850. [Google Scholar] [CrossRef] [PubMed]
- Suleiman, A.; Lithgow, B.; Dastgheib, Z.; Mansouri, B.; Moussavi, Z. Quantitative Measurement of Post-Concussion Syndrome Using Electrovestibulography. Sci. Rep. 2017, 7, 16371. [Google Scholar] [CrossRef] [PubMed]
- Dastgheib, Z.A.; Lithgow, B.; Moussavi, Z. Diagnosis of Parkinson’s Disease Using Electrovestibulography. Med. Biol. Eng. Comput. 2012, 50, 483–491. [Google Scholar] [CrossRef]
- Blakley, B.; Dastgheib, Z.A.; Lithgow, B.; Moussavi, Z. Preliminary Report: Neural Firing Patterns Specific for Meniere’s Disease. J. Otolaryngol.-Head Neck Surg. 2014, 43, 52. [Google Scholar] [CrossRef]
- Dastgheib, Z.A.; Lithgow, B.; Blakley, B.; Moussavi, Z. A New Diagnostic Vestibular Evoked Response. J. Otolaryngol. Head Neck Surg. 2015, 44, 14. [Google Scholar] [CrossRef]
- Dastgheib, Z.A.; Lithgow, B.; Blakely, B.; Moussavi, Z. Application of Vestibular Spontaneous Response as a Diagnostic Aid for Meniere’s Disease. Ann. Biomed. Eng. 2016, 44, 1672–1684. [Google Scholar] [CrossRef] [PubMed]
- Lithgow, B.; Moussavi, Z.; Fitzgerald, P. Quantitative Separation of the Depressive Phase of Bipolar Disorder and Major Depressive Disorder Using Electrovestibulography. World J. Biol. Psychiatry 2019, 20, 1–35. [Google Scholar] [CrossRef]
- Lithgow, B.J.; Dastgheib, Z.; Anssari, N.; Mansouri, B.; Blakley, B.; Ashiri, M.; Moussavi, Z. Physiological Separation of Alzheimer’s Disease and Alzheimer’s Disease with Significant Levels of Cerebrovascular Symptomology and Healthy Controls. Med. Biol. Eng. Comput. 2021, 59, 1597–1610. [Google Scholar] [CrossRef]
- Cronin, T.; Arshad, Q.; Seemungal, B.M. Vestibular Deficits in Neurodegenerative Disorders: Balance, Dizziness, and Spatial Disorientation. Front. Neurol. 2017, 8, 538. [Google Scholar] [CrossRef] [PubMed]
- Lithgow, B.J.; Dastgheib, Z.; Moussavi, Z. Baseline Prediction of rTMS Efficacy in Alzheimer Patients. Psychiatry Res. 2022, 308, 114348. [Google Scholar] [CrossRef] [PubMed]
