Association of Genetic Polymorphisms with Abdominal Aortic Aneurysm in the Processes of Apoptosis, Inflammation, and Cholesterol Metabolism
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Genotyping
2.3. Statistical Analysis
3. Results
3.1. Patient Characteristics
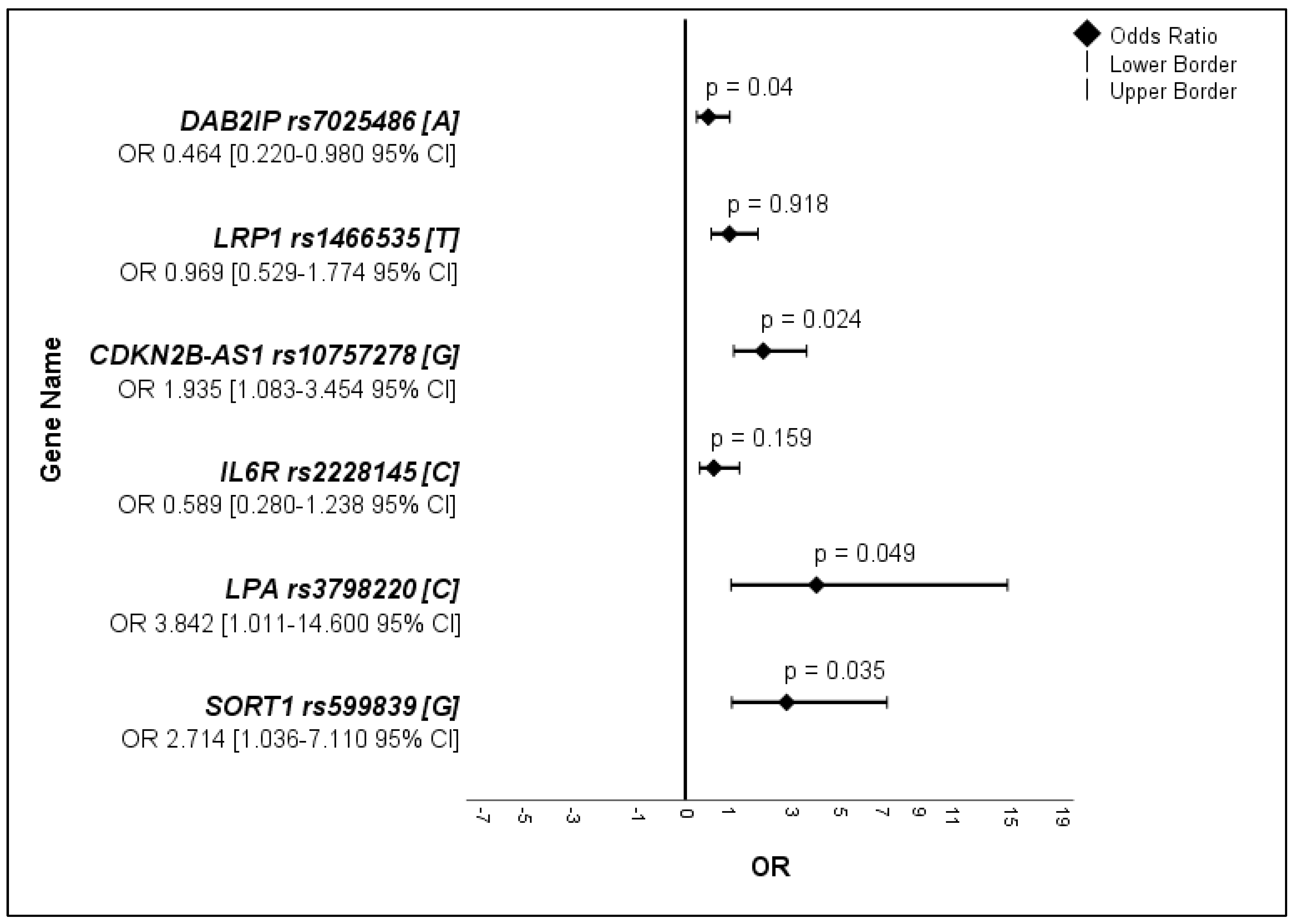
3.2. Allele Frequencies, Haplotypes, and Mutations of the SNPs
3.2.1. Allele Frequencies
3.2.2. Haplotypes
3.2.3. Mutations
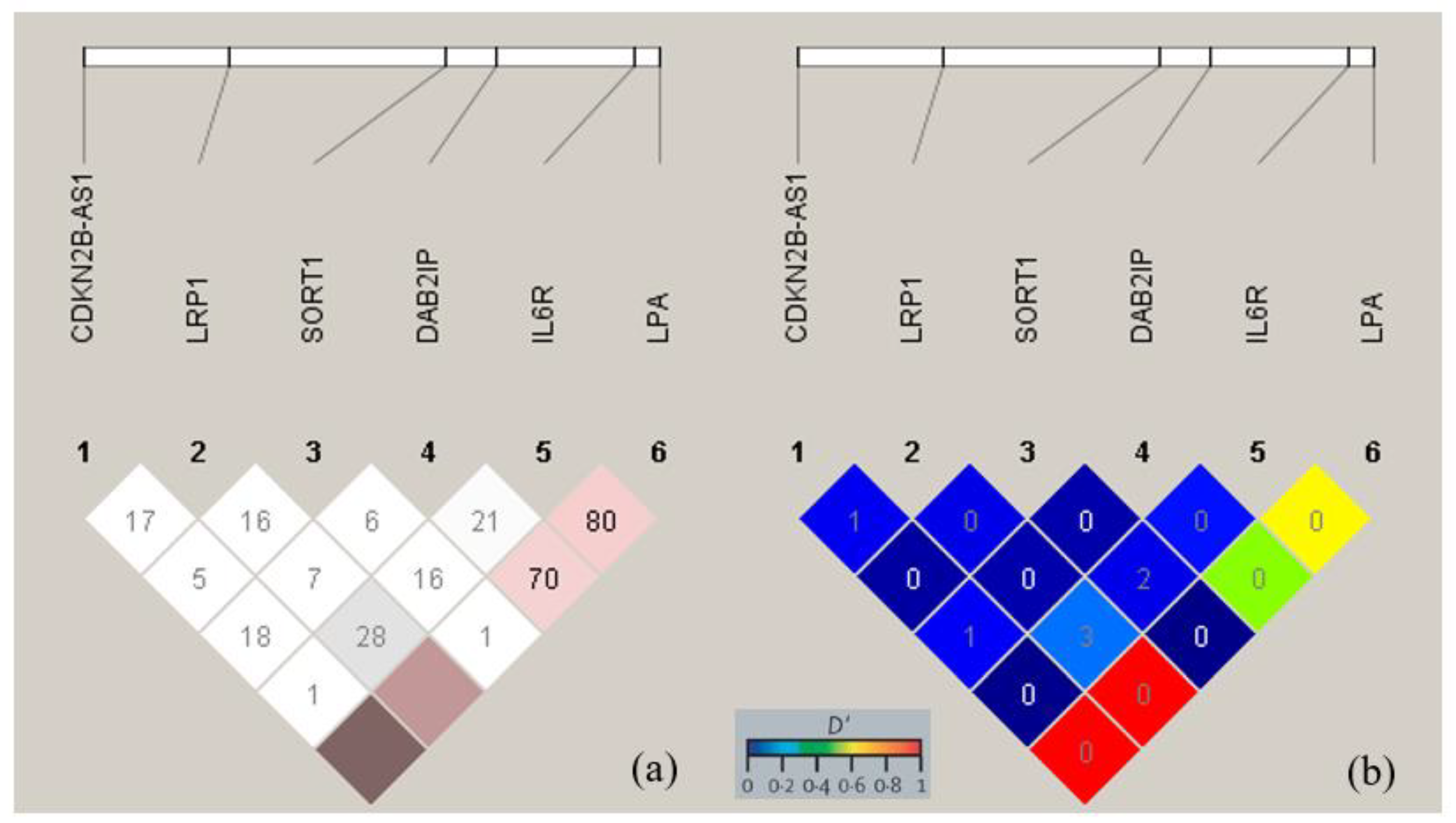
3.3. Linkage Disequilibrium
4. Discussion
4.1. Role of VSMC Apoptosis in Aneurysmal Formation–Potential Involvement of DAB2IP rs7025486[A], SORT1 rs599839[G], and CDKN2BAS rs10757278[G]
4.2. Involvement of Hypercholesterolemia in Aneurysmal Formation–Potential Roles of SORT1 rs599839 [G], LRP1 rs1466535 [T], and LPA rs3798220 [C]
4.3. Role of Inflammatory Mediators in Aneurysmal Formation–Potential Involvement of IL6R rs2228145[C] and LPA rs3798220[C]
4.4. Limitations of This Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Debus, E.S. S3-Leitlinie zum Screening, Diagnostik Therapie und Nachsorge des Bauchaortenaneurysmas. Gefässchirurgie 2018, 23, 402–403. [Google Scholar] [CrossRef]
- Trenner, M.; Salvermoser, M.; Reutersberg, B.; Busch, A.; Schmid, V.; Eckstein, H.H.; Kuehnl, A. Regional variation in endovascular treatment rate and in-hospital mortality of abdominal aortic aneurysms in Germany. Vasa 2019, 49, 107–114. [Google Scholar] [CrossRef]
- Ye, Z.; Austin, E.; Schaid, D.J.; Bailey, K.R.; Pellikka, P.A.; Kullo, I.J. A DAB2IP genotype: Sex interaction is associated with abdominal aortic aneurysm expansion. J. Investig. Med. 2017, 65, 1077–1082. [Google Scholar] [CrossRef]
- Duellman, T.; Warren, C.L.; Matsumura, J.; Yang, J. Analysis of multiple genetic polymorphisms in aggressive-growing and slow-growing abdominal aortic aneurysms. J. Vasc. Surg 2014, 60, 613–621 e613. [Google Scholar] [CrossRef]
- Li, T.; Jing, J.; Sun, L.; Jiang, B.; Xin, S.; Yang, J.; Yuan, Y. TLR4 and MMP2 polymorphisms and their associations with cardiovascular risk factors in susceptibility to aortic aneurysmal diseases. Biosci. Rep. 2019, 39, BSR20181591. [Google Scholar] [CrossRef]
- Carino, D.; Sarac, T.P.; Ziganshin, B.A.; Elefteriades, J.A. Abdominal Aortic Aneurysm: Evolving Controversies and Uncertainties. Int. J. Angiol. 2018, 27, 58–80. [Google Scholar] [CrossRef]
- Sakalihasan, N.; Defraigne, J.O.; Kerstenne, M.A.; Cheramy-Bien, J.P.; Smelser, D.T.; Tromp, G.; Kuivaniemi, H. Family members of patients with abdominal aortic aneurysms are at increased risk for aneurysms: Analysis of 618 probands and their families from the Liege AAA Family Study. Ann. Vasc. Surg. 2014, 28, 787–797. [Google Scholar] [CrossRef]
- Mathur, A.; Mohan, V.; Ameta, D.; Gaurav, B.; Haranahalli, P. Aortic aneurysm. J. Transl. Int. Med. 2016, 4, 35–41. [Google Scholar] [CrossRef]
- Bradley, D.T.; Badger, S.A.; McFarland, M.; Hughes, A.E. Abdominal Aortic Aneurysm Genetic Associations: Mostly False? A Systematic Review and Meta-analysis. Eur. J. Vasc. Endovasc. Surg. 2016, 51, 64–75. [Google Scholar] [CrossRef]
