MiRNA-Based Inspired Approach in Diagnosis of Prostate Cancer
Abstract
1. Introduction
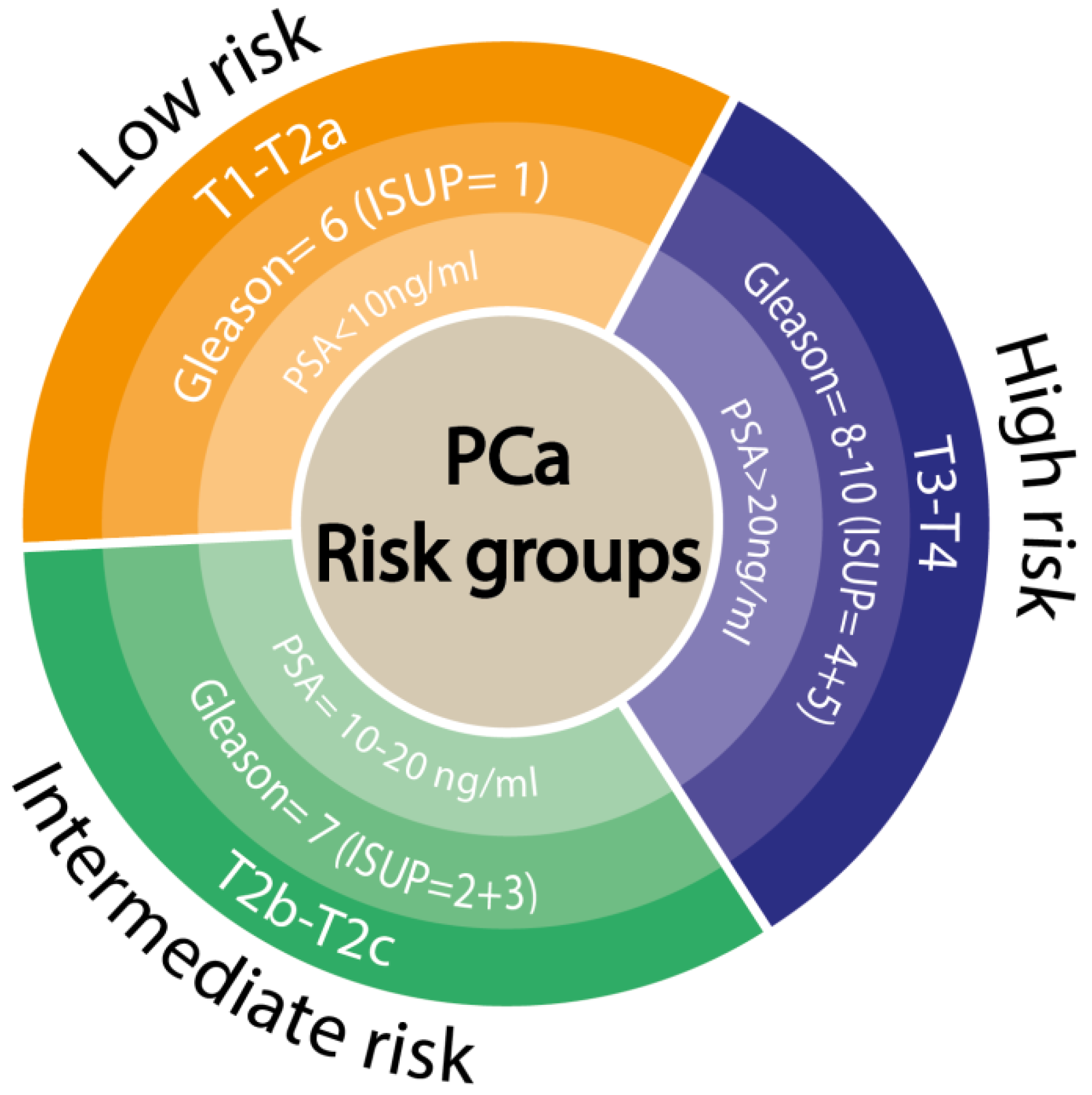
2. Current Clinical Point of View for Prostate Cancer Diagnosis
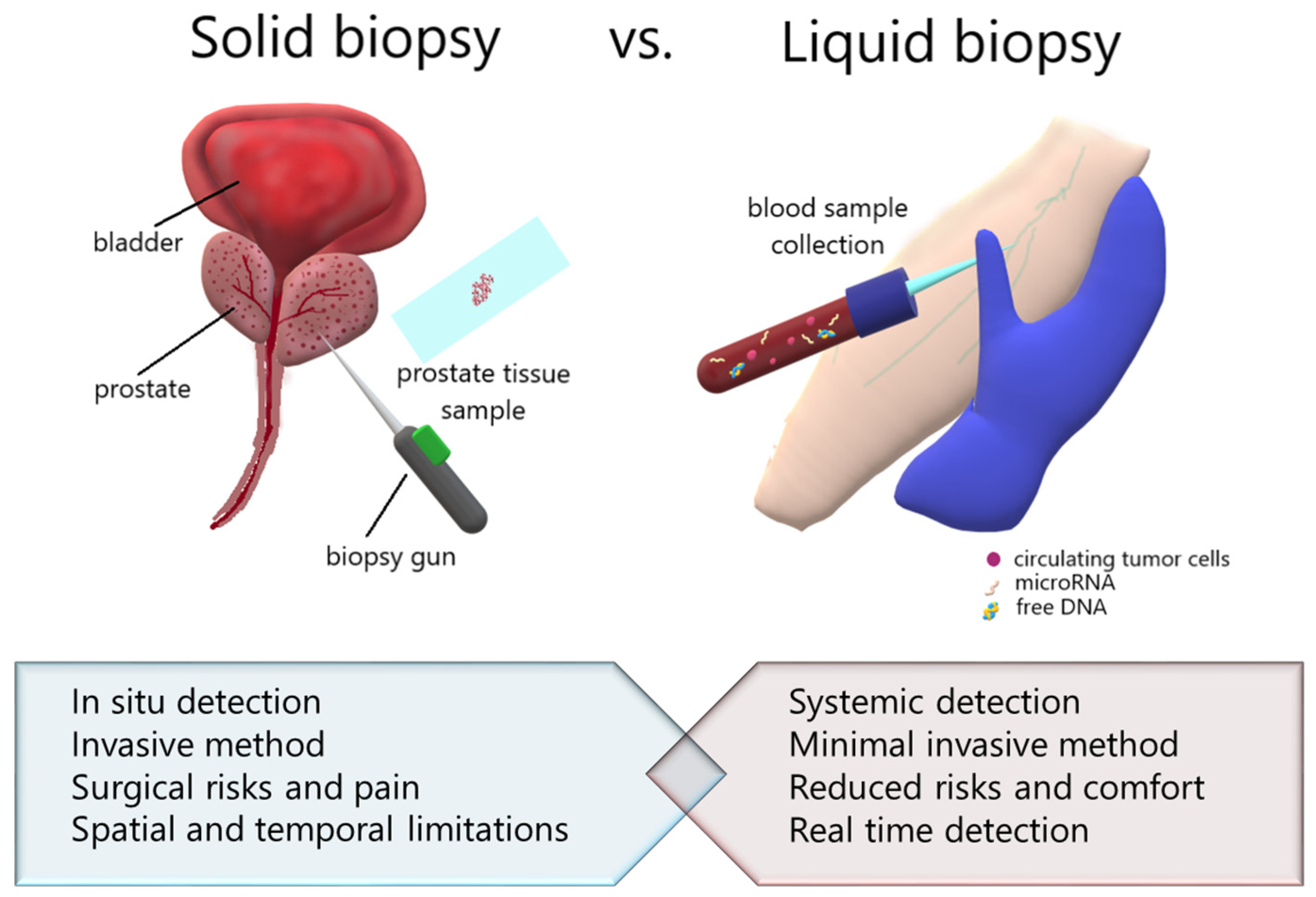
3. Liquid Biopsy vs. Tissue Biopsy—Transcriptional Point of View
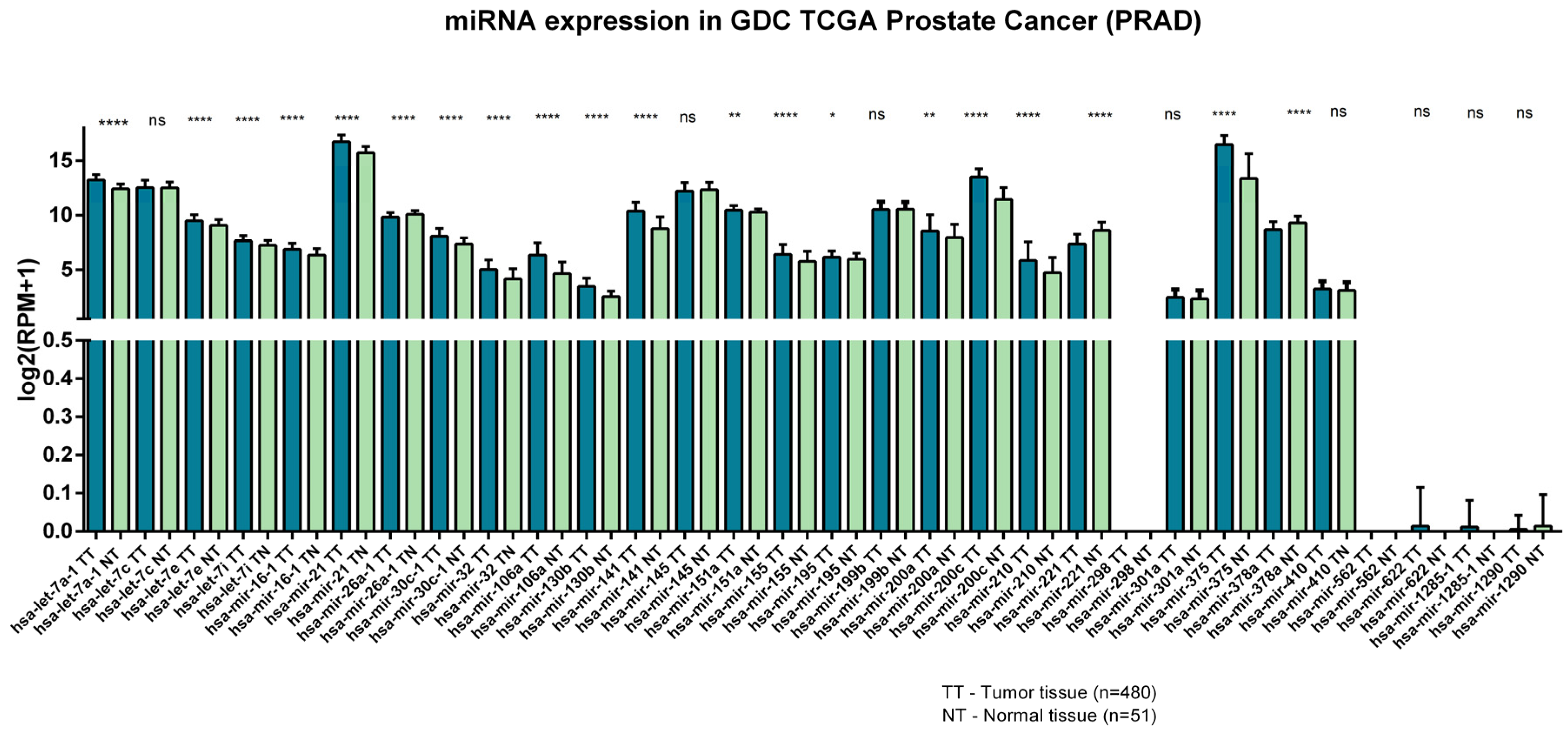
4. MiRNAs Signature in the Diagnosis of Prostate Cancer
5. MiRNAs Therapeutic Role in Prostate Cancer
6. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Patil, N.; Gaitonde, K. Clinical Perspective of Prostate Cancer. Top. Magn. Reson. Imag. 2016, 25, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.; Ma, J.; Zou, Z.; Jemal, A. Cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 9–29. [Google Scholar] [CrossRef] [PubMed]
- Taneja, S.; Shah, O. Complications of Urologic Surgery Book: Complications of Robotic Assisted Laparoscopic Radical Prostatectomy; Elsevier Health Sciences: Alpharetta, GA, USA, 2017. [Google Scholar]
- Sanda, M.G.; Dunn, R.L.; Michalski, J.; Sandler, H.M.; Northouse, L.; Hembroff, L.; Lin, X.; Greenfield, T.K.; Litwin, M.S.; Saigal, C.S.; et al. Quality of Life and Satisfaction with Outcome among Prostate-Cancer Survivors. N. Engl. J. Med. 2008, 358, 1250–1261. [Google Scholar] [CrossRef]
- Stelcer, E.; Konkol, M.; Głȩboka, A.; Suchorska, W.M. Liquid Biopsy in Oligometastatic Prostate Cancer—A Biologist’s Point of View. Front. Oncol. 2019, 9, 775. [Google Scholar] [CrossRef]
- Yin, C.; Fang, C.; Weng, H.; Yuan, C.; Wang, F. Circulating microRNAs as novel biomarkers in the diagnosis of prostate cancer: A systematic review and meta-analysis. Int. Urol. Nephrol. 2016, 48, 1087–1095. [Google Scholar] [CrossRef]
- McDonald, A.C.; Vira, M.; Shen, J.; Sanda, M.; Raman, J.D.; Liao, J.; Patil, D.; Taioli, E. Circulating microRNAs in plasma as potential biomarkers for the early detection of prostate cancer. Prostate 2018, 78, 411–418. [Google Scholar] [CrossRef]
- Wein, A.J.; Kavoussi, L.R.; Partin, A.W.; Peters, C.A. Diagnosis and Staging of Prostate Cancer. In Campbell-Walsh Urology, 11th ed.; Elsevier Health Sciences: Alpharetta, GA, USA, 2016. [Google Scholar]
- Naji, L.; Randhawa, H.; Sohani, Z.; Dennis, B.; Lautenbach, D.; Kavanagh, O.; Bawor, M.; Banfield, L.; Profetto, J. Digital Rectal Examination for Prostate Cancer Screening in Primary Care: A Systematic Review and Meta-Analysis. Ann. Fam. Med. 2018, 16, 149–154. [Google Scholar] [CrossRef]
- Smeenge, M.; Barentsz, J.; Cosgrove, D.; de la Rosette, J.; de Reijke, T.; Eggener, S.; Frauscher, F.; Kovacs, G.; Matin, S.F.; Mischi, M.; et al. Role of transrectal ultrasonography (TRUS) in focal therapy of prostate cancer: Report from a Consensus Panel. BJU Int. 2012, 110, 942–948. [Google Scholar] [CrossRef]
- Roobol, M.J.; Steyerberg, E.W.; Kranse, R.; Wolters, T.; van den Bergh, R.C.N.; Bangma, C.H.; Schröder, F.H. A Risk-Based Strategy Improves Prostate-Specific Antigen–Driven Detection of Prostate Cancer. Eur. Urol. 2010, 57, 79–85. [Google Scholar] [CrossRef]
- Epstein, J.I.; Feng, Z.; Trock, B.J.; Pierorazio, P.M. Upgrading and Downgrading of Prostate Cancer from Biopsy to Radical Prostatectomy: Incidence and Predictive Factors Using the Modified Gleason Grading System and Factoring in Tertiary Grades. Eur. Urol. 2012, 61, 1019–1024. [Google Scholar] [CrossRef]
- Borofsky, S.; George, A.K.; Gaur, S.; Bernardo, M.; Greer, M.D.; Mertan, F.V.; Taffel, M.; Moreno, V.; Merino, M.J.; Wood, B.J.; et al. What Are We Missing? False-Negative Cancers at Multiparametric MR Imaging of the Prostate. Radiology 2018, 286, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.L.; Kang, C.H.; Lee, W.C.; Chiang, P.H. Comparisons of cancer detection rate and complications between transrectal and transperineal prostate biopsy approaches—A single center preliminary study. BMC Urol. 2019, 19, 101. [Google Scholar] [CrossRef] [PubMed]
- McGrath, S.; Christidis, D.; Perera, M.; Hong, S.K.; Manning, T.; Vela, I.; Lawrentschuk, N. Prostate cancer biomarkers: Are we hitting the mark? Prostate Int. 2016, 4, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Berindan-Neagoe, I.; Calin, G.A. Molecular Pathways: MicroRNAs, Cancer Cells, and Microenvironment. Clin. Cancer Res. 2014, 20, 6247–6253. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Pop-Bica, C.; Gulei, D.; Cojocneanu-Petric, R.; Braicu, C.; Petrut, B.; Berindan-Neagoe, I. Understanding the Role of Non-Coding RNAs in Bladder Cancer: From Dark Matter to Valuable Therapeutic Targets. Int. J. Mol. Sci. 2017, 18, 1514. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Buiga, R.; Cojocneanu, R.; Buse, M.; Raduly, L.; Pop, L.A.; Chira, S.; Budisan, L.; Jurj, A.; Ciocan, C.; et al. Connecting the dots between different networks: MiRNAs associated with bladder cancer risk and progression. J. Exp. Clin. Cancer Res. 2019, 38, 433. [Google Scholar] [CrossRef]
- Cheng, H.H.; Mitchell, P.S.; Kroh, E.M.; Dowell, A.E.; Chéry, L.; Siddiqui, J.; Nelson, P.S.; Vessella, R.L.; Knudsen, B.S.; Chinnaiyan, A.M.; et al. Circulating microRNA Profiling Identifies a Subset of Metastatic Prostate Cancer Patients with Evidence of Cancer-Associated Hypoxia. PLoS ONE 2013, 8, e69239. [Google Scholar] [CrossRef]
