Preparing for Pregnancy in Women with Systemic Lupus Erythematosus—A Multidisciplinary Approach
Abstract
Highlights
- Unified, practical approach for preconception counselling of SLE women.
- What can be learned by the rheumatologist: the need for early referral for preconception counselling, risk stratification including from obstetrical point of view (preeclampsia (PE), anaesthetic evaluation), in vitro fertilisation, allowed medication during pregnancy (SLE-specific or non-specific), the need to evaluate for other autoimmune conditions includ-ing for thyroid disease.
- What can be learned by the obstetrician: how to assess activity and damage in SLE including scoring, autoantibody evaluation, and allowed vaccination from the SLE point of view.
Abstract
1. Introduction
2. Methods
3. Results
3.1. Pre-Pregnancy Counselling from the Patient’s Perspective: When, Who, What, and Why
3.2. Pre-Pregnancy Counselling from the Doctor’s Perspective: When Is the Right Time for a SLE Patient to Consider a Future Pregnancy?
3.2.1. Disease Activity and Pregnancy Planning
3.2.2. Treatment Regimens for Pre-Pregnancy and Pregnancy
3.2.3. Autoantibodies and Pregnancy Complications and Foetal/Neonatal Abnormalities
3.2.4. Vaccination
3.2.5. The Surveillance Team and Care Pathway
3.2.6. Anaesthetic Considerations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ostensen, M.; Clowse, M. Pathogenesis of preganancy complication in systemic lupus erythematosus. Curr. Opin. Rheumatol. 2013, 25, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Chighizola, G.B.; Lonati, P.A.; Meroni, P.L.; Tedesco, F. The complement system in the pathophysiology of pregnancy and in Systemic Autoimmune Rheumatic Diseases during pregnancy. Front. Immunol. 2020, 11, 2084. [Google Scholar] [CrossRef] [PubMed]
- Mor, G.; Cardenas, I. The immune system in pregnancy—A unique complexity. Am. J. Reprod. Immunol. 2010, 63, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Gluohovschi, C.; Gluhovschi, G.; Petrica, L.; Velciov, S.; Gluhovschi, A. Pregnancy associated with Systemic Lupus Erythematosus: Immune tolerance in pregnancy and its deficiency in Systemic Lupus Erythematosus—An immunological dilemma. J. Immunol. Res. 2015, 2015, 241547. [Google Scholar] [CrossRef]
- Andreoli, L.; Bertsias, G.K.; Agmon-Levin, N.; Brown, S.; Cervera, R.; Costedoat-Chalumeau, N.; Doria, A.; Fischer-Betz, R.; Forger, F.; Moraes-Fontes, M.F.; et al. EULAR recommandation for women health and the management of fammily planning, assisted reproduction, pregnancy and menoupause in patients with systemic lupus erythematosus and/or antiphospholipid syndrome. Ann. Rheum. Dis. 2017, 76, 476–485. [Google Scholar] [CrossRef]
- Andreoli, L.; Gerardi, M.C.; Fernandes, M. Disease activity assessment of rheumatic diseases during pregnancy: A comprehensive review of indices used in clinical studies. Autoimmun. Rev. 2019, 18, 164–176. [Google Scholar] [CrossRef]
- Bermas, B.L.; Sammaritano, L.R. Fertility and pregnancy in rheumatoid arthritis and systemic lupus erythematosus. Fertil. Res. Pract. 2015, 1, 13. [Google Scholar] [CrossRef]
- Clowse, M.E.; Chakravarty, E.; Costenbader, K.H. Effects of infertility, pregnancy loss and patient concerns on family size of women with rheumatoid arthritis and systemic lupus erythematosus. Arthritis Care Res. 2012, 64, 668–674. [Google Scholar] [CrossRef]
- Stephenson, J.; Vogel, C.; Hall, J.; Hutchinson, J.; Mann, S.; Duncan, H.; Woods-Townsend, K.; de Lusignan, S.; Poston, L.; Cade, J.E.; et al. Preconception healthi in England: A proposal for annual reporting with core metrics. Lancet 2019, 393, 2262–2267. [Google Scholar] [CrossRef]