- Jones, G.T.; Tromp, G.; Kuivaniemi, H.; Gretarsdottir, S.; Baas, A.F.; Giusti, B.; Strauss, E.; Van’t Hof, F.N.; Webb, T.R.; Erdman, R.; et al. Meta-Analysis of Genome-Wide Association Studies for Abdominal Aortic Aneurysm Identifies Four New Disease-Specific Risk Loci. Circ. Res. 2017, 120, 341–353. [Google Scholar] [CrossRef]
- Hinterseher, I.; Krex, D.; Kuhlisch, E.; Pilarsky, C.; Schneiders, W.; Saeger, H.D.; Bergert, H. Analysis of tissue inhibitor of metalloproteinase-2 gene polymorphisms in a caucasian population with abdominal aortic aneurysms. Zentralbl. Chir. 2008, 133, 332–337. [Google Scholar] [CrossRef]
- Delbridge, A.R.; Valente, L.J.; Strasser, A. The role of the apoptotic machinery in tumor suppression. Cold Spring Harb. Perspect Biol. 2012, 4, a008789. [Google Scholar] [CrossRef]
- Valentino, E.; Bellazzo, A.; Di Minin, G.; Sicari, D.; Apollonio, M.; Scognamiglio, G.; Di Bonito, M.; Botti, G.; Del Sal, G.; Collavin, L. Mutant p53 potentiates the oncogenic effects of insulin by inhibiting the tumor suppressor DAB2IP. Proc. Natl. Acad. Sci. USA 2017, 114, 7623–7628. [Google Scholar] [CrossRef]
- Hellenthal, F.A.; Buurman, W.A.; Wodzig, W.K.; Schurink, G.W. Biomarkers of AAA progression. Part 1: Extracellular matrix degeneration. Nat. Rev. Cardiol. 2009, 6, 464–474. [Google Scholar] [CrossRef]
- Hellenthal, F.A.; Buurman, W.A.; Wodzig, W.K.; Schurink, G.W. Biomarkers of abdominal aortic aneurysm progression. Part 2: Inflammation. Nat. Rev. Cardiol. 2009, 6, 543–552. [Google Scholar] [CrossRef]
- Paige, E.; Clement, M.; Lareyre, F.; Sweeting, M.; Raffort, J.; Grenier, C.; Finigan, A.; Harrison, J.; Peters, J.E.; Sun, B.B.; et al. Interleukin-6 Receptor Signaling and Abdominal Aortic Aneurysm Growth Rates. Circ. Genom. Precis Med. 2019, 12, e002413. [Google Scholar] [CrossRef]
- Oner, M.G.; Rokavec, M.; Kaller, M.; Bouznad, N.; Horst, D.; Kirchner, T.; Hermeking, H. Combined Inactivation of TP53 and MIR34A Promotes Colorectal Cancer Development and Progression in Mice Via Increasing Levels of IL6R and PAI1. Gastroenterology 2018, 155, 1868–1882. [Google Scholar] [CrossRef]
- Stackelberg, O.; Wolk, A.; Eliasson, K.; Hellberg, A.; Bersztel, A.; Larsson, S.C.; Orsini, N.; Wanhainen, A.; Björck, M. Lifestyle and Risk of Screening-Detected Abdominal Aortic Aneurysm in Men. J. Am. Heart Assoc. 2017, 6, e004725. [Google Scholar] [CrossRef]
- Paththinige, C.S.; Sirisena, N.D.; Dissanayake, V. Genetic determinants of inherited susceptibility to hypercholesterolemia—A comprehensive literature review. Lipids Health Dis. 2017, 16, 103. [Google Scholar] [CrossRef]
- Lu, W.; Cheng, Y.C.; Chen, K.; Wang, H.; Gerhard, G.S.; Still, C.D.; Chu, X.; Yang, R.; Parihar, A.; O’Connell, J.R.; et al. Evidence for several independent genetic variants affecting lipoprotein (a) cholesterol levels. Hum. Mol. Genet. 2015, 24, 2390–2400. [Google Scholar] [CrossRef]