- Dastgheib, Z.A.; Lithgow, B.J.; Moussavi, Z.K. An Unbiased Algorithm for Objective Separation of Alzheimer’s, Alzheimer’s Mixed with Cerebrovascular Symptomology, and Healthy Controls from One Another Using Electrovestibulography (EVestG). Med. Biol. Eng. Comput. 2022, 60, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Lithgow, B.J.; Garrett, A.L.; Moussavi, Z.M.; Gurvich, C.; Kulkarni, J.; Maller, J.J.; Fitzgerald, P.B. Major Depression and Electrovestibulography. World J. Biol. Psychiatry 2015, 16, 334–350. [Google Scholar] [CrossRef]
- Marlinsky, V.V. The Effect of Somatosensory Stimulation on Second-Order and Efferent Vestibular Neurons in the Decerebrate Decerebellate Guinea-Pig. Neuroscience 1995, 69, 661–669. [Google Scholar] [CrossRef]
- Marczynski, T.J. GABAergic Deafferentation Hypothesis of Brain Aging and Alzheimer’s Disease Revisited. Brain Res. Bull. 1998, 45, 341–379. [Google Scholar] [CrossRef]
- Rissman, R.A.; De Blas, A.L.; Armstrong, D.M. GABA(A) Receptors in Aging and Alzheimer’s Disease. J. Neurochem. 2007, 103, 1285–1292. [Google Scholar] [CrossRef]
- Govindpani, K.; Calvo-Flores Guzmán, B.; Vinnakota, C.; Waldvogel, H.J.; Faull, R.L.; Kwakowsky, A. Towards a Better Understanding of GABAergic Remodeling in Alzheimer’s Disease. Int. J. Mol. Sci. 2017, 18, 1813. [Google Scholar] [CrossRef]
- Harris, S.S.; Wolf, F.; De Strooper, B.; Busche, M.A. Tipping the Scales: Peptide-Dependent Dysregulation of Neural Circuit Dynamics in Alzheimer’s Disease. Neuron 2020, 107, 417–435. [Google Scholar] [CrossRef]
- Cortes, C.; Galindo, F.; Galicia, S.; Cebada, J.; Flores, A. Excitatory Actions of GABA in Developing Chick Vestibular Afferents: Effects on Resting Electrical Activity. Synapse 2013, 67, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Cardin, J.A. Inhibitory Interneurons Regulate Temporal Precision and Correlations in Cortical Circuits. Trends Neurosci. 2018, 41, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Yang, E.J.; Cho, K.; Lim, J.Y.; Paik, N.-J. Functional Recovery after Ischemic Stroke Is Associated with Reduced GABAergic Inhibition in the Cerebral Cortex: A GABA PET Study. Neurorehabil Neural Repair. 2014, 28, 576–583. [Google Scholar] [CrossRef]