- Chivers, B.; Boyle, J.; Lang, A.; Teede, H.J.; Moran, L.J.; Harrison, C.L. Preconception health and lifestyle behaiviours of women planning a pregnancy: A cross-sectional study. J. Clin. Med. 2020, 9, 1701. [Google Scholar] [CrossRef]
- Stephenson, J.; Heslehurst, N.; Hall, J.; Schoenaker, D.A.J.M.; Hutchinson, J.; Cade, J.E.; Poston, L.; Barrett, G.; Crozier, S.R.; Barker, M.; et al. Before the beginning: Nutrition and lifestyle in the preconception period and its importance for future health. Lancet 2018, 391, 1830–1841. [Google Scholar] [CrossRef]
- McDougall, B.; Kavanagh, K.; Stephenson, J.; Poston, L.; Flynn, A.C.; White, S.L. Health behaviours in 131,182 UK women planning pregnancy. BMC Pregnancy Childbirth 2021, 21, 530. [Google Scholar] [CrossRef] [PubMed]
- Aoki, S.; Yamamoto, Y. Systemic Lupus Erythematosus: Strategis to improve pregnancy outcomes. Int. J. Women Health 2016, 8, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Keeling, S.O.; Alabdurubalnabi, Z.; Avina-Zubieta, A.; Barr, S.; Bergeron, L.; Bernatsky, S.; Bourre-Tessier, J.; Clarke, A.; Baril-Dionne, A.; Dutz, J.; et al. Canadian Rheumatology Association recommendations for the assessment and monitoring of Systemic Lupus Erythematosus. J. Rheum. 2018, 45, 1426–1439. [Google Scholar] [CrossRef] [PubMed]
- Sammaritano, L.R.; Bermas, B.L.; Chakravarty, E.E.; Chambers, C.D.; Clowse, M.E.B.; Lockshin, M.D.; Marder, W.; Guyatt, G.; Branch, D.W.; Buyon, J.; et al. American College of Rheumatology guideline for the management of reproductive health in rheumatic and musculoskeletal diseases. Arthritis Rheumatol. 2020, 72, 529–556. [Google Scholar] [CrossRef]
- Blomjous, B.S.; de Vries, J.; Zijlstra, E.; Cramer, K.; Voskuyl, A.E.; Bultink, A.I.E.M. Desire to have children and preferences regarding to pre-pregnancy counselling in women with SLE. Rheumatology 2021, 60, 2706–2713. [Google Scholar] [CrossRef]
- Rodrigues, L.; Alves, V.L.P.; Sim-Simc, M.M.F.; Surita, F.G. Perception of women with systemic lupus erythematosus undergoing high-risk prenatal care: A qualitative study. Midwifery 2020, 87, 102715. [Google Scholar] [CrossRef]
- Giambalvo, S.; Garaffoni, C.; Silvagni, E.; Furini, F.; Rizzo, R.; Govoni, M.; Bortoluzzi, A. Factors associated with fertility abnormalities in women with systemic lupus erythematosus: A systematic review and meta-analysis. Autoimmun. Rev. 2022, 21, 103038. [Google Scholar] [CrossRef]
- Ostensen, M. New insights into sexual functioning and fertility in rheumatic diseases. Best Pract. Res. Clin. Rheumatol. 2004, 18, 219–232. [Google Scholar] [CrossRef]
- Cigni, A.; Faedda, R.; Atzeni, M.M.; Pileri, P.V.; Alagna, S.; Rovasio, P.; Satta, A.E.; Loi, M.R.; Sini, A.; Satta, V.; et al. Hormonal strategies for fertility preservation in patients receiving Cyclophosphamide to treat glomerulonephritis: A nonrandomized trial and review of the literature. Am. J. Kidney Dis. 2008, 52, 887–896. [Google Scholar] [CrossRef]
- Lateef, A.; Petri, M. Managing lupus patients during pregnancy. Best Pract. Res. Clin. Rheumatol. 2013, 27, 435–447. [Google Scholar] [CrossRef]
- Mehta, B.; Luo, Y.; Xu, J.; Sammaritano, L.; Salmon, J.; Lockshin, M.; Goodman, S.; Ibrahim, S. Trends in maternal and fetal outcomes among pregnant women with Systemic Lupus Erythematosus in the United States. Ann. Int. Med. 2019, 171, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, F.; Pirone, C.; Perricone, C.; Selntigia, A.; Orefice, V.; Pacucci, V.A.; Truglia, S.; Spinelli, F.R.; Galoppi, P.; Alessandri, C.; et al. Pregnancy outcome in systemic lupus erythematosus patients: A monocentric cohort analysis. Rheumatology 2021, 60, 1747–1754. [Google Scholar] [CrossRef] [PubMed]