- Best, M.G.; Sol, N.; Kooi, I.; Tannous, J.; Westerman, B.A.; Rustenburg, F.; Schellen, P.; Verschueren, H.; Post, E.; Koster, J.; et al. RNA-Seq of Tumor-Educated Platelets Enables Blood-Based Pan-Cancer, Multiclass, and Molecular Pathway Cancer Diagnostics. Cancer Cell 2015, 28, 666–676. [Google Scholar] [CrossRef]
- Hegemann, M.; Stenzl, A.; Bedke, J.; Chi, K.N.; Black, P.C.; Todenhöfer, T. Liquid biopsy: Ready to guide therapy in advanced prostate cancer? BJU Int. 2016, 118, 855–863. [Google Scholar] [CrossRef]
- Helsmoortel, H.; Everaert, C.; Lumen, N.; Ost, P.; Vandesompele, J. Detecting long non-coding RNA biomarkers in prostate cancer liquid biopsies: Hype or hope? Non-Cod. RNA Res. 2018, 3, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, A.; Koch, A.; Krockenberger, K.; Großhennig, A. Personalized medicine using DNA biomarkers: A review. Hum. Genet. 2012, 131, 1627–1638. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, D.T.; Lee, R.J.; Stott, S.L.; Ting, D.T.; Wittner, B.S.; Ulman, M.; Smas, M.E.; Lord, J.B.; Brannigan, B.W.; Trautwein, J.; et al. Androgen Receptor Signaling in Circulating Tumor Cells as a Marker of Hormonally Responsive Prostate Cancer. Cancer Discov. 2012, 2, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Bryant, R.J.; Pawlowski, T.; Catto, J.W.F.; Marsden, G.; Vessella, R.L.; Rhees, B.; Kuslich, C.; Visakorpi, T.; Hamdy, F.C. Changes in circulating microRNA levels associated with prostate cancer. Br. J. Cancer 2012, 106, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Ji, P.; Diederichs, S.; Wang, W.; Böing, S.; Metzger, R.; Schneider, P.M.; Tidow, N.; Brandt, B.; Buerger, H.; Bulk, E.; et al. MALAT-1, a novel noncoding RNA, and thymosin β4 predict metastasis and survival in early-stage non-small cell lung cancer. Oncogene 2003, 22, 8031–8041. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xu, X.; Lv, H.; Wen, Q.; Li, J.; Tan, L.; Li, J.; Sheng, X. The Long Noncoding RNA MALAT-1 Is Highly Expressed in Ovarian Cancer and Induces Cell Growth and Migration. PLoS ONE 2016, 11, e0155250. [Google Scholar] [CrossRef] [PubMed]
- Alix-Panabières, C.; Pantel, K. Challenges in circulating tumour cell research. Nat. Rev. Cancer 2014, 14, 623–631. [Google Scholar] [CrossRef]
- Reis, I.M.; Ramachandran, K.; Speer, C.; Gordian, E.; Singal, R. Serum GADD45a methylation is a useful biomarker to distinguish benign vs malignant prostate disease. Br. J. Cancer 2015, 113, 460–468. [Google Scholar] [CrossRef]
- Scher, H.I.; Lu, D.; Schreiber, N.A.; Louw, J.; Graf, R.P.; Vargas, H.A.; Johnson, A.; Jendrisak, A.; Bambury, R.; Danila, D.; et al. Association of AR-V7 on Circulating Tumor Cells as a Treatment-Specific Biomarker with Outcomes and Survival in Castration-Resistant Prostate Cancer. JAMA Oncol. 2016, 2, 1441. [Google Scholar] [CrossRef]
- Scher, H.I.; Graf, R.P.; Schreiber, N.A.; McLaughlin, B.; Lu, D.; Louw, J.; Danila, D.; Dugan, L.; Johnson, A.; Heller, G.; et al. Nuclear-specific AR-V7 Protein Localization is Necessary to Guide Treatment Selection in Metastatic Castration-resistant Prostate Cancer. Eur. Urol. 2017, 71, 874–882. [Google Scholar] [CrossRef]
- Clark, T.A.; Chung, J.H.; Kennedy, M.; Hughes, J.D.; Chennagiri, N.; Lieber, D.S.; Fendler, B.; Young, L.; Zhao, M.; Coyne, M.; et al. Analytical Validation of a Hybrid Capture–Based Next-Generation Sequencing Clinical Assay for Genomic Profiling of Cell-Free Circulating Tumor DNA. J. Mol. Diagn. 2018, 20, 686–702. [Google Scholar] [CrossRef]
- Hoffman, R.M. Implications of the New USPSTF Prostate Cancer Screening Recommendation—Attaining Equipoise. JAMA Int. Med. 2018, 178, 889. [Google Scholar] [CrossRef]
- Mortensen, M.M.; Høyer, S.; Ørntoft, T.F.; Sørensen, K.D.; Dyrskjøt, L.; Borre, M. High miR-449b expression in prostate cancer is associated with biochemical recurrence after radical prostatectomy. BMC Cancer 2014, 14, 859. [Google Scholar] [CrossRef]
- Fu, Y.; Cao, F. MicroRNA-125a-5p regulates cancer cell proliferation and migration through NAIF1 in prostate carcinoma. OncoTargets Ther. 2015, 8, 3827. [Google Scholar] [CrossRef]
- Qu, F.; Zheng, J.; Gan, W.; Lian, H.; He, H.; Li, W.; Yuan, T.; Yang, Y.; Li, X.; Ji, C.; et al. MiR-199a-3p suppresses proliferation and invasion of prostate cancer cells by targeting Smad1. Oncotarget 2017, 8, 52465–52473. [Google Scholar] [CrossRef]
- Fort, R.S.; Mathó, C.; Oliveira-Rizzo, C.; Garat, B.; Sotelo-Silveira, J.R.; Duhagon, M.A. An integrated view of the role of miR-130b/301b miRNA cluster in prostate cancer. Exp. Hematol. Oncol. 2018, 7, 10. [Google Scholar] [CrossRef]
- Huang, K.; Tang, Y.; He, L.; Dai, Y. MicroRNA-340 inhibits prostate cancer cell proliferation and metastasis by targeting the MDM2-p53 pathway. Oncol. Rep. 2016, 35, 887–895. [Google Scholar] [CrossRef]
- Ling, Z.; Liu, D.; Zhang, G.; Liang, Q.; Xiang, P.; Xu, Y.; Han, C.; Tao, T. miR-361-5p modulates metabolism and autophagy via the Sp1-mediated regulation of PKM2 in prostate cancer. Oncol. Rep. 2017, 38, 1621–1628. [Google Scholar] [CrossRef]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef]
- Gidlöf, O.; Andersson, P.; van der Pals, J.; Götberg, M.; Erlinge, D. Cardiospecific microRNA Plasma Levels Correlate with Troponin and Cardiac Function in Patients with ST Elevation Myocardial Infarction, Are Selectively Dependent on Renal Elimination, and Can Be Detected in Urine Samples. Cardiology 2011, 118, 217–226. [Google Scholar] [CrossRef]