- Zhang, X.; Jun, P.; Liu, B.; Li, Z. Effects of Glutamate and γ-Aminobutyric Acid of Cerebral Ischemia on Rhesus Monkey. Res. Sq. 2022; preprint. [Google Scholar] [CrossRef]
- Wang, L.; Huang, Y.; Wu, J.; Lv, G.; Zhou, L.; Jia, J. Effect of Buyang Huanwu Decoction on Amino Acid Content in Cerebrospinal Fluid of Rats during Ischemic/Reperfusion Injury. J. Pharm. Biomed. Anal. 2013, 86, 143–150. [Google Scholar] [CrossRef]
- Buia, C.; Tiesinga, P. Attentional Modulation of Firing Rate and Synchrony in a Model Cortical Network. J. Comput. Neurosci. 2006, 20, 247–264. [Google Scholar] [CrossRef]
- Graupner, M.; Reyes, A.D. Synaptic Input Correlations Leading to Membrane Potential Decorrelation of Spontaneous Activity in Cortex. J. Neurosci. 2013, 33, 15075–15085. [Google Scholar] [CrossRef]
- Helias, M.; Tetzlaff, T.; Diesmann, M. The Correlation Structure of Local Neuronal Networks Intrinsically Results from Recurrent Dynamics. PLOS Comput. Biol. 2014, 10, e1003428. [Google Scholar] [CrossRef] [PubMed]
- Tetzlaff, T.; Helias, M.; Einevoll, G.T.; Diesmann, M. Decorrelation of Neural-Network Activity by Inhibitory Feedback. PLOS Comput. Biol. 2012, 8, e1002596. [Google Scholar] [CrossRef]
- Jin, G.-S.; Li, X.-L.; Jin, Y.-Z.; Kim, M.S.; Park, B.R. Role of Peripheral Vestibular Receptors in the Control of Blood Pressure Following Hypotension. Korean J. Physiol. Pharmacol. 2018, 22, 363–368. [Google Scholar] [CrossRef]
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A Brief Screening Tool for Mild Cognitive Impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef]
- Montgomery, S.A.; Åsberg, M. A New Depression Scale Designed to Be Sensitive to Change. Br. J. Psychiatry 1979, 134, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, S.S.; Wilk, M.B. An Analysis of Variance Test for Normality (Complete Samples). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Wilcoxon, F. Individual Comparisons by Ranking Methods. Biom. Bull. 1945, 1, 80–83. [Google Scholar] [CrossRef]