- Jones, G.T.; Bown, M.J.; Gretarsdottir, S.; Romaine, S.P.; Helgadottir, A.; Yu, G.; Tromp, G.; Norman, P.E.; Jin, C.; Baas, A.F.; et al. A sequence variant associated with sortilin-1 (SORT1) on 1p13.3 is independently associated with abdominal aortic aneurysm. Hum. Mol. Genet. 2013, 22, 2941–2947. [Google Scholar] [CrossRef]
- McCarthy, N.; Bennett, M. The regulation of vascular smooth muscle cell apoptosis. Cardiovasc. Res. 2000, 45, 747–755. [Google Scholar] [CrossRef]
- Leeper, N.J.; Raiesdana, A.; Kojima, Y.; Kundu, R.K.; Cheng, H.; Maegdefessel, L.; Toh, R.; Ahn, G.O.; Ali, Z.A.; Anderson, D.R.; et al. Loss of CDKN2B Promotes p53-Dependent Smooth Muscle Cell Apoptosis and Aneurysm Formation. Arterioscler. Thromb. Vasc. Biol. 2013, 33, e1–e10. [Google Scholar] [CrossRef]
- Min, W.; Lin, Y.; Tang, S.; Yu, L.; Zhang, H.; Wan, T.; Luhn, T.; Fu, H.; Chen, H. AIP1 recruits phosphatase PP2A to ASK1 in tumor necrosis factor-induced ASK1-JNK activation. Circ. Res. 2008, 102, 840–848. [Google Scholar] [CrossRef]
- Kanehisa, M. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef]
- Gretarsdottir, S.; Baas, A.F.; Thorleifsson, G.; Holm, H.; den Heijer, M.; de Vries, J.P.; Kranendonk, S.E.; Zeebregts, C.J.; van Sterkenburg, S.M.; Geelkerken, R.H.; et al. Genome-wide association study identifies a sequence variant within the DAB2IP gene conferring susceptibility to abdominal aortic aneurysm. Nat. Genet. 2010, 42, 692–697. [Google Scholar] [CrossRef]
- Zhang, Z.; Jiang, W.; Yang, H.; Lin, Q.; Qin, X. The miR-182/SORT1 axis regulates vascular smooth muscle cell calcification in vitro and in vivo. Exp. Cell Res. 2018, 362, 324–331. [Google Scholar] [CrossRef]
- Chaikof, E.L.; Dalman, R.L.; Eskandari, M.K.; Jackson, B.M.; Lee, W.A.; Mansour, M.A.; Mastracci, T.M.; Mell, M.; Murad, M.H.; Nguyen, L.L.; et al. The Society for Vascular Surgery practice guidelines on the care of patients with an abdominal aortic aneurysm. J. Vasc. Surg. 2018, 67, 2–77.e72. [Google Scholar] [CrossRef]
- Bayoglu, B.; Cakmak, H.A.; Yuksel, H.; Can, G.; Karadag, B.; Ulutin, T.; Vural, V.A.; Cengiz, M. Chromosome 9p21 rs10757278 polymorphism is associated with the risk of metabolic syndrome. Mol. Cell Biochem. 2013, 379, 77–85. [Google Scholar] [CrossRef]
- Aylon, Y.; Oren, M. The Hippo pathway, p53 and cholesterol. Cell Cycle 2016, 15, 2248–2255. [Google Scholar] [CrossRef]
- Rowe, V.L.; Stevens, S.L.; Reddick, T.T.; Freeman, M.B.; Donnell, R.; Carroll, R.C.; Goldman, M.H. Vascular smooth muscle cell apoptosis in aneurysmal, occlusive, and normal human aortas. J. Vasc. Surg. 2000, 31, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Howatt, D.A.; Balakrishnan, A.; Graham, M.J.; Mullick, A.E.; Daugherty, A. Hypercholesterolemia Induced by a PCSK9 Gain-of-Function Mutation Augments Angiotensin II–Induced Abdominal Aortic Aneurysms in C57BL/6 Mice—Brief Report. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1753–1757. [Google Scholar] [CrossRef] [PubMed]
- Dodington, D.W.; Desai, H.R.; Woo, M. JAK/STAT—Emerging Players in Metabolism. Trends Endocrinol. Metab. 2018, 29, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Clarke, R.; Peden, J.F.; Hopewell, J.C.; Kyriakou, T.; Goel, A.; Heath, S.C.; Parish, S.; Barlera, S.; Franzosi, M.G.; Rust, S.; et al. Genetic Variants Associated with Lp(a) Lipoprotein Level and Coronary Disease. New Engl. J. Med. 2009, 361, 2518–2528. [Google Scholar] [CrossRef]