- Wang, L.; Tang, H.; Thayanithy, V.; Subramanian, S.; Oberg, A.L.; Cunningham, J.M.; Cerhan, J.; Steer, C.; Thibodeanu, S.N. Gene Networks and microRNAs Implicated in Aggressive Prostate Cancer. Cancer Res. 2009, 69, 9490–9497. [Google Scholar] [CrossRef]
- Pashaei, E.; Pashaei, E.; Ahmady, M.; Ozen, M.; Aydin, N. Meta-analysis of miRNA expression profiles for prostate cancer recurrence following radical prostatectomy. PLoS ONE 2017, 12, e0179543. [Google Scholar] [CrossRef]
- Hu, J.; Li, S.; Yu, X. Tumor suppressor miR-1-3p regulates prostate cancer proliferation by controlling E2F5 and PFTK1 expression. Eur. Urol. Suppl. 2018, 17, e503. [Google Scholar] [CrossRef]
- Tang, Y.; Pan, J.; Huang, S.; Peng, X.; Zou, X.; Luo, Y.; Ren, D.; Zhang, X.; Li, R.; He, P.; et al. Downregulation of miR-133a-3p promotes prostate cancer bone metastasis via activating PI3K/AKT signaling. J. Exp. Clin. Cancer Res. 2018, 37, 160. [Google Scholar] [CrossRef]
- Li, D.; Xia, L.; Chen, M.; Lin, C.; Wu, H.; Zhang, Y.; Pan, S.; Li, X. miR-133b, a particular member of myomiRs, coming into playing its unique pathological role in human cancer. Oncotarget 2017, 8, 50193–50208. [Google Scholar] [CrossRef]
- Chen, W.; Liu, Y.; Chen, H.; Ning, H.; Ding, K. Loss of miR-449a-caused PrLZ overexpression promotes prostate cancer metastasis. Int. J. Oncol. 2017, 51, 435–444. [Google Scholar] [CrossRef]
- Nilsson, E.M.; Laursen, K.B.; Whitchurch, J.; McWilliam, A.; Ødum, N.; Persson, J.L.; Heey, D.; Gudas, L.; Mongan, N.P. MiR137 is an androgen regulated repressor of an extended network of transcriptional coregulators. Oncotarget 2015, 6, 35710–35725. [Google Scholar] [CrossRef]
- Bian, X.; Shen, Y.; Zhang, G.; Gu, C.; Cai, Y.; Wang, C.; Zhu, Y.; Zhu, P.; Zhang, H.; Dai, B.; et al. Expression of Dicer and Its Related MiRNAs in the Progression of Prostate Cancer. PLoS ONE 2015, 10, e0120159. [Google Scholar] [CrossRef]
- Kiener, M.; Chen, L.; Krebs, M.; Grosjean, J.; Klima, I.; Kalogirou, C.; Riedmiller, H.; Kneitz, B.; Thalmann, G.N.; Snaar-Jagarska, E.; et al. miR-221-5p regulates proliferation and migration in human prostate cancer cells and reduces tumor growth in vivo. BMC Cancer 2019, 19, 627. [Google Scholar] [CrossRef]
- Ninio-Many, L.; Grossman, H.; Levi, M.; Zilber, S.; Tsarfaty, I.; Shomron, N.; Tuvar, A.; Chuderland, D.; Stemmer, S.; Ben-Aharon, I.; et al. MicroRNA miR-125a-3p modulates molecular pathway of motility and migration in prostate cancer cells. Oncoscience 2014, 1, 250–261. [Google Scholar] [CrossRef]
- Qu, Y.; Huang, X.; Li, Z.; Liu, J.; Wu, J.; Chen, D.; Zao, F.; Mu, D. miR-199a-3p Inhibits Aurora Kinase A and Attenuates Prostate Cancer Growth. Am. J. Pathol. 2014, 184, 1541–1549. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lu, X.; Wu, B.; Su, Y.; Li, J.; Wang, H. MicroRNA 363 mediated positive regulation of c-myc translation affect prostate cancer development and progress. Neoplasma 2015, 62, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Ting, H.J.; Messing, J.; Yasmin-Karim, S.; Lee, Y.F. Identification of microRNA-98 as a Therapeutic Target Inhibiting Prostate Cancer Growth and a Biomarker Induced by Vitamin D. J. Biol. Chem. 2013, 288, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Brase, J.C.; Johannes, M.; Schlomm, T.; Fälth, M.; Haese, A.; Steuber, T.; Beissbarth, T.; Kuner, R.; Sultman, H. Circulating miRNAs are correlated with tumor progression in prostate cancer. Int. J. Cancer 2011, 128, 608–616. [Google Scholar] [CrossRef]
- Chen, Z.H.; Zhang, G.L.; Li, H.R.; Luo, J.D.; Li, Z.X.; Chen, G.M.; Yang, J. A panel of five circulating microRNAs as potential biomarkers for prostate cancer. Prostate 2012, 72, 1443–1452. [Google Scholar] [CrossRef]
- Nguyen, H.C.N.; Xie, W.; Yang, M.; Hsieh, C.L.; Drouin, S.; Lee, G.S.M.; Kantoff, P.W. Expression differences of circulating microRNAs in metastatic castration resistant prostate cancer and low-risk, localized prostate cancer. Prostate 2013, 73, 346–354. [Google Scholar] [CrossRef]
- Watahiki, A.; Macfarlane, R.; Gleave, M.; Crea, F.; Wang, Y.; Helgason, C.; Chi, K.N. Plasma miRNAs as Biomarkers to Identify Patients with Castration-Resistant Metastatic Prostate Cancer. Int. J. Mol. Sci. 2013, 14, 7757–7770. [Google Scholar] [CrossRef]
- Haldrup, C.; Kosaka, N.; Ochiya, T.; Borre, M.; Høyer, S.; Orntoft, T.F.; Sorensen, K.D. Profiling of circulating microRNAs for prostate cancer biomarker discovery. Drug Deliv. Trans. Res. 2014, 4, 19–30. [Google Scholar] [CrossRef]
- Huang, X.; Yuan, T.; Liang, M.; Du, M.; Xia, S.; Dittmar, R.; Wang, D.; See, W.; Costello, B.A.; Quevedo, F.; et al. Exosomal miR-1290 and miR-375 as Prognostic Markers in Castration-resistant Prostate Cancer. Eur. Urol. 2015, 67, 33–41. [Google Scholar] [CrossRef]
- Yaman Agaoglu, F.; Kovancilar, M.; Dizdar, Y.; Darendeliler, E.; Holdenrieder, S.; Dalay, N.; Gezer, U. Investigation of miR-21, miR-141, and miR-221 in blood circulation of patients with prostate cancer. Tumor Biol. 2011, 32, 583–588. [Google Scholar] [CrossRef]