- Kalpić, D.; Hlupić, N.; Lovrić, M. Student’s t-Tests. In International Encyclopedia of Statistical Science; Lovric, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 1559–1563. ISBN 978-3-642-04898-2. [Google Scholar]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. An Introduction to Statistical Learning: With Applications in R; Springer Publishing Company Incorporated: New York, NY, USA, 2014; ISBN 978-1-4614-7137-0. [Google Scholar]
- Platt, J.C. Probabilistic Outputs for Support Vector Machines and Comparisons to Regularized Likelihood Methods. Adv. Large Margin Classif. 1999, 10, 61–74. [Google Scholar]
- Carson, N.; Leach, L.; Murphy, K.J. A Re-Examination of Montreal Cognitive Assessment (MoCA) Cutoff Scores. Int. J. Geriatr. Psychiatry 2018, 33, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Garrett, A.; Heibert, D.; Lithgow, B. Electrovestibulography: The “DC” Potential Used to Separate Meniere’s Disease and Benign Paroxysmal Positional Vertigo. In Proceedings of the 2007 29th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Lyon, France, 22–26 August 2007; Volume 2007, pp. 2381–2384. [Google Scholar] [CrossRef]
- Smith, P.F. The Growing Evidence for the Importance of the Otoliths in Spatial Memory. Front. Neural Circuits 2019, 13, 66. [Google Scholar] [CrossRef]
- Agrawal, Y.; Smith, P.F.; Rosenberg, P.B. Vestibular Impairment, Cognitive Decline and Alzheimer’s Disease: Balancing the Evidence. Aging Ment. Health 2020, 24, 705–708. [Google Scholar] [CrossRef]
- Kamil, R.J.; Jacob, A.; Ratnanather, J.T.; Resnick, S.M.; Agrawal, Y. Vestibular Function and Hippocampal Volume in the Baltimore Longitudinal Study of Aging (BLSA). Otol. Neurotol. 2018, 39, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Palop, J.J.; Mucke, L. Amyloid-Beta-Induced Neuronal Dysfunction in Alzheimer’s Disease: From Synapses toward Neural Networks. Nat. Neurosci. 2010, 13, 812–818. [Google Scholar] [CrossRef]