- Helgadottir, A.; Gretarsdottir, S.; Thorleifsson, G.; Holm, H.; Patel, R.S.; Gudnason, T.; Jones, G.T.; van Rij, A.M.; Eapen, D.J.; Baas, A.F.; et al. Apolipoprotein(a) genetic sequence variants associated with systemic atherosclerosis and coronary atherosclerotic burden but not with venous thromboembolism. J. Am. Coll. Cardiol. 2012, 60, 722–729. [Google Scholar] [CrossRef]
- Enas, E.A.; Varkey, B.; Dharmarajan, T.S.; Pare, G.; Bahl, V.K. Lipoprotein(a): An independent, genetic, and causal factor for cardiovascular disease and acute myocardial infarction. Indian Heart J. 2019, 71, 99–112. [Google Scholar] [CrossRef]
- Harrison, S.C.; Smith, A.J.; Jones, G.T.; Swerdlow, D.I.; Rampuri, R.; Bown, M.J.; Folkersen, L.; Baas, A.F.; de Borst, G.J.; Blankensteijn, J.D.; et al. Interleukin-6 receptor pathways in abdominal aortic aneurysm. European Heart Journal 2013, 34, 3707–3716. [Google Scholar] [CrossRef]
- Harrison, D.A. The JAK/STAT Pathway. Cold Spring Harb. Perspect. Biol. 2012, 4, a011205. [Google Scholar] [CrossRef]
| Parameter | AAAs, n = 148 n (%) | Controls, n = 50 n (%) | p-Value |
|---|---|---|---|
| Sex | <0.001 | ||
| Female | 19 (12.8) | 27 (54.0) | |
| Male | 129 (87.2) | 23 (46.0) | |
| Age, years | 74.8 ± 8.3 | 49.5 ± 13.2 | <0.001 |
| 19–35 | 0 (0) | 10 (20.0) | |
| 36–65 | 22 (14.9) | 36 (72.0) | |
| >65 | 126 (85.1) | 4 (8.0) | |
| Body Mass Index, kg/m2 | 27.6 ± 4.6 | 26.3 ± 5.7 | 0.097 |
| Body height in cm | 176.5 ± 7.8 | 177.5 ± 9.6 | 0.462 |
| Body weight in kg | 86.1 ± 15.0 | 83.5 ± 23.0 | 0.358 |
| Hypertension | 61 (41.2) | 10 (20) | 0.07 |
| Systolic in mmHg | 133.1 ± 20.0 | 122.6 ± 11.9 | 0.001 |
| Diastolic in mmHg | 75.9 ± 11.7 | 73.0 ± 10.6 | 0.12 |
| MAP | 94.9 ± 12.6 | 89.5 ± 9.7 | 0.177 |
| Dyslipidemia | 66 (44.6) | 5 (10.0) | <0.001 |
| Smoking history | 95 (64.2) | 11 (22) | <0.001 |
| Smoking in pack years | 34.9 ± 14.9 | 20.7 ± 13.7 | 0.003 |
| PAD | 25 (16.9) | 6 (12.0) | 0.411 |
| Relatives with AAA history | 27 (18.2) | 3 (6.0) | 0.139 |
| Parent–child relationship * | 15 (10.1) | 1 (2.0) | |
| Sibling relationship | 3 (2.0) | 1 (2.0) | |
| Twin | 5 (3.4) | 0 (0) | |
| Others | 4 (2.7) | 1 (2.0) | |
| Sex of relatives diagnosed with AAA | 0.539 | ||
| Male–male or female–female | 19 (12.8) | 2 (4.0) | |
| Male–female or female–male | 7 (4.7) | 1 (2.0) | |
| Both sexes to male/female | 1 (0.7) | 0 (0) | |
| Younger than sample | 7 (4.7) | 2 (4.0) | |
| Older than sample | 20 (13.5) | 1 (2.0) | |
| Variable | DAB2IP rs7025486 | LRP1 rs1466535 | ||
|---|---|---|---|---|
| OR [95% CI] | p-Value | OR [95% CI] | p-Value | |
| Age, years | 1.353 [0.679–2.698] | 0.389 | 1.026 [0.534–1.970] | 0.938 |
| Male sex | 1.874 [0.809–4.341] | 0.139 | 0.873 [0.424–1.796] | 0.711 |
| Smoking history | 1.661 [0.865–3.193] | 0.126 | 1.002 [0.539–1.863] | 0.995 |