- Mahn, R.; Heukamp, L.C.; Rogenhofer, S.; von Ruecker, A.; Müller, S.C.; Ellinger, J. Circulating microRNAs (miRNA) in Serum of Patients with Prostate Cancer. Urology 2011, 77, 1265.e9–1265.e16. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, J.C.; Fink, L.M.; Goodman, O.B.; Symanowski, J.T.; Vogelzang, N.J.; Ward, D.C. Comparison of Circulating MicroRNA 141 to Circulating Tumor Cells, Lactate Dehydrogenase, and Prostate-Specific Antigen for Determining Treatment Response in Patients With Metastatic Prostate Cancer. Clin. Genitourin. Cancer 2011, 9, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Selth, L.A.; Townley, S.; Gillis, J.L.; Ochnik, A.M.; Murti, K.; Macfarlane, R.J.; Chi, K.N.; Marshall, V.R.; Tilley, W.D.; Butler, L.M. Discovery of circulating microRNAs associated with human prostate cancer using a mouse model of disease. Int. J. Cancer 2012, 131, 652–661. [Google Scholar] [CrossRef] [PubMed]
- Mihelich, B.L.; Maranville, J.C.; Nolley, R.; Peehl, D.M.; Nonn, L. Elevated Serum MicroRNA Levels Associate with Absence of High-Grade Prostate Cancer in a Retrospective Cohort. PLoS ONE 2015, 10, e0124245. [Google Scholar] [CrossRef]
- Kelly, B.; Miller, N.; Sweeney, K.; Durkan, G.; Rogers, E.; Walsh, K.; Kerin, M.J. A Circulating MicroRNA Signature as a Biomarker for Prostate Cancer in a High Risk Group. J. Clin. Med. 2015, 4, 1369–1379. [Google Scholar] [CrossRef]
- Kachakova, D.; Mitkova, A.; Popov, E.; Popov, I.; Vlahova, A.; Dikov, T.; Christova, S.; Mitev, V.; Slavov, C.; Kaneva, R. Combinations of Serum Prostate-Specific Antigen and Plasma Expression Levels of let-7c, miR-30c, miR-141, and miR-375 as Potential Better Diagnostic Biomarkers for Prostate Cancer. DNA Cell Biol. 2015, 34, 189–200. [Google Scholar] [CrossRef]
- Huang, W.; Kang, X.L.; Cen, S.; Wang, Y.; Chen, X. High-Level Expression of microRNA-21 in Peripheral Blood Mononuclear Cells Is a Diagnostic and Prognostic Marker in Prostate Cancer. Genet. Test. Mol. Biomarkers 2015, 19, 469–475. [Google Scholar] [CrossRef]
- Sharova, E.; Grassi, A.; Marcer, A.; Ruggero, K.; Pinto, F.; Bassi, P.; Zanovello, P.; Zattoni, F.; D’Agostino, D.M.; Yafrate, M.; et al. A circulating miRNA assay as a first-line test for prostate cancer screening. Br. J. Cancer 2016, 114, 1362–1366. [Google Scholar] [CrossRef]
- Westermann, A.M.; Schmidt, D.; Holdenrieder, S.; Moritz, R.; Semjonow, A.; Schmidt, M.; Kristiansen, G.; Muller, S.C.; Ellinger, J. Serum microRNAs as biomarkers in patients undergoing prostate biopsy: Results from a prospective multi-center study. Anticancer Res. 2014, 34, 665–669. [Google Scholar]
- Wang, J.; Ye, H.; Zhang, D.; Hu, Y.; Yu, X.; Wang, L.; Zuo, C.; Yu, Y.; Xu, G.; Liu, S. MicroRNA-410-5p as a potential serum biomarker for the diagnosis of prostate cancer. Cancer Cell Int. 2016, 16, 12. [Google Scholar] [CrossRef]
- Chen, H.; Liu, H.; Zou, H.; Chen, R.; Dou, Y.; Sheng, S.; Dai, S.; Ai, J.; Melson, J.; Kittles, R.A.; et al. Evaluation of Plasma miR-21 and miR-152 as Diagnostic Biomarkers for Common Types of Human Cancers. J. Cancer 2016, 7, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.K.; Li, Z.; Ma, Y.Y.; Wang, J.; Zeng, X.F.; Li, R.; Kang, W.; Hao, X.K. Exosomal microRNA-141 is upregulated in the serum of prostate cancer patients. Onco Targets Ther. 2015, 9, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Kolluru, V.; Chandrasekaran, B.; Tyagi, A.; Dervishi, A.; Ankem, M.; Yan, X.; Maiying, K.; Alatassi, H.; Shaheen, S.P.; Messer, J.C.; et al. miR-301a expression: Diagnostic and prognostic marker for prostate cancer. Urol. Oncol. 2018, 36, 503.e9–503.e15. [Google Scholar] [CrossRef] [PubMed]
- Matin, F.; Jeet, V.; Moya, L.; Selth, L.A.; Chambers, S.; Clements, J.A.; Batra, J. A Plasma Biomarker Panel of Four MicroRNAs for the Diagnosis of Prostate Cancer. Sci. Rep. 2018, 8, 6653. [Google Scholar] [CrossRef]
- Urabe, F.; Matsuzaki, J.; Yamamoto, Y.; Kimura, T.; Hara, T.; Ichikawa, M.; Takizawa, S.; Aoki, Y.; Niida, S.; Sakamoto, H.; et al. Large-scale Circulating microRNA Profiling for the Liquid Biopsy of Prostate Cancer. Clin. Cancer Res. 2019, 25, 3016–3025. [Google Scholar] [CrossRef] [PubMed]
- Bidarra, D.; Constâncio, V.; Barros-Silva, D.; Ramalho-Carvalho, J.; Moreira-Barbosa, C.; Antunes, L.; Mauricio, J.; Oliveira, J.; Henrique, R.; Jeronimo, C. Circulating MicroRNAs as Biomarkers for Prostate Cancer Detection and Metastasis Development Prediction. Front. Oncol. 2019, 9, 900. [Google Scholar] [CrossRef]
- Cancer-Derived Exosomal miR-199b-5p Inhibits Distant Metastases of Prostate Cancer by Counteracting the DDR1-MAPK/ERK-EMT Pathway. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3475571 (accessed on 6 December 2019).
- Porzycki, P.; Ciszkowicz, E.; Semik, M.; Tyrka, M. Combination of three miRNA (miR-141, miR-21, and miR-375) as potential diagnostic tool for prostate cancer recognition. Int. Urol. Nephrol. 2018, 50, 1619–1626. [Google Scholar] [CrossRef]
- Kotb, S.; Mosharafa, A.; Essawi, M.; Hassan, H.; Meshref, A.; Morsy, A. Circulating miRNAs 21 and 221 as biomarkers for early diagnosis of prostate cancer. Tumor Biol. 2014, 35, 12613–12617. [Google Scholar] [CrossRef]
- Barceló, M.; Castells, M.; Bassas, L.; Vigués, F.; Larriba, S. Semen miRNAs Contained in Exosomes as Non-Invasive Biomarkers for Prostate Cancer Diagnosis. Sci. Rep. 2019, 9, 13772. [Google Scholar] [CrossRef]