- Yuen, S.Y.; Krizova, A.; Oulmet, J.M.; Pope, J.E. Pregnancy outcome in systemic lupus erythematosus (SLE) is improving: Results from a case control study and literature review. Open Rheumatol. J. 2009, 2, 89–98. [Google Scholar] [CrossRef]
- Teng, Y.K.O.; Bredewold, E.O.W.; Rabelink, T.; Huizinga, T.W.J.; Eikenboom, J.; Limper, M.; Fritsch-Stork, R.D.; Bloemenkamp, K.W.M.; Sueters, M. Evidence based approach to pre-pregnancy counselling for patients with systemic lupus erythematosus. Rheumatology 2018, 57, 1707–1720. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, A.; Eudy, A.M.; Balevic, S.J.; Clowse, M.E.B. The importance of pregnancy planning in lupus pregnancies. Lupus 2021, 30, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Skorpen, C.G.; Lydersen, S.; Gilboe, I.-M.; Skomsvoll, J.F.; Salvesen, K.; Palm, Ø.; Koksvik, H.S.S.; Jakobsen, B.; Wallenius, M. Influence of disease activity and medications on off-spring birth-weight, pre-eclampsia and preterm birth in systemic lupus erythematosus: A population-based study. Ann. Rheum. Dis. 2018, 77, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Ku, M.; Guo, S.; Shang, W.; Li, Q.; Zeng, R.; Han, M.; Ge, S.; Xu, G. Pregnancy outcomes in Chinese patients with systemic lupus erythematosus (SLE): A retrospective study of 109 pregnancies. PLoS ONE 2016, 11, e0159364. [Google Scholar]
- Mokbel, A.; Geilan, A.; AboElgheit, S. Could women with systemic lupus erythematosus (SLE) have successful pregnancy outcomes? Prospective observational study. Egypt Rheumatol. 2013, 35, 133–139. [Google Scholar] [CrossRef]
- Yang, H.; Liu, H.; Xu, D.; Zhao, L.; Wang, Q.; Leng, X.; Zheng, W.; Zhang, F.; Tang, F.; Zhang, X. Pregnancy-related systemic lupus erythematosus: Clinical features, outcome and risk factors of disease flares, a case control study. PLoS ONE 2011, 20, 829–836. [Google Scholar] [CrossRef]
- Buyon, J.P.; Kim, M.Y.; Guerra, M.M. Predictors of pregnancy outcomes in patients with lupus: A cohort study. Ann. Intern. Med. 2015, 163, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Smyth, A.; Oliveira, G.H.; Lahr, B.D.; Bailey, K.R.; Norby, S.M.; Garovic, V.D. A systematic review and meta-analysis of pregnancy outcomes in patients with systemic lupus erythematosus and lupus nephritis. Clin. J. Am. Soc. Nephrol. 2010, 5, 2060–2068. [Google Scholar] [CrossRef] [PubMed]
- Gordon, C.; Amissah-Arthur, M.B.; Gayed, M.; Brown, S.; Bruce, I.N.; D’Cruz, D.; Empson, B.; Griffiths, B.; Jayne, D.; Khamashta, M.; et al. The British Society for Rheumatology guideline for the management of systemic lupus erythematosusin adults. Rheumatology 2018, 57, 14. [Google Scholar] [CrossRef] [PubMed]
- Pons-Estel, B.A.; Bonfa, E.; Soriano, E.S.; Cardiel, M.H.; Izcovich, A.; Popoff, F.; Criniti, J.M.; Vásquez, G.; Massardo, L.; Duarte, M.; et al. First Latin American clinical practice guidelines for the treatment of systemic lupus erythematosus: Latin American Group for the study of Lupus (GLADEL, Grupo Latino Americano de Estudio del lupus)—Pan American League of Associations of Rheumatology. Ann. Rheum. Dis. 2018, 77, 1549–1557. [Google Scholar] [CrossRef] [PubMed]
- Gladman, D.D.; Ibanez, D.; Urowitz, M.B. Systemic lupus erythematosus diseasea activity index 2000. J. Rheumatol. 2002, 29, 288. [Google Scholar]
- Tselios, K.; Gladman, D.D.; Urowitz, M.B. How can we define low disease activity in systemic lupus erythematosus? Semin. Arthritis Rheum. 2019, 48, 1035–1040. [Google Scholar] [CrossRef]