| Family history of AAA | 2.146 [0.896–5.141] | 0.081 | 3.275 [1.390–7.717] | 0.005 |
| First-degree relatives | 2.300 [0.869–6.087] | 0.087 | 1.985 [0.753–5.230] | 0.159 |
| Obesity | 1.229 [0.586–2.581] | 0.585 | 0.665 [0.304–1.454] | 0.304 |
| Hypertension | 3.295 [1.704–6.374] | <0.001 | 1.365 [0.723–2.578] | 0.337 |
| Dyslipidemia | 0.967 [0.497–1.882] | 0.922 | 0.795 [0.413–1.533] | 0.494 |
| PAD | 1.218 [0.520–2.850] | 0.650 | 1.253 [0.548–2.861] | 0.593 |
| Statins | 0.967 [0.497–1.882] | 0.922 | 0.795 [0.413–1.533] | 0.494 |
| Aspirin | 0.813 [0.415–1.589] | 0.544 | 0.596 [0.305–1.166] | 0.129 |
| Clopidogrel | 3.133 [0.963–10.200] | 0.048 | 1.891 [0.574–6.227] | 0.288 |
| Warfarin | 1.020 [0.379–2.745] | 0.969 | 1.751 [0.711–4.313] | 0.219 |
| Variable | CDKN2BAS rs10757278 | IL6R rs2228145 | ||
| OR [95% CI] | p-Value | OR [95% CI] | p-Value | |
| Age, years | 1.344 [0.733–2.467] | 0.339 | 0.496 [0.228–1.078] | 0.073 |
| Male sex | 1.327 [0.668–2.639] | 0.419 | 0.482 [0.211–1.100] | 0.079 |
| Smoking history | 1.258 [0.710–2.230] | 0.431 | 0.490 [0.224–1.075] | 0.072 |
| Family history of AAA | 1.213 [0.520–2.829] | 0.654 | 0.432 [0.096–1.933] | 0.260 |
| First-degree relatives | 1.401 [0.542–3.621] | 0.484 | 0.609 [0.133–2.777] | 0.517 |
| Obesity | 0.611 [0.301–1.241] | 0.171 | 0.788 [0.301–2.058] | 0.626 |
| Hypertension | 1.063 [0.588–1.922] | 0.839 | 0.981 [0.440–2.185] | 0.962 |
| Dyslipidemia | 1.276 [0.707–2.302] | 0.419 | 0.981 [0.440–2.185] | 0.962 |
| PAD | 1.509 [0.698–3.260] | 0.293 | 0.768 [0.249–2.373] | 0.646 |
| Statins | 1.276 [0.707–2.302] | 0.419 | 0.981 [0.440–2.185] | 0.962 |
| Aspirin | 2.031 [1.126–3.665] | 0.018 | 0.931 [0.419–2.073] | 0.862 |
| Clopidogrel | 3.239 [0.941–11.151] | 0.051 | 0.473 [0.059–3.799] | 0.471 |
| Warfarin | 1.182 [0.491–2.844] | 0.709 | 0.788 [0.219–2.830] | 0.714 |
| Variable | LPA rs3798220 | SORT1 rs599839 | ||
| OR [95% CI] | p-Value | OR [95% CI] | p-Value | |
| Age, years | 0.298 [0.069–1.285] | 0.087 | 1.381 [0.620–3.075] | 0.428 |
| Male sex | 0.488 [0.112–2.123] | 0.329 | 1.572 [0.609–4.059] | 0.347 |
| Smoking history | 0.863 [0.210–3.551] | 0.838 | 1.847 [0.862–3.960] | 0.111 |
| Family history of AAA | 0.988 [0.116–8.386] | 0.991 | 1.568 [0.576–4.266] | 0.375 |
| First-degree relatives | 1.365 [0.159–11.729] | 0.561 | 2.387 [0.839–6.794] | 0.095 |
| Obesity | 2.114 [0.485–9.212] | 0.385 | 2.419 [1.101–5.314] | 0.025 |
| Hypertension | 0.245 [0.030–2.032] | 0.263 | 1.654 [0.789–3.466] | 0.180 |
| Dyslipidemia | 0.585 [0.115–2.976] | 0.714 | 1.906 [0.911–3.991] | 0.084 |
| PAD | 1.050 [1.015–1.087] | 0.362 | 0.650 [0.212–1.992] | 0.610 |
| Statins | 0.585 [0.115–2.976] | 0.513 | 1.906 [0.911–3.991] | 0.084 |
| Aspirin | 0.234 [0.028–1.942] | 0.145 | 1.175 [0.556–2.483] | 0.672 |
| Clopidogrel | 1.045 [1.014–1.077] | 0.463 | 0.927 [0.194–4.431] | 0.925 |
| Warfarin | 1.048 [1.014–1.082] | 0.600 | 0.978 [0.311–3.076] | 0.970 |
| AAAs’ Allele n = 296 | Controls’ Allele n = 100 | OR | 95% CI | p-Value | |||||
|---|---|---|---|---|---|---|---|---|---|
| Allele | n | % | n | % | |||||
