State of the Art in Exocrine Pancreatic Insufficiency
Abstract
1. Introduction
- (a)
- Pancreatic stimulation: Insufficient activation of pancreatic secretion can be caused by diseases (e.g., celiac disease) that reduce the release of cholecystokinin (CCK) from the duodenal mucosa or by pancreatic/gastrointestinal surgery. EPI can also result from the treatment of neuroendocrine tumors with somatostatin analogs, given that somatostatin is a physiological pancreatic secretion inhibitor [3,9].
- (b)
- Pancreatic juice synthesis: Damage to the pancreatic parenchyma reduces the production and secretion of pancreatic enzymes by acinar cells and of bicarbonate by pancreatic ducts. This functional loss can be caused by various diseases, including chronic pancreatitis, cystic fibrosis, pancreatic cancer, or acute necrotizing pancreatitis, or by pancreatic resection for the treatment of some of these.
- (c)
- Pancreatic juice transport: Obstruction of the passage of pancreatic juice through the pancreatic duct prevents its arrival into the intestinal lumen to carry out its digestive activity. This problem can be caused by disorders such as cystic fibrosis, with the production of a thicker secretion, or by various types of pancreatic tumor.
- (d)
- Synchronization of gastrointestinal secretions: Asynchrony in the interaction of nutrients with biliopancreatic secretions results in the incorrect digestion of foods. This problem is usually caused by anatomical changes produced by pancreatobiliary or gastrointestinal surgery. This phenomenon is also observed in patients with Crohn’s disease or short bowel syndrome. Less frequently, there is no enzyme activation despite adequate secretion and the arrival of pancreatic juice to the duodenum, as in cases of hyperchlorhydria.
2. Prevalence and Clinical Relevance
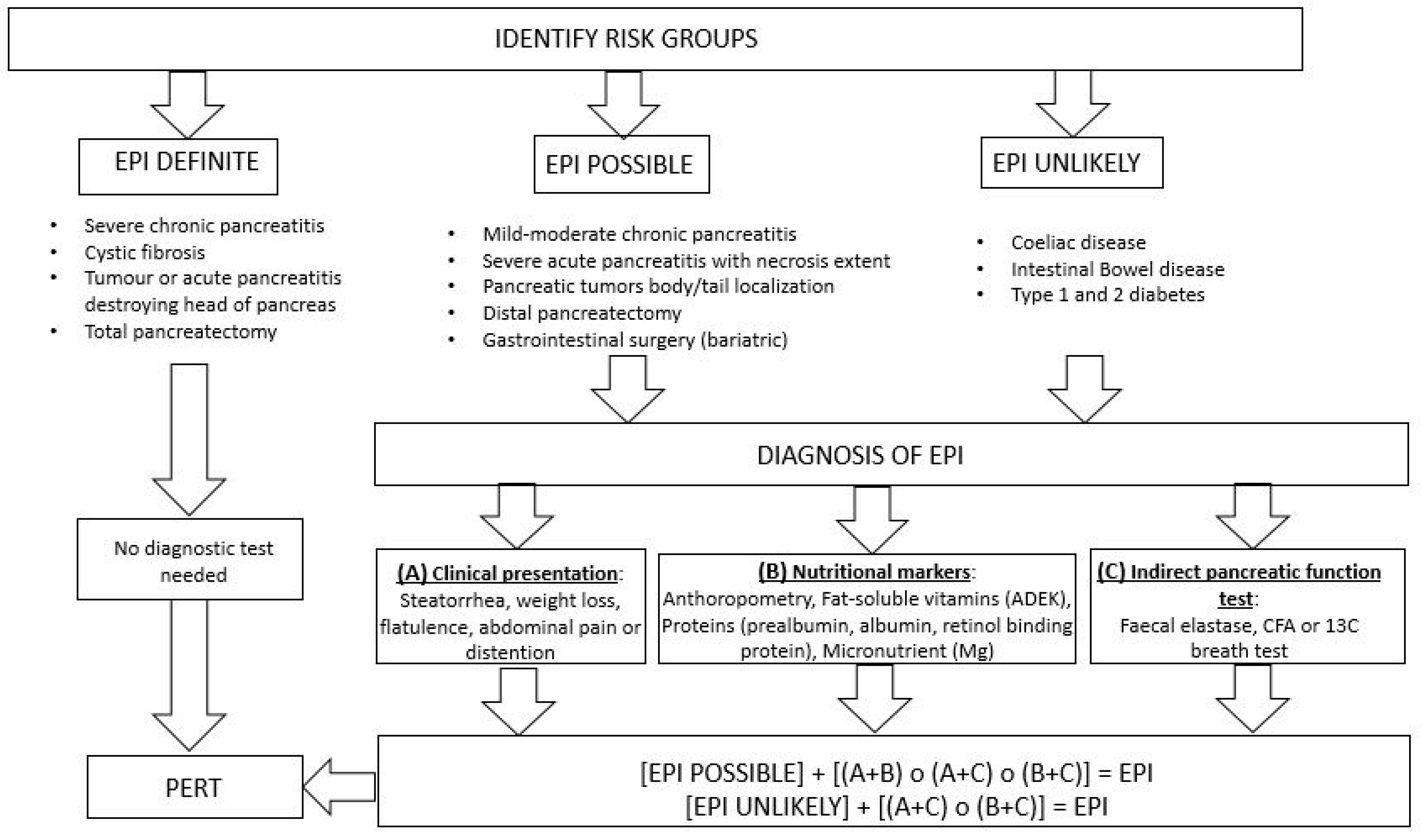
3. Diagnosis
- (a)
- There is no consensus on the cutoff point for an EPI diagnosis, which has generally been arbitrarily considered as <200 μg/g. Nevertheless, the smaller the concentration, the greater the likelihood of EPI, and very high sensitivity and specificity values have been reported for concentrations <15 μg/g. Conversely, EPI is ruled out by very high elastase values (>500 μg/g) [7].
- (b)
- The interpretation of borderline elastase values (100–200) should be guided by the observation of symptoms and malnutrition indicators [1]. A closer follow up should be considered, especially if there is persistence of the etiological factors most frequently associated with pancreatic damage (tobacco and alcohol).
- (c)