- Fanouriakis, A.; Kostopoulou, M.; Alluno, A.; Aringer, M.; Bajema, I.; Boletis, J.N.; Boletis, R.; Doria, A.; Gordon, C.; Govoni, M.; et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann. Rheum. Dis. 2019, 78, 736–745. [Google Scholar] [CrossRef]
- Buyon, J.; Kalunian, K.C.; Ramsey-Goldman, R.; Petri, M.A.; Lockshin, M.D.; Ruiz-Irastorza, G.; Khamashta, M. Assessing disease activity in SLE patients during pregnancy. Lupus 1999, 8, 677–684. [Google Scholar] [CrossRef]
- Franklyn, K.; Lau, W.C.S.; Navarra, S.V.; Louthrenoo, W.; Lateef, A.; Hamijoyo, L.; Wahono, C.S.; Le Chen, S.; Jin, O.; Morton, S.; et al. Definition and initial validation of a lupus low disease activity state (LLDAS). Ann. Rhem. Dis. 2016, 75, 1615. [Google Scholar] [CrossRef]
- Kim, J.-W.; Jung, J.-Y.; Kim, H.-A.; Yang, J.I.; Kwak, D.W.; Suh, C.-H. Lupus Low Activity State achievement is important for reducing adverse outcomes in pregnant patients with systemic lupus erythematosus. J. Rheum. 2021, 48, 5. [Google Scholar] [CrossRef]
- Gladman, D.D.; Goldsmith, C.H.; Urowitz, M.B.; Bacon, P.; Fortin, P.; Ginzler, E.; Gordon, C.; Hanly, J.G.; Isenberg, D.A.; Petri, M.; et al. The Systemic Lupus International Collalborating Clinics/American College of Rheumatology (SLICC/ACR) Damage Index for Systemic Lupus Erythematosus International Comparison. J. Rheumatol. 2000, 27, 373. [Google Scholar] [PubMed]
- Gaballa, H.A.; El-Shahawy, E.E.-D.; Atta, D.S.; Gerbash, E.F. Clinical and serological risk factors of systemic lupus erythematosus outcomes during pregnancy. Egypt Rheumatol. 2012, 34, 159–165. [Google Scholar] [CrossRef]
- Tedeschi, S.K.; Massarotti, E.; Guan, H.; Fine, A.; Bermas, B.L.; Costenbader, K.H. Specific systemic lupus erythematosus disease manifestations in the six months prior to conception are associated with similar disease manifestations during pregnancy. Lupus 2015, 24, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Irastorza, G.; Khamashta, M. Lupus and pregnancy: Ten questions and some answers. Lupus 2008, 17, 416–420. [Google Scholar] [CrossRef] [PubMed]
- de Jesus, G.R.; Mendoza-Pinto, C.; de Jesus, N.R.; Santos, F.C.D.; Klumb, E.M.; Carrasco, M.G.; Levy, R.A. Understanding and managing pregnancy in patients with lupus. Autoimmune Dis. 2015, 2015, 943490. [Google Scholar] [CrossRef]
- Skorpen, C.G.; Hoeltzenbein, M.; Tincani, A.; Fischer-Betz, R.; Elefant, E.; Chambers, C.; da Silva, J.; Nelson-Piercy, C.; Cetin, I.; Costedoat-Chalumeau, N.; et al. The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Ann. Rheum. Dis. 2016, 75, 795–810. [Google Scholar] [CrossRef]
- Flint, J.; Panchal, S.; Hurrell, A.; Van De Venne, M.; Gayed, M.; Schreiber, K.; Arthanari, S.; Cunningham, J.; Flanders, L.; Moore, L.; et al. BSR and BHPR guideline on prescribing drugs in pregnancy and breastfeeding—Part II: Analgesics and other drugs used in rheumatology practice. Rheumatology 2016, 55, 1698–1702. [Google Scholar] [CrossRef]
- Wind, M.; Hendriks, M.; van Brussel, B.T.J.; Eikenboom, J.; Allaart, C.F.; Lamb, H.J.; Siebelink, H.-M.J.; Ninaber, M.K.; van Geloven, N.; van Lith, J.M.M.; et al. Effectiveness of a multidisciplinary clinical pathaway for women with systemic lupus erythematosus and/or antiphospholipid syndrome. Lupus Sci. Med. 2021, 8, e000472. [Google Scholar] [CrossRef]
- Mendel, A.; Bernatsky, S.B.; Hanly, J.G.; Urowitz, M.; Clarke, A.E.; Romero-Diaz, J.; Gordon, C.; Bae, S.-C.; Wallace, D.J.; Merrill, J.T.; et al. Low Aspirin use and high prevalence of pre-eclampsia risk factors among pregnant women in a multinational SLE inception cohort. Ann. Rheum. Dis. 2019, 78, 1010–1012. [Google Scholar] [CrossRef]