| DAB2IP rs7025486 + 501G>A | Male | G | 212 | 71.6 | 40 | 40.0 | |||
| A | 46 | 15.5 | 6 | 6.0 | 0.691 | 0.277–1.727 | 0.427 | ||
| Female | G | 32 | 10.8 | 51 | 51.0 | ||||
| A | 6 | 2.1 | 3 | 3.0 | 0.314 | 0.073–1.344 | 0.154 | ||
| Total | G | 244 | 82.4 | 91 | 91.0 | ||||
| A | 52 | 17.6 | 9 | 9.0 | 0.464 | 0.220–0.980 | 0.040 | ||
| LRP1 rs1466535 + 504C>T | Male | C | 219 | 74.0 | 36 | 36.0 | |||
| T | 39 | 13.2 | 10 | 10.0 | 0.641 | 0.294–1.397 | 0.260 | ||
| Female | C | 28 | 9.5 | 47 | 47.0 | ||||
| T | 10 | 3.4 | 7 | 7.0 | 2.398 | 0.820–7.014 | 0.104 | ||
| Total | C | 247 | 83.4 | 83 | 83.0 | ||||
| T | 49 | 16.6 | 17 | 17.0 | 0.969 | 0.529–1.774 | 0.918 | ||
| CDKN2BAS rs10757278 + 501A>G | Male | A | 184 | 62.2 | 38 | 38.0 | |||
| G | 74 | 25.0 | 8 | 8.0 | 1.910 | 0.851–4.289 | 0.112 | ||
| Female | A | 28 | 9.5 | 45 | 45.0 | ||||
| G | 10 | 3.4 | 9 | 9.0 | 1.786 | 0.646–4.935 | 0.260 | ||
| Total | A | 212 | 71.6 | 83 | 83.0 | ||||
| G | 84 | 28.4 | 17 | 17.0 | 1.935 | 1.083–3.454 | 0.024 | ||
| IL6R rs2228145 + 501A>C | Male | A | 240 | 81.1 | 41 | 41.0 | |||
| C | 18 | 6.1 | 5 | 5.0 | 0.615 | 0.216–1.748 | 0.358 | ||
| Female | A | 34 | 11.5 | 47 | 47.0 | ||||
| C | 4 | 1.4 | 7 | 7.0 | 0.790 | 0.214–2.914 | 0.723 | ||
| Total | A | 274 | 92.6 | 88 | 88.0 | ||||
| C | 22 | 7.4 | 12 | 12.0 | 0.589 | 0.280–1.238 | 0.159 | ||
| LPA rs3798220 + 501T>C | Male | T | 254 | 85.8 | 44 | 44.0 | |||
| C | 4 | 1.4 | 2 | 2.0 | 2.886 | 0.513–16.24 | 0.226 | ||
| Female | T | 38 | 12.8 | 51 | 51.0 | ||||
| C | 0 | 0.0 | 3 | 3.0 | 0.944 | 0.885–1.008 | 0.265 | ||
| Total | T | 292 | 98.6 | 95 | 95.0 | ||||
| C | 4 | 1.4 | 5 | 5.0 | 3.842 | 1.011–14.60 | 0.049 | ||
| SORT1 rs599839 + 813A>G | Male | A | 226 | 76.4 | 44 | 44.0 | |||
| G | 32 | 10.8 | 2 | 2.0 | 3.115 | 0.720–13.47 | 0.132 | ||
| Female | A | 33 | 11.1 | 51 | 51.0 | ||||
| G | 5 | 1.7 | 3 | 3.0 | 2.576 | 0.577–11.51 | 0.268 | ||
| Total | A | 259 | 87.5 | 95 | 95.0 | ||||
| G | 37 | 12.5 | 5 | 5.0 | 2.714 | 1.036–7.110 | 0.035 | ||
| Haplotype n (%) | MAF * (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Major Homozygote | Heterozygote | Minor Homozygote | p-Value | ||||||
| AAAs | Controls | AAAs | Controls | AAAs | Controls | ||||
| DAB2IP rs7025486 + 501G>A | Male | 90 (69.8) | 19 (82.6) | 32 (24.8) | 2 (8.7) | 7 (5.4) | 2 (8.7) | ||
| Female | 14 (73.7) | 24 (88.9) | 4 (21.1) | 3 (11.1) | 1 (5.2) | 0 (0) | |||
| Total | 104 (70.3) | 43 (86.0) | 36 (24.3) | 5 (10.0) | 8 (5.4) | 2 (4.0) | 0.037 | 25.5 | |
| LRP1 rs1466535 + 504C>T | Male | 95 (73.6) | 15 (65.2) | 29 (22.5) | 6 (26.1) | 5 (3.9) | 2 (8,7) | ||
| Female | 12 (63.2) | 20 (74.1) | 4 (21.1) | 7 (25.9) | 3 (15.7) | 0 (0) | |||
| Total | 107 (72.3) | 35 (70.0) | 33 (22.3) | 13 (26.0) | 8 (5.4) | 2 (4.0) | 0.835 | 16.7 | |
| CDKN2BAS rs10757278 + 501A>G | Male | 72 (55.8) | 17 (73.9) | 40 (31.0) | 4 (17.4) | 17 (13.2) | 2 (8.7) | ||
| Female | 10 (52.6) | 19 (70.4) | 8 (42.1) | 7 (25.9) | 1 (5.3) | 1 (3.7) | |||
| Total | 82 (55.4) | 36 (72.0) | 48 (32.4) | 11 (22.0) | 18 (12.2) | 3 (6.0) | 0.037 | 10.6 | |