- Samples with a liquid consistency (Bristol scale of 5–7) should be excluded.
- (d)
- Potential causes of false positives should be ruled out, including bacterial overgrowth [8]. They may also be attributable to advanced age or conditions such as chronic kidney disease, although these associations have not been fully elucidated.
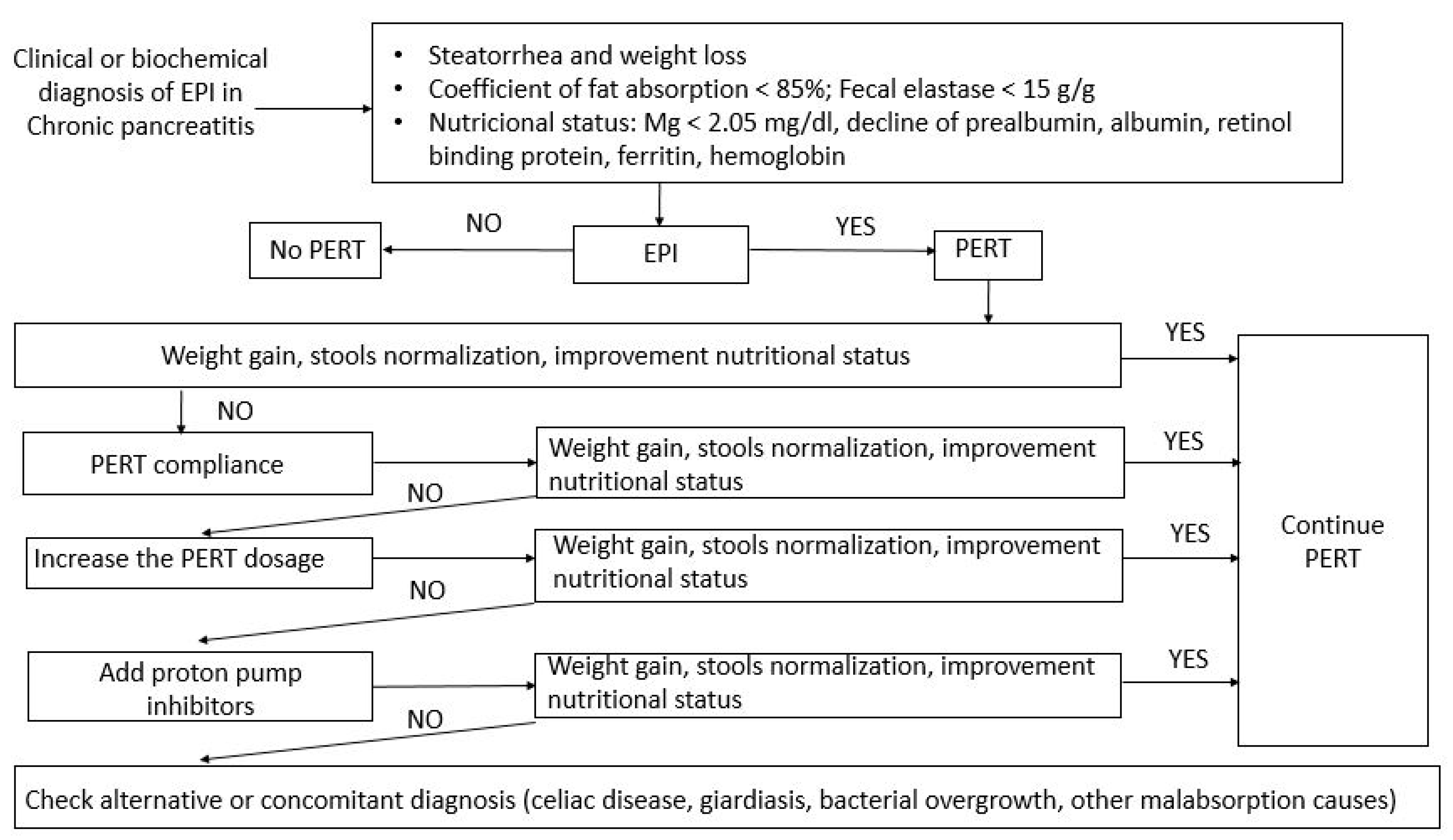
4. Treatment
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dominguez-Muñoz, J.E. Diagnosis and treatment of pancreatic exocrine insufficiency. Curr. Opin. Gastroenterol. 2018, 34, 349–354. [Google Scholar] [CrossRef]
- Perbtani, Y.; Forsmark, C.E. Update on the diagnosis and management of exocrine pancreatic insufficiency. F1000Research 2019, 8, 1991. [Google Scholar] [CrossRef] [PubMed]
- Capurso, G.; Traini, M.; Piciucchi, M.; Signoretti, M.; Arcidiacono, P.G. Exocrine pancreatic insufficiency: Prevalence, diagnosis, and management. Clin. Exp. Gastroenterol. 2019, 12, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Ghodeif, A.O.; Azer, S.A. Pancreatic insufficiency. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Forsmark, C. Diagnosis and management of exocrine pancreatic insufficiency. Curr. Treat. Options Gastroenterol. 2018, 16, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Nikfarjam, M.; Wilson, J.S.; Smith, R.S. Diagnosis and management of pancreatic exocrine insufficiency. Med. J. Aust. 2017, 207, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Löhr, J.M.; Dominguez-Munoz, E.; Rosendahl, J.; Besselink, M.; Mayerle, J.; Lerch, M.M.; Haas, S.; Akisik, F.; Kartalis, N.; Iglesias-Garcia, J.; et al. United European gastroenterology evidence-based guidelines for the diagnosis and therapy of chronic pancreatitis (HaPanEU). United Eur. Gastroenterol. J. 2017, 5, 153–199. [Google Scholar] [CrossRef]
- Rodrigues-Pinto, E.; Caldeira, A.; Soares, J.B.; Antunes, T.; Carvalho, J.R.; Costa-Maia, J.; Oliveira, P.; Azevedo, R.; Liberal, R.; Bouça Machado, T.; et al. Clube Portugues do pancreas recommendations for chronic pancreatitis: Etiology, natural history and diagnosis. Ge Port. J. Gastroenterol. 2019, 26, 346–355. [Google Scholar] [CrossRef]
- Levy, M.N.; Stanton, B.A.; Koeppen, B.M. Gastrointestinal secretions. In Berne & Levy Principles of Physiology, 4th ed.; Levy, M.N., Stanton, B.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 1, pp. 451–472. [Google Scholar]
- Duggan, S.N. Negotiating the complexities of exocrine and endocrine dysfunction in chronic pancreatitis. Proc. Nutr. Soc. 2017, 76, 484–494. [Google Scholar] [CrossRef]