- Fox, R.; Kitt, J.; Leeson, P.; Aye, C.Y.; Lewandowski, A.J. Preeclampsia: Risk factors, diagnosis, management, and the cardiovascular impact on the offspring. J. Clin. Med. 2019, 8, 1625. [Google Scholar] [CrossRef]
- Bujold, E.; Roberge, S.; Lacasse, Y. Prevention of preeclampsia and intrauterine groth restriction with aspirin started in early pregnancy: A metaanalysis. Obstet. Gynecol. 2010, 116, 402–414. [Google Scholar] [CrossRef] [PubMed]
- Tani, C.; Zucchi, D.; Haase, I.; Gerosa, M.; Larosa, M.; Cavagna, L.; Bortoluzzi, A.; Crisafulli, F.; Mucke, J.; Strigini, F.A.L.; et al. Impact of low-dose acetylsalicylic acid on pregnancy outcome in systemic lupus erythematosus: Results from a multicentre study. Lupus Sci. Med. 2022, 9, e000714. [Google Scholar] [CrossRef] [PubMed]
- Haase, I.; Schneider, M.; Brinks, R. Low dose aspirin to prevent pre-eclampsia in SLE pregnancies—Counselling helps to realize our full potential (abstract). Arthris Rheumatol. 2020, 72, 110. [Google Scholar]
- Ross, A.C.; Manson, J.E.; Abram, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: What clinicians need to know? J. Clin. Endocrinol. Metab. 2011, 96, 53. [Google Scholar] [CrossRef] [PubMed]
- Pludowski, P.; Holick, M.F.; Grant, W.B.; Konstantynowicz, J.; Mascarenhas, M.S.; Haq, A.; Povoroznyuk, V.; Balatska, N.; Barbosa, A.P.; Karonova, T.; et al. Vitamin D supplementation guidelines. J. Steroid Biochem. Mol. Biol. 2018, 175, 125–135. [Google Scholar] [CrossRef]
- Bibbins-Domingo, K.; Grossman, D.C.; Curry, S.J.; Davidson, K.W.; Epling, J.W., Jr.; García, F.A.; Kemper, A.R.; Krist, A.H.; Kurth, A.E.; Landefeld, C.S.; et al. Folic Acid supplementation for the prevention of neural tube defects: US preventive services task forcereccomandation statement. JAMA 2017, 317, 183–189. [Google Scholar]
- Morales-Martínez, F.A.; Salas-Castro, C.; García-Garza, M.R.; Valdés-Martínez, O.; García-Luna, S.M.; Garza-Elizondo, M.; Vidal-Gutiérrez, O.; Saldívar-Rodríguez, D.; Sordia-Hernández, L.H. Evaluation of the Ovarian Reserve in Women with Systemic Lupus Erythematosus. J. Fam. Reprod Health. 2021, 15, 38–44. [Google Scholar] [CrossRef]
- Orquevaux, P.; Masseau, A.; Le Guern, V.; Gayet, V.; Vauthier, D.; Guettrot-Imbert, G.; Huong, D.L.T.; Wechsler, B.; Morel, N.; Cacoub, P.; et al. In Vitro Fertilization in 37 Women with Systemic Lupus Erythematosus or Antiphospholipid Syndrome: A Series of 97 Procedures. J. Rheumatol. 2017, 44, 613–618. [Google Scholar] [CrossRef]
- Bouariu, A.; Gică, N.; Ciobanu, A.M.; Scutelnicu, A.M.; Popescu, M.R.; Panaitescu, A.M. The Potential Benefit of Hydroxychloroquine in Chronic Placental Inflammation of Unknown Etiology Associated with Adverse Pregnancy Outcomes. Healthcare 2022, 10, 168. [Google Scholar] [CrossRef]
- Hochberg, M.C. Updating the American College of Rheumatology revised criteria for tha classification of systemic lupus erythematosus. Arthritis Rheum. 1997, 40, 1725. [Google Scholar] [CrossRef]
- Petri, M.; Orbai, A.-M.; Alarcón, G.S.; Gordon, C.; Merrill, J.T.; Fortin, P.R.; Bruce, I.N.; Isenberg, D.; Wallace, D.J.; Nived, O.; et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 2012, 64, 2677–2686. [Google Scholar] [CrossRef] [PubMed]
- Aringer, M.; Costenbader, K.H.; Daikh, D.I.; Brinks, R.; Mosca, M.; Ramsey-Goldman, R.; Smolen, J.S.; Wofsy, D.; Boumpas, D.; Kamen, D.L.; et al. 2019 EULAR/ACR classification criteria for Systemic Lupus Erythematosus. Arthritis Rheumatol. 2019, 71, 1400–1412. [Google Scholar] [CrossRef] [PubMed]