| IL6R rs2228145 + 501A>C | Male | 114 (88.4) | 18 (78.3) | 12 (9.3) | 5 (21.7) | 3 (2.3) | 0 (0) | ||
| Female | 15 (78.9) | 20 (74.1) | 4 (21.1) | 7 (25.9) | 0 (0) | 0 (0) | |||
| Total | 129 (87.2) | 38 (76.0) | 16 (10.8) | 12 (24.0) | 3 (2.0) | 0 (0) | 0.046 | 15.4 | |
| LPA rs3798220 + 501T>C | Male | 125 (96.9) | 22 (95.7) | 4 (3.1) | 0 (0) | 0 (0) | 1 (4.3) | ||
| Female | 19 (100.0) | 24 (88.9) | 0 (0) | 3 (11.1) | 0 (0) | 0 (0) | |||
| Total | 144 (97.3) | 46 (92.0) | 4 (2.7) | 3 (6.0) | 0 (0) | 1 (2.0) | 0.146 | 8.6 | |
| SORT1 rs599839 + 813A>G | Male | 102 (79.0) | 21 (91.3) | 22 (17.1) | 2 (8.7) | 5 (3.9) | 0 (0) | ||
| Female | 16 (84.2) | 24 (88.9) | 1 (5.3) | 3 (11.1) | 2 (10.5) | 0 (0) | |||
| Total | 118 (79.7) | 45 (90.0) | 23 (15.5) | 5 (10.0) | 7 (4.8) | 0 (0) | 0.203 | 2.3 | |
| Mutation (n, %) | ||||||
|---|---|---|---|---|---|---|
| Occurred | Not Occurred | p-Value | ||||
| AAAs | Controls | AAAs | Controls | |||
| DAB2IP rs7025486 + 501G>A | Male | 39 (30.2) | 4 (17.4) | 90 (69.8) | 19 (82.6) | 0.314 |
| Female | 5 (26.3) | 3 (11.1) | 14 (73.7) | 24 (88.9) | 0.246 | |
| Total | 44 (29.7) | 7 (14.0) | 104 (70.4) | 43 (86.0) | 0.028 | |
| LRP1 rs1466535 + 504C>T | Male | 34 (26.4) | 8 (34.8) | 95 (73.6) | 15 (65.2) | 0.405 |
| Female | 7 (36.8) | 7 (25.9) | 12 (63.2) | 20 (74.1) | 0.428 | |
| Total | 41 (27.7) | 15 (30.0) | 107 (72.3) | 35 (70.0) | 0.755 | |
| CDKN2BAS rs10757278 + 501A>G | Male | 57 (44.2) | 6 (26.1) | 72 (55.8) | 17 (73.9) | 0.105 |
| Female | 8 (42.1) | 8 (29.6) | 11 (57.9) | 19 (70.4) | 0.382 | |
| Total | 65 (43.9) | 14 (28.0) | 83 (56.1) | 36 (72.0) | 0.047 | |
| IL6R rs2228145 + 501A>C | Male | 15 (11.6) | 5 (21.7) | 114 (88.4) | 18 (78.3) | 0.186 |
| Female | 4 (21.1) | 7 (25.9) | 15 (78.9) | 20 (74.1) | >0.995 | |
| Total | 19 (12.8) | 12 (24.0) | 129 (87.2) | 38 (76.0) | 0.073 | |
| LPA rs3798220 + 501T>C | Male | 4 (3.1) | 1 (4.3) | 125 (96.9) | 22 (95.7) | 0.565 |
| Female | 0 (0) | 3 (11.1) | 19 (100.0) | 24 (88.9) | 0.257 | |
| Total | 4 (2.7) | 4 (8.0) | 144 (97.3) | 46 (92.0) | 0.113 | |
| SORT1 rs599839 + 813A>G | Male | 27 (20.9) | 2 (8.7) | 102 (79.1) | 21 (91.3) | 0.250 |
| Female | 3 (15.8) | 3 (11.1) | 16 (84.2) | 24 (88.9) | 0.680 | |
| Total | 30 (20.3) | 5 (10.0) | 118 (79.7) | 45 (90.0) | 0.100 | |
| SNPs | Aneurysmal Sac Size | Morphology of AAA | ||||||
|---|---|---|---|---|---|---|---|---|
| Small (<50 mm) n (%) | Large (≥ 50 mm) n (%) | OR [95% CI] | p-Value * | Fusiform n (%) | Saccular n (%) | OR [95% CI] | p-Value * | |
| n = 45 | n = 103 | n = 111 | n = 19 | |||||
| DAB2IP rs7025486 | ||||||||
| GG | 34 (75.6) | 70 (68.0) | 1.457 | <0.001 | 77 (69.4) | 16 (84.2) | 2.35 | <0.001 |
| GA + AA | 11 (24.4) | 33 (32.0) | [0.66–3.23] | 34 (30.6) | 3 (15.8) | [0.64–8.62] | ||
| LRP1 rs1466535 | ||||||||
| CC | 35 (77.8) | 72 (69.9) | 1.507 | <0.001 | 81 (73.0) | 15 (78.9) | 1.389 | <0.001 |
| CT + TT | 10 (22.2) | 31 (30.1) | [0.66–3.42] | 30 (27.0) | 4 (21.1) | [0.43–4.52] | ||
| CDKN2BAS rs10757278 | ||||||||