- Hector, A.; Brouillette, J. Hyperactivity Induced by Soluble Amyloid-β Oligomers in the Early Stages of Alzheimer’s Disease. Front. Mol. Neurosci. 2020, 13, 600084. [Google Scholar] [CrossRef]
- Kelly, L.; Seifi, M.; Ma, R.; Mitchell, S.J.; Rudolph, U.; Viola, K.L.; Klein, W.L.; Lambert, J.J.; Swinny, J.D. Identification of Intraneuronal Amyloid Beta Oligomers in Locus Coeruleus Neurons of Alzheimer’s Patients and Their Potential Impact on Inhibitory Neurotransmitter Receptors and Neuronal Excitability. Neuropathol. Appl. Neurobiol. 2021, 47, 488–505. [Google Scholar] [CrossRef] [PubMed]
- Cline, E.N.; Bicca, M.A.; Viola, K.L.; Klein, W.L. The Amyloid-β Oligomer Hypothesis: Beginning of the Third Decade. J. Alzheimers Dis. 2018, 64, S567–S610. [Google Scholar] [CrossRef]
- Busche, M.A.; Eichhoff, G.; Adelsberger, H.; Abramowski, D.; Wiederhold, K.-H.; Haass, C.; Staufenbiel, M.; Konnerth, A.; Garaschuk, O. Clusters of Hyperactive Neurons near Amyloid Plaques in a Mouse Model of Alzheimer’s Disease. Science 2008, 321, 1686–1689. [Google Scholar] [CrossRef] [PubMed]
- Busche, M.A.; Chen, X.; Henning, H.A.; Reichwald, J.; Staufenbiel, M.; Sakmann, B.; Konnerth, A. Critical Role of Soluble Amyloid-β for Early Hippocampal Hyperactivity in a Mouse Model of Alzheimer’s Disease. Proc. Natl. Acad. Sci. USA 2012, 109, 8740–8745. [Google Scholar] [CrossRef] [PubMed]
- Busche, M.A.; Kekuš, M.; Adelsberger, H.; Noda, T.; Förstl, H.; Nelken, I.; Konnerth, A. Rescue of Long-Range Circuit Dysfunction in Alzheimer’s Disease Models. Nat. Neurosci. 2015, 18, 1623–1630. [Google Scholar] [CrossRef]
- Keskin, A.D.; Kekuš, M.; Adelsberger, H.; Neumann, U.; Shimshek, D.R.; Song, B.; Zott, B.; Peng, T.; Förstl, H.; Staufenbiel, M.; et al. BACE Inhibition-Dependent Repair of Alzheimer’s Pathophysiology. Proc. Natl. Acad. Sci. USA 2017, 114, 8631–8636. [Google Scholar] [CrossRef]
- Pontes, A.; Zhang, Y.; Hu, W. Novel Functions of GABA Signaling in Adult Neurogenesis. Front. Biol. 2013, 8, 496–507. [Google Scholar] [CrossRef]