- Cheng, W.S.; Tao, H.; Hu, E.P.; Liu, S.; Cai, H.R.; Tao, X.L.; Zhang, L.; Mao, J.J.; Yan, D.L. Both genes and lncRNAs can be used as biomarkers of prostate cancer by using high throughput sequencing data. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3504–3510. [Google Scholar]
- Qu, Y.; Zhang, L.; Mao, M.; Zhao, F.; Huang, X.; Yang, C.; Xiong, Y.; Mu, D. Effects of DNA zymes targeting Aurora kinase A on the growth of human prostate cancer. Cancer Gene Ther. 2008, 15, 517–525. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Noonan, E.J.; Place, R.F.; Pookot, D.; Basak, S.; Whitson, J.M.; Hirata, H.; Giardina, C.; Dahiya, R. miR-449a targets HDAC-1 and induces growth arrest in prostate cancer. Oncogene 2009, 28, 1714–1724. [Google Scholar] [CrossRef] [PubMed]
- Bertoli, G.; Cava, C.; Castiglioni, I. MicroRNAs as Biomarkers for Diagnosis, Prognosis and Theranostics in Prostate Cancer. Int. J. Mol. Sci. 2016, 17, 421. [Google Scholar] [CrossRef] [PubMed]
- Chhipa, R.R.; Wu, Y.; Ip, C. AMPK-mediated autophagy is a survival mechanism in androgen-dependent prostate cancer cells subjected to androgen deprivation and hypoxia. Cell. Signal. 2011, 23, 1466–1472. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Liu, M.; Guan, Y.; Wu, Q. Hypoxia-Responsive Mir-301a and Mir-301b Promote Radioresistance of Prostate Cancer Cells via Downregulating NDRG2. Med. Sci. Monit. 2016, 22, 2126–2132. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Bai, L. Hypoxia induced microRNA-301b-3p overexpression promotes proliferation, migration and invasion of prostate cancer cells by targeting LRP1B. Exp. Mol. Pathol. 2019, 111, 104301. [Google Scholar] [CrossRef]
- Wang, Z.; Shao, M.; Liu, Y. Promotion of Wilms’ tumor cells migration and invasion by mono-2-ethyhexyl phthalate (MEHP) via activation of NF-κB signals. Chem. Biol. Interact. 2017, 270, 1–8. [Google Scholar] [CrossRef]
- Ni, S.; Hu, J.; Duan, Y.; Shi, S.; Li, R.; Wu, H.; Qu, Y.; Li, Y. Down expression of LRP1B promotes cell migration via RhoA/Cdc42 pathway and actin cytoskeleton remodeling in renal cell cancer. Cancer Sci. 2013, 104, 817–825. [Google Scholar] [CrossRef]
- Wei, P.; Qiao, B.; Li, Q.; Han, X.; Zhang, H.; Huo, Q.; Sun, J. microRNA-340 Suppresses Tumorigenic Potential of Prostate Cancer Cells by Targeting High-Mobility Group Nucleosome-Binding Domain 5. DNA Cell Biol. 2016, 35, 33–43. [Google Scholar] [CrossRef]
- Leite, K.R.M.; Franco, M.F.; Srougi, M.; Nesrallah, L.J.; Nesrallah, A.; Bevilacqua, R.G.; Biomed, E.D.; Carvalho, H.M.; Meirelles, M.I.; Santana, I.; et al. Abnormal Expression of MDM2 in Prostate Carcinoma. Mod. Pathol. 2001, 14, 428–436. [Google Scholar] [CrossRef]
- Chen, S.Y.; Du, Y.; Song, J. MicroRNA-340 Inhibits Epithelial-Mesenchymal Transition by Impairing ROCK-1-Dependent Wnt/β-Catenin Signaling Pathway in Epithelial Cells from Human Benign Prostatic Hyperplasia. Chin. Med. J. 2018, 131, 2008–2012. [Google Scholar] [CrossRef]
- Das, S.; Roth, C.P.; Wasson, L.M.; Vishwanatha, J.K. Signal transducer and activator of transcription-6 (STAT6) is a constitutively expressed survival factor in human prostate cancer. Prostate 2007, 67, 1550–1564. [Google Scholar] [CrossRef]
- Liu, D.; Tao, T.; Xu, B.; Chen, S.; Liu, C.; Zhang, L.; Han, C.; Chen, M. MiR-361-5p acts as a tumor suppressor in prostate cancer by targeting signal transducer and activator of transcription-6(STAT6). Biochem. Biophys. Res. Commun. 2014, 445, 151–156. [Google Scholar] [CrossRef]
- Fletcher, C.E.; Sulpice, E.; Combe, S.; Shibakawa, A.; Leach, D.A.; Hamilton, M.P.; Chrysostomou, S.L.; Sharp, A.; Welti, J.; Yuan, W.; et al. Androgen receptor-modulatory microRNAs provide insight into therapy resistance and therapeutic targets in advanced prostate cancer. Oncogene 2019, 38, 5700–5724. [Google Scholar] [CrossRef]
- Hart, M.; Nolte, E.; Wach, S.; Szczyrba, J.; Taubert, H.; Rau, T.T.; Hartmann, A.; Grasser, F.A.; Wulich, B. Comparative microRNA Profiling of Prostate Carcinomas with Increasing Tumor Stage by Deep Sequencing. Mol. Cancer Res. 2014, 12, 250–263. [Google Scholar] [CrossRef]
- Lo, U.G.; Pong, R.C.; Yang, D.; Gandee, L.; Hernandez, E.; Dang, A.; Lin, C.J.; Santoyo, J.; Ma, S.; Sonavane, R.; et al. IFN-r-induced IFIT5 promotes epithelial-to-mesenchymal transition in prostate cancer via microRNA processing. Cancer Res. 2018, 79, 1098–1112. [Google Scholar] [CrossRef]
- Walter, B.A.; Valera, V.A.; Pinto, P.A.; Merino, M.J. Comprehensive microRNA Profiling of Prostate Cancer. J. Cancer 2013, 4, 350–357. [Google Scholar] [CrossRef]
- Moya, L.; Meijer, J.; Schubert, S.; Matin, F.; Batra, J. Assessment of miR-98-5p, miR-152-3p, miR-326 and miR-4289 Expression as Biomarker for Prostate Cancer Diagnosis. Int. J. Mol. Sci. 2019, 20, 1154. [Google Scholar] [CrossRef]
- Qu, Y.; Huang, W. Effects of microRNA-210 on the diagnosis and treatment of prostate cancer. Mol. Med. Rep. 2018, 18, 1740–1744. [Google Scholar] [CrossRef]
- Ishioka, J.; Matsuoka, Y.; Uehara, S.; Yasuda, Y.; Kijima, T.; Yoshida, S.; Yokoyama, M.; Saito, K.; Kihara, K.; Numao, N.; et al. Computer-aided diagnosis of prostate cancer on magnetic resonance imaging using a convolutional neural network algorithm. BJU Int. 2018, 122, 411–417. [Google Scholar] [CrossRef]