- Layer, P.; Yamamoto, H.; Kalthoff, L.; Clain, J.E.; Bakken, L.J.; DiMagno, E.P. The different courses of early- and late onset idiopathic and alcoholic chronic pancreatitis. Gastroenterology 1994, 107, 1481–1487. [Google Scholar] [CrossRef]
- Huang, W.; de la Iglesia-García, D.; Baston-Rey, I.; Calviño-Suarez, C.; Lariño-Noia, J.; Iglesias-Garcia, J.; Shi, N.; Zhang, X.; Cai, W.; Deng, L.; et al. Exocrine pancreatic insufficiency following acute pancreatitis: Systematic review and meta-analysis. Dig. Dis. Sci. 2019, 64, 1985–2005. [Google Scholar] [CrossRef]
- Hollemans, R.A.; Hallensleben, N.D.L.; Mager, D.J.; Kelder, J.C.; Besselink, M.G.; Bruno, M.J.; Verdonk, R.C.; van Santvoort, H.C. Pancreatic exocrine insufficiency following acute pancreatitis: Systematic review and study level meta-analysis. Pancreatology 2018, 18, 253–262. [Google Scholar] [CrossRef]
- Pezzilli, R.; Caccialanza, R.; Capurso, G.; Brunetti, O.; Milella, M.; Falconi, M. Pancreatic enzyme replacement therapy in pancreatic cancer. Cancers 2020, 12, 275. [Google Scholar] [CrossRef]
- Altay, M. Which factors determine exocrine pancreatic dysfunction in diabetes mellitus? World J. Gastroenterol. 2019, 25, 2699–2705. [Google Scholar] [CrossRef]
- Radlinger, B.; Ramoser, G.; Kaser, S. Exocrine Pancreatic Insufficiency in type 1 and type 2 diabetes. Curr. Diabetes Rep. 2020, 20, 18. [Google Scholar] [CrossRef]
- Fousekis, F.S.; Theopistos, V.I.; Katsanos, K.H.; Christodoulou, D.K. Pancreatic involvement inflammatory bowel disease: A review. J. Clin. Med. Res. 2018, 10, 743–751. [Google Scholar] [CrossRef]
- Iida, T.; Wagatsuma, K.; Hirayama, D.; Yokoyama, Y.; Nakase, H. The etiology of pancreatic manifestations in patients with inflammatory Bowel disease. J. Clin. Med. 2019, 8, 916. [Google Scholar] [CrossRef]
- Chaudhary, A.; Dominguez-Muñoz, J.E.; Layer, P.; Lerch, M.M. Pancreatic exocrine insufficiency as a complication of gastrointestinal surgery and the impact of pancreatic enzyme replacement therapy. Dig. Dis. 2020, 38, 53–68. [Google Scholar] [CrossRef]
- Pathanki, A.M.; Attard, J.A.; Bradley, E.; Powell-Brett, S.; Dasari, B.V.M.; Isaac, J.R.; Roberts, K.J.; Chatzizacharias, N.A. Pancreatic exocrine insufficiency after pancreáticoduodenectomy: Current evidence and management. World J. Gastrointest. Pathophysiol. 2020, 11, 20–31. [Google Scholar] [CrossRef]
- Vujasinovic, M.; Valente, R.; Thorell, A.; Rutkowski, W.; Haas, S.L.; Arnelo, U.; Martin, L.; Löhr, J.M. Pancreatic exocrine insufficiency after bariatric surgery. Nutrients 2017, 9, 1241. [Google Scholar] [CrossRef]