- Verret, L.; Mann, E.O.; Hang, G.B.; Barth, A.M.I.; Cobos, I.; Ho, K.; Devidze, N.; Masliah, E.; Kreitzer, A.C.; Mody, I.; et al. Inhibitory Interneuron Deficit Links Altered Network Activity and Cognitive Dysfunction in Alzheimer Model. Cell 2012, 149, 708–721. [Google Scholar] [CrossRef]
- Smith, P.F. Age-Related Neurochemical Changes in the Vestibular Nuclei. Front. Neurol. 2016, 7, 20. [Google Scholar] [CrossRef]
- Balaban, C.D.; Jacob, R.G.; Furman, J.M. Neurologic Bases for Comorbidity of Balance Disorders, Anxiety Disorders and Migraine: Neurotherapeutic Implications. Expert. Rev. Neurother. 2011, 11, 379–394. [Google Scholar] [CrossRef]
- Thielen, J.; Gancheva, S.; Hong, D.; Rohani Rankouhi, S.; Chen, B.; Apostolopoulou, M.; Anadol-Schmitz, E.; Roden, M.; Norris, D.G.; Tendolkar, I. Higher GABA Concentration in the Medial Prefrontal Cortex of Type 2 Diabetes Patients Is Associated with Episodic Memory Dysfunction. Hum. Brain Mapp. 2019, 40, 4287–4295. [Google Scholar] [CrossRef] [PubMed]
- Van Bussel, F.C.G.; Backes, W.H.; Hofman, P.A.M.; Puts, N.A.J.; Edden, R.A.E.; van Boxtel, M.P.J.; Schram, M.T.; Stehouwer, C.D.A.; Wildberger, J.E.; Jansen, J.F.A. Increased GABA Concentrations in Type 2 Diabetes Mellitus Are Related to Lower Cognitive Functioning. Medicine 2016, 95, e4803. [Google Scholar] [CrossRef] [PubMed]
- Sickmann, H.M.; Waagepetersen, H.S.; Schousboe, A.; Benie, A.J.; Bouman, S.D. Obesity and Type 2 Diabetes in Rats Are Associated with Altered Brain Glycogen and Amino-Acid Homeostasis. J. Cereb. Blood Flow. Metab. 2010, 30, 1527–1537. [Google Scholar] [CrossRef] [PubMed]
- Landau, S.; Everitt, B. A Handbook of Statistical Analyses Using SPSS; Chapman & Hall/CRC: Boca Raton, FL, USA, 2004. [Google Scholar]
- Pituch, K.A.; Stevens, J. Applied Multivariate Statistics for the Social Sciences: Analyses with SAS and IBM’s SPSS, 6th ed.; Routledge; Taylor & Francis Group: New York, NY, USA, 2016. [Google Scholar]
- Templeton, G.F. A Two-Step Approach for Transforming Continuous Variables to Normal: Implications and Recommendations for IS Research. Commun. Assoc. Inf. Syst. 2011, 28, 41–58. [Google Scholar] [CrossRef]
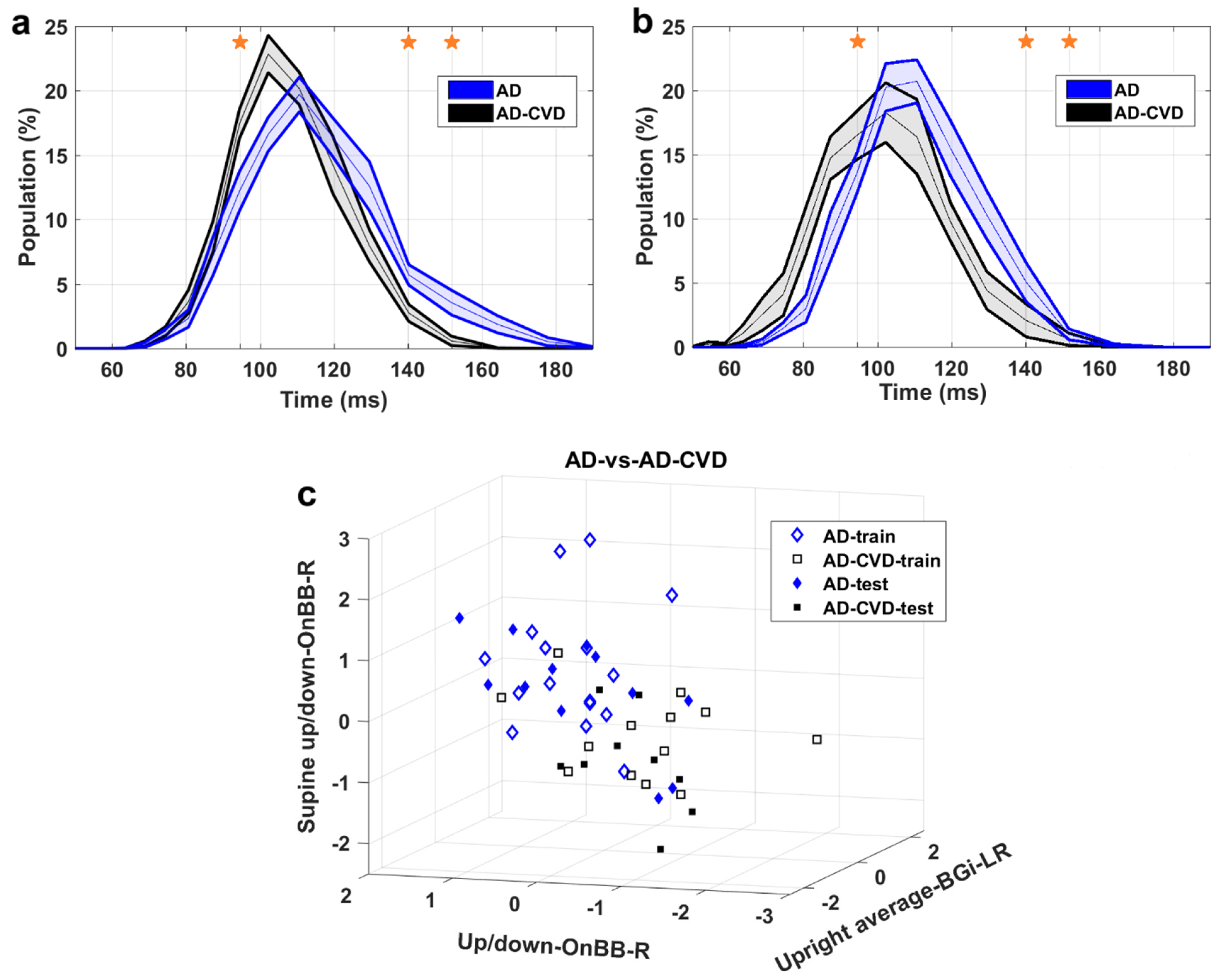