- von Klot, C.A.J.; Kuczyk, M.A. Künstliche Intelligenz und neuronale Netze in der Urologie. Der Urol. 2019, 58, 291–299. [Google Scholar] [CrossRef]
- Hamid, S.; Donaldson, I.A.; Hu, Y.; Rodell, R.; Villarini, B.; Bonmati, E.; Tranter, P.; Punwani, S.; Sidhu, H.S.; Willis, S.; et al. The SmartTarget Biopsy Trial: A Prospective, Within-person Randomised, Blinded Trial Comparing the Accuracy of Visual-registration and Magnetic Resonance Imaging/Ultrasound Image-fusion Targeted Biopsies for Prostate Cancer Risk Stratification. Eur. Urol. 2019, 75, 733–740. [Google Scholar] [CrossRef]
- Zainfeld, D.; Goldkorn, A. Liquid Biopsy in Prostate Cancer: Circulating Tumor Cells and Beyond. Cancer Treat. Res. 2018, 175, 87–104. [Google Scholar] [CrossRef]
- Campos-Fernández, E.; Barcelos, L.S.; de Souza, A.G.; Goulart, L.R.; Alonso-Goulart, V. Research landscape of liquid biopsies in prostate cancer. Am. J. Cancer Res. 2019, 9, 1309–1328. [Google Scholar]
- Xu, L.; Mao, X.; Grey, A.; Scandura, G.; Guo, T.; Burke, E.; Marzec, J.; Abdu, S.; Stankiewicz, E.; Davies, C.R.; et al. Noninvasive Detection of Clinically Significant Prostate Cancer Using Circulating Tumor Cells. J. Urol. 2020, 203, 73–82. [Google Scholar] [CrossRef]
- Broncy, L.; Paterlini-Brécho, P. Clinical Impact of Circulating Tumor Cells in Patients with Localized Prostate Cancer. Cells 2019, 8, 676. [Google Scholar] [CrossRef]
- Palmirotta, R.; Lovero, D.; Cafforio, P.; Felici, C.; Mannavola, F.; Pellè, E.; Quaresmini, D.; Tucci, M.; Silvestris, F. Liquid biopsy of cancer: A multimodal diagnostic tool in clinical oncology. Ther. Adv. Med. Oncol. 2018, 10, 175883591879463. [Google Scholar] [CrossRef]
- Huang, Q.; Wang, Y.; Chen, X.; Wang, Y.; Li, Z.; Du, S.; Wang, L.; Chen, S. Nanotechnology-Based Strategies for Early Cancer Diagnosis Using Circulating Tumor Cells as a Liquid Biopsy. Nanotheranostics 2018, 2, 21–41. [Google Scholar] [CrossRef]
- Zhang, Y.; Mi, X.; Tan, X.; Xiang, R. Recent Progress on Liquid Biopsy Analysis using Surface-Enhanced Raman Spectroscopy. Theranostics 2019, 9, 491–525. [Google Scholar] [CrossRef]
- Wang, J.; Koo, K.M.; Wang, Y.; Trau, M. Engineering State-of-the-Art Plasmonic Nanomaterials for SERS-Based Clinical Liquid Biopsy Applications. Adv. Sci. 2019, 6, 1900730. [Google Scholar] [CrossRef]
- Stefancu, A.; Moisoiu, V.; Couti, R.; Andras, I.; Rahota, R.; Crisan, D.; Pavel, I.E.; Socaciu, C.; Leopold, N.; Crisan, N. Combining SERS analysis of serum with PSA levels for improving the detection of prostate cancer. Nanomedicine 2018, 13, 2455–2467. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; O’Connor, R.; Kwizera, E.A. Gold Nanoparticle Based Platforms for Circulating Cancer Marker Detection. Nanotheranostics 2017, 1, 80–102. [Google Scholar] [CrossRef] [PubMed]
- Niciński, K.; Krajczewski, J.; Kudelski, A.; Witkowska, E.; Trzcińska-Danielewicz, J.; Girstun, A.; Kaminska, A. Detection of circulating tumor cells in blood by shell-isolated nanoparticle—Enhanced Raman spectroscopy (SHINERS) in microfluidic device. Sci. Rep. 2019, 9, 9267. [Google Scholar] [CrossRef] [PubMed]
- Onaciu, A.; Munteanu, R.A.; Moldovan, A.I.; Moldovan, C.S.; Berindan-Neagoe, I. Hydrogels Based Drug Delivery Synthesis, Characterization and Administration. Pharmaceutics 2019, 11, 432. [Google Scholar] [CrossRef] [PubMed]

| Product | Methodology | Clinical Evidence | References |
|---|---|---|---|
| Oncotype DX AR-V7 Nucleus Detect | Blood samples are analyzed for the detection of AR-V7 protein in the nucleus that determines the direction of treatment scheme in metastatic castration-resistant cancer patients. | Howard I. Scher et al conducted two studies, one in 2016 and the other one in 2017; with 161 enrolled patients diagnosed with metastatic castration-resistant prostate cancer. | [31,32] |
| Foundation One Liquid | Next generation sequencing detects clinically relevant genomic alterations like substitutions, insertion/deletions, copy number alterations and selected genetic rearrangements, in 70 oncogenes. | Clark et al. included 860 patients in a study, from which 63 had prostate cancer in 2018. | [33] |
| Sangia Total PSA Test | A blood sample from fingerstick is collected by the device and analyzed using Claros 1 Analyzer. The test is able to measure total PSA level in the blood in less than 15 minutes. | The cohort was composed of 434 enrolled patients of 50 years old or older from 10 urology clinics from U.S. Sangia Test proved a sensitivity of 84.5% with a 95% confidence interval. The sensitivity increases with almost 59% in conjunction with DRE. | [34] |
| Progensa PCA 3 Assay | The samples are represented by urine. The test is composed of two quantitative nucleic acid amplification tests that are performed in vitro: One for prostate cancer gene 3 (PCA 3) RNA and the other one for PSA RNA in order to detect the specific amplicons. The assay combines different technologies like transcription mediated amplification and hybridization protection assay in order to quantify the results. | The cohort comprised 495 male subjects enrolled from 14 clinical sites, having a median age of 67 years. From each patient were collected blood, urine and prostatic biopsies. Another study refers to a cohort of 85 patients with prostate cancer or benign hyperplasia. They concluded that DD3 mRNA is another representative biomarker for prostate cancer and its levels can be associated with PSA levels. | [35,36,37,38] |