- Singh, V.K.; Haupt, M.E.; Geller, D.E.; Hall, J.A.; Quintana Diez, P.M. Less common etiologies of exocrine pancreatic insufficiency. World J. Gastroenterol. 2017, 23, 7059–7076. [Google Scholar] [CrossRef]
- Raphael, K.L.; Chawla, S.; Kim, S.; Keith, C.G.; Propp, D.R.; Chen, Z.N.; Woods, K.E.; Keilin, S.A.; Cai, Q.; Willingham, F.F. Pancreatic insufficiency secondary to tobacco exposure: A controlled cross-sectional evaluation. Pancreas 2017, 46, 237–243. [Google Scholar] [CrossRef]
- Pezzilli, R. Chronic pancreatitis: Maldigestion, intestinal ecology and intestinal inflammation. World J. Gastroenterol. 2009, 15, 1673–1676. [Google Scholar] [CrossRef]
- Shintakuya, R.; Uemura, K.; Murakami, Y.; Kondo, N.; Nakagawa, N.; Urabe, K.; Okano, K.; Awai, K.; Higaki, T.; Sueda, T. Sarcopenia is closely associated with pancreatic exocrine insufficiency in patients with pancreatic disease. Pancreatology 2017, 17, 70–75. [Google Scholar] [CrossRef]
- Bachmann, J.; Ketterer, K.; Marsch, C.; Fechtner, K.; Krakowski-Roosen, H.; Büchler, M.W.; Friess, H.; Martignoni, M.E. Pancreatic cancer related cachexia: Influence on metabolism and correlation to weight loss and pulmonary function. BMC Cancer 2009, 9, 255. [Google Scholar] [CrossRef]
- Partelli, S.; Frulloni, L.; Minniti, C.; Bassi, C.; Barugola, G.; D’Onofrio, M.; Crippa, S.; Falconi, M. Faecal elastase-1 is an independent predictor of survival in advanced pancreatic cancer. Dig. Liver Dis. 2012, 44, 945–951. [Google Scholar] [CrossRef]
- De La Iglesia-Garcia, D.; Vallejo-Senra, N.; Lopez-Lopez, A.; Prada-Ramallal, G.; Iglesias-Garcia, J.; Larino-Noia, J.; Nieto-Garcia, L.; Dominguez-Munoz, J.E. Effect of pancreatic exocrine and endocrine insufficiency in cardiovascular events in patients with chronic pancreatitis (CP). A prospective cohort study. Pancreatology 2018, 18, S170–S171. [Google Scholar] [CrossRef]
- Dominguez-Munoz, J.E.; Phillips, M. Nutritional therapy in chronic pancreatitis. Gastroenterol. Clin. North Am. 2018, 47, 95–106. [Google Scholar] [CrossRef]
- Pezzilli, R. Applicability of a checklist for the diagnosis and treatment of severe exocrine pancreatic insufficiency: A survey on the management of pancreatic maldigestion in Italy. Panminerva Med. 2016, 58, 245–252. [Google Scholar]
- Pezzilli, R.; Capurso, G.; Falconi, M.; Frulloni, L.; Macarri, G.; Costamagna, G.; Di Leo, A.; Salacone, P.; Carroccio, A.; Zerbi, A. The applicability of a checklist for the diagnosis and treatment of exocrine pancreatic insufficiency: Results of the Italian exocrine pancreatic insufficiency registry. Pancreas 2020, 49, 793–798. [Google Scholar] [CrossRef]