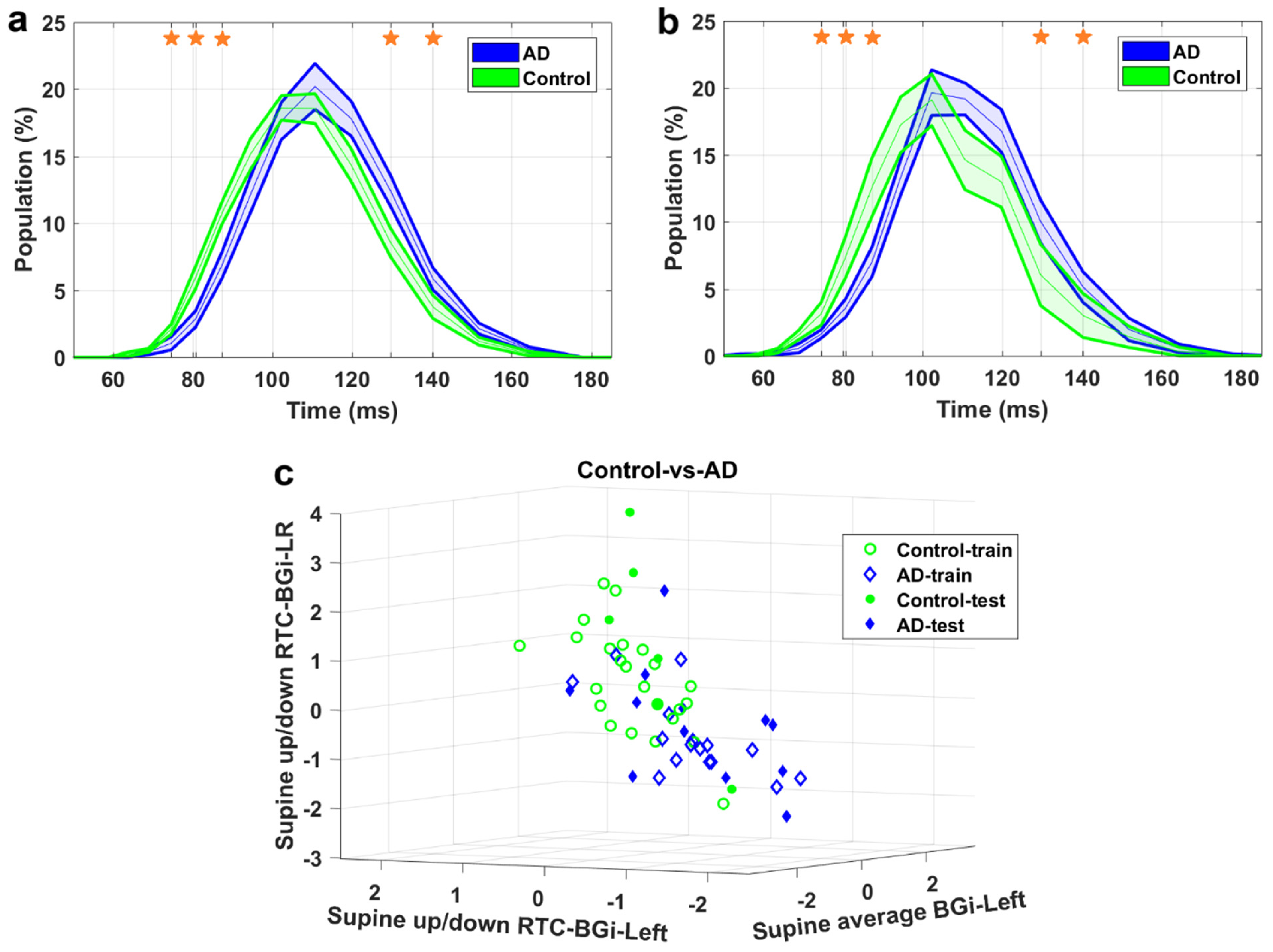
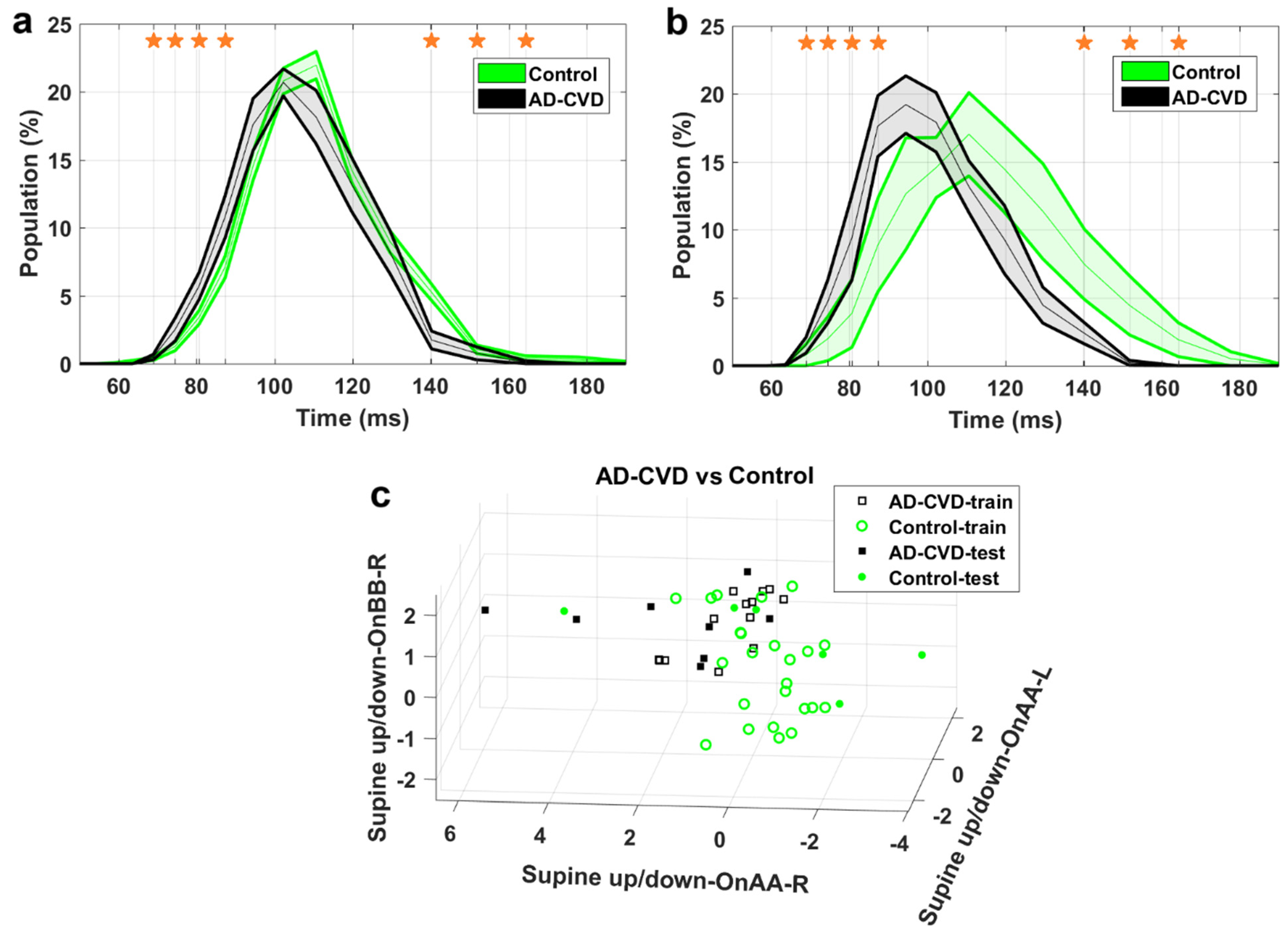
| Age (µ ± SD) | Sex | MoCA (µ ± SD) | Modified HIS (µ ± SD) | MADRS (µ ± SD) | |
|---|---|---|---|---|---|
| Control, N = 24 | 65.3 ± 7 | 9 M | 27.6 ± 1.7 | - | 2.6 ± 5.7 |
| AD, N = 16 | 72.5 ± 7.5 | 11 M | 16.4 ± 4.8 | 1.8 ± 1.2 | 1.9 ± 2.8 |
| AD-CVD, N = 13 | 75.8 ± 7.3 | 9 M | 17 ± 4.4 | 5.6 ± 1.4 | 3.1 ± 4 |
| Blind testing AD, N = 12 | 67.2 ± 7.1 | 9 M | 16 ± 6.7 | 1.3 ± 1.3 | 4.7 ± 4.7 |
| Blind testing AD-CVD, N = 9 | 71.3 ± 7.7 | 6 M | 16.8 ± 6.7 | 4.6 ± 1 | 2.2 ± 3.6 |
| Blind testing controls: | |||||
| 69.4 ± 5 | 4 M | 26 ± 2.5 | - | 4 ± 3.4 |
| 69.8 ± 4.1 | 3 M | 27 ± 1.8 | - | 3 ± 3.2 |
| Averaged Test Performances of the Binary Classifiers on Training Dataset | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| AD-vs.-AD-CVD | Control-vs.-AD | AD-CVD-vs.-Control | |||||||||
| Tilt | Sens (%) | Spec (%) | Acc (%) | Tilt | Sens (%) | Spec (%) | Acc (%) | Tilt | Sens (%) | Spec (%) | Acc (%) |
| Back/forward a | 95 | 60 | 80 | Supine up/down a | 86.7 | 80 | 82.3 | Supine up/down a | 60 | 90 | 80.3 |
| Supine up/down a | 70 | 80 | 76.7 | Back/forward | 75 | 70 | 73 | IT a | 55 | 88.3 | 77 |
| Up/down a | 80 | 70 | 75 | Supine rotation | 80 | 60 | 70.3 | Rotation | 45 | 85 | 72.1 |
| IT | 85 | 60 | 74.2 | Up/down | 86.7 | 45 | 69.7 | Back/forward | 30 | 88.3 | 68.3 |
| CT | 70 | 60 | 65.8 | IT | 76.7 | 55 | 68 | Supine rotation | 25 | 86.7 | 65.5 |
| Supine rotation | 80 | 40 | 62.5 | CT | 83.3 | 35 | 63.5 | Up/down | 10 | 85 | 59 |
| Rotation | 80 | 40 | 61.7 | Rotation | 75 | 25 | 54 | CT | 15 | 76.6 | 54.8 |
| Selected Most Informative Features of the Binary Classifiers | ||||
|---|---|---|---|---|
| Tilt | Signal Type | Segment_Side | AUC | |
| AD-vs.-AD-CVD | F1—Upright average | IH33 | BGi_LR | 0.64 |
| F2—Up/down | IH33 | OnBB_R | 0.77 | |
| F3—Supine up/down | IH33 | OnBB_R | 0.79 | |
| Control-vs.-AD | F1—Supine average | IH33 | BGi_L | 0.62 |
| F2—Supine up/down | IH33 | RTC_BGi_L | 0.78 | |