- Rufatti, A.; Tonello, M.; Visentin, M.S.; Bontadi, A.; Hoxha, A.; De Carolis, S.; Botta, A.; Salvi, S.; Nuzzo, M.; Rovere-Querini, P.; et al. Risk factors for pregnancy failure in patients with antiphospholipid syndrome treated with conventional therapies: A multicentric, case control study. Rheumatology 2011, 50, 1684–1689. [Google Scholar] [CrossRef] [PubMed]
- Miyakis, S.; Lockshin, M.D.; Atsumi, T.; Branch, D.W.; Brey, R.L.; Cervera, V.; Derksen, R.H.; DE Groot, P.G.; Koike, T.; Meroni, P.L.; et al. International consensus statement on un update of the classification criteria for definite antiphospholipid syndrome (APS). J. Thromb. Haemost. 2006, 4, 295. [Google Scholar] [CrossRef]
- Brucato, A.; Frassi, M.; Franceschini, F.; Cimaz, R.; Faden, D.; Pisoni, M.P.; Muscara, M.; Vignati, G.; Stramba-Badiale, M.; Catelli, L.; et al. Risk of congenital complete heart block in newborns of mothers with anti-Ro/SSA antibody detected by counterimmuneoelectrophoresis: A prospective study of 100 women. Arthritis Rheum 2001, 44, 1832–1835. [Google Scholar] [CrossRef]
- Veduta, A.; Panaitescu, A.M.; Ciobanu, A.M.; Neculcea, D.; Popescu, M.R.; Peltecu, G.; Cavoretto, P. Treatment of Fetal Arrhythmias. J. Clin. Med. 2021, 10, 2510. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, A.M.; Dumitru, A.E.; Gica, N.; Botezatu, R.; Peltecu, G.; Panaitescu, A.M. Benefits and Risks of IgG Transplacental Transfer. Diagnostics 2020, 10, 583. [Google Scholar] [CrossRef]
- Spence, D.; Hornberger, L.; Hamilton, R.; Silverman, E.D. Increased risk of complete congenital heart block in infants born to women with hypotyroidism and anti-Ro and/or anti-La antibodies. J. Rheumatol. 2006, 33, 167–170. [Google Scholar]
- Stagnaro-Green, A.; Akhter, E.; Yim, C.; Davies, T.F.; Magder, L.; Petri, M. Tyroid disease in pregnant women with systemic lupus erythematosus: Increased preterm delivery. Lupus 2011, 20, 690–699. [Google Scholar] [CrossRef]
- Furer, V.; Rondaan, C.; Heijstek, M.W.; Agmon-Levin, N.; Van Assen, S.; Bijl, M.; Breedveld, F.C.; D’Amelio, R.; Dougados, M.; Kapetanovic, M.C.; et al. 2019 update of EULAR recommendations for vaccination in adult patients with autoimmune inflammatory rheumatic diseases. Ann. Rheum Dis. 2020, 79, 39–52. [Google Scholar] [CrossRef]
- Garg, M.; Mufti, M.; Palmore, T.; Hasni, S. Reccomendations and barriers to vaccination in systemic lupus erythematosus. Autoimmun. Rev. 2018, 17, 990–1001. [Google Scholar] [CrossRef] [PubMed]
- Landewe, R.B.; Kroon, F.P.B.; Alunno, A.; Najm, A.; Bijlsma, J.W.J.; Burmester, G.R.R.; Caporali, R.; Combe, B.; Conway, R.; Curtis, J.R.; et al. EULAR reccomendations for the management and vaccination of people with rheumatic and musculoskeletal diseases in the context of SARS-COV-2: The November 2021 update. Ann. Rheum. Dis 2022. epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Njagu, R.; Criscione-Schreiber, L.G.; Eudy, A.; Snyderman, A.; Clowse, M.E.B. Impact of a Multifaceted educational program to improve provider skills for lupus pregnancy planning and management: A mixed-methods approach. ACR Open Rheumatol. 2020, 2, 378–387. [Google Scholar] [CrossRef]
- Ben-Menachem, E. Review article: Systemic lupus erythematosus: A review for anesthesiologists. Anesth Analg. 2010, 111, 665–676. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, S.T.; Gantz, E.; Baluch, A.R.; Kaye, R.J.; Kaye, A.D. Anesthetic considerations for the patient with systemic lupus erythematosus. Middle East J. Anaesthesiol. 2012, 21, 483–492. [Google Scholar]