- Lindkvist, B.; Dominguez-Munoz, J.E.; Luaces-Regueira, M.; Castineiras-Alvarino, M.; Nieto-Garcia, L.; Iglesias-Garcia, J. Serum nutritional markers for prediction of pancreatic exocrine insufficiency in chronic pancreatitis. Pancreatology 2012, 12, 305–310. [Google Scholar] [CrossRef]
- Afghani, E.; Sinha, A.K.; Singh, V. An overview of the diagnosis and management of nutrition in chronic pancreatitis. Nutr. Clin. Pract. 2014, 29, 295–311. [Google Scholar] [CrossRef]
- Pezzilli, R.; Andriulli, A.; Bassi, C.; Balzano, G.; Cantore, M.; Delle Fave, G.; Falconi, M. Exocrine pancreatic insufficiency in adults: A shared position statement of the Italian association for the study of the pancreas. World J. Gatroenterol. 2013, 19, 7930–7946. [Google Scholar] [CrossRef]
- Vanga, R.R.; Tansel, A.; Sidiq, S.; El-Serag, H.B.; Othman, M.O. Diagnostic performance of measurement of fecal elastase-1 in detection of exocrine pancreatic insufficiency: Systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2018, 16, 1220–1228. [Google Scholar] [CrossRef]
- Sikkens, E.C.; Cahen, D.L.; van Eijck, C.; Kuipers, E.J.; Bruno, M.J. The daily practice of pancreatic enzyme replacement therapy after pancreatic surgery: A northern European survey: Enzyme replacement after surgery. J. Gastrointest. Surg. 2012, 16, 1487–1492. [Google Scholar] [CrossRef]
- Sikkens, E.C.; Cahen, D.L.; van Eijck, C.; Kuipers, E.J.; Bruno, M.J. Patients with exocrine insufficiency due to chronic pancreatitis are undertreated: A Dutch national survey. Pancreatology 2012, 12, 71–73. [Google Scholar] [CrossRef]
- Forsmark, C.E.; Tang, G.; Tuft, M.; Xu, H.; Hughes, S.J.; Yadav, D. Oral pancreatic enzyme replacement therapy (PERT) in patients with pancreatic cancer (PCa) is infrequent and suboptimal: A national level analysis. Pancreas 2017, 46, 1400. [Google Scholar]
- Forsmark, C.; Tang, G.; Xu, H.; Yadav, D. Oral pancreatic enzyme replacement therapy (PERT) in chronic pancreatitis (CP) is infrequent and often suboptimal: A national level analysis. Gastroenterology 2017, 152, S677. [Google Scholar] [CrossRef]
- Akshintala, V.S.; Talukdar, R.; Singh, V.K.; Goggins, M. The Gut microbiome in pancreatic disease. Clin. Gastroenterol. Hepatol. 2019, 17, 290–295. [Google Scholar] [CrossRef]
- Nishiyama, H.; Nagai, T.; Kudo, M.; Okazaki, Y.; Azuma, Y.; Watanabe, T.; Goto, S.; Ogata, H.; Sakurai, T. Supplementation of pancreatic digestive enzymes alters the composition of intestinal microbiota in mice. Biochem. Biophys. Res. Commun. 2018, 495, 273–279. [Google Scholar] [CrossRef]