| 4 K Score Test | Blood samples are analyzed in order to measure four prostate cancer specific kallikreins in the blood: Total PSA, free PSA, intact PSA and human kallikrein 2. The test results undergo some algorithm processing and indicate the aggressivity of prostate cancer prior to prostate biopsy. | Lilja H.’s team included 40,379 men at ages 40, 50 and 60 years, from which they identified 1423 incident prostate cancer patients and 235 with distant metastasis. Zappala S.’s teams conducted two studies. The first study included 1012 men during 2013 and 2014. The second study included 100,000 patients suspected of having prostate cancer. The studies conclude that 4Kscore Test can provide guidance during prostate cancer detection and its results can influence the biopsy decision, therefore reducing the healthcare costs. | [39,40] |
| Sample | Cohort | MiRNAs Analyzed Upregulated/Downregulated (↑/↓) | Clinical Value | Ref. |
|---|---|---|---|---|
| Serum | Seening: 25 mCRPCa, 25 healthy men aged matched Validation set: 21 mCRPCa, 20 healthy men, aged matched | From a total of 365 miRNAs, 5 miRs were studied: miRs-141, -200a, -200c, -210, -375 ↑ | Increased levels of circulating miR-, miR-200a and miR-200c are associated with epithelial origin of prostate cancer. miR-210 represents a predictive biomarker. AUC: miR-141: 0.842 miR-200a: 0.638 miR-200c: 0.645 miR-210: 0.652 | [20] |
| Plasma derived circulating microvesicles, serum derived exosomes and microvesicles, urine | Validation set: 21 mCRPCa, 20 healthy men, aged matched | From a total of 742 miRNAs, 12 miRs were selected: miRs-107, -141, -130b, -301a, -2110, -326, -331-3p, -432, -484, -574-3p, -625 ↑ miRs-181a-2*, -572 ↓ | In metastatic PCa compared with non-recurrent PCa, miR-375 and miR-141 are significantly increased in plasma exosomes and circulating microvesicles. | [26] |
| Serum samples: 47 PCa recurrent patients, 72 non-recurrent patients | ||||
| Urine samples: 17 control patients, 70 local PCa patients, 48 advanced cancer patients | ||||
| Serum | 25 metastatic PCA patients, 25 matched healthy men | miRs-100, -125b, -141, -143, -205 and -296 were investigated and miR-141 ↑ level was of major importance. | miR-141 AUC 0.907 | [41] |
| Serum | Screening: 7 metastatic PCa, 15 localized PCa Validation set 1: 45PCa Validation set 2: 71 PCa (48N1, 23N0, 29 Gleason >8, 42 Gleason 7), 12 low risk, 12 high risk, 12 intermediate, 12 healthy controls | From 667 miRs, 5 miRs were investigated: miRs-375, -9*, -141, -200b, -516-3p ↑ | miRs-141 and -375 are associated with high Gleason score and positive lymph node status. | [56] |
| Plasma | Validation set 1: 45PCa | From a total of 1146 miRNAs, 5 miRs were selected: miRs-622, -1285 ↑ miRs-let-7e, -let-7c, -30c ↓ | miRs- 622, -1285, -let-7e, -let-7c, -30c, combined, differentiate PCa from BPH and healthy men, Area under curve (AUC) 0.924 and 0.860. | [57] |
| Validation set 2: 71 PCa (48N1, 23N0, 29 Gleason >8, 42 Gleason 7), 12 low risk, 12 high risk, 12 intermediate, 12 healthy controls | ||||
| Serum | 28 low risk localized disease, 30 high risk localized disease, 26 metastatic castration resistant PCa (mCRPCa) patients | From a total of 669 miRNAs, 4 miRs were selected. Metastatic castration resistant vs. low risk localized disease: miRs-375, -378, -141 ↑ Low risk vs. metastatic PCa: miR-409-3p ↓ | miR-375, miR-141, miR-378 were associated with disease progression. | [58] |
| Plasma | 25 localized PCa, 25 mCRPCa patients | From 742 miRs investigated, 63 miRs were found to be upregulated and 4 miRs downregulated in mCRPCa compared to localized PCa: miRs-141, -375, -200c, -126, -21, -151-3p, -152, -423-3p ↑ miRs-205 and -16 ↓ | miR-141, miR-151-3p, miR-16 can differentiate localized and mCRPCa, AUC: 0.944, sensitivity of 84%, specificity of 96% | [59] |
| Serum | 13 BPH, 11 localized PCa, 9 with lymph node or distant metastase (N+/M+), 11 CRPCa patients | From 732 miRNAs studied, 20 miRNAs were selected: miRs-107, -141, -21, -200b, -221, -30c, -346, -375, -574-3p ↑ miRs-1179, -149*, -154, -181a*, -188-5p, -31, -329, -376c, -450a, -508-5p, and -556-5p ↓ | mMiRs-let-7a*, -210, and -562 represent promising biomarkers associated with aggressive PCa. | [60] |
| Plasma exosomes | Screening: 23 CRPCa, androgen deprivation therapy (ADT) failure | miRs-30a/e-5p, -99a-5p, -let-7c, -1246, 1290, -16-5p, -125a-5p, -375 were studied and 2 miRs were representative for the study: miRs-375 and -1290 ↑ | High levels of miR-375 and miR-1290 are associated with poor overall survival. | [61] |
| Follow up: 100PCa, ADT failure | ||||
| Plasma | 51PCa (25 metastatic PCa), 20 healthy men | miRs-21, -141 and -221 were analysed and expression level of miRs-221 and -21 was found up-regulated↑. | AUC: miR-21-0.88 miR-221 0.83 did not reach PSA power for discriminating localized from metastatic PCa. | [62] |
| Serum | 18 BPH, 20 healthy men, 37 localised PCa, 8 metastatic | miRs-26a, -32, -195 and -let-7i were studied. PCa vs BPH: miRs-26a, -195 and -let-7i ↑ | miR-26a, AUC 0.703, can differentiate PCa from BPH with a sensitivity of 89%, and a specificity of 56%. Combined miRs-26a, -32,-195, -let-7i, PCA vs BPH, AUC 0.758, sensitivity 78%, specificity 67%. | [63] |