- Vyas, V.; Shukla, D.; Patil, S.; Mohite, S. Caesarean section in a case of systemic lupus erythematosus. Indian J. Anaesth. 2014, 58, 193–195. [Google Scholar]
- Scarpelli, D.G.; Mccoy, F.W.; Scott, J.K. Acute lupus erythematosus with laryngeal involvement. N Engl. J. Med. 1959, 261, 691–694. [Google Scholar] [CrossRef]
- Klemp, P.; Meyers, O.L.; Keyzer, C. Atlanto-axial subluxation in systemic lupus erythematosus: A case report. S. Afr. Med. J. 1977, 52, 331–332. [Google Scholar]
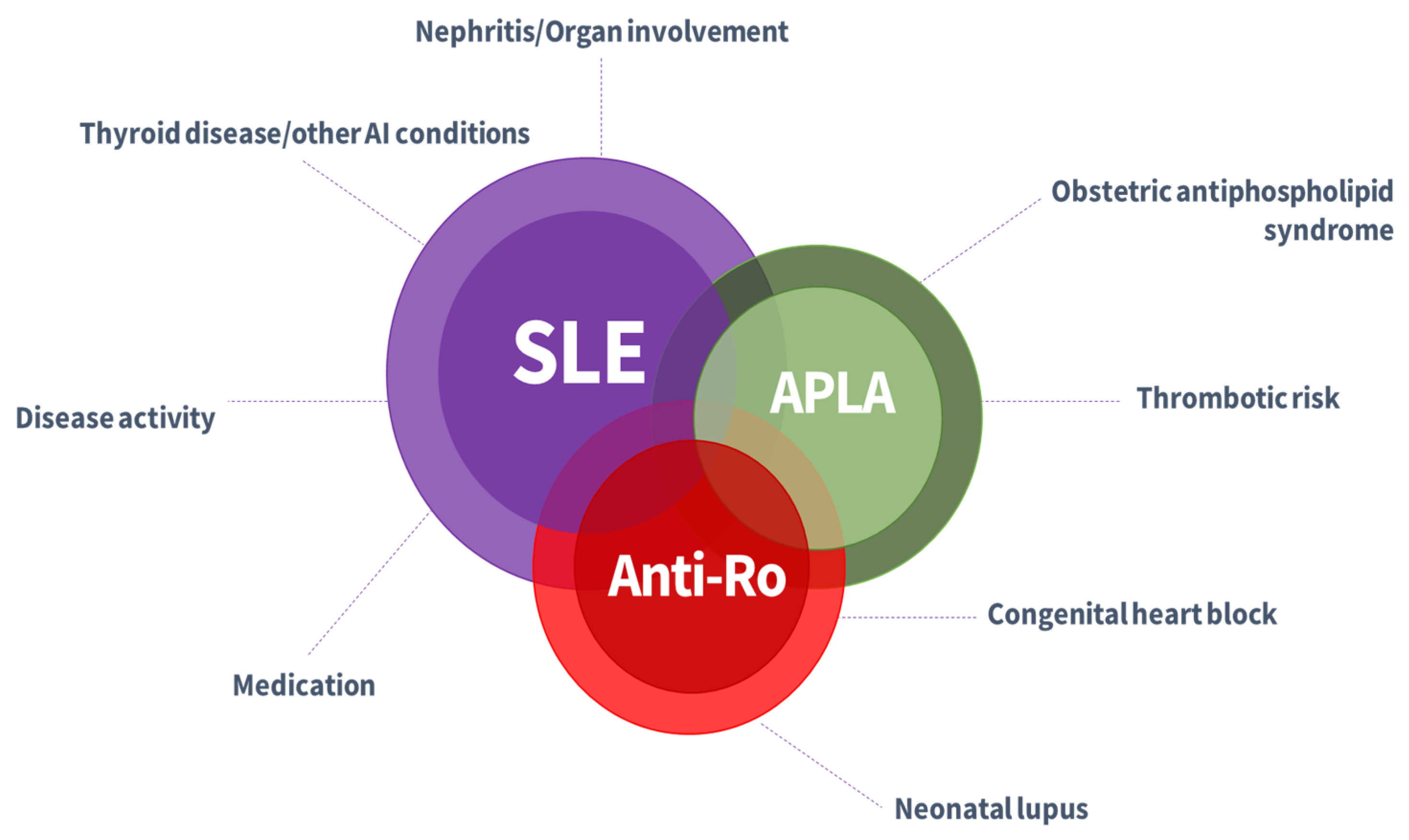
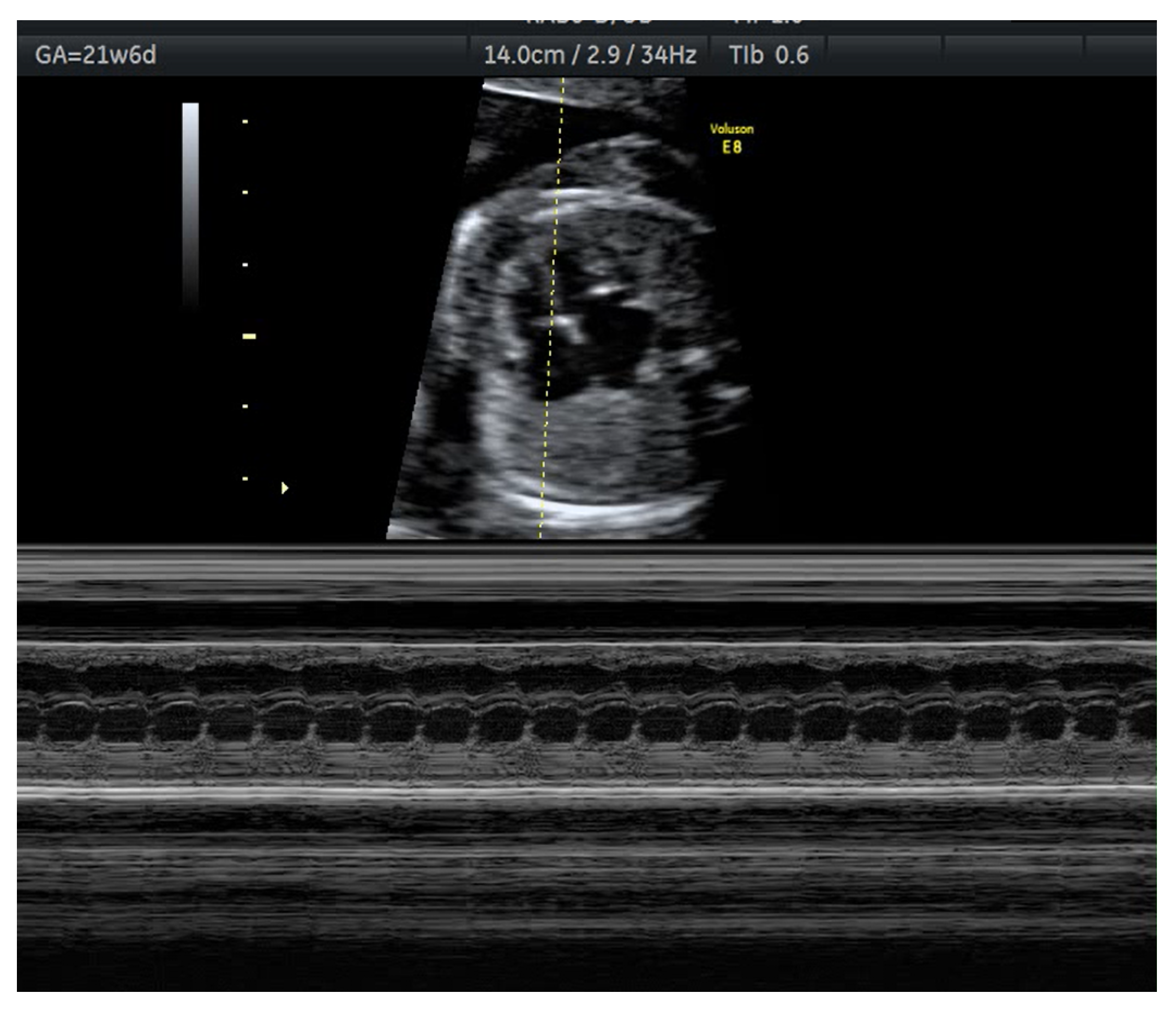
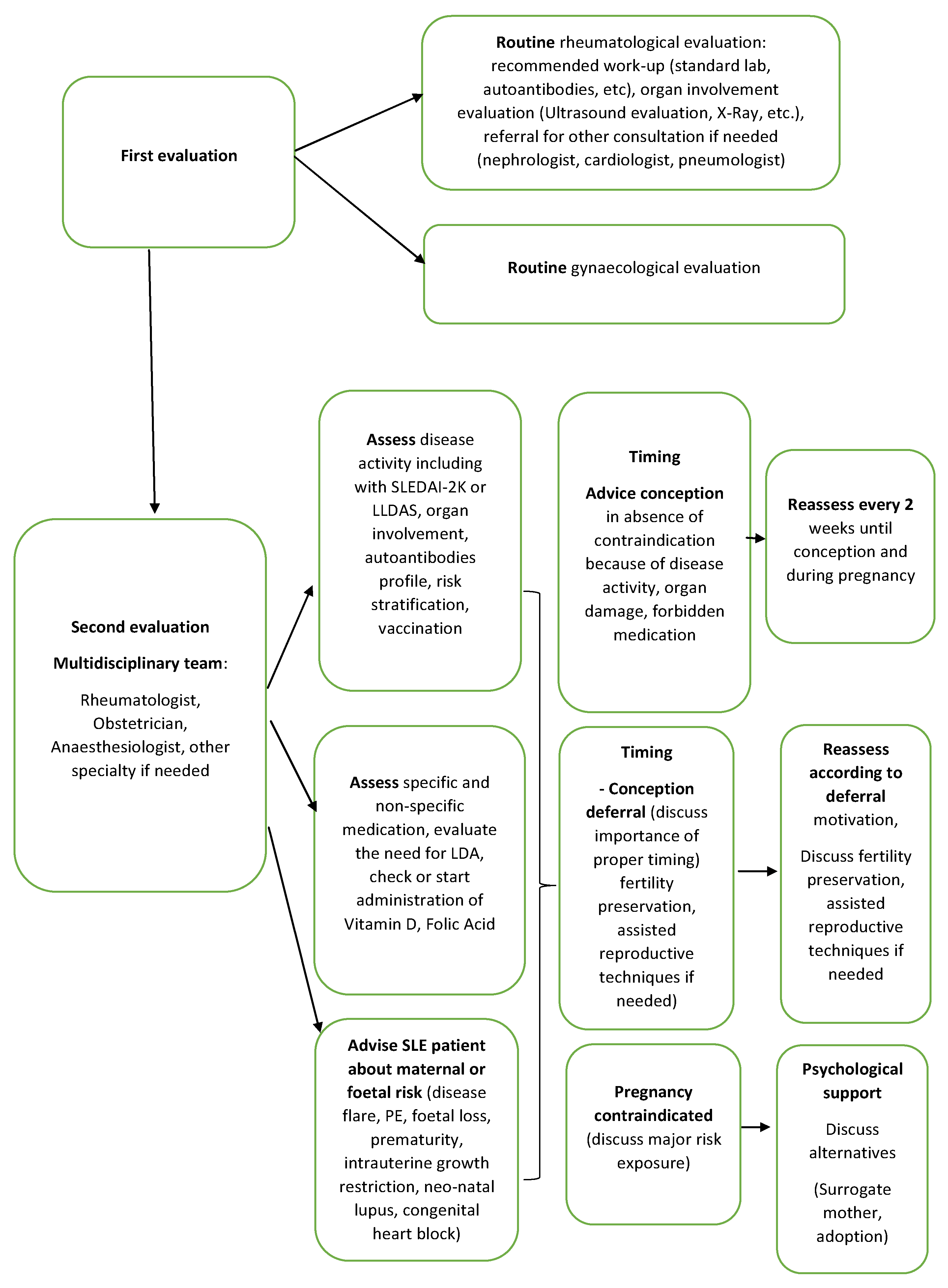
| Risk Factor | Increased Risk | References |
|---|---|---|
| SLE activity in the last 6–12 months or at conception | Flare during pregnancy Hypertension Foetal morbidity and mortality Preterm delivery Pregnancy loss | [5,30,31] |
| Lupus nephritis (anytime) | Renal flare during pregnancy Foetal loss Preterm delivery PE | [5,32] |
| Evaluation for Preconception Counselling |
|---|
| Clinical evaluation: anamnesis (important to find out if patient needed to increase or to add Prednisone or immunosuppressive therapy in the preceding 6 months for active disease) and complete physical exam |
| Lab evaluation: complete blood count, proteinuria, urinalysis, renal and hepatic function, glucose level, coagulation tests, inflammatory markers |
| Immune marker associated with active disease: low complement (C) level (C3, C4, C1q) and increased anti-double-strand DNA (anti-dsDNA) |
| Assessment of organ involvement with specific investigation (ultrasound, pulmonary function, imaging if required) and activity scores (e.g., SLEDAI-2K, LLDAS) |
| Check for existence of damage that might contraindicate pregnancy: pulmonary hypertension, low pulmonary function, cardiac failure, severe kidney failure, stroke or major thrombosis |
| Medication | Pre-Conception | References |
|---|---|---|
| Hydroxychloroquine | Recommended to all patients | [5,15,46] |
| Oral glucocorticoids | Accepted if needed at lowest effective dose, but less than 20 mg/day equivalent Prednisone | [5,15,46] |
| Azathioprine | Accepted if needed | [5,15,46] |
| Cyclosporin A and tacrolimus | Accepted if needed, caution if high blood pressure | [5,15,46] |
| Nonsteroidal antiinflamatory drugs | Accepted if needed, discontinue if there is problem with conceiving, cyclooxygenase 2 not indicated | [5,15,46] |
| Methotrexate | Stop 1–3 months prior to conception | [5,15,46] |
| Leflunomide | Stop if planning a pregnancy, washout with cholestyramine until no longer detected | [5,15,46] |
| Mycophenolate Mofetil and Mycophenolic Acid | Stop at least 6 weeks before conception to observe flare after discontinuation | [5,15,46] |
| Cyclophosphamide | Stop 3 months prior to conception | [5,15,46] |
| Available biologics (Belimumab, Rituximab) | Discontinue at conception | [15] |
| Recently approved biologic (Anifrolumab) | Not yet included in EULAR and ACR recommendations, no available data related to safety during pregnancy |
| High Risk for PE | Moderate Risk for PE |
|---|---|
| History of hypertension disease in a previous pregnancy | Nulliparous |
| Maternal disease: chronic kidney disease, autoimmune disease, diabetes, chronic hypertension | ≥40 years of age |
| Body mass index (BMI) ≥ 35 kg/m2 | |
| Family history of PE | |
| Multifetal pregnancy | |
| Pregnancy interval of more than 10 years |
| Autoantibodies Profile | Maternal Outcome | Foetal Outcome | References |
|---|---|---|---|