- Hamada, S.; Masamune, A.; Nareshima, T.; Simosegawa, T. Differences in gut microbiota profiles between autoimmune pancreatitis and chronic pancreatitis. Tohoku J. Exp. Med. 2018, 244, 113–117. [Google Scholar] [CrossRef]
- Hu, Y.; Teng, C.; Yu, S.; Wang, X.; Liang, J.; Bai, X.; Dong, L.; Song, T.; Yu, M.; Qu, J. Inonotus obliquus polysaccharide regulates gut microbiota of chronic pancreatitis in mice. Amb Expr. 2017, 7, 39. [Google Scholar] [CrossRef]
- Li, K.; Zhuo, C.; Teng, C.; Yu, S.; Wang, X.; Hu, Y.; Ren, G.; Yu, M.; Qu, J. Effects of Ganoderma lucidum polysaccharides on chronic pancreatitis and intestinal microbiota in mice. Int. J. Biol. Macromol. 2016, 93, 904–912. [Google Scholar] [CrossRef]
- Leal-Lopes, C.; Velloso, F.J.; Campopiano, J.C.; Sogayar, M.C.; Correa, R.G. Roles of comensal microbiota in pancreas homeostasis and pancreatic pathologies. J. Diabetes Res. 2015, 2015, 284680. [Google Scholar] [CrossRef]
- Memba, R.; Duggan, S.N.; Ni Chonchubhair, H.M.; Griffin, O.M.; Bashir, Y.; O’Connor, D.B.; Murphy, A.; McMahon, J.; Volcov, Y.; Ryan, B.M.; et al. The potential role of gut microbiota in pancreatic disease: A systematic review. Pancreatology 2017, 17, 867–874. [Google Scholar] [CrossRef]
- Roberts, K.J.; Bannister, C.A.; Schrem, H. Enzyme replacement improves survival among patients with pancreatic cancer: Results of a population based study. Pancreatology 2019, 19, 114–121. [Google Scholar] [CrossRef]
| Pancreatic Disorders | Extra-Pancreatic Disorders |
|---|---|
| Chronic Pancreatitis | Types 1 and 2 diabetes |
| Cystic Fibrosis | Gastrointestinal Surgery |
| Pancreatic Tumors | Celiac Disease |
| Acute Pancreatitis | Inflammatory Bowel Disease |
| Pancreatic Surgery | HIV |
| Shwachman–Diamond Syndrome | Sjögren’s Syndrome |
| Johanson–Blizzard Syndrome | Intestinal Transplant at Pediatric Age |
| Pancreatic Hemochromatosis [22] | Treatment with Somatostatin Analog |
| Trypsinogen or Enteropeptidase Deficiency | Advanced Age |
| Tobacco Habit [23] |
| Defined Causes | Likely Causes |
|---|---|
| Chronic Pancreatitis | Gastrointestinal Surgery |
| Cystic Fibrosis | Tobacco Habit [23] |
| Pancreatic Surgery | Types 1 and 2 Siabetes |
| Pancreatic Tumor/Cancer | Celiac Disease |
| Benign Main Pancreatic Duct Obstruction | Zollinger-Ellison Syndrome (gastrinoma) |
| Shwachman-Diamond Syndrome | HIV |
| Johanson-Blizzard Syndrome | Advanced Age |
| Hemochromatosis [22] | Severe Malnutrition |
| Acute Pancreatitis without Severe or Recurrent Necrosis |
| Cause of EPI | Prevalence |
|---|---|
| Chronic Pancreatitis | At the Diagnosis: 10% [7] After 10–12 Years with the Disease: 60–90% [4] |