| Plasma | 21 PCa patients | miR-141 ↑ | Changes of miR-141 are associated with clinical course. | [64] |
| Serum | 25 Healthy men, 25 mCRPCa | miRs-141, -298, -375 ↑ miR-346↓ | Expression level of miRs-141, -298 and -375 were increased in PCa. | [65] |
| Serum | 100 treated PCa, N0 (50 low risk, 50 high risk), 50 BPH | From 16 miRs, 12 miRs were detected in serum samples at high levels. miRs-96, -141, 182, 183 were not detectable in >50% of the patients. miRs-let-7a, -24, -26, -30c, -93, -103, -106a, -107, -130b, -146a, -223, -451↑ | The investigated miRNAs from serum samples are associated with low grade PCa. | [66] |
| Whole blood | 102 patients: 27 negative biopsy, 75 PCa confirmed | From 12 miRs identified, were selected: miRs-141, -145, -155 ↑ miR-let-7a ↓ | Combined miRs-let-7a, -141, -145, -155 AUC 0.783 and PPV of 80% | [67] |
| Plasma | 16 BPH, 59 PCa 11 asymptomatic young men | miRs-let-7c, -30c, -141, -375 were analysed and was noticed that miRs-141 and -375 are down-regulated↓. | AUC 0.809 for miR- 375 Combined miRs-let-7c, -30c, -141, -375 and PSA resulted an AUC 0.877 (sensitivity of 86.8% and specificity of 81.8%). | [68] |
| Peripheral blood, mononuclear cells | 75 healthy men 75 PCa patients | miR-21 ↑ | miR-21 AUC 0.9 with a sensitivity of 87.5% and specificity of 85.7%. This miR was associated with clinical stage, tumor differentiation and lymph node metastasis. | [69] |
| Plasma | 36 PCa 31 BPH patients | miRs-1061-5p, -1207-5p, -141-3p, -574-3p, -20a-5p, -21-5p, -93-5p, -2110, -130b-3p, -375 ↑ miRs-223-3p, -24-3p ↓ | miR-106a/130b AUC=0.81, miR 106a/223 AUC=0.84, PSA AUC= 0.56. | [70] |
| Serum | 133 patients: 54 PCa 79 BPH | miRs 26a-1 and -141 were investigated and was found an up-regulated ↑ expression level for miR-141 | Both miRs failed as diagnostic biomarker, miR-141 levels were increased in high Gleason scores. | [71] |
| Serum | 149 PCa, 81 BPH, 57 healthy controls 40 other urinary pathologies | miR-410-5p ↑ | miR-410-5p is a stable biomarker for PCa diagnosis and is associated with low and high-intermediate risk specific Gleason score. PCa vs healthy or other, discrimination, AUC= 0.8097 | [72] |
| Plasma | 65 PCa, 51 BPH, 74 healthy controls | miRs-21 and -152 expression levels were analysed and miR-21 has an up-regulated pattern ↑ | There was no significant difference in expression between PCa and healthy controls. | [73] |
| Serum | 20 metastatic PCa 31 PCa and 40 healthy patients | miR-141 ↑ | Discriminating PCa vs metastatic PCa, AUC 0.8694, PSA AUC 0.7758 | [74] |
| Serum | 13 BPH 28 PCa patients | miR-301a ↑ | miR-301a expression is correlated with increased Gleason score | [75] |
| Plasma | Discovery cohort: 42 PCa patients 19 controls | 372 cancer associated miRNAs were investigated and 11 miRNAs possible candidates were selected. From this selection: miRs-4289, -326, -152-3p, -98-5p ↑ | miRNA panel distinguish between PCa and healthy patients, AUC 0.88 | [76] |
| Validation cohort: 40 PCa patients 18 controls | ||||
| Serum | 809 PCa, 241 negative prostate biopsies, 500 patients with other cancers, 41 healthy controls | From 408 miRNAs were selected 38 miRNAs. From these 38 miRNAs, 18 miRNAs were identified as upregulated, from which is important to mention miRs-17-3p and -1185-2-3p, and 2 miRNAs downregulated↓ | The combination of miR-17-3p and miR-1185-2-3p achieved a high diagnostic potential with a sensitivity and specificity of 90%, AUC 0.95. | [77] |
| Plasma | Cohort 1: 98 PCa patients with radical prostatectomy | miRs-182-5p and - 375-3p ↑ in plasma samples | miR-375-3p expression level is a predictor for metastasis development, AUC 0.62 | [78] |
| Cohort 2: 252 PCa patients before treatment | ||||
| 52 healthy donors | ||||
| Plasma exosomes | 25 localized PCa, 25 with bone metastatic PCa, 10 with pelvic lymph node metastatic PCa patients | An amount of 2588 miRNAs are representative for localized and bone metastatic PCa. 582 miRNAs were differentially expressed in these groups, of which 160 up-regulated and 102 down-regulated. Significant miRNAs: miRs-361-5p, -324-5p, -139-5p, -199b-5p, -199a-3p ↓ miR-632 ↑ | Down-regulation of miR-199b-5p is associated with metastatic PCa. | [79] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Munteanu, V.C.; Munteanu, R.A.; Onaciu, A.; Berindan-Neagoe, I.; Petrut, B.; Coman, I. MiRNA-Based Inspired Approach in Diagnosis of Prostate Cancer. Medicina 2020, 56, 94. https://doi.org/10.3390/medicina56020094
Munteanu VC, Munteanu RA, Onaciu A, Berindan-Neagoe I, Petrut B, Coman I. MiRNA-Based Inspired Approach in Diagnosis of Prostate Cancer. Medicina. 2020; 56(2):94. https://doi.org/10.3390/medicina56020094
Chicago/Turabian StyleMunteanu, Vlad Cristian, Raluca Andrada Munteanu, Anca Onaciu, Ioana Berindan-Neagoe, Bogdan Petrut, and Ioan Coman. 2020. "MiRNA-Based Inspired Approach in Diagnosis of Prostate Cancer" Medicina 56, no. 2: 94. https://doi.org/10.3390/medicina56020094
APA StyleMunteanu, V. C., Munteanu, R. A., Onaciu, A., Berindan-Neagoe, I., Petrut, B., & Coman, I. (2020). MiRNA-Based Inspired Approach in Diagnosis of Prostate Cancer. Medicina, 56(2), 94. https://doi.org/10.3390/medicina56020094