| High-risk APLA profile: persistent moderate or high APLA titres, LA, multiple APLA positivity | Maternal vascular thrombotic events, PE | APS related pregnancy morbidity (unexplained spontaneous pregnancy losses before 10 weeks of gestation, preterm delivery before 34 weeks of gestation because placental insufficiency, unexplained foetal death after 10 weeks of gestation) Intrauterine growth restriction, Pre-term birth | [5,21,31,50,64] |
| Anti-Ro/SSA and anti-La/SSB antibodies | Congenital heart block Lupus neonatal | [5,15,21,68] | |
| Antithyroid antibodies: tyroid peroxidase antibody tyroglobulin antibody tyroid-stimulating immunoglobulin antibody tyroid-stimulating hormone receptor binding inhibitor immunoglobulin | Preterm delivery Increased risk of complete congenital heart block in association with anti-Ro antibodies; foetal goitre; foetal hypo- or hyperthyroidism; foetal growth restriction | [5,68,69] |
| Complete clinical evaluation (anamnesis and physical exam) to observe for active disease or contraindication |
| Lab evaluation: complete blood count, proteinuria, urinalysis, renal and hepatic function, glucose level, coagulation tests, inflammatory markers, 25(OH)D in the presence of risk factor |
| Immune markers associated with active disease: low complement level (C3, C4, C1q) and increased anti dsDNA |
| Autoantibodies associated with maternal or foetal complications: APLA, anti-Ro, anti La antibodies, antithyroid antibodies |
| Collect scores to quantify activity: SLEDAI-2K and LLDAS |
| Check for existence of damage that might contraindicate pregnancy: pulmonary hypertension, low pulmonary function, cardiac failure, severe kidney failure, stroke, or major thrombosis |
| Check specific disease medications |
| Check comorbidities medication (antihypertensive), LDA, Vitamin D, Folic Acid |
| Check approved vaccination status |
| Evaluation | Deferred Conception | Contraindication for Pregnancy |
|---|---|---|
| Active disease in the last 6 months | ✓ | |
| Active lupus nephritis (still need for teratogenic regimen) | ✓ | |
| Severe pulmonary hypertension (>50 mmHg) | ✓ | |
| Severe restrictive lung disease (forced vital capacity < 1 L) | ✓ | |
| Advanced renal insufficiency (creatinine > 2.8 mg/dL) | ✓ | |
| Advanced heart failure | ✓ | |
| Stroke or major thrombotic event in the last 6 months | ✓ | |
| Previous PE or HELLP (Homolysis, Elevated Liver enzyme and Low Plates) syndrome despite correct treatment | ✓ | |
| Forbidden medication: changed first to approved medication and weight a few months to observe | ✓ | |
| Vaccination status: missing recommended vaccines | ✓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saulescu, I.C.; Opris-Belinski, D.; Balanescu, A.R.; Pavel, B.; Gica, N.; Panaitescu, A.M. Preparing for Pregnancy in Women with Systemic Lupus Erythematosus—A Multidisciplinary Approach. Medicina 2022, 58, 1371. https://doi.org/10.3390/medicina58101371
Saulescu IC, Opris-Belinski D, Balanescu AR, Pavel B, Gica N, Panaitescu AM. Preparing for Pregnancy in Women with Systemic Lupus Erythematosus—A Multidisciplinary Approach. Medicina. 2022; 58(10):1371. https://doi.org/10.3390/medicina58101371
Chicago/Turabian StyleSaulescu, Ioana Cristina, Daniela Opris-Belinski, Andra Rodica Balanescu, Bogdan Pavel, Nicolae Gica, and Anca Maria Panaitescu. 2022. "Preparing for Pregnancy in Women with Systemic Lupus Erythematosus—A Multidisciplinary Approach" Medicina 58, no. 10: 1371. https://doi.org/10.3390/medicina58101371
APA StyleSaulescu, I. C., Opris-Belinski, D., Balanescu, A. R., Pavel, B., Gica, N., & Panaitescu, A. M. (2022). Preparing for Pregnancy in Women with Systemic Lupus Erythematosus—A Multidisciplinary Approach. Medicina, 58(10), 1371. https://doi.org/10.3390/medicina58101371