| Acute Pancreatitis [2] | Short-Term: 60% Long-Term: 33% |
| Pancreatic Tumor [2] | Unresectable: 90% Resectable: - Pre-Surgery: 20–44% - Post-Surgery: 60% |
| Pancreatic Surgery [3] | Whipple Procedure: 85–95% Distal Pancreatectomy: 20–50% |
| Cystic Fibrosis [2] | 85% (the Majority at Birth) |
| Gastrointestinal Surgery [3] | Total/Subtotal Gastrectomy: 40–80% Esophagectomy: 16% |
| Type 1 Diabetes | Severe: 10–30%; Mild–Moderate: 22–56% [4] 40% [2] |
| Type 2 Diabetes | 5–46 % [4] 27 % [2] |
| Celiac Disease | 5–80% [3] |
| Inflammatory Bowel Disease | 14–74 % [4] |
| HIV | 26–45% [4] |
| Diagnostic test type | Advantages | Disadvantages |
|---|---|---|
| Direct | ||
| Duodenal Intubation Test | High Sensitivity and Specificity Preferably with Secretin Stimulation and Measurement of Bicarbonate | Invasive Laborious Expensive Only Available in Specialist Centers |
| Endoscopic Test | ||
| Indirect | ||
| 13C-mixed triglyceride Breath Test | Simple Noninvasive Useful to Monitor the Response to PERT | Prolonged Time (6 hours) Not Widely Available False Positives (Malabsorption of Non-Pancreatic Fats and Liver or Lung Disease) Non-specific Low Sensitivity for Mild EPI |
| Stool Elastase | Very Simple Noninvasive Fast Widely Available | Not Useful for PERT Monitoring Low Sensitivity for Mild Cases False Positives (Liquid Stools, Intestinal Inflammation) Not Reliable After Pancreatic Resection |
| Fat absorption Coefficient | Gold Standard | Difficult Application/Adherence Only for Severe EPI Cases Diet of 100g Fats for 5 Days Gathering Stools for 3 Days False Positives (Malabsorption of Non-Pancreatic Fats) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diéguez-Castillo, C.; Jiménez-Luna, C.; Prados, J.; Martín-Ruiz, J.L.; Caba, O. State of the Art in Exocrine Pancreatic Insufficiency. Medicina 2020, 56, 523. https://doi.org/10.3390/medicina56100523
Diéguez-Castillo C, Jiménez-Luna C, Prados J, Martín-Ruiz JL, Caba O. State of the Art in Exocrine Pancreatic Insufficiency. Medicina. 2020; 56(10):523. https://doi.org/10.3390/medicina56100523
Chicago/Turabian StyleDiéguez-Castillo, Carmelo, Cristina Jiménez-Luna, Jose Prados, José Luis Martín-Ruiz, and Octavio Caba. 2020. "State of the Art in Exocrine Pancreatic Insufficiency" Medicina 56, no. 10: 523. https://doi.org/10.3390/medicina56100523
APA StyleDiéguez-Castillo, C., Jiménez-Luna, C., Prados, J., Martín-Ruiz, J. L., & Caba, O. (2020). State of the Art in Exocrine Pancreatic Insufficiency. Medicina, 56(10), 523. https://doi.org/10.3390/medicina56100523