| F3—Supine up/down | IH33 | RTC_BGi_LR | 0.82 | |
| AD-CVD-vs.-Control | F1—Supine up/down | IH33 | OnAA_L | 0.51 |
| F2—Supine up/down | IH33 | OnAA_R | 0.78 | |
| F3—Supine up/down | IH33 | OnBB_R | 0.51 | |
| Averaged Test Performances of the Binary Classifiers on the Blind Testing Dataset | ||||
|---|---|---|---|---|
| Sens (%) | Spec (%) | Acc (%) | AUC | |
| AD-vs.-AD-CVD | 75.11 | 88.9 | 80.9 | F1 = 0.66, F2 = 0.77, F3 = 0.79 |
| Control-vs.-AD | 87.6 | 66.4 | 74.9 | F1 = 0.62, F2 = 0.8, F3 = 0.82 |
| AD-CVD-vs.-Control | 72.5 | 67 | 70.2 | F1 = 0.5, F2 = 0.77, F3 = 0.51 |
| Train, Test Dataset Classification Results | True Class | ||||||
|---|---|---|---|---|---|---|---|
| Total Number = 54, 27 | AD | AD-CVD | Control | Sens vs. Rest (%) | Spec vs. Rest (%) | Balanced Accuracy (%) | |
| Predicted Class | AD | 15, 10 | 2, 0 | 2, 0 | 93.8, 83.3 | 89.2, 100 | 85.7, 79.6 |
| AD-CVD | 1, 2 | 11, 8 | 3, 2 | 84.6, 88.9 | 90, 77.8 | ||
| Control | 0, 0 | 0, 1 | 19, 4 | 79.2, 66.7 | 100, 95.2 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dastgheib, Z.A.; Lithgow, B.J.; Moussavi, Z.K. Evaluating the Diagnostic Value of Electrovestibulography (EVestG) in Alzheimer’s Patients with Mixed Pathology: A Pilot Study. Medicina 2023, 59, 2091. https://doi.org/10.3390/medicina59122091
Dastgheib ZA, Lithgow BJ, Moussavi ZK. Evaluating the Diagnostic Value of Electrovestibulography (EVestG) in Alzheimer’s Patients with Mixed Pathology: A Pilot Study. Medicina. 2023; 59(12):2091. https://doi.org/10.3390/medicina59122091
Chicago/Turabian StyleDastgheib, Zeinab A., Brian J. Lithgow, and Zahra K. Moussavi. 2023. "Evaluating the Diagnostic Value of Electrovestibulography (EVestG) in Alzheimer’s Patients with Mixed Pathology: A Pilot Study" Medicina 59, no. 12: 2091. https://doi.org/10.3390/medicina59122091
APA StyleDastgheib, Z. A., Lithgow, B. J., & Moussavi, Z. K. (2023). Evaluating the Diagnostic Value of Electrovestibulography (EVestG) in Alzheimer’s Patients with Mixed Pathology: A Pilot Study. Medicina, 59(12), 2091. https://doi.org/10.3390/medicina59122091