| AA | 24 (53.3) | 59 (57.3) | 0.852 | <0.001 | 62 (55.9) | 10 (52.6) | 0.878 | <0.001 |
| AG + GG | 21 (46.7) | 44 (42.7) | [0.42–1.72] | 49 (44.1) | 9 (47.4) | [0.33–2.33] | ||
| IL6R rs2228145 | ||||||||
| AA | 41 (91.1) | 88 (85.4) | 1.747 | <0.001 | 96 (86.5) | 16 (84.2) | 0.833 | <0.001 |
| AC + CC | 4 (8.9) | 15 (14.6) | [0.55–5.59] | 15 (13.5) | 3 (15.8) | [0.22–3.21] | ||
| LPA rs3798220 | ||||||||
| TT | 45 (100) | 99 (96.1) | 0.961 | 0.180 | 107 (96.4) | 19 (100) | 0.964 | 0.401 |
| TC + CC | 0 (0) | 4 (3.9) | [0.92–1.00] | 4 (3.6) | 0 (0) | [0.93–1.00] | ||
| SORT1 rs599839 | ||||||||
| AA | 35 (77.8) | 83 (80.6) | 0.843 | <0.001 | 86 (77.5) | 16 (84.2) | 1.550 | <0.001 |
| AG + GG | 10 (22.2) | 20 (19.4) | [0.36–1.98] | 25 (22.5) | 3 (15.8) | [0.42–5.75] | ||
| SNPs | Location of AAA | |||||||
| Supra/Juxtarenal n (%) | Infrarenal n (%) | OR [95% CI] | p-Value * | |||||
| n = 53 | n = 95 | |||||||
| DAB2IP rs7025486 | ||||||||
| GG | 37 (69.8) | 67 (70.5) | 1.035 | 0.317 | ||||
| GA + AA | 16 (30.2) | 28 (29.5) | [0.50–2.15] | |||||
| LRP1 rs1466535 | ||||||||
| CC | 34 (64.2) | 73 (76.8) | 1.854 | 0.138 | ||||
| CT + TT | 19 (35.8) | 22 (23.2) | [0.89–3.87] | |||||
| CDKN2BAS rs10757278 | ||||||||
| AA | 28 (52.8) | 55 (57.9) | 1.228 | 0.118 | ||||
| AG + GG | 25 (47.2) | 40 (42.1) | [0.62–2.41] | |||||
| IL6R rs2228145 | ||||||||
| AA | 47 (88.7) | 82 (86.3) | 0.805 | <0.001 | ||||
| AC + CC | 6 (11.3) | 13 (13.7) | [0.29–2.26] | |||||
| LPA rs3798220 | ||||||||
| TT | 51 (96.2) | 93 (97.9) | 1.824 | <0.001 | ||||
| TC + CC | 2 (3.8) | 2 (2.1) | [0.25–13.33] | |||||
| SORT1 rs599839 | ||||||||
| AA | 43 (81.1) | 75 (78.9) | 0.872 | 0.005 | ||||
| AG + GG | 10 (18.9) | 20 (21.1) | [0.37–2.03] | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nugroho, N.T.; Herten, M.; Torsello, G.F.; Osada, N.; Marchiori, E.; Sielker, S.; Torsello, G.B. Association of Genetic Polymorphisms with Abdominal Aortic Aneurysm in the Processes of Apoptosis, Inflammation, and Cholesterol Metabolism. Medicina 2023, 59, 1844. https://doi.org/10.3390/medicina59101844
Nugroho NT, Herten M, Torsello GF, Osada N, Marchiori E, Sielker S, Torsello GB. Association of Genetic Polymorphisms with Abdominal Aortic Aneurysm in the Processes of Apoptosis, Inflammation, and Cholesterol Metabolism. Medicina. 2023; 59(10):1844. https://doi.org/10.3390/medicina59101844
Chicago/Turabian StyleNugroho, Nyityasmono Tri, Monika Herten, Giovanni F. Torsello, Nani Osada, Elena Marchiori, Sonja Sielker, and Giovanni B. Torsello. 2023. "Association of Genetic Polymorphisms with Abdominal Aortic Aneurysm in the Processes of Apoptosis, Inflammation, and Cholesterol Metabolism" Medicina 59, no. 10: 1844. https://doi.org/10.3390/medicina59101844
APA StyleNugroho, N. T., Herten, M., Torsello, G. F., Osada, N., Marchiori, E., Sielker, S., & Torsello, G. B. (2023). Association of Genetic Polymorphisms with Abdominal Aortic Aneurysm in the Processes of Apoptosis, Inflammation, and Cholesterol Metabolism. Medicina, 59(10), 1844. https://doi.org/10.3390/medicina59101844








